Photodynamic Therapy for Glioblastoma: Potential Application of TiO2 and ZnO Nanoparticles as Photosensitizers
Abstract
1. Introduction
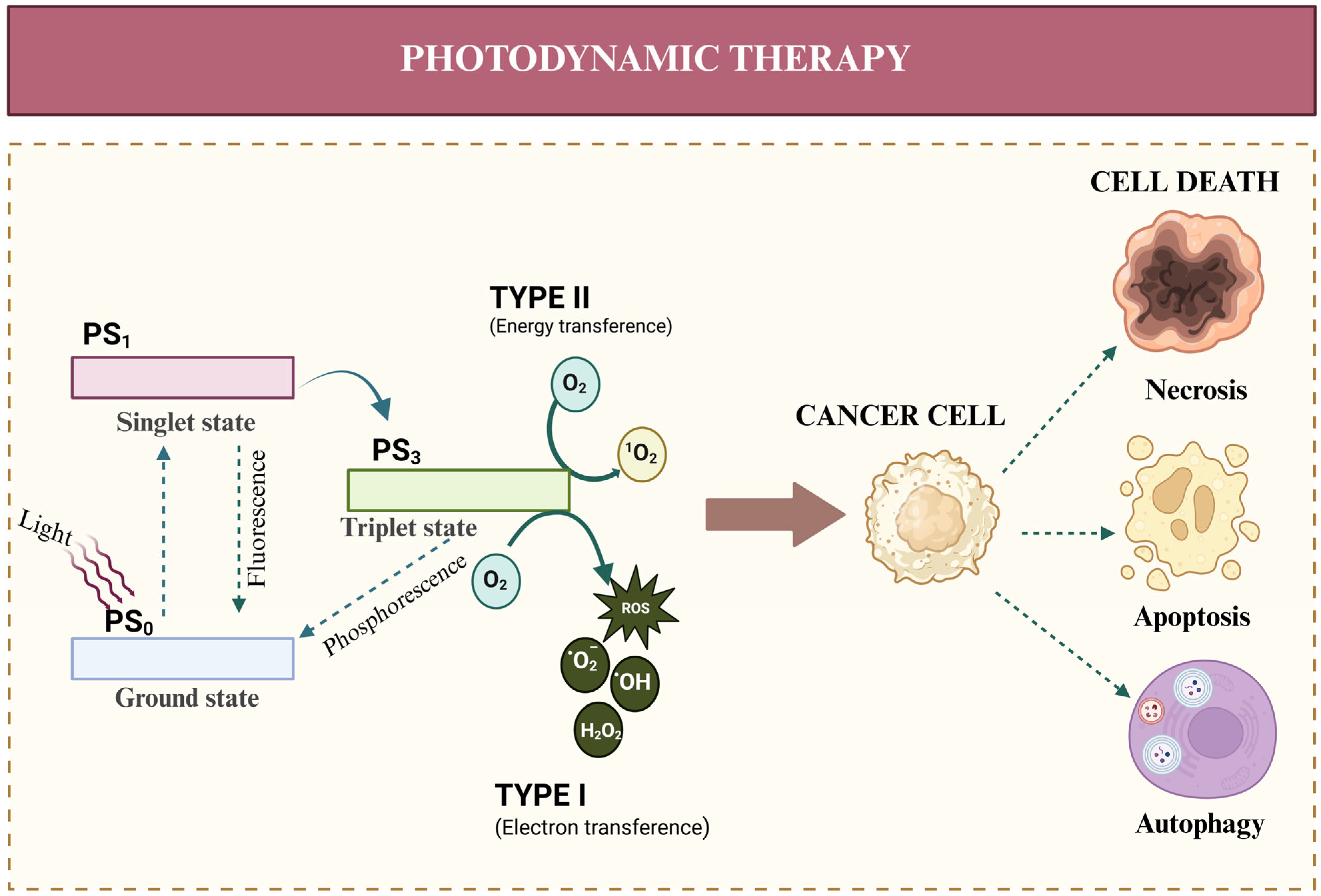
2. Photodynamic Therapy (PDT)
2.1. Type I Reactions in PDT
2.2. Type II Reactions in PDT
2.3. Types of PDT-Induced Cell Death
2.3.1. Apoptosis
2.3.2. Autophagy
2.3.3. Necroptosis
2.3.4. Pyroptosis
2.3.5. Ferroptosis
2.3.6. Cuproptosis
2.3.7. Paraptosis
2.3.8. Parthanatos
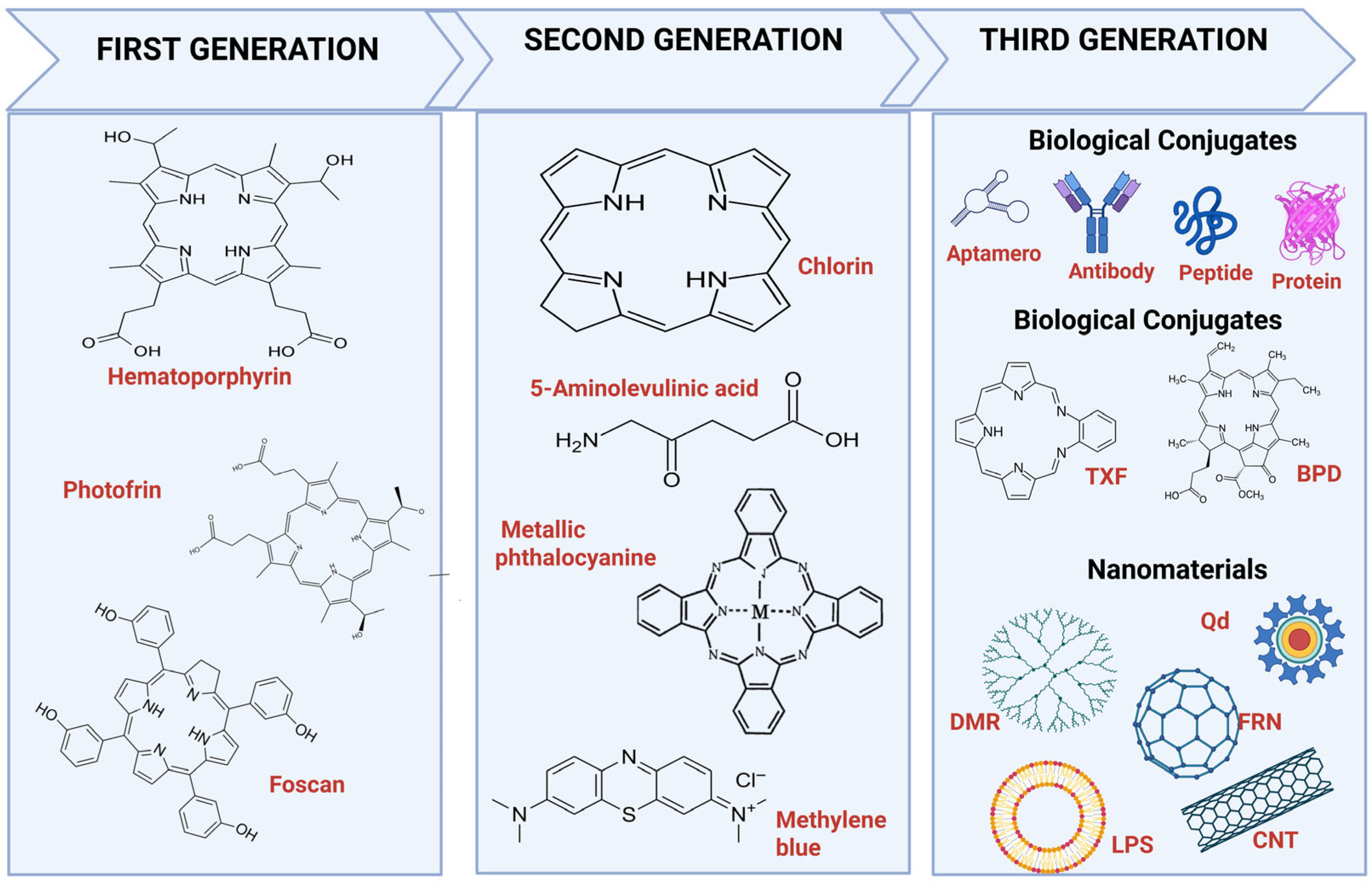
3. Photosensitizers (PSs) for PDT
3.1. First-Generation Photosensitizers in PDT for GB
3.2. Second-Generation Photosensitizers in PDT for GB
3.3. 5-ALA
3.4. Talaporfin (LS11, MACE, NPe6)
3.5. Phthalocyanines
3.6. Nanotechnology-Based Third-Generation Photosensitizers in PDT for GB
4. TiO2- and ZnO2-Based Nano-Photosensitizers
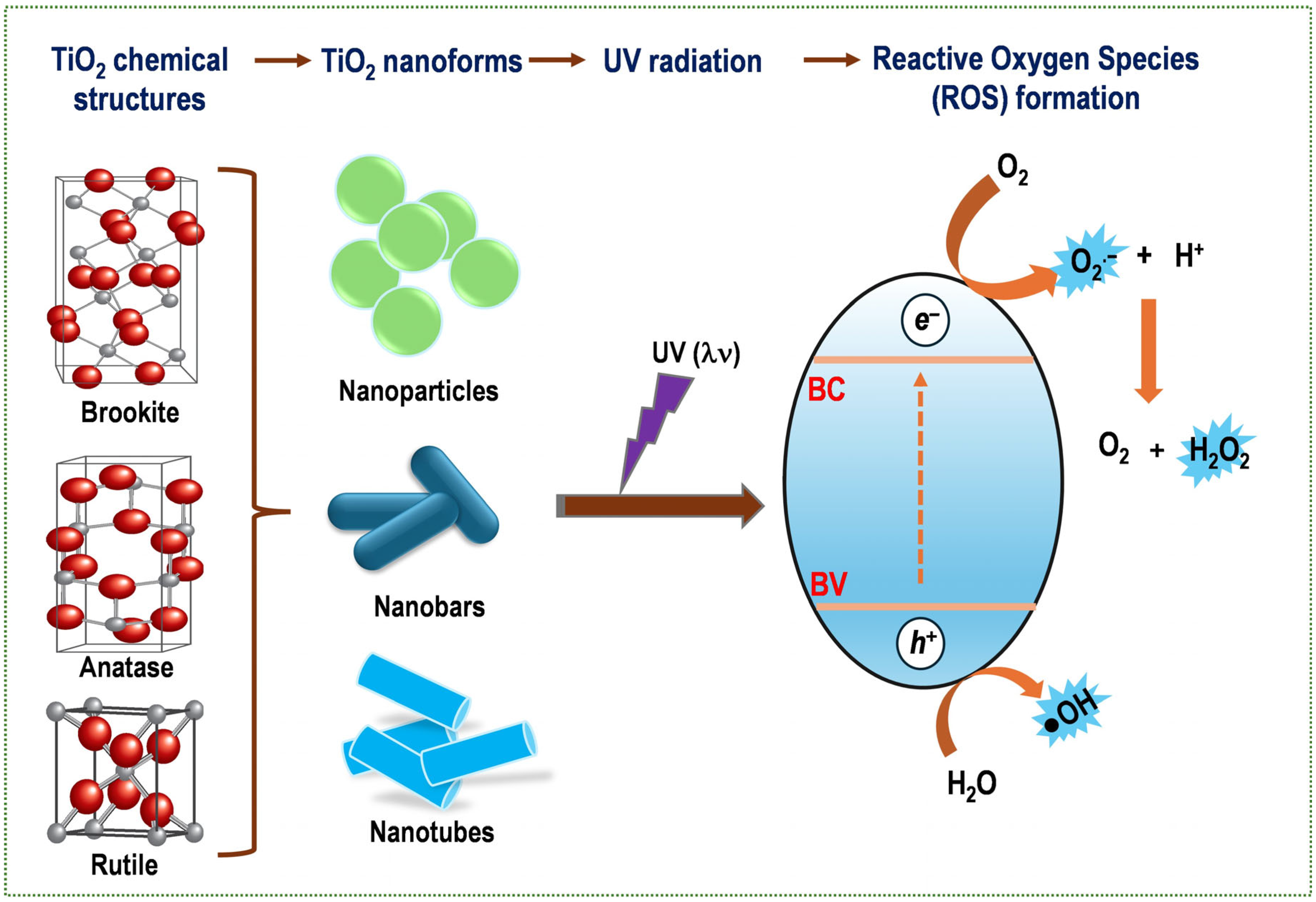
4.1. TiO2
Toxicity of TiO2NPs
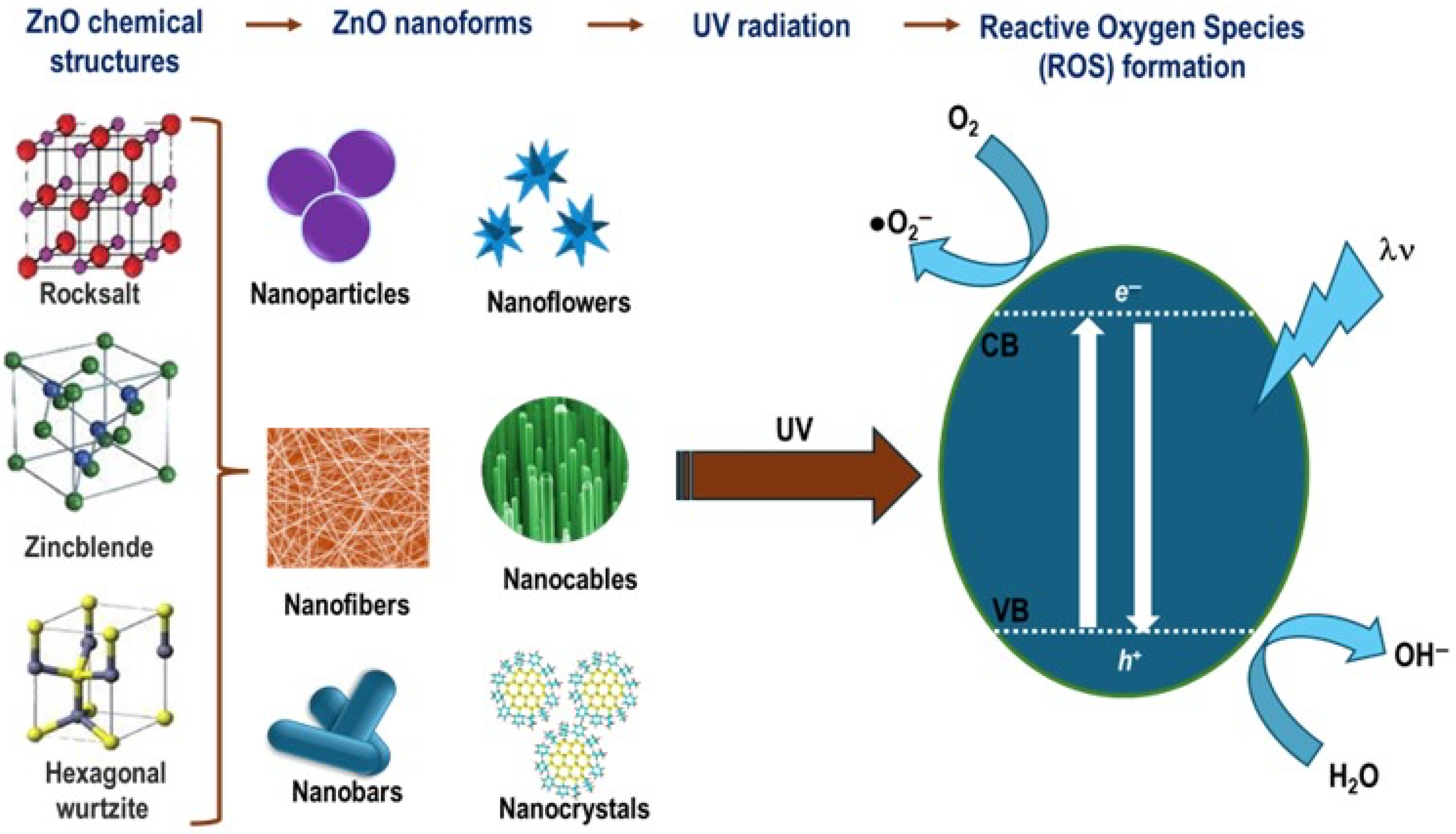
4.2. ZnO
Toxicity of ZnONPs
4.3. Protein Corona in TiO2NPs and ZnONPs
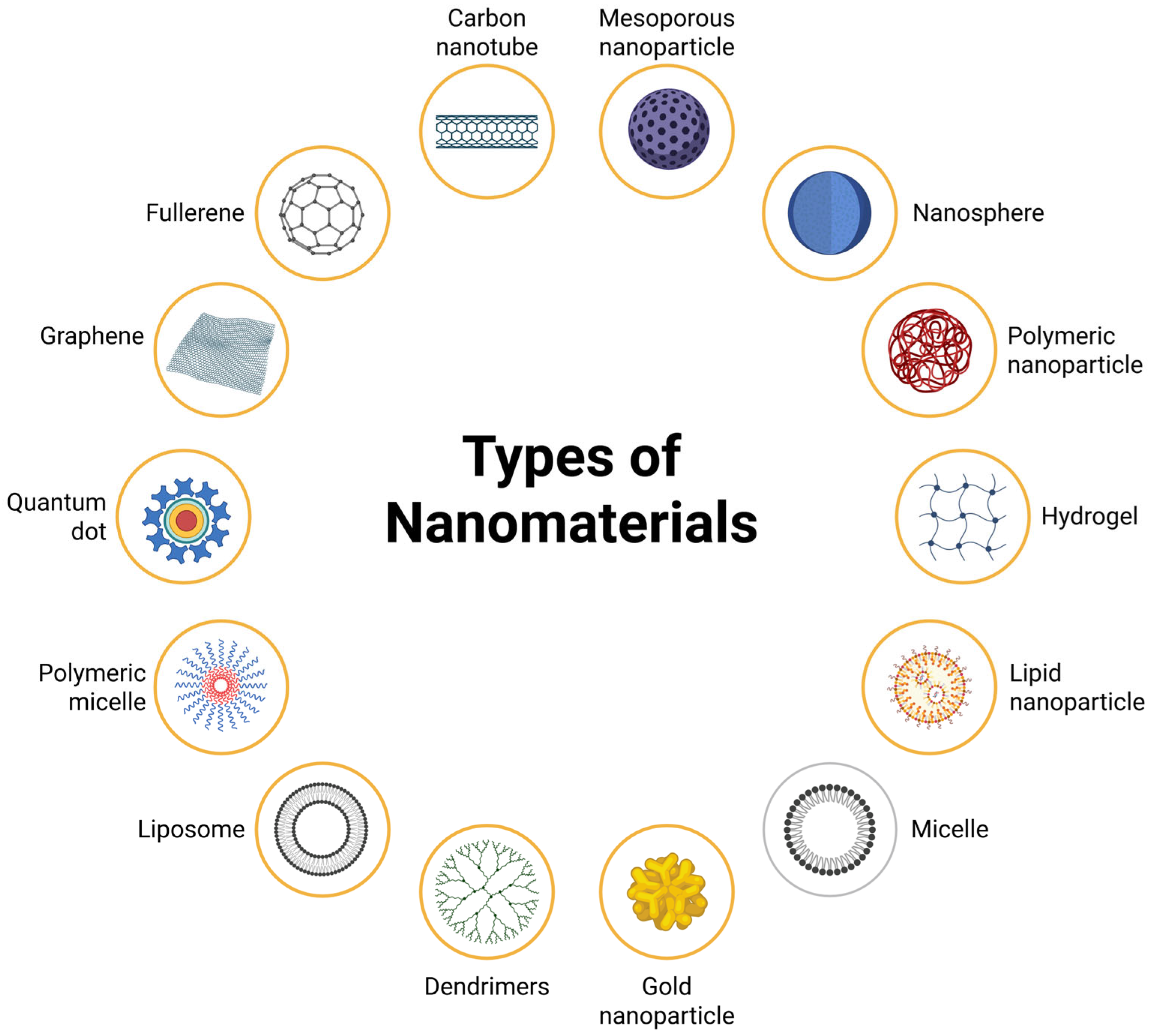
5. Cancer Therapy Using Nanomaterials
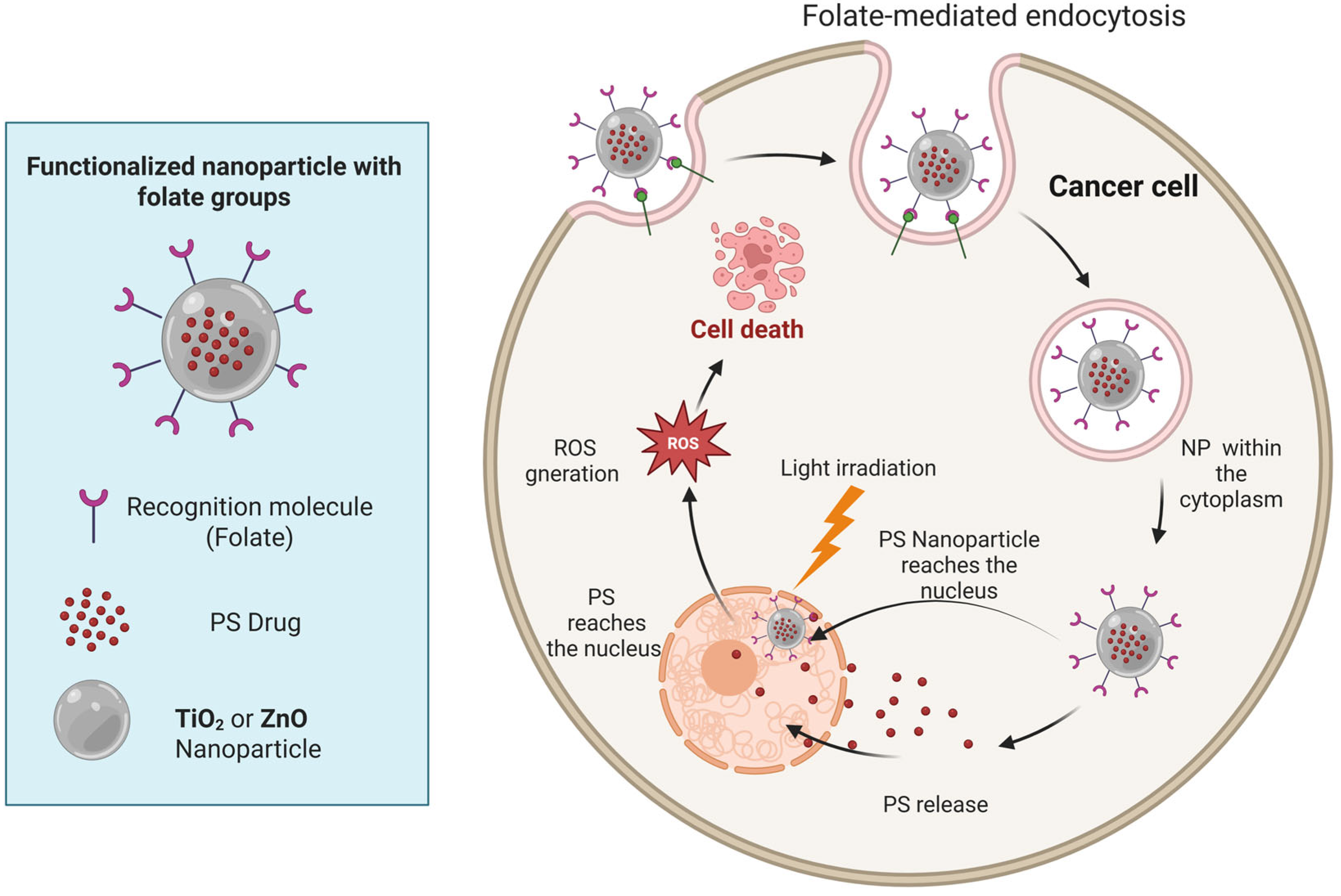
TiO2 and ZnO Nanoparticles in PDT for GB
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Long, Z.; Yi, Z.; Yan, W.; Wang, H. Trends in the immunotherapy for glioblastoma: A two-decade bibliometric analysis. Hum. Vaccines Immunother. 2025, 21, 2466299. [Google Scholar] [CrossRef] [PubMed]
- Stupp, R.; Mason, W.P.; van den Bent, M.J.; Weller, M.; Fisher, B.; Taphoorn, M.J.; Belanger, K.; Brandes, A.A.; Marosi, C.; Bogdahn, U.; et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N. Engl. J. Med. 2005, 352, 987–996. [Google Scholar] [CrossRef] [PubMed]
- Persano, L.; Rampazzo, E.; Basso, G.; Viola, G. Glioblastoma cancer stem cells: Role of the microenvironment and therapeutic targeting. Biochem. Pharmacol. 2013, 85, 612–622. [Google Scholar] [CrossRef]
- Huang, Z. A review of progress in clinical photodynamic therapy. Technol. Cancer Res. Treat. 2005, 4, 283–293. [Google Scholar] [CrossRef]
- Liu, Y.; Dai, S.; Wen, L.; Zhu, Y.; Tan, Y.; Qiu, G.; Meng, T.; Yu, F.; Yuan, H.; Hu, F. Enhancing Drug Delivery for Overcoming Angiogenesis and Improving the Phototherapy Efficacy of Glioblastoma by ICG-Loaded Glycolipid-Like Micelles. Int. J. Nanomed. 2020, 15, 2717–2732. [Google Scholar] [CrossRef]
- Aebisher, D.; Czech, S.; Dynarowicz, K.; Misiolek, M.; Komosinska-Vassev, K.; Kawczyk-Krupka, A.; Bartusik-Aebisher, D. Photodynamic Therapy: Past, Current, and Future. Int. J. Mol. Sci. 2024, 25, 11325. [Google Scholar] [CrossRef]
- Aebisher, D.; Przygorzewska, A.; Mysliwiec, A.; Dynarowicz, K.; Krupka-Olek, M.; Bozek, A.; Kawczyk-Krupka, A.; Bartusik-Aebisher, D. Current Photodynamic Therapy for Glioma Treatment: An Update. Biomedicines 2024, 12, 375. [Google Scholar] [CrossRef]
- Jiang, W.; Liang, M.; Lei, Q.; Li, G.; Wu, S. The Current Status of Photodynamic Therapy in Cancer Treatment. Cancers 2023, 15, 585. [Google Scholar] [CrossRef]
- Wu, L.; Tan, Y.; Zhang, H.; Guo, P.; Yang, D. A laser free self-luminous nanosystem for photodynamic therapy of cervical cancer cells. Photodiagn. Photodyn. Ther. 2023, 44, 103756. [Google Scholar] [CrossRef]
- da Silva, E.B., Jr.; Vasquez, M.W.M.; de Almeida Teixeira, B.C.; Neto, M.C.; Sprenger, F.; Filho, J.L.N.; Almeida-Lopes, L.; Ramina, R. Association of 5-aminolevulinic acid fluorescence guided resection with photodynamic therapy in recurrent glioblastoma: A matched cohort study. Acta Neurochir. 2024, 166, 212. [Google Scholar] [CrossRef] [PubMed]
- Wilson, B.C. Photodynamic therapy for cancer: Principles. Can. J. Gastroenterol. 2002, 16, 393–396. [Google Scholar] [CrossRef]
- Lange, C.; Bednarski, P.J. Photosensitizers for Photodynamic Therapy: Photochemistry in the Service of Oncology. Curr. Pharm. Des. 2016, 22, 6956–6974. [Google Scholar] [CrossRef]
- van Straten, D.; Mashayekhi, V.; de Bruijn, H.S.; Oliveira, S.; Robinson, D.J. Oncologic Photodynamic Therapy: Basic Principles, Current Clinical Status and Future Directions. Cancers 2017, 9, 19. [Google Scholar] [CrossRef]
- Gunaydin, G.; Gedik, M.E.; Ayan, S. Photodynamic Therapy-Current Limitations and Novel Approaches. Front. Chem. 2021, 9, 691697. [Google Scholar] [CrossRef] [PubMed]
- Correia, J.H.; Rodrigues, J.A.; Pimenta, S.; Dong, T.; Yang, Z. Photodynamic Therapy Review: Principles, Photosensitizers, Applications, and Future Directions. Pharmaceutics 2021, 13, 1332. [Google Scholar] [CrossRef] [PubMed]
- Dolmans, D.E.; Fukumura, D.; Jain, R.K. Photodynamic therapy for cancer. Nat. Rev. Cancer 2003, 3, 380–387. [Google Scholar] [CrossRef] [PubMed]
- Garapati, C.; Boddu, S.H.; Jacob, S.; Ranch, K.M.; Patel, C.; Babu, R.J.; Tiwari, A.K.; Yasin, H. Photodynamic therapy: A special emphasis on nanocarrier-mediated delivery of photosensitizers in antimicrobial therapy. Arab. J. Chem. 2023, 16, 104583. [Google Scholar] [CrossRef]
- Korbelik, M.; Heger, M.; Girotti, A.W. Participation of lipids in the tumor response to photodynamic therapy and its exploitation for therapeutic gain. J. Lipid Res. 2025, 66, 100729. [Google Scholar] [CrossRef]
- Zhu, T.; Shi, L.; Yu, C.; Dong, Y.; Qiu, F.; Shen, L.; Qian, Q.; Zhou, G.; Zhu, X. Ferroptosis Promotes Photodynamic Therapy: Supramolecular Photosensitizer-Inducer Nanodrug for Enhanced Cancer Treatment. Theranostics 2019, 9, 3293–3307. [Google Scholar] [CrossRef]
- Sai, D.L.; Lee, J.; Nguyen, D.L.; Kim, Y.P. Tailoring photosensitive ROS for advanced photodynamic therapy. Exp. Mol. Med. 2021, 53, 495–504. [Google Scholar] [CrossRef]
- Sobhani, N.; Samadani, A.A. Implications of photodynamic cancer therapy: An overview of PDT mechanisms basically and practically. J. Egypt. Natl. Cancer Inst. 2021, 33, 34. [Google Scholar] [CrossRef]
- Sultana, N.; Pathak, R.; Samanta, S.; Sarma, N.S. A comprehensive analysis of photothermal therapy (PTT) and photodynamic therapy (PDT) for the treatment of cancer. Process Biochem. 2015, 148, 17–31. [Google Scholar] [CrossRef]
- Juarranz, A.; Jaen, P.; Sanz-Rodriguez, F.; Cuevas, J.; Gonzalez, S. Photodynamic therapy of cancer. Basic principles and applications. Clin. Transl. Oncol. 2008, 10, 148–154. [Google Scholar] [CrossRef] [PubMed]
- Mishchenko, T.; Balalaeva, I.; Gorokhova, A.; Vedunova, M.; Krysko, D.V. Which cell death modality wins the contest for photodynamic therapy of cancer? Cell Death Dis. 2022, 13, 455. [Google Scholar] [CrossRef]
- de Souza, A.C.; Mencalha, A.L.; Fonseca, A.S.D.; de Paoli, F. Necroptosis as a consequence of photodynamic therapy in tumor cells. Lasers Med. Sci. 2024, 39, 267. [Google Scholar] [CrossRef]
- Miki, Y.; Akimoto, J.; Moritake, K.; Hironaka, C.; Fujiwara, Y. Photodynamic therapy using talaporfin sodium induces concentration-dependent programmed necroptosis in human glioblastoma T98G cells. Lasers Med. Sci. 2015, 30, 1739–1745. [Google Scholar] [CrossRef]
- Coupienne, I.; Fettweis, G.; Rubio, N.; Agostinis, P.; Piette, J. 5-ALA-PDT induces RIP3-dependent necrosis in glioblastoma. Photochem. Photobiol. Sci. 2011, 10, 1868–1878. [Google Scholar] [CrossRef]
