Applying Supercritical Fluid Technology to Prepare Ibuprofen Solid Dispersions with Improved Oral Bioavailability
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
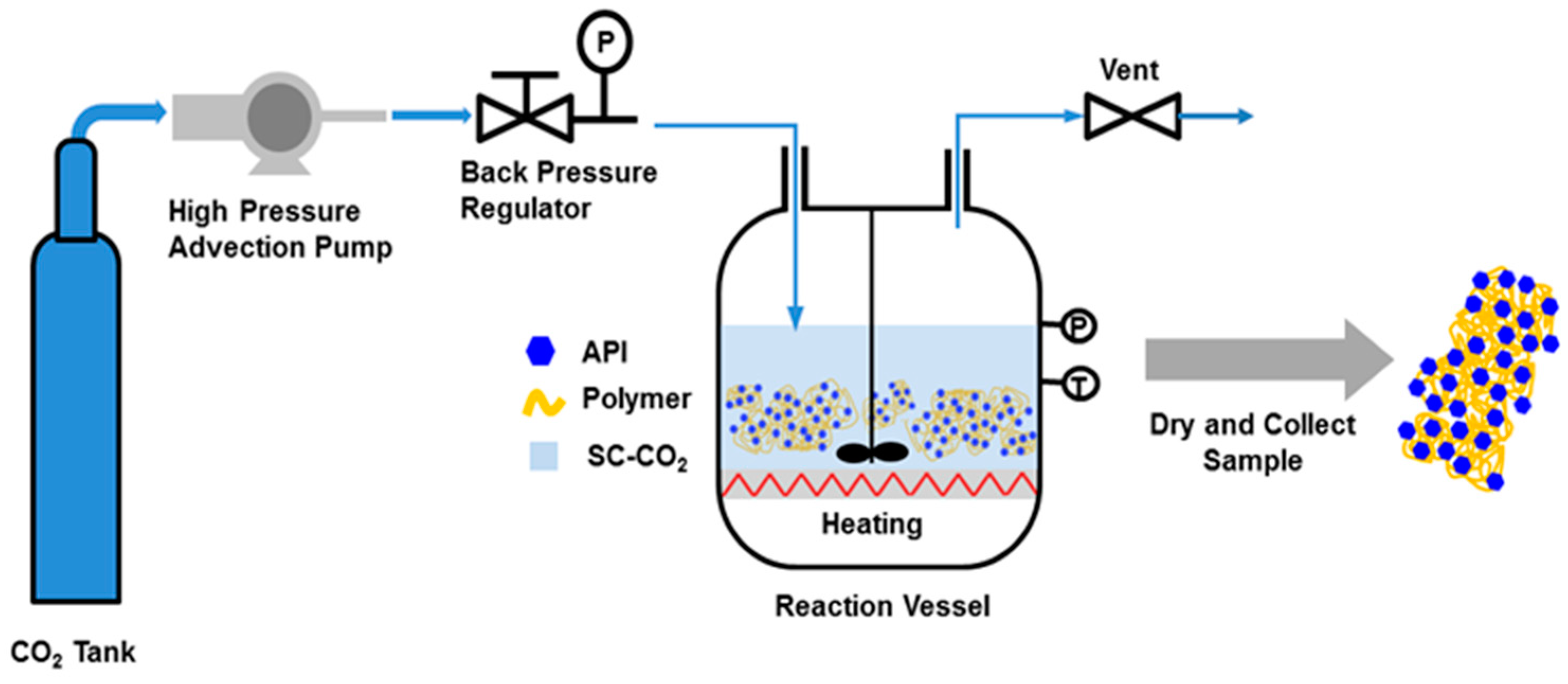
2.2. Preparation of SDs
2.3. Scanning Electron Microscopy (SEM)
2.4. Differential Scanning Calorimetry (DSC)
2.5. Powder X-Ray Diffraction (PXRD)
2.6. Dissolution Studies
2.7. Pharmacokinetic Studies in Rats
2.8. HPLC Analysis of IBU
2.9. Molecular Docking
2.10. Statistical Analysis
3. Results
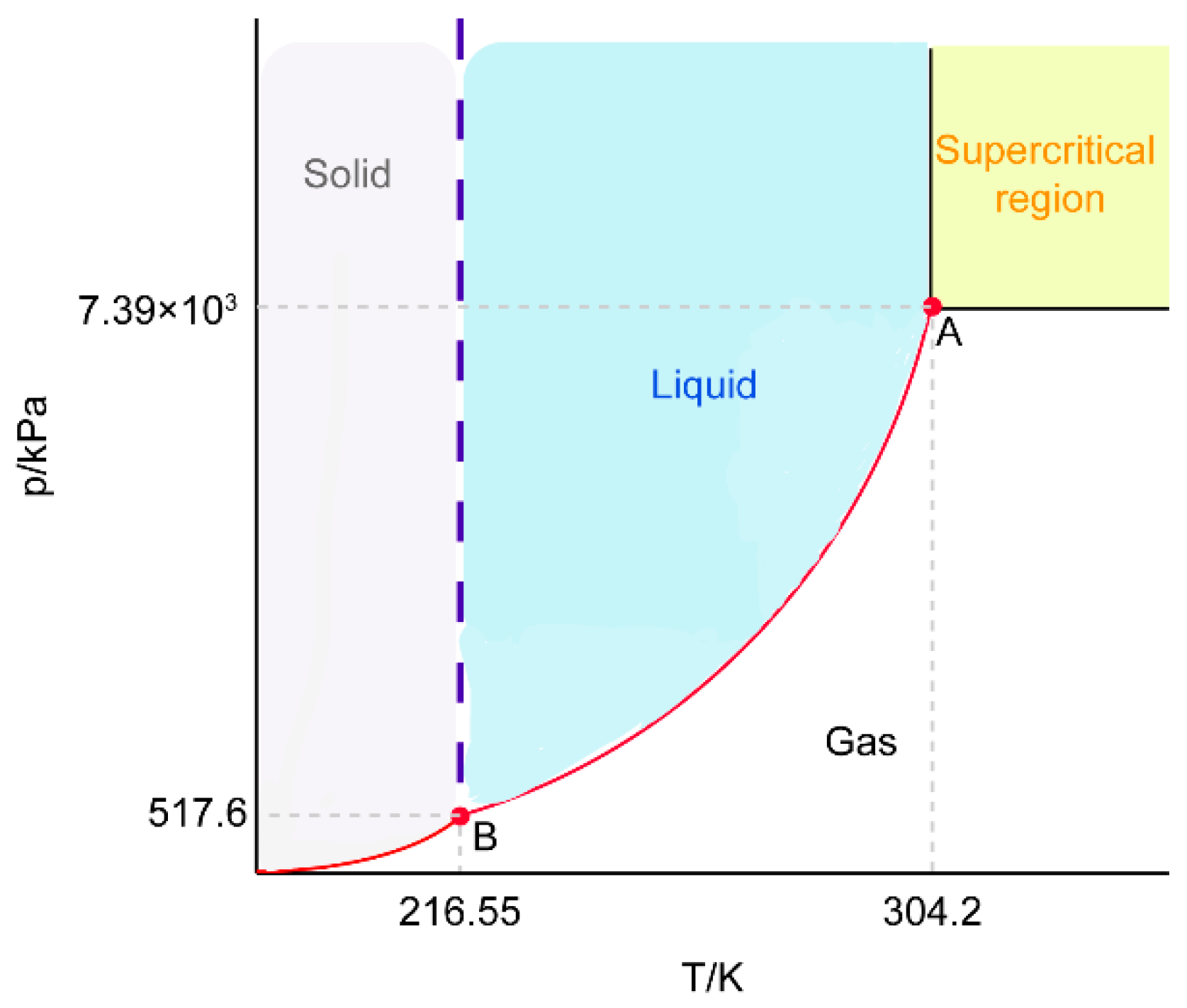
3.1. Optimization of Preparation Conditions
3.2. Characterization
3.2.1. SEM Results
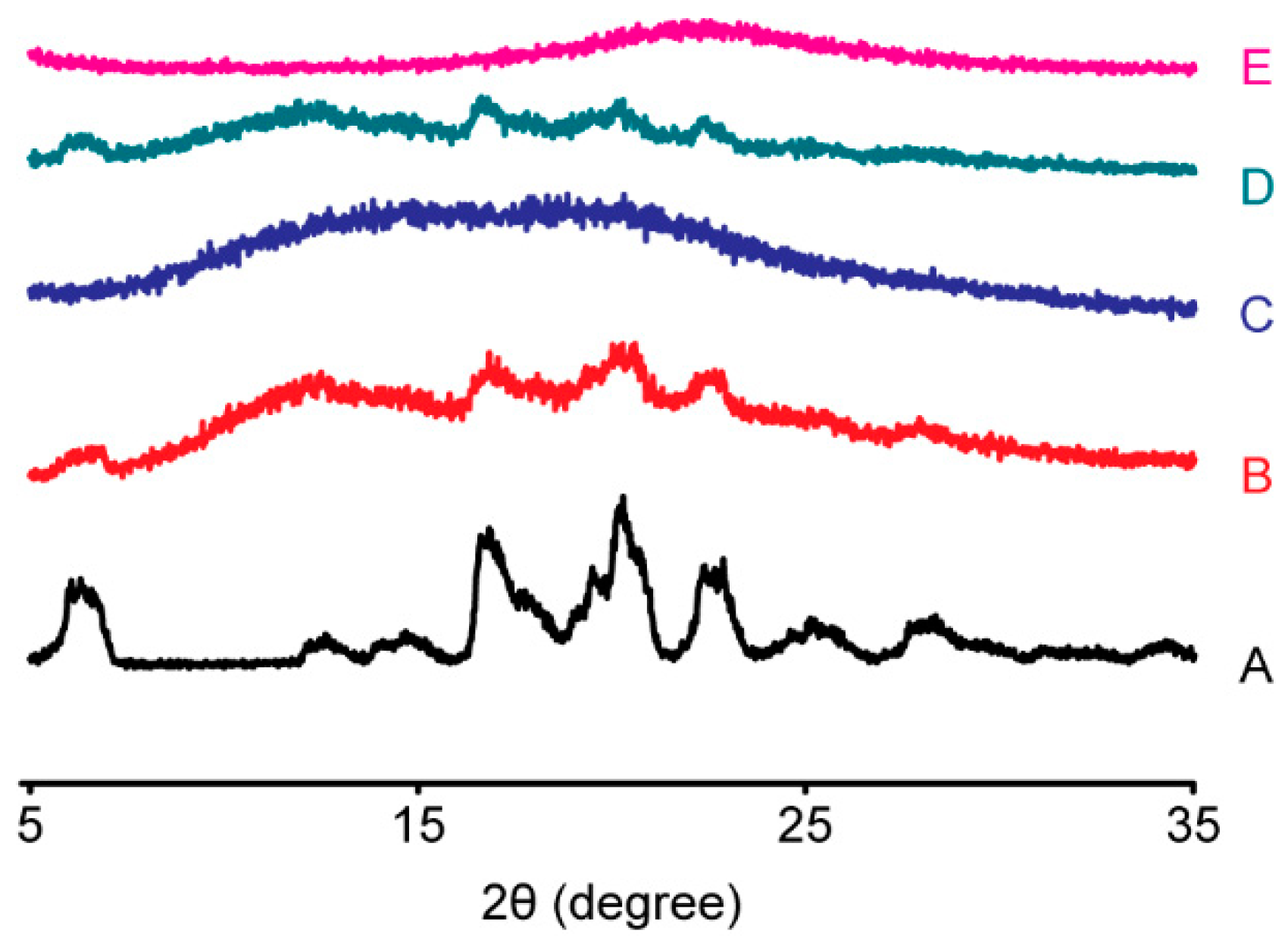
3.2.2. PXRD Analysis
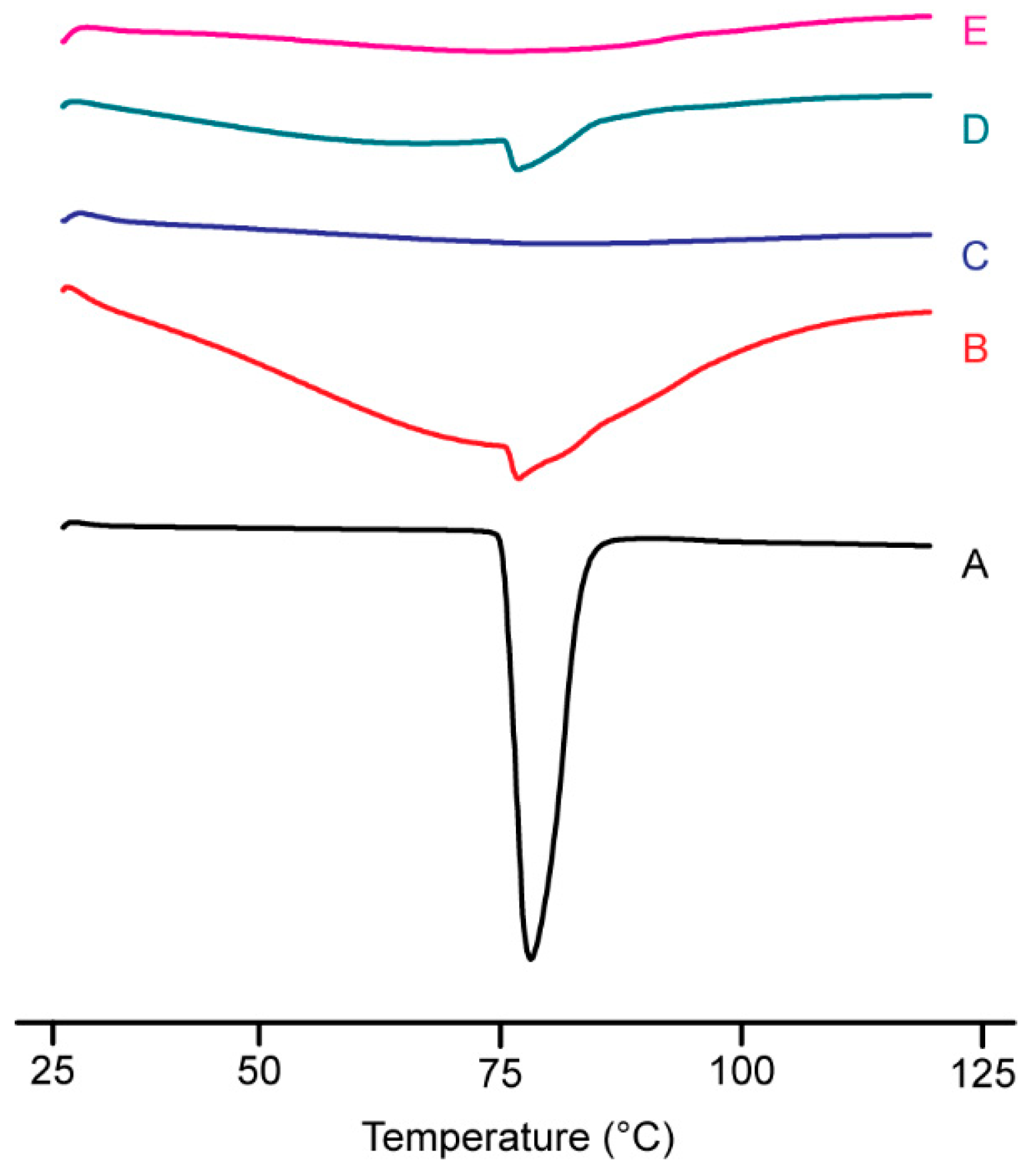
3.2.3. DSC Analysis
3.3. Dissolution Studies
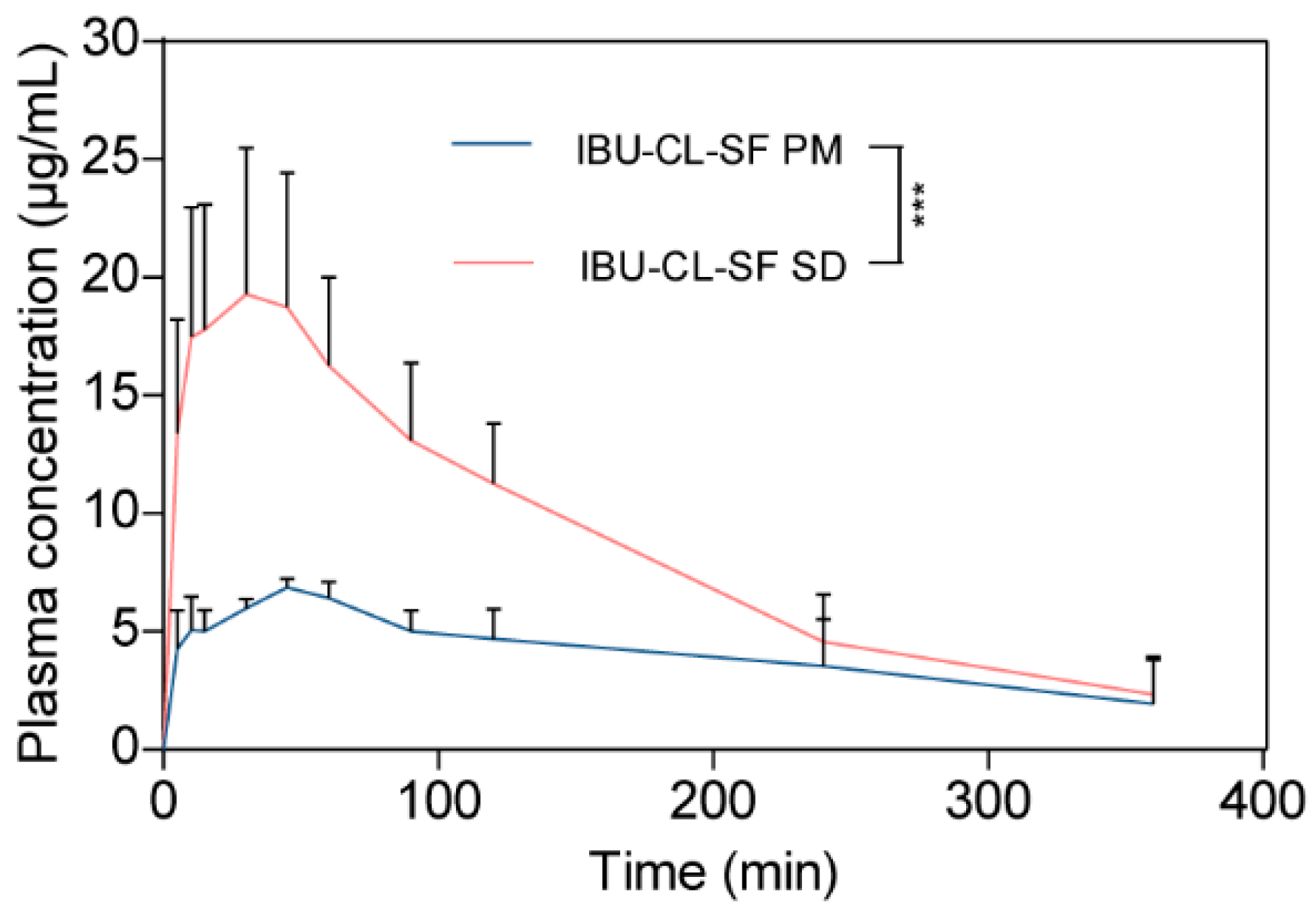
3.4. In Vivo Pharmacokinetic Evaluation
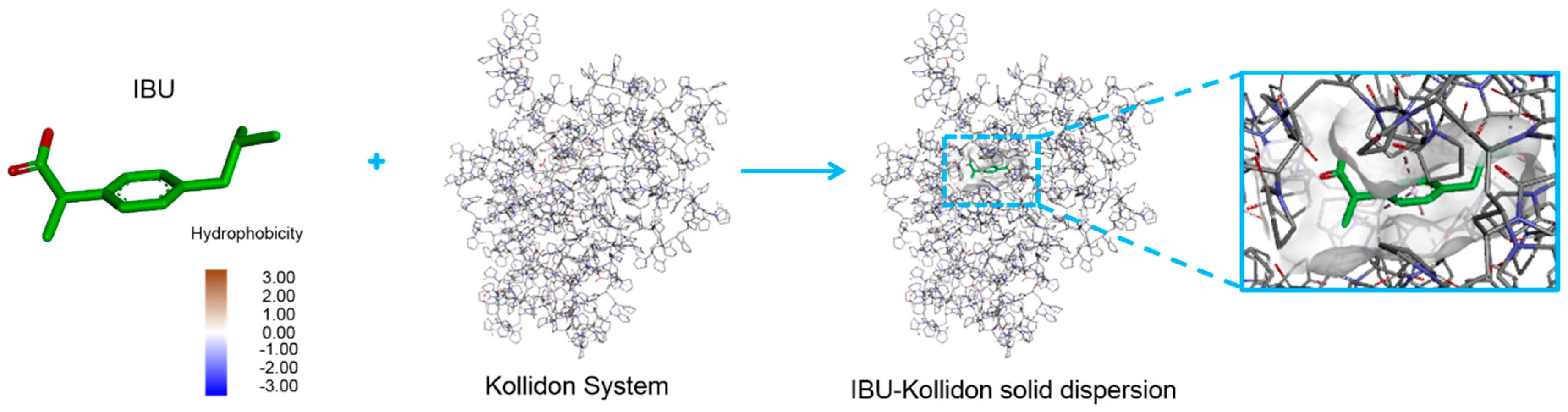
3.5. Drug–Polymer Interactions in Molecular Docking Simulation
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Brabander, C.D.; Vervaet, C.; Bortel, L.V.; Remon, J.P. Bioavailability of ibuprofen from hot-melt extruded mini-matrices. Int. J. Pharm. 2004, 271, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Horter, D.; Dressman, J.B. Influence of physicochemical properties on dissolution of drugs in the gastrointestinal tract. Adv. Drug Deliv. Rev. 2001, 46, 75–87. [Google Scholar] [CrossRef]
