Abstract
Cisplatin, a platinum-based compound, is a cornerstone of modern chemotherapy and remains widely used against a variety of solid tumors, including testicular, ovarian, lung, bladder, and head and neck cancers. Its anticancer activity is primarily attributed to the formation of DNA crosslinks, which obstruct replication and repair, ultimately leading to apoptosis. However, the clinical value of cisplatin is constrained by two major challenges: its toxic profile and the development of resistance. Cisplatin toxicity arises from its interaction not only with tumor DNA but also with proteins and nucleic acids in healthy tissues, resulting in a range of adverse effects, including, but not limited to, nephrotoxicity, ototoxicity, neurotoxicity, and gastrointestinal injury. In pediatric patients, permanent hearing loss represents a particularly debilitating complication. On the other hand, tumor cells can evade cisplatin cytotoxicity through diverse mechanisms, including reduced intracellular drug accumulation, enhanced DNA repair, detoxification by thiol-containing molecules, and alterations in apoptotic signaling. These resistance pathways severely compromise treatment outcomes and often necessitate alternative or combination strategies. This review examines the chemical structure of cisplatin, the molecular mechanisms of cisplatin cytotoxicity and cisplatin-induced resistance, as well as the main applications in cancer management and the complications associated with its clinical use.
1. Introduction
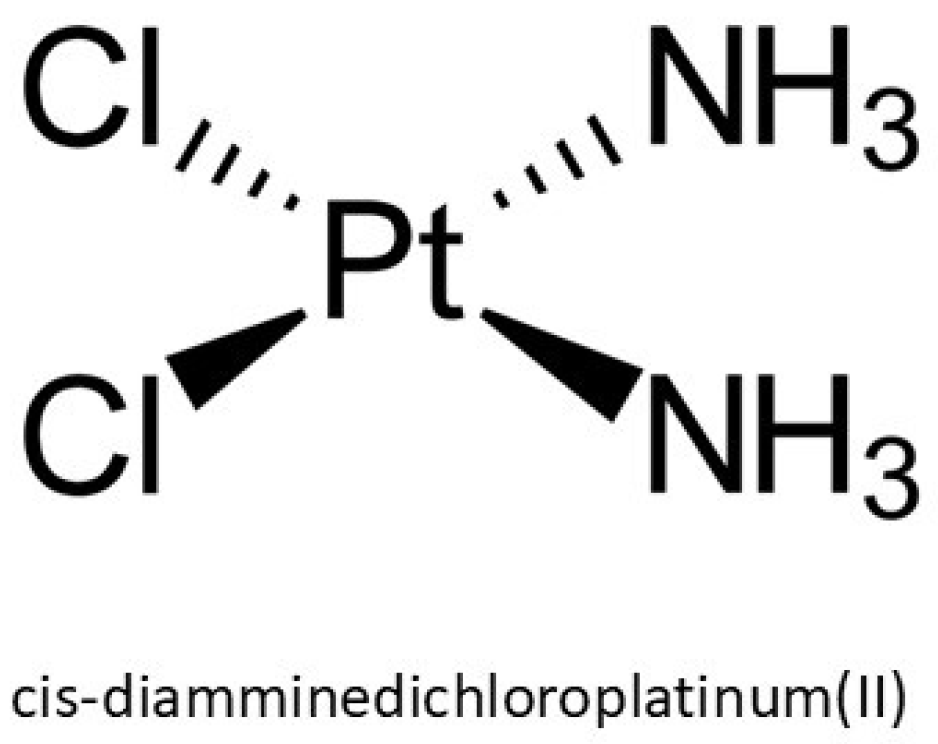
Over the past several decades, platinum-based chemotherapeutics, particularly cis-diamminedichloroplatinum(II), known as cisplatin (CAS No. 15663-27-1), have emerged as pivotal agents in the oncological pharmacopeia due to their robust cytotoxic efficacy against a spectrum of malignancies, including sarcomas and epithelial neoplasms. Structurally, cisplatin is a square planar coordination complex composed of a central platinum atom bonded to two chloride and two ammine ligands, with a molecular weight of 301.1 g/mol (Figure 1). Its physicochemical properties such as a high melting point (~270 °C), a density of 3.74 g/cm3, and aqueous solubility of 2.53 g/L at 25 °C, influence its pharmacokinetics and bioavailability [1]. Despite its chemical stability, it remains susceptible to isomerization under extended exposure, potentially diminishing its pharmacological activity. Initially synthesized by Peyrone and structurally elucidated by Werner, its therapeutic relevance was not recognized until Rosenberg’s observation of mitotic inhibition in Escherichia coli during the 1960s, which catalyzed its rapid adoption in clinical oncology and eventual FDA approval in the late 1970s [2]. The discovery that cisplatin was the compound mediating the observed biological response inspired extensive research into the therapeutic potential of platinum-based coordination complexes, as well as analogous compounds of palladium and other noble metals, for oncological applications.
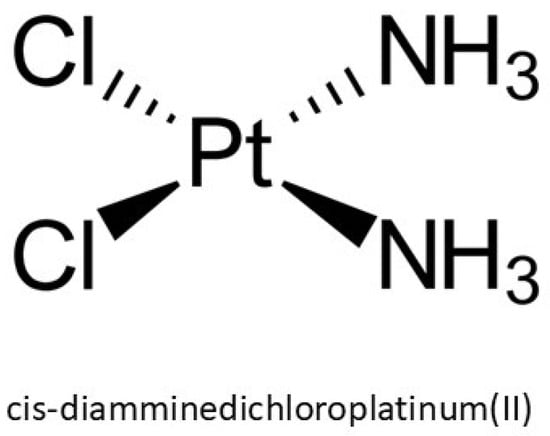
Figure 1.
Chemical structure of cisplatin.
Cisplatin’s mode of action, involving DNA crosslinking and apoptosis induction, underpins its frontline role in chemotherapeutic regimens. However, its efficacy is frequently undermined by the emergence of both intrinsic and extrinsic resistance mechanisms within tumor cells. Extrinsic resistance, often acquired through treatment-induced genomic mutations or alterations in drug target pathways, impairs cisplatin interaction with intracellular biomolecular targets. Meanwhile, intrinsic resistance, a pre-existing refractoriness, can render malignancies nonresponsive to platinum agents from the outset, posing significant hurdles to therapeutic success. Additionally, the therapeutic utility of the compound is limited by its propensity to induce dose-limiting toxicities in renal and gastrointestinal systems, necessitating combinatorial strategies to mitigate adverse effects while enhancing cytotoxic synergy. Consequently, comprehensive understanding of chemical reactivity of the cisplatin, cellular uptake, and biotransformation pathways is imperative to optimize its clinical utility and overcome resistance. As efforts continue to refine treatment protocols and develop second-generation platinum analogs, the molecular and pharmacodynamic insights into this class of drugs remain central to advancing precision oncology and overcoming the multifaceted challenges inherent to chemotherapeutic intervention.
2. Pharmacokinetics of Cisplatin
The conceptual breakthrough that catalyzed the incorporation of platinum-based molecules into chemotherapeutic protocols originated from experimental findings demonstrating that a cis-oriented platinum(II) coordination complex could effectively arrest the proliferation of both prokaryotic and mammalian cell lines, including aggressive tumor phenotypes in rat models [3,4]. Historically, skepticism toward the clinical use of heavy metals such as platinum stemmed from their inherent toxicity and associations with other hazardous elements like mercury. However, following extensive preclinical evaluations conducted under the supervision of the National Cancer Institute, this compound—subsequently designated as cisplatin—secured regulatory approval for oncological application by the late 1970s by the Food and Drug Administration [5]. The clinical armamentarium was later expanded to include structurally analogous agents such as carboplatin and oxaliplatin, each characterized by distinct pharmacokinetic profiles.
Unlike conventional small-molecule anticancer agents, platinum drugs exhibit distinct coordination chemistry in aqueous media. Under physiological pH (~7.2), cisplatin undergoes aquation, progressively replacing chloride ligands with hydroxyl groups, leading to the formation of dihydroxo species [6]. This reaction proceeds with a half-life of about one hour at body temperature (~35.5 °C). Once introduced intravenously, the majority of circulating cisplatin (68–98%) binds rapidly to plasma proteins, particularly albumin [7,8,9]. Each albumin molecule can coordinate multiple cisplatin units via histidine and methionine residues [6]. Renal elimination accounts for the excretion of both unbound cisplatin and a minor fraction of protein-associated complexes [10].
The cytotoxic effect of cisplatin and its analogues carboplatin and oxaliplatin is attributed to interference with nuclear DNA processes, preventing both duplication and repair, thereby halting cellular proliferation. For this effect to occur, the drug must traverse the plasma membrane and accumulate in the nucleus. Entry occurs largely through passive diffusion, with uptake proportional to extracellular drug levels up to approximately 3 mM [11]. A possible, though controversial, auxiliary mechanism involves copper transporters (SLC31A1 and SLC31A2). These proteins, containing methionine- and histidine-rich extracellular domains, could theoretically mimic albumin in binding platinum complexes [12]. Nonetheless, experimental evidence suggests that platinum–sulfur interactions in these transporters are neither stable nor favorable, raising doubts about their physiological role. At present, the contribution of copper transporters to platinum drug uptake remains uncertain [13].
Another proposed mechanism for cellular uptake of cisplatin involves members of the solute carrier 22 (SLC22A) family, which encompasses both organic cation transporters (OCTs) and organic anion transporters (OATs). The hypothesis concerning OCT-mediated import is grounded in the fact that, once dissolved in aqueous media, cisplatin undergoes hydrolytic transformations that yield positively charged complexes, which could represent potential substrates for cation transporters. This assumption gains plausibility from the observation that OCTs are abundantly expressed in organs and tissues commonly affected by cisplatin-induced toxicity [14]. In parallel, it has been argued that cisplatin, under physiological conditions, can also form carbonate-containing derivatives bearing negative charges. Such anionic species might be handled by OATs, which likewise belong to the SLC22A transporter family. Clinical and histological evidence supports this notion, since OATs, like OCTs, are highly expressed in renal tissue, the cochlea, peripheral nerves, and other sites where cisplatin-induced damage is prevalent [15]. Importantly, these same tissues also display expression of copper transporters, raising the unresolved possibility that both SLC22A- and SLC31A-type carriers act in concert to permit platinum drug accumulation to toxic levels [16]. From a therapeutic standpoint, the clinical use of cisplatin is constrained by its narrow effective dose window. Increasing the drug concentration beyond this range in an attempt to overcome tumor resistance is not feasible, as further intracellular accumulation would exacerbate toxicity in healthy tissues. In clinical practice, these limitations are reflected in the cumulative dosing strategies used in oncology. Cisplatin regimens typically aim for cumulative doses of approximately 180–200 mg/m2, a range broadly recognized as sufficient to achieve therapeutic benefit while minimizing unacceptable toxicity. Several clinical studies, particularly in head and neck and thoracic malignancies, indicate that cumulative doses of ≥200 mg/m2 are associated with improved treatment outcomes, although escalation beyond this level correlates with a markedly increased risk of nephrotoxicity, ototoxicity, and other dose-limiting adverse effects [17,18,19,20,21,22,23,24,25,26]. Incorporating these established dose thresholds underscores the delicate balance between efficacy and toxicity that defines cisplatin’s therapeutic window.
3. Molecular Mechanisms of Cisplatin Cytotoxicity
A variety of membrane proteins have been implicated in the trafficking of platinum-based drugs. These include ATP-dependent efflux transporters such as the multidrug resistance-associated proteins (MRPs) and the copper-transporting ATPases (ATP7A and ATP7B), as well as solute carrier systems comprising copper transporters (CTR1), aquaporins (AQP2, AQP9), and additional members of the SLC family. The efflux pump multidrug resistance protein 1 (MDR1), also known as P-glycoprotein and belonging to the ATP-binding cassette superfamily, has long been recognized for its contribution to drug resistance [27,28]. In both yeast and mammalian models, the copper transporter CTR1 has been shown to facilitate cisplatin uptake [29]. Evidence from human cells further demonstrates that exposure to cisplatin accelerates the degradation of CTR1 at the plasma membrane, which diminishes drug influx and thereby promotes resistance [30,31,32]. Functional disruption of CTR1 confers marked resistance to cisplatin in vivo, whereas elevated expression of this transporter increases intracellular platinum content and typically enhances chemosensitivity. Another protein, the transmembrane protein 205 (TMEM205), has been identified as a contributor to platinum resistance. Expression profiling reveals that TMEM205 is enriched in the liver, pancreas, and adrenal glands, and its overexpression in cisplatin-resistant (CP-r) cell lines has been associated with decreased drug sensitivity, suggesting its potential as a therapeutic biomarker or target [27]. In addition, although glucose transporter 1 (GLUT1) does not appear to serve as a direct conduit for cisplatin, aberrant localization of this protein intensifies the resistant phenotype [27,33].
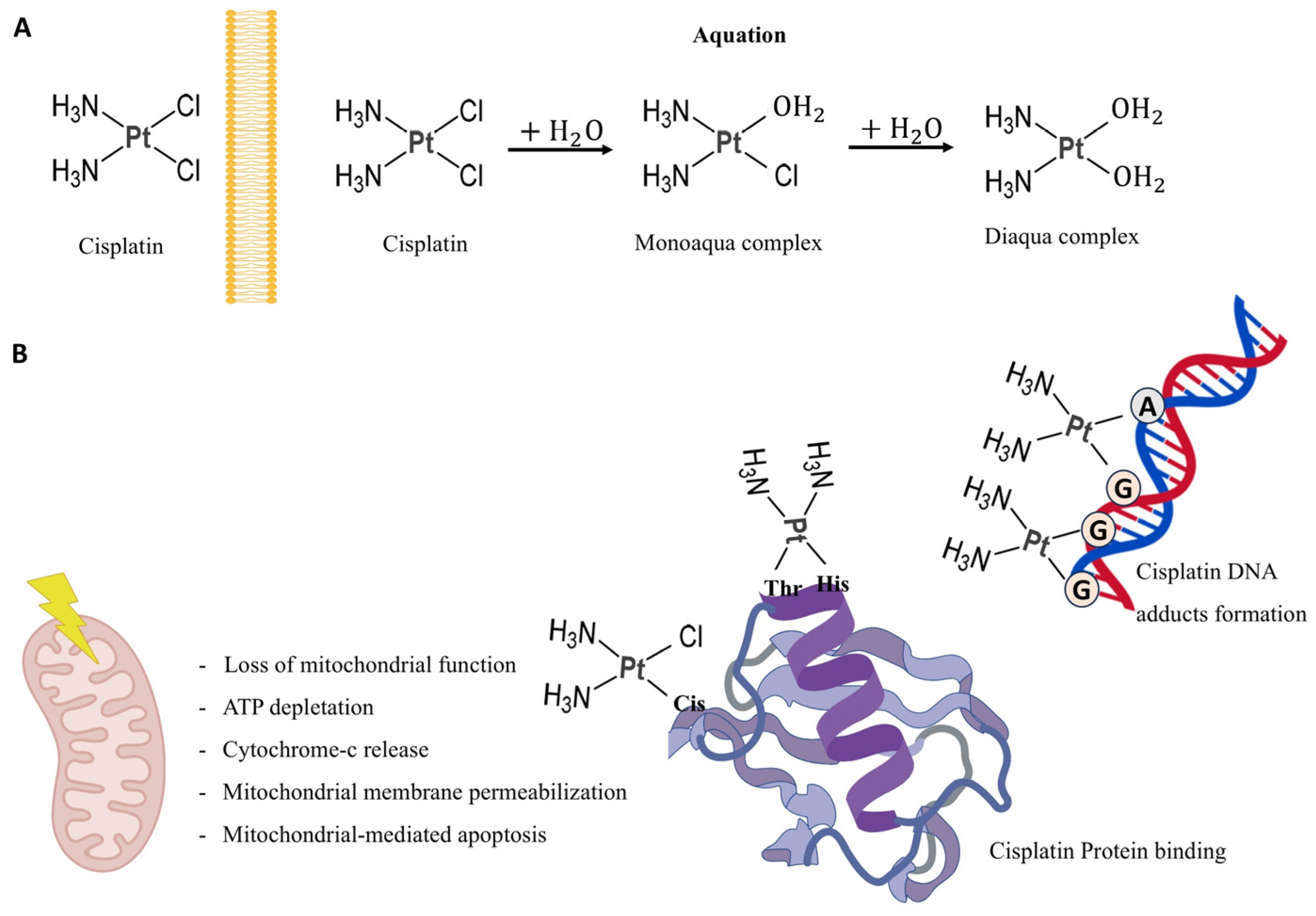
Once inside the cytoplasm, cisplatin undergoes aquation in which its chloride ligands are replaced by water molecules, producing highly electrophilic species capable of reacting with cellular nucleophiles (Figure 2).
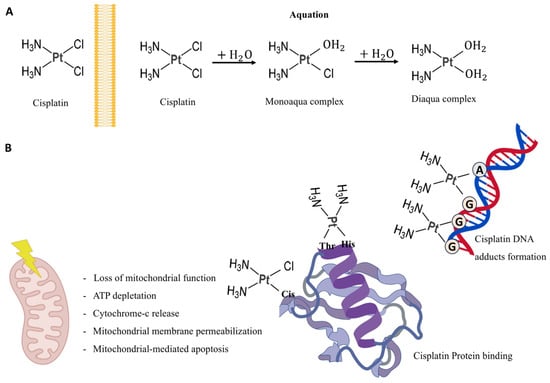
Figure 2.
Graphic representation of cisplatin modification and functions. (A) Cisplatin acquation process. (B) Cisplatin effects on mitochondria and its binding to DNA and proteins. Thr = Thirosine, His = Histidine, Cis = Cisterine, A = Adenine, G = Guanosine. Created with BioGDP.com [34].
