Nuclear Trafficking of Retroviral RNAs and Gag Proteins during Late Steps of Replication
Abstract
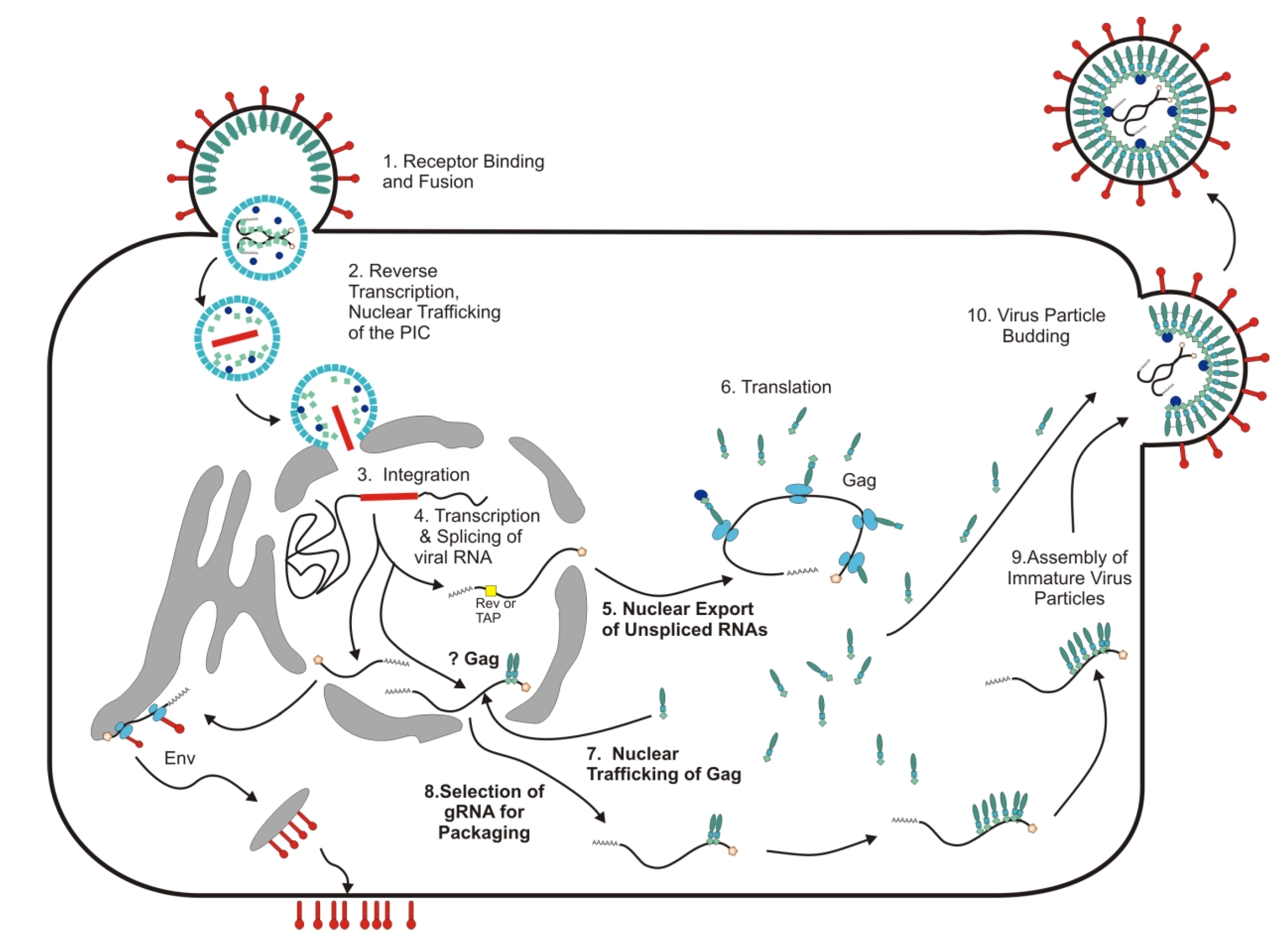
:1. Introduction: Nuclear Trafficking Events in Retrovirus Replication
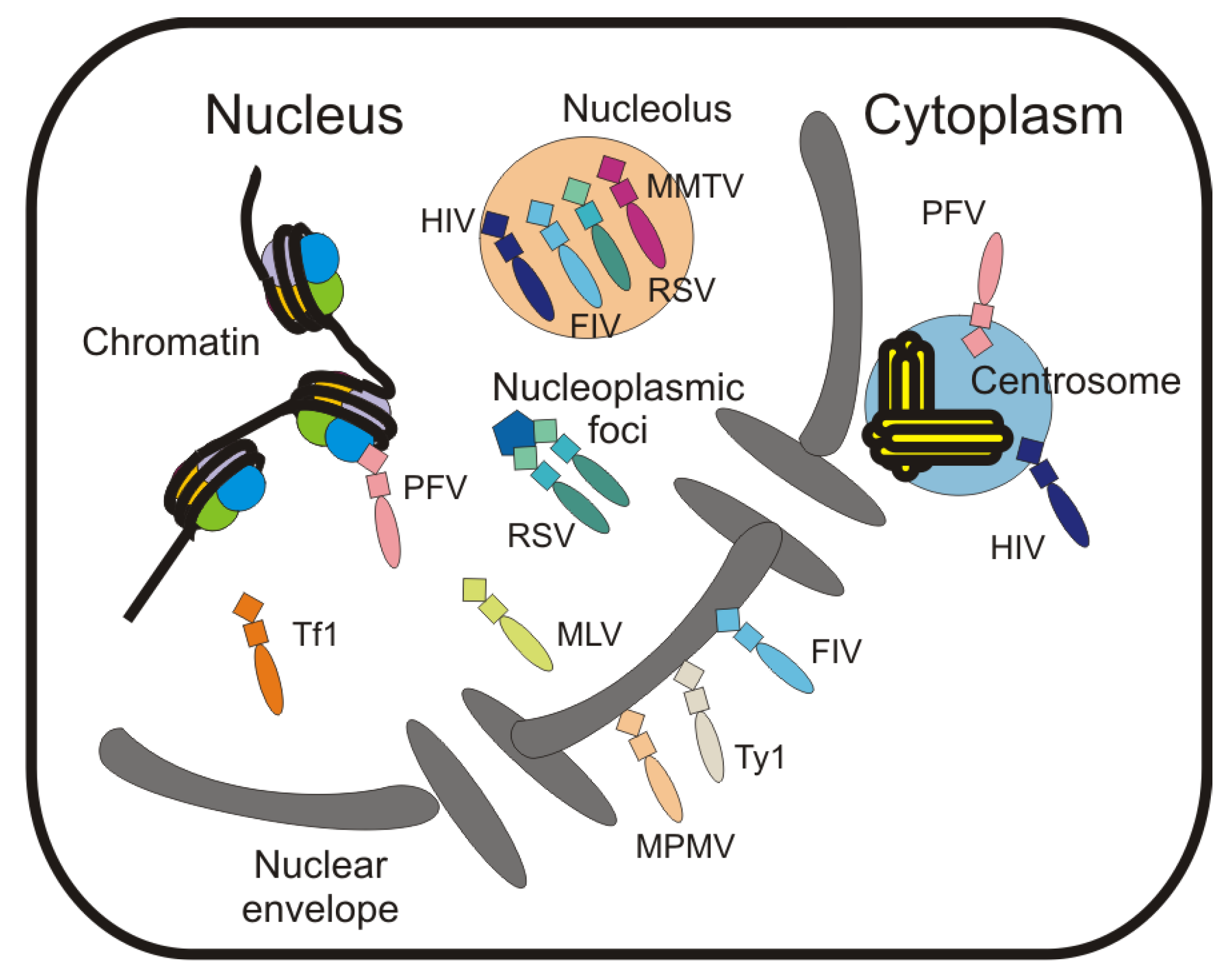
| Retroviral Protein | Localization of the Population Associated with the Nuclear Compartment | Nuclear Localization Mechanism | Nuclear Export Mechanism |
|---|---|---|---|
| Alpharetrovirus | |||
| RSV Gag | Nucleus [
20,23] Punctate Nuclear Foci [23,24] Nucleoli [24] | Imp11 (MA domain) [
25,26] TNPO3 (MA Domain) [26] Importin α/β (NC Domain) [25,26] | CRM1 [ 20,25] |
| RSV NC | Nucleoli [24] | Importin α/β [25,26] | None |
| RSV RT, β subunit | Nucleus [27] | Unknown | None |
| RSV IN | Nucleus [28] | Shares Import Pathway with Histone H1 [29] | None |
| Betaretrovirus | |||
| MMTV Gag | Nucleoli [30] | Unknown [30]; Nucleolar Localization Increased with RPL9 Overexpression [30] | No Identified NES |
| MMTV NC | Nucleoli [24,30] | Unknown; | None |
| MMTV Rem | Nucleoli [31] | Retrotranslocation from Endoplasmic Reticulum to Nucleus [32] | CRM1 [31] |
| MPMV Gag | Nuclear Pore Complex; Low Levels in Nucleus [33,34] | Unknown; Nuclear localization Increased with Ubc9 Overexpression [34] | CRM1 [35] |
| JSRV Rej | Nucleus and Nucleoli [36,37] | Unknown | Rej Function Depends on CRM1 [37] |
| Gammaretrovirus | |||
| MLV Gag | Nucleus [38] | Unknown | No Identified NES |
| MLV NC | Nucleus [24,39] Nucleoli [24,39] | Unknown | None |
| MLV p12 | Mitotic Chromatin [40,41] | Unknown | None |
| MLV IN | Nucleus and Nucleoli [39] | Unknown; Interacts withBrd4 in Nucleus [42] | None |
| Deltaretrovirus | |||
| HTLV Rex | Nucleus and Nucleoli [43,44,45] Punctate Nuclear and Nucleolar Foci [43] | Importin β [46] | CRM1 [47,48] |
| BLV Rex | Punctate Nuclear Foci [49] Nuclear Pore Complex [49] | Unknown | CRM1 [49] |
| Lentivirus | |||
| HIV-1 Gag | Nucleolar [24] | Unknown | Not CRM1 [19,35,50] |
| HIV-1 IN | Nucleus [14,51,52,53,54,55] | Importin α3 [51] Importin α [14,52] Importin 7 [56] | None |
| HIV-1 NC | Nucleoli [24] Nucleus [57,58] | Unknown | None |
| HIV-1 Rev | Nucleus andNnucleoli [44,59] Nuclear Foci [59] HIV-1 Transcription Sites [60] | Importin β [61,62] Transportin, Importin 5, and Importin 7 [63] | CRM1 [47,64] |
| HIV-1 Vpr | Nucleus [65,66] Nuclear Pore Complex [6,66] | Importin α [66] Interacts Directly with Nuclear Pore Complex [67,68] | CRM1 [65] |
| HIV-2 Rev | Nucleoli [69] | Unknown | Unknown |
| HIV-2 Vpx | Nucleoplasm [70,71] | Unknown | No Identified NES |
| FIV Gag | Nucleus [19], Nucleoli [19], and Nuclear Envelope [72] | Unknown | CRM1 [19] |
| FIV Rev | Nucleolus [73] | Unknown | CRM1 [74] |
| EAIV Rev | Nucleus [74] | Unknown | CRM1 [74] |
| BIV Rev | Nucleus and Nucleoli [75,76] | Importin α [77] | CRM1 [77] |
| MVV Rev | Nucleoli [78] | Unknown | Unknown |
| CAEV Rev | Nucleoli [79,80] | Unknown | Unknown |
| Spumaretrovirus | |||
| PFV Gag | Nucleus [21,81] Punctate Nuclear Foci [82,83] Mitotic Chromatin [82] | Binds to H2A/H2B on Mitotic Chromatin [82,83] | CRM1 [21] |
2. Nuclear Export of Unspliced and Incompletely Spliced RNAs of Complex Retroviruses
3. The Role of cis-Acting Sequences in RNA Export of Simple Retroviruses
4. Foamy Viruses Use a Unique Pathway among Retroviruses for Nuclear Export of Viral RNAs
7. Localization of Gag to Subnuclear Sites
8. Remaining Questions about Nuclear Trafficking during Retrovirus Assembly
Acknowledgments
Conflicts of Interest
References
- Jayappa, K.D.; Ao, Z.; Yao, X. The HIV–1 passage from cytoplasm to nucleus: the process involving a complex exchange between the components of HIV–1 and cellular machinery to access nucleus and successful integration. IJBMB 2012, 3, 70–85. [Google Scholar]
- Sherman, M.P.; Greene, W.C. Slipping through the door: HIV entry into the nucleus. Microbes Infect. 2002, 4, 67–73. [Google Scholar] [CrossRef]
- Suzuki, Y.; Craigie, R. The road to chromatin – nuclear entry of retroviruses. Nat. Rev. Microbiol. 2007, 5, 187–96. [Google Scholar] [CrossRef]
- Stetor, S.R.; Rausch, J.W.; Guo, M.J.; Burnham, J.P.; Boone, L.R.; Waring, M.J.; Le Grice, S.F. Characterization of (+) strand initiation and termination sequences located at the center of the equine infectious anemia virus genome. Biochem. 1999, 38, 3656–3667. [Google Scholar] [CrossRef]
- Charneau, P.; Mirambeau, G.; Roux, P.; Paulous, S.; Buc, H.; Clavel, F. HIV–1 reverse transcription. A termination step at the center of the genome. J. Mol. Biol. 1994, 241, 651–662. [Google Scholar] [CrossRef]
- Fouchier, R.A.; Meyer, B.E.; Simon, J.H.; Fischer, U.; Albright, A.V.; Gonzalez–Scarano, F.; Malim, M.H. Interaction of the human immunodeficiency virus type 1 Vpr protein with the nuclear pore complex. J. Virol. 1998, 72, 6004–6013. [Google Scholar]
- Haffar, O.K.; Popov, S.; Dubrovsky, L.; Agostini, I.; Tang, H.; Pushkarsky, T.; Nadler, S.G.; Bukrinsky, M. Two nuclear localization signals in the HIV–1 matrix protein regulate nuclear import of the HIV–1 pre–integration complex. J. Mol. Biol. 2000, 299, 359–368. [Google Scholar] [CrossRef]
- Krishnan, L.; Matreyek, K.A.; Oztop, I.; Lee, K.; Tipper, C.H.; Li, X.; Dar, M.J.; Kewalramani, V.N.; Engelman, A. The requirement for cellular transportin 3 (TNPO3 or TRN–SR2) during infection maps to human immunodeficiency virus type 1 capsid and not integrase. J. Virol. 2010, 84, 397–406. [Google Scholar] [CrossRef]
- Lee, K.; Ambrose, Z.; Martin, T.D.; Oztop, I.; Mulky, A.; Julias, J.G.; Vandegraaff, N.; Baumann, J.G.; Wang, R.; Yuen, W.; Takemura, T.; Shelton, K.; Taniuchi, I.; Li, Y.; Sodroski, J.; Littman, D.R.; Coffin, J.M.; Hughes, S.H.; Unutmaz, D.; Engelman, A.; KewalRamani, V.N. Flexible use of nuclear import pathways by HIV–1. J. Chom. 2010, 7, 221–333. [Google Scholar]
- De Iaco, A.; Luban, J. Inhibition of HIV–1 infection by TNPO3 depletion is determined by capsid and detectable after viral cDNA enters the nucleus. Retrovirol. 2011, 8, 98. [Google Scholar] [CrossRef]
- Yamashita, M.; Perez, O.; Hope, T.J.; Emerman, M. Evidence for direct involvement of the capsid protein in HIV infection of nondividing cells. PLoS Pathog. 2007, 3, 1502–1510. [Google Scholar]
- Yamashita, M.; Emerman, M. Capsid is a dominant determinant of retrovirus infectivity in nondividing cells. J. Virol. 2004, 78, 5670–5678. [Google Scholar] [CrossRef]
- Zennou, V.; Petit, C.; Guetard, D.; Nerhbass, U.; Montagnier, L.; Charneau, P. HIV–1 genome nuclear import is mediated by a central DNA flap. Cell 2000, 101, 173–185. [Google Scholar] [CrossRef]
