Abstract
Background/Objectives: The aztreonam/avibactam combination represents a promising therapeutic option for severe infections caused by multidrug-resistant Gram-negative pathogens, particularly in critically ill patients. Due to marked pharmacokinetic variability and the need to achieve joint pharmacokinetic/pharmacodynamic (PK/PD) targets of both agents, therapeutic drug monitoring (TDM) may play a pivotal role in optimizing treatment. This study aimed to develop and validate two rapid, accurate, and sensitive UHPLC–qTOF MS/MS sequential methods for quantifying aztreonam and avibactam in human plasma, suitable for routine clinical TDM. Methods: Plasma concentrations were determined by means of ultra-high-performance liquid chromatography coupled with quadrupole time-of-flight tandem mass spectrometry (UHPLC–qTOF MS/MS), operating in positive and negative electrospray ionization modes for aztreonam and avibactam, respectively. Sample preparation consisted of protein precipitation with isotopically labeled internal standards. The method’s validation was performed according to the European Medicines Agency guidelines, by assessing selectivity, linearity, precision, accuracy, recovery, matrix effects, carry-over, and stability. Clinical applicability was evaluated by reprocessing plasma samples, which were already previously collected for routine clinical practice from 20 hospitalized patients undergoing treatment with ceftazidime-avibactam plus aztreonam. Results: The methods showed excellent linearity (R2 ≥ 0.999) over ranges of 0.2–100 µg/mL for aztreonam and 0.1–50 µg/mL for avibactam. Lower limits of quantification were 0.2 µg/mL and 0.1 µg/mL, respectively. Intra- and inter-day precision and accuracy met the EMA criteria at all of the quality control levels. Extraction recovery exceeded 90% for both analytes, and matrix effects were effectively compensated by internal standards. Stability testing highlighted the need for careful sample handling, particularly for aztreonam under repeated freeze–thaw conditions. Clinical application revealed substantial inter-individual variability in steady-state concentrations. Conclusions: The validated UHPLC–qTOF MS/MS assays provide robust and sensitive sequential quantification of aztreonam and avibactam in human plasma, supporting TDM-guided dose optimization in clinical practice.
1. Introduction
Infections caused by carbapenem-resistant Enterobacterales (CPE) are an ever-growing major concern in several settings of critically ill patients [1,2]. Among CPE, those producing metallo-beta lactamases (MBLs) are particularly worrisome since the number of therapeutically effective weapons against MBL-related infections is quite limited [3,4]. In the recent past, international guidance and guidelines recommended combining aztreonam with ceftazidime-avibactam as one of the two first choices, together with cefiderocol, for treating MBL-producing Enterobacterales infections [5,6]. Specifically, aztreonam is inherently stable by itself to the MBLs, but unfortunately, it is susceptible to hydrolysis by serine β-lactamases, which are frequently co-produced by MBL-producing Gram-negative bacilli. To overcome this limitation, initially, aztreonam was extemporaneously combined with ceftazidime/avibactam to protect it from serine β-lactamase-mediated degradation, thereby restoring activity against MBL and serine β-lactamase co-producing Enterobacterales [7,8]. Recently, a new 3:1 fixed beta lactam/beta lactamase inhibitor combination (BL/BLIc) of aztreonam/avibactam was approved for this purpose and is currently being used in settings with high prevalence of these types of severe infections, occurring mainly in critically ill patients [9].
Aztreonam exhibits linear pharmacokinetics, moderate plasma protein binding, and is predominantly eliminated unchanged by the kidneys. Likewise, avibactam displays linear pharmacokinetics, low protein binding, and predominant renal elimination. Importantly, attaining a joint pharmacokinetic/pharmacodynamic (PK/PD) target is fundamental for optimal treatment with the aztreonam/avibactam combination. It has been shown that efficacy is granted whenever free aztreonam concentrations persisting for 60% of the dosing interval above the minimum inhibitory concentration (60%fT > MIC) are coupled with free avibactam concentrations persisting for 50% of the dosing interval above a target concentration of 2.5 mg/L (50%fCT > 2.5 mg/L) [10,11,12,13,14]. Unfortunately, like with other types of BL/BLIc, aztreonam/avibactam is also prone to high inter- and intra-individual pharmacokinetic variability in the critically ill patients. Factors such as sepsis-associated tissue capillary-leakage and/or transient acute kidney injury and/or augmented renal clearance may affect both the volume of distribution and the renal clearance of either aztreonam or avibactam [13,15]. This may be especially relevant in affecting an optimal joint PK/PD target attainment because, being the renal clearance of avibactam much faster than that of aztreonam, the 3:1 proportion of the aztreonam/avibactam concentrations present in the vial may vary in the critically ill patients as a consequence of the aforementioned conditions [16]. Consequently, this may lead to subtherapeutic or potentially toxic plasma concentrations, thus compromising efficacy or safety when standard dosing regimens are used. In this context, therapeutic drug monitoring (TDM) may be a valuable clinical tool for personalizing antibiotic therapy with the intent of improving the likelihood of optimal joint PK/PD target attainment and consequently the clinical outcome [17,18,19]. In this regard, implementing robust and reliable analytical methods capable of accurately quantifying both aztreonam and avibactam in biological matrices is an absolute pre-requisite for properly dealing with real-time TDM-guided dosing adaptation of aztreonam/avibactam in critically ill patients. Liquid chromatography coupled with tandem mass spectrometry (LC-MS/MS) is currently the gold standard approach for measuring antibiotics in plasma or serum, owing to its high sensitivity, specificity, and analytical robustness [20,21,22,23]. Some LC-MS/MS and UHPLC-MS/MS methods have been reported for the simultaneous determination of multiple β-lactam antibiotics, including aztreonam and avibactam, in human plasma, and have been successfully applied in the context of clinical TDM [24,25]. These methods generally comply with international bioanalytical validation guidelines and may allow for rapid and reliable quantification in routine laboratory practice. However, methods for quantifying aztreonam/avibactam in human plasma by means of high-resolution mass spectrometry platforms, namely UHPLC–quadrupole time-of-flight (qTOF) MS/MS, are still lacking. HRMS-based approaches may have several advantages compared to the traditional mass spectrometry techniques, namely high mass accuracy, enhanced selectivity and sensitivity, and the possibility of retrospectively enquiring the system about data just previously acquired, all of which may be especially valuable in complex clinical matrices and in the context of multi-analyte or exploratory analyses [26]. The aim of this study was to describe the development and validation of two rapid UHPLC–qTOF MS/MS sequential methods for the accurate and precise quantification of aztreonam/avibactam in human plasma. The proposed methods are intended to support real-time TDM in clinical practice, providing a reliable analytical tool for dose individualization and optimization of aztreonam/avibactam therapy.