- Izumoto, A.; Nishimura, T.; Hazama, H.; Ikeda, N.; Kajimoto, Y.; Awazu, K. Singlet oxygen model evaluation of interstitial photodynamic therapy with 5-aminolevulinic acid for malignant brain tumor. J. Biomed. Opt. 2019, 25, 063803. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Shen, J.; Zheng, C.; Zhu, P.; Yang, H.; Huang, Y.; Mao, X.; Yang, Z.; Hu, G.; Chen, Y. Cell death: The underlying mechanisms of photodynamic therapy for skin diseases. Interdiscip. Med. 2025, 3, e20240057. [Google Scholar] [CrossRef]
- Wang, B.; Zhou, H.; Chen, L.; Ding, Y.; Zhang, X.; Chen, H.; Liu, H.; Li, P.; Chen, Y.; Yin, C.; et al. A Mitochondria-Targeted Photosensitizer for Combined Pyroptosis and Apoptosis with NIR-II Imaging/Photoacoustic Imaging-Guided Phototherapy. Angew. Chem. Int. Ed. 2024, 63, e202408874. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.; Bisoyi, H.K.; Chen, X.M.; Liu, Z.; Chen, X.; Zhang, S.; Li, Q. Pyroptosis-Mediated Synergistic Photodynamic and Photothermal Immunotherapy Enabled by a Tumor-Membrane-Targeted Photosensitive Dimer. Adv. Mater. 2023, 35, e2300232. [Google Scholar] [CrossRef]
- Loveless, R.; Bloomquist, R.; Teng, Y. Pyroptosis at the forefront of anticancer immunity. J. Exp. Clin. Cancer Res. 2021, 40, 264. [Google Scholar] [CrossRef]
- Zhaoyun, L.; Wang, H.; Yang, C.; Zhao, X.; Hui, L.; Song, J.; Ding, K.; Fu, R. Enhancing antitumor immunity via ROS-ERS and pyroptosis-induced immunogenic cell death in multiple myeloma. J. Immunother. Cancer 2025, 13, e011717. [Google Scholar] [CrossRef]
- Demuynck, R.; Efimova, I.; Naessens, F.; Krysko, D.V. Immunogenic ferroptosis and where to find it? J. Immunother. Cancer 2021, 9, e003430. [Google Scholar] [CrossRef]
- Zhang, M.; Lei, Q.; Huang, X.; Wang, Y. Molecular mechanisms of ferroptosis and the potential therapeutic targets of ferroptosis signaling pathways for glioblastoma. Front. Pharmacol. 2022, 13, 1071897. [Google Scholar] [CrossRef]
- Otasevic, V.; Vucetic, M.; Grigorov, I.; Martinovic, V.; Stancic, A. Ferroptosis in Different Pathological Contexts Seen through the Eyes of Mitochondria. Oxidative Med. Cell. Longev. 2021, 2021, 5537330. [Google Scholar] [CrossRef] [PubMed]
- de Souza, I.; Ramalho, M.C.C.; Guedes, C.B.; Osawa, I.Y.A.; Monteiro, L.K.S.; Gomes, L.R.; Rocha, C.R.R. Ferroptosis Modulation: Potential Therapeutic Target for Glioblastoma Treatment. Int. J. Mol. Sci. 2022, 23, 6879. [Google Scholar] [CrossRef]
- Chen, Y.; Tian, H.; Zhang, X.; Nice, E.C.; Huang, C.; Zhang, H.; Zheng, S. Copper-coordination driven brain-targeting nanoassembly for efficient glioblastoma multiforme immunotherapy by cuproptosis-mediated tumor immune microenvironment reprogramming. J. Nanobiotechnol. 2024, 22, 801. [Google Scholar] [CrossRef] [PubMed]
- Qin, Z.; Yang, B.; Jin, X.; Zhao, H.; Liu, N. Cuproptosis in glioblastoma: Unveiling a novel prognostic model and therapeutic potential. Front. Oncol. 2024, 14, 1359778. [Google Scholar] [CrossRef] [PubMed]
- Cui, X.; Zheng, H.; Li, H.; Zhang, F.; Yang, L.; Ni, J.; Wang, D.; Zhang, H.; Tang, P.; Li, R.; et al. Paraptosome: A Novel Pathological Feature in Paraptotic Cell Death. bioRxiv 2024. [Google Scholar] [CrossRef]
- Kim, E.; Lee, D.M.; Seo, M.J.; Lee, H.J.; Choi, K.S. Intracellular Ca2+ Imbalance Critically Contributes to Paraptosis. Front. Cell Dev. Biol. 2020, 8, 607844. [Google Scholar] [CrossRef]
- Kunst, C.; Tumen, D.; Ernst, M.; Tews, H.C.; Muller, M.; Gulow, K. Paraptosis—A Distinct Pathway to Cell Death. Int. J. Mol. Sci. 2024, 25, 11478. [Google Scholar] [CrossRef]
- Pashootan, P.; Saadati, F.; Fahimi, H.; Rahmati, M.; Strippoli, R.; Zarrabi, A.; Cordani, M.; Moosavi, M.A. Metal-based nanoparticles in cancer therapy: Exploring photodynamic therapy and its interplay with regulated cell death pathways. Int. J. Pharm. 2024, 649, 123622. [Google Scholar] [CrossRef]
- Wang, J.; Cao, M.; Han, L.; Shangguan, P.; Liu, Y.; Zhong, Y.; Chen, C.; Wang, G.; Chen, X.; Lin, M.; et al. Blood-Brain Barrier-Penetrative Fluorescent Anticancer Agents Triggering Paraptosis and Ferroptosis for Glioblastoma Therapy. J. Am. Chem. Soc. 2024, 146, 28783–28794. [Google Scholar] [CrossRef] [PubMed]
- Kessel, D. Detection of Paraptosis After Photodynamic Therapy. Methods Mol. Biol. 2022, 2451, 711–720. [Google Scholar] [CrossRef]
- Wang, Y.; Luo, W.; Wang, Y. PARP-1 and its associated nucleases in DNA damage response. DNA Repair 2019, 81, 102651. [Google Scholar] [CrossRef] [PubMed]
- Huang, P.; Chen, G.; Jin, W.; Mao, K.; Wan, H.; He, Y. Molecular Mechanisms of Parthanatos and Its Role in Diverse Diseases. Int. J. Mol. Sci. 2022, 23, 7292. [Google Scholar] [CrossRef] [PubMed]
- Soriano, J.; Mora-Espi, I.; Alea-Reyes, M.E.; Perez-Garcia, L.; Barrios, L.; Ibanez, E.; Nogues, C. Cell Death Mechanisms in Tumoral and Non-Tumoral Human Cell Lines Triggered by Photodynamic Treatments: Apoptosis, Necrosis and Parthanatos. Sci. Rep. 2017, 7, 41340. [Google Scholar] [CrossRef] [PubMed]
- Fan, S.; Li, H.; Liu, K. Molecular prognostic of nine parthanatos death-related genes in glioma, particularly in COL8A1 identification. J. Neurochem. 2024, 168, 205–223. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Meng, X.; Bu, W. Upconversion-based photodynamic cancer therapy. Coord. Chem. Rev. 2019, 379, 82–98. [Google Scholar] [CrossRef]
- Cramer, S.W.; Chen, C.C. Photodynamic Therapy for the Treatment of Glioblastoma. Front. Surg. 2019, 6, 81. [Google Scholar] [CrossRef]
- Przygoda, M.; Bartusik-Aebisher, D.; Dynarowicz, K.; Cieslar, G.; Kawczyk-Krupka, A.; Aebisher, D. Cellular Mechanisms of Singlet Oxygen in Photodynamic Therapy. Int. J. Mol. Sci. 2023, 24, 16890. [Google Scholar] [CrossRef]
- Xie, J.; Li, H.; Zhang, T.; Song, B.; Wang, X.; Gu, Z. Recent Advances in ZnO Nanomaterial-Mediated Biological Applications and Action Mechanisms. Nanomaterials 2023, 13, 1500. [Google Scholar] [CrossRef] [PubMed]
- Mahmoudi, K.; Garvey, K.L.; Bouras, A.; Cramer, G.; Stepp, H.; Jesu Raj, J.G.; Bozec, D.; Busch, T.M.; Hadjipanayis, C.G. 5-aminolevulinic acid photodynamic therapy for the treatment of high-grade gliomas. J. Neuro-Oncol. 2019, 141, 595–607. [Google Scholar] [CrossRef] [PubMed]
- Bhanja, D.; Wilding, H.; Baroz, A.; Trifoi, M.; Shenoy, G.; Slagle-Webb, B.; Hayes, D.; Soudagar, Y.; Connor, J.; Mansouri, A. Photodynamic Therapy for Glioblastoma: Illuminating the Path toward Clinical Applicability. Cancers 2023, 15, 3427. [Google Scholar] [CrossRef] [PubMed]
- Mansoori, B.; Mohammadi, A.; Amin Doustvandi, M.; Mohammadnejad, F.; Kamari, F.; Gjerstorff, M.F.; Baradaran, B.; Hamblin, M.R. Photodynamic therapy for cancer: Role of natural products. Photodiagn. Photodyn. Ther. 2019, 26, 395–404. [Google Scholar] [CrossRef]
- Aires-Fernandes, M.; Botelho Costa, R.; Rochetti do Amaral, S.; Mussagy, C.U.; Santos-Ebinuma, V.C.; Primo, F.L. Development of Biotechnological Photosensitizers for Photodynamic Therapy: Cancer Research and Treatment-From Benchtop to Clinical Practice. Molecules 2022, 27, 6848. [Google Scholar] [CrossRef]
- Niculescu, A.-G.; Grumezescu, A.M. Photodynamic Therapy An Up-to-Date Review. Appl. Sci. 2021, 11, 3626. [Google Scholar] [CrossRef]
- Wang, Z.; Peng, H.; Shi, W.; Gan, L.U.; Zhong, L.; He, J.; Xie, L.; Wu, P.; Zhao, Y.; Deng, Z.; et al. Application of photodynamic therapy in cancer: Challenges and advancements. Biocell 2021, 45, 489–500. [Google Scholar] [CrossRef]
