Abstract
Arenaviruses have a bisegmented negative-strand RNA genome, which encodes four viral proteins: GP and NP by the S segment and L and Z by the L segment. These four viral proteins possess multiple functions in infection, replication and release of progeny viruses from infected cells. The small RING finger protein, Z protein is a matrix protein that plays a central role in viral assembly and budding. Although all arenaviruses encode Z protein, amino acid sequence alignment showed a huge variety among the species, especially at the C-terminus where the L-domain is located. Recent publications have demonstrated the interactions between viral protein and viral protein, and viral protein and host cellular protein, which facilitate transportation and assembly of viral components to sites of virus egress. This review presents a summary of current knowledge regarding arenavirus assembly and budding, in comparison with other enveloped viruses. We also refer to the restriction of arenavirus production by the antiviral cellular factor, Tetherin/BST-2.
1. Introduction
Arenaviruses are divided into two groups, Old World (OW) and New World (NW) arenaviruses, based on geographical, serological, and phylogenetic differences (Figure 1).
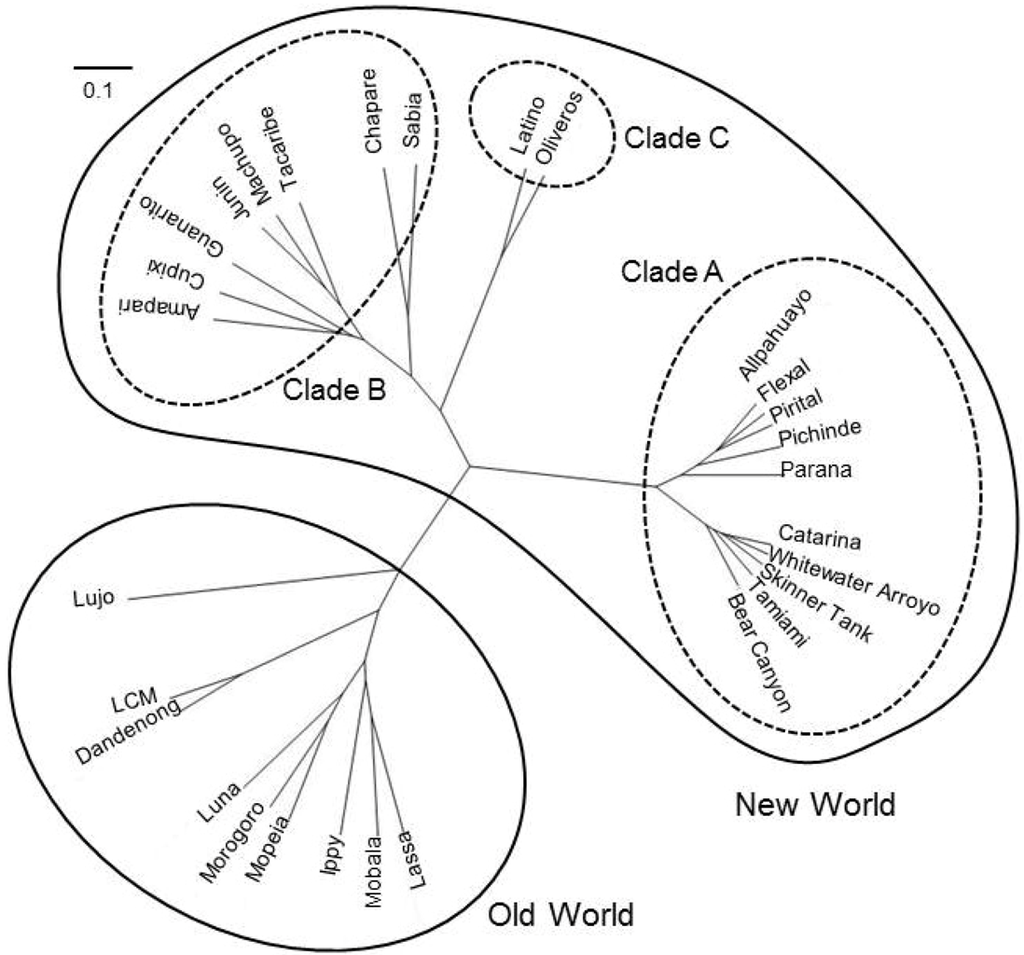
Figure 1.
Phylogenetic tree of arenavirus Z protein. Amino acid sequences of Z protein were used for analysis. The phylogenetic tree was drawn using GENETYX [1]. The scale bar indicates substitutions per site. Accession numbers for reference sequences are: ABY20731 (Dandenong), YP_516232 (Ippy), NP_694871.1 (LASV, Josiah), ABC96003 (LCMV, Armstrong), YP_002929492 (Lujo virus), YP_516228 (Mobala virus), ABC71136 (Mopeia virus), YP_003090216 (Morogoro virus), YP_004933732 (Luna virus), YP_001649213 (Allpahuayo virus), AEQ59327 (Bear Canyon virus), AEQ59329 (Catarina virus), YP_001936023 (Flexal virus), YP_001936027 (Parana virus), YP_138535 (Pichinde virus), YP_025092 (Pirital virus), AEQ59336 (Skinner Tank virus), YP_001911119 (Whitewater arroyo virus), YP_001649217 (Amapari virus), YP_001816784 (Chapare virus), YP_001649219 (Cupixi virus), NP_899220 (Guanarito virus), NP_899216 (Junin virus, XJ-13), NP_899215 (Machupo virus), YP_089659 (Sabia virus), Q88470 (Tacaribe virus), YP_001911117 (Tamiami virus), YP_001936025 (Latino virus), YP_001649215 (Oliveros virus).