- Hussain, A.; Smith, G.; Khan, K.A.; Bukhari, N.I.; Pedge, N.I.; Ermolina, I. Solubility and dissolution rate enhancement of ibuprofen by co-milling with polymeric excipients. Eur. J. Pharm. Sci. 2018, 123, 395–403. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.P.; Chuang, C.H.; Wu, P.C.; Tsai, Y.H.; Huang, Y.B.; Tzeng, C.C.; Chen, Y.L.; Gao, M.Y.; Tsai, M.J. Amsacrine analog-loaded solid lipid nanoparticle to resolve insolubility for injection delivery: Characterization and pharmacokinetics. Drug Des. Dev. Ther. 2016, 4, 1019–1028. [Google Scholar] [CrossRef] [PubMed]
- Chiu, C.H.; Chang, C.C.; Lin, S.T.; Chyau, C.C.; Peng, R.Y. Improved Hepatoprotective Effect of Liposome-Encapsulated Astaxanthin in Lipopolysaccharide-Induced Acute Hepatotoxicity. Int. J. Mol. Sci. 2016, 17, 1128. [Google Scholar] [CrossRef] [PubMed]
- Obaidat, R.M.; Alnaief, M.; Jaeger, P.T. Significant solubility of carbon dioxide in Soluplus® facilitates impregnation of ibuprofen using supercritical fluid technology. Pharm. Dev. Technol. 2017, 23, 697–705. [Google Scholar] [CrossRef] [PubMed]
- Vo, C.L.N.; Park, C.; Lee, B.J. Current trends and future perspectives of solid dispersions containing poorly water-soluble drugs. Eur. J. Pharm. Biopharm. 2013, 85, 799–813. [Google Scholar] [CrossRef]
- Sinha, S.; Ali, A.; Baboota, S.; Ahuja, A.; Kumar, D.A.; Ali, J. Solid Dispersion as an Approach for Bioavailability Enhancement of Poorly Water-Soluble Drug Ritonavir. AAPS PharmSciTech 2010, 11, 518–527. [Google Scholar] [CrossRef]
