Developing In Vitro–In Vivo Correlation for Bicalutamide Immediate-Release Dosage Forms with the Biphasic In Vitro Dissolution Test
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Single-Phase Dissolution Test
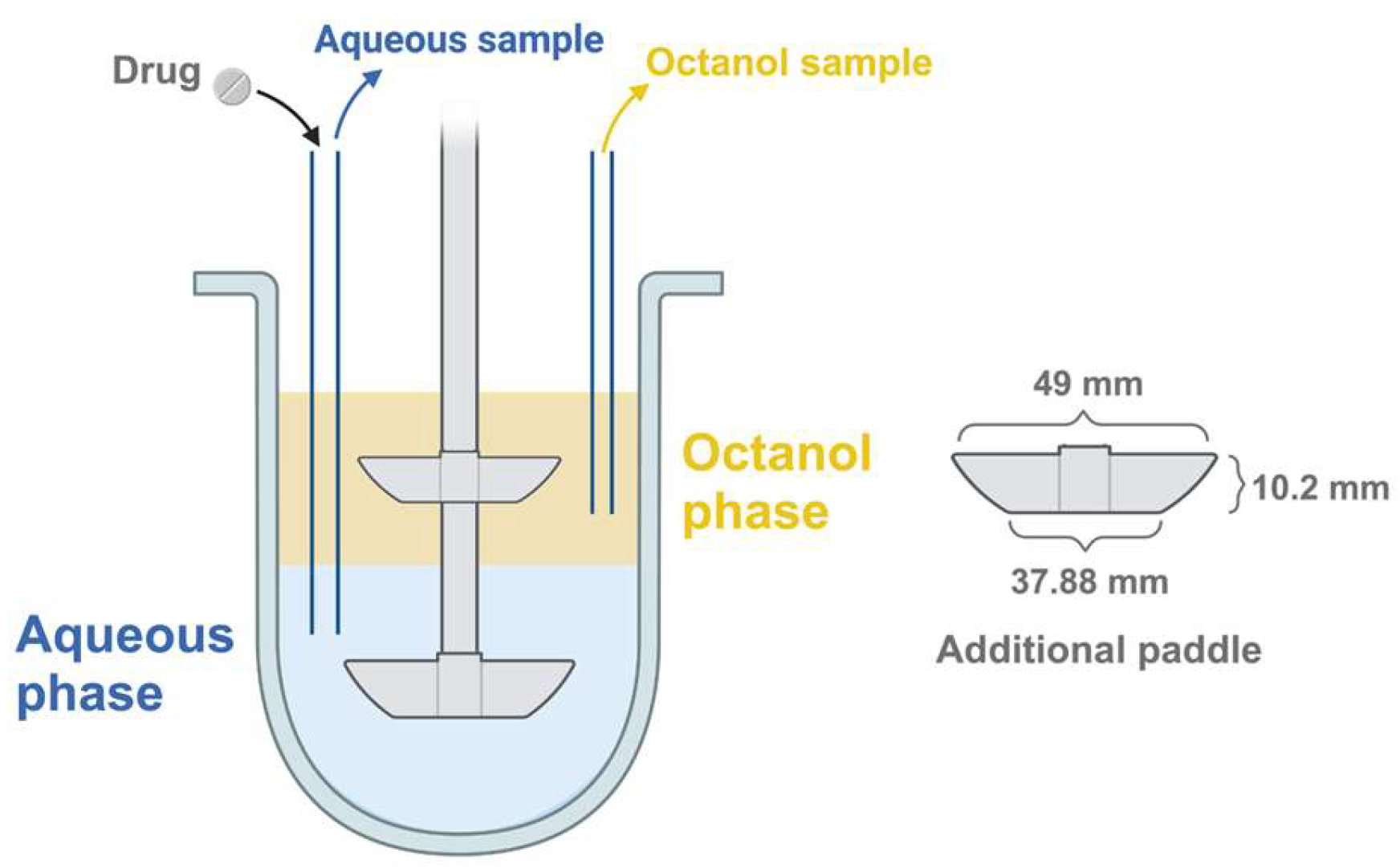
2.3. Biphasic Dissolution Test
2.4. Analytical Method
2.5. Prediction of Human Plasma Profiles with the Biphasic Dissolution Data
2.6. Data Analysis
3. Results
3.1. Single-Phase Dissolution Test
3.2. Biphasic Dissolution Test
3.3. Correlation Between Absorption and Partition Profiles
3.4. In Vivo Prediction with the Biphasic Dissolution Data
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Krollik, K.; Lehmann, A.; Wagner, C.; Kaidas, J.; Kubas, H.; Weitschies, W. The Effect of Buffer Species on Biorelevant Dissolution and Precipitation Assays-Comparison of Phosphate and Bicarbonate Buffer. Eur. J. Pharm. Biopharm. 2022, 171, 90–101. [Google Scholar] [CrossRef]
- Grady, H.; Elder, D.; Webster, G.K.; Mao, Y.; Lin, Y.; Flanagan, T.; Mann, J.; Blanchard, A.; Cohen, M.J.; Lin, J.; et al. Industry’s View on Using Quality Control, Biorelevant, and Clinically Relevant Dissolution Tests for Pharmaceutical Development, Registration, and Commercialization. J. Pharm. Sci. 2018, 107, 34–41. [Google Scholar] [CrossRef]
- Amaral Silva, D.; Al-Gousous, J.; Davies, N.M.; Bou Chacra, N.; Webster, G.K.; Lipka, E.; Amidon, G.L.; Löbenberg, R. Biphasic Dissolution as an Exploratory Method during Early Drug Product Development. Pharmaceutics 2020, 12, 420. [Google Scholar] [CrossRef] [PubMed]
- Jereb, R.; Opara, J.; Bajc, A.; Petek, B. Evaluating the Impact of Physiological Properties of the Gastrointestinal Tract on Drug In Vivo Performance Using Physiologically Based Biopharmaceutics Modeling and Virtual Clinical Trials. J. Pharm. Sci. 2021, 110, 3069–3081. [Google Scholar] [CrossRef]
- Hens, B.; Sinko, P.D.; Job, N.; Dean, M.; Al-Gousous, J.; Salehi, N.; Ziff, R.M.; Tsume, Y.; Bermejo, M.; Paixão, P.; et al. Formulation Predictive Dissolution (fPD) Testing to Advance Oral Drug Product Development: An Introduction to the US FDA Funded ‘21st Century BA/BE’ Project. Int. J. Pharm. 2018, 548, 120–127. [Google Scholar] [CrossRef]
- Pathak, S.M.; Schaefer, K.J.; Jamei, M.; Turner, D.B. Biopharmaceutic IVIVE-Mechanistic Modeling of Single- and Two-Phase In Vitro Experiments to Obtain Drug-Specific Parameters for Incorporation into PBPK Models. J. Pharm. Sci. 2019, 108, 1604–1618. [Google Scholar] [CrossRef]