Several arenaviruses cause hemorrhagic fever (HF) disease in humans. Lassa virus (LASV), Junin virus (JUNV), Machupo virus (MACV), Guanarito virus (GTOV), and Sabia virus (SABV) are the causative agents of Lassa fever (LF), Argentine HF, Bolivian HF, Venezuelan HF and Brazilian HF diseases, respectively [2]. In addition to these arenaviruses, Chapare virus and Lujo virus (LUJV) have also been reported to cause HF [3,4,5]. The prototypic arenavirus, Lymphocytic choriomeningitis virus (LCMV) has a world-wide distribution and is considered as a neglected human pathogen of clinical significance in congenital viral infections [6]. Moreover, LCMV infection of immunosuppressed individuals can result in severe disease and death [7,8]. These concerns are compounded by the lack of FDA licensed vaccines and limited existing therapeutic options. The live attenuated strain of JUNV, Candid #1, is the only arenavirus vaccine tested in humans, and has been licensed only in Argentina but is ineffective against LASV and LCMV. On the other hand, current arenavirus antiviral drug therapy is restricted to the use of the nucleoside analog ribavirin (Rib), which is only partially effective and is associated with significant side effects, including hemolytic anemia [9]. Therefore, there is now a pressing need for the development of effective anti-arenavirus therapeutic strategies. Several inhibitors of arenavirus entry have been reported [10,11,12,13,14,15,16,17]. The viral entry step is currently considered one of the most promising anti‑arenaviral targets. The success of Oseltamivir and Zanamivir, which are potent inhibitors of viral neuraminidase and prevent the release and spread of progeny virions, for the treatment of influenza clearly showed that the late steps of virus replication are also promising as antiviral targets. Recent studies have addressed the molecular mechanisms of virus budding. A number of studies yielded insight into the budding of enveloped viruses, including arenaviruses. However, this process seems to be more complicated than initially assumed. Despite of the complexity of the virus budding mechanism, there is evidence that targeting of viral budding and high-throughput screening (HTS) would be useful to identify specific anti-arenaviral drugs [18]. Utilization of arenaviral Z protein tagged with Gaussia luciferase (Gluc, 185 amino acid) at its C-terminus may be feasible for HTS [19]. This review presents a summary of current knowledge regarding arenavirus budding and the current model of arenavirus assembly and budding.
