Enhancing Pharmacokinetics and Pharmacodynamics of Rosuvastatin Calcium through the Development and Optimization of Fast-Dissolving Films
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Experimental Design
2.2.2. Preparation of RSV-FDFs
2.2.3. Characterization of RSV-FDFs
Physical Appearance
Content Uniformity, Average Weight, and Thickness
Folding Endurance, Tensile Strength, and Elongation Percentage
Surface pH
In Vitro Disintegration
In Vitro Dissolution
2.2.4. In Vivo Pharmacokinetics Evaluation on Male Wistar Rats
Study Design
Animal Handling and Blood Sampling
Chromatographic Conditions
Pharmacokinetics Data Analysis
2.2.5. In Vivo Anti-Dyslipidemic Activity
2.2.6. Statistical Analysis
3. Results and Discussion
3.1. Formulation and Evaluation of RSV-FDFs
3.1.1. Tensile Strength and Elongation Percentage
3.1.2. In Vitro Dissolution
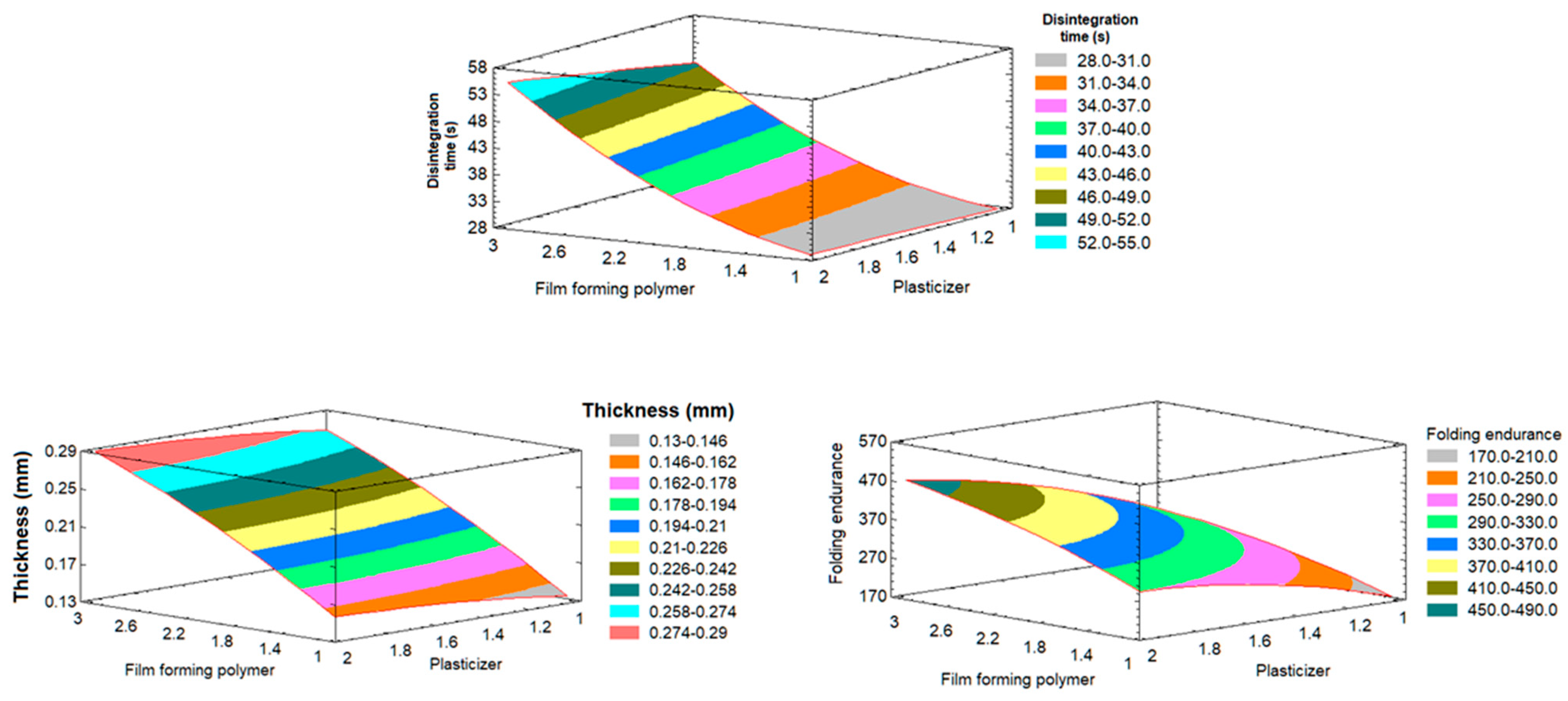
3.2. Optimization of RSV-FDFs
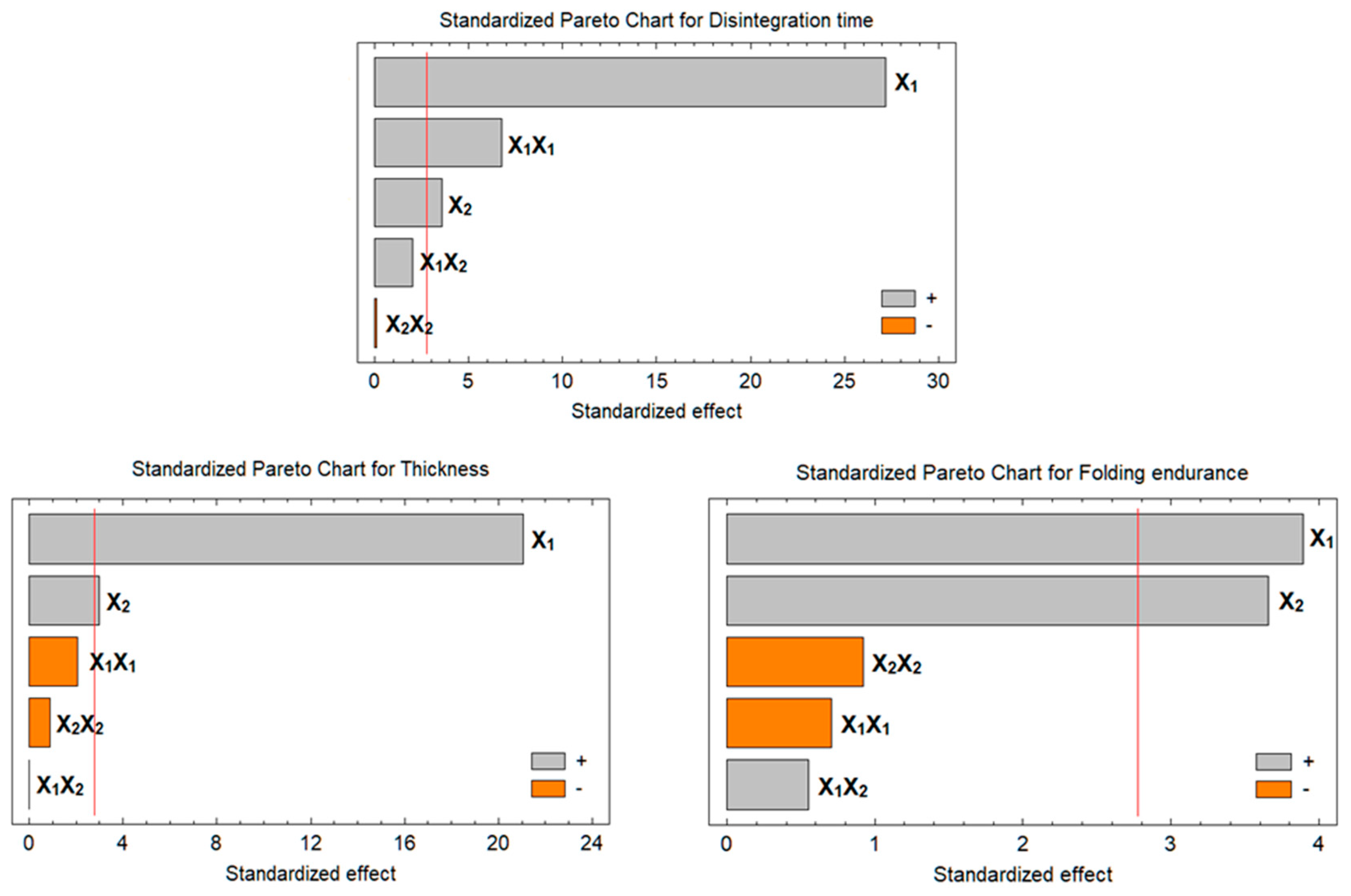
3.2.1. Estimation of the Quantitative Effects
Effect on the In Vitro Disintegration (Y1):
Effect on the Film Thickness
Effect on the Folding Endurance
3.2.2. Preparation and Evaluation of Optimized Formula
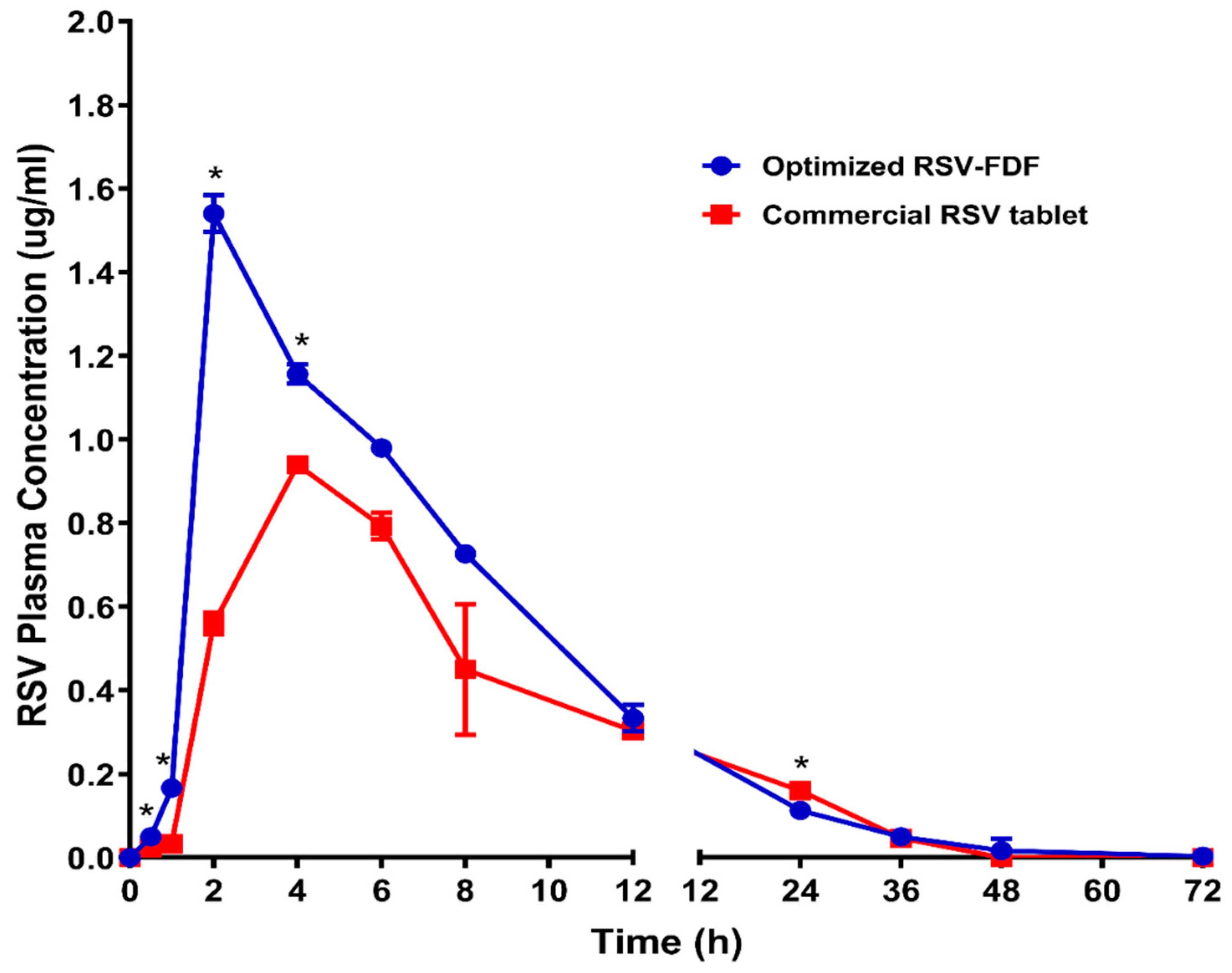
3.3. In Vivo Pharmacokinetics Evaluation
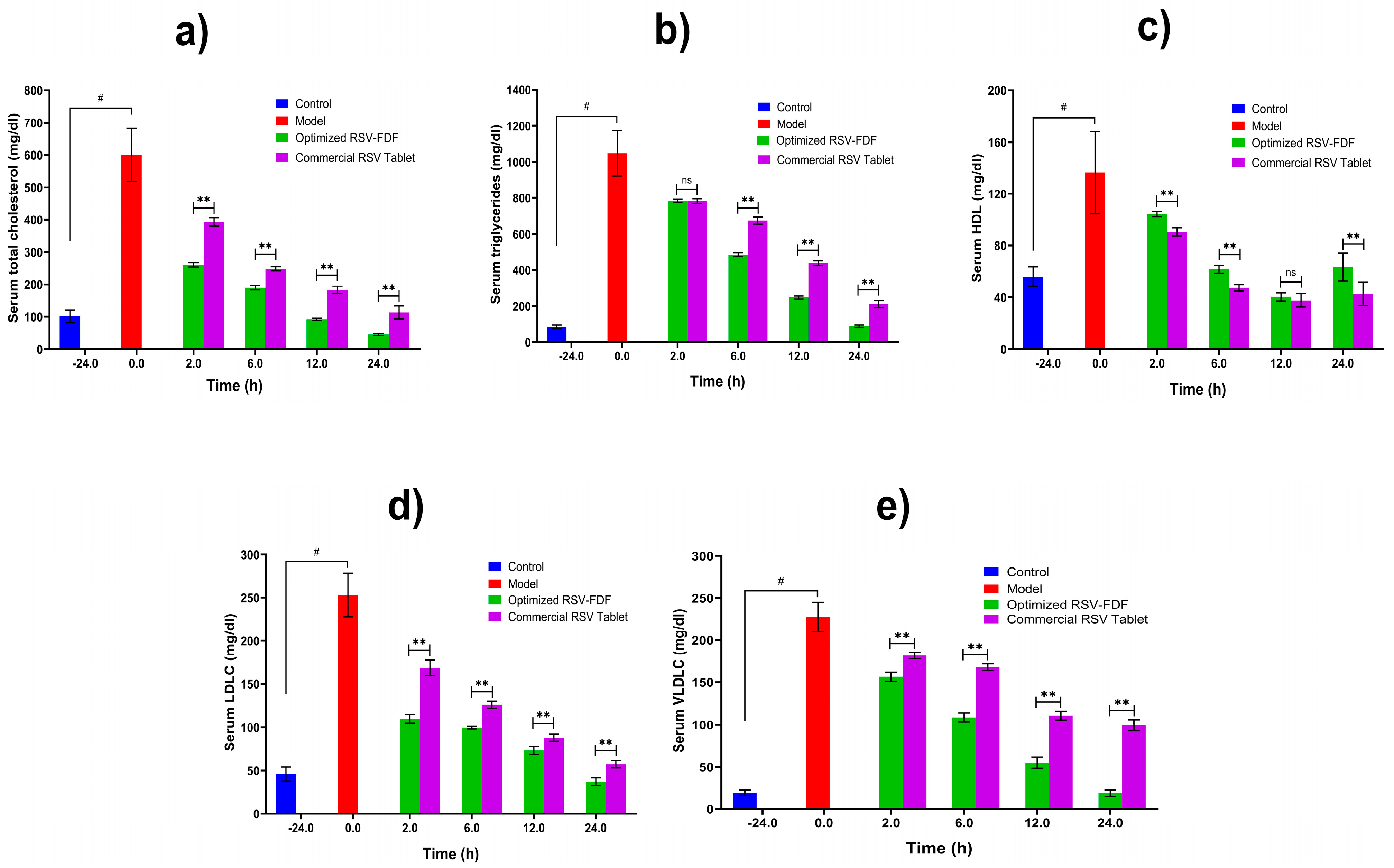
3.4. In Vivo Pharmacodynamics Evaluation
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mosca, S.; Araújo, G.; Costa, V.; Correia, J.; Bandeira, A.; Martins, E.; Mansilha, H.; Tavares, M.; Coelho, M.P. Dyslipidemia Diagnosis and Treatment: Risk Stratification in Children and Adolescents. J. Nutr. Metab. 2022, 2022, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Su, X.; Peng, H.; Chen, X.; Wu, X.; Wang, B. Hyperlipidemia and hypothyroidism. Clin. Chim. Acta 2022, 527, 61–70. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, T.A.; Elimam, H.; Alrifai, A.O.; Nadhrah, H.M.; Masoudi, L.Y.; Sairafi, W.O.; M.El-Say, K. Rosuvastatin lyophilized tablets loaded with flexible chitosomes for improved drug bioavailability, anti-hyperlipidemic and anti-oxidant activity. Int. J. Pharm. 2020, 558, 119791. [Google Scholar] [CrossRef] [PubMed]