- Diamond, I.; Granelli, S.G.; McDonagh, A.F.; Nielsen, S.; Wilson, C.B.; Jaenicke, R. Photodynamic therapy of malignant tumours. Lancet 1972, 2, 1175–1177. [Google Scholar] [CrossRef]
- Perria, C.; Capuzzo, T.; Cavagnaro, G.; Datti, R.; Francaviglia, N.; Rivano, C.; Tercero, V.E. Fast attempts at the photodynamic treatment of human gliomas. J. Neurosurg. Sci. 1980, 24, 119–129. [Google Scholar] [PubMed]
- Perria, C.; Carai, M.; Falzoi, A.; Orunesu, G.; Rocca, A.; Massarelli, G.; Francaviglia, N.; Jori, G. Photodynamic therapy of malignant brain tumors: Clinical results of, difficulties with, questions about, and future prospects for the neurosurgical applications. Neurosurgery 1988, 23, 557–563. [Google Scholar] [CrossRef]
- McCulloch, G.A.; Forbes, I.J.; See, K.L.; Cowled, P.A.; Jacka, F.J.; Ward, A.D. Phototherapy in malignant brain tumors. Prog. Clin. Biol. Res. 1984, 170, 709–717. [Google Scholar]
- Forbes, I.J.; Cowled, P.A.; Leong, A.S.; Ward, A.D.; Black, R.B.; Blake, A.J.; Jacka, F.J. Phototherapy of human tumours using haematoporphyrin derivative. Med. J. Aust. 1980, 2, 489–493. [Google Scholar] [CrossRef]
- Laws, E.R., Jr.; Cortese, D.A.; Kinsey, J.H.; Eagan, R.T.; Anderson, R.E. Photoradiation therapy in the treatment of malignant brain tumors: A phase I (feasibility) study. Neurosurgery 1981, 9, 672–678. [Google Scholar] [CrossRef]
- Kostron, H.; Fritsch, E.; Grunert, V. Photodynamic therapy of malignant brain tumours: A phase I/II trial. Br. J. Neurosurg. 1988, 2, 241–248. [Google Scholar] [CrossRef]
- Stylli, S.S.; Kaye, A.H.; MacGregor, L.; Howes, M.; Rajendra, P. Photodynamic therapy of high grade glioma—Long term survival. J. Clin. Neurosci. 2005, 12, 389–398. [Google Scholar] [CrossRef] [PubMed]
- Muller, P.J.; Wilson, B.C. Photodynamic therapy of brain tumors—A work in progress. Lasers Surg. Med. 2006, 38, 384–389. [Google Scholar] [CrossRef] [PubMed]
- Park, J.; Lee, Y.K.; Park, I.K.; Hwang, S.R. Current Limitations and Recent Progress in Nanomedicine for Clinically Available Photodynamic Therapy. Biomedicines 2021, 9, 85. [Google Scholar] [CrossRef]
- Hsia, T.; Small, J.L.; Yekula, A.; Batool, S.M.; Escobedo, A.K.; Ekanayake, E.; You, D.G.; Lee, H.; Carter, B.S.; Balaj, L. Systematic Review of Photodynamic Therapy in Gliomas. Cancers 2023, 15, 3918. [Google Scholar] [CrossRef]
- Zhang, J.; Jiang, C.; Figueiro Longo, J.P.; Azevedo, R.B.; Zhang, H.; Muehlmann, L.A. An updated overview on the development of new photosensitizers for anticancer photodynamic therapy. Acta Pharm. Sin. B 2018, 8, 137–146. [Google Scholar] [CrossRef] [PubMed]
- Cesca, B.A.; Pellicer San Martin, K.; Caverzan, M.D.; Oliveda, P.M.; Ibarra, L.E. State-of-the-art photodynamic therapy for malignant gliomas: Innovations in photosensitizers and combined therapeutic approaches. Explor. Target. Anti-Tumor Ther. 2025, 6, 1002303. [Google Scholar] [CrossRef]
- Zhang, J.; Yan, X.; Gao, X.; Hu, J.; Zhao, H.; Yang, C.; Xu, L.; Niu, Y.; Geng, M.; Wang, N.; et al. Three Decades of Photodynamic Therapy for Glioblastoma: A Comprehensive Scientometric Analysis. Photodiagn. Photodyn. Ther. 2025, 53, 104533. [Google Scholar] [CrossRef]
- Dupont, C.; Vermandel, M.; Leroy, H.A.; Quidet, M.; Lecomte, F.; Delhem, N.; Mordon, S.; Reyns, N. INtraoperative photoDYnamic Therapy for GliOblastomas (INDYGO): Study Protocol for a Phase I Clinical Trial. Neurosurgery 2019, 84, E414–E419. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Yang, L.; Yi, W.; Fan, W.; Wen, Y.; Miao, X.; Xiong, L. Combination of Fluorescence-Guided Surgery With Photodynamic Therapy for the Treatment of Cancer. Mol. Imaging 2017, 16, 1536012117722911. [Google Scholar] [CrossRef] [PubMed]
- Teng, L.; Nakada, M.; Zhao, S.G.; Endo, Y.; Furuyama, N.; Nambu, E.; Pyko, I.V.; Hayashi, Y.; Hamada, J.I. Silencing of ferrochelatase enhances 5-aminolevulinic acid-based fluorescence and photodynamic therapy efficacy. Br. J. Cancer 2011, 104, 798–807. [Google Scholar] [CrossRef]
- Stummer, W.; Stocker, S.; Novotny, A.; Heimann, A.; Sauer, O.; Kempski, O.; Plesnila, N.; Wietzorrek, J.; Reulen, H.J. In vitro and in vivo porphyrin accumulation by C6 glioma cells after exposure to 5-aminolevulinic acid. J. Photochem. Photobiol. B 1998, 45, 160–169. [Google Scholar] [CrossRef]
- Stepp, H.; Beck, T.; Pongratz, T.; Meinel, T.; Kreth, F.W.; Tonn, J.; Stummer, W. ALA and malignant glioma: Fluorescence-guided resection and photodynamic treatment. J. Environ. Pathol. Toxicol. Oncol. 2007, 26, 157–164. [Google Scholar] [CrossRef]
- Vasilev, A.; Sofi, R.; Rahman, R.; Smith, S.J.; Teschemacher, A.G.; Kasparov, S. Using Light for Therapy of Glioblastoma Multiforme (GBM). Brain Sci. 2020, 10, 75. [Google Scholar] [CrossRef]
- Fukami, S.; Akimoto, J.; Nagai, K.; Saito, Y.; Kohno, M. Photodynamic therapy using talaporfin sodium for non-totally resectable malignant glioma. Photodiagn. Photodyn. Ther. 2024, 45, 103869. [Google Scholar] [CrossRef]
- Kobayashi, T.; Nitta, M.; Shimizu, K.; Saito, T.; Tsuzuki, S.; Fukui, A.; Koriyama, S.; Kuwano, A.; Komori, T.; Masui, K.; et al. Therapeutic Options for Recurrent Glioblastoma-Efficacy of Talaporfin Sodium Mediated Photodynamic Therapy. Pharmaceutics 2022, 14, 353. [Google Scholar] [CrossRef]
- Stummer, W.; Pichlmeier, U.; Meinel, T.; Wiestler, O.D.; Zanella, F.; Reulen, H.J.; Group, A.L.-G.S. Fluorescence-guided surgery with 5-aminolevulinic acid for resection of malignant glioma: A randomised controlled multicentre phase III trial. Lancet Oncol. 2006, 7, 392–401. [Google Scholar] [CrossRef] [PubMed]
- Hadjipanayis, C.G.; Stummer, W. 5-ALA and FDA approval for glioma surgery. J. Neuro-Oncol. 2019, 141, 479–486. [Google Scholar] [CrossRef] [PubMed]
- Muragaki, Y.; Akimoto, J.; Maruyama, T.; Iseki, H.; Ikuta, S.; Nitta, M.; Maebayashi, K.; Saito, T.; Okada, Y.; Kaneko, S.; et al. Phase II clinical study on intraoperative photodynamic therapy with talaporfin sodium and semiconductor laser in patients with malignant brain tumors. J. Neurosurg. 2013, 119, 845–852. [Google Scholar] [CrossRef]
- Namatame, H.; Akimoto, J.; Matsumura, H.; Haraoka, J.; Aizawa, K. Photodynamic therapy of C6-implanted glioma cells in the rat brain employing second-generation photosensitizer talaporfin sodium. Photodiagn. Photodyn. Ther. 2008, 5, 198–209. [Google Scholar] [CrossRef]
- Akimoto, J.; Haraoka, J.; Aizawa, K. Preliminary clinical report on safety and efficacy of photodynamic therapy using talaporfin sodium for malignant gliomas. Photodiagn. Photodyn. Ther. 2012, 9, 91–99. [Google Scholar] [CrossRef]
- Akimoto, J. Photodynamic Therapy for Malignant Brain Tumors. Neurol. Med. Chir. 2016, 56, 151–157. [Google Scholar] [CrossRef]
- Akimoto, J.; Fukami, S.; Ichikawa, M.; Mohamed, A.; Kohno, M. Intraoperative Photodiagnosis for Malignant Glioma Using Photosensitizer Talaporfin Sodium. Front. Surg. 2019, 6, 12. [Google Scholar] [CrossRef]
- Castilho-Fernandes, A.; Lopes, T.G.; Primo, F.L.; Pinto, M.R.; Tedesco, A.C. Photodynamic process induced by chloro-aluminum phthalocyanine nanoemulsion in glioblastoma. Photodiagn. Photodyn. Ther. 2017, 19, 221–228. [Google Scholar] [CrossRef] [PubMed]
- Neagu, M.; Constantin, C.; Tampa, M.; Matei, C.; Lupu, A.; Manole, E.; Ion, R.M.; Fenga, C.; Tsatsakis, A.M. Toxicological and efficacy assessment of post-transition metal (Indium) phthalocyanine for photodynamic therapy in neuroblastoma. Oncotarget 2016, 7, 69718–69732. [Google Scholar] [CrossRef]