- Gallay, P.; Hope, T.; Chin, D.; Trono, D. HIV–1 infection of nondividing cells through the recognition of integrase by the importin/karyopherin pathway. Proc. Natl. Acad. Sci. U.S.A. 1997, 94, 9825–9830. [Google Scholar] [CrossRef]
- Schaller, T.; Ocwieja, K.E.; Rasaiyaah, J.; Price, A.J.; Brady, T.L.; Roth, S.L.; Hue, S.; Fletcher, A.J.; Lee, K.; KewalRamani, V.N.; Noursadeghi, M.; Jenner, R.G.; James, L.C.; Bushman, F.D.; Towers, G.J. HIV–1 capsid–cyclophilin interactions determine nuclear import pathway, integration targeting and replication efficiency. PLoS Pathog. 2011, 7, e1002439. [Google Scholar] [CrossRef]
- Butsch, M.; Boris–Lawrie, K. Destiny of unspliced retroviral RNA: ribosome and/or virion? J. Virol. 2002, 76, 3089–3094. [Google Scholar] [CrossRef]
- Schmid, M.; Jensen, T.H. Quality control of mRNP in the nucleus. Chromosoma 2008, 117, 419–429. [Google Scholar] [CrossRef]
- D'Souza, V.; Summers, M.F. How retroviruses select their genomes. Nat. Rev. Microbiol. 2005, 3, 643–655. [Google Scholar] [CrossRef]
- Kemler, I.; Saenz, D.; Poeschla, E. Feline immunodeficiency virus Gag is a nuclear shuttling protein. J. Virol. 2012, 86, 8402–8411. [Google Scholar] [CrossRef]
- Scheifele, L.Z.; Garbitt, R.; Rhoads, J.D.; Parent, L.J. Nuclear entry and CRM1–dependent nuclear export of the Rous sarcoma virus Gag polyprotein. Proc. Natl. Acad. Sci. U.S.A. 2002, 99, 3944–3949. [Google Scholar]
- Renault, N.; Tobaly–Tapiero, J.; Paris, J.; Giron, M.L.; Coiffic, A.; Roingeard, P.; Saib, A. A nuclear export signal within the structural Gag protein is required for prototype foamy virus replication. Retrovirol. 2011, 8, 6. [Google Scholar] [CrossRef]
- Bann, D.V. Intracellular Trafficking of Retroviral Gag and RNA During Late Replication. Doctoral Dissertation, The Pennsylvania State University College of Medicine, Hershey, PA, USA, 2013. [Google Scholar]
- Kenney, S.P.; Lochmann, T.L.; Schmid, C.L.; Parent, L.J. Intermolecular Interactions between Retroviral Gag Proteins in the Nucleus. J. Virol. 2007, 82, 683–691. [Google Scholar]
- Lochmann, T.L.; Bann, D.V.; Ryan, E.P.; Beyer, A.R.; Mao, A.; Cochrane, A.; Parent, L.J. NC–mediated nucleolar localization of retroviral gag proteins. Virus Res. 2013, 171, 304–318. [Google Scholar] [CrossRef]
- Gudleski, N.; Flanagan, J.M.; Ryan, E.P.; Bewly, M.C.; Parent, L.J. Directionality of nucleocytoplasmic transport of the retroviral gag protein depends on sequential binding of karyopherins and viral RNA. Proc. Natl. Acad. Sci. U.S.A. 2010, 107, 9358–9363. [Google Scholar]
- Butterfield–Gerson, K.L.; Scheifele, L.Z.; Ryan, E.P.; Hopper, A.K.; Parent, L.J. Importin–beta Family Members Mediate Alpharetrovirus Gag Nuclear Entry via Interactions with Matrix and Nucleocapsid. J. Virol. 2006, 80, 1798–1806. [Google Scholar] [CrossRef]
- Werner, S.; Hindmarsh, P.; Napirei, M.; Vogel–Bachmayr, K.; Wohrl, B.M. Subcellular localization and integration activities of rous sarcoma virus reverse transcriptase. J. Virol. 2002, 76, 6205–6212. [Google Scholar] [CrossRef]
- Kukolj, G.; Jones, K.S.; Skalka, A.M. Subcellular localization of avian sarcoma virus and human immunodeficiency virus type 1 integrases. J. Virol. 1997, 71, 843–847. [Google Scholar]
- Andrake, M.D.; Sauter, M.M.; Boland, K.; Goldstein, A.D.; Hussein, M.; Skalka, A.M. Nuclear import of Avian Sarcoma Virus integrase is facilitated by host cell factors. Retrovirol. 2008, 5, 73. [Google Scholar] [CrossRef]
- Beyer, A.R.; Bann, D.V.; Rice, B.; Pultz, I.S.; Kane, M.; Goff, S.P.; Golovkina, T.V.; Parent, L.J. Nucleolar trafficking of the mouse mammary tumor virus gag protein induced by interaction with ribosomal protein L9. J. Virol. 2013, 87, 1069–1082. [Google Scholar] [CrossRef]
- Indik, S.; Gunzburg, W.H.; Salmons, B.; Rouault, F. A novel, mouse mammary tumor virus encoded protein with Rev–like properties. Virology 2005, 337, 1–6. [Google Scholar] [CrossRef]
- Byun, H.; Halani, N.; Mertz, J.A.; Ali, A.F.; Lozano, M.M.; Dudley, J.P. Retroviral Rem protein requires processing by signal peptidase and retrotranslocation for nuclear function. Proc. Natl. Acad. Sci. U.S.A. 2010, 107, 12287–12292. [Google Scholar]
- Bohl, C.; Brown, S.; Weldon, R. The pp24 phosphoprotein of Mason–Pfizer monkey virus contributes to viral genome packaging. Retrovirol. 2005, 2, 68. [Google Scholar] [CrossRef]
- Weldon Jr, R.A.; Sarkar, P.; Brown, S.M.; Weldon, S.K. Mason–Pfizer monkey virus Gag proteins interact with the human sumo conjugating enzyme, hUbc9. Virology 2003, 314, 62–73. [Google Scholar] [CrossRef]
- Baluyot, M.F.; Grosse, S.A.; Lyddon, T.D.; Janaka, S.K.; Johnson, M.C. CRM1–dependent trafficking of retroviral Gag proteins revisited. J. Virol. 2012, 86, 4696–4700. [Google Scholar] [CrossRef]
- Hofacre, A.; Nitta, T.; Fan, H. Jaagsiekte sheep retrovirus encodes a regulatory factor, Rej, required for synthesis of Gag protein. J. Virol. 2009, 83, 12483–12498. [Google Scholar] [CrossRef]
- Caporale, M.; Arnaud, F.; Mura, M.; Golder, M.; Murgia, C.; Palmarini, M. The signal peptide of a simple retrovirus envelope functions as a posttranscriptional regulator of viral gene expression. J. Virol. 2009, 83, 4591–4604. [Google Scholar] [CrossRef]
- Nash, M.A.; Meyer, M.K.; Decker, G.L.; Arlinghaus, R.B. A Subset of Pr65gag Is Nucleus Associated in Murine Leukemia Virus–Infected Cells. J. Virol. 1993, 67, 1350–1356. [Google Scholar]
- Risco, C.; Menendez–Arias, L.; Copeland, T.D.; Pinto da Silva, P.; Oroszlan, S. Intracellular transport of the murine leukemia virus during acute infection of NIH 3T3 cells: nuclear import of nucleocapsid protein and integrase. J. Cell Sci. 1995, 108, 3039–3050. [Google Scholar]
- Elis, E.; Ehrlich, M.; Prizan–Ravid, A.; Laham–Karam, N.; Bacharach, E. p12 Tethers the Murine Leukemia Virus Pre–integration Complex to Mitotic Chromosomes. PLoS Pathog. 2012, 8, e1003103. [Google Scholar] [CrossRef]
- Schneider, W.M.; Brzezinski, J.D.; Aiyer, S.; Malani, N.; Gyuricza, M.; Bushman, F.D.; Roth, M.J. Viral DNA tethering domains complement replication–defective mutations in the p12 protein of MuLV Gag. Proc. Natl. Acad. Sci. U.S.A. 2013, 110, 9487–9492. [Google Scholar]
- Sharma, A.; Larue, R.C.; Plumb, M.R.; Malani, N.; Male, F.; Slaughter, A.; Kessl, J.J.; Shkriabai, N.; Coward, E.; Aiyer, S.S.; Green, P.L.; Wu, L.; Roth, M.J.; Bushman, F.D.; Kvaratskhelia, M. BET proteins promote efficient murine leukemia virus integration at transcription start sites. Proc. Natl. Acad. Sci. U.S.A. 2013, 110, 12036–12041. [Google Scholar] [CrossRef]