- Phillips, D.J.; Pygall, S.R.; Cooper, V.B.; Mann, J.C. Overcoming Sink Limitations in Dissolution Testing: A Review of Traditional Methods and the Potential Utility of Biphasic Systems. J. Pharm. Pharmacol. 2012, 64, 1549–1559. [Google Scholar] [CrossRef]
- Niebergall, P.J.; Patil, M.Y.; Sugita, E.T. Simultaneous Determination of Dissolution and Partitioning Rates In Vitro. J. Pharm. Sci. 1967, 56, 943–947. [Google Scholar] [CrossRef] [PubMed]
- Hoa, N.T.; Kinget, R. Design and Evaluation of Two-Phase Partition-Dissolution Method and Its Use in Evaluating Artemisinin Tablets. J. Pharm. Sci. 1996, 85, 1060–1063. [Google Scholar] [CrossRef] [PubMed]
- Gabriels, M.; Plaizier-Vercammen, J. Design of a Dissolution System for the Evaluation of the Release Rate Characteristics of Artemether and Dihydroartemisinin from Tablets. Int. J. Pharm. 2004, 274, 245–260. [Google Scholar] [CrossRef]
- Vangani, S.; Li, X.; Zhou, P.; Del-Barrio, M.-A.; Chiu, R.; Cauchon, N.; Gao, P.; Medina, C.; Jasti, B. Dissolution of Poorly Water-Soluble Drugs in Biphasic Media Using USP 4 and Fiber Optic System. Clin. Res. Regul. Aff. 2009, 26, 8–19. [Google Scholar] [CrossRef]
- Panchagnula, R.; Thomas, N.S. Biopharmaceutics and Pharmacokinetics in Drug Research. Int. J. Pharm. 2000, 201, 131–150. [Google Scholar] [CrossRef] [PubMed]
- Grundy, J.S.; Anderson, K.E.; Rogers, J.A.; Foster, R.T. Studies on Dissolution Testing of the Nifedipine Gastrointestinal Therapeutic System. I. Description of a Two-Phase In Vitro Dissolution Test. J. Control. Release 1997, 48, 1–8. [Google Scholar] [CrossRef]
- Gibaldi, M.; Feldman, S. Establishment of Sink Conditions in Dissolution Rate Determinations. Theoretical Considerations and Application to Nondisintegrating Dosage Forms. J. Pharm. Sci. 1967, 56, 1238–1242. [Google Scholar] [CrossRef]
- Stead, J.A.; Freeman, M.; John, E.G.; Ward, G.T.; Whiting, B. Ibuprofen Tablets: Dissolution and Bioavailability Studies. Int. J. Pharm. 1983, 14, 59–72. [Google Scholar] [CrossRef]
- Frank, K.J.; Locher, K.; Zecevic, D.E.; Fleth, J.; Wagner, K.G. In Vivo Predictive Mini-Scale Dissolution for Weak Bases: Advantages of pH-Shift in Combination with an Absorptive Compartment. Eur. J. Pharm. Sci. 2014, 61, 32–39. [Google Scholar] [CrossRef]
