Treatment in Patients with Psoriatic Disease and Rheumatoid Arthritis: Seven Case Reports
Abstract
1. Introduction
2. Cases
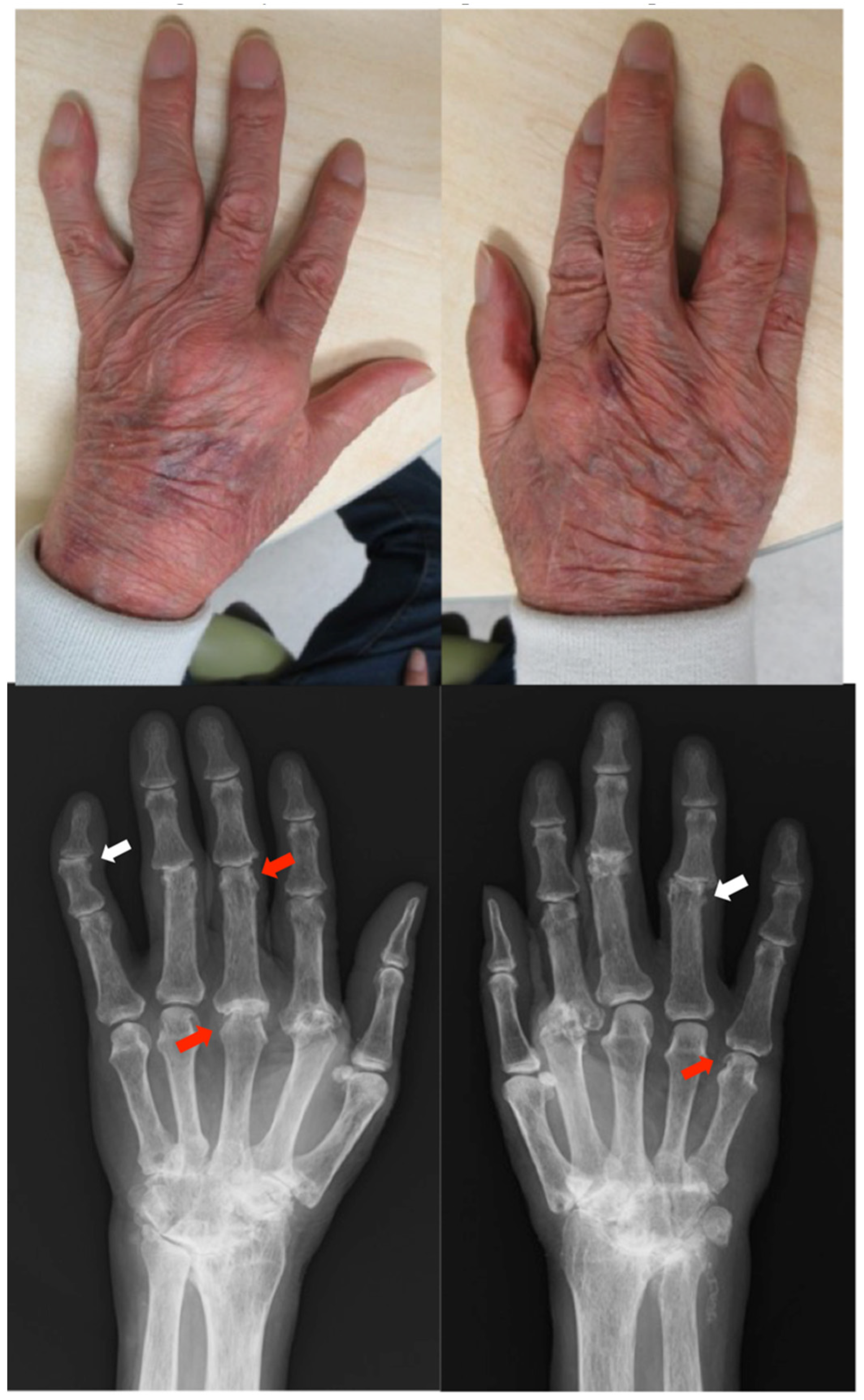
2.1. Case 1
Patient: Man in his 70s
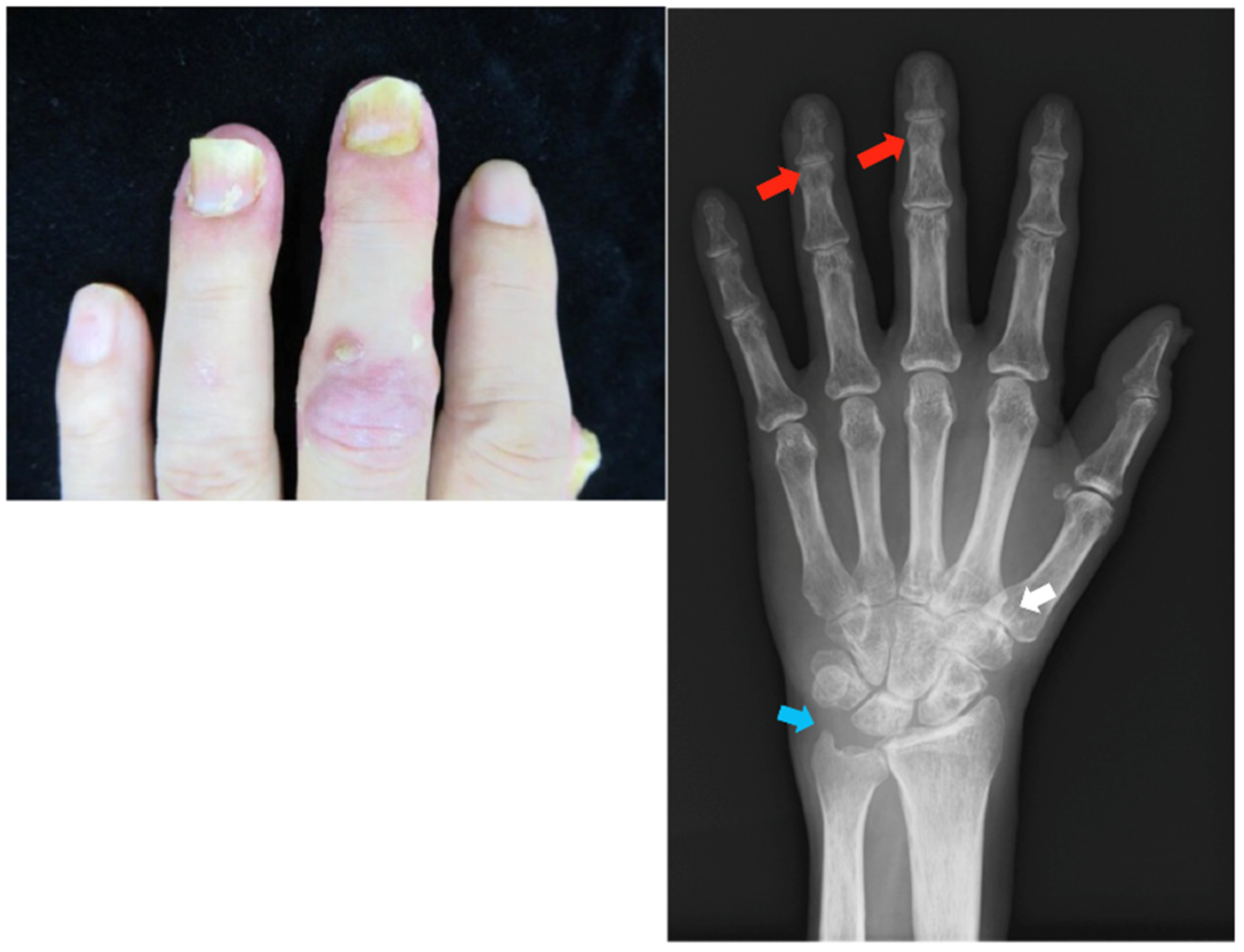
2.2. Case 2
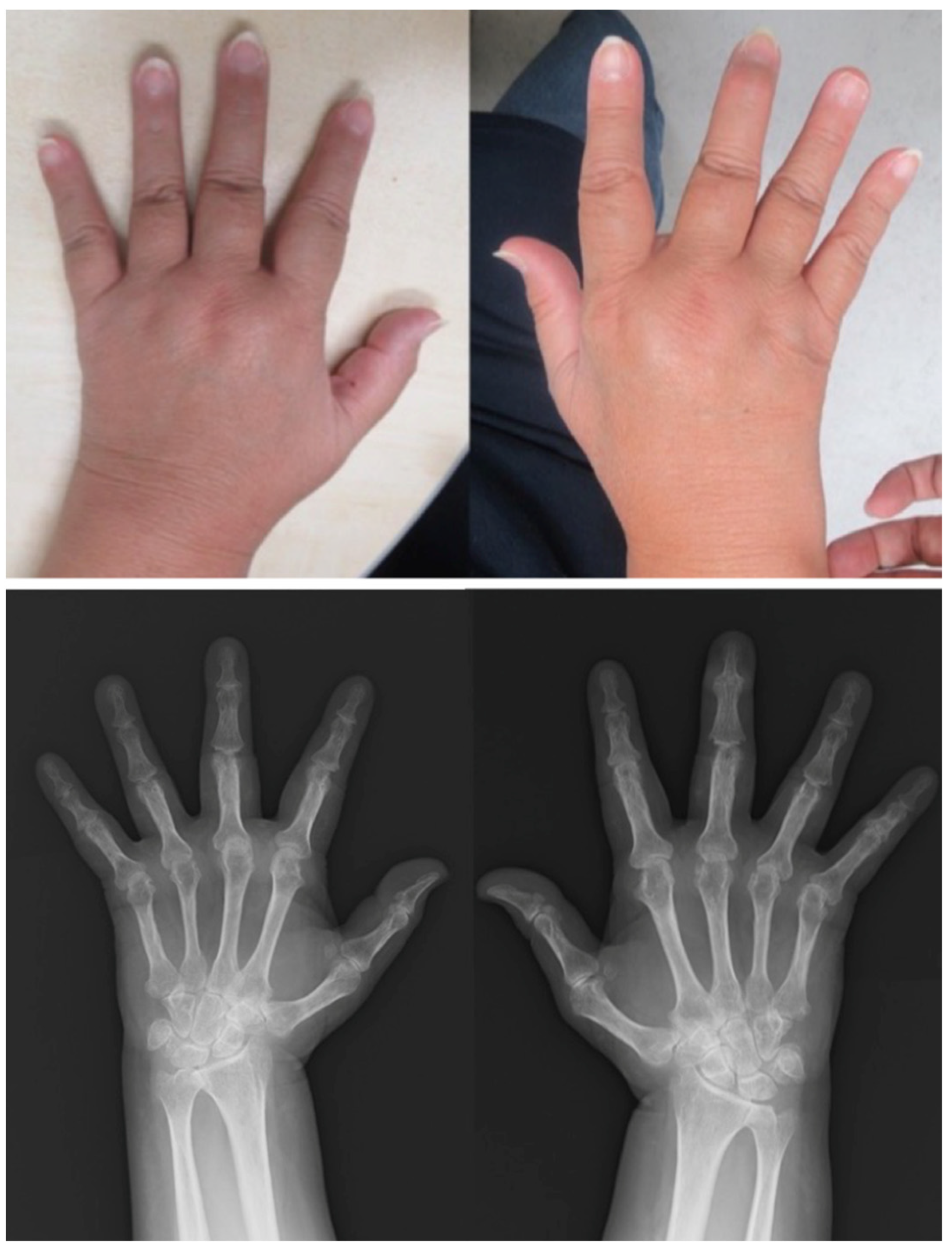
Patient: Man in his 60s
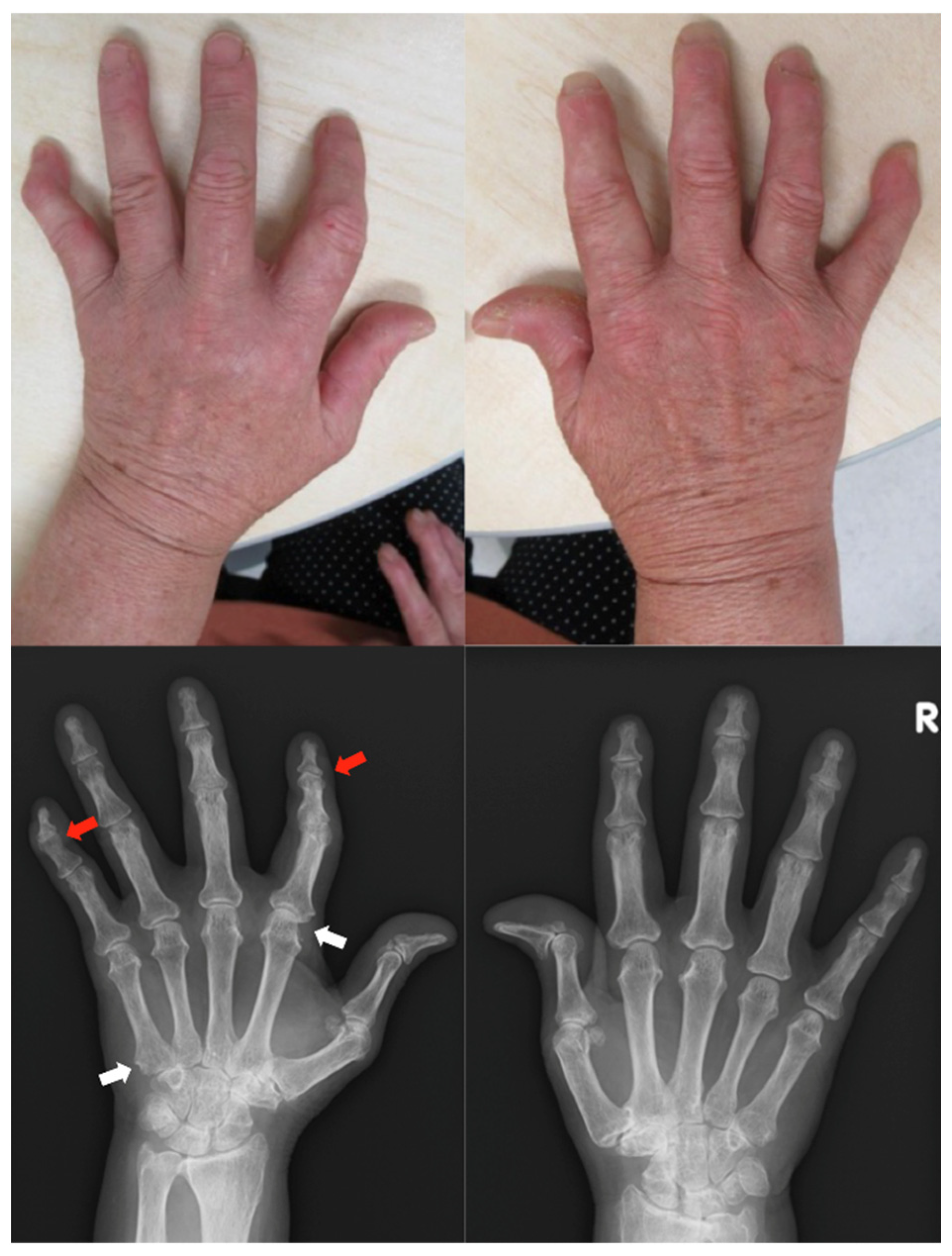
2.3. Case 3
Patient: Woman in her 60s
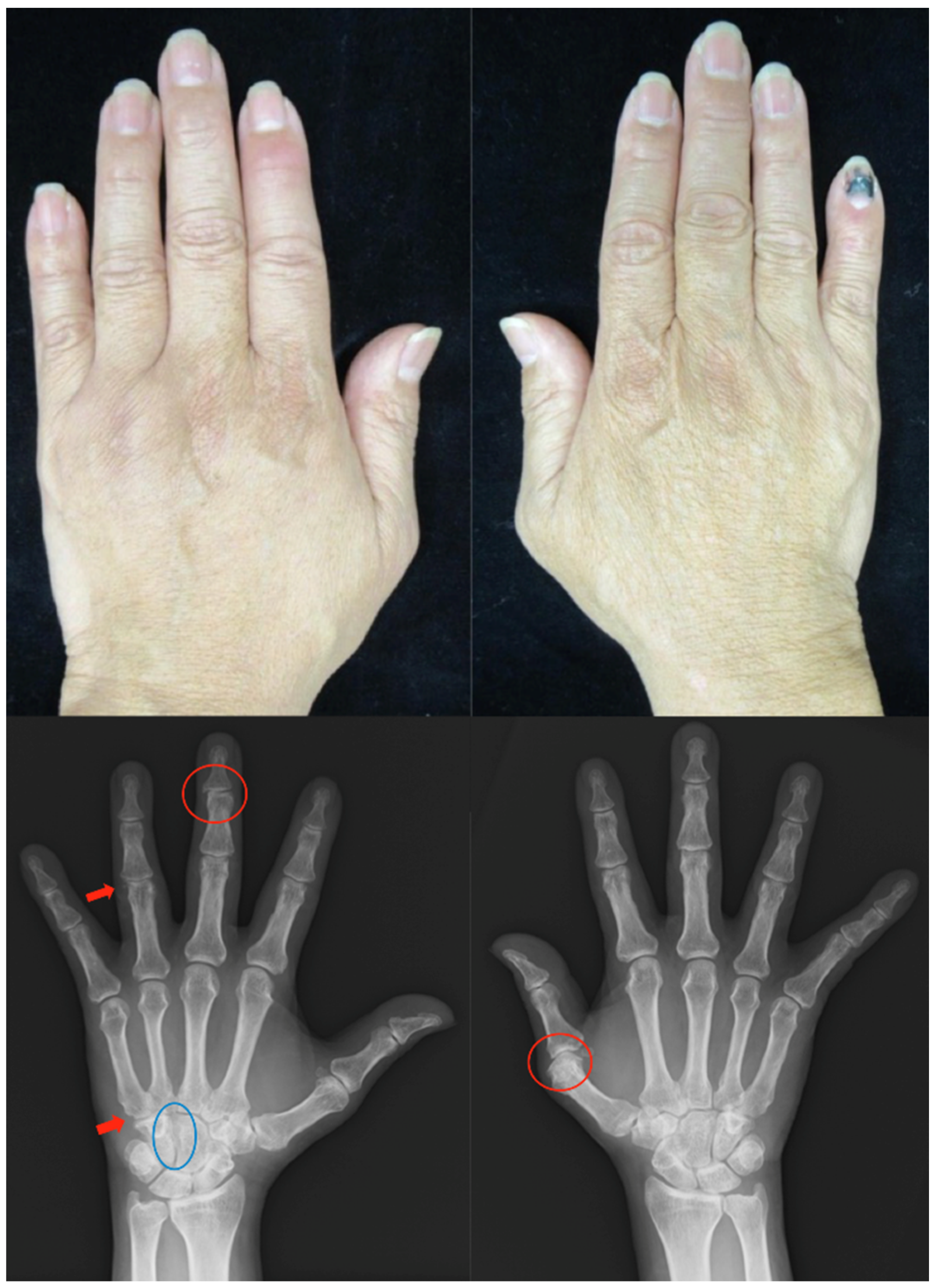
2.4. Case 4
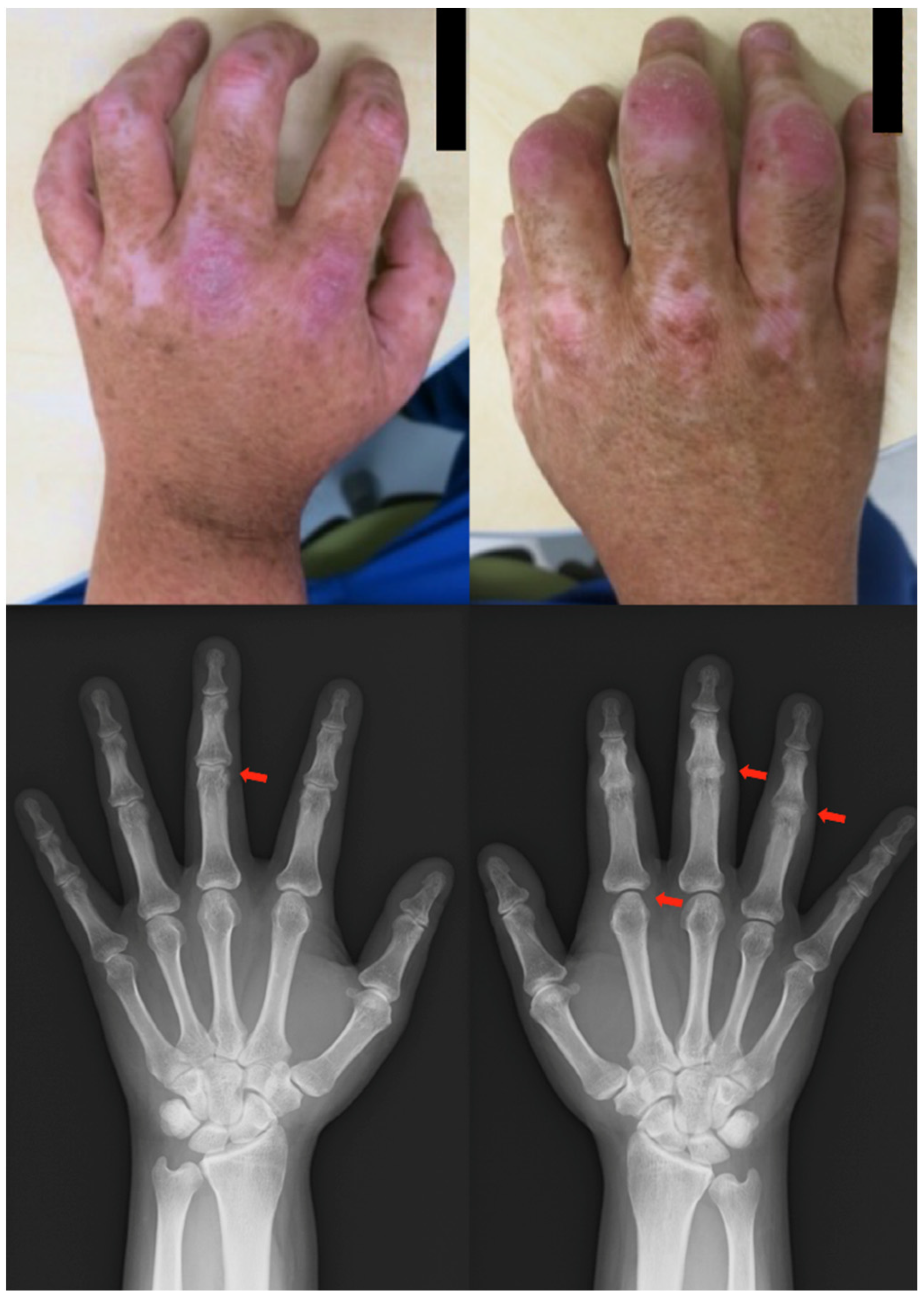
Patient: Man in his 50s
2.5. Case 5
Patient: Woman in her 50s
2.6. Case 6
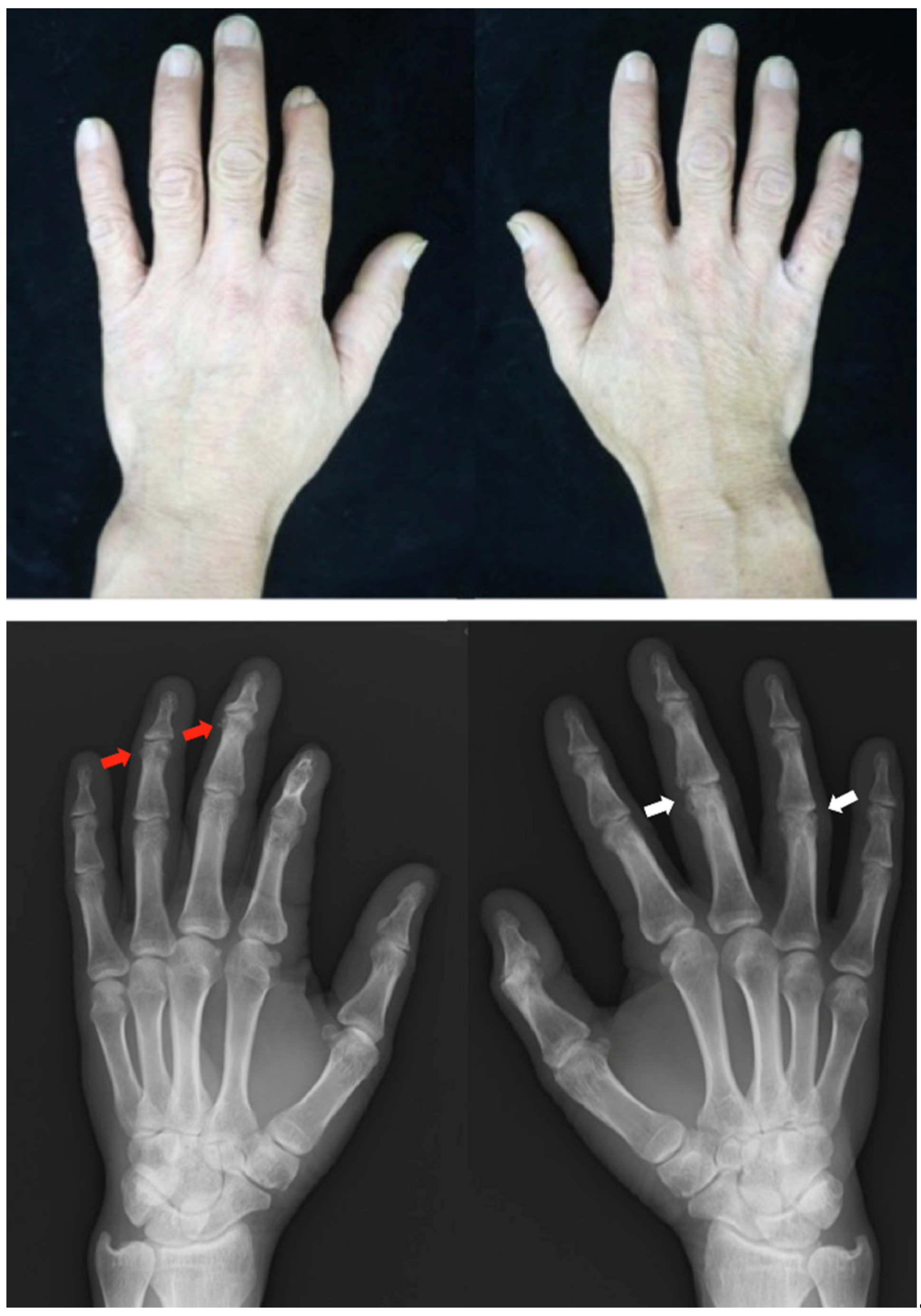
Patient: Man in his 40s
2.7. Case 7
Patient: Man in his 50s
2.8. Summary of the Cases
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Terui, T.; Nakagawa, H.; Etoh, T.; Ozawa, A. A survey of the status of psoriasis conducted using information obtained from health insurance claims provided by health insurance societies. Rinshoiyaku 2014, 30, 279–285. [Google Scholar]
- Kubota, K.; Kamijima, Y.; Sato, T.; Ooba, N.; Koide, D.; Iizuka, H.; Nakagawa, H. Epidemiology of psoriasis and palmoplantar pustulosis: A nationwide study using the Japanese national claims database. BMJ Open 2015, 5, e006450. [Google Scholar] [CrossRef] [PubMed]
