Rapid Quantification and Validation of Lipid Concentrations within Liposomes
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Manufacturing of Liposomes
2.3. HPLC Method
2.4. Standard Solutions
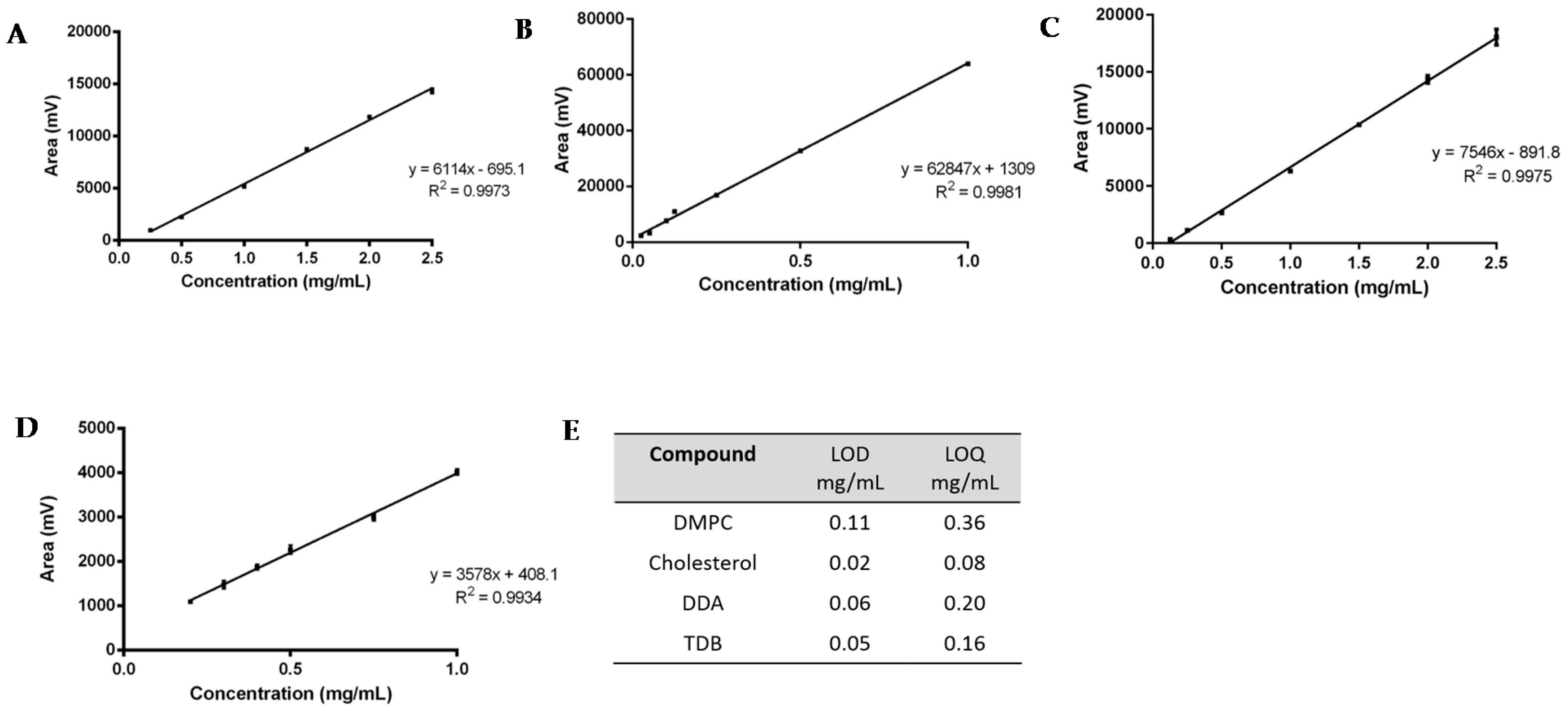
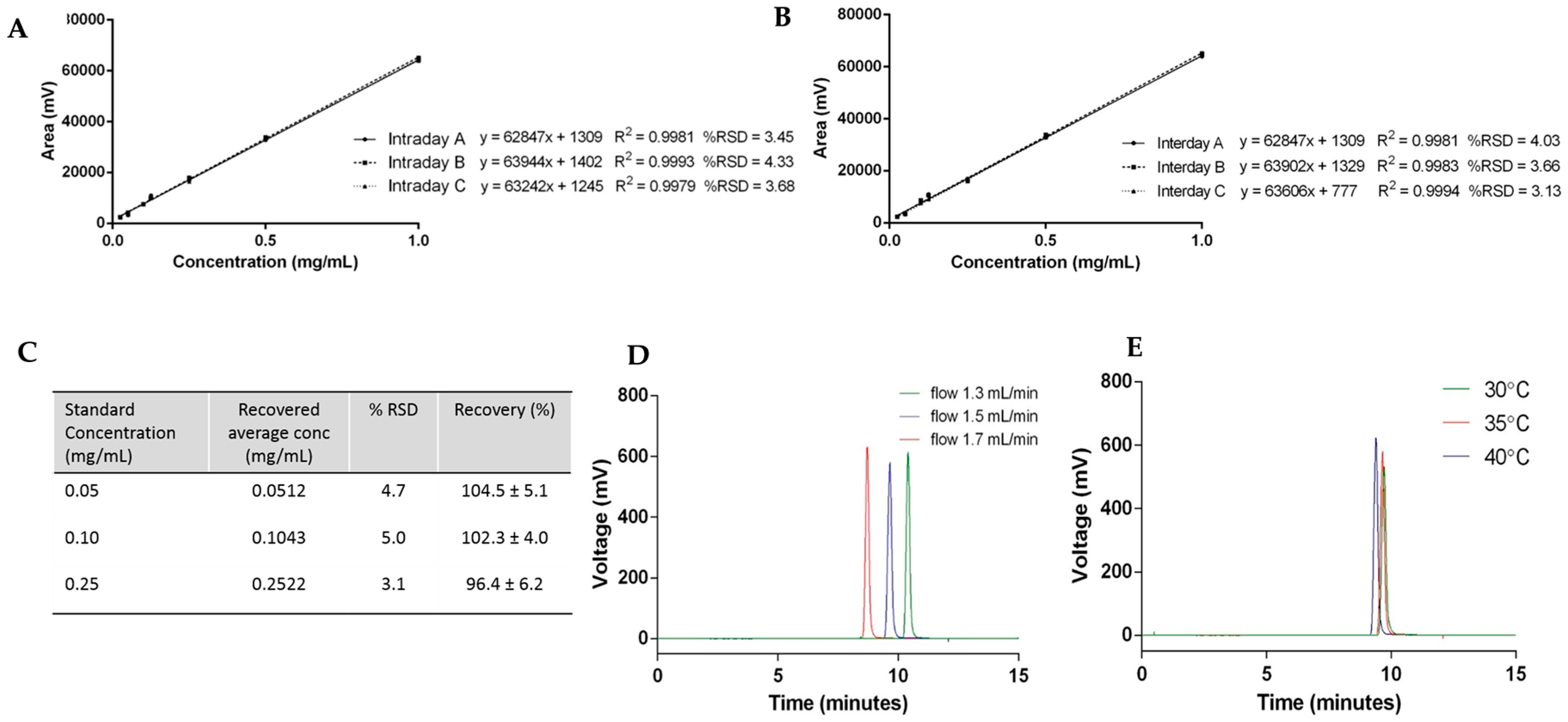
2.5. Method Validation
2.6. HPLC Lipid Degradation Method
3. Results and Discussion
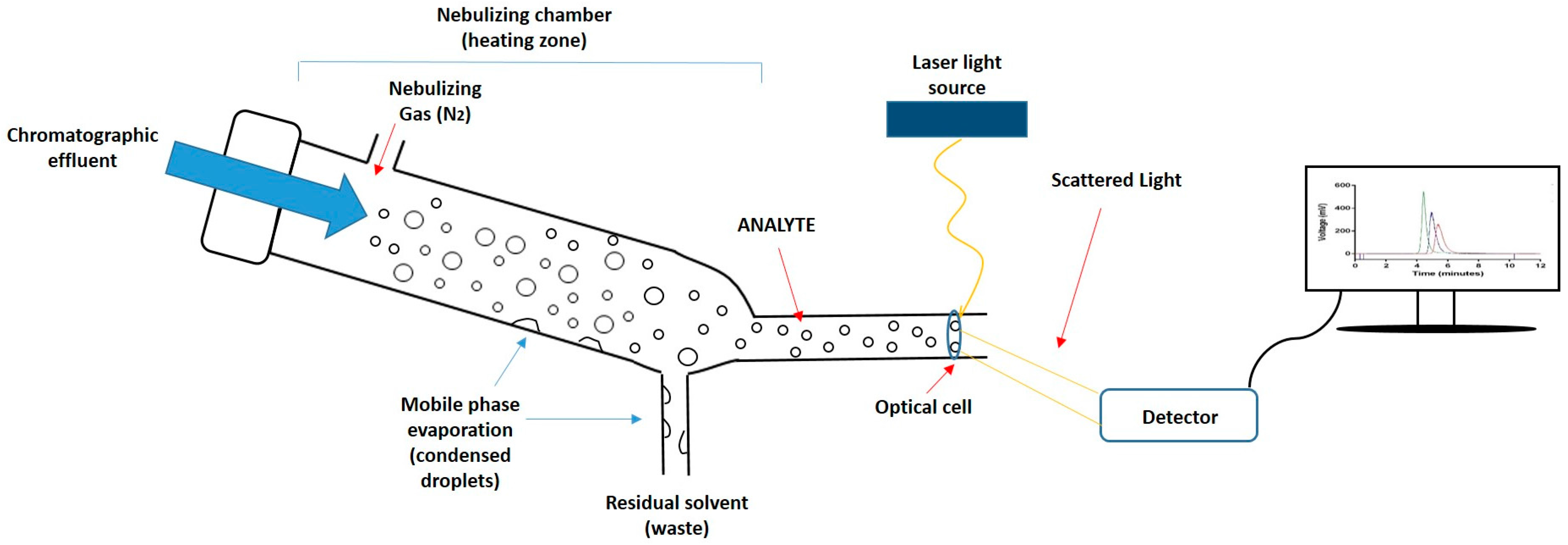
3.1. Lipid Quantification Using HPLC-ELSD: Method Development
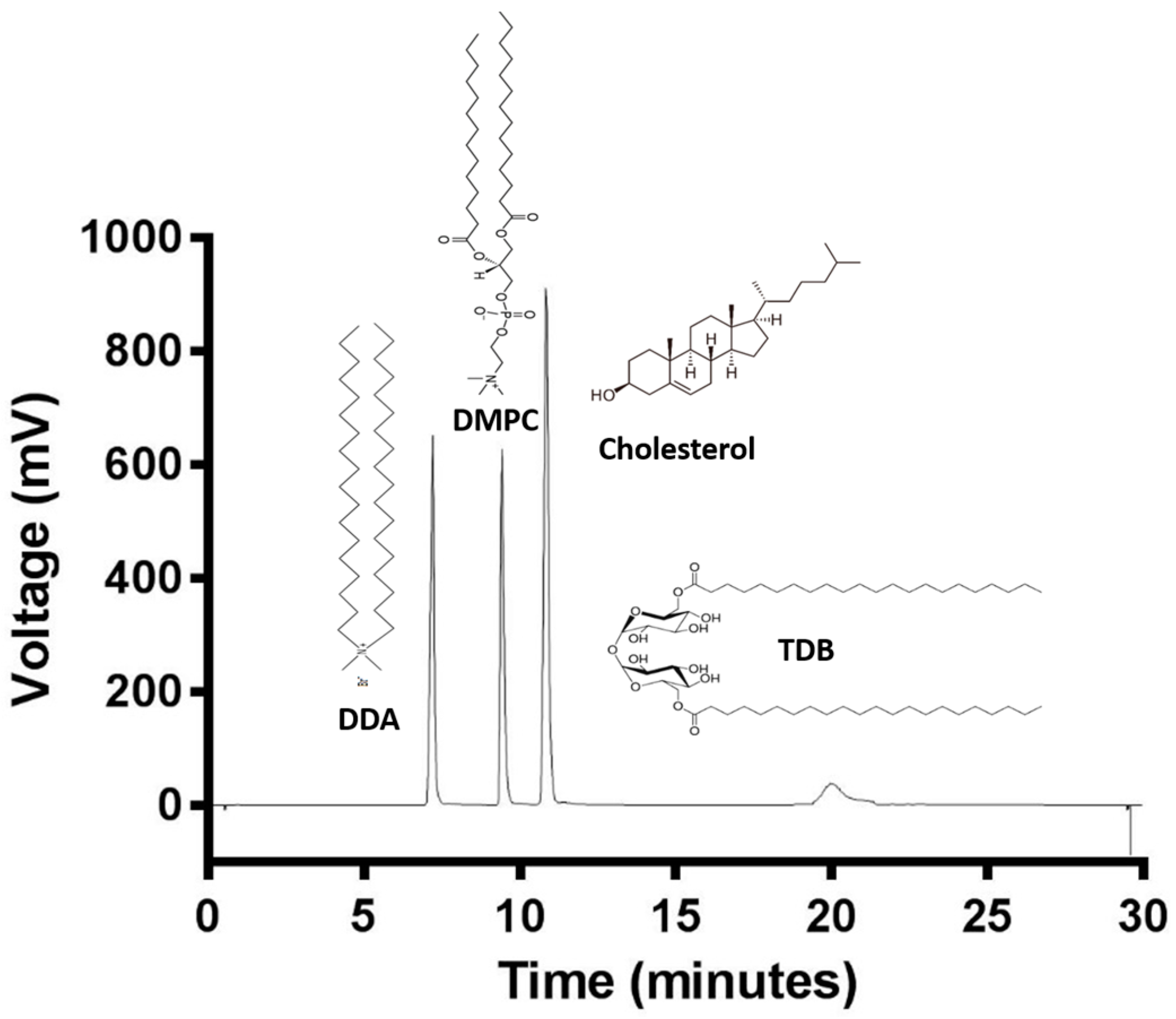
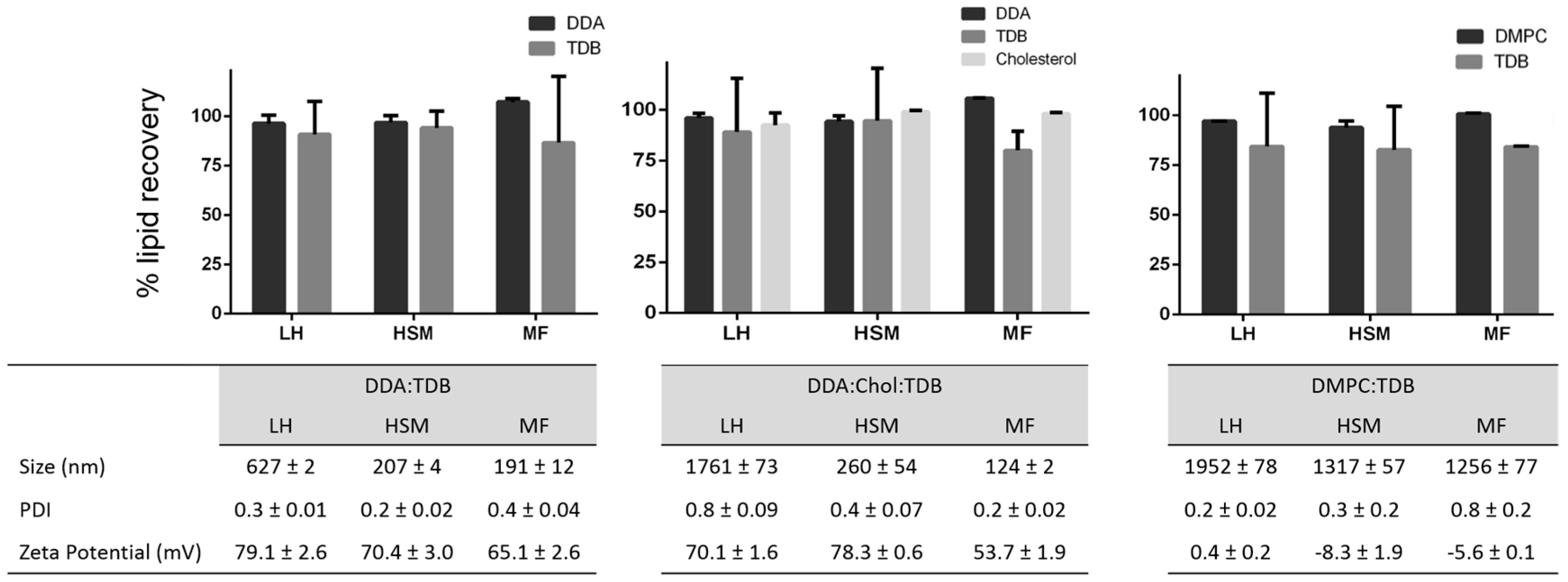
3.2. Quantification of Lipids within Liposomal Adjuvant Formulations
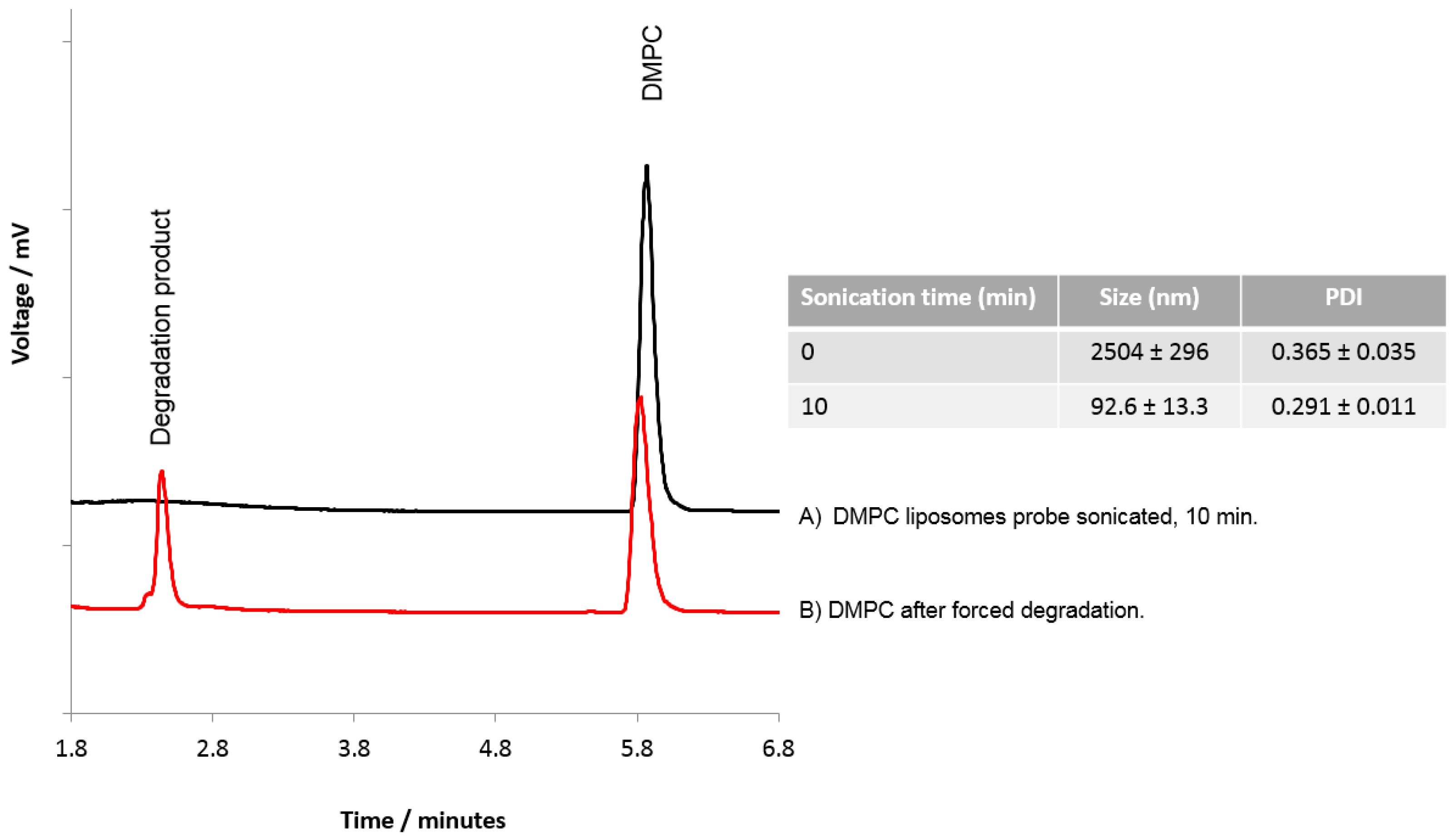
3.3. The Use of HPLC-ELSD to Assess Lipid Degradation in Liposome Formulations
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Supporting Information Available
References
- Gall, D. The adjuvant activity of aliphatic nitrogenous bases. Immunology 1966, 11, 369–386. [Google Scholar] [PubMed]