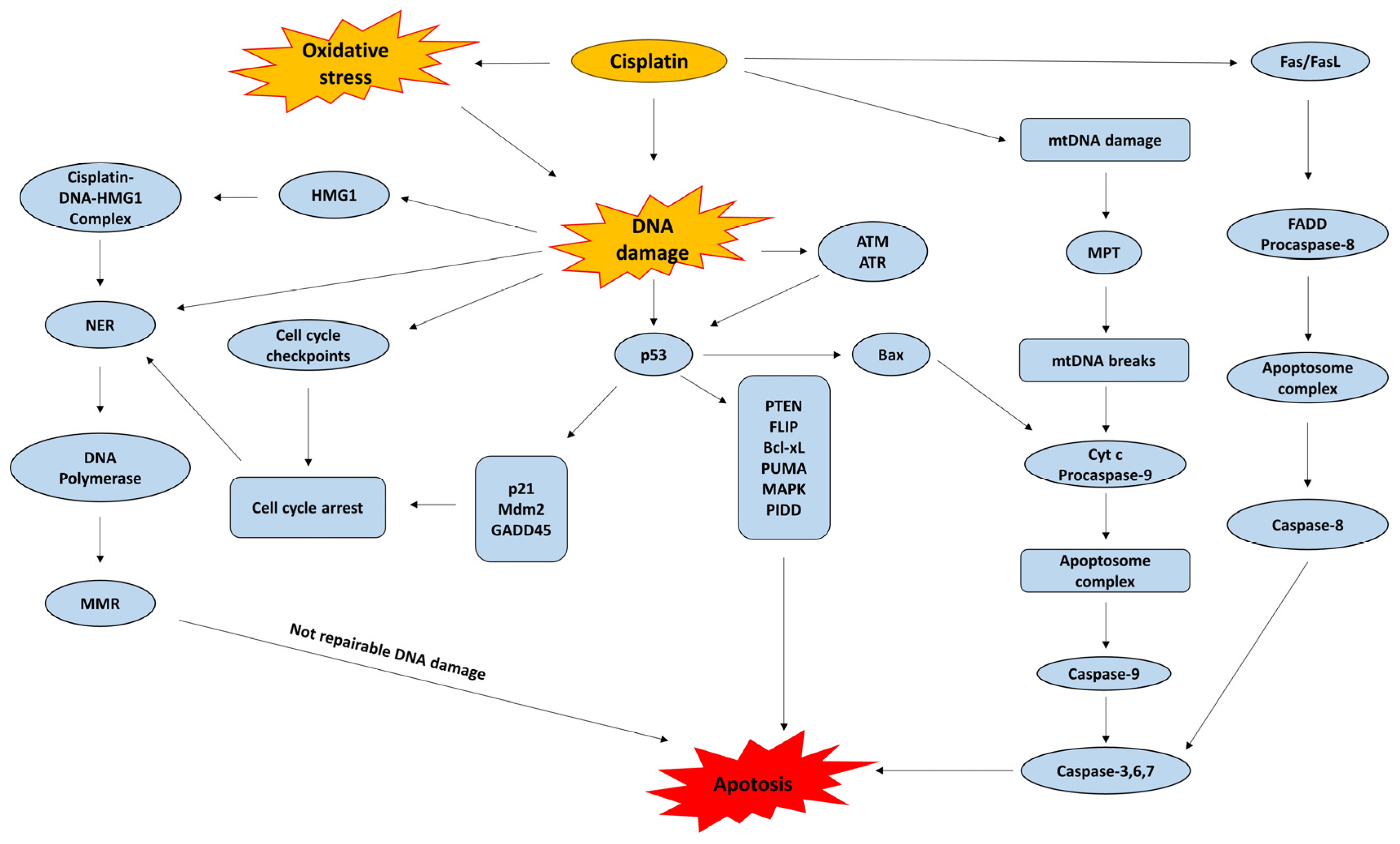
These activated forms interact readily with sulfhydryl groups in proteins and with nitrogen atoms in nucleic acids. The primary biological target is DNA, where cisplatin preferentially coordinates with the N7 position of purine bases [35]. The most prevalent lesions are 1,2-intrastrand cross-links involving adjacent guanine residues d(GpG), which account for about 90% of adducts, and adenine-guanine cross-links d(ApG), comprising roughly 10%. Additional DNA distortions include 1,3-intrastrand d(GpXpG) adducts, inter-strand cross-links, and nonproductive binding events, all of which contribute to cytotoxicity. The centrality of DNA as a damage target is underscored by the hypersensitivity to cisplatin observed in organisms lacking proficient DNA repair pathways, spanning both prokaryotic and eukaryotic systems [36,37]. The accumulation of cisplatin-induced DNA lesions initiates a cascade of downstream apoptotic signaling pathways, which constitutes a central mechanistic determinant of its anticancer efficacy (Figure 3).
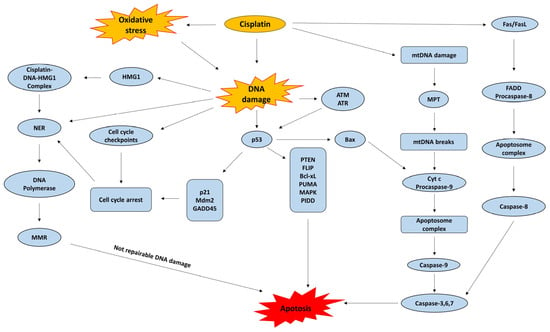
Figure 3.
Cisplatin mechanisms of action. NER: Nucleotide Excision Repair; MMR: Mismatch Repair; p53: Tumor protein p53; p21: Cyclin-dependent kinase inhibitor 1 (CDKN1A); Mdm2: Mouse double minute 2 homolog; GADD45: Growth Arrest and DNA Damage-inducible protein 45; HMG1: High-Mobility Group Box 1 protein; ATM: Ataxia Telangiectasia Mutated; ATR: ATM and Rad3-related; PTEN: Phosphatase and Tensin Homolog; FLIP: FLICE-like inhibitory protein; Bcl-xL: B-cell lymphoma—extra large; PUMA: p53 upregulated modulator of apoptosis; MAPK: Mitogen-Activated Protein Kinase; PIDD: p53-induced protein with a death domain; Bax: Bcl-2-associated X protein; mtDNA: Mitochondrial DNA; MPT: Mitochondrial Permeability Transition; Cyt c: Cytochrome c; Fas/FasL: Fas receptor/Fas ligand; FADD: Fas-Associated protein with Death Domain.
In healthy cells, intracellular reactive oxygen species (ROS) levels are tightly regulated through a dynamic balance between their generation and clearance. This homeostasis is largely sustained by enzymatic and nonenzymatic defense systems, including reduced glutathione (GSH), superoxide dismutase (SOD), and catalase (CAT) [38]. When this balance collapses under conditions of oxidative stress, excessive ROS accumulation can provoke irreversible injury to macromolecules such as proteins, lipids, and nucleic acids, thereby favoring the onset and progression of several cancerous and non-cancerous diseases [39,40]. Compared with non-malignant cells, tumor cells typically exist in a state of heightened oxidative pressure, a consequence of aberrant signaling cascades, elevated metabolic flux, and defects in mitochondrial function. Among the mechanisms underlying the cytotoxic effects of cisplatin, ROS generation is regarded as a central pathway. Mitochondria are particularly vulnerable to this insult, where cisplatin exposure leads to oxidation of critical sulfhydryl groups in mitochondrial proteins, suppression of calcium uptake, and dissipation of the mitochondrial membrane potential [41].
Oxidative stress disrupts essential cellular processes, and cisplatin contributes to this imbalance by stimulating the production of ROS in addition to its well-established genotoxic activity. The induction of cell death by cisplatin involves the rapid engagement of multiple signaling cascades, the exact nature of which varies depending on the cellular context and tumor type. Both the level of cis-diamminedichloroplatinum(II) exposure and the duration of treatment are critical determinants of the extent of ROS accumulation [42]. The regulation of intracellular redox balance relies heavily on thiol-containing molecules, whose sulfhydryl (-SH) groups play a central role in detoxification. However, under certain conditions, these thiols may undergo oxidation to form thiyl radicals. Such reactive intermediates can subsequently react with molecular oxygen, amplifying the generation of ROS and exacerbating oxidative damage [43].
Elevated concentrations of ROS are capable of initiating programmed cell death through both receptor-mediated (extrinsic) and mitochondria-dependent (intrinsic) mechanisms [44]. Within the extrinsic cascade, ROS production occurs as a proximal signal downstream of Fas ligand engagement, facilitating phosphorylation-dependent activation of Fas. This step is required for recruitment of adaptor molecules such as Fas-associated protein with a death domain and the subsequent activation of caspase-8, ultimately driving apoptosis [45,46]. In the intrinsic pathway, ROS promote the release of cytochrome c from mitochondria by interfering with the regulatory balance of Bcl-2 family proteins—enhancing the activity of pro-apoptotic members such as Bax and Bak while compromising the function of anti-apoptotic counterparts including Bcl-2 and Bcl-xL [47,48,49]. At even higher intracellular ROS burdens, cells may undergo not only apoptosis but also necrosis, reflecting overwhelming oxidative injury [50]. Furthermore, ROS have been implicated in triggering autophagy, a catabolic survival-to-death mechanism characterized by the sequestration of damaged organelles and protein aggregates into autophagosomes for lysosomal degradation [51].
In addition to apoptosis, increasing evidence indicates that cisplatin can trigger multiple forms of regulated necrosis, including ferroptosis, necroptosis and pyroptosis, depending on the cellular context and the intensity of oxidative and metabolic stress [52].
Pyroptosis is an inflammatory form of programmed cell death orchestrated by gasdermin (GSDM) family proteins, particularly GSDMD and GSDME, whose activation culminates in membrane pore formation and the release of proinflammatory cytokines [53]. Central to this mechanism is the NLRP3 inflammasome, a complex of NLRP3, ASC, and pro-caspase-1, which governs caspase-mediated cleavage of gasdermins and thereby initiates pore formation and cell lysis [54]. Cisplatin has emerged as a potent upstream driver of this pathway in the inner ear, where it stimulates NLRP3 activation, enhances GSDMD/GSDME processing, and accelerates pyroptotic death in cochlear sensory and supporting cells [55,56]. Thus, reduction in pyroptosis may induce cisplatin resistance in neoplastic cells [57,58,59].
Ferroptosis, an iron-dependent, lipid peroxidation-driven form of regulated necrosis, is increasingly recognized as a contributor to cisplatin toxicity. By depleting glutathione and impairing mitochondrial redox balance, cisplatin promotes accumulation of lipid hydroperoxides, thereby lowering the threshold for ferroptotic death [60]. A recent study demonstrated that triggering ferroptosis can counteract cisplatin resistance in gastric cancer. Increased ATF3 expression drives ferroptotic cell death by inhibiting the Nrf2–Keap1–xCT pathway, thereby limiting tumor progression and enhancing cisplatin sensitivity in gastric cancer cells [61].
Necroptosis is a regulated cell death program that shares morphological features with unregulated necrosis but is driven by a defined molecular apparatus distinct from apoptosis. Its core effector module consists of RIP1, RIP3, and MLKL, which assemble into a cytotoxic complex that disrupts membrane integrity and promotes inflammatory mediator release [62]. Cisplatin acts as a potent upstream initiator of this cascade, particularly within renal proximal tubular cells where the drug preferentially accumulates [63]. Increasing intracellular cisplatin burden intensifies necroptotic signaling, elevates RIP1 and RIP3 activity, and amplifies cytokine production, thereby accelerating tissue injury and contributing to cisplatin-induced acute kidney injury [64]. Targeted suppression of necroptosis, either via deletion of RIP1, RIP3, or MLKL or through pharmacologic inhibition, markedly attenuates cisplatin-associated renal toxicity without diminishing its anticancer efficacy [65]. This direct functional linkage positions the necroptotic pathway as a central mediator of cisplatin nephrotoxicity. Cisplatin-driven necroptosis also intersects with apoptosis and autophagy, revealing a broader regulatory network. The proapoptotic signaling of cisplatin can impair autophagic flux, reducing a key survival mechanism and increasing reliance on necroptotic cell death.
Collectively, these alternative programmed necrotic pathways do not replace apoptotic signaling but operate in parallel or downstream of it, particularly under conditions of severe oxidative stress [60,66,67]. Their contribution may enhance antitumor cytotoxicity but also exacerbate organ-specific toxicity, reinforcing the complexity of the cell-death landscape of cisplatin.
When intracellular chloride concentration is low, cisplatin undergoes aquation, producing highly reactive ionic species such as the monoaqua complex [cis-(NH3)2PtCl(H2O)]+ and the diaqua complex [cis-(NH3)2Pt(H2O)2]2+ [68,69,70]. These hydrolyzed derivatives exhibit reactivities up to a thousand-fold greater than the parent drug, largely due to their ability to interfere with mitochondrial oxidative metabolism by uncoupling oxidative phosphorylation [71]. This mitochondrial dysfunction is accompanied by calcium efflux, leading to transient elevations in cytosolic calcium concentration, which severely perturbs calcium homeostasis and compromises normal cellular physiology.
Mitochondrial glutathione (GSH) plays a central role in sustaining redox balance and preserving enzymatic thiol groups in their reduced, active state. Loss of this protective system results in irreversible enzyme inactivation. Cisplatin toxicity, especially in renal tissue, has been linked to depletion of both GSH and protein-associated sulfhydryl groups. Nicotinamide adenine dinucleotide (NADH), another crucial cofactor required for maintaining reduced thiols, also decreases after cisplatin exposure. The combined reduction of GSH and NADH compromises mitochondrial dehydrogenase activity, uncouples oxidative phosphorylation, and promotes hydroxyl radical formation, thereby amplifying oxidative stress [72]. These reactive species initiate the peroxidation of polyunsaturated lipids and structural proteins, leading to a self-amplifying lipid peroxidation cascade that ultimately results in irreversible damage to mitochondrial and cellular membranes [73,74].
Ultimately, disruption of calcium balance and oxidative injury converge to drive key pathological events, including loss of mitochondrial function, ATP depletion, impairment of vital cofactors, and collapse of energy metabolism. These processes culminate in apoptotic signaling and, in severe cases, necrotic cell death. Interestingly, modulation of intracellular calcium levels through exogenous supplementation has been proposed as a possible protective strategy, since calcium may compete with cisplatin for binding sites and thereby attenuate certain toxic side effects [35,75].
Programmed cell death via apoptosis is a highly regulated molecular process that requires energy and is marked by specific structural transformations, including nuclear condensation, cell shrinkage, external display of phosphatidylserine, and membrane blebbing. The central regulators of this cascade are caspases, a conserved protease family that cleaves substrates after aspartate residues [76,77]. Caspases are subdivided into initiators, which include caspase-8 and caspase-9, and executioners, such as caspase-3 and caspase-7. Initiator caspases function as sensors that respond to upstream apoptotic stimuli: caspase-8 is recruited and activated at plasma membrane death receptors, while caspase-9 is activated within the apoptosome after mitochondrial cytochrome-c release. Once activated, executioner caspases dismantle the cell by targeting key nuclear and cytoplasmic proteins, such as PARP and inhibitors of caspase-activated DNases [78,79].
Cisplatin is particularly effective in driving apoptosis, and failure of apoptotic signaling has been directly associated with the emergence of drug resistance. Apoptosis can proceed through two primary molecular routes [80]. The extrinsic pathway begins when ligands of the TNF-α receptor superfamily bind to their cognate receptors, leading to receptor clustering and recruitment of adaptor proteins that nucleate the death-inducing signaling complex (DISC). This complex facilitates procaspase-8 activation, thereby launching the apoptotic cascade. The intrinsic pathway, in contrast, is triggered by intracellular stresses such as cisplatin-induced DNA lesions, which compromise mitochondrial integrity and provoke cytochrome-c efflux. Cytochrome-c then interacts with APAF-1 to assemble the apoptosome, resulting in procaspase-9 activation. The balance of pro-apoptotic (e.g., Bax, Bak) and anti-apoptotic (e.g., Bcl-2, Bcl-xL) Bcl-2 family proteins dictates the release of cytochrome-c, thus serving as a decisive checkpoint for mitochondrial apoptosis [81]. Cisplatin activates a spectrum of stress-responsive signaling networks, and the cellular outcome—either apoptotic death or the acquisition of resistance—depends on how these pathways are integrated and executed [80,82].
Protein kinase C (PKC) represents a group of phospholipid-activated enzymes that phosphorylate numerous downstream substrates and thereby influence cell growth, apoptosis, and stress signaling [83,84]. The functional contribution of PKC to cisplatin response remains controversial: in some cellular models, PKC activation enhances susceptibility to cisplatin, while in others, drug sensitivity requires PKC inhibition or downregulation [85,86,87,88]. These divergent outcomes likely reflect differences in isozyme-specific signaling. Distinct PKC isoforms can modulate pathways related to apoptosis, DNA repair capacity, and drug transport, meaning that the effect of PKC activity on cisplatin resistance is not universal but instead depends on which isoforms are dominant in a given cellular context [83,89].
Mitogen-activated protein kinases (MAPKs) constitute a conserved family of serine/threonine kinases that integrate extracellular stimuli to control processes such as proliferation, differentiation, and survival [90,91,92]. Cisplatin exposure has been consistently linked to activation of the extracellular signal-regulated kinase (ERK) pathway, though experimental outcomes differ on whether ERK activity promotes resistance to drug-induced apoptosis or instead enhances cisplatin-mediated cytotoxicity [93,94,95,96,97].
Notably, ERK activation occurs upstream of p53 signaling: ERK directly phosphorylates p53, resulting in transcriptional induction of target genes such as p21, growth arrest and DNA damage-inducible 45 (GADD45), and Mdm2 [98]. Through these interactions, ERK activation can enforce cell cycle arrest, which provides an interval for repair of cisplatin-induced DNA lesions in a p53-dependent manner.
