Preparation of Liposomal Formulations for Ocular Delivery of Thymoquinone: In Vitro Evaluation in HCEC-2 e HConEC Cells
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Chromatography Conditions and Instruments
HPLC Method for TQ and FITC
2.3. Preparation of Liposomal Formulations
2.4. Characterization of Liposomes
2.4.1. Physical Characterization
2.4.2. Morphological Characterization
2.5. Determination of Encapsulation Efficiency and Loading Capacity
2.6. Storage Stability
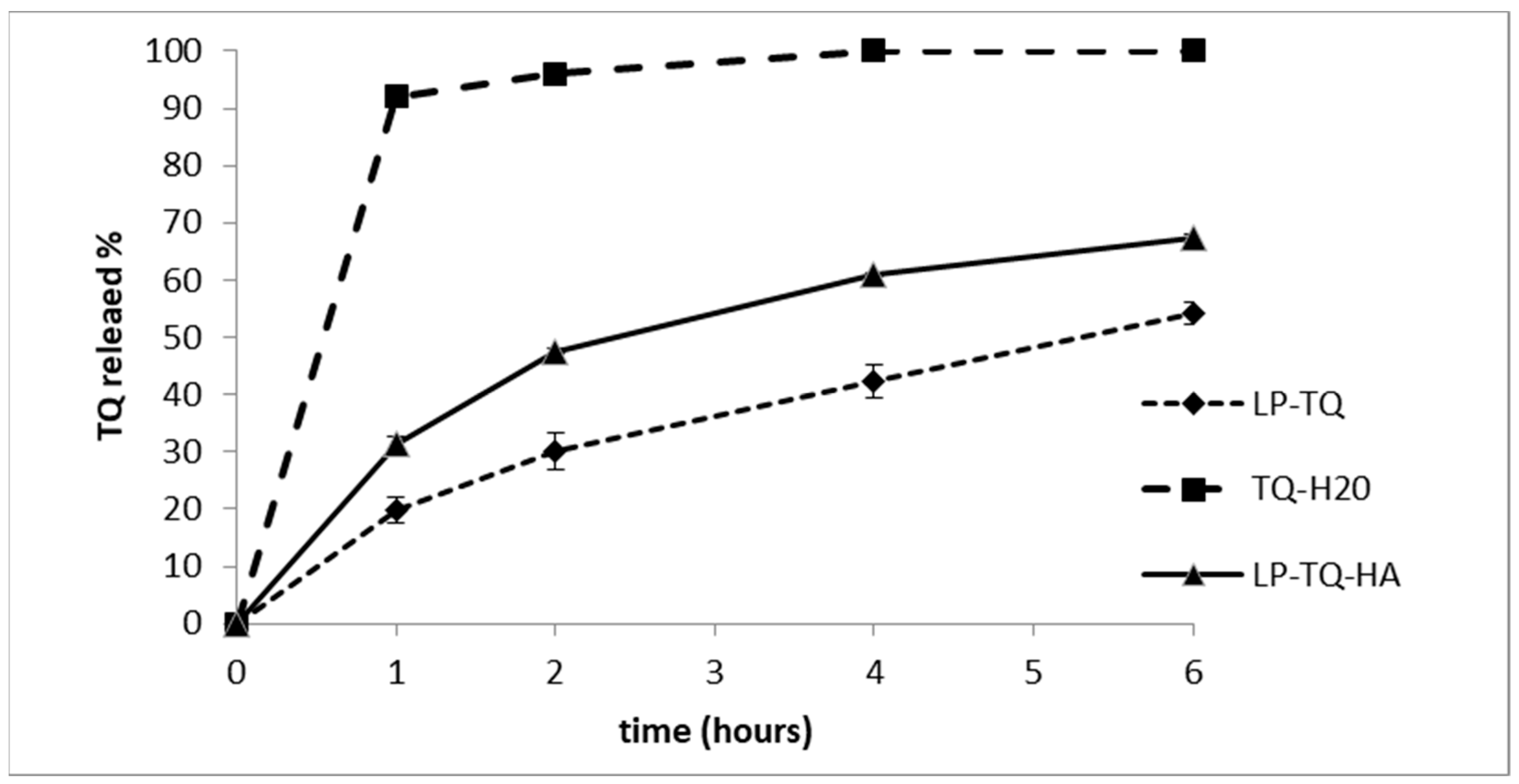
2.7. In Vitro Release Study
2.8. Cell Culture Studies
2.8.1. Human Corneal Epithelial Cells (HCE-2)
2.8.2. Human Conjunctival Epithelial Cells (HConEC)
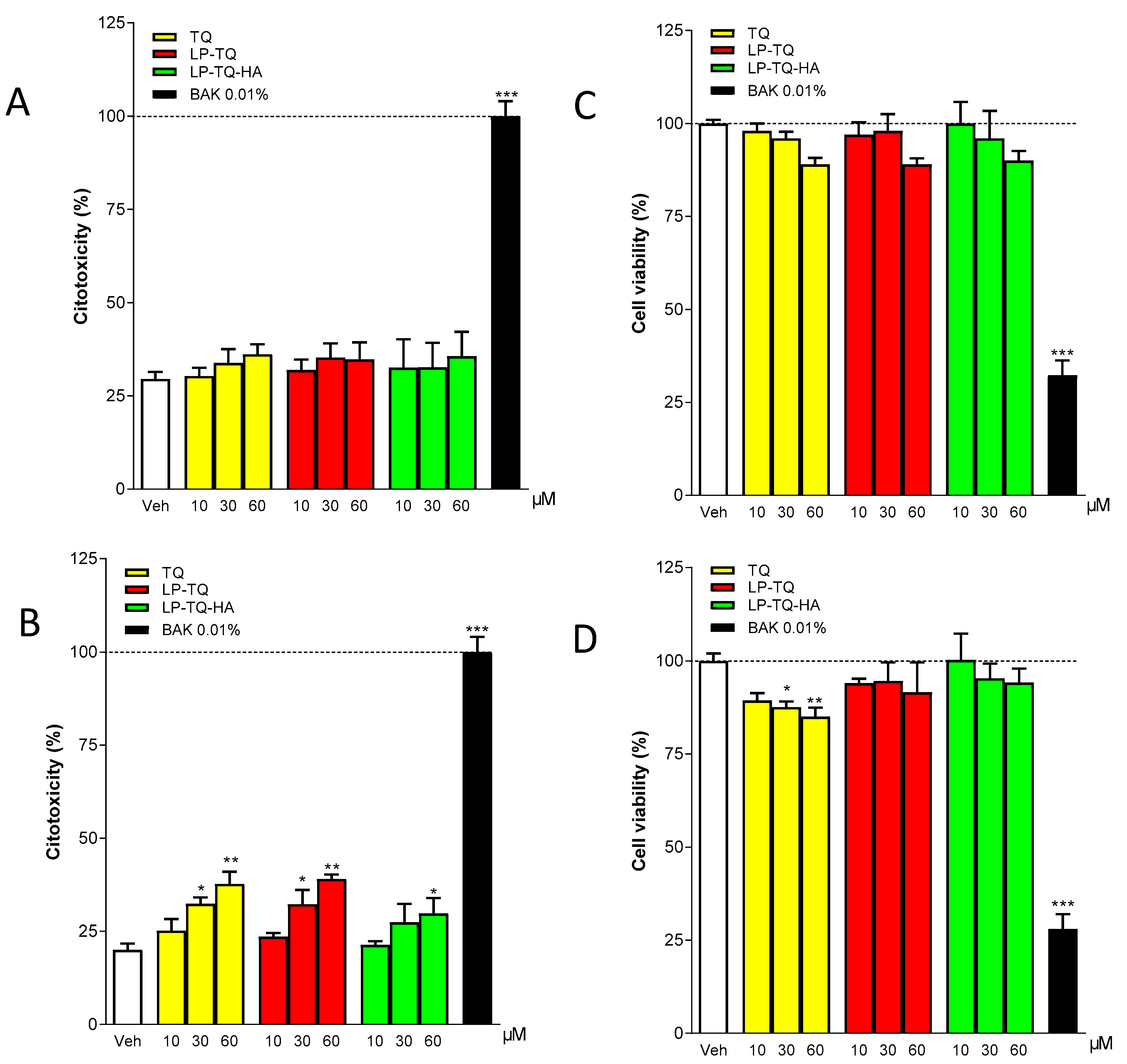
2.8.3. Analysis of In Vitro Cytotoxicity
MTT Assay
LDH Assay
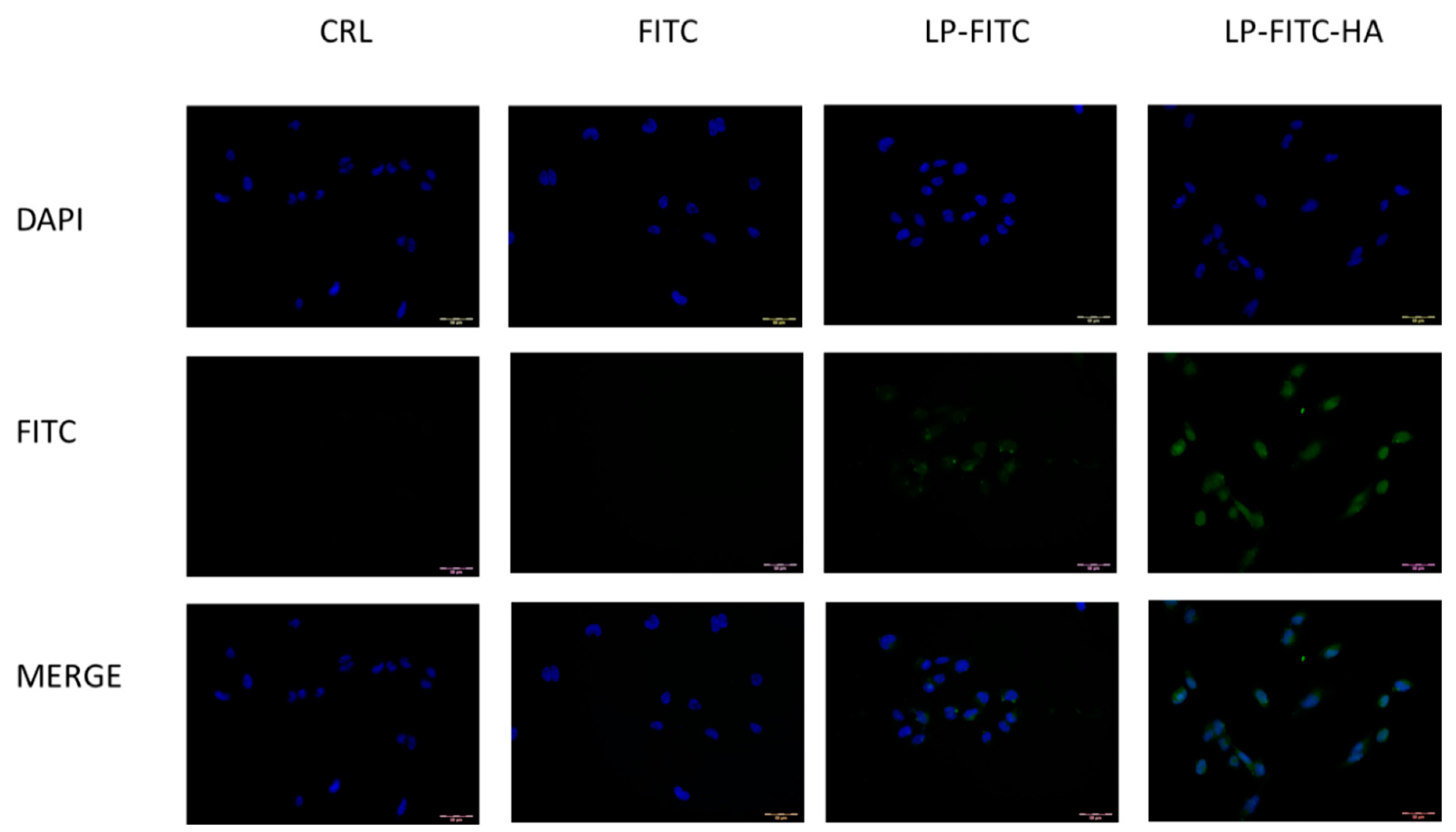
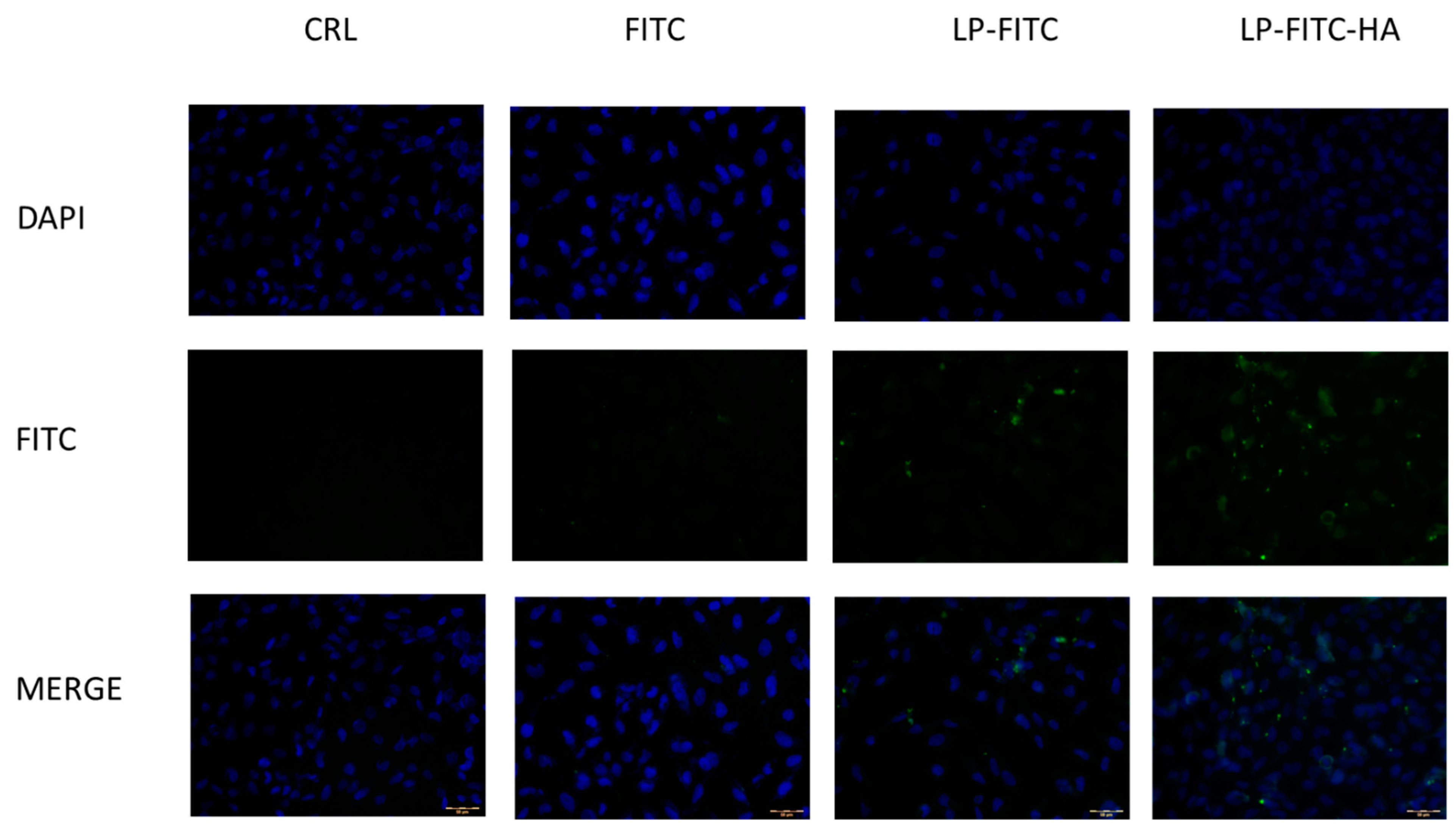
2.8.4. Cellular Uptake Studies
3. Results and Discussion
3.1. Preparation and Characterization of TQ and FITC Liposomal Formulations
3.1.1. Physical Parameters of Liposomal Formulations
3.1.2. Chemical Characterization of Liposomal Formulations
3.1.3. Characterization of Optimized Liposomal Formulations
3.1.4. Physical Parameters of HA-Coated Liposomes
3.2. Morphology
3.3. Stability Studies
Storage Stability Study of TQ Liposomes
3.4. In Vitro Release Study
3.5. Cytotoxicity Studies in Human Epithelial Cells of Cornea (HCEC-2) and Conjunctiva (HConEC)
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yimer, E.M.; Tuem, K.B.; Karim, A.; Ur-Rehman, N.; Anwar, F. Nigella sativa L. (Black Cumin): A Promising Natural Remedy for Wide Range of Illnesses. Evid.-Based Complement. Altern. Med. 2019, 2019, 1528635. [Google Scholar] [CrossRef]
- Gupta, B.; Ghosh, K.K.; Gupta, R.C. Chapter 39—Thymoquinone. In Nutraceuticals, 1st ed.; Gupta, R.C., Ed.; Academic Press: London, UK, 2016. [Google Scholar] [CrossRef]