- Ziaee, A.; Albadarin, A.B.; Padrela, L.; Faucher, A.; O’Reilly, E.J.; Walker, G.M. Spray drying ternary amorphous solid dispersions of ibuprofen—An investigation into critical formulation and processing parameters. Eur. J. Pharm. Biopharm. 2017, 120, 43–51. [Google Scholar] [CrossRef]
- Herbrink, M.; Schellens, J.H.M. Improving the solubility of nilotinib through novel spray-dried solid dispersions. Int. J. Pharm. 2017, 529, 294–302. [Google Scholar] [CrossRef]
- Nijlen, T.V.; Brennan, K.; Blaton, N.; Mooter, G.V. Improvement of the dissolution rate of artemisinin by means of supercritical fluid technology and solid dispersions. Int. J. Pharm. 2003, 254, 173–181. [Google Scholar] [CrossRef]
- Badens, E.; Majerik, V.; Horváth, G.; Szokonya, L.; Bosc, N.; Teillaud, E.; Charbit, G. Comparison of solid dispersions produced by supercritical antisolvent and spray-freezing technologies. Int. J. Pharm. 2009, 377, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Park, J.; Cho, W.; Cha, K.H.; Ahn, J.; Han, K.; Hwang, S.J. Solubilization of the poorly watersoluble drug, telmisartan, using supercritical anti-solvent (SAS) process. Int. J. Pharm. 2013, 441, 50–55. [Google Scholar] [CrossRef] [PubMed]
- Cardea, S.; Baldino, L.; Scognamiglio, M.; Reverchon, E. 3D PLLA/ibuprofen composite scaffolds obtained by a supercritical fluids assisted process. J. Mater. Sci. Mater. Med. 2014, 25, 989–998. [Google Scholar] [CrossRef] [PubMed]