- Shih, H.-J.; Lin, K.-H.; Wen, Y.-C.; Fan, Y.-C.; Tsai, P.-S.; Huang, C.-J. Increased risk of bladder cancer in young adult men with hyperlipidemia: A population-based cohort study. Medicine 2021, 100, e28125. [Google Scholar] [CrossRef]
- Staels, B.; Dallongeville, J.; Auwerx, J.; Schoonjans, K.; Leitersdorf, E.; Fruchart, J.-C. Mechanism of action of fibrates on lipid and lipoprotein metabolism. Circulation 1998, 98, 2088–2093. [Google Scholar] [CrossRef] [PubMed]
- Lennernäs, H.; Fager, G. Pharmacodynamics and Pharmacokinetics of the HMG-CoA Reductase Inhibitors. Clin. Pharmacokinet. 1997, 32, 403–425. [Google Scholar] [CrossRef] [PubMed]
- Alshora, D.H.; Ibrahim, M.A.; Elzayat, E.; Almeanazel, O.T.; Alanazi, F. Rosuvastatin calcium nanoparticles: Improving bioavailability by formulation and stabilization codesign. PLoS ONE 2018, 13, e0200218. [Google Scholar] [CrossRef]
- Chizner, M.A.; Duvall, W.L. Highlights of prescribing information crestor (rosuvastatin calcium). Cardiovasc. Rev. Rep. 2003, 24, 591. [Google Scholar]
- Scott, L.J.; Curran, M.P.; Figgitt, D.P. Rosuvastatin. Am. J. Cardiovasc. Drugs 2004, 4, 117–138. [Google Scholar] [CrossRef]
- Elsayed, I.; El-Dahmy, R.M.; Elshafeey, A.H.; El Gawad, N.A.A.; El Gazayerly, O.N. Tripling the bioavailability of rosuvastatin calcium through development and optimization of an In-Situ forming nanovesicular system. Pharmaceutics 2019, 11, 275. [Google Scholar] [CrossRef]
- Butt, S.; Hasan, S.M.F.; Hassan, M.M.; Alkharfy, K.M.; Neau, S.H. Directly compressed rosuvastatin calcium tablets that offer hydrotropic and micellar solubilization for improved dissolution rate and extent of drug release. Saudi Pharm. J. 2019, 27, 619–628. [Google Scholar] [CrossRef] [PubMed]