Cellular stress responses strongly influence cisplatin-triggered apoptosis, in large part through activation of the p38 MAPK signaling cascade. One identified downstream target of this pathway is the epidermal growth factor receptor (EGFR), whose internalization upon cisplatin exposure occurs following p38-dependent phosphorylation [99]. p38 MAPK also exerts pro-apoptotic effects through regulation of the adaptor protein p18, which forms complexes with p53 and promotes transcription of the pro-death genes PUMA and NOXA [100]. Collectively, these findings underscore the pivotal role of p38 MAPK as a mediator of cisplatin-induced apoptotic signaling.
The c-Jun N-terminal kinase (JNK), also referred to as stress-activated protein kinase, is a central effector of signaling pathways triggered by environmental and genotoxic stressors. DNA damage, including lesions produced by platinum-based agents, serves as a potent activator of JNK. Both stereoisomeric forms of cisplatin (cis and trans) have been shown to stimulate JNK signaling cascades, highlighting the pathway’s broad responsiveness to platinum-induced stress [101]. A pivotal link between JNK activity and apoptosis is mediated through p73, a pro-apoptotic transcription factor and structural homolog of p53. Upon activation, p73 physically associates with JNK, forming a functional complex that enhances transcription of death-promoting genes, thereby reinforcing cisplatin-induced cell death [102,103]. Beyond its interaction with p73, JNK also interfaces with mitochondrial apoptotic regulators, modulating the activity of Bcl-2 family proteins and facilitating cytochrome c release. In parallel, JNK contributes to death receptor signaling, where it amplifies caspase activation and apoptosis induction. These converging inputs position JNK as an essential signaling hub that integrates nuclear DNA damage with both extrinsic and intrinsic apoptotic machinery during cisplatin treatment.
Akt, a serine/threonine kinase downstream of phosphoinositide 3-kinase (PI3K), is a central regulator of prosurvival signaling [104]. Following cisplatin exposure, DNA damage has been shown to activate Akt-dependent phosphorylation of the pro-apoptotic Bcl-2 family protein BAD at serine-136 [105]. This modification of BAD is observed in both cisplatin-sensitive and -resistant tumor cells, and its phosphorylation is essential to sustain viability after drug treatment. Mechanistically, BAD regulation involves dual input from distinct kinase pathways: ERK signaling mediates phosphorylation at serine-112, while PI3K/Akt controls phosphorylation at serine-136. Inhibition of either cascade sensitizes ovarian carcinoma cells to cisplatin, underscoring the cooperative role of ERK and Akt in converging on BAD to neutralize its apoptotic function [105].
Cisplatin response also engages the p53 axis. p53, a transcription factor with a normally short half-life, is stabilized and activated through phosphorylation by the ATM kinase in response to DNA strand breaks. Once activated, p53 induces Mdm2, an E3 ubiquitin ligase that contributes to feedback regulation of its stability. Beyond this autoregulatory loop, p53 transactivates numerous genes involved in DNA repair, cell cycle arrest, and apoptosis. In cisplatin-treated cells, p53-dependent death pathways include degradation of the anti-apoptotic FLIP protein, direct antagonism of Bcl-xL function, induction of PTEN, and suppression of AMPK signaling [106]. Interestingly, cisplatin cytotoxicity is not limited to p53-proficient cells; p53-deficient systems can still undergo apoptosis upon treatment. This indicates that alternative, p53-independent mechanisms exist to mediate stress responses and cell death following cisplatin-induced DNA lesions.
4. Mechanisms of Cisplatin Resistance
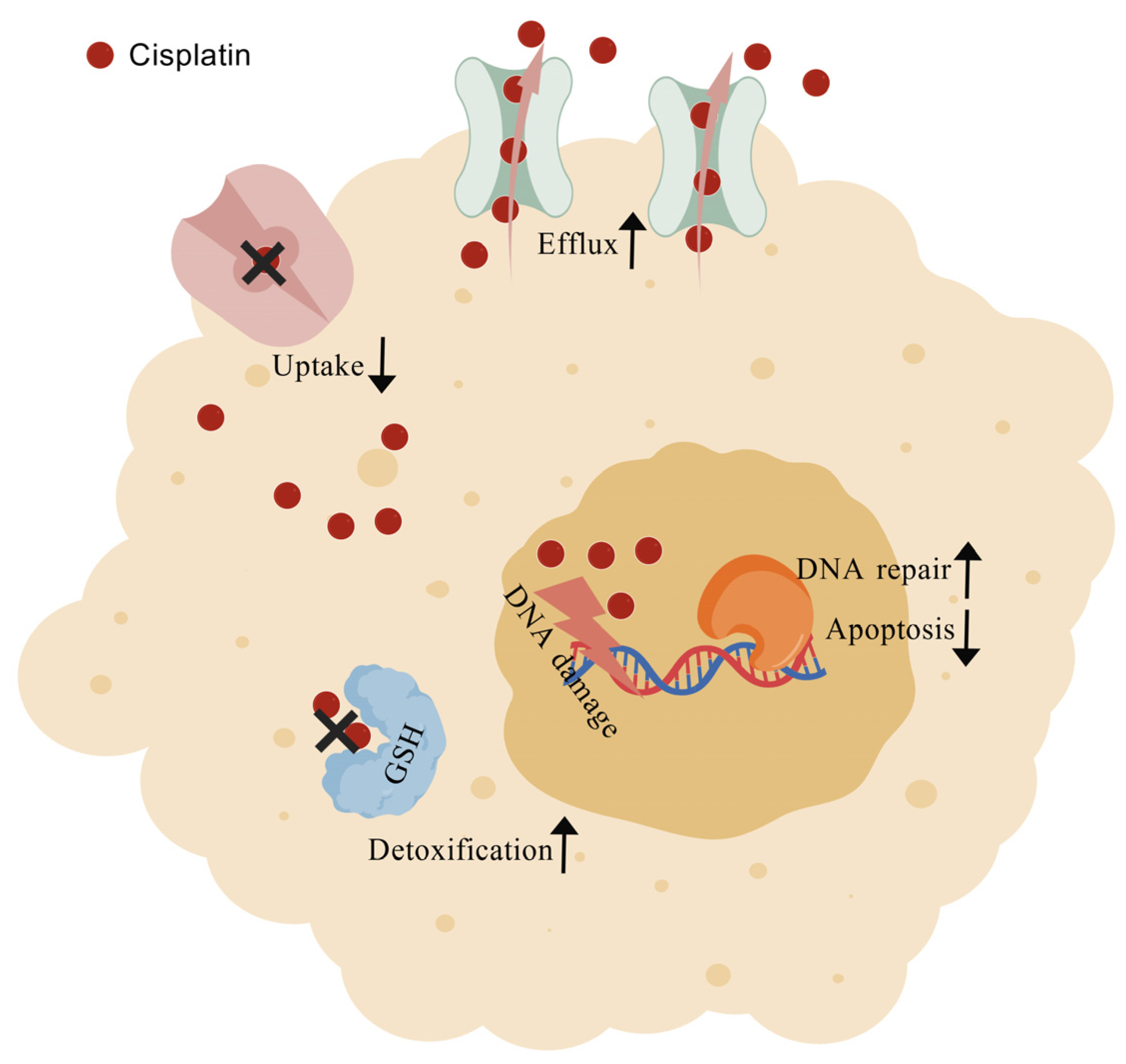
One of the biggest challenges in oncology remains the emergence of resistance to platinum-based chemotherapeutics such as cisplatin. Alongside dose-limiting toxicities, this phenomenon continues to undermine the efficacy of these drugs in the management of solid tumors. Experimental data from in vitro models indicate that resistance to cisplatin can arise through diverse epigenetic and cellular modifications. Mechanisms include diminished intracellular drug accumulation—achieved through active efflux, intracellular sequestration, or impaired uptake—together with detoxification by glutathione conjugates, metallothioneins, and other antioxidant systems (Figure 4). Additional resistance routes involve elevated DNA repair capacity via nucleotide excision and mismatch repair pathways, aberrant DNA methylation, altered membrane trafficking due to cytoskeletal disorganization, overexpression of molecular chaperones, dysregulated expression of microRNAs, and changes in transcription factors or small GTPases [107]. A consequence of reduced cisplatin uptake or retention is a substantial decrease in platinum–DNA adduct formation, which undermines cytotoxic efficiency. For instance, cisplatin-resistant hepatoma 7404-CP20 cells display up to a nine-fold reduction in overall platinum–DNA adducts and a twelve-fold reduction in rRNA gene-specific cross-links, despite removal kinetics comparable to parental 7404 cells [108]. Cisplatin also provokes endoplasmic reticulum stress and apoptotic signals independent of nuclear involvement [109]. Since the drug functions as both a DNA-damaging agent and apoptosis inducer, alterations in heat-shock protein (HSP) expression are strongly implicated in resistance. Elevated HSP60 has been detected in cervical and liver carcinoma [27], while HSP27, HSP70, HSP72, GRP78, and HSP90 upregulation has been documented in resistant ovarian [110,111], cervical [112], colon [113], breast [114], and laryngeal carcinoma models [42].
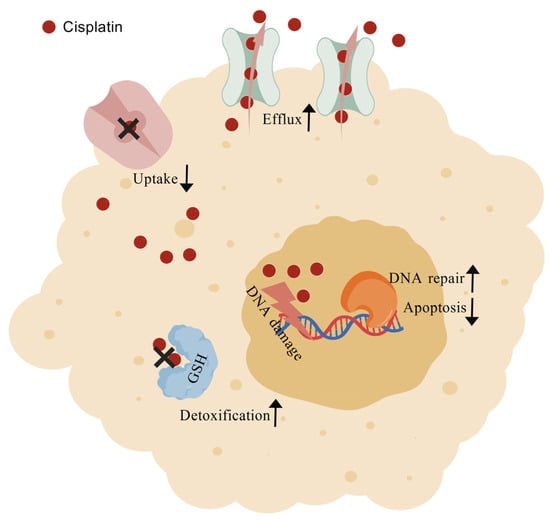
Figure 4.
Graphic representation of cisplatin resistant mechanisms. Created with BioGDP.com [34].
Members of the Rho family of small GTP-binding proteins—RhoA, RhoB, RhoC, and Rac—also contribute to cisplatin resistance. Reduced expression of Rab5, Rac1, and RhoA correlates with impaired accumulation of radiolabeled cisplatin in resistant cells [107]. Specific ribosomal proteins are likewise implicated: L36 has been linked with resistance in carcinoma cells [115], while proteomic analysis in MCF-7 breast cancer cells revealed decreased P0 expression [116], and ectopic expression of L37 can blunt p53-mediated DNA damage responses [117]. Transcriptional regulators also play prominent roles, including Y-box binding protein-1, CCAAT-binding factor 2, activating transcription factor 4, zinc finger protein 143, nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB), microphthalmia-associated transcription factor (MITF), forkhead box O (FoxO), and the mitochondrial transcription factor A (TFAM) [27]. Nuclear factor erythroid 2-related factor 2 (Nrf2) is a key transcription factor that plays a key role antioxidant response and is involved in the onset and progression of several cancerous and non-cancerous diseases [118,119,120] and is emerging as a pivotal determinant: Nrf2-deficient murine embryonic fibroblasts and cisplatin-resistant SK-OV ovarian cancer cells display a clear correlation between Nrf2 levels and drug resistance. Silencing or loss of Nrf2 enhances cisplatin cytotoxicity and apoptosis compared with controls [121]. This effect of Nrf2 in inducing cisplatin resistance and cancer progression has been demonstrated in several malignancies [122,123,124]. Finally, upregulation of GCF2 in KB-3-1 cells reduces RhoA expression, perturbs actin–filamin organization, causes mislocalization of the efflux transporter MRP1, and lowers cisplatin accumulation, collectively producing a three-fold increase in drug resistance [27].
Evidence suggests that alterations in several signaling cascades contribute to the acquisition of cisplatin resistance. These include modifications in protein kinase C isoforms and activation of MAPK or small GTP-binding proteins and such changes may arise directly from drug exposure or through drug-independent adaptive responses. More recently, a mechanism involving the nuclear translocation of the glucocorticoid receptor following cisplatin binding has been described. This event upregulates MAST1, a kinase that promotes platinum resistance by reactivating MEK1 via phosphorylation at Ser221 [125]. The result is inhibition of apoptotic signaling and enhanced cellular proliferation. The clinical relevance of this pathway is underscored by the observation that pharmacological inhibition of MAST1 with agents such as lestaurtinib reinstates apoptosis and decreases tumor growth [126].
At the molecular level, epigenetic reprogramming contributes substantially to resistance phenotypes. Mechanisms include increased detoxification through glutathione, metallothioneins, or antioxidant conjugates, altered trafficking of membrane proteins, rearrangements of cytoskeletal structures, and deregulation of transcription factors and small GTPases (Figure 4). Platinum sequestration, reduced intracellular uptake, and enhanced efflux also belong to this spectrum. Although robust preclinical and clinical data support reduced uptake and enhanced efflux as contributors to resistance, the clinical significance of detoxification-mediated inactivation remains less definitive [27]. To classify these mechanisms systematically, Galluzzi and colleagues proposed their subdivision into pre-target, on-target, post-target, and off-target categories [127]. Pre-target events encompass reduced drug import, increased export, and enhanced conjugation to detoxifying molecules such as glutathione and metallothioneins. On-target mechanisms primarily involve alterations in DNA damage processing. These include elevated nucleotide excision repair activity, defects in mismatch repair, upregulation of homologous recombination, and increased tolerance to DNA adducts through translesion synthesis (Figure 4). In fact, the polymerases POLH, POLI, POLK, REV1, REV3, and REV7 cooperate in translesion synthesis to bypass DNA crosslinks. Experimental data reveal that loss of POLH or REV3 sensitizes tumor cells to cisplatin, whereas REV3 overexpression has the opposite effect, underscoring the therapeutic potential of modulating this pathway [128,129]. In addition, the possibility that cytoplasmic proteins may sequester cisplatin and thereby attenuate its DNA-damaging effects remains underexplored. Post-target resistance is associated with signaling alterations downstream of DNA damage that regulate apoptosis. Normally, cisplatin-induced inter- and intra-strand crosslinks activate apoptotic cascades, but mutations or epigenetic changes in these pathways confer survival advantages. MAPK and p53 signaling are among the most frequently implicated routes. Notably, p53 dysfunction explains the paradoxical observation that tetraploid cells often exhibit greater tolerance to DNA-damaging agents compared with diploid counterparts [130]. Dysregulation of apoptosis-related proteins, such as the upregulation of survivin via a PI3K/AKT1-dependent pathway, has been correlated with diminished cisplatin sensitivity and poor patient survival in multiple malignancies, including ovarian and esophageal cancers [131]. Off-target mechanisms include signaling networks not directly engaged by cisplatin yet capable of compensating for its cytotoxic effects. Key examples are the dual-specificity kinase DYRK1B, which participates in differentiation programs, and ERBB2/HER2, which activates pro-survival pathways through MAPK and PI3K/AKT. Glutathione again emerges as a central player, as it modulates not only pre-target detoxification but also redox balance, thereby suppressing apoptosis and enhancing tolerance to oxidative stress. This places glutathione at the intersection of multiple resistance pathways [127].
Finally, increasing attention has been directed toward the role of STAT3 signaling, a key pathway involved in the onset and progression of several diseases [132,133]. In fact, it has been demonstrated that STAT3 contributes to tumor cell survival, proliferation, angiogenesis, immune evasion, and importantly, drug resistance. Persistent activation of STAT3, for instance via S1PR1 upregulation as observed in gastric cancer, leads to constitutive cisplatin resistance [134]. Inhibition of the S1PR1–STAT3 signaling loop has been shown to restore drug sensitivity, highlighting this axis as a potential therapeutic target to overcome resistance [135].
5. Therapeutic Applications of Cisplatin in Oncology
5.1. Ovarian Cancer
Among gynecologic malignancies, ovarian carcinoma represents one of the most prevalent and lethal forms of disease in women, with mortality rates surpassing those of other cancers affecting the female reproductive tract [136,137]. Although the precise molecular and environmental determinants of ovarian tumorigenesis remain unresolved, familial predisposition—particularly in patients with a history of breast or colorectal cancer—appears to play an important contributory role [123,138]. A major clinical challenge is the absence of reliable screening programs and the nonspecific nature of early manifestations, which frequently leads to late-stage diagnosis. Standard management typically involves surgical resection of the tumor mass, followed by systemic chemotherapy to eradicate residual microscopic disease [139]. Despite dose-limiting toxicities, cisplatin continues to be regarded as a cornerstone chemotherapeutic agent in ovarian cancer therapy. However, one of its major limitations lies in the propensity of tumors to recur and to acquire resistance following initial therapeutic response. To counteract this phenomenon, cisplatin is increasingly administered in combinatorial regimens with bioactive agents such as honey venom, trichostatin A or 5-aza-2′-deoxycytidine, and withaferin A, which aim to enhance antitumor efficacy while reducing the likelihood of resistance [140,141,142]. In addition, many other synthetic and natural molecules are being investigated for their ability to synergistically enhance cisplatin cytotoxicity [143,144,145,146].
5.2. Breast Cancer
Breast carcinoma remains a principal contributor to cancer-related mortality among women on a global scale. In advanced or highly aggressive forms of the disease, chemotherapy constitutes the primary therapeutic intervention, with the objective of prolonging patient survival [147]. Over the years, multiple chemotherapeutic compounds have been introduced in an attempt to overcome the persistent challenge posed by breast cancer [148]. The majority of these pharmacological agents act by indiscriminately targeting populations of rapidly proliferating cells, a mechanism that results in significant collateral tissue damage and accounts for their classification as cytotoxic drugs. Within this group, cisplatin has achieved prominence as a versatile antineoplastic agent, demonstrating clinical utility. Cisplatin exerts its antitumor activity in breast cancer primarily through the formation of covalent DNA adducts that disrupt replication and transcription, producing replication fork stalling and extensive genomic stress that exceed the capacity of tumor cells to repair the damage. This DNA injury serves as the central initiating event for activation of the intrinsic apoptotic pathway, where p53 signaling, induction of BH3-only proteins, and mitochondrial outer-membrane permeabilization converge to drive caspase activation and apoptotic cell death. Tumors with defects in high-fidelity DNA repair, particularly homologous recombination, display heightened susceptibility to cisplatin-induced cytotoxicity; the article highlights, for example, the critical role of BRCA1/2 and RAD51-mediated repair in determining whether cells can tolerate platinum-induced DNA crosslinks or instead undergo apoptosis [149]. Beyond direct genotoxicity, cisplatin affects multiple stress-response pathways that modulate treatment outcome. Activation of the MAPK family members JNK and p38 enhances pro-apoptotic signaling under sustained stress, whereas ERK activation can promote survival, underlying how pathway-specific signaling biases influence cellular fate following platinum exposure. Finally, metabolic state also impact cisplatin efficacy since glycolytic reprogramming and elevated antioxidant capacity can buffer the oxidative stress generated downstream of cisplatin, whereas impaired redox homeostasis sensitizes cancer cells to platinum-induced damage [149,150].