- Jain, A.; Dhruw, L.; Sinha, P.; Pradhan, A.; Sharma, R.; Gupta, B. Chapter 52—Thymoquinone. In Nutraceuticals, 2nd ed.; Gupta, R.C., Lall, R., Srivastava, A., Eds.; Academic Press: London, UK, 2021; pp. 891–901. [Google Scholar] [CrossRef]
- Farkhondeh, T.; Samarghandian, S.; Shahri, A.M.P.; Samini, F. The Neuroprotective Effects of Thymoquinone: A Review. Dose-Response 2018, 16. [Google Scholar] [CrossRef]
- El-Far, A.H.; Al Jaouni, S.K.; Li, W.; Mousa, S.A. Protective Roles of Thymoquinone Nanoformulations: Potential Nanonutraceuticals in Human Diseases. Nutrients 2018, 10, 1369. [Google Scholar] [CrossRef]
- Hu, X.; Liang, Y.; Zhao, B.; Wang, Y. Thymoquinone protects human retinal pigment epithelial cells against hydrogen peroxide induced oxidative stress and apoptosis. J. Cell. Biochem. 2019, 120, 4514–4522. [Google Scholar] [CrossRef]
- Kocatürk, T.; Erkan, E.; Meteoğlu, I.; Ekici, M.; Büyüköztürk, A.K.; Yavaşoğlu, I.; Çakmak, H.; Dayanır, V.; Balkaya, M. Effects of Topical Thymoquinone in an Experimental Dry Eye Model. Turk. J. Ophthalmol. 2018, 48, 281–287. [Google Scholar] [CrossRef]
- Salem, M.L. Immunomodulatory and therapeutic properties of the Nigella sativa L. seed. Int. Immunopharmacol. 2005, 5, 1749–1770. [Google Scholar] [CrossRef]
- Hayat, K.; Asim, M.R.; Nawaz, M.; Li, M.; Zhang, L.; Sun, N. Ameliorative Effect of Thymoquinone on Ovalbumin-induced Allergic Conjunctivitis in Balb/c Mice. Curr. Eye Res. 2011, 36, 591–598. [Google Scholar] [CrossRef]
- Landucci, E.; Mazzantini, C.; Buonvicino, D.; Pellegrini-Giampietro, D.; Bergonzi, M. Neuroprotective Effects of Thymoquinone by the Modulation of ER Stress and Apoptotic Pathway in In Vitro Model of Excitotoxicity. Molecules 2021, 26, 1592. [Google Scholar] [CrossRef]
- Bergonzi, M.C.; Vasarri, M.; Marroncini, G.; Barletta, E.; Degl’Innocenti, D. Thymoquinone-Loaded Soluplus®-Solutol® HS15 Mixed Micelles: Preparation, In Vitro Characterization, and Effect on the SH-SY5Y Cell Migration. Molecules 2020, 25, 4707. [Google Scholar] [CrossRef] [PubMed]