- Olds, G.R.; Chedid, L.; Lederer, E.; Mahmoud, A.A.F. Induction of resistance to schistosoma mansoni by natural cord factor and synthetic lower homologues. J. Infect. Dis. 1980, 141, 473–478. [Google Scholar] [CrossRef] [PubMed]
- Pimm, M.V.; Baldwin, R.W.; Polonsky, J.; Lederer, E. Immunotherapy of an ascitic rat hepatoma with cord factor (trehalose-6, 6′-dimycolate) and synthetic analogues. Int. J. Cancer 1979, 24, 780–785. [Google Scholar] [CrossRef] [PubMed]
- Klinguer, C.; Beck, A.; De-Lys, P.; Bussat, M.C.; Blaecke, A.; Derouet, F.; Bonnefoy, J.Y.; Nguyen, T.N.; Corvaı̈a, N.; Velin, D. Lipophilic quaternary ammonium salt acts as a mucosal adjuvant when co-administered by the nasal route with vaccine antigens. Vaccine 2001, 19, 4236–4244. [Google Scholar] [CrossRef]
- Vangala, A.; Bramwell, V.W.; McNeil, S.; Christensen, D.; Agger, E.M.; Perrie, Y. Comparison of vesicle based antigen delivery systems for delivery of hepatitis b surface antigen. J. Control. Release 2007, 119, 102–110. [Google Scholar] [CrossRef] [PubMed]
- Esposito, E.; Sebben, S.; Cortesi, R.; Menegatti, E.; Nastruzzi, C. Preparation and characterization of cationic microspheres for gene delivery. Int. J. Pharm. 1999, 189, 29–41. [Google Scholar] [CrossRef]
