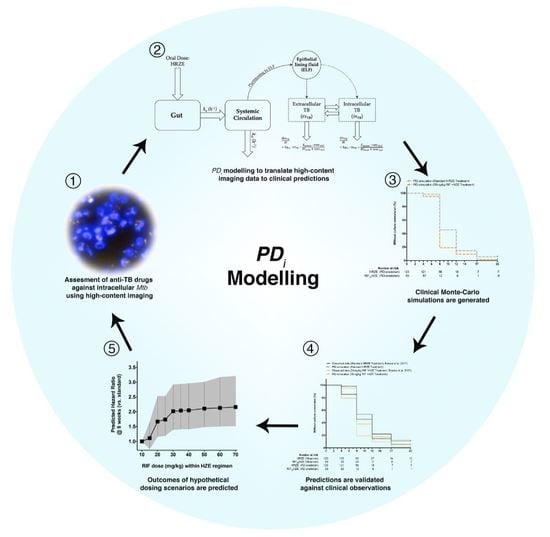
Intracellular PD Modelling (PDi) for the Prediction of Clinical Activity of Increased Rifampicin Dosing
Abstract
1. Introduction
2. Materials and Methods
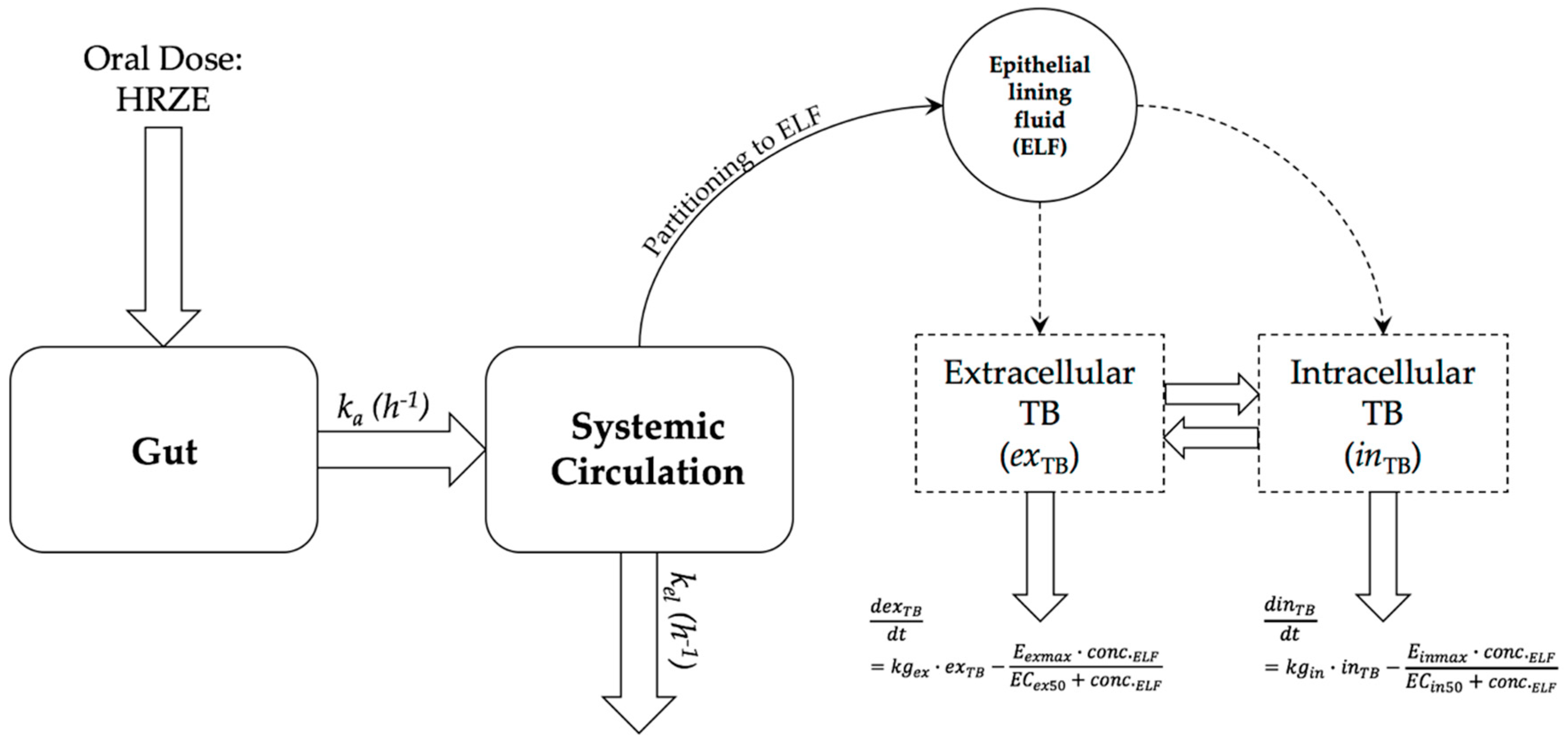
2.1. PDi Modelling
2.2. Hazard Ratio Calculation
2.3. PK Monte Carlo Simulations
3. Results
3.1. PDi Modelling Predicts Efficacy of Standard and High-Dose RIF-Containing Treatments
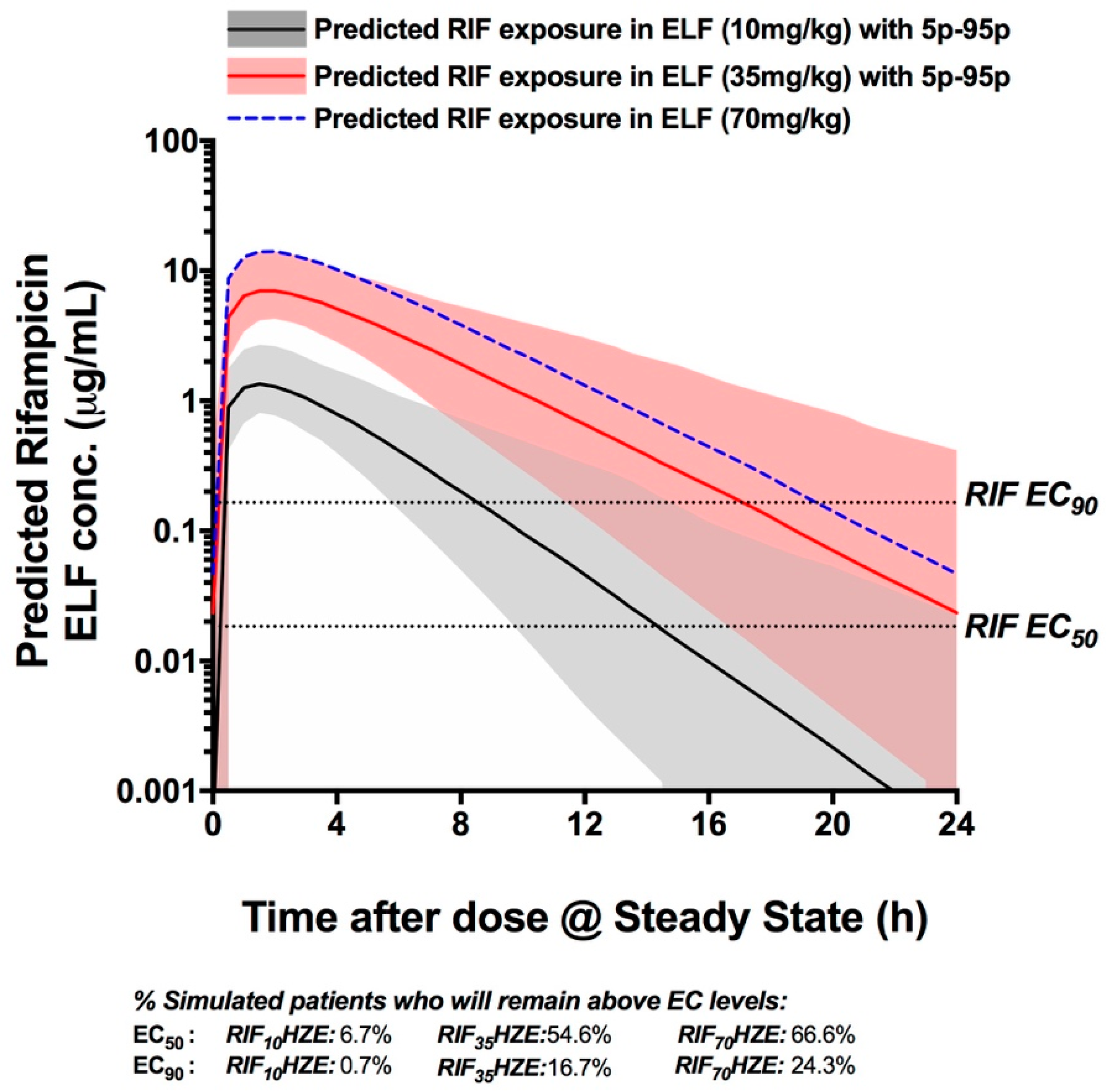
3.2. PDi Modelling Suggests No Further Reduction in Time to Culture Conversion Rates by Increasing RIF Dose Beyond 35 mg/kg
3.3. PDi Modelling Predicts a Dose-Effect Relationship of RIF
3.4. PK Simulation Analysis
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Global Tuberculosis Report; World Health Organization: Geneva, Switzerland, 2017. [Google Scholar]
- World Health Organization. United Nations General Assembly on Ending Tuberculosis; World Health Organization: Geneva, Switzerland, 2018. [Google Scholar]
- Controlled clinical trial of four short-course regimens of chemotherapy for two durations in the treatment of pulmonary tuberculosis: First report: Third East African/British Medical Research Councils study. Am. Rev. Respir. Dis. 1978, 118, 39–48. [CrossRef]
- Hu, Y.; Liu, A.; Ortega-Muro, F.; Alameda-Martin, L.; Mitchison, D.; Coates, A. High-dose rifampicin kills persisters, shortens treatment duration, and reduces relapse rate in vitro and in vivo. Front. Microbiol. 2015, 6, 641. [Google Scholar] [CrossRef] [PubMed]