- Thiry, J.; Lebrun, P.; Vinassa, C.; Adam, M.; Netchacoviych, L.; Ziemons, E.; Hubert, P.; Krier, F.; Evrard, B. Continuous Production of Itraconazole-based Solid Dispersions by Hot Melt Extrusion: Preformulation, Optimization and Design Space Determination. Int. J. Pharm. 2016, 515, 114–124. [Google Scholar] [CrossRef] [PubMed]
- Chmiel, K.; Knapik-kowalczuk, J.; Jurkiewicz, K.; Sawwichi, W.; Jachowicz, R.; Paluch, M. A New Method To Identify Physically Stable Concentration of Amorphous Solid Dispersions (I): Case of Flutamide + Kollidon VA64. Mol. Pharm. 2017, 14, 3370–3380. [Google Scholar] [CrossRef] [PubMed]
- Djuris, J.; Milovanovic, S.; Medarevic, D.; Dobricic, V.; Dapčević, A.; Ibric, S. Selection of the suitable polymer for supercritical fluid assisted preparation of carvedilol solid dispersions. Int. J. Pharm. 2018, 554, 190–200. [Google Scholar] [CrossRef]
- Xu, L.; Li, S.M.; Sunada, H. Preparation and evaluation of Ibuprofen solid dispersion systems with kollidon particles using a pulse combustion dryer system. Chem. Pharm. Bull. 2007, 55, 1545–1550. [Google Scholar] [CrossRef]
- Newa, M.; Bhandari, K.H.; Kim, J.O.; Im, J.S.; Kim, J.A.; Yoo, B.K.; Woo, J.S.; Choi, H.G.; Yong, C.S. Enhancement of Solubility, Dissolution and Bioavailability of Ibuprofen in Solid Dispersion Systems. Chem. Pharm. Bull. 2008, 56, 569–574. [Google Scholar] [CrossRef]