- Nosaka, T.; Miyazaki, Y.; Takamatsu, T.; Sano, K.; Nakai, M.; Fujita, S.; Martin, T.E.; Hatanaka, M. The Post–transcriptional Regulator Rex of the Human T–Cell Leukemia Virus Type I Is Present as Nucleolar Speckles in Infected Cells. Exp. Cell Res. 1995, 219, 122–129. [Google Scholar] [CrossRef]
- Kubota, S.; Siomi, H.; Satoh, T.; Endo, S.; Maki, M.; Hatanaka, M. Functional similarity of HIV–I rev and HTLV–I rex proteins: identification of a new nucleolar–targeting signal in rev protein. BBRC 1989, 162, 963–970. [Google Scholar]
- Siomi, H.; Shida, H.; Nam, S.H.; Nosaka, T.; Maki, M.; Hatanaka, M. Sequence requirements for nucleolar localization of human T cell leukemia virus type I pX protein, which regulates viral RNA processing. Cell 1988, 55, 197–209. [Google Scholar] [CrossRef]
- Palmeri, D.; Malim, M.H. Importin β Can Mediate the Nuclear Import of an Arginine–Rich Nuclear Localization Signal in the Absence of Importin α. Mol. Cell. Biol. 1999, 19, 1218–1225. [Google Scholar]
- Neville, M.; Stutz, F.; Lee, L.; Davis, L.I.; Rosbash, M. The importin–beta family member Crm1p bridges the interaction between Rev and the nuclear pore complex during nuclear export. Curr. Biol. 1997, 7, 767–775. [Google Scholar]
- Hakata, Y.; Umemoto, T.; Matsushita, S.; Shida, H. Involvement of human CRM1 (exportin 1) in the export and multimerization of the Rex protein of human T–cell leukemia virus type 1. J. Virol. 1998, 72, 6602–6607. [Google Scholar]
- Choi, E.A.; Hope, T.J. Mutational analysis of bovine leukemia virus Rex: Identification of a dominant–negative inhibitor. J. Virol. 2005, 79, 7172–7181. [Google Scholar] [CrossRef]
- Grewe, B.; Hoffmann, B.; Ohs, I.; Blissenbach, M.; Brandt, S.; Tippler, B.; Grunwald, T.; Uberla, K. Cytoplasmic utilization of human immunodeficiency virus type 1 genomic RNA is not dependent on a nuclear interaction with gag. J. Virol. 2012, 86, 2990–3002. [Google Scholar] [CrossRef]
- Ao, Z.; Danappa Jayappa, K.; Wang, B.; Zheng, Y.; Kung, S.; Rassart, E.; Depping, R.; Kohler, M.; Cohen, E.A.; Yao, X. Importin alpha3 interacts with HIV–1 integrase and contributes to HIV–1 nuclear import and replication. J. Virol. 2010, 84, 8650–8663. [Google Scholar] [CrossRef]
- Hearps, A.C.; Jans, D.A. HIV–1 integrase is capable of targeting DNA to the nucleus via an importin alpha/beta–dependent mechanism. Biochem. J. 2006, 398, 475–484. [Google Scholar] [CrossRef]
- Bouyac–Bertoia, M.; Dvorin, J.D.; Fouchier, R.A.; Jenkins, Y.; Meyer, B.E.; Wu, L.I.; Emerman, M.; Malim, M.H. HIV–1 infection requires a functional integrase NLS. Mol. Cell. 2001, 7, 1025–1035. [Google Scholar] [CrossRef]
- Depienne, C.; Mousnier, A.; Leh, H.; Le Rouzic, E.; Dormont, D.; Benichou, S.; Dargemont, C. Characterization of the nuclear import pathway for HIV–1 integrase. J. Biol. Chem. 2001, 276, 18102–18107. [Google Scholar]
- Petit, C.; Schwartz, O.; Mammano, F. The karyophilic properties of human immunodeficiency virus type 1 integrase are not required for nuclear import of proviral DNA. J. Virol. 2000, 74, 7119–7126. [Google Scholar] [CrossRef]
- Ao, Z.; Huang, G.; Yao, H.; Xu, Z.; Labine, M.; Cochrane, A.W.; Yao, X. Interaction of human immunodeficiency virus type 1 integrase with cellular nuclear import receptor importin 7 and its impact on viral replication. J. Biol. Chem. 2007, 282, 13456–13467. [Google Scholar] [CrossRef]
- Zhang, J.; Crumpacker, C.S. Human immunodeficiency virus type 1 nucleocapsid protein nuclear localization mediates early viral mRNA expression. J. Virol. 2002, 76, 10444–10454. [Google Scholar] [CrossRef]
- Gallay, P.; Swingler, S.; Song, J.; Bushman, F.; Trono, D. HIV nuclear import is governed by the phosphotyrosine–mediated binding of matrix to the core domain of integrase. Cell 1995, 83, 569–576. [Google Scholar] [CrossRef]
- Kalland, K.H.; Szilvay, A.M.; Langhoff, E.; Haukenes, G. Subcellular distribution of human immunodeficiency virus type 1 Rev and colocalization of Rev with RNA splicing factors in a speckled pattern in the nucleoplasm. J. Virol. 1994, 68, 1475–1485. [Google Scholar]
- Kula, A.; Gharu, L.; Marcello, A. HIV–1 pre–mRNA commitment to Rev mediated export through PSF and Matrin 3. Virology 2013, 435, 329–340. [Google Scholar] [CrossRef]
- Truant, R.; Cullen, B.R. The arginine–rich domains present in human immunodeficiency virus type 1 Tat and Rev function as direct importin beta–dependent nuclear localization signals. Mol. Cell. Biol. 1999, 19, 1210–1217. [Google Scholar]
- Henderson, B.R.; Percipalle, P. Interactions between HIV Rev and nuclear import and export factors: the Rev nuclear localisation signal mediates specific binding to human importin–beta. J. Mol. Biol. 1997, 274, 693–707. [Google Scholar] [CrossRef]
- Arnold, M.; Nath, A.; Hauber, J.; Kehlenbach, R.H. Multiple importins function as nuclear transport receptors for the Rev protein of human immunodeficiency virus type 1. J. Biol. Chem. 2006, 281, 20883–20890. [Google Scholar] [CrossRef]
- Fornerod, M.; Ohno, M.; Yoshida, M.; Mattaj, I.W. CRM1 is an export receptor for leucine–rich nuclear export signals. Cell 1997, 90, 1051–1060. [Google Scholar] [CrossRef]
- Sherman, M.P.; de Noronha, C.M.; Heusch, M.I.; Greene, S.; Greene, W.C. Nucleocytoplasmic shuttling by human immunodeficiency virus type 1 Vpr. J. Virol. 2001, 75, 1522–1532. [Google Scholar] [CrossRef]
- Vodicka, M.A.; Koepp, D.M.; Silver, P.A.; Emerman, M. HIV–1 Vpr interacts with the nuclear transport pathway to promote macrophage infection. Gene Dev. 1998, 12, 175–185. [Google Scholar] [CrossRef]
- Jenkins, Y.; McEntee, M.; Weis, K.; Greene, W.C. Characterization of HIV–1 vpr nuclear import: analysis of signals and pathways. J. Cell Biol. 1998, 143, 875–885. [Google Scholar] [CrossRef]
- Le Rouzic, E.; Mousnier, A.; Rustum, C.; Stutz, F.; Hallberg, E.; Dargemont, C.; Benichou, S. Docking of HIV–1 Vpr to the nuclear envelope is mediated by the interaction with the nucleoporin hCG1. J. Biol. Chem. 2002, 277, 45091–45098. [Google Scholar]
- Dillon, P.J.; Nelbock, P.; Perkins, A.; Rosen, C.A. Structural and functional analysis of the human immunodeficiency virus type 2 Rev protein. J. Virol. 1991, 65, 445–449. [Google Scholar]
- Belshan, M.; Ratner, L. Identification of the nuclear localization signal of human immunodeficiency virus type 2 Vpx. Virology 2003, 311, 7–15. [Google Scholar] [CrossRef]
- Pancio, H.A.; Vander Heyden, N.; Ratner, L. The C–terminal proline–rich tail of human immunodeficiency virus type 2 Vpx is necessary for nuclear localization of the viral preintegration complex in nondividing cells. J. Virol. 2000, 74, 6162–6167. [Google Scholar] [CrossRef]