- Brandt, L.; Elhay, M.; Rosenkrands, I.; Lindblad, E.B.; Andersen, P. Esat-6 subunit vaccination against mycobacterium tuberculosis. Infect. Immun. 2000, 68, 791–795. [Google Scholar] [CrossRef] [PubMed]
- Holten-Andersen, L.; Doherty, T.M.; Korsholm, K.S.; Andersen, P. Combination of the cationic surfactant dimethyl dioctadecyl ammonium bromide and synthetic mycobacterial cord factor as an efficient adjuvant for tuberculosis subunit vaccines. Infect. Immun. 2004, 72, 1608–1617. [Google Scholar] [CrossRef] [PubMed]
- Rosenkrands, I.; Agger, E.M.; Olsen, A.W.; Korsholm, K.S.; Andersen, C.S.; Jensen, K.T.; Andersen, P. Cationic liposomes containing mycobacterial lipids: A new powerful th1 adjuvant system. Infect. Immun. 2005, 73, 5817–5826. [Google Scholar] [CrossRef] [PubMed]
- Davidsen, J.; Rosenkrands, I.; Christensen, D.; Vangala, A.; Kirby, D.; Perrie, Y.; Agger, E.M.; Andersen, P. Characterization of cationic liposomes based on dimethyldioctadecylammonium and synthetic cord factor from M. tuberculosis (trehalose 6,6′-dibehenate)—a novel adjuvant inducing both strong CMI and antibody responses. Biochim. Biophys. Acta 2005, 1718, 22–31. [Google Scholar] [CrossRef] [PubMed]
- Lesnefsky, E.J.; Stoll, M.S.K.; Minkler, P.E.; Hoppel, C.L. Separation and quantitation of phospholipids and lysophospholipids by high-performance liquid chromatography. Anal. Biochem. 2000, 285, 246–254. [Google Scholar] [CrossRef] [PubMed]
- Patton, G.M.; Fasulo, J.M.; Robins, S.J. Separation of phospholipids and individual molecular species of phospholipids by high-performance liquid chromatography. J. Lipid Res. 1982, 23, 190–196. [Google Scholar] [PubMed]
- Holland, W.L.; Stauter, E.C.; Stith, B.J. Quantification of phosphatidic acid and lysophosphatidic acid by HPLC with evaporative light-scattering detection. J. Lipid Res. 2003, 44, 854–858. [Google Scholar] [CrossRef] [PubMed]
- Fagan, P.; Wijesundera, C. Liquid chromatographic analysis of milk phospholipids with on-line pre-concentration. J. Chromatogr. 2004, 1054, 241–249. [Google Scholar] [CrossRef]