5.3. Lung Cancer
Lung cancer continues to rank among the most prevalent and deadly forms of malignancy worldwide. Small-cell lung carcinoma (SCLC) accounts for approximately 15% of all diagnosed cases, and the current therapeutic backbone for this subtype relies heavily on platinum-based regimens [151]. Among these, cisplatin and carboplatin remain the principal agents utilized in chemotherapy protocols for SCLC [152]. Cisplatin is frequently favored in clinical trials owing to its potent antineoplastic properties; however, its clinical application is often limited by severe toxicities, notably nephrotoxicity [153,154] and gastrointestinal side effects such as nausea and emesis [155]. To mitigate renal complications, cisplatin administration requires rigorous hydration strategies and careful monitoring of urine output. Given these challenges, carboplatin is frequently adopted as an alternative, offering comparable efficacy while circumventing the need for aggressive fluid management.
For patients with localized non-small-cell lung cancer (NSCLC), surgical resection constitutes the standard therapeutic approach, with adjuvant cisplatin-based chemotherapy recommended in stage II and III disease. The Lung Adjuvant Cisplatin Evaluation (LACE) meta-analysis, which pooled data from the five largest randomized trials, demonstrated a 5.3% absolute improvement in 5-year overall survival following cisplatin-based adjuvant therapy, reinforcing the value of this approach [156].
In addition to treatment strategies, tumor biology has highlighted cluster of differentiation 133 (CD133), a membrane glycoprotein associated with stem cell phenotypes, as a putative marker of tumor-initiating cells. Notably, studies have reported that populations expressing both CD133 and epithelial-specific antigen (ESA) are significantly enriched in primary NSCLC tissues compared to normal lung counterparts [157].
5.4. Head and Neck Squamous Cell Carcinoma
Head and neck squamous cell carcinoma (HNSCC) remains a prevalent and aggressive form of cancer, with global incidence figures exceeding 600,000 new diagnoses annually and may be preceded by potentially malignant disorders [158,159]. Despite advancements in therapeutic approaches such as surgical intervention, radiotherapy, and chemotherapeutic regimens, the disease continues to carry a poor prognosis [160,161]. The overall survival rate at five years has remained stagnant at around 50%, underscoring the persistent clinical challenges in its management [162].
While cisplatin is commonly incorporated into treatment strategies, its effectiveness as a monotherapy for HNSCC has been limited [162,163,164]. To enhance therapeutic outcomes, researchers have explored combination therapies involving cisplatin and other agents, such as methotrexate, vinblastine, doxorubicin, and gemcitabine. Comparative clinical studies investigating these combinations—particularly in metastatic urothelial cancer contexts—have been pivotal in evaluating the efficacy of multi-drug protocols over single-agent cisplatin use. In HNSCC, the acquisition of stem-like characteristics has been strongly correlated with the upregulation of markers such as SOX2, Bmi-1, CD44, Nanog, and aldehyde dehydrogenase. The presence of these markers defines a subpopulation of cells that demonstrates heightened tolerance to cisplatin therapy [165]. This phenomenon highlights a critical feature of cancer stem cells (CSCs) in the context of therapeutic resistance, and as a result, extensive research efforts have been directed toward elucidating the functional significance of these stemness-associated markers in cisplatin-resistant HNSCC [166,167,168].
5.5. Glioblastoma Multiforme
Glioblastoma multiforme (GBM) is the most aggressive and frequently diagnosed primary malignant tumor of the central nervous system, characterized by diffuse infiltration, rapid progression, and almost inevitable recurrence [169]. Despite advances in surgical techniques and radiotherapy, prognosis remains dismal, with median overall survival rarely exceeding 15–18 months even under the current standard of care combining surgery, radiotherapy, and temozolomide [170]. Given its broad antitumor efficacy across solid malignancies, cisplatin has been explored as an adjunct or salvage chemotherapeutic agent in GBM, particularly in recurrent disease or pediatric brain tumors. In the context of GBM, cisplatin has been administered both as a single agent and in combination regimens, often with alkylating agents, topoisomerase inhibitors, or novel radiosensitizers, in an attempt to overcome the intrinsic chemoresistance of glioma cells [171]. Nevertheless, systemic dosing is frequently limited by toxicity, necessitating careful hydration protocols and renal function monitoring to minimize nephrotoxic risk.
One of the greatest challenges in applying cisplatin for GBM lies in the intrinsic and acquired resistance mechanisms of the tumor. Glioblastoma cells exhibit upregulated DNA repair pathways, particularly nucleotide excision repair and mismatch repair components, which facilitate the removal of cisplatin-induced DNA adducts. Overexpression of the DNA repair enzyme O6-methylguanine-DNA methyltransferase (MGMT), well known for its role in resistance to temozolomide, has also been implicated in reducing cisplatin cytotoxicity [172]. Additionally, glioma stem-like cells (GSCs) display enhanced drug efflux through ATP-binding cassette (ABC) transporters, elevated antioxidant defenses, and activation of survival signaling cascades such as PI3K/AKT and NF-κB, further diminishing cisplatin sensitivity. These resistance mechanisms collectively explain why clinical responses to cisplatin in GBM are often transient and incomplete [173].
Toxicity remains another major limitation of cisplatin therapy in glioblastoma [174]. In neuro-oncology, adverse events are particularly problematic because patients often require prolonged treatment courses and are already debilitated by neurological deficits. As a result, carboplatin, a less nephrotoxic analog, has occasionally been employed as a substitute, though evidence suggests it may offer reduced potency in GBM.
5.6. Skin Cancers
Cisplatin has been investigated in the treatment of both melanoma and non-melanoma skin cancers (NMSC). In melanoma, cisplatin was among the earliest chemotherapeutic agents to show in vitro cytotoxicity; however, clinical outcomes have been disappointing, as the disease is largely refractory to platinum-based therapy [175,176]. Multiple mechanisms underlie this resistance, including enhanced DNA repair through nucleotide excision repair (NER), upregulation of survival signaling pathways such as PI3K/AKT and MAPK, increased expression of anti-apoptotic proteins, and drug efflux mediated by ATP-binding cassette (ABC) transporters. Clinical regimens incorporating cisplatin have demonstrated transient responses but without significant improvements in overall survival. By contrast, in non-melanoma skin cancers, particularly cutaneous squamous cell carcinoma (cSCC), cisplatin has retained a more prominent role. Cisplatin is frequently administered in combination with 5-fluorouracil (5-FU), and is also utilized as a radiosensitizer in concurrent chemoradiation protocols, especially for advanced head and neck squamous cell carcinoma, which shares biological features with cSCC. Response rates in this context are significantly higher than those observed in melanoma, although resistance and toxicity still limit long-term efficacy [177].
5.7. Gastric Cancer
Gastric cancer, arising from the epithelial lining of the gastric mucosa, represents a major malignancy of the digestive system and it ranks as the fifth most prevalent cancer globally, with an incidence of 5.6%, and is responsible for 7.7% of cancer-related mortality [178]. Cisplatin exerts its cytotoxic activity in gastric cancer primarily through the generation of intra- and interstrand DNA crosslinks that impede replication and transcription, imposing a genotoxic burden that often surpasses the repair capacity of malignant cells [179]. This damage activates the DNA damage response, leading to replication fork collapse, checkpoint activation, and engagement of intrinsic apoptotic pathways when repair mechanisms are overwhelmed. Mitochondrial dysfunction and oxidative stress further amplify this response, reinforcing the apoptotic cascade [179]. Effective cisplatin activity therefore depends on the integrity of pro-apoptotic machinery, including mitochondrial permeabilization, cytochrome c release, and caspase activation. Gastric tumors exhibiting intact apoptotic signaling are more susceptible to cisplatin, whereas those with upregulated anti-apoptotic factors or survival pathways such as ERK, NF-κB, or PI3K/Akt, can diminish apoptotic output and reduce drug sensitivity [180,181,182,183,184,185].
In advanced gastric cancer, the integration of chemotherapy with targeted therapeutic approaches has been shown to extend overall survival and enhance patient quality of life. Among the chemotherapeutic agents employed, cisplatin has long served as a cornerstone in first-line regimens [186]. Nonetheless, the therapeutic benefits of cisplatin are frequently undermined by the emergence of drug resistance and the substantial toxicity profile, both of which remain critical barriers to improving long-term outcomes in gastric cancer management [187,188].
5.8. Testicular Cancer
Cisplatin itself was formally approved by the FDA in 1978 for use against testicular malignancies [189]. Testicular cancers are classified into seminomas and non-seminomas, both of which primarily affect young men and, despite their shared germ cell origin, these entities differ markedly in differentiation status, biological behavior, and therapeutic responsiveness. Seminomas can arise across a wide range of ages, but they usually progress and metastasize more gradually compared to non-seminomas. At the molecular level, seminomas are characterized by limited DNA damage repair capacity and a pronounced propensity to undergo apoptosis following genotoxic stress. Standard management of seminomas relies on cisplatin-based chemotherapy, with approximately 85% of patients with advanced disease achieving remission after three to four treatment cycles [190]. The exceptional sensitivity of seminomas to cisplatin has been attributed to inefficient nucleotide excision repair and an intact p53-mediated apoptotic response, which together amplify the cytotoxic impact of platinum-induced DNA crosslinks [191].
Non-seminomas are more frequently diagnosed in men from their late teens to early thirties and consist of four main histological variants: embryonal carcinoma, yolk sac tumor, choriocarcinoma, and teratoma. These components often coexist within the same tumor, contributing to biological heterogeneity and variable treatment responses. Among these, teratomas are most effectively treated with combination therapy incorporating bleomycin, etoposide, and cisplatin, producing cure rates above 90% [192]. While most non-seminomatous elements are highly chemosensitive, teratomatous components display intrinsic resistance to cisplatin and frequently require surgical resection to achieve durable disease control [191]. Overall, the extraordinary clinical success of cisplatin in testicular cancer reflects a convergence of biological vulnerabilities, including impaired DNA repair, low tolerance for replication stress, and robust activation of apoptotic pathways, which collectively distinguish germ cell tumors from most other solid malignancies.
5.9. Bladder Cancer
Bladder cancer represents the tenth most prevalent malignancy worldwide, with an estimated global incidence of 573,000 new diagnoses and approximately 213,000 disease-related deaths annually [178,193]. Since their introduction in the late 1980s, cisplatin-based combination regimens have constituted the standard adjuvant therapeutic approach for patients with metastatic bladder cancer, and cisplatin continues to serve as a fundamental component of systemic treatment [194]. Despite this, clinical efficacy remains restricted, with objective responses observed in only about 35% of metastatic cases. Furthermore, even among initially responsive patients, the eventual acquisition of cisplatin resistance is almost inevitable [194,195]. This phenomenon of drug resistance is recognized as a principal determinant underlying the poor overall prognosis associated with advanced bladder cancer.
6. Cisplatin-Induced Organ Toxicity
A major limitation of cisplatin therapy is the broad spectrum of toxicities it induces. These effects arise directly from the covalent interaction of cisplatin (and carboplatin) with DNA purine residues, as well as indirectly from drug-triggered increases in oxidative stress once endogenous scavenging defenses become engaged in adduct formation. Although virtually any organ can be susceptible to injury given the generalized mode of action, the liver, kidney, heart, auditory system, and peripheral nerves are among the most consistently affected. Despite considerable progress in delineating the cellular and molecular mechanisms by which cisplatin, carboplatin, and oxaliplatin exert their toxic effects, predicting the specific organs at risk in a given patient remains challenging. The severity and pattern of organ involvement are highly variable, and few clinical trials have addressed these questions due to inherent difficulties such as heterogeneous patient populations, differences in treatment regimens, and limitations in control groups or blinding strategies. These same challenges extend to evaluating adjunctive therapies intended to mitigate toxicity. Magnesium supplementation has shown some promise in reducing the incidence of acute kidney injury and oliguria, but more comprehensive evidence is needed to establish its protective efficacy [196].
Hepatic injury is frequently associated with high-dose cisplatin [197]. Mechanistically, hepatotoxicity is linked to oxidative stress characterized by depletion of glutathione and the production of reactive aldehydes, including malondialdehyde, generated via CYP2E1-mediated lipid peroxidation [198]. In such cases, serum enzymes—transaminases and alkaline phosphatase—remain the most sensitive markers of parenchymal and biliary damage, whereas conjugated and unconjugated bilirubin levels provide functional assessment of hepatic impairment [199].
Cardiotoxicity is another clinically relevant concern. Reliable indicators of myocardial damage include elevated cytosolic lactate dehydrogenase and creatine kinase, with increases in serum troponin C signaling irreversible cardiomyocyte necrosis [200].
The kidney represents the primary route of cisplatin elimination and, consequently, a major target of injury. Renal accumulation of cisplatin can reach concentrations several times higher than plasma levels, particularly within proximal tubular epithelial cells where uptake is mediated by copper transporter CTR1 and organic cation transporter OCT2 [29,201]. The clinical manifestation is acute kidney injury, frequently associated with oliguria. Hydration therapy remains the cornerstone of prevention. Both short-term and low-volume hydration protocols have been shown to be effective and safe, even in patients treated with intermediate or high doses [202]. Adjunct magnesium administration in such regimens can further reduce nephrotoxicity, although its benefit appears limited at very high cisplatin doses. Mannitol, by inducing osmotic diuresis, is often employed in high-dose regimens, while furosemide use is more controversial due to risks of electrolyte imbalance and secondary hypomagnesemia, which may paradoxically worsen renal injury [203]. Additional support for the protective role of magnesium comes from studies showing that supplementation with 8–16 mEq in short-term, low-volume hydration protocols significantly reduces nephrotoxicity at intermediate cisplatin doses [202]. Alternative agents have been tested with mixed outcomes: glutathione provided a degree of protection comparable to magnesium, whereas dopamine was ineffective. Interestingly, magnesium supplementation seems less effective in patients receiving high-dose cisplatin, where mannitol has demonstrated greater benefit. As an osmotic diuretic, mannitol enhances urine output and thereby reduces tubular cisplatin accumulation; however, it is only recommended in high-dose regimens (≈100 mg/m2), since its strong diuretic effect can otherwise lead to dehydration [202]. Replacement of mannitol with furosemide is not well supported by clinical evidence, and carries additional risks [204,205]. By blocking the Na+/K+/2Cl− cotransporter in the loop of Henle, furosemide may precipitate hypokalemia and secondary hypomagnesemia. This imbalance can further stimulate OCT2 activity, enhancing cisplatin uptake into proximal tubular epithelial cells and paradoxically exacerbating nephrotoxicity [206].
Cisplatin-induced neuropathy manifests primarily as a peripheral sensory neuropathy, with a symmetrical distribution in the extremities that can progress to the knees and elbows [207]. Symptoms include paresthesia, pain, decreased vibratory sensation, mechanical allodynia, and gait disturbances. These deficits can persist or even worsen after therapy cessation, significantly impairing quality of life and sometimes necessitating discontinuation of treatment [208]. In severe cases, degeneration of large myelinated fibers in the dorsal columns may occur, giving rise to Lhermitte’s sign [209]. Effective treatments are lacking: antioxidants such as vitamin E and α-lipoic acid, beneficial in diabetic neuropathy, have not proven successful in this context [210].
Ototoxicity represents another major complication, characterized by bilateral, progressive, high-frequency hearing loss, often accompanied by tinnitus and ear pain [211]. Cisplatin accumulation in cochlear cells is facilitated by CTR1 and OCT2, the same transporters involved in nephrotoxicity. The injury is mediated by oxidative stress and depletion of antioxidant defenses. Pediatric patients appear especially vulnerable, though sodium thiosulfate has shown some protective effect when carefully timed to avoid inactivation of cisplatin’s antitumor activity [212]. Amifostine, another thiol compound, has produced inconsistent results, offering reliable protection against nephrotoxicity, but only partial or inconsistent benefit in reducing ototoxicity or neuropathy [210].
While peripheral neuropathy is the predominant neurotoxic effect, central nervous system complications such as posterior reversible encephalopathy syndrome (PRES) have also been described, particularly in patients receiving cisplatin for brain malignancies. These cases suggest that cisplatin may cross or disrupt the blood–brain barrier, possibly facilitated by pro-inflammatory cytokines and oxidative stress [210]. Symptoms typically reverse after cessation of therapy, though further studies are required to clarify mechanisms.
Beyond the major target organs, other tissues—including the gastrointestinal tract, bone marrow, and reproductive system—are also subject to cisplatin-induced injury, though to a lesser extent. Together, these toxicities underscore the delicate balance between therapeutic efficacy and systemic harm that defines cisplatin-based chemotherapy.