- Bilia, A.R.; Piazzini, V.; Risaliti, L.; Vanti, G.; Casamonti, M.; Wang, M.; Bergonzi, M.C. Nanocarriers: A Successful Tool to Increase Solubility, Stability and Optimise Bioefficacy of Natural Constituents. Curr. Med. Chem. 2019, 26, 4631–4656. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, R.; Iezhitsa, I.; Agarwal, P.; Nasir, N.A.A.; Razali, N.; Alyautdin, R.; Ismail, N.M. Liposomes in topical ophthalmic drug delivery: An update. Drug Deliv. 2014, 23, 1075–1091. [Google Scholar] [CrossRef]
- Ibrahim, M.M.; Tawfique, S.A.H.; Mahdy, M.M. Liposomal diltiazem HCl as ocular drug delivery system for glaucoma. Drug Dev. Ind. Pharm. 2014, 40, 765–773. [Google Scholar] [CrossRef] [PubMed]
- Achouri, D.; Alhanout, K.; Piccerelle, P.; Andrieu, V. Recent advances in ocular drug delivery. Drug Dev. Ind. Pharm. 2013, 39, 1599–1617. [Google Scholar] [CrossRef]
- Lalu, L.; Tambe, V.; Pradhan, D.; Nayak, K.; Bagchi, S.; Maheshwari, R.; Kalia, K.; Tekade, R.K. Novel nanosystems for the treatment of ocular inflammation: Current paradigms and future research directions. J. Control. Release 2017, 268, 19–39. [Google Scholar] [CrossRef]
- Fahmy, H.M.; Saad, E.A.E.-M.S.; Sabra, N.M.; El-Gohary, A.A.; Mohamed, F.F.; Gaber, M.H. Treatment merits of Latanoprost/Thymoquinone—Encapsulated liposome for glaucomatus rabbits. Int. J. Pharm. 2018, 548, 597–608. [Google Scholar] [CrossRef]
- Antimisiaris, S.G.; Marazioti, A.; Kannavou, M.; Natsaridis, E.; Gkartziou, F.; Kogkos, G.; Mourtas, S. Overcoming barriers by local drug delivery with liposomes. Adv. Drug Deliv. Rev. 2021, 174, 53–86. [Google Scholar] [CrossRef]
- García-Posadas, L.; Contreras-Ruiz, L.; López-García, A.; Álvarez, S.V.; Maldonado, M.J.; Diebold, Y. Hyaluronan receptors in the human ocular surface: A descriptive and comparative study of RHAMM and CD44 in tissues, cell lines and freshly collected samples. Histochem. Cell Biol. 2011, 137, 165–176. [Google Scholar] [CrossRef]