4. Intracellular Transport of Other Viral Components
4.1. Transport of vRNA and NP to the Budding Site
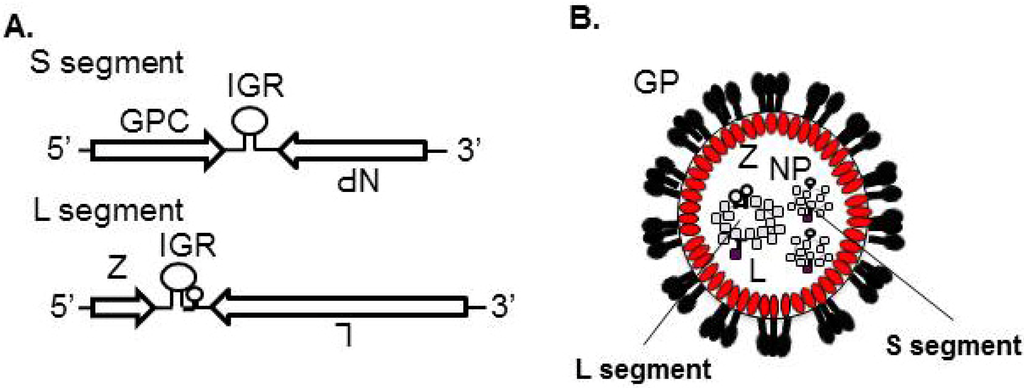
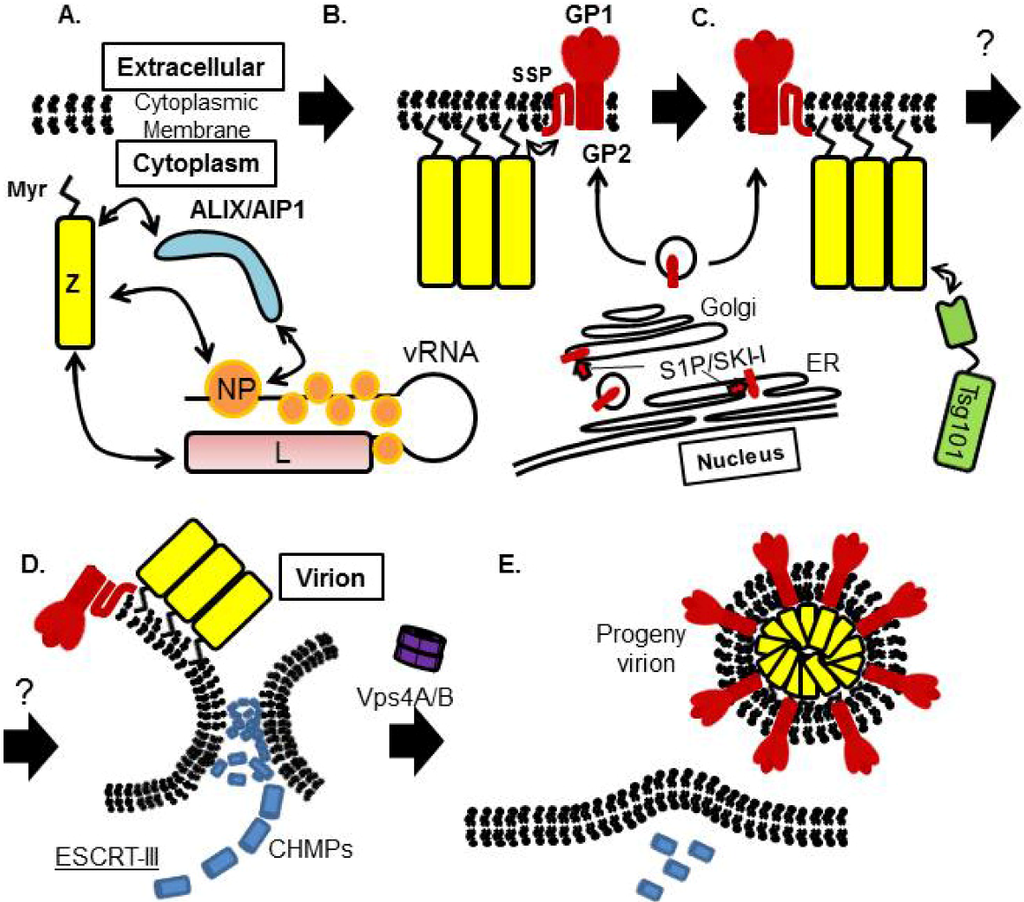
All the virion components must be concentrated at the site of budding, but the mechanism underlying this process is largely unknown for arenaviruses. The presence of IGR in vRNA, which is a hairpin structure aligned between two viral coding genes in both L and S segments, is one of the features of arenavirus [2] (Figure 2A). IGR plays a critical role in LCMV genome incorporation [101]. Previous studies showed that Z interacts with NP, L, and GP [40,49,85,89,90,91,102]. Through the interaction between Z and NP/L, vRNP may be recruited into the virion (Figure 8).
The contribution of NP to LCMV and LASV Z-mediated budding has not been known, although LCMV and LASV NP interact with LCMV and LASV Z, respectively. On the other hand, some arenaviruses’ NP were reported to contribute to the assembly and budding processes. One example is MOPV NP. ALIX/AIP1, one of the ESCRT components, can interact with both MOPV NP and Z, and therefore bridges the interaction between NP and Z [41] (Figure 4A). This interaction facilitates MOPV Z-mediated budding. Another example is TCRV NP. TCRV Z-mediated budding is enhanced by the co-expression of TCRV NP, which was not observed with JUNV Z and NP [49]. These observations suggest that arenavirus Z protein may utilize a different mechanism to facilitate budding.
Although we previously showed that Tsg101 is important for MARV VP40-induced VLP budding and the PPPY motif located in VP40 is crucial for Tsg101-mediated budding and incorporation into VP40-induced VLP [66], it is unlikely that MARV VP40-Tsg101 interaction is direct [103] (Figure 4B). MARV NP was reported to interact with Tsg101, and facilitate VP40-induced VLP production [104]. Based on these data, it is possible that viral proteins can recruit ESCRT components with several different mechanisms and facilitate budding.