- Stylli, S.; Hill, J.; Sawyer, W.; Kaye, A. Aluminium phthalocyanine mediated photodynamic therapy in experimental malignant glioma. J. Clin. Neurosci. 1995, 2, 146–151. [Google Scholar] [CrossRef]
- Velazquez, F.N.; Miretti, M.; Baumgartner, M.T.; Caputto, B.L.; Tempesti, T.C.; Prucca, C.G. Effectiveness of ZnPc and of an amine derivative to inactivate Glioblastoma cells by Photodynamic Therapy: An in vitro comparative study. Sci. Rep. 2019, 9, 3010. [Google Scholar] [CrossRef]
- Kharroubi Lakouas, D.; Huglo, D.; Mordon, S.; Vermandel, M. Nuclear medicine for photodynamic therapy in cancer: Planning, monitoring and nuclear PDT. Photodiagn. Photodyn. Ther. 2017, 18, 236–243. [Google Scholar] [CrossRef]
- Domka, W.; Bartusik-Aebisher, D.; Rudy, I.; Dynarowicz, K.; Pieta, K.; Aebisher, D. Photodynamic therapy in brain cancer: Mechanisms, clinical and preclinical studies and therapeutic challenges. Front. Chem. 2023, 11, 1250621. [Google Scholar] [CrossRef] [PubMed]
- Shibata, S.; Shinozaki, N.; Suganami, A.; Ikegami, S.; Kinoshita, Y.; Hasegawa, R.; Kentaro, H.; Okamoto, Y.; Aoki, I.; Tamura, Y.; et al. Photo-immune therapy with liposomally formulated phospholipid-conjugated indocyanine green induces specific antitumor responses with heat shock protein-70 expression in a glioblastoma model. Oncotarget 2019, 10, 175–183. [Google Scholar] [CrossRef] [PubMed]
- Luiza Andreazza, N.; Vevert-Bizet, C.; Bourg-Heckly, G.; Sureau, F.; Jose Salvador, M.; Bonneau, S. Berberine as a photosensitizing agent for antitumoral photodynamic therapy: Insights into its association to low density lipoproteins. Int. J. Pharm. 2016, 510, 240–249. [Google Scholar] [CrossRef]
- Zhu, X.; Zhou, H.; Liu, Y.; Wen, Y.; Wei, C.; Yu, Q.; Liu, J. Transferrin/aptamer conjugated mesoporous ruthenium nanosystem for redox-controlled and targeted chemo-photodynamic therapy of glioma. Acta Biomater. 2018, 82, 143–157. [Google Scholar] [CrossRef]
- Dong, C.; Yi, Q.; Fang, W.; Zhang, J. A mini review of nanomaterials on photodynamic therapy. J. Photochem. Photobiol. C Photochem. Rev. 2023, 54, 100568. [Google Scholar] [CrossRef]
- Bhaumik, J.; Mittal, A.K.; Banerjee, A.; Chisti, Y.; Banerjee, U.C. Applications of phototheranostic nanoagents in photodynamic therapy. Nano Res. 2015, 8, 1373–1394. [Google Scholar] [CrossRef]
- Silva Adaya, D.; Aguirre-Cruz, L.; Guevara, J.; Ortiz-Islas, E. Nanobiomaterials’ applications in neurodegenerative diseases. J. Biomater. Appl. 2017, 31, 953–984. [Google Scholar] [CrossRef] [PubMed]
- Fatima, J.; Siddique, Y.H. Application of Nanocomposites and Nanoparticles in Treating Neurodegenerative Disorders. CNS Neurol. Disord. Drug Targets 2024, 23, 1217–1233. [Google Scholar] [CrossRef]
- Perrin, C.L.; Agranat, I.; Bagno, A.; Braslavsky, S.E.; Fernandes, P.A.; Gal, J.F.; Lloyd-Jones, G.C.; Mayr, H.; Murdoch, J.R.; Nudelman, N.S.; et al. Glossary of terms used in physical organic chemistry (IUPAC Recommendations 2021). Pure Appl. Chem. 2022, 94, 353–534. [Google Scholar] [CrossRef]
- Pawar, V.; Maske, P.; Khan, A.; Ghosh, A.; Keshari, R.; Bhatt, M.; Srivastava, R. Responsive Nanostructure for Targeted Drug Delivery. J. Nanotheranostics 2023, 4, 55–85. [Google Scholar] [CrossRef]
- Park, W.; Shin, H.; Choi, B.; Rhim, W.K.; Na, K.; Han, D.K. Advanced hybrid nanomaterials for biomedical applications. Prog. Mater. Sci. 2020, 114, 100686. [Google Scholar] [CrossRef]
- Mabrouk, M.; Das, D.B.; Salem, Z.A.; Beherei, H.H. Nanomaterials for Biomedical Applications: Production, Characterisations, Recent Trends and Difficulties. Molecules 2021, 26, 1077. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.T.; Sui, S.Y.; He, Y.X.; Yu, C.H.; Peng, Q. Nanomaterials-based photosensitizers and delivery systems for photodynamic cancer therapy. Biomater. Adv. 2022, 135, 212725. [Google Scholar] [CrossRef]
- Debele, T.A.; Peng, S.; Tsai, H.C. Drug Carrier for Photodynamic Cancer Therapy. Int. J. Mol. Sci. 2015, 16, 22094–22136. [Google Scholar] [CrossRef]
- Calixto, G.M.; Bernegossi, J.; de Freitas, L.M.; Fontana, C.R.; Chorilli, M. Nanotechnology-Based Drug Delivery Systems for Photodynamic Therapy of Cancer: A Review. Molecules 2016, 21, 342. [Google Scholar] [CrossRef] [PubMed]
- Rejinold, N.S.; Choi, G.; Choy, J.H. Recent trends in nano photochemo therapy approaches and future scopes. Coord. Chem. Rev. 2020, 411, 213252. [Google Scholar] [CrossRef]
- Wang, J.; Wu, X.; Shen, P.; Wang, J.; Shen, Y.; Shen, Y.; Webster, T.J.; Deng, J. Applications of Inorganic Nanomaterials in Photothermal Therapy Based on Combinational Cancer Treatment. Int. J. Nanomed. 2020, 15, 1903–1914. [Google Scholar] [CrossRef] [PubMed]
- Lv, Z.; He, S.; Wang, Y.; Zhu, X. Noble Metal Nanomaterials for NIR-Triggered Photothermal Therapy in Cancer. Adv. Healthc. Mater. 2021, 10, e2001806. [Google Scholar] [CrossRef] [PubMed]
- Kwiatkowski, S.; Knap, B.; Przystupski, D.; Saczko, J.; Kedzierska, E.; Knap-Czop, K.; Kotlinska, J.; Michel, O.; Kotowski, K.; Kulbacka, J. Photodynamic therapy—Mechanisms, photosensitizers and combinations. Biomed. Pharmacother. 2018, 106, 1098–1107. [Google Scholar] [CrossRef]
- Ahirwar, S.; Mallick, S.; Bahadur, D. Photodynamic therapy using graphene quantum dot derivatives. J. Solid State Chem. 2020, 282, 121107. [Google Scholar] [CrossRef]
- Lin, Z.; Jiang, B.P.; Liang, J.; Wen, C.; Shen, X.C. Phycocyanin functionalized single-walled carbon nanohorns hybrid for near-infrared light-mediated cancer phototheranostics. Carbon 2019, 143, 814–827. [Google Scholar] [CrossRef]
- Rossi, F.; Bedogni, E.; Bigi, F.; Rimoldi, T.; Cristofolini, L.; Pinelli, S.; Alinovi, R.; Negri, M.; Dhanabalan, S.C.; Attolini, G.; et al. Porphyrin conjugated SiC/SiOx nanowires for X-ray-excited photodynamic therapy. Sci. Rep. 2015, 5, 7606. [Google Scholar] [CrossRef]
- Hao, C.; Wang, X.; Jia, X.; Liu, T.; Sun, J.; Yan, Z. The applications of two-dimensional materials and the derivative quantum dots in photodynamic therapy. APL Mater. 2022, 10, 021104. [Google Scholar] [CrossRef]
- Younis, M.R.; He, G.; Qu, J.; Lin, J.; Huang, P.; Xia, X.H. Inorganic Nanomaterials with Intrinsic Singlet Oxygen Generation for Photodynamic Therapy. Adv. Sci. 2021, 8, e2102587. [Google Scholar] [CrossRef]
- Qin, X.; Tang, Q.; Jiang, X.; Zhang, J.; Wang, B.; Liu, X.; Zhang, Y.; Zou, Z.; Chen, C. Zinc Oxide Nanoparticles Induce Ferroptotic Neuronal Cell Death in vitro and in vivo. Int. J. Nanomed. 2020, 15, 5299–5315. [Google Scholar] [CrossRef] [PubMed]
- Qidwai, A.; Annu; Nabi, B.; Kotta, S.; Narang, J.K.; Baboota, S.; Ali, J. Role of nanocarriers in photodynamic therapy. Photodiagn. Photodyn. Ther. 2020, 30, 101782. [Google Scholar] [CrossRef]
- Rui, L.-L.; Cao, H.-L.; Xue, Y.-D.; Liu, L.-C.; Xu, L.; Gao, Y.; Zhang, W.-A. Functional organic nanoparticles for photodynamic therapy. Chin. Chem. Lett. 2016, 27, 1412–1420. [Google Scholar] [CrossRef]
- Mokwena, M.G.; Kruger, C.A.; Ivan, M.T.; Heidi, A. A review of nanoparticle photosensitizer drug delivery uptake systems for photodynamic treatment of lung cancer. Photodiagn. Photodyn. Ther. 2018, 22, 147–154. [Google Scholar] [CrossRef]