7. Conclusions and Perspectives
Cisplatin is a powerful chemotherapeutic agent frequently employed in managing a variety of solid malignancies. It plays a crucial role in treating cancers such as those of the lung, breast, brain, ovary, kidney, testis, and the hematopoietic system. Its primary mechanism involves interfering with cellular DNA, leading to disrupted replication and cell division, ultimately resulting in programmed cell death. Beyond its direct genotoxic effects, cisplatin also triggers oxidative damage through reactive oxygen species generation, promotes cell cycle arrest via p53 activation, suppresses oncogene expression, and activates multiple apoptotic pathways. Despite its clinical success, the use of cisplatin often leads to harmful effects on vital organs, including the liver, kidneys, heart, nervous system, and blood cells. Furthermore, tumor recurrence and resistance to cisplatin remain significant hurdles. To mitigate these challenges, combination regimens incorporating cisplatin and other agents have gained traction, aiming to improve efficacy and minimize toxicity. Emerging evidence supports multi-targeted approaches that not only enhance drug responsiveness but also combat inflammation and limit cellular uptake of cisplatin, paving the way for more effective and safer cancer treatments. In addition to its well-established cytotoxic effects, accumulating evidence suggests that cisplatin can modulate antitumor immunity by promoting forms of immunogenic cell death characterized by the release of danger-associated molecular patterns, enhanced antigen presentation, and activation of dendritic cells [213]. These immunomodulatory effects may contribute to treatment efficacy in selected contexts and provide a biological rationale for combining cisplatin with immune checkpoint inhibitors or other immunotherapies. Looking ahead, major challenges include reducing cisplatin-associated organ toxicity, preventing or reversing drug resistance, and identifying biomarkers that enable patient stratification and personalized dosing strategies. Parallel efforts aimed at developing next-generation platinum compounds with improved selectivity, as well as rational combination regimens that exploit vulnerabilities in DNA repair, redox balance, and immune signaling pathways, are likely to shape the future clinical use of platinum-based chemotherapy.
Author Contributions
Conceptualization, R.C.; methodology, M.C., V.P. (Valentina Pozzi) and V.S.; software, A.M.; resources, R.C.; writing—original draft preparation, R.C., M.C., V.P. (Valentina Pozzi), V.P. (Veronica Pompei), A.P., A.R. and S.F.; writing—review and editing, R.C., D.S. and G.T.; visualization, V.S. and A.P.; supervision, G.T. and R.C. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
No new data were created or analyzed in this study.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| ABC | ATP-binding cassette transporters |
| AKT/PKB | Protein kinase B |
| APAF-1 | Apoptotic protease-activating factor 1 |
| ATP7A/ATP7B | Copper-transporting ATPases |
| AQP | Aquaporin |
| ATM | Ataxia Telangiectasia Mutated |
| ATR | ATM and Rad3-related |
| BAD | Bcl-2-associated agonist of cell death |
| Bax | Bcl-2-associated X protein |
| Bcl-2/Bcl-xL | B-cell lymphoma proteins (anti-apoptotic family members) |
| CAT | Catalase |
| CD133 | Cluster of Differentiation 133 |
| CP-r | Cisplatin-resistant |
| CSCs | cancer stem cells |
| CTR1 | Copper transporter 1 |
| Cyt c | Cytochrome c |
| DISC | Death-inducing signaling complex |
| EGFR | Epidermal growth factor receptor |
| ERK | Extracellular signal-regulated kinase |
| ESA | Epithelial-specific antigen |
| FADD | Fas-associated death domain protein |
| FoxO | Forkhead box O |
| Fas/FasL | Fas receptor/Fas ligand |
| GADD45 | Growth arrest and DNA damage-inducible 45 |
| GSDM | gasdermin |
| GLUT1 | Glucose transporter 1 |
| GBM | Glioblastoma multiforme |
| GRP78 | Glucose-regulated protein 78 |
| GSCs | glioma stem-like cells |
| GSH | Glutathione |
| HER2/ERBB2 | Human epidermal growth factor receptor 2 |
| HMG1 | High Mobility Group Box 1 protein |
| HSP | Heat shock proteins (HSP27, HSP60, HSP70, HSP72, HSP90) |
| JNK | c-Jun N-terminal kinase |
| MAPK | Mitogen-activated protein kinase |
| MAST1 | Microtubule-associated serine/threonine kinase 1 |
| Mdm2 | Mouse double minute 2 homolog |
| MGMT | O6-methylguanine-DNA methyltransferase |
| MITF | Microphthalmia-associated transcription factor |
| MMR | Mismatch Repair |
| MPT | Mitochondrial permeability transition |
| MDR1 | Multidrug Resistance protein 1/P-glycoprotein |
| MRP | Multidrug resistance associated proteins |
| mtDNA | Mitochondrial DNA |
| NADH | Nicotinamide adenine dinucleotide (reduced form) |
| NER | Nucleotide excision repair |
| NF-κB | Nuclear factor kappa-light-chain-enhancer of activated B cells |
| NMSC | Non-melanoma skin cancer |
| NOXA | Pro-apoptotic Bcl-2 family protein |
| Nrf2 | Nuclear factor erythroid 2–related factor 2 |
| NSCLC | Non-small-cell lung cancer |
| OAT | Organic anion transporter |
| OCT | Organic cation transporter |
| PARP | Poly(ADP-ribose) polymerase |
| PI3K | Phosphoinositide 3-kinase |
| PIDD | p53-induced protein with death domain |
| PKC | Protein kinase C |
| POLH/POLI/POLK/REV1/REV3/REV7 | Translesion DNA polymerases |
| PTEN | Phosphatase and tensin homolog |
| PUMA | p53 upregulated modulator of apoptosis |
| ROS | Reactive oxygen species |
| SLC | Solute carrier transporters (SLC22, SLC31 families) |
| SOD | Superoxide dismutase |
| S1PR1 | Sphingosine-1-phosphate receptor 1 |
| STAT3 | Signal transducer and activator of transcription 3 |
| TFAM | Mitochondrial transcription factor A |
| TMEM205 | Transmembrane protein 205 |
| TNF-α | Tumor necrosis factor alpha |
| 5-FU | 5-fluorouracil |
References
- Cohen, S.M.; Lippard, S.J. Cisplatin: From DNA damage to cancer chemotherapy. Prog. Nucleic Acid Res. Mol. Biol. 2001, 67, 93–130. [Google Scholar] [CrossRef]
- Rosenberg, B.; Vancamp, L.; Krigas, T. Inhibition of Cell Division in Escherichia Coli by Electrolysis Products from a Platinum Electrode. Nature 1965, 205, 698–699. [Google Scholar] [CrossRef]
- Rosenberg, B.; Van Camp, L.; Grimley, E.B.; Thomson, A.J. The inhibition of growth or cell division in Escherichia coli by different ionic species of platinum(IV) complexes. J. Biol. Chem. 1967, 242, 1347–1352. [Google Scholar] [CrossRef]
- Rosenberg, B.; VanCamp, L.; Trosko, J.E.; Mansour, V.H. Platinum compounds: A new class of potent antitumour agents. Nature 1969, 222, 385–386. [Google Scholar] [CrossRef]
- Ghosh, S. Cisplatin: The first metal based anticancer drug. Bioorg. Chem. 2019, 88, 102925. [Google Scholar] [CrossRef] [PubMed]
- Makovec, T. Cisplatin and beyond: Molecular mechanisms of action and drug resistance development in cancer chemotherapy. Radiol. Oncol. 2019, 53, 148–158. [Google Scholar] [CrossRef] [PubMed]
- Ferraro, G.; Massai, L.; Messori, L.; Merlino, A. Cisplatin binding to human serum albumin: A structural study. Chem. Commun. 2015, 51, 9436–9439. [Google Scholar] [CrossRef]
- Kato, R.; Sato, T.; Iwamoto, A.; Yamazaki, T.; Nakashiro, S.; Yoshikai, S.; Fujimoto, A.; Imano, H.; Ijiri, Y.; Mino, Y.; et al. Interaction of platinum agents, cisplatin, carboplatin and oxaliplatin against albumin in vivo rats and in vitro study using inductively coupled plasma-mass spectrometory. Biopharm. Drug Dispos. 2019, 40, 242–249. [Google Scholar] [CrossRef]
- Yin, W.J.; Huang, Y.J.; Zhu, Q.; Lin, X.Q.; Piao, H.L.; Yu, Q.Q.; Lai, C.H.; Zhou, G.L.; Zhou, L.Y.; Liu, K.; et al. Hypoalbuminemia and cisplatin-induced acute kidney injury. Front. Pharmacol. 2024, 15, 1510477. [Google Scholar] [CrossRef] [PubMed]
- Gouyette, A.; Apchin, A.; Foka, M.; Richard, J.M. Pharmacokinetics of intra-arterial and intravenous cisplatin in head and neck cancer patients. Eur. J. Cancer Clin. Oncol. 1986, 22, 257–263. [Google Scholar] [CrossRef]
- Hall, M.D.; Okabe, M.; Shen, D.W.; Liang, X.J.; Gottesman, M.M. The role of cellular accumulation in determining sensitivity to platinum-based chemotherapy. Annu. Rev. Pharmacol. Toxicol. 2008, 48, 495–535. [Google Scholar] [CrossRef]
- Eljack, N.D.; Ma, H.Y.; Drucker, J.; Shen, C.; Hambley, T.W.; New, E.J.; Friedrich, T.; Clarke, R.J. Mechanisms of cell uptake and toxicity of the anticancer drug cisplatin. Metallomics 2014, 6, 2126–2133. [Google Scholar] [CrossRef]
- Arnesano, F.; Scintilla, S.; Natile, G. Interaction between platinum complexes and a methionine motif found in copper transport proteins. Angew. Chem. Int. Ed. Engl. 2007, 46, 9062–9064. [Google Scholar] [CrossRef]
- Yonezawa, A.; Masuda, S.; Yokoo, S.; Katsura, T.; Inui, K. Cisplatin and oxaliplatin, but not carboplatin and nedaplatin, are substrates for human organic cation transporters (SLC22A1-3 and multidrug and toxin extrusion family). J. Pharmacol. Exp. Ther. 2006, 319, 879–886. [Google Scholar] [CrossRef] [PubMed]
- Nieskens, T.T.G.; Peters, J.G.P.; Dabaghie, D.; Korte, D.; Jansen, K.; Van Asbeck, A.H.; Tavraz, N.N.; Friedrich, T.; Russel, F.G.M.; Masereeuw, R.; et al. Expression of Organic Anion Transporter 1 or 3 in Human Kidney Proximal Tubule Cells Reduces Cisplatin Sensitivity. Drug Metab. Dispos. 2018, 46, 592–599. [Google Scholar] [CrossRef] [PubMed]
- Desai, V.; Kaler, S.G. Role of copper in human neurological disorders. Am. J. Clin. Nutr. 2008, 88, 855S–858S. [Google Scholar] [CrossRef]
- Strojan, P.; Vermorken, J.B.; Beitler, J.J.; Saba, N.F.; Haigentz, M., Jr.; Bossi, P.; Worden, F.P.; Langendijk, J.A.; Eisbruch, A.; Mendenhall, W.M.; et al. Cumulative cisplatin dose in concurrent chemoradiotherapy for head and neck cancer: A systematic review. Head Neck 2016, 38, E2151–E2158. [Google Scholar] [CrossRef]
- Szturz, P.; Cristina, V.; Herrera Gomez, R.G.; Bourhis, J.; Simon, C.; Vermorken, J.B. Cisplatin Eligibility Issues and Alternative Regimens in Locoregionally Advanced Head and Neck Cancer: Recommendations for Clinical Practice. Front. Oncol. 2019, 9, 464. [Google Scholar] [CrossRef]
- Al-Mamgani, A.; de Ridder, M.; Navran, A.; Klop, W.M.; de Boer, J.P.; Tesselaar, M.E. The impact of cumulative dose of cisplatin on outcome of patients with head and neck squamous cell carcinoma. Eur. Arch. Otorhinolaryngol. 2017, 274, 3757–3765. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.S.; Tang, L.Q.; Zhang, L.; Chen, Q.Y.; Liu, L.T.; Guo, L.; Mo, H.Y.; Luo, D.H.; Huang, P.Y.; Xiang, Y.Q.; et al. The impact of the cumulative dose of cisplatin during concurrent chemoradiotherapy on the clinical outcomes of patients with advanced-stage nasopharyngeal carcinoma in an era of intensity-modulated radiotherapy. BMC Cancer 2015, 15, 977. [Google Scholar] [CrossRef]
- Bossi, P.; Kiyota, N.; Mesia, R.; Rosenberg, A.J.; Martino, R.; Fazer-Posorske, C.; Salmio, S.; Schroeder, A.; Yom, S.S. Adverse event profiles and management of cisplatin-based chemoradiotherapy in locally advanced head and neck squamous cell carcinoma: A systematic review. Oral. Oncol. 2025, 169, 107596. [Google Scholar] [CrossRef]
- Chovanec, M.; Abu Zaid, M.; Hanna, N.; El-Kouri, N.; Einhorn, L.H.; Albany, C. Long-term toxicity of cisplatin in germ-cell tumor survivors. Ann. Oncol. 2017, 28, 2670–2679. [Google Scholar] [CrossRef] [PubMed]
- Peng, H.; Chen, L.; Li, W.F.; Guo, R.; Mao, Y.P.; Zhang, Y.; Zhang, F.; Liu, L.Z.; Tian, L.; Lin, A.H.; et al. The Cumulative Cisplatin Dose Affects the Long-Term Survival Outcomes of Patients with Nasopharyngeal Carcinoma Receiving Concurrent Chemoradiotherapy. Sci. Rep. 2016, 6, 24332. [Google Scholar] [CrossRef] [PubMed]
- Ngamphaiboon, N.; Dechaphunkul, A.; Setakornnukul, J.; Dechaphunkul, T.; Jiratrachu, R.; Suktitipat, B.; Jiarpinitnun, C.; Pattaranutaporn, P.; Danchaivijitr, P. Optimal cumulative dose of cisplatin for concurrent chemoradiotherapy among patients with non-metastatic nasopharyngeal carcinoma: A multicenter analysis in Thailand. BMC Cancer 2020, 20, 518. [Google Scholar] [CrossRef]
- Paken, J.; Govender, C.D.; Pillay, M.; Feyasa, M.; Sewram, V. Cisplatin-associated ototoxicity amongst cervical cancer patients: A prospective cohort study in south Africa. PLoS ONE 2023, 18, e0283639. [Google Scholar] [CrossRef] [PubMed]
- Clemens, E.; de Vries, A.C.; Pluijm, S.F.; Am Zehnhoff-Dinnesen, A.; Tissing, W.J.; Loonen, J.J.; van Dulmen-den Broeder, E.; Bresters, D.; Versluys, B.; Kremer, L.C.; et al. Determinants of ototoxicity in 451 platinum-treated Dutch survivors of childhood cancer: A DCOG late-effects study. Eur. J. Cancer 2016, 69, 77–85. [Google Scholar] [CrossRef]
- Shen, D.W.; Pouliot, L.M.; Hall, M.D.; Gottesman, M.M. Cisplatin resistance: A cellular self-defense mechanism resulting from multiple epigenetic and genetic changes. Pharmacol. Rev. 2012, 64, 706–721. [Google Scholar] [CrossRef]
- Abdolmaleki, S.; Khaksar, S.; Aliabadi, A.; Panjehpour, A.; Motieiyan, E.; Marabello, D.; Faraji, M.H.; Beihaghi, M. Cytotoxicity and mechanism of action of metal complexes: An overview. Toxicology 2023, 492, 153516. [Google Scholar] [CrossRef]
- Ishida, S.; Lee, J.; Thiele, D.J.; Herskowitz, I. Uptake of the anticancer drug cisplatin mediated by the copper transporter Ctr1 in yeast and mammals. Proc. Natl. Acad. Sci. USA 2002, 99, 14298–14302. [Google Scholar] [CrossRef]
- Holzer, A.K.; Manorek, G.H.; Howell, S.B. Contribution of the major copper influx transporter CTR1 to the cellular accumulation of cisplatin, carboplatin, and oxaliplatin. Mol. Pharmacol. 2006, 70, 1390–1394. [Google Scholar] [CrossRef]
- Lin, X.; Okuda, T.; Holzer, A.; Howell, S.B. The copper transporter CTR1 regulates cisplatin uptake in Saccharomyces cerevisiae. Mol. Pharmacol. 2002, 62, 1154–1159. [Google Scholar] [CrossRef]