- Fernandes-Cunha, G.M.; Na, K.-S.; Putra, I.; Lee, H.J.; Hull, S.; Cheng, Y.-C.; Blanco, I.J.; Eslani, M.; Djalilian, A.R.; Myung, D. Corneal Wound Healing Effects of Mesenchymal Stem Cell Secretome Delivered Within a Viscoelastic Gel Carrier. Stem Cells Transl. Med. 2019, 8, 478–489. [Google Scholar] [CrossRef]
- Piazzini, V.; Cinci, L.; D’Ambrosio, M.; Luceri, C.; Bilia, A.R.; Bergonzi, M.C. Solid Lipid Nanoparticles and Chitosan-coated Solid Lipid Nanoparticles as Promising Tool for Silybin Delivery: Formulation, Characterization, and In vitro Evaluation. Curr. Drug Deliv. 2018, 16, 142–152. [Google Scholar] [CrossRef] [PubMed]
- Ryman, B.E.; Tyrrell, D.A. Liposomes—Methodology and applications. Front. Biol. 1979, 48, 549–574. [Google Scholar]
- Righeschi, C.; Coronnello, M.; Mastrantoni, A.; Isacchi, B.; Bergonzi, M.C.; Mini, E.; Bilia, A.R. Strategy to provide a useful solution to effective delivery of dihydroartemisinin: Development, characterization and in vitro studies of liposomal formulations. Colloids Surf. B Biointerfaces 2014, 116, 121–127. [Google Scholar] [CrossRef] [PubMed]
- Mady, M.M.; Darwish, M.M.; Khalil, S.; Khalil, W.M. Biophysical studies on chitosan-coated liposomes. Eur. Biophys. J. 2009, 38, 1127–1133. [Google Scholar] [CrossRef]
- Mencucci, R.; Favuzza, E.; Becatti, M.; Tani, A.; Mazzantini, C.; Vignapiano, R.; Fiorillo, C.; Pellegrini-Giampietro, D.; Manetti, M.; Marini, M.; et al. Co-expression of the SARS-CoV-2 entry receptors ACE2 and TMPRSS2 in healthy human conjunctiva. Exp. Eye Res. 2021, 205, 108527. [Google Scholar] [CrossRef]
- Mencucci, R.; Favuzza, E.; Bottino, P.; Mazzantini, C.; Zanotto, E.; Pellegrini-Giampietro, D.E.; Landucci, E. A new ophthalmic formulation containing antiseptics and dexpanthenol: In vitro antimicrobial activity and effects on corneal and conjunctival epithelial cells. Exp. Eye Res. 2020, 201, 108269. [Google Scholar] [CrossRef]