- Ohara, Y.; Kishimoto, M.; Takizawa, N.; Yoshida, K.; Okada, M.; Eto, H.; Deshpande, G.A.; Ritchlin, C.T.; Tanaka, A.; Higashiyama, M.; et al. Prevalence and Clinical Characteristics of Psoriatic Arthritis in Japan. J. Rheumatol. 2015, 42, 1439–1442. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, A.; Sakai, R.; Inoue, E.; Harigai, M. Prevalence of patients with rheumatoid arthritis and age-stratified trends in clinical characteristics and treatment, based on the National Database of Health Insurance Claims and Specific Health Checkups of Japan. Int. J. Rheum. Dis. 2020, 23, 1676–1684. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.J.; Nguyen, T.U.; Poon, K.Y.; Herrinton, L.J. The association of psoriasis with autoimmune diseases. J. Am. Acad. Dermatol. 2012, 67, 924–930. [Google Scholar] [CrossRef] [PubMed]
- Zhu, B.; Edson-Heredia, E.; Gatz, J.L.; Guo, J.; Shuler, C.L. Treatment patterns and health care costs for patients with psoriatic arthritis on biologic therapy: A retrospective cohort study. Clin. Ther. 2013, 35, 1376–1385. [Google Scholar] [CrossRef] [PubMed]
- Edson-Heredia, E.; Zhu, B.; Lefevre, C.; Wang, M.; Barrett, A.; Bushe, C.J.; Cox, A.; Wu, J.J.; Maeda-Chubachi, T. Prevalence and incidence rates of cardiovascular, autoimmune, and other diseases in patients with psoriatic or psoriatic arthritis: A retrospective study using Clinical Practice Research Datalink. J. Eur. Acad. Dermatol. Venereol. 2015, 29, 955–963. [Google Scholar] [CrossRef]
- Tsai, T.F.; Wang, T.S.; Hung, S.T.; Tsai, P.I.; Schenkel, B.; Zhang, M.; Tang, C.H. Epidemiology and comorbidities of psoriasis patients in a national database in Taiwan. J. Dermatol. Sci. 2011, 63, 40–46. [Google Scholar] [CrossRef]