- Morris, G.M.; Huey, R.; Lindstrom, W.; Sanner, M.F.; Belew, R.K.; Goodsell, D.S.; Olson, A.J. Autodock4 and AutoDockTools4: Automated docking with selective receptor flexibility. J. Comput. Chem. 2009, 30, 2785–2791. [Google Scholar] [CrossRef]
- Zhang, Y.; Huo, M.; Zhou, J.; Zou, A.; Li, W.; Yao, C.; Xie, S. DDSolver: An Add-In Program for Modeling and Comparison of Drug Dissolution Profiles. AAPS J. 2010, 12, 263–271. [Google Scholar] [CrossRef] [PubMed]
- Nan, S.; Wang, T.; Yan, X. Synthesis and investigation of a self-assembled hydrogel based on hydroxyethyl cellulose and its in vitro ibuprofen drug release characteristics. RSC Adv. 2017, 7, 9500–9511. [Google Scholar] [CrossRef]
- Csapó, E.; Juhász, A.; Varga, N.; Sebok, D.; Hornok, V.; Janovák, L.; Dekany, I. Thermodynamic and kinetic characterization of pH-dependent interactions between bovine serum albumin and ibuprofen in 2D and 3D systems. Colloids Surf. A Physicochem. Eng. Asp. 2016, 504, 471–478. [Google Scholar] [CrossRef]
- Al-Khateeb, L.A.; Hakami, W.; Salam, M.A. Removal of non-steroidal anti-inflammatory drugs from water using high surface area nanographene: Kinetic and thermodynamic studies. J. Mol. Liq. 2017, 241, 733–741. [Google Scholar] [CrossRef]
- Haruo, S.; Kazumi, D.; Hiroshi, E.; Yorinobu, Y.; Sunada, H.; Otauka, A. Solid-State Interaction of Ibuprofen with Polyvinylpyrrolidone. Chem. Pharm. Bull. 1995, 43, 988–993. [Google Scholar] [CrossRef]