4.2. Transport of GP to the Plasma Membrane
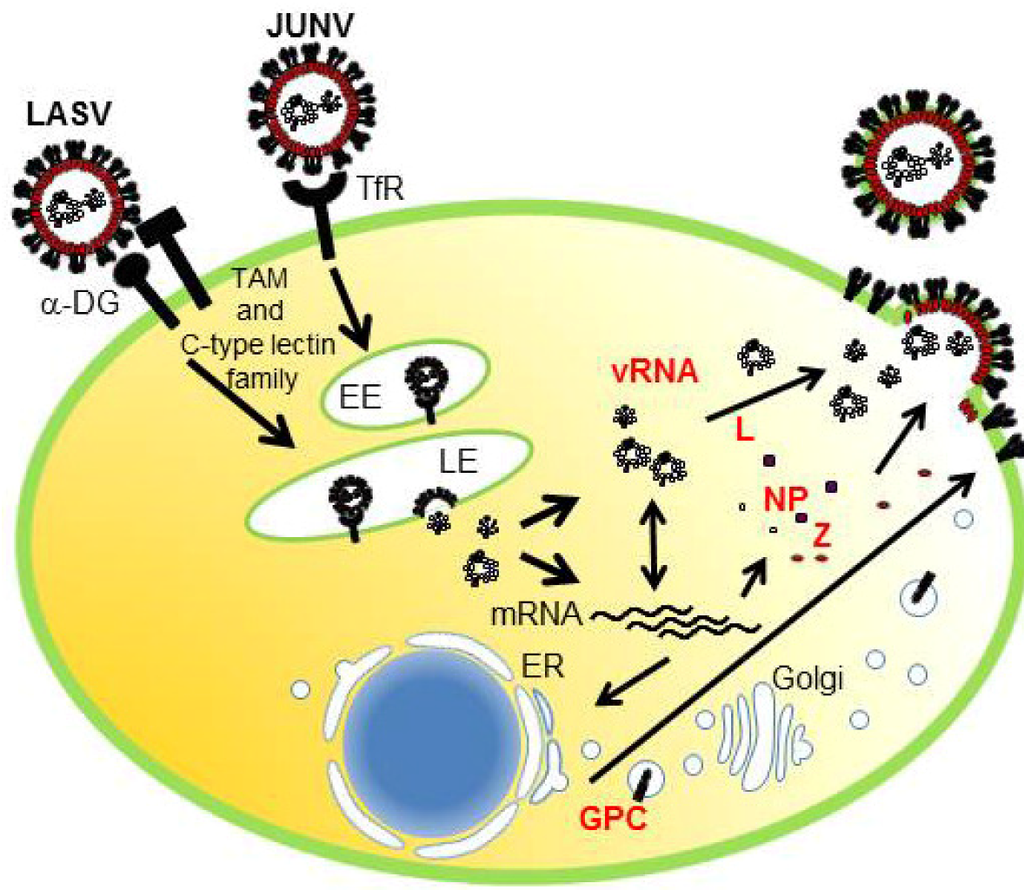
GP is a viral surface glycoprotein that is responsible for binding to the cellular receptor and for the subsequent fusion event (Figure 3). LASV, LCMV [20], as well as clade C of NW arenaviruses [105] use α-DG as the primary receptor, whereas clade B of NW arenaviruses utilize TfR1 [21,106] (Figure 3). Therefore, the incorporation of GP into the virion is a critical process to produce infectious progeny virions. GPC is cleaved by a cellular site 1 protease (S1P/SKI-1) at either ER or Golgi [107,108,109,110], to form GP trimer complex [108,111] (Figure 5). Interestingly, this GP cleavage by S1P/SKI-1 is necessary to incorporate GP into virions, but not transportation to the cell membrane [23]. Recently, it was shown that the S1P/SKI-1 processing is different between GPC and cellular substrates, such as SREBP2 and ATF6, based on the dependency on S1P/SKI-1 autoprocessing [110]. This S1P/SKI-1 cleavage is also important for GP oligomerization [112]. Thus GPC cleavage by S1P/SKI-1 is an attractive target for arenavirus therapy [113,114,115,116,117]. Indeed, we and others have reported that the small chemical compound PF-429242, which targets S1P/SKI-1, can be a leading compound to combat arenavirus [115,118]. In addition, stable signal peptide (SSP) located at the N-terminus of GPC was shown to play a critical role in GPC trafficking of LCMV and JUNV [119,120] (Figure 5), as well as its own expression, processing, cis-acting, and fusion activity [121,122]. SSP was also showed to interact with myristoylated Z [102]. The N-glycosylation of GP is also important for its expression, cleavage, and fusion activity, and these influence virion infectivity [123,124].
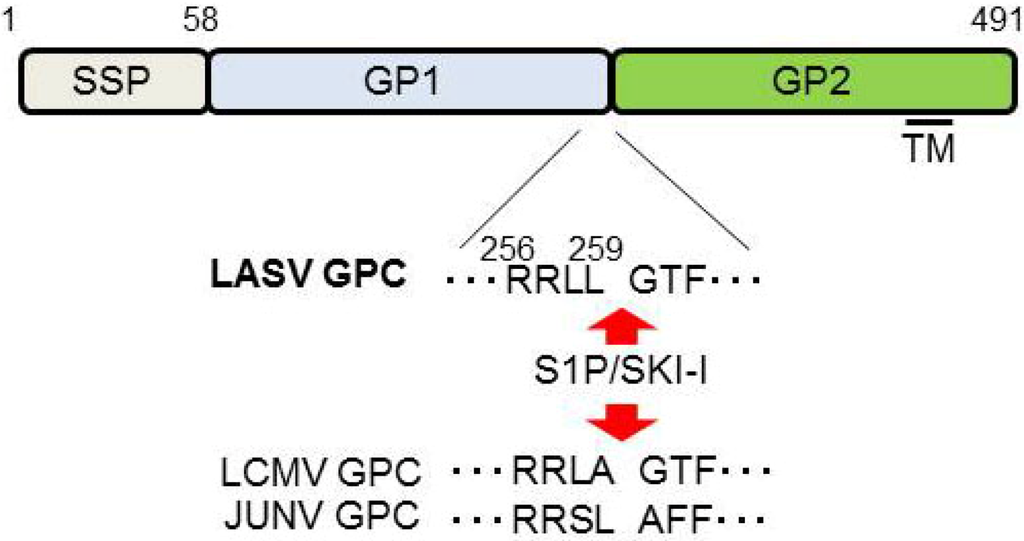
Figure 5.