- Lin, J.T.; McKeon, T.A. Separation of intact phosphatidylcholine molecular species by high performance liquid chromatography. J. Liq. Chromatogr. Relat. Technol. 2000, 23, 813–829. [Google Scholar] [CrossRef]
- Mazzella, N.; Molinet, J.; Syakti, A.D.; Dodi, A.; Doumenq, P.; Artaud, J.; Bertrand, J.-C. Bacterial phospholipid molecular species analysis by ion-pair reversed-phase HPLC/ESI/MS. J. Lipid Res. 2004, 45, 1355–1363. [Google Scholar] [CrossRef] [PubMed]
- Christie, W.W. Rapid separation and quantification of lipid classes by high performance liquid chromatography and mass (light-scattering) detection. J. Lipid Res. 1985, 26, 507–512. [Google Scholar] [PubMed]
- Lasic, D.D.; Barenholz, Y. Handbook of Nonmedical Applications of Liposomes; CRC Press: Boca Raton, FL, USA, 1996. [Google Scholar]
- Bharath, S. Pharmaceutical Technology: Concepts and Applications; Pearson Education India: Noida, India, 2013. [Google Scholar]
- Mills, J.K.; Needham, D. Lysolipid incorporation in dipalmitoylphosphatidylcholine bilayer membranes enhances the ion permeability and drug release rates at the membrane phase transition. Biochim. Biophys. Acta 2005, 1716, 77–96. [Google Scholar] [CrossRef] [PubMed]
- Crowe, J.H.; McKersie, B.D.; Crowe, L.M. Effects of free fatty acids and transition temperature on the stability of dry liposomes. Biochim. Biophys. Acta 1989, 979, 7–10. [Google Scholar] [CrossRef]
- Toh, M.-R.; Chiu, G.N.C. Liposomes as sterile preparations and limitations of sterilisation techniques in liposomal manufacturing. Asian J. Pharm. Sci. 2013, 8, 88–95. [Google Scholar] [CrossRef]
- Gabrielska, J.; Sarapuk, J.; Przestalski, S. Antioxidant protection of egg lecithin liposomes during sonication. Z. Naturforsch. 1995, 50, 561–564. [Google Scholar]
- Samuni, A.M.; Lipman, A.; Barenholz, Y. Damage to liposomal lipids: Protection by antioxidants and cholesterol-mediated dehydration. Chem. Phys. Lipids 2000, 105, 121–134. [Google Scholar] [CrossRef]