- Lim, C.K.; Heo, J.; Shin, S.; Jeong, K.; Seo, Y.H.; Jang, W.D.; Park, C.R.; Park, S.Y.; Kim, S.; Kwon, I.C. Nanophotosensitizers toward advanced photodynamic therapy of Cancer. Cancer Lett. 2013, 334, 176–187. [Google Scholar] [CrossRef]
- Li, Z.; Zhou, Z.; Wang, Y.; Wang, J.; Zhou, L.; Cheng, H.B.; Yoon, J. Activatable nano-photosensitizers for precise photodynamic cancer therapy. Coord. Chem. Rev. 2023, 493, 215324. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhao, M.; Cheng, D.; Zhu, J.; Cheng, B.; Miao, M.; Li, Q.; Miao, Q. Self-Assembled Type I Nanophotosensitizer with NIR-II Fluorescence Emission for Imaging-Guided Targeted Phototherapy of Glioblastoma. ACS Appl. Nano Mater. 2024, 7, 22117–22129. [Google Scholar] [CrossRef]
- Cesca, B.A.; Caverzan, M.D.; Lamberti, M.J.; Ibarra, L.E. Enhancing Therapeutic Approaches in Glioblastoma with Pro-Oxidant Treatments and Synergistic Combinations: In Vitro Experience of Doxorubicin and Photodynamic Therapy. Int. J. Mol. Sci. 2024, 25, 7525. [Google Scholar] [CrossRef] [PubMed]
- Dixit, S.; Novak, T.; Miller, K.; Zhu, Y.; Kenney, M.E.; Broome, A.M. Transferrin receptor-targeted theranostic gold nanoparticles for photosensitizer delivery in brain tumors. Nanoscale 2015, 7, 1782–1790. [Google Scholar] [CrossRef]
- Barbosa, H.F.G.; Piva, H.L.; Matsuo, F.S.; de Lima, S.C.G.; de Souza, L.E.B.; Osako, M.K.; Tedesco, A.C. Hybrid lipid-biopolymer nanocarrier as a strategy for GBM photodynamic therapy (PDT). Int. J. Biol. Macromol. 2023, 242 Pt 1, 124647. [Google Scholar] [CrossRef]
- Comincini, S.; Manai, F.; Sorrenti, M.; Perteghella, S.; D’Amato, C.; Miele, D.; Catenacci, L.; Bonferoni, M.C. Development of Berberine-Loaded Nanoparticles for Astrocytoma Cells Administration and Photodynamic Therapy Stimulation. Pharmaceutics 2023, 15, 1078. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Zhang, J.Z. Optical properties and applications of hybrid semiconductor nanomaterials. Coord. Chem. Rev. 2009, 253, 3015–3041. [Google Scholar] [CrossRef]
- Escudero, A.; Carrillo-Carrión, C.; Castillejos, M.C.; Romero-Ben, E.; Rosales-Barrios, C.; Khiar, N. Photodynamic therapy: Photosensitizers and nanostructures. Mater. Chem. Front. 2021, 5, 3788–3812. [Google Scholar] [CrossRef]
- Zhang, H.; Shan, Y.; Dong, L. A comparison of TiO2 and ZnO nanoparticles as photosensitizers in photodynamic therapy for cancer. J. Biomed. Nanotechnol. 2014, 10, 1450–1457. [Google Scholar] [CrossRef]
- Reghunath, S.; Pinheiro, D.; Devi KR, S. A review of hierarchical nanostructures of TiO2: Advances and applications. Appl. Surf. Sci. Adv. 2021, 3, 100063. [Google Scholar] [CrossRef]
- Wang, M.; Hou, Z.; Al Kheraif, A.A.; Xing, B.; Lin, J. Mini Review of TiO2-Based Multifunctional Nanocomposites for Near-Infrared Light-Responsive Phototherapy. Adv. Healthc. Mater. 2018, 7, e1800351. [Google Scholar] [CrossRef]
- Parrino, F.P.F.; Camera-Roda, G.; Loddo, V.; Palmisano, L. Properties of titanium dioxide. In Titanium Dioxide (Tio2) and Its Applications, 1st ed.; Parrino, F., Palmisano, L., Eds.; Elsevier: Amsterdam, The Netherlands, 2021; pp. 13–66. [Google Scholar]
- Nyamukamba, P.; Okoh, O.; Mungondori, H.; Taziwa, R.; Zinya, S. Synthetic Methods for Titanium Dioxide Nanoparticles: A Review. In Titanium Dioxide—Material for a Sustainable Environment; Yang, D., Ed.; IntechOpen: London, UK, 2018; pp. 151–175. [Google Scholar]
- Jafari, S.; Mahyad, B.; Hashemzadeh, H.; Janfaza, S.; Gholikhani, T.; Tayebi, L. Biomedical Applications of TiO2 Nanostructures: Recent Advances. Int. J. Nanomed. 2020, 15, 3447–3470. [Google Scholar] [CrossRef] [PubMed]
- Sargazi, S.; Simge, E.R.; Gelen, S.S.; Rahdar, A.; Bilal, M.; Arshad, R.; Ajalli, N.; Khan, M.F.A.; Pandey, S. Application of titanium dioxide nanoparticles in photothermal and photodynamic therapy of cancer: An updated and comprehensive review. J. Drug Deliv. Sci. Technol. 2022, 755, 103605. [Google Scholar] [CrossRef]
- Tonelli, F.M.P.; Tonelli, F.C.P.; Cordeiro, H.G. TiO2 Nanoparticles in Cancer Therapy as Nanocarriers in Paclitaxel’s Delivery and Nanosensitizers in Phototherapies and/or Sonodynamic Therapy. Curr. Pharm. Biotechnol. 2023, 25, 133–143. [Google Scholar] [CrossRef]
- Cesmeli, S.; Biray Avci, C. Application of titanium dioxide (TiO2) nanoparticles in cancer therapies. J. Drug Target. 2019, 27, 762–766. [Google Scholar] [CrossRef]
- Ni, W.; Li, M.; Cui, J.; Xing, Z.; Li, Z.; Wu, X.; Song, E.; Gong, M.; Zhou, W. 808 nm light triggered black TiO2 nanoparticles for killing of bladder cancer cells. Mater. Sci. Eng. C Mater. Biol. Appl. 2017, 81, 252–260. [Google Scholar] [CrossRef]
- Li, J.; Wang, X.; Shao, Y.; Lu, X.; Chen, B. A Novel Exploration of a Combination of Gambogic Acid with TiO2 Nanofibers: The Photodynamic Effect for HepG2 Cell Proliferation. Materials 2014, 7, 6865–6878. [Google Scholar] [CrossRef] [PubMed]
- Zheng, K.; Chen, R.; Sun, Y.; Tan, Z.; Liu, Y.; Cheng, X.; Leng, J.; Guo, Z.; Xu, P. Cantharidin-loaded functional mesoporous titanium peroxide nanoparticles for non-small cell lung cancer targeted chemotherapy combined with high effective photodynamic therapy. Thorac. Cancer 2020, 11, 1476–1486. [Google Scholar] [CrossRef] [PubMed]
- Shah, Z.; Nazir, S.; Mazhar, K.; Abbasi, R.; Samokhvalov, I.M. PEGylated doped- and undoped-TiO(2) nanoparticles for photodynamic Therapy of cancers. Photodiagn. Photodyn. Ther. 2019, 27, 173–183. [Google Scholar] [CrossRef]
- Yurt, F.; Ocakoglu, K.; Ince, M.; Colak, S.G.; Er, O.; Soylu, H.M.; Gunduz, C.; Biray Avci, C.; Caliskan Kurt, C. Photodynamic therapy and nuclear imaging activities of zinc phthalocyanine-integrated TiO2 nanoparticles in breast and cervical tumors. Chem. Biol. Drug Des. 2018, 91, 789–796. [Google Scholar] [CrossRef]
- Huang, K.; Chen, L.; Liao, M.; Xiong, J. The Photocatalytic Inactivation Effect of Fe-Doped TiO2 Nanocomposites on Leukemic HL60 Cells-Based Photodynamic Therapy. Int. J. Photoenergy 2012, 2012, 367072. [Google Scholar] [CrossRef]
- Huang, K.; Chen, L.; Deng, J.; Xiong, J. Enhanced Visible-Light Photocatalytic Performance of Nanosized Anatase TiO2 Doped with CdS Quantum Dots for Cancer-Cell Treatment. J. Nanomater. 2012, 2012, 720491. [Google Scholar] [CrossRef]
- Matijević, M.; Nešić, M.; Stepić, M.; Radoičić, M.; Šaponjić, Z.; Petković, M. Light controllable TiO2-Ru nanocomposite system encapsulated in phospholipid unilamellar vesicles for anti-cancer photodynamic therapy. Opt. Quantum Electron. 2018, 50, 232. [Google Scholar] [CrossRef]
- Jiménez, V.A.; Moreno, N.; Guzmán, L.; Torres, C.C.; Campos, C.H.; Alderete, J.B. Visible-light-responsive folate-conjugated titania and alumina nanotubes for photodynamic therapy applications. J. Mater. Sci. 2020, 55, 6976–6991. [Google Scholar] [CrossRef]
- Dong, M.; Sun, X.; Bu, T.; Zhang, H.; Wang, J.; He, K.; Li, L.; Li, Z. 3D/2D TMSs/TiO2 nanofibers heterojunctions for photodynamic-photothermal and oxidase-like synergistic antibacterial therapy co-driven by VIS and NIR biowindows. Compos. B Eng. 2022, 230, 109498. [Google Scholar] [CrossRef]
- Uribe-Robles, M.; Ortiz-Islas, E.; Rodriguez-Perez, E.; Lim, T.; Martinez-Morales, A.A. TiO2 hollow nanospheres functionalized with folic acid and ZnPc for targeted photodynamic therapy in glioblastoma cancer. MRS Commun. 2019, 9, 1242–1248. [Google Scholar] [CrossRef]