- Schett, G.; Lories, R.J.; D’Agostino, M.A.; Elewaut, D.; Kirkham, B.; Soriano, E.R.; McGonagle, D. Enthesitis: From pathophysiology to treatment. Nat. Rev. Rheumatol. 2017, 13, 731–741. [Google Scholar] [CrossRef]
- Merola, J.F.; Espinoza, L.R.; Fleischmann, R. Distinguishing rheumatoid arthritis from psoriatic arthritis. RMD Open 2018, 4, e000656. [Google Scholar] [CrossRef]
- Ito, T.; Takahashi, H.; Kawada, A.; Iizuka, H.; Nakagawa, H.; Japanese Society For Psoriasis Research. Epidemiological survey from 2009 to 2012 of psoriatic patients in Japanese Society for Psoriasis Research. J. Dermatol. 2018, 45, 293–301. [Google Scholar] [CrossRef] [PubMed]
- Aletaha, D.; Smolen, J.S. Diagnosis and Management of Rheumatoid Arthritis: A Review. JAMA 2018, 320, 1360–1372. [Google Scholar]
- Prodanovich, S.; Kirsner, R.S.; Kravetz, J.D.; Ma, F.; Martinez, L.; Federman, D.G. Association of psoriasis with coronary artery, cerebrovascular, and peripheral vascular diseases and mortality. Arch. Dermatol. 2009, 145, 700–703. [Google Scholar] [CrossRef] [PubMed]
- Yamanaka, K.; Nakanishi, T.; Saito, H.; Maruyama, J.; Isoda, K.; Yokochi, A.; Imanaka-Yoshida, K.; Tsuda, K.; Kakeda, M.; Okamoto, R.; et al. Persistent release of IL-1s from skin is associated with systemic cardio-vascular disease, emaciation and systemic amyloidosis: The potential of anti-IL-1 therapy for systemic inflammatory diseases. PLoS ONE 2014, 9, e104479. [Google Scholar] [CrossRef] [PubMed]