- Gabr, M.M.; Mortada, S.M.; Sallam, M.A. Carboxylate cross-linked cyclodextrin: A nanoporous scaffold for enhancement of rosuvastatin oral bioavailability. Eur. J. Pharm. Sci. 2018, 111, 1–12. [Google Scholar] [CrossRef] [PubMed]
- González, R.; Peña, M.; Torres, N.S.; Torrado, G. Design, development, and characterization of amorphous rosuvastatin calcium tablets. PLoS ONE 2022, 17, e0265263. [Google Scholar] [CrossRef] [PubMed]
- El-Say, K.M.; Felimban, R.; Tayeb, H.H.; Chaudhary, A.G.; Omar, A.M.; Rizg, W.Y.; Alnadwi, F.H.; I Abd-Allah, F.; Ahmed, T. Pairing 3D-Printing with Nanotechnology to Manage Metabolic Syndrome. Int. J. Nanomed. 2022, 17, 1783–1801. [Google Scholar] [CrossRef] [PubMed]
- Zaki, R.M.; Alfadhel, M.; Seshadri, V.D.; Albagami, F.; Alrobaian, M.; Tawati, S.M.; Warsi, M.H.; Almurshedi, A.S. Fabrication and characterization of orodispersible films loaded with solid dispersion to enhance Rosuvastatin calcium bioavailability. Saudi Pharm. J. 2023, 31, 135–146. [Google Scholar] [CrossRef]
- Rédai, E.-M.; Antonoaea, P.; Todoran, N.; Vlad, R.A.; Bîrsan, M.; Tătaru, A.; Ciurba, A. Development and evaluation of fluoxetine fast dissolving films: An alternative for noncompliance in pediatric patients. Processes 2021, 9, 778. [Google Scholar] [CrossRef]
- Reveny, J.; Tanuwijaya, J.; Remalya, A. Formulation of Orally Dissolving Film (ODF) Metoclopramide Using Hydroxy Propyl Methyl Cellulose and Polyvinyl Alcohol with Solvent Casting Method. Int. J. ChemTech Res. 2017, 10, 316–321. [Google Scholar]
- Hamza, M. Development and Evaluation of Orodispersible Films of Lamotrigine: Hydroxypropyl Β Cyclodextrin Inclusion Complex. Al-Azhar J. Pharm. Sci. 2017, 56, 31–46. [Google Scholar] [CrossRef]
- Koland, M.; Sandeep, V.; Charyulu, N. Fast Dissolving Sublingual Films of Ondansetron Hydrochloride: Effect of Additives on in vitro Drug Release and Mucosal Permeation. J. Young- Pharm. 2010, 2, 216–222. [Google Scholar] [CrossRef]