- Locher, K.; Borghardt, J.M.; Frank, K.J.; Kloft, C.; Wagner, K.G. Evolution of a Mini-Scale Biphasic Dissolution Model: Impact of Model Parameters on Partitioning of Dissolved API and Modelling of In Vivo-Relevant Kinetics. Eur. J. Pharm. Biopharm. 2016, 105, 166–175. [Google Scholar] [CrossRef]
- O’Dwyer, P.J.; Box, K.J.; Imanidis, G.; Vertzoni, M.; Reppas, C. On the Usefulness of Four In Vitro Methods in Assessing the Intraluminal Performance of Poorly Soluble, Ionisable Compounds in the Fasted State. Eur. J. Pharm. Sci. 2022, 168, 106034. [Google Scholar] [CrossRef]
- Blackledge, G.R.P. Clinical Progress with a New Antiandrogen, Casodex™ (Bicalutamide). Eur. Urol. 1996, 29 (Suppl. S2), 96–104. [Google Scholar] [CrossRef] [PubMed]
- Cockshott, I.D. Bicalutamide: Clinical Pharmacokinetics and Metabolism. Clin. Pharmacokinet. 2004, 43, 855–878. [Google Scholar] [CrossRef] [PubMed]
- McKillop, D.; Boyle, G.W.; Cockshott, I.D.; Jones, D.C.; Phillips, P.J.; Yates, R.A. Metabolism and Enantioselective Pharmacokinetics of Casodex in Man. Xenobiotica 1993, 23, 1241–1253. [Google Scholar] [CrossRef] [PubMed]
- Boyle, G.W.; McKillop, D.; Phillips, P.J.; Harding, J.R.; Pickford, R.; McCormick, A.D. Metabolism of Casodex in Laboratory Animals. Xenobiotica 1993, 23, 781–798. [Google Scholar] [CrossRef]
- Wellington, K.; Keam, S.J. Bicalutamide 150 mg: A Review of Its Use in the Treatment of Locally Advanced Prostate Cancer. Drugs 2006, 66, 837–850. [Google Scholar] [CrossRef]
- Golfar, Y.; Shayanfar, A. Prediction of Biopharmaceutical Drug Disposition Classification System (BDDCS) by Structural Parameters. J. Pharm Pharm Sci. 2019, 22, 247–269. [Google Scholar] [CrossRef]
- Bamfo, N.O.; Hosey-Cojocari, C.; Benet, L.Z.; Remsberg, C.M. Examination of Urinary Excretion of Unchanged Drug in Humans and Preclinical Animal Models: Increasing the Predictability of Poor Metabolism in Humans. Pharm Res. 2021, 38, 1139–1156. [Google Scholar] [CrossRef]
- Pokharkar, V.B.; Malhi, T.; Mandpe, L. Bicalutamide nanocrystals with improved oral bioavailability: In vitro and in vivo evaluation. Pharm. Dev. Technol. 2013, 18, 660–666. [Google Scholar] [CrossRef]
- Incecayir, T.; Demir, M.E. In Vivo Relevance of a Biphasic In Vitro Dissolution Test for the Immediate Release Tablet Formulations of Lamotrigine. Pharmaceutics 2023, 15, 2474. [Google Scholar] [CrossRef] [PubMed]
