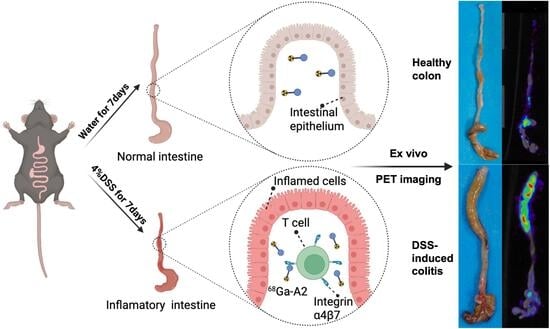
A Novel 68Ga-Labeled Integrin α4β7-Targeted Radiopharmaceutical for PET/CT Imaging of DSS-Induced Murine Colitis
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents and Materials
2.2. 68Ga Radiolabeling and Quality Control of 68Ga-A2
2.3. Determination of the Partition Coefficient (LogD) and in Vitro Stability of 68Ga-A2
2.4. In Vitro Binding Specificity and Affinity Determination
2.5. Establishment of DSS-Induced Colitis Murine Model
2.6. PET/CT Imaging and Ex Vivo Biodistribution Study in DSS-Induced Murine Colitis Model
2.7. Hematoxylin-Eosin (HE) Staining and Immunohistochemical (IHC) Analysis of Colon Tissues
2.8. Statistical Analysis
2.9. Utilization of GenAI Tools in This Study
3. Results
3.1. 68Ga-Radiolabeling and In Vitro Stability of 68Ga-A2
3.2. In Vitro Characterization of 68Ga-A2
3.3. Induction and Confirmation of DSS-Induced Colitis
3.4. PET/CT Imaging of 68Ga-A2 in DSS-Induced Colitis Model
3.5. Ex Vivo Biodistribution Study of 68Ga-A2 in DSS-Induced Colitis Model
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Vieujean, S.; Jairath, V.; Peyrin-Biroulet, L.; Dubinsky, M.; Iacucci, M.; Magro, F.; Danese, S. Understanding the Therapeutic Toolkit for Inflammatory Bowel Disease. Nat. Rev. Gastroenterol. Hepatol. 2025, 22, 371–394. [Google Scholar] [CrossRef]
- Ramos, G.P.; Papadakis, K.A. Mechanisms of Disease: Inflammatory Bowel Diseases. Mayo Clin. Proc. 2019, 94, 155–165. [Google Scholar] [CrossRef]
- Lamb, C.A.; O’Byrne, S.; Keir, M.E.; Butcher, E.C. Gut-Selective Integrin-Targeted Therapies for Inflammatory Bowel Disease. J. Crohns Colitis 2018, 12, S653–S668. [Google Scholar] [CrossRef]
- Hong, S.M.; Baek, D.H. Diagnostic Procedures for Inflammatory Bowel Disease: Laboratory, Endoscopy, Pathology, Imaging, and Beyond. Diagnostics 2024, 14, 1384. [Google Scholar] [CrossRef]
- Nett, A.; Velayos, F.; McQuaid, K. Quality Bowel Preparation for Surveillance Colonoscopy in Patients with Inflammatory Bowel Disease Is a Must. Gastrointest. Endosc. Clin. N. Am. 2014, 24, 379–392. [Google Scholar] [CrossRef]
- Molinié, F.; Gower-Rousseau, C.; Yzet, T.; Merle, V.; Grandbastien, B.; Marti, R.; Lerebours, E.; Dupas, J.-L.; Colombel, J.-F.; Salomez, J.-L.; et al. Opposite Evolution in Incidence of Crohn’s Disease and Ulcerative Colitis in Northern France (1988–1999). Gut 2004, 53, 843–848. [Google Scholar] [CrossRef]
- Dmochowska, N.; Wardill, H.R.; Hughes, P.A. Advances in Imaging Specific Mediators of Inflammatory Bowel Disease. Int. J. Mol. Sci. 2018, 19, 2471. [Google Scholar] [CrossRef]