- Kankala, R.K.; Zhang, Y.S.; Wang, S.B.; Lee, C.H.; Chen, A.Z. Supercritical Fluid Technology: An Emphasis on Drug Delivery and Related Biomedical Applications. Adv. Healthc. Mater. 2017, 6, 1–31. [Google Scholar] [CrossRef] [PubMed]
- Skrdla, P.J.; Floyd, P.D.; Dell’Orco, P.C. Practical Estimation of Amorphous Solubility Enhancement Using Thermoanalytical Data: Determination of the Amorphous/Crystalline Solubility Ratio for Pure Indomethacin and Felodipine. J. Pharm. Sci. 2016, 105, 2625–2630. [Google Scholar] [CrossRef]
- Ainurofiq, A.; Mauludin, R.; Mudhakir, D. A Novel Desloratadine-Benzoic Acid Co-Amorphous Solid: Preparation, Characterization, and Stability Evaluation. Pharmaceutics 2018, 10, 85. [Google Scholar] [CrossRef]
- An, J.H.; Lim, C.; Kiyonga, A.; Mo, K.; Park, M.; Youn, W.; Choi, W.; Suh, Y.G.; Jung, K. Co-Amorphous Screening for the Solubility Enhancement of Poorly Water-Soluble Mirabegron and Investigation of Their Intermolecular Interactions and Dissolution Behaviors. Pharmaceutics 2018, 10, 149. [Google Scholar] [CrossRef]
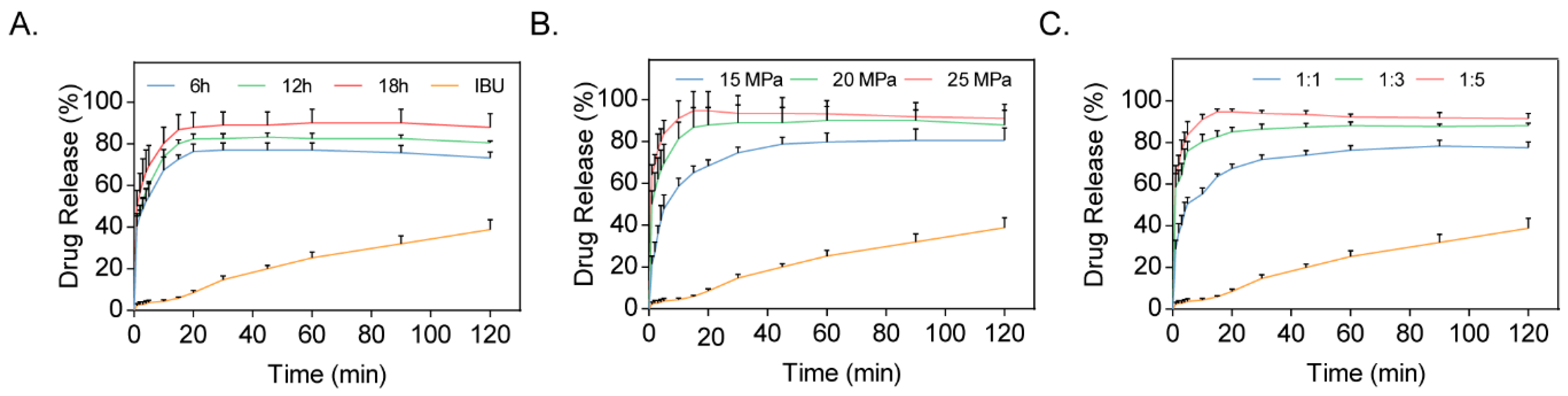


| Group | Pressure (MPa) | Reaction Time (h) | Drug-to-Carrier Ratio | Cumulative Percentage of Drug Release (%) |
|---|---|---|---|---|
| 1 | 20 | 6 | 1:5 | 73.21 |
| 2 | 20 | 12 | 1:5 | 80.44 |
| 3 | 20 | 18 | 1:5 | 87.90 |
| 4 | 15 | 18 | 1:5 | 80.58 |
| 5 | 25 | 18 | 1:5 | 91.05 |
| 6 | 20 | 18 | 1:1 | 77.45 |
| 7 | 20 | 18 | 1:3 | 91.45 |