- Volkova, T.V.; Simonova, O.R.; Perlovich, G.L. Physicochemical Profile of Antiandrogen Drug Bicalutamide: Solubility, Distribution, Permeability. Pharmaceutics 2022, 14, 674. [Google Scholar] [CrossRef] [PubMed]
- Wagner, J.G. Estimation of Theophylline Absorption Rate by Means of the Wagner-Nelson Equation. J. Allergy Clin. Immunol. 1986, 78, 681–688. [Google Scholar] [CrossRef]
- Wagner, J.G. Fundamentals of Clinical Pharmacokinetics, 2nd ed.; Drug Intelligence Publications: Hamilton, IL, USA, 1979; pp. 174–176. [Google Scholar]
- United States Food and Drug Administration, Department of Health and Human Services, Center for Drug Evaluation and Research (CDER). FDA Guidance. In Dissolution Testing of Immediate Release Solid Oral Dosage Forms, Guidance for Industry; U.S. Government Printing Office: Washington, DC, USA, 1997. [Google Scholar]
- The United States Pharmacopeial Convention. USP Dissolution Methods Database; The United States Pharmacopeial Convention: North Bethesda, MD, USA, 2025; Available online: https://www.usp.org/resources/dissolution-methods-database (accessed on 14 March 2025).
- Tsume, Y.; Mudie, D.M.; Langguth, P.; Amidon, G.E.; Amidon, G.L. The Biopharmaceutics Classification System: Subclasses for In Vivo Predictive Dissolution (IPD) Methodology and IVIVC. Eur. J. Pharm. Sci. 2014, 57, 152–163. [Google Scholar] [CrossRef]
- Tsume, Y.; Igawa, N.; Drelich, A.J.; Ruan, H.; Amidon, G.E.; Amidon, G.L. The In Vivo Predictive Dissolution for Immediate Release Dosage of Donepezil and Danazol, BCS Class IIc Drugs, with the GIS and the USP II with Biphasic Dissolution Apparatus. J. Drug Deliv. Sci. Technol. 2020, 56, 100920. [Google Scholar] [CrossRef]
- Deng, J.; Staufenbiel, S.; Hao, S.; Wang, B.; Dashevskiy, A.; Bodmeier, R. Development of a Discriminative Biphasic In Vitro Dissolution Test and Correlation with In Vivo Pharmacokinetic Studies for Differently Formulated Racecadotril Granules. J. Control. Release 2017, 255, 202–209. [Google Scholar] [CrossRef] [PubMed]
- U.S. Food and Drug Administration (FDA). Center for Drug Evaluation and Research (CDER). Guidance for Industry: Statistical Approaches to Establishing Bioequivalence. 2001. Available online: https://www.fda.gov/media/70958/download (accessed on 24 June 2025).