- Wang, X.; Lou, Q.; Fan, T.; Zhang, Q.; Yang, X.; Liu, H.; Fan, R. Copper transporter Ctr1 contributes to enhancement of the sensitivity of cisplatin in esophageal squamous cell carcinoma. Transl. Oncol. 2023, 29, 101626. [Google Scholar] [CrossRef]
- Schiavoni, V.; Emanuelli, M.; Campagna, R.; Cecati, M.; Sartini, D.; Milanese, G.; Galosi, A.B.; Pozzi, V.; Salvolini, E. Paraoxonase-2 shRNA-mediated gene silencing suppresses proliferation and migration, while promotes chemosensitivity in clear cell renal cell carcinoma cell lines. J. Cell. Biochem. 2024, 125, e30572. [Google Scholar] [CrossRef]
- Jiang, S.; Li, H.; Zhang, L.; Mu, W.; Zhang, Y.; Chen, T.; Wu, J.; Tang, H.; Zheng, S.; Liu, Y.; et al. Generic Diagramming Platform (GDP): A comprehensive database of high-quality biomedical graphics. Nucleic Acids Res. 2025, 53, D1670–D1676. [Google Scholar] [CrossRef] [PubMed]
- Dasari, S.; Tchounwou, P.B. Cisplatin in cancer therapy: Molecular mechanisms of action. Eur. J. Pharmacol. 2014, 740, 364–378. [Google Scholar] [CrossRef] [PubMed]
- Lehmann, D.M.; Williams, W.C. Physiological responses to cisplatin using a mouse hypersensitivity model. Inhal. Toxicol. 2020, 32, 68–78. [Google Scholar] [CrossRef] [PubMed]
- Fraval, H.N.; Rawlings, C.J.; Roberts, J.J. Increased sensitivity of UV-repair-deficient human cells to DNA bound platinum products which unlike thymine dimers are not recognized by an endonuclease extracted from Micrococcus luteus. Mutat. Res. 1978, 51, 121–132. [Google Scholar] [CrossRef]
- Ferreira, M.J.; Rodrigues, T.A.; Pedrosa, A.G.; Silva, A.R.; Vilarinho, B.G.; Francisco, T.; Azevedo, J.E. Glutathione and peroxisome redox homeostasis. Redox Biol. 2023, 67, 102917. [Google Scholar] [CrossRef]
- Cooke, M.S.; Evans, M.D.; Dizdaroglu, M.; Lunec, J. Oxidative DNA damage: Mechanisms, mutation, and disease. FASEB J. 2003, 17, 1195–1214. [Google Scholar] [CrossRef]
- Campagna, R.; Serritelli, E.N.; Salvolini, E.; Schiavoni, V.; Cecati, M.; Sartini, D.; Pozzi, V.; Emanuelli, M. Contribution of the Paraoxonase-2 Enzyme to Cancer Cell Metabolism and Phenotypes. Biomolecules 2024, 14, 208. [Google Scholar] [CrossRef]
- Saad, S.Y.; Najjar, T.A.; Alashari, M. Role of non-selective adenosine receptor blockade and phosphodiesterase inhibition in cisplatin-induced nephrogonadal toxicity in rats. Clin. Exp. Pharmacol. Physiol. 2004, 31, 862–867. [Google Scholar] [CrossRef] [PubMed]
- Brozovic, A.; Ambriovic-Ristov, A.; Osmak, M. The relationship between cisplatin-induced reactive oxygen species, glutathione, and BCL-2 and resistance to cisplatin. Crit. Rev. Toxicol. 2010, 40, 347–359. [Google Scholar] [CrossRef]
- Desoize, B. Cancer and metals and metal compounds: Part I--carcinogenesis. Crit. Rev. Oncol. Hematol. 2002, 42, 1–3. [Google Scholar] [CrossRef]
- Ozben, T. Oxidative stress and apoptosis: Impact on cancer therapy. J. Pharm. Sci. 2007, 96, 2181–2196. [Google Scholar] [CrossRef]
- Ikner, A.; Ashkenazi, A. TWEAK induces apoptosis through a death-signaling complex comprising receptor-interacting protein 1 (RIP1), Fas-associated death domain (FADD), and caspase-8. J. Biol. Chem. 2011, 286, 21546–21554. [Google Scholar] [CrossRef]
- Zhang, M.; Lee, S.J.; An, C.; Xu, J.F.; Joshi, B.; Nabi, I.R.; Choi, A.M.; Jin, Y. Caveolin-1 mediates Fas-BID signaling in hyperoxia-induced apoptosis. Free Radic. Biol. Med. 2011, 50, 1252–1262. [Google Scholar] [CrossRef]
- Chen, Y.; Li, N.; Yang, J.; Li, K.; Tang, M.; Zhao, X.; Guo, W.; Tong, A.; Nie, C.; Peng, Y.; et al. PUMA overexpression dissociates thioredoxin from ASK1 to activate the JNK/BCL-2/BCL-XL pathway augmenting apoptosis in ovarian cancer. Biochim. Biophys. Acta Mol. Basis. Dis. 2022, 1868, 166553. [Google Scholar] [CrossRef] [PubMed]
- Franklin, J.L. Redox regulation of the intrinsic pathway in neuronal apoptosis. Antioxid. Redox Signal. 2011, 14, 1437–1448. [Google Scholar] [CrossRef]
- Pelicano, H.; Carney, D.; Huang, P. ROS stress in cancer cells and therapeutic implications. Drug Resist. Updat. 2004, 7, 97–110. [Google Scholar] [CrossRef] [PubMed]
- Higuchi, Y. Chromosomal DNA fragmentation in apoptosis and necrosis induced by oxidative stress. Biochem. Pharmacol. 2003, 66, 1527–1535. [Google Scholar] [CrossRef]
- Li, L.; Tan, J.; Miao, Y.; Lei, P.; Zhang, Q. ROS and Autophagy: Interactions and Molecular Regulatory Mechanisms. Cell. Mol. Neurobiol. 2015, 35, 615–621. [Google Scholar] [CrossRef]
- Dai, D.; Chen, C.; Lu, C.; Guo, Y.; Li, Q.; Sun, C. Apoptosis, autophagy, ferroptosis, and pyroptosis in cisplatin-induced ototoxicity and protective agents. Front. Pharmacol. 2024, 15, 1430469. [Google Scholar] [CrossRef]
- de Vasconcelos, N.M.; Van Opdenbosch, N.; Van Gorp, H.; Parthoens, E.; Lamkanfi, M. Single-cell analysis of pyroptosis dynamics reveals conserved GSDMD-mediated subcellular events that precede plasma membrane rupture. Cell Death Differ. 2019, 26, 146–161. [Google Scholar] [CrossRef]
- Sun, L.; Ma, W.; Gao, W.; Xing, Y.; Chen, L.; Xia, Z.; Zhang, Z.; Dai, Z. Propofol directly induces caspase-1-dependent macrophage pyroptosis through the NLRP3-ASC inflammasome. Cell Death Dis. 2019, 10, 542. [Google Scholar] [CrossRef] [PubMed]
- Yu, W.; Zong, S.; Zhou, P.; Wei, J.; Wang, E.; Ming, R.; Xiao, H. Cochlear Marginal Cell Pyroptosis Is Induced by Cisplatin via NLRP3 Inflammasome Activation. Front. Immunol. 2022, 13, 823439. [Google Scholar] [CrossRef]
- Yu, R.; Wang, K.; Luo, W.; Jiang, H. Knockdown and mutation of Pou4f3 gene mutation promotes pyroptosis of cochleae in cisplatin-induced deafness mice by NLRP3/caspase-3/GSDME pathway. Toxicology 2022, 482, 153368. [Google Scholar] [CrossRef]
- Li, R.Y.; Zheng, Z.Y.; Li, Z.M.; Heng, J.H.; Zheng, Y.Q.; Deng, D.X.; Xu, X.E.; Liao, L.D.; Lin, W.; Xu, H.Y.; et al. Cisplatin-induced pyroptosis is mediated via the CAPN1/CAPN2-BAK/BAX-caspase-9-caspase-3-GSDME axis in esophageal cancer. Chem. Biol. Interact. 2022, 361, 109967. [Google Scholar] [CrossRef]
- Zhang, F.; Zhu, T.; Gan, L.; Du, Y.; Liu, L.; Ding, H.; Chen, X.; Guan, Y. FTO triggers NLRP3/GSDMD-dependent pyroptosis to enhance cisplatin-sensitivity in ovarian cancer. Cell Signal. 2025, 131, 111698. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Z.; Li, Z.; Gu, L.; Li, L.; Gao, Q.; Zhang, X.; Fu, J.; Guo, Y.; Li, Q.; Shen, X.; et al. Ophiopogonin B alleviates cisplatin resistance of lung cancer cells by inducing Caspase-1/GSDMD dependent pyroptosis. J. Cancer 2022, 13, 715–727. [Google Scholar] [CrossRef] [PubMed]
- Jian, B.; Pang, J.; Xiong, H.; Zhang, W.; Zhan, T.; Su, Z.; Lin, H.; Zhang, H.; He, W.; Zheng, Y. Autophagy-dependent ferroptosis contributes to cisplatin-induced hearing loss. Toxicol. Lett. 2021, 350, 249–260. [Google Scholar] [CrossRef]
- Fu, D.; Wang, C.; Yu, L.; Yu, R. Induction of ferroptosis by ATF3 elevation alleviates cisplatin resistance in gastric cancer by restraining Nrf2/Keap1/xCT signaling. Cell. Mol. Biol. Lett. 2021, 26, 26. [Google Scholar] [CrossRef]
- Barbosa, L.A.; Fiuza, P.P.; Borges, L.J.; Rolim, F.A.; Andrade, M.B.; Luz, N.F.; Quintela-Carvalho, G.; Lima, J.B.; Almeida, R.P.; Chan, F.K.; et al. RIPK1-RIPK3-MLKL-Associated Necroptosis Drives Leishmania infantum Killing in Neutrophils. Front. Immunol. 2018, 9, 1818. [Google Scholar] [CrossRef]
- Tristao, V.R.; Pessoa, E.A.; Nakamichi, R.; Reis, L.A.; Batista, M.C.; Durao Junior Mde, S.; Monte, J.C. Synergistic effect of apoptosis and necroptosis inhibitors in cisplatin-induced nephrotoxicity. Apoptosis 2016, 21, 51–59. [Google Scholar] [CrossRef]
- Alassaf, N.; Attia, H. Autophagy and necroptosis in cisplatin-induced acute kidney injury: Recent advances regarding their role and therapeutic potential. Front. Pharmacol. 2023, 14, 1103062. [Google Scholar] [CrossRef] [PubMed]
- Meng, X.M.; Li, H.D.; Wu, W.F.; Ming-Kuen Tang, P.; Ren, G.L.; Gao, L.; Li, X.F.; Yang, Y.; Xu, T.; Ma, T.T.; et al. Wogonin protects against cisplatin-induced acute kidney injury by targeting RIPK1-mediated necroptosis. Lab. Investig. 2018, 98, 79–94. [Google Scholar] [CrossRef] [PubMed]
- Ramkumar, V.; Mukherjea, D.; Dhukhwa, A.; Rybak, L.P. Oxidative Stress and Inflammation Caused by Cisplatin Ototoxicity. Antioxidants 2021, 10, 1919. [Google Scholar] [CrossRef]
- Dong, X.Q.; Chu, L.K.; Cao, X.; Xiong, Q.W.; Mao, Y.M.; Chen, C.H.; Bi, Y.L.; Liu, J.; Yan, X.M. Glutathione metabolism rewiring protects renal tubule cells against cisplatin-induced apoptosis and ferroptosis. Redox Rep. 2023, 28, 2152607. [Google Scholar] [CrossRef]
- Andersson, A.; Hedenmalm, H.; Elfsson, B.; Ehrsson, H. Determination of the acid dissociation constant for cis-diammineaquachloroplatinum(II) ion. A hydrolysis product of cisplatin. J. Pharm. Sci. 1994, 83, 859–862. [Google Scholar] [CrossRef]
- Jennerwein, M.; Andrews, P.A. Effect of intracellular chloride on the cellular pharmacodynamics of cis-diamminedichloroplatinum(II). Drug Metab. Dispos. 1995, 23, 178–184. [Google Scholar]
- Aggarwal, S.K.; Broomhead, J.A.; Fairlie, D.P.; Whitehouse, M.W. Platinum drugs: Combined anti-lymphoproliferative and nephrotoxicity assay in rats. Cancer Chemother. Pharmacol. 1980, 4, 249–258. [Google Scholar] [CrossRef]
- Aggarwal, S.K. A histochemical approach to the mechanism of action of cisplatin and its analogues. J. Histochem. Cytochem. 1993, 41, 1053–1073. [Google Scholar] [CrossRef]
- Cadenas, E.; Davies, K.J. Mitochondrial free radical generation, oxidative stress, and aging. Free Radic. Biol. Med. 2000, 29, 222–230. [Google Scholar] [CrossRef]
- Aggarwal, S.K. Calcium modulation of toxicities due to Cisplatin. Met. Based Drugs 1998, 5, 77–81. [Google Scholar] [CrossRef] [PubMed]
- Niki, E. Biomarkers of lipid peroxidation in clinical material. Biochim. Biophys. Acta 2014, 1840, 809–817. [Google Scholar] [CrossRef] [PubMed]
- Minzi, O.M.S.; Lyimo, T.E.; Furia, F.F.; Marealle, A.I.; Kilonzi, M.; Bwire, G.M.; Malichewe, C. Electrolytes supplementation can decrease the risk of nephrotoxicity in patients with solid tumors undergoing chemotherapy with cisplatin. BMC Pharmacol. Toxicol. 2020, 21, 69. [Google Scholar] [CrossRef] [PubMed]
- Moyer, A.; Tanaka, K.; Cheng, E.H. Apoptosis in Cancer Biology and Therapy. Annu. Rev. Pathol. 2025, 20, 303–328. [Google Scholar] [CrossRef]
- Elmore, S. Apoptosis: A review of programmed cell death. Toxicol. Pathol. 2007, 35, 495–516. [Google Scholar] [CrossRef] [PubMed]
- Yuan, J.; Ofengeim, D. A guide to cell death pathways. Nat. Rev. Mol. Cell Biol. 2024, 25, 379–395. [Google Scholar] [CrossRef]
- Dorstyn, L.; Akey, C.W.; Kumar, S. New insights into apoptosome structure and function. Cell Death Differ. 2018, 25, 1194–1208. [Google Scholar] [CrossRef]
- Gonzalez, V.M.; Fuertes, M.A.; Alonso, C.; Perez, J.M. Is cisplatin-induced cell death always produced by apoptosis? Mol. Pharmacol. 2001, 59, 657–663. [Google Scholar] [CrossRef]
- Kashyap, D.; Garg, V.K.; Goel, N. Intrinsic and extrinsic pathways of apoptosis: Role in cancer development and prognosis. Adv. Protein Chem. Struct. Biol. 2021, 125, 73–120. [Google Scholar] [CrossRef]
- Carneiro, B.A.; El-Deiry, W.S. Targeting apoptosis in cancer therapy. Nat. Rev. Clin. Oncol. 2020, 17, 395–417. [Google Scholar] [CrossRef]
- Isakov, N. Protein kinase C (PKC) isoforms in cancer, tumor promotion and tumor suppression. Semin. Cancer Biol. 2018, 48, 36–52. [Google Scholar] [CrossRef] [PubMed]
- Martiny-Baron, G.; Fabbro, D. Classical PKC isoforms in cancer. Pharmacol. Res. 2007, 55, 477–486. [Google Scholar] [CrossRef]
- Li, N.; Zhang, W. Protein kinase C beta inhibits autophagy and sensitizes cervical cancer Hela cells to cisplatin. Biosci. Rep. 2017, 37, BSR20160445. [Google Scholar] [CrossRef]
- Zhang, Y.; Yonezawa, A.; Nakagawa, S.; Imai, S.; Denda, M.; Omura, T.; Nakagawa, T.; Matsubara, K. Cisplatin, rather than oxaliplatin, increases paracellular permeability of LLC-PK1 cells via activating protein kinase C. Drug Metab. Pharmacokinet 2020, 35, 111–116. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Pan, J.; Xiang, X.; Liu, Y.; Dong, G.; Livingston, M.J.; Chen, J.K.; Yin, X.M.; Dong, Z. Protein Kinase Cdelta Suppresses Autophagy to Induce Kidney Cell Apoptosis in Cisplatin Nephrotoxicity. J. Am. Soc. Nephrol. 2017, 28, 1131–1144. [Google Scholar] [CrossRef] [PubMed]
- Muscella, A.; Urso, L.; Calabriso, N.; Vetrugno, C.; Rochira, A.; Storelli, C.; Marsigliante, S. Anti-apoptotic effects of protein kinase C-delta and c-fos in cisplatin-treated thyroid cells. Br. J. Pharmacol. 2009, 156, 751–763. [Google Scholar] [CrossRef]
- Reyland, M.E. Protein kinase C isoforms: Multi-functional regulators of cell life and death. Front. Biosci. 2009, 14, 2386–2399. [Google Scholar] [CrossRef]
- Johnson, G.L.; Lapadat, R. Mitogen-activated protein kinase pathways mediated by ERK, JNK, and p38 protein kinases. Science 2002, 298, 1911–1912. [Google Scholar] [CrossRef]
- Ronkina, N.; Gaestel, M. MAPK-Activated Protein Kinases: Servant or Partner? Annu. Rev. Biochem. 2022, 91, 505–540. [Google Scholar] [CrossRef] [PubMed]
- Runchel, C.; Matsuzawa, A.; Ichijo, H. Mitogen-activated protein kinases in mammalian oxidative stress responses. Antioxid. Redox Signal. 2011, 15, 205–218. [Google Scholar] [CrossRef]
- Tang, M.K.; Zhou, H.Y.; Yam, J.W.; Wong, A.S. c-Met overexpression contributes to the acquired apoptotic resistance of nonadherent ovarian cancer cells through a cross talk mediated by phosphatidylinositol 3-kinase and extracellular signal-regulated kinase 1/2. Neoplasia 2010, 12, 128–138. [Google Scholar] [CrossRef]
- Basu, A.; Tu, H. Activation of ERK during DNA damage-induced apoptosis involves protein kinase Cdelta. Biochem. Biophys. Res. Commun. 2005, 334, 1068–1073. [Google Scholar] [CrossRef]
- Tao, C.; Lin, H.; Chen, S. The regulation of ERK and p-ERK expression by cisplatin and sorafenib in gastric cancer cells. Gene 2014, 552, 106–115. [Google Scholar] [CrossRef]