- Cho, H.-W.; Baek, S.-H.; Lee, B.-J.; Jin, H.-E. Orodispersible polymer films with the poorly water-soluble drug, olanzapine: Hot-Melt pneumatic extrusion for single-process 3D printing. Pharmaceutics 2020, 12, 692. [Google Scholar] [CrossRef]
- Abdulelah, F.M.; Abdulbaqi, M.R. Fast dissolving film nanocrystal (FDFN) preparation as a new trend for solubility enhancement of poorly soluble class ii drug tenoxicam. AVFT–Arch. Venez. Farmacol. Ter. 2021, 40, 333–339. [Google Scholar]
- Hoffmann, E.M.; Breitenbach, A.; Breitkreutz, J.; Pharm, D. Advances in orodispersible films for drug delivery. Expert Opin. Drug Deliv. 2011, 8, 299–316. [Google Scholar] [CrossRef] [PubMed]
- Geng, Y.; Zhou, F.; Williams, G.R. Developing and scaling up fast-dissolving electrospun formulations based on poly(vinylpyrrolidone) and ketoprofen. J. Drug Deliv. Sci. Technol. 2020, 61, 102138. [Google Scholar] [CrossRef]
- Adrover, A.; Varani, G.; Paolicelli, P.; Petralito, S.; Di Muzio, L.; Casadei, M.A.; Tho, I. Experimental and modeling study of drug release from HPMC-based erodible oral thin films. Pharmaceutics 2018, 10, 222. [Google Scholar] [CrossRef] [PubMed]
- Hosny, K.M.; El-Say, K.M.; Ahmed, O.A. Optimized sildenafil citrate fast orodissolvable film: A promising formula for overcoming the barriers hindering erectile dysfunction treatment. Drug Deliv. 2014, 23, 355–361. [Google Scholar] [CrossRef] [PubMed]
- Rathod, S.; Phansekar, M.; Bhagwan, A.; Surve, G. A Review on mouth dissolving tablets. Indian Drugs 2013, 50, 5–14. [Google Scholar] [CrossRef]
- Maher, E.M.; Ali, A.M.A.; Salem, H.F.; Abdelrahman, A.A. In vitro/in vivo evaluation of an optimized fast dissolving oral film containing olanzapine co-amorphous dispersion with selected carboxylic acids. Drug Deliv. 2016, 23, 3088–3100. [Google Scholar] [CrossRef]
- Wasilewska, K.; Winnicka, K. How to assess orodispersible film quality? A review of applied methods and their modifications. Acta Pharm. 2019, 69, 155–176. [Google Scholar] [CrossRef]