- Reis, A.; Spickett, C.M. Chemistry of phospholipid oxidation. Biochim. Biophys. Acta 2012, 1818, 2374–2387. [Google Scholar] [CrossRef] [PubMed]
- Bangham, A.; Standish, M.M.; Watkins, J. Diffusion of univalent ions across the lamellae of swollen phospholipids. J. Mol. Biol. 1965, 13, IN26–252–IN27. [Google Scholar] [CrossRef]
- Andreasen, L.V.; Wood, G.K.; Christensen, D. Methods for Producing Liposomes. WO2013004234 A2, 10 January 2013. [Google Scholar]
- Kastner, E.; Verma, V.; Lowry, D.; Perrie, Y. Microfluidic-controlled manufacture of liposomes for the solubilisation of a poorly water soluble drug. Int. J. Pharm. 2015, 485, 122–130. [Google Scholar] [CrossRef] [PubMed]
- Zhong, Z.; Ji, Q.; Zhang, J.A. Analysis of Cationic Liposomes by Reversed-Phase HPLC with Evaporative Light-Scattering Detection. J. Pharm. Biomed. Anal. 2010, 51, 947–951. [Google Scholar] [CrossRef] [PubMed]
- US, F. Q2 (R1) validation of analytical procedures: Text and methodology. Available online: http://www.fda.gov/Drugs/GuidanceComplianceRegulatoryInformation/Guidances/ucm265700.htm (accessed on 2 September 2016).