2. Materials and Methods
2.1. Chemicals and Reagents
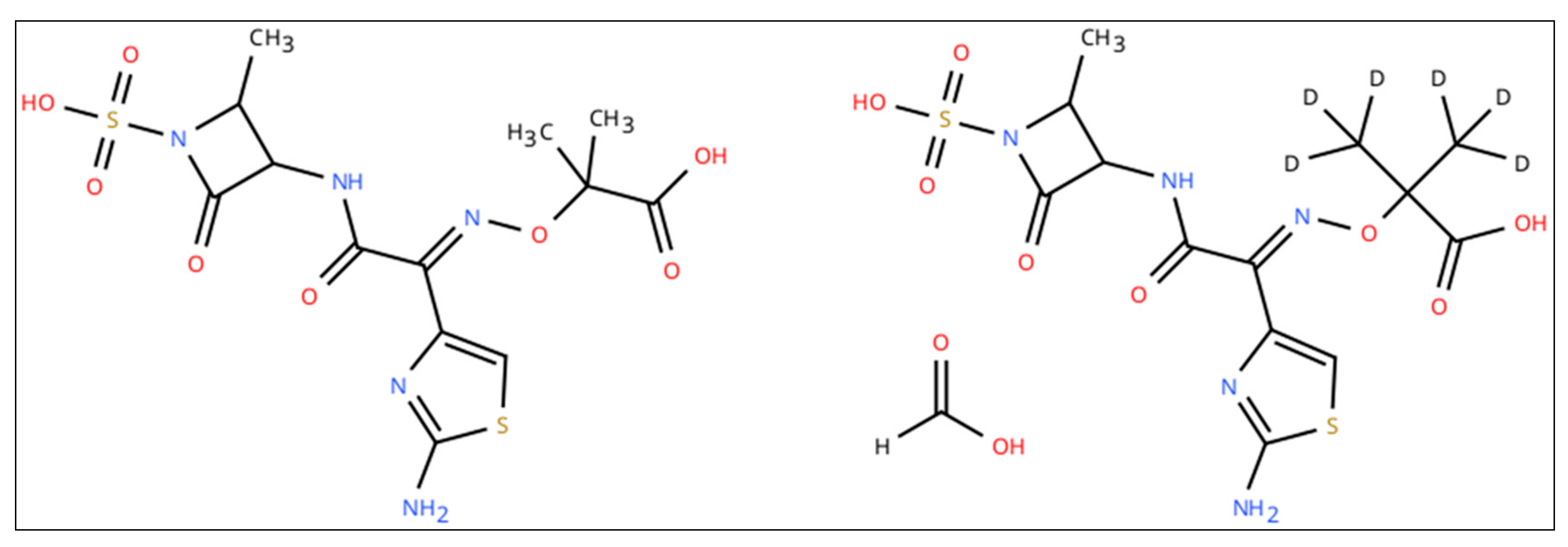
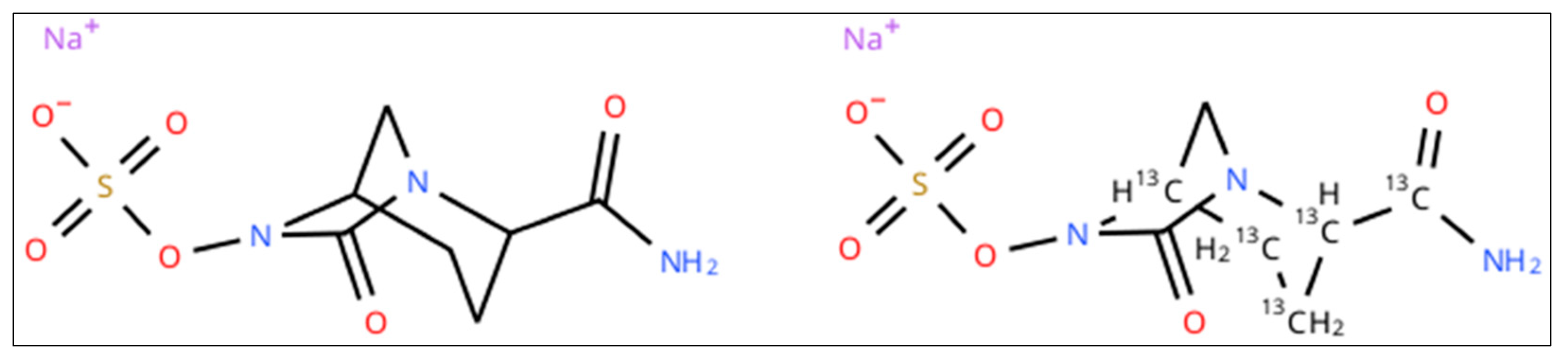
Lyophilized standard of aztreonam, [13C5]-avibactam sodium salt and [2H6]-aztreonam formate salt (chemical structures shown in Figure 1 and Figure 2) were purchased from Alsachim Shimadzu Chemistry & Diagnostics (Illkirch, France). Avibactam powder (Figure 1) and drug-free human plasma were provided by Merck KgaA (Darmstadt, Germany). Liquid chromatography–MS/MS-grade reagents were purchased from Thermo-Fisher Scientific (Milan, Italy).
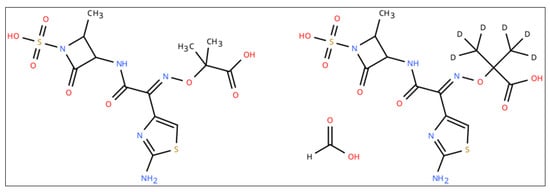
Figure 1.
Chemical structure of aztreonam (left) and [2H6]-aztreonam formate salt (right).
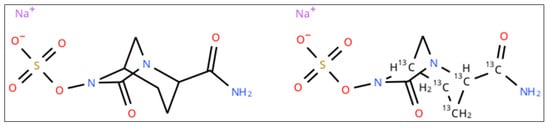
Figure 2.
Chemical structure of avibactam (left) and [13C5]-avibactam sodium salt (right).
2.2. Stock Solutions, Calibrators and Quality Control Samples
Aztreonam and avibactam stock solutions at a concentration of 1 mg/mL and 5 mg/mL, respectively, were prepared by dissolving the analyte powders in DMSO. Solutions of calibrators for the calibration curves and of independent quality controls (QCs) were prepared in MilliQ water (Sigma-Aldrich, Merck, Darmstadt, Germany) at different concentrations as serial dilutions from the respective stock solution, by spiking drug-free plasma in each of them. Calibration ranges covered from 0.2 to 100 μg/mL (calibration points: 0.2–1–5–10–25–50–100 μg/mL) for aztreonam and from 0.1 to 50 μg/mL (calibration points: 0.1–1–2.5–5–10–25–50 μg/mL) for avibactam. QC samples were prepared at the concentrations of 2.5 μg/mL (LQC), 20 μg/mL (MQC) and 80 μg/mL (HQC) for aztreonam and 1.5 μg/mL (LQC), 8 μg/mL (MQC) and 40 μg/mL (HQC) for avibactam. The dynamic ranges of the calibration curves were based on the expected therapeutic plasma concentrations of the analytes in critically ill patients receiving treatment by extended infusion over 3 h. A mixed solution containing 1 μg/mL of [2H6]-aztreonam formate salt plus 1 μg/mL of [13C5]-avibactam in methanol was used as internal standard (IS). All solvents and matrix solutions were stored at −80 °C.
2.3. Sample Pre-Treatment
For sample preparation, 10 µL of human plasma diluted with 80 µL of ultrapure water was added to 300 µL of the methanol internal standard (IS) solution. The mixture was kept at room temperature, vortexed for 15 s and then centrifuged at 13,000 rpm for 5 min. After centrifugation, 100 µL of the clear supernatant was transferred to an autosampler vial, and a 2 µL aliquot was injected into the LC–MS/MS system.
2.4. Instrumentation and Analysis
Samples were analyzed by means of a Shimadzu Nexera U-HPLC system equipped with binary pumps for mobile-phase delivery and coupled with a Quadrupole Time-of-Flight mass spectrometer X500 QTOF (AB SCIEX, Marlborough, MA, USA). Data were acquired in Multiple Reaction Monitoring (MRM) mode, with electrospray ionization (ESI) operating in the positive mode for the analysis of aztreonam and in the negative mode for that of avibactam. Separation was performed by means of an Agilent Poroshell 120 EC-C18 (2.1 × 50 mm, 1.9 µm) column for aztreonam and a Poroshell 120 PFP (2.1 × 50 mm, 2.7 µm) column for avibactam, ensuring the elution of the analytes at a specific and reproducible retention time. Both columns were kept at 45 °C and coupled with an autosampler kept at 4 °C. Analyte separation was achieved by using a binary pump system with a linear gradient elution from mobile-phase A [water with 0.2% formic acid, v/v] to mobile-phase B [methanol/acetonitrile 50:50 with 0.2% formic acid, v/v] at a flow rate of 0.5 mL/min (Table 1 and Table 2).