- Vishvakarma, P. Design and development of montelukast sodium fast dissolving films for better therapeutic efficacy. J. Chil. Chem. Soc. 2018, 63, 3988–3993. [Google Scholar] [CrossRef]
- Kumar, A.; Verma, R.; Jain, V. Formulation Development and Evaluation of Fast Dissolving Oral Film of Dolasetron Mesylate. Asian J. Pharm. Educ. Res. 2019, 8, 38. [Google Scholar] [CrossRef]
- Liew KBin Tan, Y.T.F.; Peh, K.K. Effect of polymer, plasticizer and filler on orally disintegrating film. Drug. Dev. Ind. Pharm. 2014, 40, 110–119. [Google Scholar] [CrossRef] [PubMed]
- Elshafeey, A.H.; El-Dahmy, R.M. Formulation and development of oral fast-dissolving films loaded with nanosuspension to augment paroxetine bioavailability: In vitro characterization, ex vivo permeation, and pharmacokinetic evaluation in healthy human volunteers. Pharmaceutics 2021, 13, 1869. [Google Scholar] [CrossRef] [PubMed]
- Olechno, K.; Maciejewski, B.; Głowacz, K.; Lenik, J.; Ciosek-Skibińska, P.; Basa, A.; Winnicka, K. Orodispersible Films with Rupatadine Fumarate Enclosed in Ethylcellulose Microparticles as Drug Delivery Platform with Taste-Masking Effect. Materials 2022, 15, 2126. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.-Y.; Lee, C.-J.; Lin, Y.-Y. Drug-polymer interaction affecting the mechanical properties, adhesion strength and release kinetics of piroxicam-loaded Eudragit E films plasticized with different plasticizers. J. Control. Release 1995, 33, 375–381. Available online: http://www.sciencedirect.com/science/article/pii/0168365994001098 (accessed on 23 October 2023). [CrossRef]
- Pichayakorn, W.; Suksaeree, J.; Boonme, P.; Amnuaikit, T.; Taweepreda, W.; Ritthidej, G.C. Deproteinized natural rubber film forming polymeric solutions for nicotine transdermal delivery. Pharm. Dev. Technol. 2011, 18, 1111–1121. [Google Scholar] [CrossRef] [PubMed]
- Rujivipat, S.; Bodmeier, R. Moisture plasticization for enteric Eudragit® L30D-55-coated pellets prior to compression into tablets. Eur. J. Pharm. Biopharm. 2012, 81, 223–229. [Google Scholar] [CrossRef]