| Equations | IBU-CL SD | IBU-CL-SF SD | IBU | IBU-CL PM | IBU-CL-SF PM |
|---|---|---|---|---|---|
| Zero-order F = C + kt | F = 51.441 + 0.376t r2 = 0.2997 | F = 71.947 + 0.266t r2 = 0.0875 | F = 2.131 + 0.332t r2 = 0.9769 | F = 16.276 + 0.369t r2 = 0.7279 | F = 25.417 + 0.288t r2 = 0.5396 |
| First-order F = a × (1 − e−kt) | F = 78.064 × (1 − e−0.366t) r2 = 0.9474 | F = 90.550 × (1 − e−0.862t) r2 = 0.9980 | F = 57.736 × (1−e−0.009t) r2 = 0.9915 | F = 45.904 × (1−e−0.076t) r2 = 0.9328 | F = 44.493 × (1 − e−0.247t) r2 = 0.9328 |
| Weibull F = 100×{1 − e[−(t^β) / α]} | F = 100 × {1 − e[− (t^0.294) / 1.873]} r2 = 0.9630 | F = 100 × {1 − e[− (t^0.278) / 0.934]} r2 = 0.9742 | F= 100 × {1 − e[− (t^0.869) / 128.345]} r2 = 0.9912 | F = 100 × {1 − e[− (t^0.410) / 9.318]} r2 = 0.9858 | F = 100 × {1 − e[ −(t^0.248) / 4.333]} r2 = 0.9794 |
| Makoid-Banakar F = k × tn × e−bt | F = 39.238 × t0.251 × e−0.004t r2 = 0.9906 | F = 66.349 × t0.135 × e−0.003t r2 = 0.9870 | F = 0.637 × t0.936 × e−0.003t r2 = 0.9910 | F = 8.811 × t0.455 × e−0.004t r2 = 0.9969 | F = 19.688 × t0.244 × e−0.002t r2 = 0.9844 |
| Peppas-Sahlin F = k1 × t0.5 + k2t | F = 26.104 × t0.5 − 1.821t r2 = 0.8292 | F = 34.669 × t0.5 − 2.603t r2 = 0.3378 | F = 1.237 × t0.5 + 0.223t r2 = 0.9875 | F = 8.968 × t0.5 − 0.404t r2 = 0.9967 | F =12.862 × t0.5 − 0.794t r2 = 0.8805 |
| Korsmeyer-Peppas F = k × tn | F = 45.960 × t0.140 r2 = 0.9294 | F = 72.461 × t0.064 r2 = 0.9460 | F = 1.020 × t0.766 r2 = 0.9895 | F = 11.220 × t0.329 r2 = 0.9771 | F = 21.366 × t0.192 r2 = 0.9762 |