Table 1.
UHPLC method for aztreonam by means of the 120 EC-C18 column.

Table 2.
UHPLC method for avibactam by means of the 120 PFP column.
Data acquisition was carried out in MRM mode, with electrospray ionization (ESI) operating in positive or in negative mode, as needed. The MS/MS parameters for positive polarization and for negative polarization are summarized in Table 3 and Table 4, respectively. Chromatographic data acquisition, peak integration, and quantification were performed by means of the Sciex OS 3.1.6 software (AB SCIEX, Marlborough, MA, USA).

Table 3.
MS/MS parameters for positive polarization.

Table 4.
MS/MS parameters for negative polarization.
2.5. Methods Validation
The two methods were validated in accordance with the European Medicines Agency (EMA) guidelines for bioanalytical method validation [27]. Validation parameters included selectivity, linearity, accuracy, precision, lower limit of quantification (LLOQ), recovery, matrix effects and stability.
2.5.1. Selectivity and Carry-Over
Twenty plasma samples were tested to check for the absence of both the analytes at the retention times and the deuterated IS of any response attributable to endogenous or exogenous components of the matrix. The carry-over was assessed by injecting a blank sample immediately after finishing the highest calibrator run (ULoQ), and it was considered negligible whenever a signal intensity < 20% of that of the LLOQ was detected, as recommended by the EMA guidelines [27].
2.5.2. Linearity and Limit of Quantification (LoQ)
Calibration curves were constructed by means of weighted (1/x) linear regression. Linearity assessment was based on back-calculated concentrations of the calibration points deviating maximum ±15% from the nominal values (±20% at the LLOQ), in agreement with the ICH M10 guidelines. Acceptance of calibration curve linearity was verified with correlation coefficients (R2) ≥ 0.999. The limit of quantification (LoQ) was defined as the lowest calibrator within the dynamic range (namely 0.2 µg/mL for aztreonam and 0.1 µg/mL for avibactam) exhibiting a signal-to-noise ratio (S/N) > 10.
2.5.3. Precision and Accuracy
Precision (expressed as mean CV%) and accuracy (expressed as mean BIAS%) were established by analyzing the LLOQ, the LQC, the MQC, and the HQC five times within the same day (intra-day) across three separate inter-day analytical sessions.
2.5.4. Matrix Effect and Extraction Recovery
The matrix effect (ME) and the extraction recovery (ER) were assessed at the three quality control (QC) levels (namely, LQC, MQC, and HQC). The ME was assessed quantitatively by comparing plasma samples spiked with aztreonam and avibactam to water samples spiked with the same analytes. The analyte ratios obtained from plasma and water were compared, and the mean values and coefficients of variation (CVs) were calculated across six independent batches. The ER was determined by comparing plasma extracts spiked with both the analytes and the IS with those spiked with only the IS and expressed as a percentage. The compensated ME was calculated as the analyte-to-IS area ratio, and to be considered negligible, it had to be <15%.
2.5.5. Stability
The stability of aztreonam and of avibactam in human plasma was assessed at three concentration levels within the calibration range (LQC, MQC, and HQC) under different storage conditions. In accordance with laboratory needs and routine practice, the tests assessed the stability of the extracts stored at 4 °C for 24 h and for 5 days, as well as that of the matrix samples after three complete freeze–thaw cycles from −80 °C to 25 °C. Measured sample concentrations before and after storage were compared with nominal values, and the stability was defined as acceptable if the effective concentrations were within ±15% of the nominal concentrations.
2.5.6. Application to Clinical Samples