- Kemler, I.; Meehan, A.; Poeschla, E.M. Live–Cell Coimaging of the Genomic RNAs and Gag Proteins of Two Lentiviruses. J. Virol. 2010, 84, 6352–6366. [Google Scholar] [CrossRef]
- Phillips, T.R.; Lamont, C.; Konings, D.A.; Shacklett, B.L.; Hamson, C.A.; Luciw, P.A.; Elder, J.H. Identification of the Rev transactivation and Rev–responsive elements of feline immunodeficiency virus. J. Virol. 1992, 66, 5464–5471. [Google Scholar]
- Otero, G.C.; Harris, M.E.; Donello, J.E.; Hope, T.J. Leptomycin B Inhibits Equine Infectious Anemia Virus Rev and Feline Immunodeficiency Virus Rev Function but Not the Function of the Hepatitis B Virus Posttranscriptional Regulatory Element. J. Virol. 1998, 72, 7593–7597. [Google Scholar]
- Oberste, M.S.; Williamson, J.C.; Greenwood, J.D.; Nagashima, K.; Copeland, T.D.; Gonda, M.A. Characterization of Bovine Immunodeficiency Virus Rev Cdnas and Identification and Subcellular–Localization of the Rev Protein. J. Virol. 1993, 67, 6395–6405. [Google Scholar]
- Gomez Corredor, A.; Archambault, D. The bovine immunodeficiency virus Rev protein: identification of a novel nuclear import pathway and nuclear export signal among retroviral Rev/Rev–like proteins. J. Virol. 2012, 86, 4892–4905. [Google Scholar]
- Corredor, A.G.; Archambault, D. The bovine immunodeficiency virus Rev protein: identification of a novel nuclear import pathway and nuclear export signal among retroviral Rev/Rev–like proteins. J. Virol. 2012, 86, 4892–4905. [Google Scholar] [CrossRef]
- Tiley, L.S.; Brown, P.H.; Le, S.Y.; Maizel, J.V.; Clements, J.E.; Cullen, B.R. Visna virus encodes a post–transcriptional regulator of viral structural gene expression. Proc. Natl. Acad. Sci. U.S.A. 1990, 87, 7497–7501. [Google Scholar]
- Schoborg, R.V.; Saltarelli, M.J.; Clements, J.E. A Rev Protein Is Expressed in Caprine Arthritis–Encephalitis Virus (Caev)–Infected Cells and Is Required for Efficient Viral Replication. Virology 1994, 202, 1–15. [Google Scholar] [CrossRef]
- Saltarelli, M.J.; Schoborg, R.; Pavlakis, G.N.; Clements, J.E. Identification of the caprine arthritis encephalitis virus Rev protein and its cis–acting Rev–responsive element. Virology 1994, 199, 47–55. [Google Scholar] [CrossRef]
- Schliephake, A.W.; Rethwilm, A. Nuclear localization of foamy virus Gag precursor protein. J. Virol. 1994, 68, 4946–4954. [Google Scholar]
- Tobaly–Tapiero, J.; Bittoun, P.; Lehmann–Che, J.; Delelis, O.; Giron, M.L.; de The, H.; Saib, A. Chromatin tethering of incoming foamy virus by the structural Gag protein. Traffic 2008, 9, 1717–1727. [Google Scholar] [CrossRef]
- Müllers, E.; Stirnnagel, K.; Kaulfuss, S.; Lindemann, D. Prototype Foamy Virus Gag Nuclear Localization: a Novel Pathway among Retroviruses. J. Virol. 2011, 85, 9276–9285. [Google Scholar]
- Kiyomasu, T.; Miyazawa, T.; Furuya, T.; Shibata, R.; Sakai, H.; Sakuragi, J.; Fukasawa, M.; Maki, N.; Hasegawa, A.; Mikami, T.; et al. Identification of feline immunodeficiency virus rev gene activity. J. Virol. 1991, 65, 4539–4542. [Google Scholar]
- Hirsch, V.M.; Olmsted, R.A.; Murphey–Corb, M.; Purcell, R.H.; Johnson, P.R. An African primate lentivirus (SIVsm) closely related to HIV–2. Nature 1989, 339, 389–392. [Google Scholar] [CrossRef]
- Cheng, S.M.; Blume, M.; Lee, S.G.; Hung, P.P.; Hirsch, V.M.; Johnson, P.R. Coexpression of biologically active simian immunodeficiency virus (SIV) Rev and Env in an SV40 system: the SIV rev gene regulates env expression. Virology 1990, 177, 816–819. [Google Scholar] [CrossRef]
- Gomez Corredor, A.; Archambault, D. The bovine immunodeficiency virus rev protein: identification of a novel lentiviral bipartite nuclear localization signal harboring an atypical spacer sequence. J. Virol. 2009, 83, 12842–12853. [Google Scholar] [CrossRef]
- Stephens, R.M.; Derse, D.; Rice, N.R. Cloning and characterization of cDNAs encoding equine infectious anemia virus tat and putative Rev proteins. J. Virol. 1990, 64, 3716–3725. [Google Scholar]
- Fridell, R.A.; Partin, K.M.; Carpenter, S.; Cullen, B.R. Identification of the activation domain of equine infectious anemia virus rev. J. Virol. 1993, 67, 7317–7323. [Google Scholar]
- Fischer, U.; Meyer, S.; Teufel, M.; Heckel, C.; Luhrmann, R.; Rautmann, G. Evidence that HIV–1 Rev directly promotes the nuclear export of unspliced RNA. EMBO J. 1994, 13, 4105–4112. [Google Scholar]
- Guyader, M.; Emerman, M.; Sonigo, P.; Clavel, F.; Montagnier, L.; Alizon, M. Genome organization and transactivation of the human immunodeficiency virus type 2. Nature 1987, 326, 662–669. [Google Scholar] [CrossRef]
- Malim, M.H.; Hauber, J.; Le, S.Y.; Maizel, J.V.; Cullen, B.R. The HIV–1 rev trans–activator acts through a structured target sequence to activate nuclear export of unspliced viral mRNA. Nature 1989, 338, 254–257. [Google Scholar] [CrossRef]
- Garvey, K.J.; Oberste, M.S.; Elser, J.E.; Braun, M.J.; Gonda, M.A. Nucleotide sequence and genome organization of biologically active proviruses of the bovine immunodeficiency–like virus. Virology 1990, 175, 391–409. [Google Scholar] [CrossRef]
- Mertz, J.A.; Simper, M.S.; Lozano, M.M.; Payne, S.M.; Dudley, J.P. Mouse mammary tumor virus encodes a self–regulatory RNA export protein and is a complex retrovirus. J. Virol. 2005, 79, 14737–14747. [Google Scholar] [CrossRef]
- Hidaka, M.; Inoue, J.; Yoshida, M.; Seiki, M. Post–transcriptional regulator (rex) of HTLV–1 initiates expression of viral structural proteins but suppresses expression of regulatory proteins. EMBO J. 1988, 7, 519–523. [Google Scholar]
- Sagata, N.; Yasunaga, T.; Tsuzuku–Kawamura, J.; Ohishi, K.; Ogawa, Y.; Ikawa, Y. Complete nucleotide sequence of the genome of bovine leukemia virus: its evolutionary relationship to other retroviruses. Proc. Natl. Acad. Sci. U.S.A. 1985, 82, 677–681. [Google Scholar]
- Dultz, E.; Hildenbeutel, M.; Martoglio, B.; Hochman, J.; Dobberstein, B.; Kapp, K. The signal peptide of the mouse mammary tumor virus Rem protein is released from the endoplasmic reticulum membrane and accumulates in nucleoli. J. Biol. Chem. 2008, 283, 9966–9976. [Google Scholar] [CrossRef]
- Nosaka, T.; Siomi, H.; Adachi, Y.; Ishibashi, M.; Kubota, S.; Maki, M.; Hatanaka, M. Nucleolar targeting signal of human T–cell leukemia virus type I rex–encoded protein is essential for cytoplasmic accumulation of unspliced viral mRNA. Proc. Natl. Acad. Sci. U.S.A. 1989, 86, 9798–9802. [Google Scholar] [CrossRef]
- Nitta, T.; Hofacre, A.; Hull, S.; Fan, H. Identification and mutational analysis of a Rej response element in Jaagsiekte sheep retrovirus RNA. J. Virol. 2009, 83, 12499–12511. [Google Scholar] [CrossRef]