- Nesseem, D.I.; Eid, S.; El-Houseny, S. Development of novel transdermal self-adhesive films for tenoxicam, an anti-inflammatory drug. Life Sci. 2011, 89, 430–438. [Google Scholar] [CrossRef]
- Bhupinder, B.; Sarita, J. Formulation and evaluation of fast dissolving sublingual films of Rizatriptan Benzoate. Int. J. Drug. Dev. Res. 2012, 4, 133–143. [Google Scholar]
- Choi, M.-J.; Woo, M.R.; Choi, H.-G.; Jin, S.G. Effects of Polymers on the Drug Solubility and Dissolution Enhancement of Poorly Water-Soluble Rivaroxaban. Int. J. Mol. Sci. 2022, 23, 9491. [Google Scholar] [CrossRef]
- Al-Mogherah, A.I.; Ibrahim, M.A.; Hassan, M.A. Optimization and evaluation of venlafaxine hydrochloride fast dissolving oral films. Saudi Pharm. J. 2020, 28, 1374–1382. [Google Scholar] [CrossRef]
- Centkowska, K.; Ławrecka, E.; Sznitowska, M. Technology of orodispersible polymer films with micronized loratadine—influence of different drug loadings on film properties. Pharmaceutics 2020, 12, 250. [Google Scholar] [CrossRef] [PubMed]
- Basu, B.; Mankad, A.; Dutta, A. Methylphenidate Fast Dissolving Films: Development, Optimization Using Simplex Centroid Design and In Vitro Characterization. Turk. J. Pharm. Sci. 2022, 19, 251–266. [Google Scholar] [CrossRef] [PubMed]
- Bharti, K.; Mittal, P.; Mishra, B. Formulation and characterization of fast dissolving oral films containing buspirone hydrochloride nanoparticles using design of experiment. J. Drug Deliv. Sci. Technol. 2018, 49, 420–432. [Google Scholar] [CrossRef]
| Independent Variables | Levels | ||
| Low | Medium | High | |
| X1 = film-forming polymer (HPMC) % | 1 | 2 | 3 |
| X2 = plasticizer (PEG 400) % | 1 | 1.5 | 2 |
| Dependent Variables | Constraints | ||
| Low | High | Goal | |
| Y1 = disintegration time (s) | 26 | 62 | Minimize |
| Y2 = thickness (mm) | 0.11 | 0.31 | Minimize |
| Y3 = folding endurance | 155 | 456 | Maximize |
| Run Code | Independent Variables | Dependent Variables | |||