- Boeree, M.J.; Diacon, A.H.; Dawson, R.; Narunsky, K.; du Bois, J.; Venter, A.; Phillips, P.P.; Gillespie, S.H.; McHugh, T.D.; Hoelscher, M.; et al. A dose-ranging trial to optimize the dose of rifampin in the treatment of tuberculosis. Am. J. Respir. Crit. Care Med. 2015, 191, 1058–1065. [Google Scholar] [CrossRef] [PubMed]
- de Steenwinkel, J.E.; Aarnoutse, R.E.; de Knegt, G.J.; ten Kate, M.T.; Teulen, M.; Verbrugh, H.A.; Boeree, M.J.; van Soolingen, D.; Bakker-Woudenberg, I.A. Optimization of the rifampin dosage to improve the therapeutic efficacy in tuberculosis treatment using a murine model. Am. J. Respir. Crit. Care Med. 2013, 187, 1127–1134. [Google Scholar] [CrossRef] [PubMed]
- Velasquez, G.E.; Brooks, M.B.; Coit, J.M.; Pertinez, H.; Vargas Vasquez, D.; Sanchez Garavito, E.; Calderon, R.I.; Jimenez, J.; Tintaya, K.; Peloquin, C.A.; et al. Efficacy and Safety of High-Dose Rifampin in Pulmonary Tuberculosis. A Randomized Controlled Trial. Am. J. Respir. Crit. Care Med. 2018, 198, 657–666. [Google Scholar] [CrossRef]
- Jayaram, R.; Gaonkar, S.; Kaur, P.; Suresh, B.L.; Mahesh, B.N.; Jayashree, R.; Nandi, V.; Bharat, S.; Shandil, R.K.; Kantharaj, E.; et al. Pharmacokinetics-pharmacodynamics of rifampin in an aerosol infection model of tuberculosis. Antimicrob. Agents ChemoTher. 2003, 47, 2118–2124. [Google Scholar] [CrossRef]
- Goutelle, S.; Bourguignon, L.; Maire, P.H.; Van Guilder, M.; Conte, J.E., Jr.; Jelliffe, R.W. Population modeling and Monte Carlo simulation study of the pharmacokinetics and antituberculosis pharmacodynamics of rifampin in lungs. Antimicrob. Agents ChemoTher. 2009, 53, 2974–2981. [Google Scholar] [CrossRef]
- Kreis, B.; Pretet, S.; Birenbaum, J.; Guibout, P.; Hazeman, J.J.; Orin, E.; Perdrizet, S.; Weil, J. Two three-month treatment regimens for pulmonary tuberculosis. Bull. Int. Union Tuberc. 1976, 51, 71–75. [Google Scholar]