- Cullen, B.R.; Hauber, J.; Campbell, K.; Sodroski, J.G.; Haseltine, W.A.; Rosen, C.A. Subcellular localization of the human immunodeficiency virus trans–acting art gene product. J. Virol. 1988, 62, 2498–2501. [Google Scholar]
- Kjems, J.; Brown, M.; Chang, D.D.; Sharp, P.A. Structural analysis of the interaction between the human immunodeficiency virus Rev protein and the Rev response element. Proc. Natl. Acad. Sci. U.S.A. 1991, 88, 683–687. [Google Scholar] [CrossRef]
- Daugherty, M.D.; Booth, D.S.; Jayaraman, B.; Cheng, Y.; Frankel, A.D. HIV Rev response element (RRE) directs assembly of the Rev homooligomer into discrete asymmetric complexes. Proc. Natl. Acad. Sci. U.S.A. 2010, 107, 12481–12486. [Google Scholar]
- Tiley, L.S.; Malim, M.H.; Tewary, H.K.; Stockley, P.G.; Cullen, B.R. Identification of a high–affinity RNA–binding site for the human immunodeficiency virus type 1 Rev protein. Proc. Natl. Acad. Sci. U.S.A. 1992, 89, 758–762. [Google Scholar]
- Cook, K.S.; Fisk, G.J.; Hauber, J.; Usman, N.; Daly, T.J.; Rusche, J.R. Characterization of HIV–1 REV protein: binding stoichiometry and minimal RNA substrate. Nucleic. Acids. Res. 1991, 19, 1577–1583. [Google Scholar] [CrossRef]
- Pollard, V.W.; Malim, M.H. The HIV–1 Rev protein. Annu. Rev. Microbiol. 1998, 52, 491–532. [Google Scholar] [CrossRef]
- Daelemans, D.; Costes, S.V.; Cho, E.H.; Erwin–Cohen, R.A.; Lockett, S.; Pavlakis, G.N. In vivo HIV–1 Rev multimerization in the nucleolus and cytoplasm identified by fluorescence resonance energy transfer. J. Biol. Chem. 2004, 279, 50167–50175. [Google Scholar]
- Michienzi, A.; Cagnon, L.; Bahner, I.; Rossi, J.J. Ribozyme–mediated inhibition of HIV 1 suggests nucleolar trafficking of HIV–1 RNA. Proc. Natl. Acad. Sci. U.S.A. 2000, 97, 8955–8960. [Google Scholar] [CrossRef]
- Buonomo, S.B.C.; Michienzi, A.; De Angelis, F.G.; Bozzoni, I. The Rev protein is able to transport to the cytoplasm small nucleolar RNAs containing a Rev binding element. Rna 1999, 5, 993–1002. [Google Scholar] [CrossRef]
- Fischer, U.; Pollard, V.W.; Luhrmann, R.; Teufel, M.; Michael, M.W.; Dreyfuss, G.; Malim, M.H. Rev–mediated nuclear export of RNA is dominant over nuclear retention and is coupled to the Ran–GTPase cycle. Nucleic. Acids. Res. 1999, 27, 4128–4134. [Google Scholar] [CrossRef]
- Favaro, J.P.; Borg, K.T.; Arrigo, S.J.; Schmidt, M.G. Effect of Rev on the intranuclear localization of HIV–1 unspliced RNA. Virology 1998, 249, 286–296. [Google Scholar] [CrossRef]
- Shopland, L.S.; Johnson, C.V.; Byron, M.; McNeil, J.; Lawrence, J.B. Clustering of multiple specific genes and gene–rich R–bands around SC–35 domains: evidence for local euchromatic neighborhoods. J. Cell. Biol. 2003, 162, 981–990. [Google Scholar] [CrossRef]
- Moen, P.T.; Johnson, C.V.; Byron, M.; Shopland, L.S.; de la Serna, I.L.; Imbalzano, A.N.; Lawrence, J.B. Repositioning of muscle–specific genes relative to the periphery of SC–35 domains during skeletal myogenesis. Mol. Biol. Cell. 2004, 15, 197–206. [Google Scholar]
- Szczerbal, I.; Bridger, J.M. Association of adipogenic genes with SC–35 domains during porcine adipogenesis. Chromosome Res. 2010, 18, 887–895. [Google Scholar] [CrossRef]
- Spector, D.L.; Lamond, A.I. Nuclear Speckles. In Csh Perspect Biol; 2011; Vol. 3. [Google Scholar]
- Sutherland, H.; Bickmore, W.A. Transcription factories: gene expression in unions? Nat. Rev. Genet. 2009, 10, 457–466. [Google Scholar] [CrossRef]
- Listerman, I.; Sapra, A.K.; Neugebauer, K.M. Cotranscriptional coupling of splicing factor recruitment and precursor messenger RNA splicing in mammalian cells. Nat. Struct. Mol. Biol. 2006, 13, 815–822. [Google Scholar] [CrossRef]
- Iacampo, S.; Cochrane, A. Human immunodeficiency virus type 1 Rev function requires continued synthesis of its target mRNA. J. Virol. 1996, 70, 8332–8339. [Google Scholar]
- Naji, S.; Ambrus, G.; Cimermancic, P.; Reyes, J.R.; Johnson, J.R.; Filbrandt, R.; Huber, M.D.; Vesely, P.; Krogan, N.J.; Yates, J.R.; Saphire, A.C.; Gerace, L. Host Cell Interactome of HIV–1 Rev Includes RNA Helicases Involved in Multiple Facets of Virus Production. In Mol Cell Proteomics; 2012; Vol. 11. [Google Scholar]
- Nekhai, S.; Jeang, K.T. Transcriptional and post–transcriptional regulation of HIV–1 gene expression: role of cellular factors for Tat and Rev. Future Microbiol. 2006, 1, 417–426. [Google Scholar] [CrossRef]
- Hadian, K.; Vincendeau, M.; Mausbacher, N.; Nagel, D.; Hauck, S.M.; Ueffing, M.; Loyter, A.; Werner, T.; Wolff, H.; Brack–Werner, R. Identification of a heterogeneous nuclear ribonucleoprotein–recognition region in the HIV Rev protein. J. Biol. Chem. 2009, 284, 33384–33391. [Google Scholar] [CrossRef]
- Ruhl, M.; Himmelspach, M.; Bahr, G.M.; Hammerschmid, F.; Jaksche, H.; Wolff, B.; Aschauer, H.; Farrington, G.K.; Probst, H.; Bevec, D.; et al. Eukaryotic initiation factor 5A is a cellular target of the human immunodeficiency virus type 1 Rev activation domain mediating trans–activation. J. Cell Biol. 1993, 123, 1309–1320. [Google Scholar] [CrossRef]
- Schatz, O.; Oft, M.; Dascher, C.; Schebesta, M.; Rosorius, O.; Jaksche, H.; Dobrovnik, M.; Bevec, D.; Hauber, J. Interaction of the HIV–1 rev cofactor eukaryotic initiation factor 5A with ribosomal protein L5. Proc. Natl. Acad. Sci. U.S.A. 1998, 95, 1607–1612. [Google Scholar] [CrossRef]
- Yasuda–Inoue, M.; Kuroki, M.; Ariumi, Y. Distinct DDX DEAD–box RNA helicases cooperate to modulate the HIV–1 Rev function. Biochem. Biophys. Res. Commun. 2013, 434, 803–808. [Google Scholar] [CrossRef]
- Fang, J.; Kubota, S.; Yang, B.; Zhou, N.; Zhang, H.; Godbout, R.; Pomerantz, R.J. A DEAD box protein facilitates HIV–1 replication as a cellular co–factor of Rev. Virology 2004, 330, 471–480. [Google Scholar] [CrossRef]
- Zhou, X.X.; Luo, J.; Mills, L.; Wu, S.X.; Pan, T.; Geng, G.N.; Zhang, J.; Luo, H.H.; Liu, C.; Zhang, H. DDX5 Facilitates HIV–1 Replication as a Cellular Co–Factor of Rev. Plos One 2013, 8, (5). [Google Scholar]
- Edgcomb, S.P.; Carmel, A.B.; Naji, S.; Ambrus–Aikelin, G.; Reyes, J.R.; Saphire, A.C.; Gerace, L.; Williamson, J.R. DDX1 is an RNA–dependent ATPase involved in HIV–1 Rev function and virus replication. J. Mol. Biol. 2012, 415, 61–74. [Google Scholar] [CrossRef]
- Robertson–Anderson, R.M.; Wang, J.; Edgcomb, S.P.; Carmel, A.B.; Williamson, J.R.; Millar, D.P. Single–molecule studies reveal that DEAD box protein DDX1 promotes oligomerization of HIV–1 Rev on the Rev response element. J. Mol. Biol. 2011, 410, 959–971. [Google Scholar] [CrossRef]