|---|---|---|---|---|---|
| HPMC % (X1) | PEG 400 (X2) | Disintegration Time (Y1), s | Thickness (Y2), mm | Folding Endurance (Y3) | |
| F1 | 0.59 | 1.5 | 26 | 0.11 | 189 |
| F2 | 3.0 | 2.0 | 55 | 0.28 | 420 |
| F3 | 3.0 | 1.0 | 49 | 0.27 | 320 |
| F4 | 2.0 | 1.5 | 36 | 0.22 | 350 |
| F5 | 2.0 | 1.5 | 37 | 0.23 | 354 |
| F6 | 3.41 | 1.5 | 62 | 0.31 | 444 |
| F7 | 1.0 | 1.0 | 29 | 0.14 | 229 |
| F8 | 2.0 | 2.21 | 38 | 0.24 | 456 |
| F9 | 1.0 | 2.0 | 30 | 0.15 | 269 |
| F10 | 2.0 | 0.79 | 34 | 0.20 | 155 |
| Run Code | Surface pH | Average Weight (g) | Tensile Strength (N/cm2) | Percent Elongation (%) | Dissolution after 2 min (%) | RSV Content (%) |
|---|---|---|---|---|---|---|
| F1 | 6.5 | 0.02 | 1.765 | 10 | 98.61 | 103.69 |
| F2 | 6.53 | 0.11 | 1.852 | 80 | 105.22 | 105.39 |
| F3 | 6.6 | 0.10 | 1.843 | 64 | 105.56 | 103.88 |
| F4 | 6.62 | 0.08 | 1.814 | 74 | 101.79 | 104.91 |
| F5 | 6.6 | 0.08 | 1.816 | 72 | 105.69 | 105.16 |
| F6 | 6.6 | 0.12 | 1.872 | 88 | 101.52 | 105.37 |
| F7 | 6.58 | 0.03 | 1.758 | 16 | 98.68 | 104.72 |
| F8 | 6.61 | 0.09 | 1.828 | 76 | 100.33 | 105.33 |
| F9 | 6.62 | 0.03 | 1.778 | 70 | 97.22 | 105.55 |
| F10 | 6.58 | 0.07 | 1.807 | 20 | 102.65 | 104.97 |
| Factors | Disintegration Time (Y1), s | Thickness (Y2), mm | Folding Endurance (Y3) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Estimate | F-Ratio | p-Value | Estimate | F-Ratio | p-Value | Estimate | F-Ratio | p-Value | |
| X1 | 23.98 | 739.58 | 0.0001 * | 0.14 | 443.28 | 0.0001 * | 150.66 | 15.17 | 0.0176 * |
| X2 | 3.16 | 12.88 | 0.0230 * | 0.02 | 8.82 | 0.0412 * | 141.42 | 13.36 | 0.0217 * |
| X1X1 | 7.88 | 45.59 | 0.0025 * | −0.02 | 4.21 | 0.1094 | −36.25 | 0.50 | 0.5178 |
| X1X2 | 2.50 | 4.02 | 0.1155 | 0.00 | 0.00 | 1.0000 | 30.00 | 0.30 | 0.6126 |
| X2X2 | −0.13 | 0.01 | 0.9198 | −0.01 | 0.77 | 0.4288 | −47.25 | 0.85 | 0.4081 |
| R2 | 99.51 | 99.13 | 88.17 | ||||||
| Adj. R2 | 98.89 | 98.04 | 73.37 | ||||||