- Rezazadeh, F.; Kilcline, A.P.; Viola, N.T. Imaging Agents for PET of Inflammatory Bowel Disease: A Review. J. Nucl. Med. 2023, 64, 1858–1864. [Google Scholar] [CrossRef] [PubMed]
- Salehi Farid, A.; Rowley, J.E.; Allen, H.H.; Kruger, I.G.; Tavakolpour, S.; Neeley, K.; Cong, M.; Shahbazian, H.; Dorafshani, N.; Berrada, A.; et al. CD45-PET Is a Robust, Non-Invasive Tool for Imaging Inflammation. Nature 2025, 639, 214–224. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Ran, J.; Hu, X.; Lv, C.; You, J.; Sun, D.; Chen, L.; Tang, Y.; Li, H.; Hu, D.; et al. 18F-FAPI PET/CT for Early Detection and Severity Assessment of Intestinal Fibrosis in a Mouse Model. Inflamm. Bowel Dis. 2025, 31, 2019–2026. [Google Scholar] [CrossRef] [PubMed]
- Pan, Q.; Xu, H.; Liu, S.; Zhang, H.; Zhang, S.; Li, J.; Li, F.; Luo, Y. Head-to-Head Comparison of 68Ga-FAPI-04 and 18F-FDG PET/CT for the Assessment of Crohn’s Disease: A Prospective Pilot Study. Clin. Nucl. Med. 2025, 50, 473. [Google Scholar] [CrossRef]
- Li, X.; Liu, Y.; Zhang, Z.; Hai, W.; Pan, Y.; Zhang, Y. Exendin-4 Imaging Based on Gastrointestinal GLP-1R Targets for IBD Diagnosis and Efficacy Assessment. Eur. J. Nucl. Med. Mol. Imaging 2025, 52, 3891–3902. [Google Scholar] [CrossRef]
- Heidari, P.; Haj-Mirzaian, A.; Prabhu, S.; Ataeinia, B.; Esfahani, S.A.; Mahmood, U. Granzyme B PET Imaging for Assessment of Disease Activity in Inflammatory Bowel Disease. J. Nucl. Med. 2024, 65, 1137–1143. [Google Scholar] [CrossRef]
- Chen, Y.; Yuan, H.; Tan, X.; Shang, Y.; Sun, X.; Wang, P.; Jiang, L. CXCR4-Targeted 68 Ga-Pentixafor PET/CT Imaging in Inflammatory Bowel Disease. Clin. Nucl. Med. 2024, 49, 817–821. [Google Scholar] [CrossRef]
- Bhowmik, A.A.; Heikkilä, T.R.H.; Polari, L.; Virta, J.; Liljenbäck, H.; Moisio, O.; Li, X.-G.; Viitanen, R.; Jalkanen, S.; Koffert, J.; et al. Detection of Intestinal Inflammation by Vascular Adhesion Protein-1-Targeted [68Ga]Ga-DOTA-Siglec-9 Positron Emission Tomography in Murine Models of Inflammatory Bowel Disease. Mol. Imaging Biol. 2024, 26, 322–333. [Google Scholar] [CrossRef]
- Ismail, M.S.; Peters, D.E.; Rowe, S.P.; Salavati, A.; Sharma, S.; Anders, R.A.; Pomper, M.; Slusher, B.S.; Selaru, F.M. PSMA-Targeted PET Radiotracer [18F]DCFPyL as an Imaging Biomarker in Inflammatory Bowel Disease. Clin. Exp. Gastroenterol. 2023, 16, 237–247. [Google Scholar] [CrossRef] [PubMed]
- Aarntzen, E.H.J.G.; Hermsen, R.; Drenth, J.P.H.; Boerman, O.C.; Oyen, W.J.G. 99mTc-CXCL8 SPECT to Monitor Disease Activity in Inflammatory Bowel Disease. J. Nucl. Med. 2016, 57, 398–403. [Google Scholar] [CrossRef] [PubMed]