| Parameters | IBU-CL-SF PM | IBU-CL-SF SD |
|---|---|---|
| Cmax (mg/L) | 6.98 ± 0.18 | 20.6 ± 5.4 |
| Tmax (min) | 48 ± 6.7 | 33 ± 12.55 |
| AUC(0–t) (mg/L × min) | 1476.4 ± 411.7 | 3203.7 ± 450.9 |
| MRT(0–t) (min) | 140.5 ± 30.8 | 115.7 ± 23.8 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Han, F.; Zhang, W.; Wang, Y.; Xi, Z.; Chen, L.; Li, S.; Xu, L. Applying Supercritical Fluid Technology to Prepare Ibuprofen Solid Dispersions with Improved Oral Bioavailability. Pharmaceutics 2019, 11, 67. https://doi.org/10.3390/pharmaceutics11020067
Han F, Zhang W, Wang Y, Xi Z, Chen L, Li S, Xu L. Applying Supercritical Fluid Technology to Prepare Ibuprofen Solid Dispersions with Improved Oral Bioavailability. Pharmaceutics. 2019; 11(2):67. https://doi.org/10.3390/pharmaceutics11020067
Chicago/Turabian StyleHan, Fei, Wei Zhang, Ying Wang, Ziyue Xi, Lu Chen, Sanming Li, and Lu Xu. 2019. "Applying Supercritical Fluid Technology to Prepare Ibuprofen Solid Dispersions with Improved Oral Bioavailability" Pharmaceutics 11, no. 2: 67. https://doi.org/10.3390/pharmaceutics11020067
APA StyleHan, F., Zhang, W., Wang, Y., Xi, Z., Chen, L., Li, S., & Xu, L. (2019). Applying Supercritical Fluid Technology to Prepare Ibuprofen Solid Dispersions with Improved Oral Bioavailability. Pharmaceutics, 11(2), 67. https://doi.org/10.3390/pharmaceutics11020067