- Diacon, A.H.; Patientia, R.F.; Venter, A.; van Helden, P.D.; Smith, P.J.; McIlleron, H.; Maritz, J.S.; Donald, P.R. Early bactericidal activity of high-dose rifampin in patients with pulmonary tuberculosis evidenced by positive sputum smears. Antimicrob. Agents ChemoTher. 2007, 51, 2994–2996. [Google Scholar] [CrossRef]
- Peloquin, C. What is the ‘right’ dose of rifampin? Int. J. Tuberc. Lung Dis. 2003, 7, 3–5. [Google Scholar]
- van Ingen, J.; Aarnoutse, R.E.; Donald, P.R.; Diacon, A.H.; Dawson, R.; Plemper van Balen, G.; Gillespie, S.H.; Boeree, M.J. Why Do We Use 600 mg of Rifampicin in Tuberculosis Treatment? Clin. Infect. Dis. 2011, 52, e194–e199. [Google Scholar] [CrossRef] [PubMed]
- Boeree, M.J.; Heinrich, N.; Aarnoutse, R.; Diacon, A.H.; Dawson, R.; Rehal, S.; Kibiki, G.S.; Churchyard, G.; Sanne, I.; Ntinginya, N.E.; et al. High-dose rifampicin, moxifloxacin, and SQ109 for treating tuberculosis: A multi-arm, multi-stage randomised controlled trial. Lancet Infect. Dis. 2017, 17, 39–49. [Google Scholar] [CrossRef]
- te Brake, L.H.M.; Aarnoutse, R.; Svensson, E.M.; Svensson, R.J.; Simonsson, U.S.H.; Boeree, M.J.; Hoelscher, M.; Heinrich, N.; Konsten, S.; Churchyard, G.; et al. The Potential for Treatment Shortening With Higher Rifampicin Doses: Relating Drug Exposure to Treatment Response in Patients With Pulmonary Tuberculosis. Clin. Infect. Dis. 2018, 67, 34–41. [Google Scholar] [CrossRef]
- Aljayyoussi, G.; Jenkins, V.A.; Sharma, R.; Ardrey, A.; Donnellan, S.; Ward, S.A.; Biagini, G.A. Pharmacokinetic-Pharmacodynamic modelling of intracellular Mycobacterium tuberculosis growth and kill rates is predictive of clinical treatment duration. Sci. Rep. 2017, 7, 502. [Google Scholar] [CrossRef] [PubMed]