- Jovani, M.; Danese, S. Vedolizumab for the Treatment of IBD: A Selective Therapeutic Approach Targeting Pathogenic A4b7 Cells. Curr. Drug Targets 2013, 14, 1433–1443. [Google Scholar] [CrossRef] [PubMed]
- Peyrin-Biroulet, L.; Danese, S.; Argollo, M.; Pouillon, L.; Peppas, S.; Gonzalez-Lorenzo, M.; Lytras, T.; Bonovas, S. Loss of Response to Vedolizumab and Ability of Dose Intensification to Restore Response in Patients with Crohn’s Disease or Ulcerative Colitis: A Systematic Review and Meta-Analysis. Clin. Gastroenterol. Hepatol. Off. Clin. Pract. J. Am. Gastroenterol. Assoc. 2019, 17, 838–846.e2. [Google Scholar] [CrossRef]
- Bhandari, A.; Patel, D.V.; Zemede, G.; Frederick, B.T.; Mattheakis, L.C. α4β7 Integrin Thioether Peptide Antagonists. U.S. Patent US9714270B2, 25 July 2017. [Google Scholar]
- Hall, L.J.; Faivre, E.; Quinlan, A.; Shanahan, F.; Nally, K.; Melgar, S. Induction and Activation of Adaptive Immune Populations During Acute and Chronic Phases of a Murine Model of Experimental Colitis. Dig. Dis. Sci. 2011, 56, 79–89. [Google Scholar] [CrossRef]
- Bonfiglio, R.; Galli, F.; Varani, M.; Scimeca, M.; Borri, F.; Fazi, S.; Cicconi, R.; Mattei, M.; Campagna, G.; Schönberger, T.; et al. Extensive Histopathological Characterization of Inflamed Bowel in the Dextran Sulfate Sodium Mouse Model with Emphasis on Clinically Relevant Biomarkers and Targets for Drug Development. Int. J. Mol. Sci. 2021, 22, 2028. [Google Scholar] [CrossRef]
- Dearling, J.L.J.; Park, E.J.; Dunning, P.; Baker, A.; Fahey, F.; Treves, S.T.; Soriano, S.G.; Shimaoka, M.; Packard, A.B.; Peer, D. Detection of Intestinal Inflammation by MicroPET Imaging Using a 64Cu-Labeled Anti-Beta7 Integrin Antibody. Inflamm. Bowel Dis. 2010, 16, 1458–1466. [Google Scholar] [CrossRef]
- Dearling, J.L.J.; Daka, A.; Veiga, N.; Peer, D.; Packard, A.B. Colitis ImmunoPET: Defining Target Cell Populations and Optimizing Pharmacokinetics. Inflamm. Bowel Dis. 2016, 22, 529–538. [Google Scholar] [CrossRef]
- Signore, A.; Bonfiglio, R.; Varani, M.; Galli, F.; Campagna, G.; Desco, M.; Cussó, L.; Mattei, M.; Wunder, A.; Borri, F.; et al. Radioimmune Imaging of α4β7 Integrin and TNFα for Diagnostic and Therapeutic Applications in Inflammatory Bowel Disease. Pharmaceutics 2023, 15, 817. [Google Scholar] [CrossRef]
- Freise, A.C.; Zettlitz, K.A.; Salazar, F.B.; Tavaré, R.; Tsai, W.-T.K.; Chatziioannou, A.F.; Rozengurt, N.; Braun, J.; Wu, A.M. Immuno-PET in Inflammatory Bowel Disease: Imaging CD4-Positive T Cells in a Murine Model of Colitis. J. Nucl. Med. 2018, 59, 980–985. [Google Scholar] [CrossRef] [PubMed]