The developed LC–MS/MS sequential methods were validated by measuring the plasma concentrations of aztreonam/avibactam in reprocessed samples which were already previously collected for routine clinical practice from 20 hospitalized patients undergoing treatment with ceftazidime-avibactam plus aztreonam according to the retrospective observational study approved by the Ethics Committee of the IRCCS Azienda Ospedaliero-Universitaria di Bologna (No. 442/2021/Oss/AOUBo on 22 June 2021, EM232–2022/Oss/AOUBo on 16 March 2022, and EM449–2023/Oss/AOUBo on 17 April 2024). Informed written consent was waived due to the retrospective and observational nature of the study, according to the article 110-bis comma 4 of Legislative Decree No. 196/2003 rev. 2024 of Italian Law regulating retrospective study.
3. Results
3.1. Optimization of Analytical Conditions
Single-charge positive and negative ion mass transitions were selected for optimal sensitivity and specificity by scrutinizing the MS/MS fragmentation pattern spectra of the analytes and by comparing them with those reported in the literature [21,23,28]. The specific MRM transition parameters are summarized in Table 5.

Table 5.
Specific multiple reaction monitoring (MRM) transition parameters used for aztreonam, avibactam, and their respective internal standards.
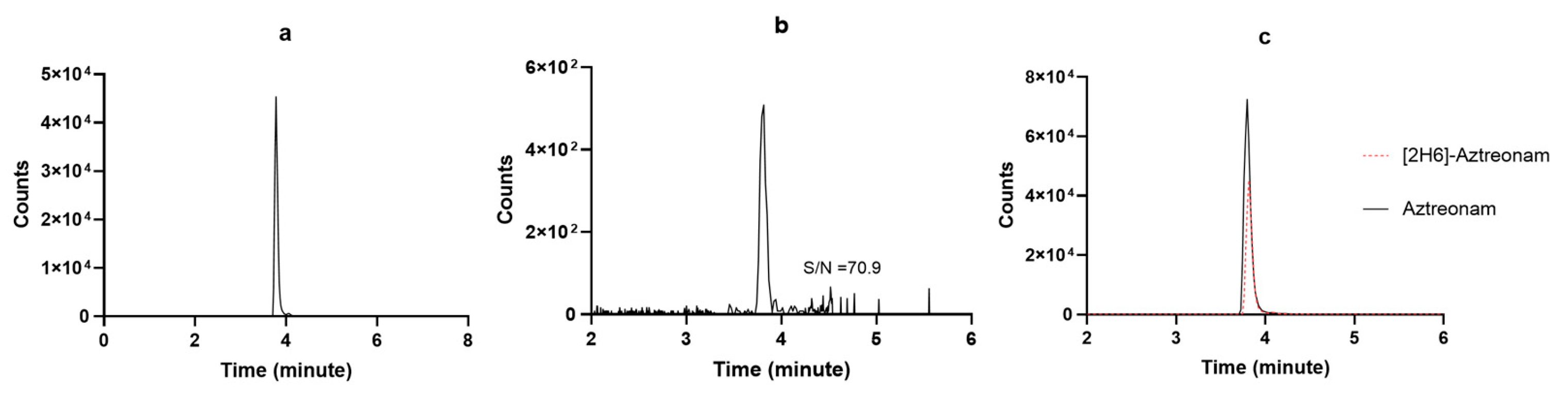
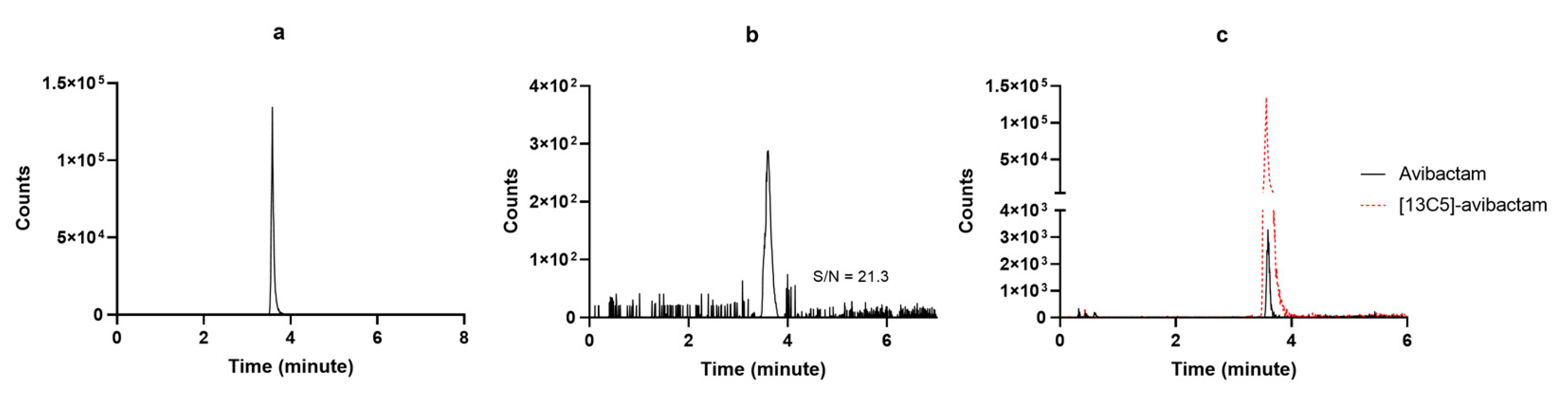
Method development started by testing preliminarily if the chromatographic conditions available in our lab for measuring other BL/BLIc could have fit for our purpose. Since this was not the case, we applied several adaptations and the final chromatographic conditions were definitely selected once reaching high-resolution MS detection based on adequate retention times, peak symmetry, signal intensity, and reproducibility under gradient conditions. The chosen stationary phases provided the best overall balance between chromatographic performance and analytical robustness for each analyte within the selected instrumental configuration. The selected LC elution conditions were good enough for granting high quality and sharp shape of the chromatographic peaks, as shown in Figure 3 and Figure 4. The retention times were constant and reproducible, thus supporting an optimal inter-run column reconditioning. The MRM chromatograms obtained by analyzing a drug-free plasma sample (blank), detecting only the specific signal of [2H6]-aztreonam formate salt and of [13C5]-avibactam, are depicted in Figure 3a and Figure 4a, respectively. This confirms both the high specificity of the MRM transitions and the purity of the IS solutions. The MRM chromatograms obtained by analyzing the LLOQ samples of aztreonam and avibactam are depicted in Figure 3b and Figure 4b, respectively. The S/N ratios were high (70.9 and 21.3, respectively), thus confirming the high sensitivity of the methods. Real sample MRM chromatograms of aztreonam and avibactam (Figure 3c and Figure 4c) show that for both peaks, shapes were sharp and the resolutions were optimal, with no isobaric interference coming from interferences generated by endogenous components and/or by any other drug. Noteworthy, the IS peak areas almost overlapped with those of the analytes concentrations expected in patients.
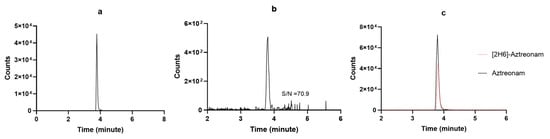
Figure 3.
MRM chromatograms of (a) [2H6]-aztreonam formate salt; (b) a LLOQ aztreonam sample with the respective S/N ratio; (c) a real patient sample (black continuous line) overlayed with the IS peak (red dotted line).
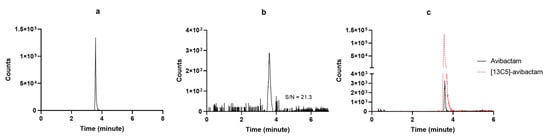
Figure 4.
MRM chromatograms of (a) [13C5]-avibactam sodium salt; (b) a LLOQ avibactam sample with the respective S/N ratio; (c) a real patient sample (black continuous line) overlayed with the IS peak (red dotted line).
3.2. Method Validation
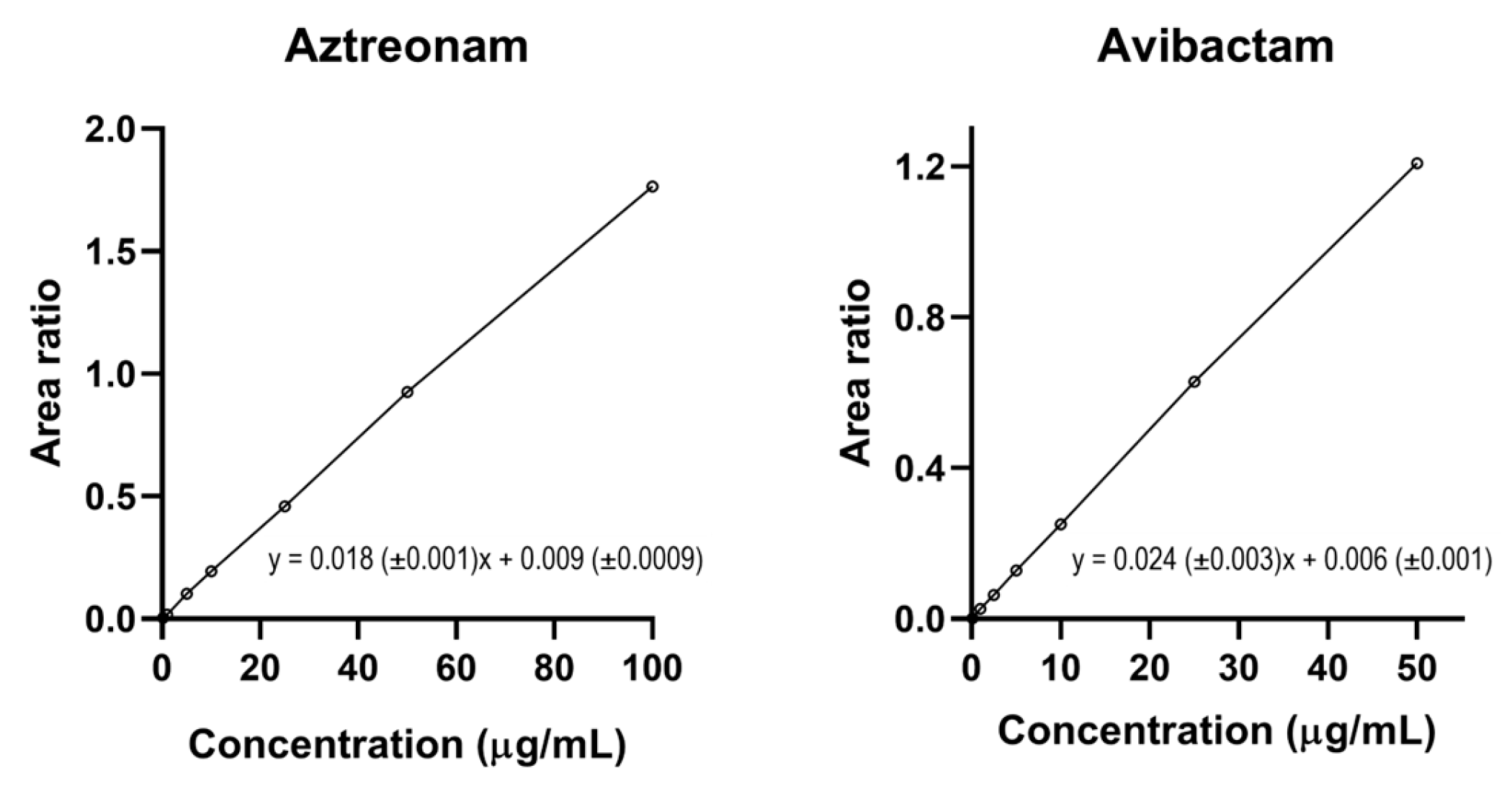
The sequential methods were validated according to the EMA guidelines showed excellent selectivity and specificity with no interfering peaks at the optimized chromatographic conditions. Thanks to the small injection volume (2 µL), the carry-over was absent. The linear regression fit of the calibration curves was excellent (with R2 ≥ 0.999) for both aztreonam and avibactam within the respective optimized dynamic range (0.2–100 μg/mL for aztreonam and 0.1–50 μg/mL for avibactam), as shown in Figure 5. The results of the intra-day and inter-day test of precision and accuracy were always within the acceptable ±15% ranges for both aztreonam and avibactam, as recommended by the EMA Guidelines [27] (Table 6 and Table 7, respectively).