- Neely, M.N.; van Guilder, M.G.; Yamada, W.M.; Schumitzky, A.; Jelliffe, R.W. Accurate detection of outliers and subpopulations with Pmetrics, a nonparametric and parametric pharmacometric modeling and simulation package for R. Ther. Drug Monit. 2012, 34, 467–476. [Google Scholar] [CrossRef] [PubMed]
- Conte, J.E., Jr.; Golden, J.A.; Duncan, S.; McKenna, E.; Zurlinden, E. Intrapulmonary concentrations of pyrazinamide. Antimicrob. Agents ChemoTher. 1999, 43, 1329–1333. [Google Scholar] [CrossRef] [PubMed]
- Conte, J.E., Jr.; Golden, J.A.; McQuitty, M.; Kipps, J.; Duncan, S.; McKenna, E.; Zurlinden, E. Effects of gender, AIDS, and acetylator status on intrapulmonary concentrations of isoniazid. Antimicrob. Agents ChemoTher. 2002, 46, 2358–2364. [Google Scholar] [CrossRef]
- Kiem, S.; Schentag, J.J. Interpretation of antibiotic concentration ratios measured in epithelial lining fluid. Antimicrob. Agents ChemoTher. 2008, 52, 24–36. [Google Scholar] [CrossRef]
- Conte, J.E., Jr.; Golden, J.A.; Kipps, J.; Lin, E.T.; Zurlinden, E. Effects of AIDS and gender on steady-state plasma and intrapulmonary ethambutol concentrations. Antimicrob. Agents ChemoTher. 2001, 45, 2891–2896. [Google Scholar] [CrossRef]
- Conte, J.E.; Golden, J.A.; Kipps, J.E.; Lin, E.T.; Zurlinden, E. Effect of sex and AIDS status on the plasma and intrapulmonary pharmacokinetics of rifampicin. Clin. Pharmacokinet 2004, 43, 395–404. [Google Scholar] [CrossRef]