- Jahn, A.; Reiner, J.E.; Vreeland, W.N.; DeVoe, D.L.; Locascio, L.E.; Gaitan, M. Preparation of nanoparticles by continuous-flow microfluidics. J. Nanoparticle Res. 2008, 10, 925–934. [Google Scholar] [CrossRef]
- Belliveau, N.M.; Huft, J.; Lin, P.J.C.; Chen, S.; Leung, A.K.K.; Leaver, T.J.; Wild, A.W.; Lee, J.B.; Taylor, R.J.; Tam, Y.K.; et al. Microfluidic synthesis of highly potent limit-size lipid nanoparticles for in vivo delivery of sirna. Mol. Ther. Nucleic Acids 2012, 1, e37. [Google Scholar] [CrossRef] [PubMed]
- Zhigaltsev, I.V.; Belliveau, N.; Hafez, I.; Leung, A.K.K.; Huft, J.; Hansen, C.; Cullis, P.R. Bottom-up design and synthesis of limit size lipid nanoparticle systems with aqueous and triglyceride cores using millisecond microfluidic mixing. Langmuir 2012, 28, 3633–3640. [Google Scholar] [CrossRef] [PubMed]
- Grit, M.; de Smidt, J.H.; Struijke, A.; Crommelin, D.J.A. Hydrolysis of phosphatidylcholine in aqueous liposome dispersions. Int. J. Pharm. 1989, 50, 1–6. [Google Scholar] [CrossRef]
- Almog, R.; Forward, R.; Samsonoff, C. Stability of sonicated aqueous suspensions of phospholipids under air. Chem. Phys. Lipids 1991, 60, 93–99. [Google Scholar] [CrossRef]
| Product Name | Drug Delivered | Lipid Composition |
|---|---|---|
| AmBisome® | Amphotericin B | Hydrogenated soy phosphatidylcholine, cholesterol, disteraroylphosphatidyl glycerol, and α tocopherol |
| Caelyx®/Doxil® | Doxorubicin | PEGylated distearoylphosphatidyl ethanolamine, hydrogenated soy phosphatidylcholine, and cholesterol |
| DaunoXome® | Daunorubicin | Distearylphosphatidylcholine and cholesterol |
| Definity® | Octafluoropropane | Dipalmitoylphosphatidic acid, dipalmitoylphosphatidylcholine, and PEG-500 dipalmitoyl phosphatidyletholamine |
| DepoCyte® | Cytarabine | Dioleoylphosphatidylcholine, cholesterol, dipalmitoylphosphatidylglycerol, and triolein |
| DepoDur® | Morphine sulfate | Dioleoyl phosphatidylcholine, dipalmitoyl phosphatidylglycerol, cholesterol, and triolein |
| Epaxal® | Inactivated hepatitis A virus | Phosphatidylcholine, cephalin, and purified virus surface antigens |
| Inflexal V® | Influenza haemagglutinin glycoprotein and neuraminidase | Similar to Epaxal; phosphatidylcholine, cephalin, and purified virus surface antigens |
| Myocet® | Doxorubicin | Egg phosphatidylcholine and cholesterol |
| Visudyne® | Vereporfin | Dimyristoylphosphatidylcholine and egg phosphatidylglycerol |
| Time (min) | % Eluent A (0.1% TFA in dH2O) | % Eluent B (MeOH) | Flow Rate (mL/min) |
|---|---|---|---|
| 0 | 15 | 85 | 1.5 |
| 6 | 0 | 100 | 1.5 |
| 25 | 0 | 100 | 1.5 |
| 26 | 15 | 85 | 1.5 |
| 35 | 15 | 85 | 1.5 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Roces, C.B.; Kastner, E.; Stone, P.; Lowry, D.; Perrie, Y. Rapid Quantification and Validation of Lipid Concentrations within Liposomes. Pharmaceutics 2016, 8, 29. https://doi.org/10.3390/pharmaceutics8030029
Roces CB, Kastner E, Stone P, Lowry D, Perrie Y. Rapid Quantification and Validation of Lipid Concentrations within Liposomes. Pharmaceutics. 2016; 8(3):29. https://doi.org/10.3390/pharmaceutics8030029
Chicago/Turabian StyleRoces, Carla B., Elisabeth Kastner, Peter Stone, Deborah Lowry, and Yvonne Perrie. 2016. "Rapid Quantification and Validation of Lipid Concentrations within Liposomes" Pharmaceutics 8, no. 3: 29. https://doi.org/10.3390/pharmaceutics8030029
APA StyleRoces, C. B., Kastner, E., Stone, P., Lowry, D., & Perrie, Y. (2016). Rapid Quantification and Validation of Lipid Concentrations within Liposomes. Pharmaceutics, 8(3), 29. https://doi.org/10.3390/pharmaceutics8030029