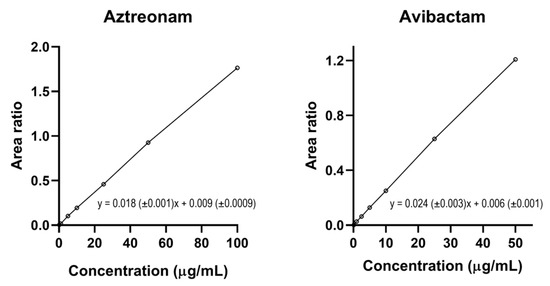
Figure 5.
Calibration curves of aztreonam and of avibactam and their respective linear equation based on 7 incremental calibrators each obtained by plotting the aztreonam/[2H6]-aztreonam and the avibactam/[13C5]-avibactam area ratio over concentration, respectively.

Table 6.
Intra-day and inter-day average (Avg) precision and accuracy evaluated at four concentration levels (LLOQ, LQC, MQC, and HQC) for aztreonam.

Table 7.
Intra-day and inter-day average (Avg) precision and accuracy evaluated at four concentration levels (LLOQ, LQC, MQC, and HQC) for avibactam.
The findings of %ME, %IS-normalized ME, and %ER of the LQC, the MQC, and the HQC of aztreonam and avibactam are summarized in Table 8 and Table 9, respectively. At all of the tested QC levels, the %ME was negative for aztreonam with a moderate signal suppression, whereas it was positive for avibactam with a slight signal increase. After IS normalization, the %ME values matched the criteria established by the EMA for validation. The %ER was very high for both aztreonam and avibactam, exceeding 90% at all of the tested concentrations. These findings may support the need for an IS to provide an accurate quantification across the entire tested dynamic range.

Table 8.
Average matrix effect (ME%), IS-normalized matrix effect (IS-ME%) and extraction recovery (ER%) of aztreonam measured at three QC levels.

Table 9.
Average matrix effect (ME%), IS-normalized matrix effect (IS-ME%) and extraction recovery (ER%) of avibactam measured at three QC levels.
The findings concerning the test of stability of aztreonam and avibactam are summarized in Table 10 and Table 11, respectively. Specifically, when the LQC, the MQC and the HQC were kept in the autosampler at 4 °C, aztreonam was stable for up to three days and avibactam for up to five days. Regarding the freeze–thaw stability, aztreonam was completely stable after the first cycle, showed borderline stability after the second cycle, and was markedly unstable after the third cycle. Conversely, avibactam easily withstood up to three freeze–thaw cycles.

Table 10.
Stability of aztreonam at different storage conditions.

Table 11.
Stability of avibactam at different storage conditions.
3.3. Clinical Application
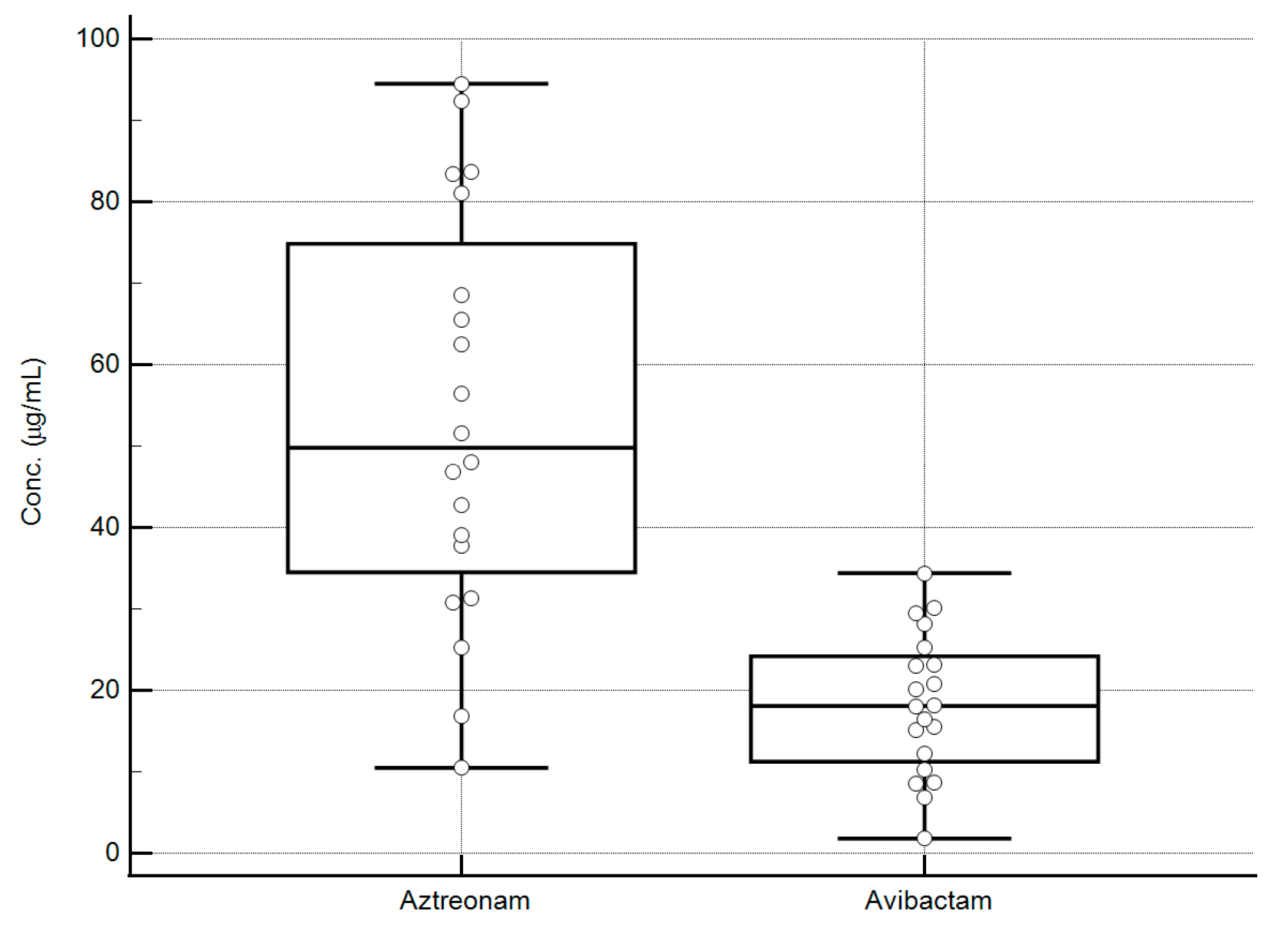
All aztreonam/avibactam concentrations measured in the critically ill patients’ plasma using the developed LC-MS/MS sequential methods were within the validated dynamic ranges. The distribution of the plasma concentrations of the analytes was quite widespread, as shown in the box and whisker plots of Figure 6. This may support the usefulness of these methods for applying real-time TDM-guided optimization of aztreonam/avibactam therapy.