Schematic representation of LASV GPC. Cleavage sites for S1P/SKI-1 are indicated by the red arrows. The sequences of LCMV and JUNV cleavage sites are also indicated. SSP: stable signal peptide; TM: Transmembrane domain.
5. Roles of Phosphatidylinositol (PI) at the PM, Myristoylation of Viral Matrix Protein, and PI3K on Viral Budding
PIs are lipids distributed in the membrane that are highly regulated by both phosphatases and kinases. Phosphatidylinositol (4, 5) (PI (4, 5) P2) is the most abundant, and is specifically localized on the cytoplasmic leaflet of the PM, and catalyzed by class I PI3Ks to generate PI (3, 4, 5) P3. PI (4, 5) P2 was shown to act as a platform for HIV-1 budding [87,125,126]. The monomeric HIV-1 Gag was proposed to interact with PM through PI (4, 5) P2, thus triggering a conformational change in Gag, followed by myristoylation of G2 [127,128]. This HIV-1 Gag myristoylation has been characterized in detail [129]. It has also been shown that HIV-1 Gag is myristoylated by N-myristoyltransferase (NMT)-1 and -2 [130]. In addition, pH [131] and calmodulin (CaM) binding to the Gag MA domain [132,133] are also important for Gag myristoylation. Furthermore, RNA has been proposed to be required for this Gag-PM binding [134]. Once Gag is myristoylated at PM, Gag-Gag multimerization occurs, and increases the strength of PM-Gag interaction [135,136]. Similarly, oligomerization of TCRV Z is also dependent on its myristoylation [90]. As arenavirus Z is also myristoylated [48,137,138], it is likely that PI acts as a platform for Z-PM interaction. Recently, we demonstrated that Phosphatidylinositol 3-kinase (PI3K) is involved in arenavirus budding [118]. In this study, we used the pan class I PI3K inhibitor, LY294002, as well as dual PI3K/mTOR inhibitor, BEZ-235, which is currently in clinical trial as an anti-solid tumor drug [139]. Based on our results, mTOR seems not to be involved in this activity. Which class of PI3K contributes and the underlying mechanism remain to be elucidated. Nonetheless, we showed that targeting of PI3K can be a novel approach to combat arenavirus infections.
6. Involvement of ESCRT Machinery in the Budding of Enveloped Viruses
The ESCRT pathway exists in all eukaryotes and consists of six heterooligomeric complexes (ESCRT-0, ESCRT-I, ESCRT-II, ESCRT-III, ALIX/AIP1, and VPS4A/B). These complexes are recruited sequentially to membranes and function in protein sorting, membrane remodeling, membrane fission, etc. [46,140]. The ubiquitinated endosomal cargo is first recognized and bound by HRS in ESCRT-0 complex at the membrane, resulting concentration of ubiquitinated cargo on the endosomal membranes (Figure 6AI). In the next step, the cargo is bound by ESCRT-I, through Ubiquitin E2 Variant (UEV) domain of Tsg101, and ESCRT-II is recruited to the membrane. These ESCRT-I and ‑II complexes together induce bud formation (Figure 6AII,III). Finally, ESCRT-III complex mediates membrane scission from the cytosolic side of the bud. ESCRT-III is disassembled and recycled by the VPS4A/B/LIP5 complex (Figure 6AIV,V).
Since the discovery of the interaction between Tsg101 and HIV-1 Gag, and that this interaction facilitates HIV-1 budding, many of the enveloped viruses have been shown to utilize the ESCRT pathway to bud from the cell. The PT/SAP motif within viral matrix protein recognizes the UEV domain of Tsg101 and ride on the ESCRT pathway. The importance of Tsg101 in viral budding has been reported for a variety of viruses [42,44,141]. Interestingly, VSV M possess PT/SAP motif, but neither Tsg101 nor Vps4 are required for replication [142].
ESCRT-II, which contains two copies of EAP20 and a single copy each of EAP30 and EAP45, interacts with both ESCRT-I and -III (Figure 6AIII). However, it has been shown that depletion of EAP45 by RNAi did not affect either HIV-1 budding or cytokinesis [143]. This result indicated that ESCRT-II may have a specific function only in MVB vesicle formation (Figure 6).