- Hong, S.-S.; Kim, S.H.; Lim, S.-J. Effects of triglycerides on the hydrophobic drug loading capacity of saturated phosphatidylcholine-based liposomes. Int. J. Pharm. 2015, 483, 142–150. [Google Scholar] [CrossRef]
- Hsieh, Y.-F.; Chen, T.-L.; Wang, Y.-T.; Chang, J.-H.; Chang, H.-M. Properties of Liposomes Prepared with Various Lipids. J. Food Sci. 2002, 67, 2808–2813. [Google Scholar] [CrossRef]
- Gilbert, E.; Roussel, L.; Serre, C.; Sandouk, R.; Salmon, D.; Kirilov, P.; Haftek, M.; Falson, F.; Pirot, F. Percutaneous absorption of benzophenone-3 loaded lipid nanoparticles and polymeric nanocapsules: A comparative study. Int. J. Pharm. 2016, 504, 48–58. [Google Scholar] [CrossRef]
- Nasr, A.M.; Qushawy, M.K.; Elkhoudary, M.M.; Gawish, A.Y.; Elhady, S.S.; Swidan, S.A. Quality by Design for the Development and Analysis of Enhanced In-Situ Forming Vesicles for the Improvement of the Bioavailability of Fexofenadine HCl In Vitro and In Vivo. Pharmaceutics 2020, 12, 409. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, B.K.; Hardenia, S.S. Microemulsion Drug Delivery System: For Oral Bioavailability Enhancement of Glipizide. J. Adv. Pharm. Educ. Res. 2011, 1, 195–200. [Google Scholar]
- Yeo, L.K.; Chaw, C.S.; Elkordy, A.A. The Effects of Hydration Parameters and Co-Surfactants on Methylene Blue-Loaded Niosomes Prepared by the Thin Film Hydration Method. Pharmaceutics 2019, 12, 46. [Google Scholar] [CrossRef]
- Zeng, W.; Li, Q.; Wan, T.; Liu, C.; Pan, W.; Wu, Z.; Zhang, G.; Pan, J.; Qin, M.; Lin, Y.; et al. Hyaluronic acid-coated niosomes facilitate tacrolimus ocular delivery: Mucoadhesion, precorneal retention, aqueous humor pharmacokinetics, and transcorneal permeability. Colloids Surf. B Biointerfaces 2016, 141, 28–35. [Google Scholar] [CrossRef]
- Ghosh, P.; Hutadilok, N.; Adam, N.; Lentini, A. Interactions of hyaluronan (hyaluronic acid) with phospholipids as determined by gel permeation chromatography, multi-angle laser-light-scattering photometry and 1H-NMR spectroscopy. Int. J. Biol. Macromol. 1994, 16, 237–244. [Google Scholar] [CrossRef]