- Yedavalli, V.S.; Neuveut, C.; Chi, Y.H.; Kleiman, L.; Jeang, K.T. Requirement of DDX3 DEAD box RNA helicase for HIV–1 Rev–RRE export function. Cell 2004, 119, 381–392. [Google Scholar] [CrossRef]
- Yedavalli, V.S.R.K.; Jeang, K.T. Matrin 3 is a co–factor for HIV–1 Rev in regulating post–transcriptional viral gene expression. In Retrovirology; 2011; Vol. 8. [Google Scholar]
- Salton, M.; Elkon, R.; Borodina, T.; Davydov, A.; Yaspo, M.L.; Halperin, E.; Shiloh, Y. Matrin 3 Binds and Stabilizes mRNA. In Plos One; 2011; Vol. 6. [Google Scholar]
- Butin–Israeli, V.; Adam, S.A.; Goldman, A.E.; Goldman, R.D. Nuclear lamin functions and disease. Trends Genet. 2012, 28, 464–471. [Google Scholar] [CrossRef]
- Ciejek, E.M.; Tsai, M.J.; Omalley, B.W. Actively Transcribed Genes Are Associated with the Nuclear Matrix. Nature 1983, 306, 607–609. [Google Scholar] [CrossRef]
- Malecki, M.; Malecki, B. Nuclear routing networks span between nuclear pore complexes and genomic DNA to guide nucleoplasmic trafficking of biomolecules. In J Fertili In Vitro, 2013/01/01 ed.; 2012; Vol. 2. [Google Scholar]
- Malecki, M.; Malecki, B. Routing of Biomolecules and Transgenes' Vectors in Nuclei of Oocytes. J. Fertili. In Vitro 2012, 2, 108–118. [Google Scholar]
- Zeitz, M.J.; Malyavantham, K.S.; Seifert, B.; Berezney, R. Matrin 3: chromosomal distribution and protein interactions. J. Cell. Biochem. 2009, 108, 125–33. [Google Scholar] [CrossRef]
- Malyavantham, K.S.; Bhattacharya, S.; Barbeitos, M.; Mukherjee, L.; Xu, J.; Fackelmayer, F.O.; Berezney, R. Identifying functional neighborhoods within the cell nucleus: proximity analysis of early S–phase replicating chromatin domains to sites of transcription, RNA polymerase II, HP1gamma, matrin 3 and SAF–A. J. Cell. Biochem. 2008, 105, 391–403. [Google Scholar] [CrossRef]
- Bray, M.; Prasad, S.; Dubay, J.W.; Hunter, E.; Jeang, K.–T.; Rekosh, D.; Hammarskjöld, M.L. A small element from the Mason–Pfizer monkey virus genome makes human immunodeficiency virus type 1 expression and replication Rev–independent. Proc. Natl. Acad. Sci. U.S.A. 1994, 91, 1256–1260. [Google Scholar]
- Ernst, R.K.; Bray, M.; Rekosh, D.; Hammarskjold, M.L. A structured retroviral RNA element that mediates nucleocytoplasmic export of intron–containing RNA. Mol. Cell. Biol. 1997, 17, 135–144. [Google Scholar]
- Grüter, P.; Tabernero, C.; von Kobbe, C.; Schmitt, C.; Saavedra, C.; Bachi, A.; Wilm, M.; Felber, B.K.; Izaurralde, E. TAP, the Human Homolog of Mex67p, Mediates CTE–Dependent RNA Export from the Nucleus. Mol. Cell. 1998, 1, 649–659. [Google Scholar] [CrossRef]
- Katahira, J.; Strasser, K.; Podtelejnikov, A.; Mann, M.; Jung, J.U.; Hurt, E. The Mex67p–mediated nuclear mRNA export pathway is conserved from yeast to human. EMBO J. 1999, 18, 2593–2609. [Google Scholar] [CrossRef]
- Saavedra, C.; Felber, B.; Izaurralde, E. The simian retrovirus–1 constitutive transport element, unlike the HIV–1 RRE, uses factors required for cellular mRNA export. Curr. Biol. 1997, 7, 619–628. [Google Scholar] [CrossRef]
- Pasquinelli, A.E.; Ernst, R.K.; Lund, E.; Grimm, C.; Zapp, M.L.; Rekosh, D.; Hammarskjold, M.L.; Dahlberg, J.E. The constitutive transport element (CTE) of Mason–Pfizer monkey virus (MPMV) accesses a cellular mRNA export pathway. EMBO J. 1997, 16, 7500–7510. [Google Scholar] [CrossRef]
- Sergef, A.; Sharma, K.; Doye, V.; Hellwig, A.; Huber, J.; Lührmann, R.; Hurt, E. Mex67p, a novel factor for nuclear mRNA export, binds to both poly(A)+ RNA and nuclear pores. EMBO J. 1997, 16, 3256–3271. [Google Scholar] [CrossRef]
- Kang, Y.; Cullen, B.R. The human Tap protein is a nuclear mRNA export factor that contains novel RNA–binding and nucleocytoplasmic transport sequences. Genes Dev. 1999, 13, 1126–1139. [Google Scholar] [CrossRef]
- Stutz, F.; Bachi, A.; Doerks, T.; Braun, I.C.; Séraphin, B.; Wilm, M.; Bork, P.; Izaurralde, E. REF, an evolutionarily conserved family of hnRNP–like proteins, interacts with TAP/Mex67p and participates in mRNA nuclear export. Rna 2000, 6, 638–650. [Google Scholar] [CrossRef]
- Teplova, M.; Wohlbold, L.; Khin, N.W.; Izaurralde, E.; Patel, D.J. Structure–function studies of nucleocytoplasmic transport of retroviral genomic RNA by mRNA export factor TAP. Nature 2011, 18, 990–998. [Google Scholar]
- Schwartz, D.E.; Tizard, R.; Gilbert, W. Nucleotide Sequence of Rous Sarcoma Virus. Cell 1983, 32, 853–869. [Google Scholar] [CrossRef]
- Ogert, R.A.; Lee, L.H.; Beemon, K. Avian Retroviral RNA Element Promotes Unspliced RNA Accumulation in the Cytoplasm. J. Virol. 1996, 70, 3834–3843. [Google Scholar]
- Omer, C.A.; Pogue–Geile, K.; Guntaka, R.; Staskus, K.A.; Faras, A.J. Involvement of Directly Repeated Sequences in the Generation of Deletions of the Avian Sarcoma Virus src Gene. J. Virol. 1983, 47, 380–382. [Google Scholar]
- Yamamoto, T.; Tyagi, J.S.; Fagan, J.B.; Jay, G.; de Crombrugghe, B.; Pastan, I. Molecular Mechanism for the Capture and Excision of the Transforming Gene of Avian Sarcoma Virus as Suggested by Analysis of Recombinant Clones. J. Virol. 1980, 35, 436–443. [Google Scholar]
- Aschoff, J.M.; Foster, D.; Coffin, J.M. Point mutations in the avian sarcoma/leukosis virus 3' untranslated region result in a packaging defect. J. Virol. 1999, 73, 7421–7429. [Google Scholar]
- Simpson, S.B.; Guo, W.; Winistorfer, S.C.; Craven, R.C.; Stoltzfus, C.M. The Upstream Direct Repeat Sequence of Prague A Rous Sarcoma Virus is Deficient in Mediating Efficiient Gag Assembly and Particle Release. Virology 1998, 247, 86–96. [Google Scholar] [CrossRef]
- Simpson, S.B.; Zhang, L.; Craven, R.C.; Stoltzfus, C.M. Rous sarcoma virus direct repeat cis elements exert effects at several points in the virus life cycle. J. Virol. 1997, 71, 9150–9156. [Google Scholar]
- Paca, R.E.; Ogert, R.A.; Hibbert, C.S.; Izaurralde, E.; Beemon, K. Rous Sarcoma Virus DR Postranscriptional Elements Use a Novel RNA Export Pathway. J. Virol. 2000, 74, 9507–9514. [Google Scholar] [CrossRef]
- LeBlanc, J.J.; Uddowla, S.; Abraham, B.; Clatterbuck, S.; Beemon, K.L. Tap and Dbp5, but not Gag, are involved in DR–mediated nuclear export of unspliced Rous sarcoma virus RNA. Virology 2007, 363, 376–386. [Google Scholar] [CrossRef]
- Garbitt–Hirst, R.; Kenney, S.P.; Parent, L.J. Genetic Evidence for a Connection between Rous Sarcoma Virus Gag Nuclear Trafficking and Genomic RNA Packaging. J. Virol. 2009, 83, 6790–6797. [Google Scholar] [CrossRef]
- Mullers, E. The foamy virus Gag proteins: what makes them different? Viruses 2013, 5, 1023–1041. [Google Scholar] [CrossRef]
- Linial, M.L. Foamy Viruses Are Unconventional Retroviruses. J. Virol. 1999, 73, 1747–1755. [Google Scholar]