- Sun, C.Y.; Cao, D.; Ren, Q.N.; Zhang, S.S.; Zhou, N.N.; Mai, S.J.; Feng, B.; Wang, H.Y. Combination Treatment With Inhibitors of ERK and Autophagy Enhances Antitumor Activity of Betulinic Acid in Non-small-Cell Lung Cancer In Vivo and In Vitro. Front. Pharmacol. 2021, 12, 684243. [Google Scholar] [CrossRef]
- Wang, J.; Zhou, J.Y.; Wu, G.S. ERK-dependent MKP-1-mediated cisplatin resistance in human ovarian cancer cells. Cancer Res. 2007, 67, 11933–11941. [Google Scholar] [CrossRef]
- DeHaan, R.D.; Yazlovitskaya, E.M.; Persons, D.L. Regulation of p53 target gene expression by cisplatin-induced extracellular signal-regulated kinase. Cancer Chemother. Pharmacol. 2001, 48, 383–388. [Google Scholar] [CrossRef] [PubMed]
- Winograd-Katz, S.E.; Levitzki, A. Cisplatin induces PKB/Akt activation and p38(MAPK) phosphorylation of the EGF receptor. Oncogene 2006, 25, 7381–7390. [Google Scholar] [CrossRef] [PubMed]
- Cuadrado, A.; Lafarga, V.; Cheung, P.C.; Dolado, I.; Llanos, S.; Cohen, P.; Nebreda, A.R. A new p38 MAP kinase-regulated transcriptional coactivator that stimulates p53-dependent apoptosis. EMBO J. 2007, 26, 2115–2126. [Google Scholar] [CrossRef]
- Minerva; Bhat, A.; Verma, S.; Chander, G.; Jamwal, R.S.; Sharma, B.; Bhat, A.; Katyal, T.; Kumar, R.; Shah, R. Cisplatin-based combination therapy for cancer. J. Cancer Res. Ther. 2023, 19, 530–536. [Google Scholar] [CrossRef]
- Jones, E.V.; Dickman, M.J.; Whitmarsh, A.J. Regulation of p73-mediated apoptosis by c-Jun N-terminal kinase. Biochem. J. 2007, 405, 617–623. [Google Scholar] [CrossRef]
- Toh, W.H.; Siddique, M.M.; Boominathan, L.; Lin, K.W.; Sabapathy, K. c-Jun regulates the stability and activity of the p53 homologue, p73. J. Biol. Chem. 2004, 279, 44713–44722. [Google Scholar] [CrossRef] [PubMed]
- Revathidevi, S.; Munirajan, A.K. Akt in cancer: Mediator and more. Semin. Cancer Biol. 2019, 59, 80–91. [Google Scholar] [CrossRef]
- Hayakawa, J.; Ohmichi, M.; Kurachi, H.; Kanda, Y.; Hisamoto, K.; Nishio, Y.; Adachi, K.; Tasaka, K.; Kanzaki, T.; Murata, Y. Inhibition of BAD phosphorylation either at serine 112 via extracellular signal-regulated protein kinase cascade or at serine 136 via Akt cascade sensitizes human ovarian cancer cells to cisplatin. Cancer Res. 2000, 60, 5988–5994. [Google Scholar] [PubMed]
- Basu, A.; Krishnamurthy, S. Cellular responses to Cisplatin-induced DNA damage. J. Nucleic Acids 2010, 2010, 201367. [Google Scholar] [CrossRef] [PubMed]
- Shen, D.W.; Liang, X.J.; Gawinowicz, M.A.; Gottesman, M.M. Identification of cytoskeletal [14C]carboplatin-binding proteins reveals reduced expression and disorganization of actin and filamin in cisplatin-resistant cell lines. Mol. Pharmacol. 2004, 66, 789–793. [Google Scholar] [CrossRef]
- Johnson, S.W.; Shen, D.; Pastan, I.; Gottesman, M.M.; Hamilton, T.C. Cross-resistance, cisplatin accumulation, and platinum-DNA adduct formation and removal in cisplatin-sensitive and -resistant human hepatoma cell lines. Exp. Cell Res. 1996, 226, 133–139. [Google Scholar] [CrossRef]
- Mandic, A.; Hansson, J.; Linder, S.; Shoshan, M.C. Cisplatin induces endoplasmic reticulum stress and nucleus-independent apoptotic signaling. J. Biol. Chem. 2003, 278, 9100–9106. [Google Scholar] [CrossRef]
- Liu, J.R.; Opipari, A.W.; Tan, L.; Jiang, Y.; Zhang, Y.; Tang, H.; Nunez, G. Dysfunctional apoptosome activation in ovarian cancer: Implications for chemoresistance. Cancer Res. 2002, 62, 924–931. [Google Scholar]
- Arts, H.J.; Hollema, H.; Lemstra, W.; Willemse, P.H.; De Vries, E.G.; Kampinga, H.H.; Van der Zee, A.G. Heat-shock-protein-27 (hsp27) expression in ovarian carcinoma: Relation in response to chemotherapy and prognosis. Int. J. Cancer 1999, 84, 234–238. [Google Scholar] [CrossRef]
- Huang, T.G.; Ip, S.M.; Yeung, W.S.; Ngan, H.Y. Changes in p21WAF1, pRb, Mdm-2, Bax and Bcl-2 expression in cervical cancer cell lines transfected with a p53 expressing adenovirus. Eur. J. Cancer 2000, 36, 249–256. [Google Scholar] [CrossRef]
- Belfi, C.A.; Chatterjee, S.; Gosky, D.M.; Berger, S.J.; Berger, N.A. Increased sensitivity of human colon cancer cells to DNA cross-linking agents after GRP78 up-regulation. Biochem. Biophys. Res. Commun. 1999, 257, 361–368. [Google Scholar] [CrossRef]
- Vargas-Roig, L.M.; Gago, F.E.; Tello, O.; Aznar, J.C.; Ciocca, D.R. Heat shock protein expression and drug resistance in breast cancer patients treated with induction chemotherapy. Int. J. Cancer 1998, 79, 468–475. [Google Scholar] [CrossRef]
- Shen, D.W.; Liang, X.J.; Suzuki, T.; Gottesman, M.M. Identification by functional cloning from a retroviral cDNA library of cDNAs for ribosomal protein L36 and the 10-kDa heat shock protein that confer cisplatin resistance. Mol. Pharmacol. 2006, 69, 1383–1388. [Google Scholar] [CrossRef] [PubMed]
- Smith, L.; Welham, K.J.; Watson, M.B.; Drew, P.J.; Lind, M.J.; Cawkwell, L. The proteomic analysis of cisplatin resistance in breast cancer cells. Oncol. Res. 2007, 16, 497–506. [Google Scholar] [CrossRef]
- Llanos, S.; Serrano, M. Depletion of ribosomal protein L37 occurs in response to DNA damage and activates p53 through the L11/MDM2 pathway. Cell Cycle 2010, 9, 4005–4012. [Google Scholar] [CrossRef]
- Tossetta, G.; Fantone, S.; Marzioni, D.; Mazzucchelli, R. Cellular Modulators of the NRF2/KEAP1 Signaling Pathway in Prostate Cancer. Front. Biosci. 2023, 28, 143. [Google Scholar] [CrossRef]
- Marzioni, D.; Mazzucchelli, R.; Fantone, S.; Tossetta, G. NRF2 modulation in TRAMP mice: An in vivo model of prostate cancer. Mol. Biol. Rep. 2022, 50, 871–881. [Google Scholar] [CrossRef] [PubMed]
- Tossetta, G.; Marzioni, D. Natural and synthetic compounds in Ovarian Cancer: A focus on NRF2/KEAP1 pathway. Pharmacol. Res. 2022, 183, 106365. [Google Scholar] [CrossRef] [PubMed]
- Cho, J.M.; Manandhar, S.; Lee, H.R.; Park, H.M.; Kwak, M.K. Role of the Nrf2-antioxidant system in cytotoxicity mediated by anticancer cisplatin: Implication to cancer cell resistance. Cancer Lett. 2008, 260, 96–108. [Google Scholar] [CrossRef] [PubMed]
- Fantone, S.; Marzioni, D.; Tossetta, G. NRF2/KEAP1 signaling inhibitors in gynecologic cancers. Expert Rev. Anticancer Ther. 2024, 24, 1191–1194. [Google Scholar] [CrossRef] [PubMed]
- Tossetta, G.; Fantone, S.; Goteri, G.; Giannubilo, S.R.; Ciavattini, A.; Marzioni, D. The Role of NQO1 in Ovarian Cancer. Int. J. Mol. Sci. 2023, 24, 7839. [Google Scholar] [CrossRef]
- Tossetta, G.; Marzioni, D. Targeting the NRF2/KEAP1 pathway in cervical and endometrial cancers. Eur. J. Pharmacol. 2023, 941, 175503. [Google Scholar] [CrossRef]
- Pan, C.; Kang, J.; Hwang, J.S.; Li, J.; Boese, A.C.; Wang, X.; Yang, L.; Boggon, T.J.; Chen, G.Z.; Saba, N.F.; et al. Cisplatin-mediated activation of glucocorticoid receptor induces platinum resistance via MAST1. Nat. Commun. 2021, 12, 4960. [Google Scholar] [CrossRef]
- Jin, L.; Chun, J.; Pan, C.; Li, D.; Lin, R.; Alesi, G.N.; Wang, X.; Kang, H.B.; Song, L.; Wang, D.; et al. MAST1 Drives Cisplatin Resistance in Human Cancers by Rewiring cRaf-Independent MEK Activation. Cancer Cell 2018, 34, 315–330 e317. [Google Scholar] [CrossRef]
- Galluzzi, L.; Senovilla, L.; Vitale, I.; Michels, J.; Martins, I.; Kepp, O.; Castedo, M.; Kroemer, G. Molecular mechanisms of cisplatin resistance. Oncogene 2012, 31, 1869–1883. [Google Scholar] [CrossRef] [PubMed]
- Wittschieben, J.P.; Reshmi, S.C.; Gollin, S.M.; Wood, R.D. Loss of DNA polymerase zeta causes chromosomal instability in mammalian cells. Cancer Res. 2006, 66, 134–142. [Google Scholar] [CrossRef]
- Wang, H.; Zhang, S.Y.; Wang, S.; Lu, J.; Wu, W.; Weng, L.; Chen, D.; Zhang, Y.; Lu, Z.; Yang, J.; et al. REV3L confers chemoresistance to cisplatin in human gliomas: The potential of its RNAi for synergistic therapy. Neuro. Oncol. 2009, 11, 790–802. [Google Scholar] [CrossRef]
- Castedo, M.; Coquelle, A.; Vivet, S.; Vitale, I.; Kauffmann, A.; Dessen, P.; Pequignot, M.O.; Casares, N.; Valent, A.; Mouhamad, S.; et al. Apoptosis regulation in tetraploid cancer cells. EMBO J. 2006, 25, 2584–2595. [Google Scholar] [CrossRef]
- Kato, J.; Kuwabara, Y.; Mitani, M.; Shinoda, N.; Sato, A.; Toyama, T.; Mitsui, A.; Nishiwaki, T.; Moriyama, S.; Kudo, J.; et al. Expression of survivin in esophageal cancer: Correlation with the prognosis and response to chemotherapy. Int. J. Cancer 2001, 95, 92–95. [Google Scholar] [CrossRef]
- Aptullahoglu, E.; Kaygusuz, E. Synergistic MDM2-STAT3 Inhibition Demonstrates Strong Anti-Leukemic Efficacy in Acute Lymphoblastic Leukemia. Int. J. Mol. Sci. 2025, 26, 8648. [Google Scholar] [CrossRef]
- Feng, Y.; Chen, F.; Mu, C.; Wang, L.; Jiang, Y.; Liu, D.; Li, D.; Liang, C.; Zhai, Y.; Yang, T.; et al. M1 macrophages enhance breast cancer chemoresistance via JAK-STAT3 signaling. Biochim. Biophys. Acta Mol. Basis. Dis. 2026, 1872, 168056. [Google Scholar] [CrossRef]
- Yu, H.; Lee, H.; Herrmann, A.; Buettner, R.; Jove, R. Revisiting STAT3 signalling in cancer: New and unexpected biological functions. Nat. Rev. Cancer 2014, 14, 736–746. [Google Scholar] [CrossRef] [PubMed]
- Bosch-Barrera, J.; Queralt, B.; Menendez, J.A. Targeting STAT3 with silibinin to improve cancer therapeutics. Cancer Treat. Rev. 2017, 58, 61–69. [Google Scholar] [CrossRef] [PubMed]
- Lynch, H.T.; Casey, M.J.; Snyder, C.L.; Bewtra, C.; Lynch, J.F.; Butts, M.; Godwin, A.K. Hereditary ovarian carcinoma: Heterogeneity, molecular genetics, pathology, and management. Mol. Oncol. 2009, 3, 97–137. [Google Scholar] [CrossRef] [PubMed]
- Tossetta, G.; Inversetti, A. Ovarian Cancer: Advances in Pathophysiology and Therapies. Int. J. Mol. Sci. 2023, 24, 8930. [Google Scholar] [CrossRef]
- Tossetta, G.; Fantone, S.; Montanari, E.; Marzioni, D.; Goteri, G. Role of NRF2 in Ovarian Cancer. Antioxidants 2022, 11, 663. [Google Scholar] [CrossRef]
- Fantone, S.; Piani, F.; Olivieri, F.; Rippo, M.R.; Sirico, A.; Di Simone, N.; Marzioni, D.; Tossetta, G. Role of SLC7A11/xCT in Ovarian Cancer. Int. J. Mol. Sci. 2024, 25, 587. [Google Scholar] [CrossRef]
- Alizadehnohi, M.; Nabiuni, M.; Nazari, Z.; Safaeinejad, Z.; Irian, S. The synergistic cytotoxic effect of cisplatin and honey bee venom on human ovarian cancer cell line A2780cp. J. Venom Res. 2012, 3, 22–27. [Google Scholar]
- Meng, F.; Sun, G.; Zhong, M.; Yu, Y.; Brewer, M.A. Anticancer efficacy of cisplatin and trichostatin A or 5-aza-2′-deoxycytidine on ovarian cancer. Br. J. Cancer 2013, 108, 579–586. [Google Scholar] [CrossRef]
- Kakar, S.S.; Jala, V.R.; Fong, M.Y. Synergistic cytotoxic action of cisplatin and withaferin A on ovarian cancer cell lines. Biochem. Biophys. Res. Commun. 2012, 423, 819–825. [Google Scholar] [CrossRef]
- Ni, M.; Zhou, J.; Zhu, Z.; Xu, Q.; Yin, Z.; Wang, Y.; Zheng, Z.; Zhao, H. Shikonin and cisplatin synergistically overcome cisplatin resistance of ovarian cancer by inducing ferroptosis via upregulation of HMOX1 to promote Fe(2+) accumulation. Phytomedicine 2023, 112, 154701. [Google Scholar] [CrossRef]
- Sue, S.H.; Tseng, W.C.; Wu, Z.S.; Huang, S.M.; Chen, J.L.; Wu, Z.F.; Lai, H.C. The synergistic mechanisms of propofol with cisplatin or doxorubicin in human ovarian cancer cells. J. Ovarian Res. 2024, 17, 187. [Google Scholar] [CrossRef]
- Duan, J.; Zhang, Z.; Du, J.; Zhang, J.; Li, M.; Li, C. Esomeprazole Alleviates Cisplatin Resistance by Inhibiting the AKT/mTOR Pathway in Ovarian Cancer Cells. Onco. Targets Ther. 2023, 16, 425–440. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Liu, W.; Zhan, X.; Zhong, Y.; Feng, Y.; Cao, Q.; Tan, B. Synergistic effect of Tripterygium glycosides and cisplatin on drug-resistant human epithelial ovarian cancer via ILK/GSK3beta/Slug signal pathway. Am. J. Transl. Res. 2022, 14, 2051–2062. [Google Scholar] [PubMed]
- Veronesi, U.; Boyle, P.; Goldhirsch, A.; Orecchia, R.; Viale, G. Breast cancer. Lancet 2005, 365, 1727–1741. [Google Scholar] [CrossRef] [PubMed]
- Campagna, R.; Pozzi, V.; Giorgini, S.; Morichetti, D.; Goteri, G.; Sartini, D.; Serritelli, E.N.; Emanuelli, M. Paraoxonase-2 is upregulated in triple negative breast cancer and contributes to tumor progression and chemoresistance. Hum. Cell 2023, 36, 1108–1119. [Google Scholar] [CrossRef]
- Hashemi, M.; Khosroshahi, E.M.; Chegini, M.K.; Asadi, S.; Hamyani, Z.; Jafari, Y.A.; Rezaei, F.; Eskadehi, R.K.; Kojoori, K.K.; Jamshidian, F.; et al. Mechanistic insights into cisplatin response in breast tumors: Molecular determinants and drug/nanotechnology-based therapeutic opportunities. Mutat. Res. Rev. Mutat. Res. 2024, 794, 108513. [Google Scholar] [CrossRef]
- Siddik, Z.H. Cisplatin: Mode of cytotoxic action and molecular basis of resistance. Oncogene 2003, 22, 7265–7279. [Google Scholar] [CrossRef]
- Brody, H. Lung cancer. Nature 2020, 587, S7. [Google Scholar] [CrossRef]
- Rossi, A.; Di Maio, M.; Chiodini, P.; Rudd, R.M.; Okamoto, H.; Skarlos, D.V.; Fruh, M.; Qian, W.; Tamura, T.; Samantas, E.; et al. Carboplatin- or cisplatin-based chemotherapy in first-line treatment of small-cell lung cancer: The COCIS meta-analysis of individual patient data. J. Clin. Oncol. 2012, 30, 1692–1698. [Google Scholar] [CrossRef]
- Mense, E.S.; Smit, A.A.J.; Crul, M.; Franssen, E.J.F. The effect of rapid infusion of cisplatin on nephrotoxicity in patients with lung carcinoma. J. Clin. Pharm. Ther. 2019, 44, 249–257. [Google Scholar] [CrossRef]
- Iwasaki, Y.; Nagata, K.; Nakanishi, M.; Natuhara, A.; Kubota, Y.; Ueda, M.; Arimoto, T.; Hara, H. Double-cycle, high-dose ifosfamide, carboplatin, and etoposide followed by peripheral blood stem-cell transplantation for small cell lung cancer. Chest 2005, 128, 2268–2273. [Google Scholar] [CrossRef]
- Kosmas, C.; Tsavaris, N.B.; Malamos, N.A.; Vadiaka, M.; Koufos, C. Phase II study of paclitaxel, ifosfamide, and cisplatin as second-line treatment in relapsed small-cell lung cancer. J. Clin. Oncol. 2001, 19, 119–126. [Google Scholar] [CrossRef]