- de Knegt, G.J.; Dickinson, L.; Pertinez, H.; Evangelopoulos, D.; McHugh, T.D.; Bakker-Woudenberg, I.; Davies, G.R.; de Steenwinkel, J.E.M. Assessment of treatment response by colony forming units, time to culture positivity and the molecular bacterial load assay compared in a mouse tuberculosis model. Tuberculosis (Edinb) 2017, 105, 113–118. [Google Scholar] [CrossRef] [PubMed]
- Wilkins, J.J.; Savic, R.M.; Karlsson, M.O.; Langdon, G.; McIlleron, H.; Pillai, G.; Smith, P.J.; Simonsson, U.S. Population pharmacokinetics of rifampin in pulmonary tuberculosis patients, including a semimechanistic model to describe variable absorption. Antimicrob. Agents ChemoTher. 2008, 52, 2138–2148. [Google Scholar] [CrossRef] [PubMed]
- Jonsson, S.; Davidse, A.; Wilkins, J.; Van der Walt, J.S.; Simonsson, U.S.; Karlsson, M.O.; Smith, P.; McIlleron, H. Population pharmacokinetics of ethambutol in South African tuberculosis patients. Antimicrob. Agents ChemoTher. 2011, 55, 4230–4237. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wilkins, J.J.; Langdon, G.; McIlleron, H.; Pillai, G.; Smith, P.J.; Simonsson, U.S. Variability in the population pharmacokinetics of isoniazid in South African tuberculosis patients. Br. J. Clin. Pharmacol. 2011, 72, 51–62. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2017. [Google Scholar]
- Svensson, R.J.; Aarnoutse, R.E.; Diacon, A.H.; Dawson, R.; Gillespie, S.H.; Boeree, M.J.; Simonsson, U.S.H. A Population Pharmacokinetic Model Incorporating Saturable Pharmacokinetics and Autoinduction for High Rifampicin Doses. Clin. Pharmacol. Ther. 2018, 103, 674–683. [Google Scholar] [CrossRef] [PubMed]
- IBM Corp. IBM SPSS Statistics for Windows, Version 24.0, Released 2016; IBM Corp.: Armonk, NY, USA, 2016.