- Wynendaele, E.; Bracke, N.; Stalmans, S.; De Spiegeleer, B. Development of Peptide and Protein Based Radiopharmaceuticals. Curr. Pharm. Des. 2014, 20, 2250–2267. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Merlin, D. Unveiling Colitis: A Journey through the Dextran Sodium Sulfate-Induced Model. Inflamm. Bowel Dis. 2024, 30, 844–853. [Google Scholar] [CrossRef] [PubMed]
- Chassaing, B.; Aitken, J.D.; Malleshappa, M.; Vijay-Kumar, M. Dextran Sulfate Sodium (DSS)-Induced Colitis in Mice. Curr. Protoc. Immunol. 2014, 104, 15.25.1–15.25.14. [Google Scholar] [CrossRef]
- Codesido, J.; García-Varela, L.; García-Otero, X.; Bouzón-Barreiro, S.; Gómez-Lado, N.; Toja-Camba, F.J.; Mondelo-García, C.; Lazaré, H.; Torres, J.B.; Vidal-Otero, J.; et al. PET Biodistribution Study of Subcutaneous and Intravenous Administration of Adalimumab in an Inflammatory Bowel Disease Model. Int. J. Pharm. 2025, 669, 125011. [Google Scholar] [CrossRef]
- Yan, G.; Wang, X.; Fan, Y.; Lin, J.; Yan, J.; Wang, L.; Pan, D.; Xu, Y.; Yang, M. Immuno-PET Imaging of TNF-α in Colitis Using 89Zr-DFO-Infliximab. Mol. Pharm. 2022, 19, 3632–3639. [Google Scholar] [CrossRef]
- Kumar, R.; Melmed, G.Y.; Gu, P. Imaging in Inflammatory Bowel Disease. Rheum. Dis. Clin. N. Am. 2024, 50, 721–733. [Google Scholar] [CrossRef]
- Borhani, A.; Afyouni, S.; Attari, M.M.A.; Mohseni, A.; Catalano, O.; Kamel, I.R. PET/MR Enterography in Inflammatory Bowel Disease: A Review of Applications and Technical Considerations. Eur. J. Radiol. 2023, 163, 110846. [Google Scholar] [CrossRef] [PubMed]
- Shaban, N.; Hoad, C.L.; Naim, I.; Alshammari, M.; Radford, S.J.; Clarke, C.; Marciani, L.; Moran, G. Imaging in Inflammatory Bowel Disease: Current and Future Perspectives. Frontline Gastroenterol. 2022, 13, e28–e34. [Google Scholar] [CrossRef] [PubMed]
- Brodersen, J.B.; Hess, S. FDG-PET/CT in Inflammatory Bowel Disease: Is There a Future? PET Clin. 2020, 15, 153–162. [Google Scholar] [CrossRef] [PubMed]
- Shah, S.C.; Itzkowitz, S.H. Colorectal Cancer in Inflammatory Bowel Disease: Mechanisms and Management. Gastroenterology 2022, 162, 715–730.e3. [Google Scholar] [CrossRef]
- Di, L. Strategic Approaches to Optimizing Peptide ADME Properties. AAPS J. 2015, 17, 134–143. [Google Scholar] [CrossRef]
- Lau, J.; Jacobson, O.; Niu, G.; Lin, K.-S.; Bénard, F.; Chen, X. Bench to Bedside: Albumin Binders for Improved Cancer Radioligand Therapies. Bioconjug. Chem. 2019, 30, 487–502. [Google Scholar] [CrossRef]