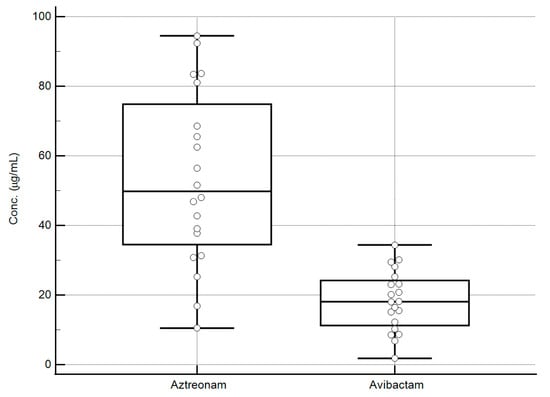
Figure 6.
Box and whisker plots of the steady-state concentration of aztreonam/avibactam measured in the plasma of 20 hospitalized patients. The values obtained range from 10.5 to 94.5 µg/mL for aztreonam and from 1.8 to 34.4 µg/mL for avibactam, falling well within the validation ranges of the analytical methods. Box represents median and interquartile range for each analyte, external lines represent the range of concentrations (min–max) for each analyte, whereas the circles represent the different concentrations measured for each analyte.
4. Discussion
In this study, two sensitive, accurate, and robust UHPLC–qTOF MS/MS sequential methods were developed and fully validated for quantifying aztreonam/avibactam in human plasma, with the specific aim of supporting TDM in clinical practice. The analytical performance achieved, particularly in terms of sensitivity, precision, and rapid turnaround time, compares favorably with previously published LC-MS/MS methods for β-lactam antibiotics, which are increasingly used to guide individualized dosing strategies in critically ill patients [17,18]. The linearity over a wide range of concentrations of the calibration curves and the specific LLOQ of these methods, namely among the lowest reported, may enable reliable measurement across wide concentration ranges encompassing both subtherapeutic and potentially supratherapeutic levels, regardless of using small plasma volumes [24,29,30,31]. The relevance of such analytical sensitivity is underscored by the pronounced intra- and inter-individual pharmacokinetic variability that aztreonam/avibactam—and likewise other BL/BLIc—may exhibit in hospitalized critically ill patients, driven by altered renal clearance and changes in volume of distribution [10,32,33]. Additionally, since the efficacy of aztreonam/avibactam is strictly dependent on the attainment of an optimal joint PK/PD target, the different variations in the renal clearance of avibactam compared to that of aztreonam potentially occurring in the critically ill patients could lead to major changes in the 3:1 aztreonam/avibactam proportion present in the vial, thus causing suboptimal joint PK/PD target attainment [12,13]. In this context, the present methods provide a valuable tool for real-time dose adaptation, complementing emerging clinical evidence supporting β-lactam TDM as a means to improve clinical outcomes and reduce the risk of therapeutic failure. It is noteworthy that the use of isotopically labeled internal standards enabled effective correction for plasma matrix variability, namely a major critical issue occurring in heterogeneous populations such as critically ill patients [29]. This may further improve the robustness and the reliability of the developed methods. Noteworthy, our methods of measuring aztreonam/avibactam were developed by using a high-resolution qTOF platform combined with a UPLC system. This approach may offer several advantages compared with the only other LC-MS/MS method available in the literature, which was based instead on a conventional triple-quadrupole approach [24]. Specifically, the high accuracy of mass detection may enhance the possibility of properly differentiating the target analytes from potential matrix interferents, even when the latter have very similar m/z values [26]. Furthermore, the quantification limits are significantly lower for both analytes, being even one order of magnitude lower for aztreonam, thus enabling the ability to work with small sample volumes. Finally, the qTOF platform, by recording a full scan of all of the ions generated from the sample and not only the signals of those of specific interest, may allow for the re-examination of the saved data files at a later stage. This could be especially valuable, for example, whenever searching for any other new potential interferent not originally taken into consideration. This may be especially valuable in the presence of complex clinical matrices and/or in the case of patients receiving multiple concomitant therapies, in which the risk of analytical interferences might be non-negligible [21]. The successful application of this method on clinical samples collected from patients having a wide inter-individual variability of concentrations may support the suitability and the utility of the methods for routine clinical use, especially considering the ever-growing role that aztreonam/avibactam has acquired in the treatment of MBL-producing Enterobacterales-related infections [25].
We recognize that the need to use two sequential chromatographic methods based on different stationary phases for maximizing analytical robustness and reliability could be a limitation. Unfortunately, the simultaneous measuring of aztreonam and avibactam on a single column was unfeasible due to the markedly different physicochemical properties and retention behaviors of the two analytes. However, it should not be overlooked that both the sample preparation protocol and the UHPLC–qTOF MS/MS analytical platform were shared by the two methods, and this may be time-saving when operating in routine therapeutic drug monitoring. Incurred sample reanalysis (ISR) of real-world clinical samples used for validating the analytical methods was not performed. We are aware that the ICH M10 guidelines recommend using this approach for testing bioanalytical methods supporting pharmacokinetic studies during pivotal clinical trials. Our aim was simply to validate the analytical method by showing that it was feasible for measuring residual clinical samples coming from routine therapeutic drug monitoring. Although we recognize that ISR could have added some value, its absence does not affect in any way the reliability, robustness and reproducibility of our sequential methods, as witnessed by the positive outcome of all the procedures for bioanalytical method validation recommended by the EMA guidelines.
Finally, it should not be overlooked that the incomplete stability of the two analytes under different testing conditions needs particular attention and may limit the application of these methods for real-time TDM in some settings. Both aztreonam and avibactam were stable for at least 3 days when kept at +4 °C. This means that after collecting blood samples from the patients, it is preferable to keep them at +4 °C during transport. At the same time, keeping sample extracts at +4 °C in the refrigerator could allow for reanalysis several times in the subsequent 2–3 days whenever needed. Conversely, the freeze–thaw tests showed that although avibactam withstood three freeze–thaw cycles, unfortunately, aztreonam was stable for only one thawing cycle. Consequently, whenever samples are delivered from a remote hospital to a hub lab, it is recommended that blood samples should be centrifuged locally and the supernatant, after being separated, should be stored in dry ice during transport until processing, so that they may undergo a maximum of one freeze–thaw cycle.
5. Conclusions
In conclusion, the two developed and validated UPLC–qTOF MS/MS sequential methods for measuring aztreonam/avibactam in human plasma were rapid, sensitive, and accurate, granting high performance, reliability, and short running times. Consequently, this may support their use as a robust bioanalytical platform suitable for real-time TDM-guided dose adjustment of aztreonam/avibactam in routine clinical practice.