- Xie, J.; Pan, X.; Wang, M.; Yao, L.; Liang, X.; Ma, J.; Fei, Y.; Wang, P.N.; Mi, L. Targeting and Photodynamic Killing of Cancer Cell by Nitrogen-Doped Titanium Dioxide Coupled with Folic Acid. Nanomaterials 2016, 6, 113. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Santos, A.; Evdokiou, A.; Losic, D. An overview of nanotoxicity and nanomedicine research: Principles, progress and implications for cancer therapy. J. Mater. Chem. B 2015, 3, 7153–7172. [Google Scholar] [CrossRef]
- Liu, X.; Sui, B.; Sun, J. Size- and shape-dependent effects of titanium dioxide nanoparticles on the permeabilization of the blood-brain barrier. J. Mater. Chem. B 2017, 5, 9558–9570. [Google Scholar] [CrossRef] [PubMed]
- Hong, F.; Yu, X.; Wu, N.; Zhang, Y.Q. Progress of in vivo studies on the systemic toxicities induced by titanium dioxide nanoparticles. Toxicol. Res. 2017, 6, 115–133. [Google Scholar] [CrossRef]
- Aschner, M.; Skalny, A.V.; Santamaria, A.; Buha Djordjevic, A.; Tizabi, Y.; Jiang, Y.; Lu, R.; Virgolini, M.B.; Tinkov, A.A. From Mechanisms to Implications: Understanding the Molecular Neurotoxicity of Titanium Dioxide Nanoparticles. Front. Biosci. 2023, 28, 204. [Google Scholar] [CrossRef] [PubMed]
- Santonastaso, M.; Mottola, F.; Iovine, C.; Genualdo, V.; Montano, L.; Piscopo, M.; Palmieri, I.; Rocco, L. Protective Effects of Anthocyanin and α-Tocopherol Against Titanium Dioxide Nanoparticle-Induced DNA Damage in Human Sperm Cells. Expo. Health 2025, 17, 523–535. [Google Scholar] [CrossRef]
- Tedja, R.; Marquis, C.; Lim, M.; Amal, R. Biological impacts of TiO2 on human lung cell lines A549 and H1299: Particle size distribution effects. J. Nanopart. Res. 2011, 13, 3801–3813. [Google Scholar] [CrossRef]
- Bacova, J.; Knotek, P.; Kopecka, K.; Hromadko, L.; Capek, J.; Nyvltova, P.; Bruckova, L.; Schroterova, L.; Sestakova, B.; Palarcik, J.; et al. Evaluating the Use of TiO2 Nanoparticles for Toxicity Testing in Pulmonary A549 Cells. Int. J. Nanomed. 2022, 17, 4211–4225. [Google Scholar] [CrossRef]
- Valdiglesias, V.; Costa, C.; Sharma, V.; Kilic, G.; Pasaro, E.; Teixeira, J.P.; Dhawan, A.; Laffon, B. Comparative study on effects of two different types of titanium dioxide nanoparticles on human neuronal cells. Food Chem. Toxicol. 2013, 57, 352–361. [Google Scholar] [CrossRef]
- Hong, F.; Mu, X.; Ze, Y.; Li, W.; Zhou, Y.; Ji, J. Damage to the Blood Brain Barrier Structure and Function from Nano Titanium Dioxide Exposure Involves the Destruction of Key Tight Junction Proteins in the Mouse Brain. J. Biomed. Nanotechnol. 2021, 17, 1068–1078. [Google Scholar] [CrossRef]
- Grissa, I.; ElGhoul, J.; Mrimi, R.; Mir, L.E.; Cheikh, H.B.; Horcajada, P. In deep evaluation of the neurotoxicity of orally administered TiO2 nanoparticles. Brain Res. Bull. 2020, 155, 119–128. [Google Scholar] [CrossRef]
- Wu, T.; Tang, M. The inflammatory response to silver and titanium dioxide nanoparticles in the central nervous system. Nanomedicine 2018, 13, 233–249. [Google Scholar] [CrossRef]
- Krawczynska, A.; Dziendzikowska, K.; Gromadzka-Ostrowska, J.; Lankoff, A.; Herman, A.P.; Oczkowski, M.; Krolikowski, T.; Wilczak, J.; Wojewodzka, M.; Kruszewski, M. Silver and titanium dioxide nanoparticles alter oxidative/inflammatory response and renin-angiotensin system in brain. Food Chem. Toxicol. 2015, 85, 96–105. [Google Scholar] [CrossRef] [PubMed]
- Huerta-Garcia, E.; Perez-Arizti, J.A.; Marquez-Ramirez, S.G.; Delgado-Buenrostro, N.L.; Chirino, Y.I.; Iglesias, G.G.; Lopez-Marure, R. Titanium dioxide nanoparticles induce strong oxidative stress and mitochondrial damage in glial cells. Free Radic. Biol. Med. 2014, 73, 84–94. [Google Scholar] [CrossRef]
- Perez-Arizti, J.A.; Ventura-Gallegos, J.L.; Galvan Juarez, R.E.; Ramos-Godinez, M.D.P.; Colin-Val, Z.; Lopez-Marure, R. Titanium dioxide nanoparticles promote oxidative stress, autophagy and reduce NLRP3 in primary rat astrocytes. Chem.-Biol. Interact. 2020, 317, 108966. [Google Scholar] [CrossRef]
- Kim, I.; Viswanathan, K.; Kasi, G.; Thanakkasaranee, S.; Sadeghi, K.; Seo, J. ZnO Nanostructures in Active Antibacterial Food Packaging: Preparation Methods, Antimicrobial Mechanisms, Safety Issues, Future Prospects, and Challenges. Food Rev. Int. 2020, 38, 537–565. [Google Scholar] [CrossRef]
- Sharma, D.K.; Shukla, S.; Sharma, K.K.; Kumar, V. A review on ZnO: Fundamental properties and applications. Mater. Today Proc. 2022, 49, 3028–3035. [Google Scholar] [CrossRef]
- Martinez-Carmona, M.; Gun’ko, Y.; Vallet-Regi, M. ZnO Nanostructures for Drug Delivery and Theranostic Applications. Nanomaterials 2018, 8, 268. [Google Scholar] [CrossRef]
- Lepot, N.; Van Bael, M.K.; Van den Rul, H.; D’Haen, J.; Peeters, R.; Franco, D.; Mullens, J. Synthesis of ZnO nanorods from aqueous solution. Mater. Lett. 2007, 61, 2624–2627. [Google Scholar] [CrossRef]
- Xi, Y.; Hu, C.G.; Han, X.Y.; Xiong, Y.F.; Gao, P.X.; Liu, G.B. Hydrothermal synthesis of ZnO nanobelts and gas sensitivity property. Solid State Commun. 2007, 141, 506–509. [Google Scholar] [CrossRef]
- Tripathi, R.M.; Bhadwal, A.S.; Gupta, R.K.; Singh, P.; Shrivastav, A.; Shrivastav, B.R. ZnO nanoflowers: Novel biogenic synthesis and enhanced photocatalytic activity. J. Photochem. Photobiol. B 2014, 141, 288–295. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.C.; Cai, W. Synthesis and characterization of ZnO nanorings with ZnO nanowires array aligned at the inner surface without catalyst. J. Cryst. Growth 2008, 310, 843–846. [Google Scholar] [CrossRef]
- Zhang, Y.; Ram, M.K.; Stefanakos, E.K.; Goswami, D.Y. Synthesis, Characterization, and Applications of ZnO Nanowires. J. Nanomater. 2012, 2012, 624520. [Google Scholar] [CrossRef]
- Baskoutas, S. Special Issue: Zinc Oxide Nanostructures: Synthesis and Characterization. Materials 2018, 11, 873. [Google Scholar] [CrossRef]
- Obeng, E.; Feng, J.; Wang, D.; Zheng, D.; Xiang, B.; Shen, J. Multifunctional phototheranostic agent ZnO@Ag for anti-infection through photothermal/photodynamic therapy. Front. Chem. 2022, 10, 1054739. [Google Scholar] [CrossRef]
- Nagi, J.S.; Skorenko, K.; Bernier, W.; Jones, W.E.; Doiron, A.L. Near Infrared-Activated Dye-Linked ZnO Nanoparticles Release Reactive Oxygen Species for Potential Use in Photodynamic Therapy. Materials 2019, 13, 17. [Google Scholar] [CrossRef] [PubMed]
- Liao, C.; Jin, Y.; Li, Y.; Tjong, S.C. Interactions of Zinc Oxide Nanostructures with Mammalian Cells: Cytotoxicity and Photocatalytic Toxicity. Int. J. Mol. Sci. 2020, 21, 6305. [Google Scholar] [CrossRef]
- Li, J.; Guo, D.; Wang, X.; Wang, H.; Jiang, H.; Chen, B. The Photodynamic Effect of Different Size ZnO Nanoparticles on Cancer Cell Proliferation In Vitro. Nanoscale Res. Lett. 2010, 5, 1063–1071. [Google Scholar] [CrossRef]
- Fakhar-e-Alam, M.; Firdous, S.; Atif, M.; Khan, Y.; Zaidi, S.S.Z.; Suleman, R.; Rehman, A.; Khan, R.U.; Nawaz, M.; Ikram, M. The Potential Applications of ZnO Nanoparticles Conjugated with ALA and Photofrin as a Biomarker in HepG2 Cells. Laser Phys. 2011, 21, 2156–2164. [Google Scholar] [CrossRef]
- Raha, S.; Ahmaruzzaman, M. ZnO nanostructured materials and their potential applications: Progress, challenges and perspectives. Nanoscale Adv. 2022, 4, 1868–1925. [Google Scholar] [CrossRef]