- van Zyl-Smit, R.N.; Binder, A.; Meldau, R.; Mishra, H.; Semple, P.L.; Theron, G.; Peter, J.; Whitelaw, A.; Sharma, S.K.; Warren, R.; et al. Comparison of quantitative techniques including Xpert MTB/RIF to evaluate mycobacterial burden. PLoS ONE 2011, 6, e28815. [Google Scholar] [CrossRef] [PubMed]
- Fox, W.; Nunn, A.J. The cost of antituberculous drug regimens. Am. Rev. Respir. Dis. 1979, 120, 503–509. [Google Scholar] [CrossRef] [PubMed]
- Wicha, S.G.; Clewe, O.; Svensson, R.J.; Gillespie, S.H.; Hu, Y.; Coates, A.R.M.; Simonsson, U.S.H. Forecasting Clinical Dose-Response From Preclinical Studies in Tuberculosis Research: Translational Predictions With Rifampicin. Clin. Pharmacol. Ther. 2018, 104, 1208–1218. [Google Scholar] [CrossRef] [PubMed]
- Bartelink, I.H.; Zhang, N.; Keizer, R.J.; Strydom, N.; Converse, P.J.; Dooley, K.E.; Nuermberger, E.L.; Savic, R.M. New Paradigm for Translational Modeling to Predict Long-term Tuberculosis Treatment Response. Clin. Transl. Sci. 2017, 10, 366–379. [Google Scholar] [CrossRef]
- Acocella, G. Pharmacokinetics and metabolism of rifampin in humans. Rev. Infect. Dis. 1983, 5 (Suppl. 3), S428–S432. [Google Scholar] [CrossRef]
- Decroix, G.; Kreis, B.; Sors, C.; Birembaum, J.; Le Lirzin, M.; Canetti, G. Comparison between regimes of rifampicin-isoniazid administered daily and administered twice a week (initial results of a comparative study conducted in 4 medical services of the Parisian region). Rev. Tuberc. Pneumol. 1969, 33, 751–768. [Google Scholar]


| Boeree et al. (2017) [14] (Observed) | PDi Prediction | ||||
|---|---|---|---|---|---|
| Standard HRZE | H35RZE | Standard HRZE | H35RZE | H70RZE | |
| Total in Analysis | 123 | 63 | 123 | 63 | 63 |
| Hazard ratio over 8 weeks (CI) * | N/A | 1.73 (1.07–2.82), p = 0.004 (unadjusted) | |||
| N/A | 2.06 (1.26–3.38), p = 0.004 (adjusted) | N/A | 2.04 (1.41–2.94), p < 0.001 | 2.16 (1.50–3.12), p < 0.001 | |
| Hazard ratio over 12 weeks (CI) | N/A | 1.46 (1.02–2.11), p = 0.04 (unadjusted) | |||
| N/A | 1.78 (1.22–2.58), p = 0.003 (adjusted) | N/A | 1.68 (1.21–2.32), p < 0.001 | 1.86 (1.35–2.57), p < 0.001 | |
| No. of culture conversions during 26-weeks (MGIT **) (% of patients) | 101 (82%) | 51 (81%) | 104 (94%) | 62 (99%) | 63 (100%) |
| Median time to culture conversion (IQR) *** | 62 (4–83) | 48 (34–69) | 55 (41–76) | 40 (34–51) | 39 (32–48) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aljayyoussi, G.; Donnellan, S.; Ward, S.A.; Biagini, G.A. Intracellular PD Modelling (PDi) for the Prediction of Clinical Activity of Increased Rifampicin Dosing. Pharmaceutics 2019, 11, 278. https://doi.org/10.3390/pharmaceutics11060278
Aljayyoussi G, Donnellan S, Ward SA, Biagini GA. Intracellular PD Modelling (PDi) for the Prediction of Clinical Activity of Increased Rifampicin Dosing. Pharmaceutics. 2019; 11(6):278. https://doi.org/10.3390/pharmaceutics11060278
Chicago/Turabian StyleAljayyoussi, Ghaith, Samantha Donnellan, Stephen A. Ward, and Giancarlo A. Biagini. 2019. "Intracellular PD Modelling (PDi) for the Prediction of Clinical Activity of Increased Rifampicin Dosing" Pharmaceutics 11, no. 6: 278. https://doi.org/10.3390/pharmaceutics11060278
APA StyleAljayyoussi, G., Donnellan, S., Ward, S. A., & Biagini, G. A. (2019). Intracellular PD Modelling (PDi) for the Prediction of Clinical Activity of Increased Rifampicin Dosing. Pharmaceutics, 11(6), 278. https://doi.org/10.3390/pharmaceutics11060278