- Gaete, C.G.; Tsapis, N.; Silva, L.; Bourgaux, C.; Fattal, E. Morphology, structure and supramolecular organization of hybrid 1,2-dipalmitoyl-sn-glycero-3-phosphatidylcholine–hyaluronic acid microparticles prepared by spray drying. Eur. J. Pharm. Sci. 2008, 34, 12–21. [Google Scholar] [CrossRef] [PubMed]
- Ionov, R.; El-Abed, A.; Goldmann, M.; Peretti, P. Interactions of lipid monolayers with the natural biopolymer hyaluronic acid. Biochim. Biophys. Acta (BBA)-Biomembr. 2004, 1667, 200–207. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Mayol, L.; Quaglia, F.; Borzacchiello, A.; Ambrosio, L.; La Rotonda, M.I. A novel poloxamers/hyaluronic acid in situ forming hydrogel for drug delivery: Rheological, mucoadhesive and in vitro release properties. Eur. J. Pharm. Biopharm. 2008, 70, 199–206. [Google Scholar] [CrossRef] [PubMed]
- El Kechai, N.; Mamelle, E.; Nguyen, Y.; Huang, N.; Nicolas, V.; Chaminade, P.; Yen-Nicolaÿ, S.; Gueutin, C.; Granger, B.; Ferrary, E.; et al. Hyaluronic acid liposomal gel sustains delivery of a corticoid to the inner ear. J. Control. Release 2016, 226, 248–257. [Google Scholar] [CrossRef]
- Jeon, S.; Yoo, C.Y.; Park, S.N. Improved stability and skin permeability of sodium hyaluronate-chitosan multilayered liposomes by Layer-by-Layer electrostatic deposition for quercetin delivery. Colloids Surf. B Biointerfaces 2015, 129, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Calvo, P.; Remuñán-López, C.; Vila-Jato, J.L.; Alonso, M.J. Novel hydrophilic chitosan-polyethylene oxide nanoparticles as protein carriers. J. Appl. Polym. Sci. 1997, 63, 125–132. [Google Scholar] [CrossRef]
- Arpicco, S.; Lerda, C.; Pozza, E.D.; Costanzo, C.; Tsapis, N.; Stella, B.; Donadelli, M.; Dando, I.; Fattal, E.; Cattel, L.; et al. Hyaluronic acid-coated liposomes for active targeting of gemcitabine. Eur. J. Pharm. Biopharm. 2013, 85, 373–380. [Google Scholar] [CrossRef]