| SEE | 1.25 | 0.009 | 54.71 | ||||||
| MAE | 0.68 | 0.005 | 28.98 | ||||||
| Parameter | Unit | Optimized RSV-FDFs | Marketed RSV Tablets | ||
|---|---|---|---|---|---|
| Average | STDEV | Average | STDEV | ||
| Lambda_z | 1/h | 0.072 | 0.020 | 0.084 | 0.004 |
| t1/2 | h | 10.172 | 2.716 | 8.231 | 0.417 |
| Tmax | h | 2.000 | 0.000 | 4.000 | 0.000 |
| Cmax | µg/mL | 1.540 * | 0.044 | 0.940 | 0.017 |
| AUC0–t | µg/mL.h | 13.680 * | 0.622 | 10.320 | 0.531 |
| AUC0–inf | µg/mL.h | 14.178 * | 0.331 | 10.874 | 0.589 |
| AUMC0–inf | µg/mL.h2 | 166.681 | 27.408 | 141.365 | 6.273 |
| MRT0–inf | h | 11.731 | 1.656 | 13.005 | 0.188 |
| Vz | (mg)/(µg/mL) | 20.629 | 5.057 | 21.894 | 1.803 |
| Cl | (mg)/(µg/mL)/h | 1.411 * | 0.033 | 1.843 | 0.102 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ashraf, I.; Hanna, P.A.; Gad, S.; Abd-Allah, F.I.; El-Say, K.M. Enhancing Pharmacokinetics and Pharmacodynamics of Rosuvastatin Calcium through the Development and Optimization of Fast-Dissolving Films. Pharmaceutics 2023, 15, 2640. https://doi.org/10.3390/pharmaceutics15112640
Ashraf I, Hanna PA, Gad S, Abd-Allah FI, El-Say KM. Enhancing Pharmacokinetics and Pharmacodynamics of Rosuvastatin Calcium through the Development and Optimization of Fast-Dissolving Films. Pharmaceutics. 2023; 15(11):2640. https://doi.org/10.3390/pharmaceutics15112640
Chicago/Turabian StyleAshraf, Ibrahim, Pierre A. Hanna, Shadeed Gad, Fathy I. Abd-Allah, and Khalid M. El-Say. 2023. "Enhancing Pharmacokinetics and Pharmacodynamics of Rosuvastatin Calcium through the Development and Optimization of Fast-Dissolving Films" Pharmaceutics 15, no. 11: 2640. https://doi.org/10.3390/pharmaceutics15112640
APA StyleAshraf, I., Hanna, P. A., Gad, S., Abd-Allah, F. I., & El-Say, K. M. (2023). Enhancing Pharmacokinetics and Pharmacodynamics of Rosuvastatin Calcium through the Development and Optimization of Fast-Dissolving Films. Pharmaceutics, 15(11), 2640. https://doi.org/10.3390/pharmaceutics15112640