- Kato, S.; Matsushima, Y.; Mizutani, K.; Kawakita, F.; Fujimoto, M.; Okada, K.; Kondo, M.; Habe, K.; Suzuki, H.; Mizutani, H.; et al. The Stenosis of Cerebral Arteries and Impaired Brain Glucose Uptake by Long-Lasting Inflammatory Cytokine Release from Dermatitis Is Rescued by Anti-IL-1 Therapy. J. Invest. Dermatol. 2018, 138, 2280–2283. [Google Scholar] [CrossRef] [PubMed]
- Yamanaka, K.; Mizutani, H. “Inflammatory skin march”: IL-1-mediated skin inflammation, atopic dermatitis, and psoriasis to cardiovascular events. J. Allergy Clin. Immunol. 2015, 136, 823–824. [Google Scholar] [CrossRef]
- Mizutani, K.; Shirakami, E.; Ichishi, M.; Matsushima, Y.; Umaoka, A.; Okada, K.; Yamaguchi, Y.; Watanabe, M.; Morita, E.; Yamanaka, K. Systemic Dermatitis Model Mice Exhibit Atrophy of Visceral Adipose Tissue and Increase Stromal Cells via Skin-Derived Inflammatory Cytokines. Int. J. Mol. Sci. 2020, 21, 3367. [Google Scholar] [CrossRef]
- Mizutani, K.; Isono, K.; Matsushima, Y.; Okada, K.; Umaoka, A.; Iida, S.; Habe, K.; Hagimori, K.; Yamazaki, H.; Yamanaka, K. Inflammatory Skin-Derived Cytokines Accelerate Osteoporosis in Mice with Persistent Skin Inflammation. Int. J. Mol. Sci. 2020, 21, 3620. [Google Scholar] [CrossRef]