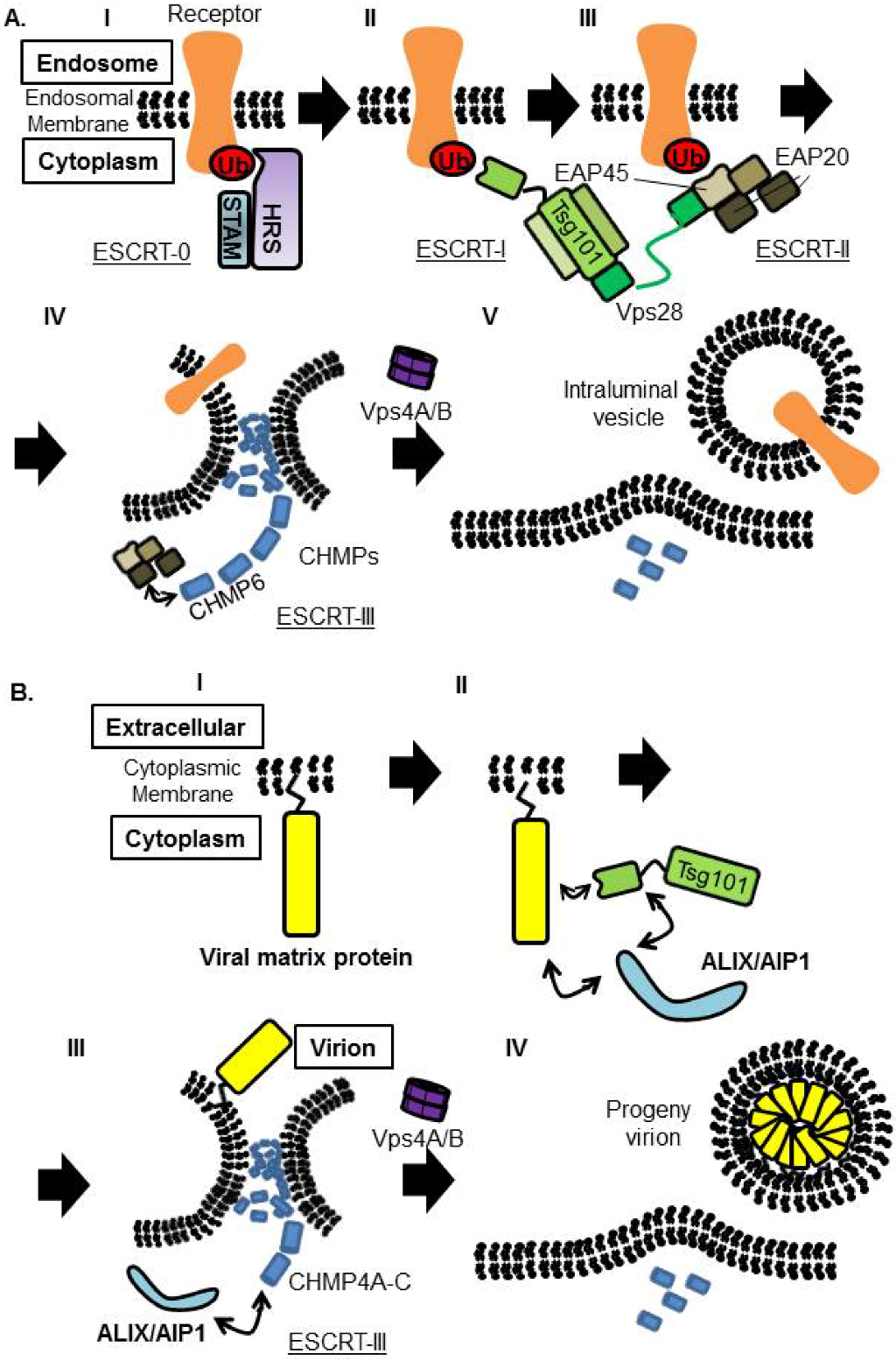
Figure 6.
Proposed model of receptor sorting and virus budding. (A) Model of receptor sorting. Ubiquitinated receptor is recognized by ESCRT-0, followed by Tsg101, one of the ESCRT-I components. ESCRT-II is proposed to connect ESCRT-I and ESCRT-III. ESCRT‑III and the AAA ATPase Vps4A/B trigger the membrane scission to form intraluminal vesicle inside the endosome. (B) Viral matrix protein attached to PM and is recognized by Tsg101. Tsg101 binds to ALIX/AIP1 directly, and ALIX/AIP1 also binds to CHMP4, members of the ESCRT-III components to induce vesicle formation.It is not fully understand how ESCRT-I and ESCRT-III is connected in the virus budding procedure. ESCRT-III complex is disassembled by Vps4A/B, for recycling and initiation of new rounds of sorting or budding.
Although Tsg101 is the most important factor to initiate budding when viral matrix protein possesses the PT/SAP motif, interaction with ALIX/AIP1, which also bridges Tsg101 and CHMP4, can overcome Tsg101 deficiency [144,145] (Figure 6B). EIAV Gag does not possess the PT/SAP motif, but the YPDL motif in EIAV Gag binds directly to the V domain of ALIX/AIP1 and facilitates its budding [144] (Figure 4B and Figure 6B). It was also reported that ALIX/AIP1 played an important role in bridging MOPV Z and NP [40,41]. We showed previously that ALIX/AIP1 does not contribute to LASV Z-mediated budding based on analysis using siRNA specific for ALIX/AIP1 [82], but we did not examine whether ALIX/AIP1 contributes to other processes.
ESCRT-III is comprised on charged MVB proteins (CHMPs) (Figure 6). In humans, twelve proteins were reported to compose the ESCRT-III complex. These ESCRT-III proteins play a critical role in membrane fission by assembling into helical filaments within the membrane neck structure. Recently, it was shown that utilization of these CHMPs is regulated in a sophisticated manner between HIV-1 budding and cytokinesis, as only some are required for HIV-1 budding, but most play a significant role in cytokinesis [146].