- Al Durdunji, A.; AlKhatib, H.S.; Al-Ghazawi, M. Development of a Biphasic Dissolution Test for Deferasirox Dispersible Tablets and Its Application in Establishing an In Vitro-In Vivo Correlation. Eur. J. Pharm. Biopharm. 2016, 102, 9–18. [Google Scholar] [CrossRef]
- Xu, H.; Vela, S.; Shi, Y.; Marroum, P.; Gao, P. In Vitro Characterization of Ritonavir Drug Products and Correlation to Human In Vivo Performance. Mol. Pharm. 2017, 14, 3801–3814. [Google Scholar] [CrossRef]
- Xu, H.; Shi, Y.; Vela, S.; Marroum, P.; Gao, P. Developing Quantitative In Vitro-In Vivo Correlation for Fenofibrate Immediate-Release Formulations with the Biphasic Dissolution-Partition Test Method. J. Pharm. Sci. 2018, 107, 476–487. [Google Scholar] [CrossRef]
- Denninger, A.; Westedt, U.; Wagner, K.G. Shared IVIVR for Five Commercial Enabling Formulations Using the BiPHa+ Biphasic Dissolution Assay. Pharmaceutics 2021, 13, 285. [Google Scholar] [CrossRef] [PubMed]






| Subject | ka (h−1) 1,# | kd (h−1) 2,# | Vd/FF* 3,# (mL) |
|---|---|---|---|
| 1 | 0.0485 | 0.00910 | 55,948 |
| 2 | 0.247 | 0.00570 | 48,685 |
| 3 | 0.0321 | 0.00830 | 38,374 |
| 4 | 0.608 | 0.00750 | 54,048 |
| 5 | 0.290 | 0.00580 | 43,811 |
| Mean SD 4 | 0.245 0.233 | 0.00730 0.0015 | 48,173 7251 |
| CV% 5 | 95.1 | 20.5 | 15.0 |
| Bioavailability Criteria | Test | Reference |
|---|---|---|
| Mean ± SD 1 | Mean ± SD | |
| AUC2 (µg/mL.h) | 157 ± 41.0 | 151 ± 40.3 ° |
| Cmax 3 (ng/mL) | 815 ± 175 | 855 ± 167 * |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ozaksun, N.T.; Incecayir, T. Developing In Vitro–In Vivo Correlation for Bicalutamide Immediate-Release Dosage Forms with the Biphasic In Vitro Dissolution Test. Pharmaceutics 2025, 17, 1126. https://doi.org/10.3390/pharmaceutics17091126
Ozaksun NT, Incecayir T. Developing In Vitro–In Vivo Correlation for Bicalutamide Immediate-Release Dosage Forms with the Biphasic In Vitro Dissolution Test. Pharmaceutics. 2025; 17(9):1126. https://doi.org/10.3390/pharmaceutics17091126
Chicago/Turabian StyleOzaksun, Nihal Tugce, and Tuba Incecayir. 2025. "Developing In Vitro–In Vivo Correlation for Bicalutamide Immediate-Release Dosage Forms with the Biphasic In Vitro Dissolution Test" Pharmaceutics 17, no. 9: 1126. https://doi.org/10.3390/pharmaceutics17091126
APA StyleOzaksun, N. T., & Incecayir, T. (2025). Developing In Vitro–In Vivo Correlation for Bicalutamide Immediate-Release Dosage Forms with the Biphasic In Vitro Dissolution Test. Pharmaceutics, 17(9), 1126. https://doi.org/10.3390/pharmaceutics17091126