- Umaoka, A.; Takeuchi, H.; Mizutani, K.; Seo, N.; Matsushima, Y.; Habe, K.; Hagimori, K.; Yamaguchi, Y.; Ikeda, T.; Yamanaka, K. Skin Inflammation and Testicular Function: Dermatitis Causes Male Infertility via Skin-Derived Cytokines. Biomedicines 2020, 8, 293. [Google Scholar] [CrossRef]
- Nakanishi, T.; Mizutani, K.; Iida, S.; Matsushima, Y.; Umaoka, A.; Kondo, M.; Habe, K.; Yamanaka, K. Janus Kinase Inhibitors Ameliorated Gastrointestinal Amyloidosis and Hypoalbuminemia in Persistent Dermatitis Mouse Model. Int. J. Mol. Sci. 2021, 23, 28. [Google Scholar] [CrossRef]
- Iida, S.; Nakanishi, T.; Momose, F.; Ichishi, M.; Mizutani, K.; Matsushima, Y.; Umaoka, A.; Kondo, M.; Habe, K.; Hirokawa, Y.; et al. IL-17A Is the Critical Cytokine for Liver and Spleen Amyloidosis in Inflammatory Skin Disease. Int. J. Mol. Sci. 2022, 23, 5726. [Google Scholar] [CrossRef] [PubMed]
- Dauden, E.; Castaneda, S.; Suarez, C.; Garcia-Campayo, J.; Blasco, A.J.; Aguilar, M.D.; Ferrandiz, C.; Puig, L.; Sanchez-Carazo, J.L.; Working Group on Comorbidity in Psoriasis. Clinical practice guideline for an integrated approach to comorbidity in patients with psoriasis. J. Eur. Acad. Dermatol. Venereol. 2013, 27, 1387–1404. [Google Scholar] [PubMed]
- Perera, G.K.; Di Meglio, P.; Nestle, F.O. Psoriasis. Annu. Rev. Pathol. 2012, 7, 385–422. [Google Scholar]