- Enssle, J.; Jordan, I.; Mauer, B.; Rethwilm, A. Foamy virus reverse transcriptase is expressed independently from the Gag protein. Proc. Natl. Acad. Sci. U.S.A. 1996, 93, 4137–4141. [Google Scholar] [CrossRef]
- Lochelt, M.; Flugel, R.M. The human foamy virus pol gene is expressed as a Pro–Pol polyprotein and not as a gag–pol fusion protein. J. Virol. 1996, 70, 1033–1040. [Google Scholar]
- Yu, S.F.; Edelmann, K.; Strong, R.K.; Moebes, A.; Rethwilm, A.; Linial, M.L. The carboxyl terminus of the human foamy virus Gag protein contains separable nucleic acid binding and nuclear transport domains. J. Virol. 1996, 70, 8255–8262. [Google Scholar]
- Jordan, I.; Enssle, J.; Güttler, E.; Mauer, B.; Rethwilm, A. Expression of Human Foamy Virus Reverse Transcriptase Involves a Spliced pol mRNA. Virology 1996, 224, 314–319. [Google Scholar] [CrossRef]
- Keller, A.; Partin, K.M.; Löchelt, M.; Bannert, H.; Flügel, R.M.; Cullen, B.R. Characterization of the Transcriptional trans Activator of Human Foamy Retrovirus. J. Virol. 1991, 65, 2589–2594. [Google Scholar]
- Baunach, G.; Maurer, B.; Hahn, H.; Kranz, M.; Rethwilm, A. Functional Analysis of Human Foamy Virus Accessory Reading Frames. J. Virol. 1993, 67, 5411–5418. [Google Scholar]
- Bodem, J.; Schied, T.; Gabriel, R.; Rammling, M.; Rethwilm, A. Foamy virus nuclear RNA export is distinct from that of other retroviruses. J. Virol. 2011, 85, 2333–2341. [Google Scholar]
- Gallouzi, I.E.; Steitz, J.A. Delineation of mRNA export pathways by the use of cell–permeable peptides. Science 2001, 294, 1895–1901. [Google Scholar] [CrossRef]
- Brennan, C.M.; Gallouzi, I.–E.; Steitz, J.A. Protein Ligands to HuR Modulate Its Interaction with target mRNAs In Vivo. J. Cell Biol. 2000, 151, 1–13. [Google Scholar]
- Yu, S.F.; Lujan, P.; Jackson, D.L.; Emerman, M.; Linial, M.L. The DEAD–box RNA helicase DDX6 is required for efficient encapsidation of a retroviral genome. PLoS Pathog 2011, 7, e1002303. [Google Scholar] [CrossRef]
- Erlwein, O.; Bieniasz, P.D.; McClure, M.O. Sequences in pol are required for transfer of human foamy virus–based vectors. J. Virol. 1998, 72, 5510–5516. [Google Scholar]
- Scheifele, L.Z.; Ryan, E.P.; Parent, L.J. Detailed Mapping of the Nuclear Export Signal in the Rous Sarcoma Virus Gag Protein. J. Virol. 2005, 79, 8732–8741. [Google Scholar] [CrossRef]
- Brun, S.; Solignat, M.; Gay, B.; Bernard, E.; Chaloin, L.; Fenard, D.; Devaux, C.; Chazal, N.; Briant, L. VSV–G pseudotyping rescues HIV–1 CA mutations that impair core assembly or stability. Retrovirol. 2008, 5, 57. [Google Scholar] [CrossRef]
- Thys, W.; De Houwer, S.; Demeulemeester, J.; Taltynov, O.; Vancraenenbroeck, R.; Gerard, M.; De Rijck, J.; Gijsbers, R.; Christ, F.; Debyser, Z. Interplay between HIV entry and transportin–SR2 dependency. Retrovirol. 2011, 8, 7. [Google Scholar] [CrossRef]
- Garbitt, R.A.; Bone, K.R.; Parent, L.J. Insertion of a Classical Nuclear Import Signal into the Matrix Domain of the Rous Sarcoma Virus Gag Protein Interferes with Virus Replication. J. Virol. 2004, 78, 13534–13542. [Google Scholar] [CrossRef]
- Kataoka, N.; Bachorik, J.L.; Dreyfuss, G. Transportin–SR, a Nuclear Import Receptor for SR Proteins. J. Cell. Biol. 1999, 145, 1145–1152. [Google Scholar] [CrossRef]
- Lai, M.C.; Lin, R.I.; Tarn, W.Y. Transportin–SR2 mediates nuclear import of phosphorylated SR proteins. Proc. Natl. Acad. Sci. U.S.A. 2001, 98, 10154–10159. [Google Scholar] [CrossRef]
- Lai, M.C.; Lin, R.I.; Huang, S.Y.; Tsai, C.W.; Tarn, W.Y. A human importin–beta family protein, transportin–SR2, interacts with the phosphorylated RS domain of SR proteins. J. Biol. Chem. 2000, 275, 7950–7957. [Google Scholar]
- Flynn, J.A.; An, W.; King, S.R.; Telesnitsky, A. Nonrandom dimerization of murine leukemia virus genomic RNAs. J. Virol. 2004, 78, 12129–12139. [Google Scholar] [CrossRef]
- Flynn, J.A.; Telesnitsky, A. Two distinct Moloney murine leukemia virus RNAs produced from a single locus dimerize at random. Virology 2006, 344, 391–400. [Google Scholar] [CrossRef]
- Kharytonchyk, S.A.; Kireyeva, A.I.; Osipovich, A.B.; Fomin, I.K. Evidence for preferential copackaging of Moloney murine leukemia virus genomic RNAs transcribed in the same chromosomal site. Retrovirol. 2005, 2, 3. [Google Scholar] [CrossRef]
- Rasmussen, S.V.; Pedersen, F.S. Co–localization of gammaretroviral RNAs at their transcription site favours co–packaging. J. Gen. Virol. 2006, 87, 2279–2289. [Google Scholar] [CrossRef]
- Maurel, S.; Mougel, M. Murine leukemia virus RNA dimerization is coupled to transcription and splicing processes. Retrovirol. 2010, 7, 64. [Google Scholar] [CrossRef]
- Levin, J.G.; Rosenak, M. J. Synthesis of murine leukemia virus proteins associated with virions assembled in actinomycin D–treated cells: evidence for persistence of viral messenger RNA. Proc. Natl. Acad. Sci. U.S.A. 1976, 73, 1154–1158. [Google Scholar] [CrossRef]
- Levin, J.G.; Grimley, P.M.; Ramseur, J.M.; Berezesky, I.K. Deficiency of 60 to 70S RNA in murine leukemia virus particles assembled in cells treated with actinomycin D. J. Virol. 1974, 14, 152–161. [Google Scholar]
- Dorman, N.; Lever, A. Comparison of viral genomic RNA sorting mechanisms in human immunodeficiency virus type 1 (HIV–1), HIV–2, and Moloney murine leukemia virus. J. Virol. 2000, 74, 11413–11417. [Google Scholar] [CrossRef]
- Messer, L.I.; Levin, J.G.; Chattopadhyay, S.K. Metabolism of viral RNA in murine leukemia virus–infected cells; evidence for differential stability of viral message and virion precursor RNA. J. Virol. 1981, 40, 683–690. [Google Scholar]
- D'Souza, V.; Summers, M.F. Structural basis for packaging the dimeric genome of Moloney murine leukaemia virus. Nature 2004, 431, 586–590. [Google Scholar] [CrossRef]
- Garcia, E.L.; Onafuwa–Nuga, A.; Sim, S.; King, S.R.; Wolin, S.L.; Telesnitsky, A. Packaging of host mY RNAs by murine leukemia virus may occur early in Y RNA biogenesis. J. Virol. 2009, 83, 12526–12534. [Google Scholar] [CrossRef]
- Poole, E.; Strappe, P.; Mok, H.P.; Hicks, R.; Lever, A.M. HIV–1 Gag–RNA interaction occurs at a perinuclear/centrosomal site; analysis by confocal microscopy and FRET. Traffic 2005, 6, 741–755. [Google Scholar] [CrossRef]
- Yu, S.F.; Eastman, S.W.; Linial, M.L. Foamy virus capsid assembly occurs at a pericentriolar region through a cytoplasmic targeting/retention signal in Gag. Traffic 2006, 7, 966–977. [Google Scholar] [CrossRef]
- Molle, D.; Segura–Morales, C.; Camus, G.; Berlioz–Torrent, C.; Kjems, J.; Basyuk, E.; Bertrand, E. Endosomal trafficking of HIV–1 gag and genomic RNAs regulates viral egress. J. Biol. Chem. 2009, 284, 19727–19743. [Google Scholar]