Author Contributions
Methodology, I.T. and F.P.; validation, I.T., B.G. and R.D.P.; formal analysis, I.T., B.G., R.D.P., M.G. and F.P.; investigation, I.T., B.G. and R.D.P.; data curation, I.T., B.G., R.D.P., M.G. and F.P.; writing—original draft preparation, I.T.; writing—review and editing, M.G. and F.P. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The study was conducted according to the guidelines of the Declaration of Helsinki and approved by the Ethics Committee of the IRCCS Azienda Ospedaliero-Universitaria di Bologna (No. 442/2021/Oss/AOUBo on 22 June 2021, EM232–2022/Oss/AOUBo on 16 March 2022, and EM449–2023/Oss/AOUBo on 17 April 2024). Informed written consent was waived due to the retrospective and observational nature of the study, according to the article 110-bis comma 4 of Legislative Decree No. 196/2003 rev. 2024 of Italian Law regulating retrospective study.
Informed Consent Statement
Informed written consent was waived due to the retrospective and observational nature of the study, according to the article 110-bis comma 4 of Legislative Decree No. 196/2003 rev. 2024 of Italian Law regulating retrospective study.
Data Availability Statement
The data presented in this study are available upon request from the corresponding author. The data are not publicly available due to privacy restrictions.
Conflicts of Interest
M.G. received personal fees from Angelini, AdvanzPharma, and Viatris; F.P. participated in a speaker bureau for Angelini, BeiGene, Gilead, InfectoPharm, Menarini, Merck Sharp & Dohme, Pfizer, and Shionogi and in advisory boards for BeiGene, Merck Sharp & Dohme, Pfizer, and Viatris. The authors report no other conflicts of interest in this work.
Abbreviations
The following abbreviations are used in this manuscript:
| TDM | Therapeutic drug monitoring |
| UHPLC–qTOF-MS/MS | Ultraperformance Liquid Chromatography-quadrupole-Time-of-Flight-Mass Spectrometry |
| EMA | European Medicine Agency |
| MIC | Minimum inhibitory concentration |
| LC | Liquid chromatography |
| PK/PD | Pharmacokinetic/pharmacodynamic |
| DMSO | Dimethyl sulfoxide |
| QC | Quality control |
| LQC | Low quality control |
| MQC | Medium quality control |
| HQC | High quality control |
| CI | Continuous infusion |
| IS | Internal standard |
| MRM | Multiple Reaction Monitoring |
| ESI | Electrospray Ionization |
| DP | Declustering potential |
| CE | Collision energy |
| LLOQ | Low Limit of Quantification |
| ULoQ | Upper Limit of Quantification |
| S/N | Signal-to-noise ratio |
| CV | Coefficient of variation |
| ER | Extraction Recovery |
| ME | Matrix Effect |
| AVG | Average |
| CONC | Concentration |
| ESBLs | Extended-spectrum β-lactamases |
| MBLs | Metallo β-lactamases |
| CAD | Collisionally Activated Dissociation gas |
References
- Falcone, M.; Tiseo, G.; Carbonara, S.; Marino, A.; Di Caprio, G.; Carretta, A.; Mularoni, A.; Mariani, M.F.; Maraolo, A.E.; Scotto, R.; et al. Mortality Attributable to Bloodstream Infections Caused by Different Carbapenem-Resistant Gram-Negative Bacilli: Results from a Nationwide Study in Italy (ALARICO Network). Clin. Infect. Dis. 2023, 76, 2059–2069. [Google Scholar] [CrossRef]
- Lee, Y.-L.; Chen, H.-M.; Hii, I.-M.; Hsueh, P.-R. Carbapenemase-Producing Enterobacterales Infections: Recent Advances in Diagnosis and Treatment. Int. J. Antimicrob. Agents 2022, 59, 106528. [Google Scholar] [CrossRef]
- Kanj, S.S.; Kantecki, M.; Arhin, F.F.; Gheorghe, M. Epidemiology and Outcomes Associated with MBL-Producing Enterobacterales: A Systematic Literature Review. Int. J. Antimicrob. Agents 2025, 65, 107449. [Google Scholar] [CrossRef]
- Hidalgo-Tenorio, C.; Bou, G.; Oliver, A.; Rodríguez-Aguirregabiria, M.; Salavert, M.; Martínez-Martínez, L. The Challenge of Treating Infections Caused by Metallo-β-Lactamase-Producing Gram-Negative Bacteria: A Narrative Review. Drugs 2024, 84, 1519–1539. [Google Scholar] [CrossRef]
- Paul, M.; Carrara, E.; Retamar, P.; Tängdén, T.; Bitterman, R.; Bonomo, R.A.; de Waele, J.; Daikos, G.L.; Akova, M.; Harbarth, S.; et al. European Society of Clinical Microbiology and Infectious Diseases (ESCMID) Guidelines for the Treatment of Infections Caused by Multidrug-Resistant Gram-Negative Bacilli (Endorsed by European Society of Intensive Care Medicine). Clin. Microbiol. Infect. 2022, 28, 521–547. [Google Scholar] [CrossRef] [PubMed]
- Tamma, P.D.; Heil, E.L.; Justo, J.A.; Mathers, A.J.; Satlin, M.J.; Bonomo, R.A. Infectious Diseases Society of America 2024 Guidance on the Treatment of Antimicrobial-Resistant Gram-Negative Infections. Clin. Infect. Dis. 2024, ciae403. [Google Scholar] [CrossRef]
- Falcone, M.; Daikos, G.L.; Tiseo, G.; Bassoulis, D.; Giordano, C.; Galfo, V.; Leonildi, A.; Tagliaferri, E.; Barnini, S.; Sani, S.; et al. Efficacy of Ceftazidime-Avibactam Plus Aztreonam in Patients with Bloodstream Infections Caused by Metallo-β-Lactamase–Producing Enterobacterales. Clin. Infect. Dis. 2021, 72, 1871–1878. [Google Scholar] [CrossRef]
- Zhang, B.; Zhu, Z.; Jia, W.; Qu, F.; Huang, B.; Shan, B.; Yu, H.; Tang, Y.; Chen, L.; Du, H. In Vitro Activity of Aztreonam-Avibactam against Metallo-β-Lactamase-Producing Enterobacteriaceae-A Multicenter Study in China. Int. J. Infect. Dis. 2020, 97, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Al Musawa, M.; Bleick, C.R.; Herbin, S.R.; Caniff, K.E.; Van Helden, S.R.; Rybak, M.J. Aztreonam-Avibactam: The Dynamic Duo against Multidrug-Resistant Gram-Negative Pathogens. Pharmacotherapy 2024, 44, 927–938. [Google Scholar] [CrossRef] [PubMed]
- Pai Mangalore, R.; Peel, T.N.; Udy, A.A.; Peleg, A.Y. The Clinical Application of Beta-Lactam Antibiotic Therapeutic Drug Monitoring in the Critical Care Setting. J. Antimicrob. Chemother. 2023, 78, 2395–2405. [Google Scholar] [CrossRef]
- Ramsey, C.; MacGowan, A.P. A Review of the Pharmacokinetics and Pharmacodynamics of Aztreonam. J. Antimicrob. Chemother. 2016, 71, 2704–2712. [Google Scholar] [CrossRef] [PubMed]