- Roubille, C.; Richer, V.; Starnino, T.; McCourt, C.; McFarlane, A.; Fleming, P.; Siu, S.; Kraft, J.; Lynde, C.; Pope, J.; et al. Evidence-based Recommendations for the Management of Comorbidities in Rheumatoid Arthritis, Psoriasis, and Psoriatic Arthritis: Expert Opinion of the Canadian Dermatology-Rheumatology Comorbidity Initiative. J. Rheumatol. 2015, 42, 1767–1780. [Google Scholar] [CrossRef] [PubMed]
- Han, C.; Robinson, D.W., Jr.; Hackett, M.V.; Paramore, L.C.; Fraeman, K.H.; Bala, M.V. Cardiovascular disease and risk factors in patients with rheumatoid arthritis, psoriatic arthritis, and ankylosing spondylitis. J. Rheumatol. 2006, 33, 2167–2172. [Google Scholar]
- Figus, F.A.; Piga, M.; Azzolin, I.; McConnell, R.; Iagnocco, A. Rheumatoid arthritis: Extra-articular manifestations and comorbidities. Autoimmun. Rev. 2021, 20, 102776. [Google Scholar]
- Perez-Chada, L.M.; Merola, J.F. Comorbidities associated with psoriatic arthritis: Review and update. Clin. Immunol. 2020, 214, 108397. [Google Scholar]
- Veale, D.J.; Fearon, U. What makes psoriatic and rheumatoid arthritis so different? RMD Open 2015, 1, e000025. [Google Scholar] [CrossRef]
- Popescu, C.; Zofota, S.; Bojinca, V.; Ionescu, R. Anti-cyclic citrullinated peptide antibodies in psoriatic arthritis--cross-sectional study and literature review. J. Med. Life. 2013, 6, 376–382. [Google Scholar]
- Verheul, M.K.; Fearon, U.; Trouw, L.A.; Veale, D.J. Biomarkers for rheumatoid and psoriatic arthritis. Clin. Immunol. 2015, 161, 2–10. [Google Scholar] [CrossRef]