- Basyuk, E.; Galli, T.; Mougel, M.; Blanchard, J.M.; Sitbon, M.; Bertrand, E. Retroviral genomic RNAs are transported to the plasma membrane by endosomal vesicles. Dev. Cell. 2003, 5, 161–174. [Google Scholar] [CrossRef]
- Checkley, M.A.; Mitchell, J.A.; Eizenstat, L.D.; Lockett, S.J.; Garfinkel, D.J. Ty1 gag enhances the stability and nuclear export of Ty1 mRNA. Traffic 2013, 14, 57–69. [Google Scholar]
- Jouvenet, N.; Bieniasz, P.D.; Simon, S.M. Imaging the biogenesis of individual HIV–1 virions in live cells. Nature 2008, 454, 236–240. [Google Scholar] [CrossRef]
- Jouvenet, N.; Simon, S.M.; Bieniasz, P.D. Imaging the interaction of HIV–1 genomes and Gag during assembly of individual viral particles. Proc. Natl. Acad. Sci. U.S.A. 2009, 106, 19114–19119. [Google Scholar] [CrossRef]
- Jouvenet, N.; Simon, S.M.; Bieniasz, P.D. Visualizing HIV–1 assembly. J. Mol. Biol. 2011, 410, 501–511. [Google Scholar] [CrossRef]
- Kutluay, S.B.; Bieniasz, P.D. Analysis of the initiating events in HIV–1 particle assembly and genome packaging. PLoS Pathog. 2010, 6, e1001200. [Google Scholar] [CrossRef]
- Levesque, K.; Halvorsen, M.; Abrahamyan, L.; Chatel–Chaix, L.; Poupon, V.; Gordon, H.; DesGroseillers, L.; Gatignol, A.; Mouland, A.J. Trafficking of HIV–1 RNA is mediated by heterogeneous nuclear ribonucleoprotein A2 expression and impacts on viral assembly. Traffic 2006, 7, 1177–1193. [Google Scholar] [CrossRef]
- Lund, N.; Milev, M.P.; Wong, R.; Sanmuganantham, T.; Woolaway, K.; Chabot, B.; Abou Elela, S.; Mouland, A.J.; Cochrane, A. Differential effects of hnRNP D/AUF1 isoforms on HIV–1 gene expression. Nucleic. Acids. Res. 2012, 40, 3663–3675. [Google Scholar] [CrossRef]
- Stoltzfus, C.M.; Madsen, J.M. Role of viral splicing elements and cellular RNA binding proteins in regulation of HIV–1 alternative RNA splicing. Curr. HIV Res. 2006, 4, 43–55. [Google Scholar] [CrossRef]
- Lehmann, M.; Milev, M.P.; Abrahamyan, L.; Yao, X.J.; Pante, N.; Mouland, A.J. Intracellular transport of human immunodeficiency virus type 1 genomic RNA and viral production are dependent on dynein motor function and late endosome positioning. J. Biol. Chem. 2009, 284, 14572–14585. [Google Scholar] [CrossRef]
- Malagon, F.; Jensen, T.H. The T body, a new cytoplasmic RNA granule in Saccharomyces cerevisiae. Mol. Cell. Biol. 2008, 28, 6022–6032. [Google Scholar] [CrossRef]
- Checkley, M.A.; Nagashima, K.; Lockett, S.J.; Nyswaner, K.M.; Garfinkel, D.J. P–body components are required for Ty1 retrotransposition during assembly of retrotransposition–competent virus–like particles. Mol. Cell. Biol. 2010, 30, 382–398. [Google Scholar] [CrossRef]
- Dutko, J.A.; Kenny, A.E.; Gamache, E.R.; Curcio, M.J. 5' to 3' mRNA decay factors colocalize with Ty1 gag and human APOBEC3G and promote Ty1 retrotransposition. J. Virol. 2010, 84, 5052–5066. [Google Scholar] [CrossRef]
- Malagon, F.; Jensen, T.H. T–body formation precedes virus–like particle maturation in S. cerevisiae. RNA Biol. 2011, 8, 184–189. [Google Scholar] [CrossRef]
- Larsen, L.S.; Beliakova–Bethell, N.; Bilanchone, V.; Zhang, M.; Lamsa, A.; Dasilva, R.; Hatfield, G.W.; Nagashima, K.; Sandmeyer, S. Ty3 nucleocapsid controls localization of particle assembly. J. Virol. 2008, 82, 2501–2514. [Google Scholar] [CrossRef]
- Dang, V.D.; Levin, H.L. Nuclear import of the retrotransposon Tf1 is governed by a nuclear localization signal that possesses a unique requirement for the FXFG nuclear pore factor Nup124p. Mol. Cell. Biol. 2000, 20, 7798–7812. [Google Scholar] [CrossRef]
- Hiscox, J.A. The nucleolus – a gateway to viral infection? Arch. Virol. 2002, 147, 1077–1089. [Google Scholar] [CrossRef]
- Politz, J.C.; Yarovoi, S.; Kilroy, S.M.; Gowda, K.; Zwieb, C.; Pederson, T. Signal recognition particle components in the nucleolus. Proc. Natl. Acad. Sci. U.S.A. 2000, 97, 55–60. [Google Scholar] [CrossRef]
- Jacobson, M.R.; Pederson, T. Localization of signal recognition particle RNA in the nucleolus of mammalian cells. Proc. Natl. Acad. Sci. U.S.A. 1998, 95, 7981–7986. [Google Scholar] [CrossRef]
- Keene, S.E.; King, S.R.; Telesnitsky, A. 7SL RNA is retained in HIV–1 minimal virus–like particles as an S–domain fragment. J. Virol. 2010, 84, 9070–9077. [Google Scholar]
- Keene, S.E.; Telesnitsky, A. cis–Acting determinants of 7SL RNA packaging by HIV–1. J. Virol. 2012, 86, 7934–7942. [Google Scholar] [CrossRef]
- Onafuwa–Nuga, A.A.; Telesnitsky, A.; King, S.R. 7SL RNA, but not the 54–kd signal recognition particle protein, is an abundant component of both infectious HIV–1 and minimal virus–like particles. Rna 2006, 12, 542–546. [Google Scholar] [CrossRef]
- Rulli, S.J., Jr.; Hibbert, C.S.; Mirro, J.; Pederson, T.; Biswal, S.; Rein, A. Selective and nonselective packaging of cellular RNAs in retrovirus particles. J. Virol. 2007, 81, 6623–6631. [Google Scholar] [CrossRef]
- Giles, K.E.; Caputi, M.; Beemon, K.L. Packaging and reverse transcription of snRNAs by retroviruses may generate pseudogenes. Rna 2004, 10, 299–307. [Google Scholar] [CrossRef]
- Sawyer, R.C.; Hanafusa, H. Comparison of the Small RNAs of Polymerase–Deficient and Polymerase–Positive Rous Sarcoma Virus and Another Species of Avian Retrovirus. J. Virol. 1979, 29, 863–871. [Google Scholar]
- Faras, A.J.; Garapin, A.C.; Levinson, W.E.; Bishop, J.M.; Goodman, H.M. Characterization of low–molecular–weight RNAs associated with 70s RNA of Rous sarcoma virus. J. Virol. 1973, 12, 334–342. [Google Scholar]
- Buzdin, A.; Gogvadze, E.; Lebrun, M.H. Chimeric retrogenes suggest a role for the nucleolus in LINE amplification. FEBS Lett 2007, 581, 2877–2882. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Stake, M.S.; Bann, D.V.; Kaddis, R.J.; Parent, L.J. Nuclear Trafficking of Retroviral RNAs and Gag Proteins during Late Steps of Replication. Viruses 2013, 5, 2767-2795. https://doi.org/10.3390/v5112767
Stake MS, Bann DV, Kaddis RJ, Parent LJ. Nuclear Trafficking of Retroviral RNAs and Gag Proteins during Late Steps of Replication. Viruses. 2013; 5(11):2767-2795. https://doi.org/10.3390/v5112767
Chicago/Turabian StyleStake, Matthew S., Darrin V. Bann, Rebecca J. Kaddis, and Leslie J. Parent. 2013. "Nuclear Trafficking of Retroviral RNAs and Gag Proteins during Late Steps of Replication" Viruses 5, no. 11: 2767-2795. https://doi.org/10.3390/v5112767
APA StyleStake, M. S., Bann, D. V., Kaddis, R. J., & Parent, L. J. (2013). Nuclear Trafficking of Retroviral RNAs and Gag Proteins during Late Steps of Replication. Viruses, 5(11), 2767-2795. https://doi.org/10.3390/v5112767