- Pignon, J.P.; Tribodet, H.; Scagliotti, G.V.; Douillard, J.Y.; Shepherd, F.A.; Stephens, R.J.; Dunant, A.; Torri, V.; Rosell, R.; Seymour, L.; et al. Lung adjuvant cisplatin evaluation: A pooled analysis by the LACE Collaborative Group. J. Clin. Oncol. 2008, 26, 3552–3559. [Google Scholar] [CrossRef]
- Bertolini, G.; Roz, L.; Perego, P.; Tortoreto, M.; Fontanella, E.; Gatti, L.; Pratesi, G.; Fabbri, A.; Andriani, F.; Tinelli, S.; et al. Highly tumorigenic lung cancer CD133+ cells display stem-like features and are spared by cisplatin treatment. Proc. Natl. Acad. Sci. USA 2009, 106, 16281–16286. [Google Scholar] [CrossRef] [PubMed]
- Polizzi, A.; Tartaglia, G.M.; Santonocito, S.; Alibrandi, A.; Verzi, A.E.; Isola, G. Impact of Topical Fluocinonide on Oral Lichen Planus Evolution: Randomized Controlled Clinical Trial. Oral. Dis. 2025, 31, 510–521. [Google Scholar] [CrossRef]
- Polizzi, A.; Santonocito, S.; Distefano, A.; De Pasquale, R.; Alibrandi, A.; Alanazi, A.M.; Li Volti, G.; Isola, G. Analysis of oral lichen planus severity on micro-RNA linked with malignant transformation risks. Oral. Dis. 2024, 30, 2918–2928. [Google Scholar] [CrossRef] [PubMed]
- Vengaloor Thomas, T.; Packianathan, S.; Bhanat, E.; Albert, A.; Abraham, A.; Gordy, X.; Kanakamedala, M.; Mehta, D.; Vijayakumar, S. Oligometastatic head and neck cancer: Comprehensive review. Head Neck 2020, 42, 2194–2201. [Google Scholar] [CrossRef] [PubMed]
- Campagna, R.; Pozzi, V.; Salvucci, A.; Togni, L.; Mascitti, M.; Sartini, D.; Salvolini, E.; Santarelli, A.; Lo Muzio, L.; Emanuelli, M. Paraoxonase-2 expression in oral squamous cell carcinoma. Hum. Cell 2023, 36, 1211–1213. [Google Scholar] [CrossRef]
- Campagna, R.; Belloni, A.; Pozzi, V.; Salvucci, A.; Notarstefano, V.; Togni, L.; Mascitti, M.; Sartini, D.; Giorgini, E.; Salvolini, E.; et al. Role Played by Paraoxonase-2 Enzyme in Cell Viability, Proliferation and Sensitivity to Chemotherapy of Oral Squamous Cell Carcinoma Cell Lines. Int. J. Mol. Sci. 2022, 24, 338. [Google Scholar] [CrossRef]
- Belloni, A.; Campagna, R.; Notarstefano, V.; Pozzi, V.; Orilisi, G.; Pompei, V.; Togni, L.; Mascitti, M.; Sartini, D.; Giorgini, E.; et al. Deepening Cisplatin sensitivity on Oral Squamous cell Carcinoma cell lines after PON2 knockdown: A FTIRM investigation. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2025, 330, 125726. [Google Scholar] [CrossRef]
- Kim, S.S.; Liu, H.C.; Mell, L.K. Treatment Considerations for Patients with Locoregionally Advanced Head and Neck Cancer with a Contraindication to Cisplatin. Curr. Treat. Options Oncol. 2023, 24, 147–161. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.; Li, B.; Wang, J.; Tan, Y.; Xu, M.; Xu, W.; Lu, H. New advances into cisplatin resistance in head and neck squamous carcinoma: Mechanisms and therapeutic aspects. Biomed. Pharmacother. 2023, 163, 114778. [Google Scholar] [CrossRef]
- Oshimori, N. Cancer stem cells and their niche in the progression of squamous cell carcinoma. Cancer Sci. 2020, 111, 3985–3992. [Google Scholar] [CrossRef] [PubMed]
- Salem, A.; Salo, T. Identity matters: Cancer stem cells and tumour plasticity in head and neck squamous cell carcinoma. Expert Rev. Mol. Med. 2023, 25, e8. [Google Scholar] [CrossRef]
- Lima de Oliveira, J.; More Milan, T.; Longo Bighetti-Trevisan, R.; Fernandes, R.R.; Machado Leopoldino, A.; Oliveira de Almeida, L. Epithelial-mesenchymal transition and cancer stem cells: A route to acquired cisplatin resistance through epigenetics in HNSCC. Oral. Dis. 2023, 29, 1991–2005. [Google Scholar] [CrossRef] [PubMed]
- Wirsching, H.G.; Galanis, E.; Weller, M. Glioblastoma. Handb. Clin. Neurol. 2016, 134, 381–397. [Google Scholar] [CrossRef]
- Wang, Y.; Kong, X.; Guo, Y.; Wang, R.; Ma, W. Continuous dose-intense temozolomide and cisplatin in recurrent glioblastoma patients. Medicine 2017, 96, e6261. [Google Scholar] [CrossRef]
- Roberts, N.B.; Wadajkar, A.S.; Winkles, J.A.; Davila, E.; Kim, A.J.; Woodworth, G.F. Repurposing platinum-based chemotherapies for multi-modal treatment of glioblastoma. Oncoimmunology 2016, 5, e1208876. [Google Scholar] [CrossRef][Green Version]
- Hegi, M.E.; Diserens, A.C.; Gorlia, T.; Hamou, M.F.; de Tribolet, N.; Weller, M.; Kros, J.M.; Hainfellner, J.A.; Mason, W.; Mariani, L.; et al. MGMT gene silencing and benefit from temozolomide in glioblastoma. N. Engl. J. Med. 2005, 352, 997–1003. [Google Scholar] [CrossRef] [PubMed]
- Roda, D.; Veiga, P.; Melo, J.B.; Carreira, I.M.; Ribeiro, I.P. Principles in the Management of Glioblastoma. Genes 2024, 15, 501. [Google Scholar] [CrossRef]
- Charest, G.; Sanche, L.; Fortin, D.; Mathieu, D.; Paquette, B. Glioblastoma treatment: Bypassing the toxicity of platinum compounds by using liposomal formulation and increasing treatment efficiency with concomitant radiotherapy. Int. J. Radiat. Oncol. Biol. Phys. 2012, 84, 244–249. [Google Scholar] [CrossRef] [PubMed]
- Campagna, R.; Bacchetti, T.; Salvolini, E.; Pozzi, V.; Molinelli, E.; Brisigotti, V.; Sartini, D.; Campanati, A.; Ferretti, G.; Offidani, A.; et al. Paraoxonase-2 Silencing Enhances Sensitivity of A375 Melanoma Cells to Treatment with Cisplatin. Antioxidants 2020, 9, 1238. [Google Scholar] [CrossRef]
- Campagna, R.; Salvolini, E.; Pompei, V.; Pozzi, V.; Salvucci, A.; Molinelli, E.; Brisigotti, V.; Sartini, D.; Campanati, A.; Offidani, A.; et al. Nicotinamide N-methyltransferase gene silencing enhances chemosensitivity of melanoma cell lines. Pigment Cell Melanoma Res. 2021, 34, 1039–1048. [Google Scholar] [CrossRef] [PubMed]
- Trodello, C.; Pepper, J.P.; Wong, M.; Wysong, A. Cisplatin and Cetuximab Treatment for Metastatic Cutaneous Squamous Cell Carcinoma: A Systematic Review. Dermatol. Surg. 2017, 43, 40–49. [Google Scholar] [CrossRef]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Gupta, J.; Ahmed, A.T.; Tayyib, N.A.; Zabibah, R.S.; Shomurodov, Q.; Kadheim, M.N.; Alsaikhan, F.; Ramaiah, P.; Chinnasamy, L.; Samarghandian, S. A state-of-art of underlying molecular mechanisms and pharmacological interventions/nanotherapeutics for cisplatin resistance in gastric cancer. Biomed. Pharmacother. 2023, 166, 115337. [Google Scholar] [CrossRef]
- Fu, X.; Feng, J.; Zeng, D.; Ding, Y.; Yu, C.; Yang, B. PAK4 confers cisplatin resistance in gastric cancer cells via PI3K/Akt- and MEK/ERK-dependent pathways. Biosci. Rep. 2014, 34, e00094. [Google Scholar] [CrossRef]
- Wei, J.; Wang, R.; Lu, Y.; He, S.; Ding, Y. Flotillin-1 promotes progression and dampens chemosensitivity to cisplatin in gastric cancer via ERK and AKT signaling pathways. Eur. J. Pharmacol. 2022, 916, 174631. [Google Scholar] [CrossRef]
- Liu, H.; Li, P.; Li, B.; Sun, P.; Zhang, J.; Wang, B.; Jia, B. RKIP promotes cisplatin-induced gastric cancer cell death through NF-kappaB/Snail pathway. Tumour Biol. 2015, 36, 1445–1453. [Google Scholar] [CrossRef] [PubMed]
- Lin, X.M.; Li, S.; Zhou, C.; Li, R.Z.; Wang, H.; Luo, W.; Huang, Y.S.; Chen, L.K.; Cai, J.L.; Wang, T.X.; et al. Cisplatin induces chemoresistance through the PTGS2-mediated anti-apoptosis in gastric cancer. Int. J. Biochem. Cell Biol. 2019, 116, 105610. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Cai, S.; Deng, Y.; Zhang, Z.; Zhou, X.; Su, Y.; Xu, D. PD-1/PD-L1 enhanced cisplatin resistance in gastric cancer through PI3K/AKT mediated P-gp expression. Int. Immunopharmacol. 2021, 94, 107443. [Google Scholar] [CrossRef]
- Li, J.; Zhang, Y.; Ye, F.; Qian, P.; Qin, Z.; Li, D.; Ye, L.; Feng, L. DKK1 Promotes Epithelial-Mesenchymal Transition and Cisplatin Resistance in Gastric Cancer via Activation of the PI3K/AKT Pathway. Cancers 2023, 15, 4756. [Google Scholar] [CrossRef] [PubMed]
- Hall, C.E.; Maegawa, F.; Patel, A.D.; Lin, E. Management of Gastric Cancer. Am. Surg. 2023, 89, 2713–2720. [Google Scholar] [CrossRef]
- Liu, C.; Li, S.; Tang, Y. Mechanism of cisplatin resistance in gastric cancer and associated microRNAs. Cancer Chemother. Pharmacol. 2023, 92, 329–340. [Google Scholar] [CrossRef]
- Pozzi, V.; Campagna, R.; Sartini, D.; Emanuelli, M. Nicotinamide N-Methyltransferase as Promising Tool for Management of Gastrointestinal Neoplasms. Biomolecules 2022, 12, 1173. [Google Scholar] [CrossRef]
- Rozencweig, M.; von Hoff, D.D.; Slavik, M.; Muggia, F.M. Cis-diamminedichloroplatinum (II). A new anticancer drug. Ann. Intern. Med. 1977, 86, 803–812. [Google Scholar] [CrossRef]
- Patel, P.M.; Patel, H.D.; Koehne, E.L.; Doshi, C.; Belshoff, A.; Seffren, C.M.; Baker, M.; Gorbonos, A.; Gupta, G. Contemporary Trends in Presentation and Management of Spermatocytic Seminoma. Urology 2020, 146, 177–182. [Google Scholar] [CrossRef]
- de Vries, G.; Rosas-Plaza, X.; van Vugt, M.; Gietema, J.A.; de Jong, S. Testicular cancer: Determinants of cisplatin sensitivity and novel therapeutic opportunities. Cancer Treat. Rev. 2020, 88, 102054. [Google Scholar] [CrossRef]
- Chavarriaga, J.; Nappi, L.; Papachristofilou, A.; Conduit, C.; Hamilton, R.J. Testicular cancer. Lancet 2025, 406, 76–90. [Google Scholar] [CrossRef]
- Tossetta, G.; Mazzucchelli, R. Pathology of Urologic Cancers. Cancers 2022, 14, 3751. [Google Scholar] [CrossRef]
- Fumarola, S.; Cecati, M.; Sartini, D.; Ferretti, G.; Milanese, G.; Galosi, A.B.; Pozzi, V.; Campagna, R.; Morresi, C.; Emanuelli, M.; et al. Bladder Cancer Chemosensitivity is Affected by Paraoxonase-2 Expression. Antioxidants 2020, 9, 175. [Google Scholar] [CrossRef] [PubMed]
- Shi, Z.D.; Hao, L.; Han, X.X.; Wu, Z.X.; Pang, K.; Dong, Y.; Qin, J.X.; Wang, G.Y.; Zhang, X.M.; Xia, T.; et al. Targeting HNRNPU to overcome cisplatin resistance in bladder cancer. Mol. Cancer 2022, 21, 37. [Google Scholar] [CrossRef]
- Li, Q.Q.; Liu, M.Z.; Hu, Y.H.; Liu, H.; He, Z.Y.; Lin, H.X. Definitive concomitant chemoradiotherapy with docetaxel and cisplatin in squamous esophageal carcinoma. Dis. Esophagus 2010, 23, 253–259. [Google Scholar] [CrossRef]
- dos Santos, N.A.; Martins, N.M.; Curti, C.; Pires Bianchi Mde, L.; dos Santos, A.C. Dimethylthiourea protects against mitochondrial oxidative damage induced by cisplatin in liver of rats. Chem. Biol. Interact. 2007, 170, 177–186. [Google Scholar] [CrossRef] [PubMed]
- Yilmaz, H.R.; Sogut, S.; Ozyurt, B.; Ozugurlu, F.; Sahin, S.; Isik, B.; Uz, E.; Ozyurt, H. The activities of liver adenosine deaminase, xanthine oxidase, catalase, superoxide dismutase enzymes and the levels of malondialdehyde and nitric oxide after cisplatin toxicity in rats: Protective effect of caffeic acid phenethyl ester. Toxicol. Ind. Health 2005, 21, 67–73. [Google Scholar] [CrossRef]
- Iseri, S.; Ercan, F.; Gedik, N.; Yuksel, M.; Alican, I. Simvastatin attenuates cisplatin-induced kidney and liver damage in rats. Toxicology 2007, 230, 256–264. [Google Scholar] [CrossRef]
- Al-Majed, A.A.; Sayed-Ahmed, M.M.; Al-Yahya, A.A.; Aleisa, A.M.; Al-Rejaie, S.S.; Al-Shabanah, O.A. Propionyl-L-carnitine prevents the progression of cisplatin-induced cardiomyopathy in a carnitine-depleted rat model. Pharmacol. Res. 2006, 53, 278–286. [Google Scholar] [CrossRef] [PubMed]
- Kuhlmann, M.K.; Burkhardt, G.; Kohler, H. Insights into potential cellular mechanisms of cisplatin nephrotoxicity and their clinical application. Nephrol. Dial. Transplant 1997, 12, 2478–2480. [Google Scholar] [CrossRef]
- Crona, D.J.; Faso, A.; Nishijima, T.F.; McGraw, K.A.; Galsky, M.D.; Milowsky, M.I. A Systematic Review of Strategies to Prevent Cisplatin-Induced Nephrotoxicity. Oncologist 2017, 22, 609–619. [Google Scholar] [CrossRef]
- Money, M.E.; Hamroun, A.; Shu, Y.; Matthews, C.; Ahmed Eltayeb, S.; Ciarimboli, G.; Metz, C.N. Case Report and Supporting Documentation: Acute Kidney Injury Manifested as Oliguria Is Reduced by Intravenous Magnesium Before Cisplatin. Front. Oncol. 2021, 11, 607574. [Google Scholar] [CrossRef]
- Morgan, K.P.; Snavely, A.C.; Wind, L.S.; Buie, L.W.; Grilley-Olson, J.; Walko, C.M.; Weiss, J. Rates of Renal Toxicity in Cancer Patients Receiving Cisplatin With and Without Mannitol. Ann. Pharmacother. 2014, 48, 863–869. [Google Scholar] [CrossRef]
- Ostrow, S.; Egorin, M.J.; Hahn, D.; Markus, S.; Aisner, J.; Chang, P.; LeRoy, A.; Bachur, N.R.; Wiernik, P.H. High-dose cisplatin therapy using mannitol versus furosemide diuresis: Comparative pharmacokinetics and toxicity. Cancer Treat. Rep. 1981, 65, 73–78. [Google Scholar]
- Reyes, A.J. Loop diuretics versus others in the treatment of congestive heart failure after myocardial infarction. Cardiovasc. Drugs Ther. 1993, 7, 869–876. [Google Scholar] [CrossRef] [PubMed]
- Brouwers, E.E.; Huitema, A.D.; Boogerd, W.; Beijnen, J.H.; Schellens, J.H. Persistent neuropathy after treatment with cisplatin and oxaliplatin. Acta Oncol. 2009, 48, 832–841. [Google Scholar] [CrossRef]
- Avan, A.; Postma, T.J.; Ceresa, C.; Avan, A.; Cavaletti, G.; Giovannetti, E.; Peters, G.J. Platinum-induced neurotoxicity and preventive strategies: Past, present, and future. Oncologist 2015, 20, 411–432. [Google Scholar] [CrossRef] [PubMed]
- Areti, A.; Yerra, V.G.; Naidu, V.; Kumar, A. Oxidative stress and nerve damage: Role in chemotherapy induced peripheral neuropathy. Redox Biol. 2014, 2, 289–295. [Google Scholar] [CrossRef]
- Santos, N.; Ferreira, R.S.; Santos, A.C.D. Overview of cisplatin-induced neurotoxicity and ototoxicity, and the protective agents. Food Chem. Toxicol. 2020, 136, 111079. [Google Scholar] [CrossRef] [PubMed]
- Waissbluth, S.; Peleva, E.; Daniel, S.J. Platinum-induced ototoxicity: A review of prevailing ototoxicity criteria. Eur. Arch. Otorhinolaryngol. 2017, 274, 1187–1196. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Womer, R.B.; Silber, J.H. Predicting cisplatin ototoxicity in children: The influence of age and the cumulative dose. Eur. J. Cancer 2004, 40, 2445–2451. [Google Scholar] [CrossRef] [PubMed]
- de Biasi, A.R.; Villena-Vargas, J.; Adusumilli, P.S. Cisplatin-induced antitumor immunomodulation: A review of preclinical and clinical evidence. Clin. Cancer Res. 2014, 20, 5384–5391. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.