- Moustafa, M.A.; Elnaggar, Y.; El-Refaie, W.M.; Abdallah, O.Y. Hyalugel-integrated liposomes as a novel ocular nanosized delivery system of fluconazole with promising prolonged effect. Int. J. Pharm. 2017, 534, 14–24. [Google Scholar] [CrossRef]
- Lin, J.; Wu, H.; Wang, Y.; Lin, J.; Chen, Q.; Zhu, X. Preparation and ocular pharmacokinetics of hyaluronan acid-modified mucoadhesive liposomes. Drug Deliv. 2016, 23, 1144–1151. [Google Scholar] [CrossRef] [PubMed]
- He, M.; Zhao, Z.; Yin, L.; Tang, C.; Yin, C. Hyaluronic acid coated poly(butyl cyanoacrylate) nanoparticles as anticancer drug carriers. Int. J. Pharm. 2009, 373, 165–173. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.J.; Jung, M.-Y.; Pak, H.-J.; Park, J.-H.; Kim, M.; Chuck, R.S.; Park, C.Y. Development of a novel hyaluronic acid membrane for the treatment of ocular surface diseases. Sci. Rep. 2021, 11, 2351. [Google Scholar] [CrossRef] [PubMed]


| Sample (4:1) | Size (nm) | PdI |
|---|---|---|
| PC:CH | 85 ± 0 | 0.18 ± 0.01 |
| PC:Plurol Oleique | 141 ± 1 | 0.17 ± 0.02 |
| PC:Maisine | 232 ± 1 | 0.07 ± 0.01 |
| PC:Trascutol P | 182 ± 3 | 0.32 ± 0.03 |
| PC:Capryol 90 | 516 ± 1 | 0.61 ± 0.01 |
| PC:Lauroglycol 90 | 640 ± 7 | 0.64 ± 0.04 |
| Sample | EE% |
|---|---|
| PC:CH 4:1 | 40 ± 0 |
| PC:Plurol Oleique 4:1 | 63 ± 5 |
| PC:Plurol Oleique 6:1 | 73 ± 3 |
| PC:Maisine 4:1 | 53 ± 6 |
| PC:Trascutol P 4:1 | 40 ± 0 |
| Sample | Size (nm) | PdI | Zeta-Potential (mV) | EE% |
|---|---|---|---|---|
| LP | 114 ± 3 | 0.23 ± 0.01 | −23 ± 1 | |
| LP-TQ | 146 ± 2 | 0.15 ± 0.03 | −26 ± 3 | 73 ± 3 |
| LP-TQ-HA | 166 ± 3 | 0.27 ± 0.01 | −36 ± 1 | 73 ± 4 |
| LP-FITC | 106± 2 | 0.22 ± 0.01 | −25 ± 2 | 98 ± 1 |
| LP-FITC-HA | 118 ± 1 | 0.25 ± 0.01 | −34 ± 1 | 97 ± 2 |
| HA % w/v | Size (nm) | PdI |
|---|---|---|
| 0.1% | 166 ± 3 | 0.27 ± 0.01 |
| 0.2% | 558 ± 17 | 0.53 ± 0.07 |
| 0.5% | 422 ± 23 | 0.49 ± 0.12 |