- Ali, A.; Suhail, M.; Mathew, S.; Shah, M.A.; Harakeh, S.M.; Ahmad, S.; Kazmi, Z.; Alhamdan, M.A.; Chaudhary, A.; Damanhouri, G.A.; et al. Nanomaterial Induced Immune Responses and Cytotoxicity. J. Nanosci. Nanotechnol. 2016, 16, 40–57. [Google Scholar] [CrossRef] [PubMed]
- Aschner, M.; Skalny, A.V.; Lu, R.; Martins, A.C.; Tsatsakis, A.; Miroshnikov, S.A.; Santamaria, A.; Tinkov, A.A. Molecular mechanisms of zinc oxide nanoparticles neurotoxicity. Chem. Biol. Interact. 2024, 403, 111245. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.; Zhang, P.; Luo, Y.; Xie, H.Q.; Chakraborty, S.; Monikh, F.A.; Bu, L.; Liu, Y.; Ma, Y.; Zhang, Z.; et al. Valsami-Jones. Intranasal exposure to ZnO nanoparticles induces alterations in cholinergic neurotransmission in rat brain. Nano Today 2020, 35, 100977. [Google Scholar] [CrossRef]
- Singh, S. Zinc oxide nanoparticles impacts: Cytotoxicity, genotoxicity, developmental toxicity, and neurotoxicity. Toxicol. Mech. Methods 2019, 29, 300–311. [Google Scholar] [CrossRef]
- Pandurangan, M.; Kim, D.H. In vitro toxicity of zinc oxide nanoparticles: A review. J. Nanopart. Res. 2015, 17, 158. [Google Scholar] [CrossRef]
- Kahil, N.; Abouzeinab, N.S.; Hussein, M.A.A.; Khalil, M.I. Intraperitoneal hepatorenal toxicity of zinc oxide and nickel oxide nanoparticles in rats: A systematic review. Nanotoxicology 2024, 18, 583–598. [Google Scholar] [CrossRef]
- Fujihara, J.; Nishimoto, N. Review of Zinc Oxide Nanoparticles: Toxicokinetics, Tissue Distribution for Various Exposure Routes, Toxicological Effects, Toxicity Mechanism in Mammals, and an Approach for Toxicity Reduction. Biol. Trace Elem. Res. 2024, 202, 9–23. [Google Scholar] [CrossRef] [PubMed]
- Niska, K.; Muszynska, B.; Kowalski, S.; Tomaszewska, E.; Narajczyk, M.; Pawlowska, M.; Majewski, P.W.; Augustin, E.; Inkielewicz-Stepniak, I. Cytotoxicity of ZnO nanoparticles in human melanocyte cells in the presence or absence of UV radiation: A preliminary comparative study with TiO2 nanoparticles. Toxicol. Vitr. 2025, 106, 106051. [Google Scholar] [CrossRef] [PubMed]
- Bakhori, S.K.M.; Mahmud, S.; Mohamad, D.; Masudi, S.A.M.; Seeni, A. Cytotoxicity determination of nano-zinc oxide eugenol on human gingival fibroblast cells. Mater. Chem. Phys. 2021, 268, 124649. [Google Scholar] [CrossRef]
- Mittag, A.; Hoera, C.; Kampfe, A.; Westermann, M.; Kuckelkorn, J.; Schneider, T.; Glei, M. Cellular Uptake and Toxicological Effects of Differently Sized Zinc Oxide Nanoparticles in Intestinal Cells. Toxics 2021, 9, 96. [Google Scholar] [CrossRef]
- Chen, A.; Feng, X.; Sun, T.; Zhang, Y.; An, S.; Shao, L. Evaluation of the effect of time on the distribution of zinc oxide nanoparticles in tissues of rats and mice: A systematic review. IET Nanobiotechnol. 2016, 10, 97–106. [Google Scholar] [CrossRef]
- Chen, A.; Liang, H.; Liu, J.; Ou, L.; Wei, L.; Wu, J.; Lai, X.; Han, X.; Shao, L. Central neurotoxicity induced by the instillation of ZnO and TiO2 nanoparticles through the taste nerve pathway. Nanomedicine 2017, 12, 2453–2470. [Google Scholar] [CrossRef]
- Li, Y.; Lu, Y.; Li, J.; Li, M.; Gou, H.; Sun, X.; Xu, X.; Song, B.; Li, Z.; Ma, Y. Screening of low-toxic zinc oxide nanomaterials and study the apoptosis mechanism of NSC-34 cells. Biotechnol. J. 2024, 19, e2300443. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Jeong, M.S.; Kim, D.Y.; Her, S.; Wie, M.B. Zinc oxide nanoparticles induce lipoxygenase-mediated apoptosis and necrosis in human neuroblastoma SH-SY5Y cells. Neurochem. Int. 2015, 90, 204–214. [Google Scholar] [CrossRef] [PubMed]
- Sruthi, S.; Mohanan, P.V. Investigation on cellular interactions of astrocytes with zinc oxide nanoparticles using rat C6 cell lines. Colloids Surf. B Biointerfaces 2015, 133, 1–11. [Google Scholar] [CrossRef]
- Hamdi, E.; Muniz-Gonzalez, A.B.; Hidouri, S.; Bermejo, A.M.; Sakly, M.; Venero, C.; Amara, S. Prevention of neurotoxicity and cognitive impairment induced by zinc nanoparticles by oral administration of saffron extract. J. Anim. Physiol. Anim. Nutr. 2023, 107, 1473–1494. [Google Scholar] [CrossRef]
- Zhang, J.; Qin, X.; Wang, B.; Xu, G.; Qin, Z.; Wang, J.; Wu, L.; Ju, X.; Bose, D.D.; Qiu, F.; et al. Zinc oxide nanoparticles harness autophagy to induce cell death in lung epithelial cells. Cell Death Dis. 2017, 8, e2954. [Google Scholar] [CrossRef]
- Rafiei, S.; Riazi, G.H.; Afrasiabi, A.; Dadras, A.; Khajeloo, M.; Shahriary, L.; Eskandari, G.; Modaresi, S.M.S. Modaresi. Zinc and copper oxide nanoparticles decrease synaptosomal glutamate uptake: An in vitro study. J. Iran. Chem. Soc. 2015, 12, 87–94. [Google Scholar] [CrossRef]
- Liu, J.; Kang, Y.; Yin, S.; Song, B.; Wei, L.; Chen, L.; Shao, L. Zinc oxide nanoparticles induce toxic responses in human neuroblastoma SHSY5Y cells in a size-dependent manner. Int. J. Nanomed. 2017, 12, 8085–8099. [Google Scholar] [CrossRef] [PubMed]
- Tomak, A.; Cesmeli, S.; Hanoglu, B.D.; Winkler, D.; Oksel Karakus, C. Nanoparticle-protein corona complex: Understanding multiple interactions between environmental factors, corona formation, and biological activity. Nanotoxicology 2021, 15, 1331–1357. [Google Scholar] [CrossRef]
- Garcia-Alvarez, R.; Vallet-Regi, M. Hard and Soft Protein Corona of Nanomaterials: Analysis and Relevance. Nanomaterials 2021, 11, 888. [Google Scholar] [CrossRef]
- Kose, O.; Stalet, M.; Leclerc, L.; Forest, V. Influence of the physicochemical features of TiO2 nanoparticles on the formation of a protein corona and impact on cytotoxicity. RSC Adv. 2020, 10, 43950–43959. [Google Scholar] [CrossRef]
- Poulsen, K.M.; Albright, M.C.; Niemuth, N.J.; Tighe, R.M.; Payne, C.K. Interaction of TiO2 nanoparticles with lung fluid proteins and the resulting macrophage inflammatory response. Environ. Sci. Nano 2023, 10, 2427–2436. [Google Scholar] [CrossRef]
- Bhunia, A.K.; Saha, S.; Kamilya, T. Microscopic and spectroscopic study of the corona formation and unfolding of human haemoglobin in presence of ZnO nanoparticles. Luminescence 2020, 35, 144–155. [Google Scholar] [CrossRef]
- Hsiao, I.L.; Huang, Y.J. Effects of serum on cytotoxicity of nano- and micro-sized ZnO particles. J. Nanopart. Res. 2013, 15, 1829. [Google Scholar] [CrossRef]
- Shaikh, A.J. Molecular targeting agents in cancer therapy: Science and society. Asian Pac. J. Cancer Prev. 2012, 13, 1705–1708. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Qin, W.; Huang, G.; Chen, Z.; Zhang, Y. Nanomaterials in Targeting Cancer Stem Cells for Cancer Therapy. Front. Pharmacol. 2017, 8, 1. [Google Scholar] [CrossRef] [PubMed]
- Pan, Y.; Ma, S.; Cao, K.; Zhou, S.; Zhao, A.; Li, M.; Qian, F.; Zhu, C. Therapeutic approaches targeting cancer stem cells. J. Cancer Res. Ther. 2018, 14, 1469–1475. [Google Scholar] [CrossRef]
- Dwivedi, A.R.; Thakur, A.; Kumar, V.; Skvortsova, I.; Kumar, V. Targeting Cancer Stem Cells Pathways for the Effective Treatment of Cancer. Curr. Drug Targets 2020, 21, 258–278. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Ning, Q.; Mo, Z.; Tang, S. A promising cancer diagnosis and treatment strategy: Targeted cancer therapy and imaging based on antibody fragment. Artif. Cells Nanomed. Biotechnol. 2019, 47, 3621–3630. [Google Scholar] [CrossRef]
- Kholodenko, R.V.; Kalinovsky, D.V.; Doronin, I.I.; Ponomarev, E.D.; Kholodenko, I.V. Antibody Fragments as Potential Biopharmaceuticals for Cancer Therapy: Success and Limitations. Curr. Med. Chem. 2019, 26, 396–426. [Google Scholar] [CrossRef]