- Shen, R.; Ren, X.; Jing, R.; Shen, X.; Chen, J.; Ju, S.; Yang, C. Rheumatoid Factor, Anti-Cyclic Citrullinated Peptide Antibody, C-Reactive Protein, and Erythrocyte Sedimentation Rate for the Clinical Diagnosis of Rheumatoid Arthritis. Lab. Med. 2015, 46, 226–229. [Google Scholar] [CrossRef] [PubMed]
- Pincus, T.; Gibson, K.A.; Castrejón, I. Update on methotrexate as the anchor drug for rheumatoid arthritis. Bull. Hosp. Jt. Dis. 2013, 71, S9–S19. [Google Scholar]
- Gossec, L.; Baraliakos, X.; Kerschbaumer, A.; de Wit, M.; McInnes, I.; Dougados, M.; Primdahl, J.; McGonagle, D.G.; Aletaha, D.; Balanescu, A.; et al. EULAR recommendations for the management of psoriatic arthritis with pharmacological therapies: 2019 update. Ann. Rheum. Dis. 2020, 79, 700–712. [Google Scholar] [PubMed]
- Smolen, J.S.; Landewe, R.B.M.; Bijlsma, J.W.J.; Burmester, G.R.; Dougados, M.; Kerschbaumer, A.; McInnes, I.B.; Sepriano, A.; van Vollenhoven, R.F.; de Wit, M.; et al. EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs: 2019 update. Ann. Rheum. Dis. 2020, 79, 685–699. [Google Scholar] [CrossRef]
- Taams, L.S. Interleukin-17 in rheumatoid arthritis: Trials and tribulations. J. Exp. Med. 2020, 217, e20192048. [Google Scholar] [CrossRef]
| Patient | Psoriatic Skin Lesion | Joint Involvement | Laboratory Data | Therapy | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Age/Sex | Duration (Years) | Nail | Duration (Years) | Axial | CASPAR ≥ 3 | RF | ACPA | Systemic | Biologic Agent | |