| Sample | Months | Size (nm) | PdI | EE% |
|---|---|---|---|---|
| LP-TQ | 0 | 146 ± 2 | 0.15 ± 0.03 | 73 ± 3 |
| 1 | 143 ± 2 | 0.18 ± 0.03 | 69 ± 2 | |
| 2 | 113 ± 3 | 0.23 ± 0.00 | 50 ± 1 | |
| LP-TQ-HA | 0 | 166 ± 3 | 0.27 ± 0.01 | 73 ± 4 |
| 1 | 160 ± 0 | 0.21 ± 0.01 | 71 ± 2 | |
| 2 | 156 ± 1 | 0.18 ± 0.01 | 68 ± 3 |
| Sample | Days | Size (nm) | PdI | EE% |
|---|---|---|---|---|
| LP-FITC | 0 | 106 ± 2 | 0.22 ± 0.01 | 98 ± 1 |
| 6 | 112 ± 1 | 0.23 ± 0.01 | 98 ± 0 | |
| LP-FITC-HA | 0 | 118 ± 1 | 0.25 ± 0.01 | 98 ± 4 |
| 6 | 125 ± 1 | 0.27 ± 0.01 | 97 ± 1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Landucci, E.; Bonomolo, F.; De Stefani, C.; Mazzantini, C.; Pellegrini-Giampietro, D.E.; Bilia, A.R.; Bergonzi, M.C. Preparation of Liposomal Formulations for Ocular Delivery of Thymoquinone: In Vitro Evaluation in HCEC-2 e HConEC Cells. Pharmaceutics 2021, 13, 2093. https://doi.org/10.3390/pharmaceutics13122093
Landucci E, Bonomolo F, De Stefani C, Mazzantini C, Pellegrini-Giampietro DE, Bilia AR, Bergonzi MC. Preparation of Liposomal Formulations for Ocular Delivery of Thymoquinone: In Vitro Evaluation in HCEC-2 e HConEC Cells. Pharmaceutics. 2021; 13(12):2093. https://doi.org/10.3390/pharmaceutics13122093
Chicago/Turabian StyleLanducci, Elisa, Francesca Bonomolo, Chiara De Stefani, Costanza Mazzantini, Domenico Edoardo Pellegrini-Giampietro, Anna Rita Bilia, and Maria Camilla Bergonzi. 2021. "Preparation of Liposomal Formulations for Ocular Delivery of Thymoquinone: In Vitro Evaluation in HCEC-2 e HConEC Cells" Pharmaceutics 13, no. 12: 2093. https://doi.org/10.3390/pharmaceutics13122093
APA StyleLanducci, E., Bonomolo, F., De Stefani, C., Mazzantini, C., Pellegrini-Giampietro, D. E., Bilia, A. R., & Bergonzi, M. C. (2021). Preparation of Liposomal Formulations for Ocular Delivery of Thymoquinone: In Vitro Evaluation in HCEC-2 e HConEC Cells. Pharmaceutics, 13(12), 2093. https://doi.org/10.3390/pharmaceutics13122093