- Kang, H.; Hu, S.; Cho, M.H.; Hong, S.H.; Choi, Y.; Choi, H.S. Theranostic Nanosystems for Targeted Cancer Therapy. Nano Today 2018, 23, 59–72. [Google Scholar] [CrossRef]
- Raj, S.; Khurana, S.; Choudhari, R.; Kesari, K.K.; Kamal, M.A.; Garg, N.; Ruokolainen, J.; Das, B.C.; Kumar, D. Specific targeting cancer cells with nanoparticles and drug delivery in cancer therapy. Semin. Cancer Biol. 2021, 69, 166–177. [Google Scholar] [CrossRef]
- Cheng, Z.; Li, M.; Dey, R.; Chen, Y. Nanomaterials for cancer therapy: Current progress and perspectives. J. Hematol. Oncol. 2021, 14, 85. [Google Scholar] [CrossRef]
- Marfavi, Z.H.; Farhadi, M.; Jameie, S.B.; Zahmatkeshan, M.; Pirhajati, V.; Jameie, M. Glioblastoma U-87MG tumour cells suppressed by ZnO folic acid-conjugated nanoparticles: An in vitro study. Artif. Cells Nanomed. Biotechnol. 2019, 47, 2783–2790. [Google Scholar] [CrossRef]
- Sadri, A.; Changizi, V.; Eivazadeh, N. Evaluation of glioblastoma (U87) treatment with ZnO nanoparticle and X-ray in spheroid culture model using MTT assay. Radiat. Phys. Chem. 2015, 115, 17–21. [Google Scholar] [CrossRef]
- Zheng, M.; Wang, S.; Liu, Z.; Xie, L.; Deng, Y. Development of temozolomide coated nano zinc oxide for reversing the resistance of malignant glioma stem cells. Mater. Sci. Eng. C Mater. Biol. Appl. 2018, 83, 44–50. [Google Scholar] [CrossRef]
- Narmani, A.; Rezvani, M.; Farhood, B.; Darkhor, P.; Mohammadnejad, J.; Amini, B.; Refahi, S.; Abdi Goushbolagh, N. Folic acid functionalized nanoparticles as pharmaceutical carriers in drug delivery systems. Drug Dev. Res. 2019, 80, 404–424. [Google Scholar] [CrossRef]
- Movileanu, C.; Anghelache, M.; Turtoi, M.; Voicu, G.; Neacsu, I.A.; Ficai, D.; Trusca, R.; Oprea, O.; Ficai, A.; Andronescu, E.; et al. Folic acid-decorated PEGylated magnetite nanoparticles as efficient drug carriers to tumor cells overexpressing folic acid receptor. Int. J. Pharm. 2022, 625, 122064. [Google Scholar] [CrossRef]
- Low, P.S.; Kularatne, S.A. Folate-targeted therapeutic and imaging agents for cancer. Curr. Opin. Chem. Biol. 2009, 13, 256–262. [Google Scholar] [CrossRef]
- Aung, W.; Tsuji, A.B.; Hanaoka, K.; Higashi, T. Folate receptor-targeted near-infrared photodynamic therapy for folate receptor-overexpressing tumors. World J. Clin. Oncol. 2022, 13, 880–895. [Google Scholar] [CrossRef] [PubMed]
- McCord, E.; Pawar, S.; Koneru, T.; Tatiparti, K.; Sau, S.; Iyer, A.K. Folate Receptors’ Expression in Gliomas May Possess Potential Nanoparticle-Based Drug Delivery Opportunities. ACS Omega 2021, 6, 4111–4118. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Li, F.; Yi, J.; Gu, C.; Fan, L.; Qiao, Y.; Tao, Y.; Cheng, C.; Wu, H. Folate-decorated maleilated pullulan-doxorubicin conjugate for active tumor-targeted drug delivery. Eur. J. Pharm. Sci. 2011, 42, 517–526. [Google Scholar] [CrossRef] [PubMed]
- Ahmadi, M.; Ritter, C.A.; von Woedtke, T.; Bekeschus, S.; Wende, K. Package delivered: Folate receptor-mediated transporters in cancer therapy and diagnosis. Chem. Sci. 2024, 15, 1966–2006. [Google Scholar] [CrossRef]
- Song, C.; Wang, G.Y.; Kong, D.M. A facile fluorescence method for versatile biomolecular detection based on pristine alpha-Fe2O3 nanoparticle-induced fluorescence quenching. Biosens. Bioelectron. 2015, 68, 239–244. [Google Scholar] [CrossRef]
- Affatigato, L.; Licciardi, M.; Bonamore, A.; Martorana, A.; Incocciati, A.; Boffi, A.; Militello, V. Ferritin-Coated SPIONs as New Cancer Cell Targeted Magnetic Nanocarrier. Molecules 2023, 28, 1163. [Google Scholar] [CrossRef] [PubMed]
- Laurent, S.; Saei, A.A.; Behzadi, S.; Panahifar, A.; Mahmoudi, M. Superparamagnetic iron oxide nanoparticles for delivery of therapeutic agents: Opportunities and challenges. Expert Opin. Drug Deliv. 2014, 11, 1449–1470. [Google Scholar] [CrossRef]
- Huang, P.; Wang, C.; Deng, H.; Zhou, Y.; Chen, X. Surface Engineering of Nanoparticles toward Cancer Theranostics. Acc. Chem. Res. 2023, 56, 1766–1779. [Google Scholar] [CrossRef]
- Castillo, J.J.; Rindzevicius, T.; Rozo, C.E.; Boisen, A. Adsorption and vibrational study of folic acid on gold nanopillar structures using surface-enhanced Raman scattering spectroscopy. Nanomater. Nanotechnol. 2015, 5, 29. [Google Scholar] [CrossRef]
- Kanwal, U.; Irfan Bukhari, N.; Ovais, M.; Abass, N.; Hussain, K.; Raza, A. Advances in nano-delivery systems for doxorubicin: An updated insight. J. Drug Target. 2018, 26, 296–310. [Google Scholar] [CrossRef] [PubMed]
- Krais, A.; Wortmann, L.; Hermanns, L.; Feliu, N.; Vahter, M.; Stucky, S.; Mathur, S.; Fadeel, B. Targeted uptake of folic acid-functionalized iron oxide nanoparticles by ovarian cancer cells in the presence but not in the absence of serum. Nanomedicine 2014, 10, 1421–1431. [Google Scholar] [CrossRef]
- Hsu, T.I.; Chen, Y.P.; Zhang, R.L.; Chen, Z.A.; Wu, C.H.; Chang, W.C.; Mou, C.Y.; Chan, H.W.; Wu, S.H. Overcoming the Blood-Brain Tumor Barrier with Docetaxel-Loaded Mesoporous Silica Nanoparticles for Treatment of Temozolomide-Resistant Glioblastoma. ACS Appl. Mater. Interfaces 2024, 16, 21722–21735. [Google Scholar] [CrossRef]
- Khosravian, P.; Shafiee Ardestani, M.; Khoobi, M.; Ostad, S.N.; Dorkoosh, F.A.; Akbari Javar, H.; Amanlou, M. Mesoporous silica nanoparticles functionalized with folic acid/methionine for active targeted delivery of docetaxel. Onco Targets Ther. 2016, 9, 7315–7330. [Google Scholar] [CrossRef] [PubMed]
- Jardon-Guadarrama, G.; Manriquez-Ramirez, M.E.; Rodriguez-Perez, C.E.; Diaz-Ruiz, A.; de Los Angeles Martinez-Cardenas, M.; Mata-Bermudez, A.; Rios, C.; Ortiz-Islas, E. TiO2-ZnPc nanoparticles functionalized with folic acid as a target photosensitizer for photodynamic therapy against glioblastoma cells. J. Mater. Sci. Mater. Med. 2024, 35, 51. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ortiz-Islas, E.; Manríquez-Ramírez, M.E.; Montes, P.; Rodríguez-Pérez, C.E.; Ruiz-Sanchez, E.; Carvajal-Aguilera, K.; Campos-Peña, V. Photodynamic Therapy for Glioblastoma: Potential Application of TiO2 and ZnO Nanoparticles as Photosensitizers. Pharmaceutics 2025, 17, 1132. https://doi.org/10.3390/pharmaceutics17091132
Ortiz-Islas E, Manríquez-Ramírez ME, Montes P, Rodríguez-Pérez CE, Ruiz-Sanchez E, Carvajal-Aguilera K, Campos-Peña V. Photodynamic Therapy for Glioblastoma: Potential Application of TiO2 and ZnO Nanoparticles as Photosensitizers. Pharmaceutics. 2025; 17(9):1132. https://doi.org/10.3390/pharmaceutics17091132
Chicago/Turabian StyleOrtiz-Islas, Emma, María Elena Manríquez-Ramírez, Pedro Montes, Citlali Ekaterina Rodríguez-Pérez, Elizabeth Ruiz-Sanchez, Karla Carvajal-Aguilera, and Victoria Campos-Peña. 2025. "Photodynamic Therapy for Glioblastoma: Potential Application of TiO2 and ZnO Nanoparticles as Photosensitizers" Pharmaceutics 17, no. 9: 1132. https://doi.org/10.3390/pharmaceutics17091132
APA StyleOrtiz-Islas, E., Manríquez-Ramírez, M. E., Montes, P., Rodríguez-Pérez, C. E., Ruiz-Sanchez, E., Carvajal-Aguilera, K., & Campos-Peña, V. (2025). Photodynamic Therapy for Glioblastoma: Potential Application of TiO2 and ZnO Nanoparticles as Photosensitizers. Pharmaceutics, 17(9), 1132. https://doi.org/10.3390/pharmaceutics17091132