| 1 | 77/M | 30 | - | 33 | - | + | 1100 | 150 | NSAIDs, MTX, & PSL | SEC |
| 2 | 60/M | 2 | + | 1 | + | + | 279 | NA | NSAIDs | SEC |
| 3 | 67/F | 18 | - | 2 | - | N | 100 | 435 | MTX, PSL, & CyA | ADA & CZP GUS, SEC, |
| 4 | 58/M | 1 | + | - | + | 68 | 88.2 | NSAIDs, MTX, & PSL | CZP, & TCZ | |
| 5 | 57/F | 20 | + | 8 | - | + | 30 | 55.6 | NSAIDs, MTX, SASP, & IGU | IFX, ADA, & IXE |
| 6 | 47/M | 16 | - | 10 | - | + | negative to 7 | 5.8 | NSAIDs, MTX, PSL, SASP, & CyA | IFX, BRO, & IXE |
| 7 | 56/M | 3 | + | 8 | - | + | negative | 787.9 | NSAIDs & MTX | ADA, ABA, & IXE |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Akeda, T.; Yamanaka, K. Treatment in Patients with Psoriatic Disease and Rheumatoid Arthritis: Seven Case Reports. Clin. Pract. 2023, 13, 177-189. https://doi.org/10.3390/clinpract13010016
Akeda T, Yamanaka K. Treatment in Patients with Psoriatic Disease and Rheumatoid Arthritis: Seven Case Reports. Clinics and Practice. 2023; 13(1):177-189. https://doi.org/10.3390/clinpract13010016
Chicago/Turabian StyleAkeda, Tomoko, and Keiichi Yamanaka. 2023. "Treatment in Patients with Psoriatic Disease and Rheumatoid Arthritis: Seven Case Reports" Clinics and Practice 13, no. 1: 177-189. https://doi.org/10.3390/clinpract13010016
APA StyleAkeda, T., & Yamanaka, K. (2023). Treatment in Patients with Psoriatic Disease and Rheumatoid Arthritis: Seven Case Reports. Clinics and Practice, 13(1), 177-189. https://doi.org/10.3390/clinpract13010016