Vps4 is an AAA-ATPase, and involved in disassembly and recycling of ESCRT-III complex. Vps4 is compromised by Vps4A and Vps4B, and binds to LIP5, which serves as an activator of Vps4 assembly and ATPase activities (Figure 6).
HCET-type Nedd4-like E3 ligases are recognized to bind to the PPXY motif of viral matrix protein through its WW domain (Figure 4B). The WW domain consists of 38–40 amino acids containing two widely spaced tryptophans (W) and binds to a variety of proteins containing the PPXY motif. Although E3 ligase does not belong to ESCRT components, many studies have demonstrated the importance of E3 ligase in viral budding. Nonetheless, the precise roles of these E3 ligases in the viral budding process are still unknown. It is obvious that there is some specificity of this WW domain to bind and regulate PPXY motif-mediated budding, as the specific WW domain acts in a dominant negative manner for the budding. We showed previously that the Nedd4.1 WW domain does not inhibit LASV Z-mediated budding [82], but this WW domain inhibited EBOV and MARV VP40‑mediated budding [147,148]. Gag proteins of many retroviruses, including HTLV-1, RSV, MoMLV, and M‑PMV, have been shown to interact with specific E3 ligases and be regulated [149,150,151,152,153,154,155]. Noteworthy, overexpression of Nedd4.2 (or Nedd4L) enhanced its budding, although HIV-1 Gag does not possess canonical PPXY motif [156,157], suggesting an important role of E3 ligase on HIV-1 replication.
LASV Z possesses the PPPY motif which is critical for virus budding [81]. However, the E3 ligase which specifically regulates the LASV budding has not been identified. The Arrestin-related trafficking (ART) proteins have been reported to bridge ESCRT machinery and E3 ligases. Arenaviral Z proteins, which possess PPXY motif such as LASV, might be connected to the ESCRT machinery via ART proteins, together with the PTAP motif-Tsg101 interaction [45,158].
7. Regulation of Viral Assembly and Budding by IFN
7.1. Regulation of Virus Budding by Interferon-Stimulated Genes (ISGs)
IFN is a well-characterized host immune molecule that triggers expression of hundreds of so-called interferon stimulated genes (ISGs). In general, many of the viruses encode specific proteins that antagonize innate immune responses [159,160]. The tripartite motif containing 5α (TRIM5α) has been shown to be induced by IFN-α, and is well known as a HIV-1 entry host restriction factor [161,162,163]. In addition, TRIM5α has been shown to regulate HIV-1 budding [164,165]. Recent reports have suggested that not all of the ISGs show anti-viral activity, but rather exhibit specificity among viruses [166,167]. A wide scale experiment was performed to determine the antiviral activities of ISGs against hepatitis C virus, yellow fever virus, West Nile virus, chikungunya virus, Venezuelan equine encephalitis virus, and HIV-1. The authors screened more than 380 ISGs independently to determine their antiviral activities against these selected viruses, and demonstrated the diversity of these functions [166]. A total of 288 ISGs were then tested against VSV and murine gammmaherpes virus 68 (MHV-68) [167]. It is not clear whether these ISGs contribute to inhibition of arenvirus assembly, budding, and release. Therefore further analysis is required.
7.3. Inhibition of Virus Release by the Interferon-Induced Cellular Protein, Tetherin
As described above, some interferon-induced proteins clearly show antiviral activity specifically at the late stage of virus replication, which included ISG15 and TRIM5α [164,165,168,169]. Recently, one of these ISGs, named Tetherin (also known as BST-2, CD317, or HM1.24), has been reported as an inhibitory cellular factor against HIV-1 [175,176]. We and other groups reported that Tetherin shows antiviral activity against other retroviruses, filoviruses, and arenaviruses [177,178]. Tetherin is constitutively expressed in terminally differentiated B cells, bone marrow stromal cells, and pDC, and is also broadly induced by treatment with Type I and Type II IFNs in various cell types [176]. Therefore, Tetherin is thought to be involved in antiviral host defense as one of the innate immunity mechanisms. In addition, Tetherin expression has been shown to be increased in multiple myeloma [179], endometrial cancer [180], primary lung cancer [181], and glioma cells [182]. Therefore, several groups have attempted immunotherapy using anti-Tetherin antibodies, which mediate antibody-dependent cellular cytotoxicity [181,183].
