Constrictive Pericarditis–A Cloak Camouflaging Lymphoma
Abstract
1. Introduction
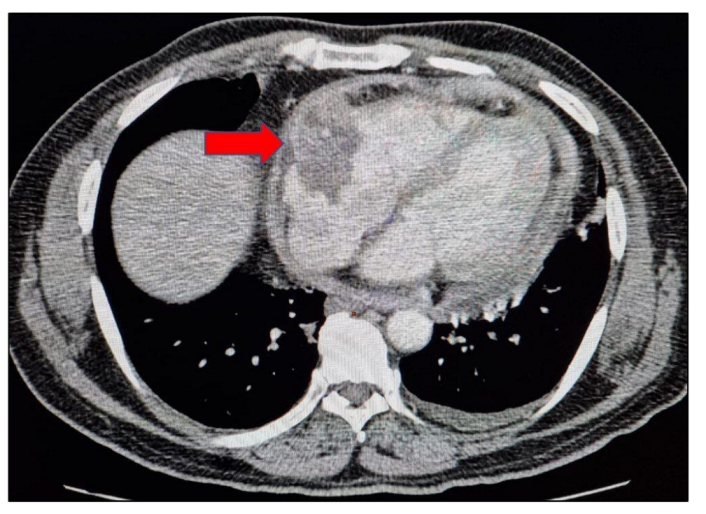
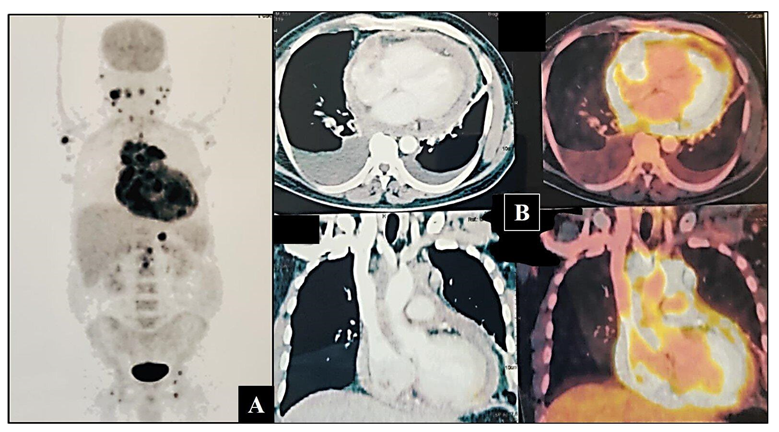
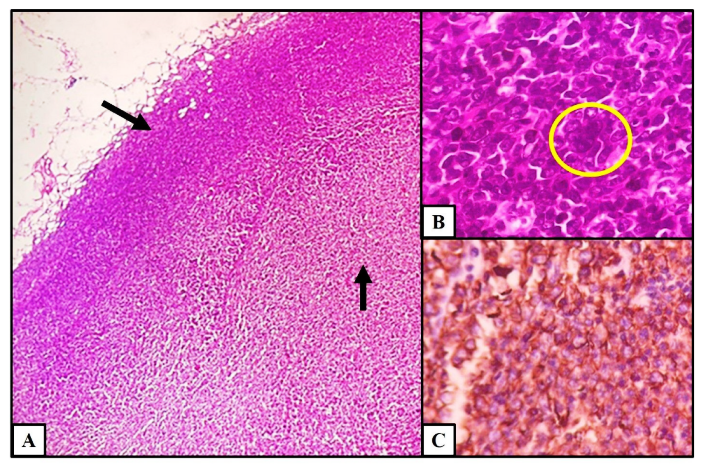
2. Case Presentation
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Travis, W.D.; Brambilla, E.; Burke, A.P.; Marx, A.; Nicholson, A.G. Introduction to the 2015 World Health Organization Classification of Tumors of the Lung, Pleura, Thymus, and Heart. J. Thorac. Oncol. 2015, 10, 1240–1242. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.W.; Chang, J.H.; Yeh, S.P.; Lu, C.R. Primary cardiac B-cell lymphoma with atrioventricular block and paroxysmal ventricular tachycardia. J. Cardiothorac. Surg. 2012, 7, 70. [Google Scholar] [CrossRef] [PubMed]
- Vitolo, U.; Seymour, J.F.; Martelli, M.; Illerhaus, G.; Illidge, T.; Zucca, E.; Campo, E.; Ladetto, M.; ESMO Guidelines Committee. Extranodal diffuse large B-cell lymphoma (DLBCL) and primary mediastinal B-cell lymphoma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2016, 27, v91–v102. [Google Scholar] [CrossRef] [PubMed]
- Boussios, S.; Zerdes, I.; Vassou, A.; Bareta, E.; Seraj, E.; Papoudou-Bai, A.; Pavlidis, N.; Batistatou, A.; Pentheroudakis, G. Extranodal diffuse large B-cell lymphomas: A retrospective case series and review of the literature. Hematol. Rep. 2018, 10, 7070. [Google Scholar] [CrossRef] [PubMed]
- Reynen, K. Cardiac myxomas. N. Engl. J. Med. 1995, 333, 1610–1617. [Google Scholar] [CrossRef] [PubMed]
- Zanelli, M.; Zizzo, M.; Montanaro, M.; Gomes, V.; Martino, G.; De Marco, L.; Fraternali Orcioni, G.; Martelli, M.P.; Ascani, S. Fibrin-associated large B-cell lymphoma: First case report within a cerebral artery aneurysm and literature review. BMC Cancer 2019, 19, 916. [Google Scholar] [CrossRef] [PubMed]
- Zanelli, M.; Zizzo, M.; De Marco, L.; Bisagni, A.; Ascani, S. Fibrin-associated diffuse large B-cell lymphoma. Br. J. Haematol. 2019, 185, 397. [Google Scholar] [CrossRef] [PubMed]
- Tapan, U.; Pestana, J.B.; Lee, J.C.; Lerner, A. Epstein-Barr virus-associated diffuse large B-cell lymphoma arising in atrial myxoma: A proposal for a modified therapeutic approach. Leuk. Lymphoma 2015, 56, 505–507. [Google Scholar] [CrossRef] [PubMed]
- Johri, A.; Baetz, T.; Isotalo, P.A.; Nolan, R.L.; Sanfilippo, A.J.; Ropchan, G. Primary cardiac diffuse large B cell lymphoma presenting with superior vena cava syndrome. Can. J. Cardiol. 2009, 25, e210–e212. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Qian, S.; Shi, P.; Liu, L.; Yang, F. A presentation, treatment, and survival analysis of primary cardiac lymphoma cases reported from 2009 to 2019. Int. J. Hematol. 2020, 112, 65–73. [Google Scholar] [CrossRef] [PubMed]
- Jeudy, J.; Burke, A.P.; Frazier, A.A. Cardiac Lymphoma. Radiol. Clin. N. Am. 2016, 54, 689–710. [Google Scholar] [CrossRef] [PubMed]
- Chia, A.X.F.; Zhao, Z.; Lim, S.L. Primary cardiac lymphoma. BMJ Case Rep. 2019, 12, e230468. [Google Scholar] [CrossRef] [PubMed]