- Xie, R.; Rogers, H.; Chow, J.W.; Soto, E.; Raber, S.R. Population Pharmacokinetic/Pharmacodynamic Modeling to Optimize Aztreonam-Avibactam Dose Regimens for Adult Patients. Antimicrob. Agents Chemother. 2025, 69, e0195024. [Google Scholar] [CrossRef] [PubMed]
- Das, S.; Riccobene, T.; Carrothers, T.J.; Wright, J.G.; MacPherson, M.; Cristinacce, A.; McFadyen, L.; Xie, R.; Luckey, A.; Raber, S. Dose Selection for Aztreonam-Avibactam, Including Adjustments for Renal Impairment, for Phase IIa and Phase III Evaluation. Eur. J. Clin. Pharmacol. 2024, 80, 529–543. [Google Scholar] [CrossRef]
- Tarral, A.; Merdjan, H. Effect of Age and Sex on the Pharmacokinetics and Safety of Avibactam in Healthy Volunteers. Antimicrob. Agents Chemother. 2015, 37, 877–886. [Google Scholar] [CrossRef]
- Morales Castro, D.; Dresser, L.; Granton, J.; Fan, E. Pharmacokinetic Alterations Associated with Critical Illness. Clin. Pharmacokinet. 2023, 62, 209–220. [Google Scholar] [CrossRef]
- Lodise, T.P.; O’Donnell, J.N.; Raja, S.; Guptill, J.T.; Zaharoff, S.; Schwager, N.; Fowler, V.G.; Beresnev, T.; Wall, A.; Wiegand, K.; et al. Safety of Ceftazidime-Avibactam in Combination with Aztreonam (COMBINE) in a Phase I, Open-Label Study in Healthy Adult Volunteers. Antimicrob. Agents Chemother. 2022, 66, e0093522. [Google Scholar] [CrossRef]
- Fratoni, A.J.; Nicolau, D.P.; Kuti, J.L. A Guide to Therapeutic Drug Monitoring of β-Lactam Antibiotics. Pharmacotherapy 2021, 41, 220–233. [Google Scholar] [CrossRef]
- Carlier, M.; Stove, V.; Wallis, S.C.; De Waele, J.J.; Verstraete, A.G.; Lipman, J.; Roberts, J.A. Assays for Therapeutic Drug Monitoring of β-Lactam Antibiotics: A Structured Review. Int. J. Antimicrob. Agents 2015, 46, 367–375. [Google Scholar] [CrossRef] [PubMed]
- Gatti, M.; Pea, F. Pharmacokinetic/Pharmacodynamic Target Attainment in Critically Ill Renal Patients on Antimicrobial Usage: Focus on Novel Beta-Lactams and Beta Lactams/Beta-Lactamase Inhibitors. Expert Rev. Clin. Pharmacol. 2021, 14, 583–599. [Google Scholar] [CrossRef]
- Lu, W.; Pan, M.; Ke, H.; Liang, J.; Liang, W.; Yu, P.; Zhang, P.; Wang, Q. An LC-MS/MS Method for the Simultaneous Determination of 18 Antibacterial Drugs in Human Plasma and Its Application in Therapeutic Drug Monitoring. Front. Pharmacol. 2022, 13, 1044234. [Google Scholar] [CrossRef]
- Feliu, C.; Konecki, C.; Candau, T.; Vautier, D.; Haudecoeur, C.; Gozalo, C.; Cazaubon, Y.; Djerada, Z. Quantification of 15 Antibiotics Widely Used in the Critical Care Unit with a LC-MS/MS System: An Easy Method to Perform a Daily Therapeutic Drug Monitoring. Pharmaceuticals 2021, 14, 1214. [Google Scholar] [CrossRef]
- Caro, Y.S.; Cámara, M.S.; De Zan, M.M. A Review of Bioanalytical Methods for the Therapeutic Drug Monitoring of β-Lactam Antibiotics in Critically Ill Patients: Evaluation of the Approaches Used to Develop and Validate Quality Attributes. Talanta 2020, 210, 120619. [Google Scholar] [CrossRef]
- Mula, J.; Chiara, F.; Manca, A.; Palermiti, A.; Maiese, D.; Cusato, J.; Simiele, M.; De Rosa, F.G.; Di Perri, G.; De Nicolò, A.; et al. Analytical Validation of a Novel UHPLC-MS/MS Method for 19 Antibiotics Quantification in Plasma: Implementation in a LC-MS/MS Kit. Biomed. Pharmacother. 2023, 163, 114790. [Google Scholar] [CrossRef]
- Magréault, S.; Jaureguy, F.; Zahar, J.-R.; Méchaï, F.; Toinon, D.; Cohen, Y.; Carbonnelle, E.; Jullien, V. Automated HPLC-MS/MS Assay for the Simultaneous Determination of Ten Plasma Antibiotic Concentrations. J. Chromatogr. B 2022, 1211, 123496. [Google Scholar] [CrossRef]
- Hölsken, O.; Sponheuer, K.; Weber, F.; Martens-Lobenhoffer, J.; Bode-Böger, S.M.; Kloft, C.; Treskatsch, S.; Angermair, S. First Clinical Application of Aztreonam–Avibactam in Treating Carbapenem-Resistant Enterobacterales: Insights from Therapeutic Drug Monitoring and Pharmacokinetic Simulations. J. Pers. Med. 2024, 14, 1135. [Google Scholar] [CrossRef] [PubMed]
- Géhin, C.; Holman, S.W. Advances in High-Resolution Mass Spectrometry Applied to Pharmaceuticals in 2020: A Whole New Age of Information. Anal. Sci. Adv. 2021, 2, 142–156. [Google Scholar] [CrossRef]
- European Medicine Agency. ICH M10 on Bioanalytical Method Validation—Scientific Guideline; European Medicines Agency: Amsterdam, The Netherlands, 2019. [Google Scholar]
- Forier, K.; Heck, V.V.; Carlier, M.; Braeckel, E.V.; Daele, S.V.; Baets, F.D.; Schelstraete, P.; Haerynck, F.; Stove, V.; Simaey, L.V.; et al. Development and Validation of an LC Tandem MS Assay for the Quantification of B-Lactam Antibiotics in the Sputum of Cystic Fibrosis Patients. J. Antimicrob. Chemother. 2017, 73, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Van Vooren, S.; Verstraete, A.G. A Sensitive and High-Throughput Quantitative Liquid Chromatography High-Resolution Mass Spectrometry Method for Therapeutic Drug Monitoring of 10 β-Lactam Antibiotics, Linezolid and Two β-Lactamase Inhibitors in Human Plasma. Biomed. Chromatogr. 2021, 35, e5092. [Google Scholar] [CrossRef] [PubMed]
- Goh, K.K.-K.; Toh, W.G.-H.; Hee, D.K.-H.; Ting, E.Z.-W.; Chua, N.G.S.; Zulkifli, F.I.B.; Sin, L.-J.; Tan, T.-T.; Kwa, A.L.-H.; Lim, T.-P. Quantification of Fosfomycin in Combination with Nine Antibiotics in Human Plasma and Cation-Adjusted Mueller-Hinton II Broth via LCMS. Antibiotics 2022, 11, 54. [Google Scholar] [CrossRef]
- Cheng, Y.; Chen, M.; Zhang, B.; Lin, H.; Li, X.; Cai, Y.; Zhang, H.; Que, W.; Liu, M.; Qiu, H. Rapid, Simple, and Economical LC–MS/MS Method for Simultaneous Determination of Ceftazidime and Avibactam in Human Plasma and Its Application in Therapeutic Drug Monitoring. J. Clin. Pharm. Ther. 2022, 47, 1426–1437. [Google Scholar] [CrossRef]
- Bush, K.; Bradford, P.A. β-Lactams and β-Lactamase Inhibitors: An Overview. Cold Spring Harb. Perspect. Med. 2016, 6, a025247. [Google Scholar] [CrossRef] [PubMed]
- Roberts, J.A.; Abdul-Aziz, M.H.; Lipman, J.; Mouton, J.W.; Vinks, A.A.; Felton, T.W.; Hope, W.W.; Farkas, A.; Neely, M.N.; Schentag, J.J.; et al. Individualised Antibiotic Dosing for Patients Who Are Critically Ill: Challenges and Potential Solutions. Lancet Infect. Dis. 2014, 14, 498–509. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.