- Davis, R.A.; Hausner, S.H.; Harris, R.; Sutcliffe, J.L. A Comparison of Evans Blue and 4-(p-Iodophenyl)Butyryl Albumin Binding Moieties on an Integrin αvβ6 Binding Peptide. Pharmaceutics 2022, 14, 745. [Google Scholar] [CrossRef]
- Lu, Q.; Yang, M.-F.; Liang, Y.-J.; Xu, J.; Xu, H.-M.; Nie, Y.-Q.; Wang, L.-S.; Yao, J.; Li, D.-F. Immunology of Inflammatory Bowel Disease: Molecular Mechanisms and Therapeutics. J. Inflamm. Res. 2022, 15, 1825–1844. [Google Scholar] [CrossRef] [PubMed]
- Bábíčková, J.; Tóthová, Ľ.; Lengyelová, E.; Bartoňová, A.; Hodosy, J.; Gardlík, R.; Celec, P. Sex Differences in Experimentally Induced Colitis in Mice: A Role for Estrogens. Inflammation 2015, 38, 1996–2006. [Google Scholar] [CrossRef]





| Tissue | Group 1 DSS (n = 4) | Group 2 Water (n = 3) | Group 3 DSS (Block) (n = 3) | p Value | ||
|---|---|---|---|---|---|---|
| 1 vs. 3 | 1 vs. 2 | 2 vs. 3 | ||||
| Blood | 0.34 ± 0.08 | 0.38 ± 0.08 | 0.14 ± 0.02 | 0.0081 | 0.5512 | 0.0057 |
| Lung | 0.74 ± 0.24 | 2.14 ± 0.23 | 0.16 ± 0.03 | 0.0095 | 0.0005 | 0.0001 |
| Heart | 0.10 ± 0.03 | 0.13 ± 0.02 | 0.05 ± 0.00 | 0.0303 | 0.2356 | 0.0033 |
| Liver | 0.16 ± 0.04 | 0.17 ± 0.04 | 0.11 ± 0.01 | 0.0602 | 0.8765 | 0.0372 |
| Spleen | 0.19 ± 0.05 | 0.32 ± 0.11 | 0.08 ± 0.01 | 0.0114 | 0.0925 | 0.0220 |
| Muscle | 0.07 ± 0.01 | 0.08 ± 0.01 | 0.04 ± 0.01 | 0.0074 | 0.2409 | 0.0041 |
| Bone | 0.29 ± 0.14 | 0.13 ± 0.01 | 0.12 ± 0.03 | 0.0910 | 0.1109 | 0.4249 |
| Colon | 0.20 ± 0.05 | 0.11 ± 0.03 | 0.10 ± 0.03 | 0.0283 | 0.0450 | 0.5581 |
| Kidney | 2.99 ± 0.36 | 4.04 ± 1.23 | 1.81 ± 0.18 | 0.0039 | 0.1581 | 0.0359 |
| Stomach | 0.24 ± 0.19 | 0.14 ± 0.04 | 0.07 ± 0.01 | 0.2052 | 0.4260 | 0.0624 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, G.; Zhang, H.; Huo, L. A Novel 68Ga-Labeled Integrin α4β7-Targeted Radiopharmaceutical for PET/CT Imaging of DSS-Induced Murine Colitis. Pharmaceutics 2025, 17, 1591. https://doi.org/10.3390/pharmaceutics17121591
Yang G, Zhang H, Huo L. A Novel 68Ga-Labeled Integrin α4β7-Targeted Radiopharmaceutical for PET/CT Imaging of DSS-Induced Murine Colitis. Pharmaceutics. 2025; 17(12):1591. https://doi.org/10.3390/pharmaceutics17121591
Chicago/Turabian StyleYang, Guangjie, Haiqiong Zhang, and Li Huo. 2025. "A Novel 68Ga-Labeled Integrin α4β7-Targeted Radiopharmaceutical for PET/CT Imaging of DSS-Induced Murine Colitis" Pharmaceutics 17, no. 12: 1591. https://doi.org/10.3390/pharmaceutics17121591
APA StyleYang, G., Zhang, H., & Huo, L. (2025). A Novel 68Ga-Labeled Integrin α4β7-Targeted Radiopharmaceutical for PET/CT Imaging of DSS-Induced Murine Colitis. Pharmaceutics, 17(12), 1591. https://doi.org/10.3390/pharmaceutics17121591