- Petrich, A.; Cho, S.I.; Billett, H. Primary cardiac lymphoma: An analysis of presentation, treatment, and outcome patterns. Cancer 2011, 117, 581–589. [Google Scholar] [CrossRef] [PubMed]
- Nascimento, A.F.; Winters, G.L.; Pinkus, G.S. Primary cardiac lymphoma: Clinical, histologic, immunophenotypic, and genotypic features of 5 cases of a rare disorder. Am. J. Surg. Pathol. 2007, 31, 1344–1350. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Ren, C.; Wang, Y. Primary Lymphoma of the Heart: A Case Report of Surgical Treatment and Review of the Literature. Heart Surg. Forum 2019, 22, E225–E228. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.N.; Lai, C.H.; Lu, L.F.; Lin, H.H. Fever of unknown origin from a left atrial myxoma: An immunologic basis and cytokine association. South. Med. J. 2011, 104, 360–362. [Google Scholar] [CrossRef] [PubMed]
- Fukunaga, H.; Tatewaki, Y.; Mutoh, T.; Shimomura, H.; Yamamoto, S.; Terao, C.; Totsune, T.; Nakagawa, M.; Taki, Y. A Case of Low-Grade Primary Cardiac Lymphoma with Pericardial Effusion Diagnosed by Combined 18F-Fluorodeoxyglucose Positron Emission Tomography and Computed Tomography (FDG-PET/CT) Imaging and Effusion Cytology. Am. J. Case Rep. 2018, 19, 292–295. [Google Scholar] [CrossRef] [PubMed]
- Yoshihara, S.; Naito, M.; Tanioka, F.; Matsunaga, M. A case of primary cardiac lymphoma: In vivo imaging and pathologic correlation. Eur. Heart J. Cardiovasc. Imaging 2013, 14, 1027. [Google Scholar] [CrossRef] [PubMed]
- Colavolpe, C.; Ebbo, M.; Trousse, D.; Khibri, H.; Franques, J.; Chetaille, B.; Coso, D.; Ouvrier, M.J.; Gastaud, L.; Guedj, E.; et al. FDG-PET/CT is a pivotal imaging modality to diagnose rare intravascular large B-cell lymphoma: Case report and review of literature. Hematol. Oncol. 2015, 33, 99–109. [Google Scholar] [CrossRef] [PubMed]
- Chin, J.Y.; Chung, M.H.; Kim, J.J.; Lee, J.H.; Kim, J.H.; Maeng, I.H.; Jung, S.Y.; Hwang, H.J.; Lee, J.B.; Youn, H.J. Extensive primary cardiac lymphoma diagnosed by percutaneous endomyocardial biopsy. J. Cardiovasc. Ultrasound. 2009, 17, 141–144. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Venugopala, D.; Dsouza, N.V.; Acharya, V.; Rai, M.; Venkataramana, C.G.; Boussios, S. Constrictive Pericarditis–A Cloak Camouflaging Lymphoma. Hematol. Rep. 2023, 15, 166-171. https://doi.org/10.3390/hematolrep15010017
Venugopala D, Dsouza NV, Acharya V, Rai M, Venkataramana CG, Boussios S. Constrictive Pericarditis–A Cloak Camouflaging Lymphoma. Hematology Reports. 2023; 15(1):166-171. https://doi.org/10.3390/hematolrep15010017
Chicago/Turabian StyleVenugopala, Delanthabettu, Nikhil Victor Dsouza, Vishak Acharya, Maneesh Rai, Chaithra Gowthuvalli Venkataramana, and Stergios Boussios. 2023. "Constrictive Pericarditis–A Cloak Camouflaging Lymphoma" Hematology Reports 15, no. 1: 166-171. https://doi.org/10.3390/hematolrep15010017
APA StyleVenugopala, D., Dsouza, N. V., Acharya, V., Rai, M., Venkataramana, C. G., & Boussios, S. (2023). Constrictive Pericarditis–A Cloak Camouflaging Lymphoma. Hematology Reports, 15(1), 166-171. https://doi.org/10.3390/hematolrep15010017