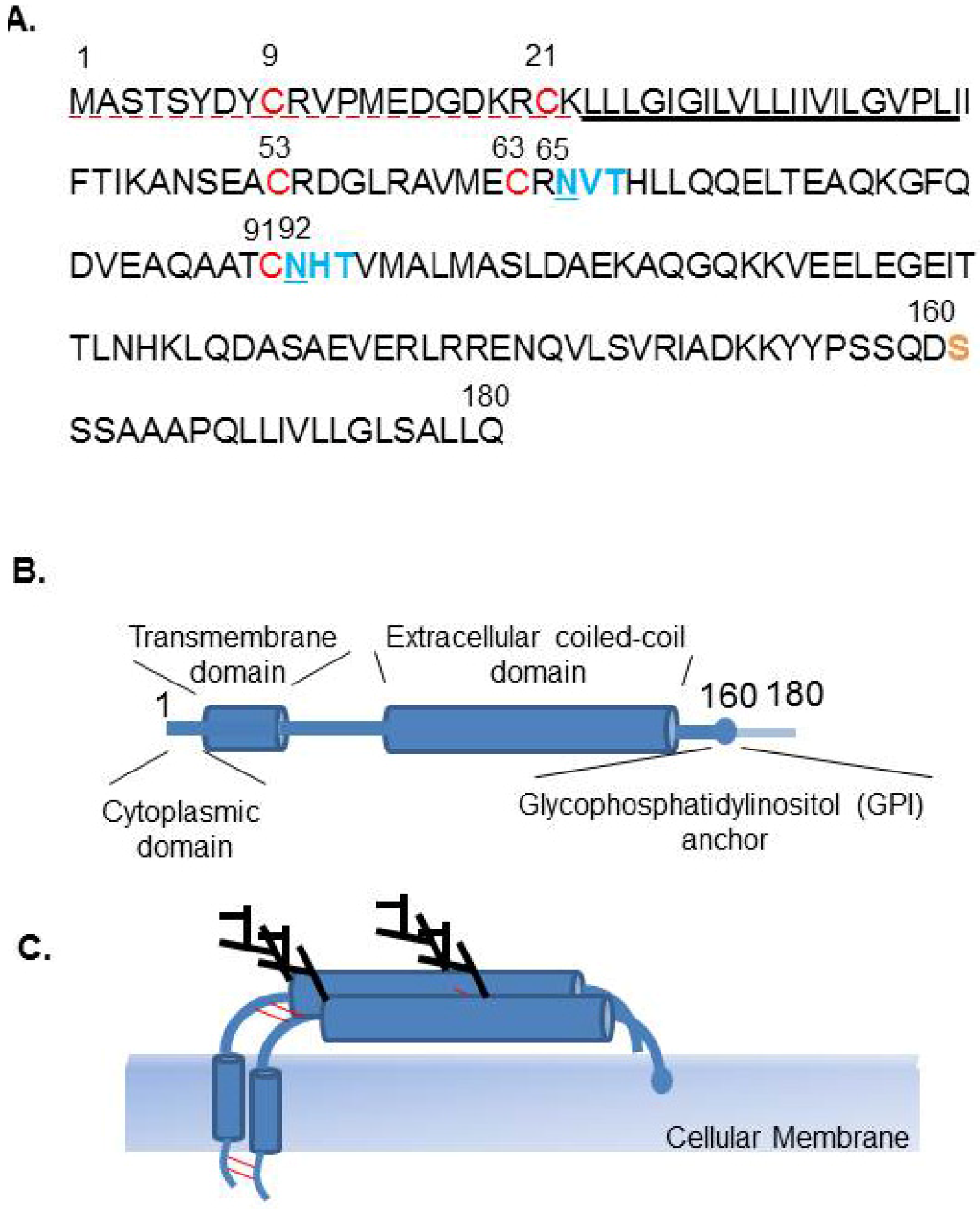
Figure 7.
Characteristics of human Tetherin. (A) Amino acid sequence of human Tetherin (accession number NP_004326). Dashed red underline (1-22) indicates the cytoplasmic domain (CD), underline indicates the transmembrane domain (TM). Red font (C9, C21, C53, C63, and C91) indicates amino acids that may be responsible for disulfide bond formation and dimerization. Blue font indicates potential N-linked glycosylation motif (N‑X-S/T) and site (underlined). Serine (S) at position 160 is indicated as the site of cleavage prior to the C-terminal addition of a GPI anchor in yellow font. (B) Schematic representation of the domain structure of Tetherin, a type II transmembrane protein. (C) Tetherin forms a homodimer, through a C-C disulfide bond. Two sites of N‑glycosilation are also indicated.
Tetherin is a type II transmembrane protein, consisting of four domains, i.e., an N-terminal cytoplasmic domain (CD), a single transmembrane domain (TM), an extracellular domain, and a putative C-terminal glycosyl phosphatidylinositol (GPI) anchor (Figure 7A–C), which is present on the cell surface and in perinuclear compartments. Tetherin is anchored in the cell membrane at both ends, and combined high-resolution crystallography and small-angle X-ray scattering-based modeling indicated that the Tetherin ectodomain forms a parallel coiled-coil homodimer [184,185,186,187,188]. Therefore, it has been suggested that Tetherin inhibits progeny virus release by directly tethering virions to cells, briefly by anchoring one end of the molecule on the cell membrane and the other end on the viral envelope. Progeny virions released from cells could also be directly tethered to each other by Tetherin. Electron microscopy was performed to confirm this model, and based on the distances between tethered virions and cells, the membrane-spanning model was proposed as the model of action of Tetherin [176,189,190]. Interestingly, an artificial Tetherin-like protein, composed by domain replacement of TfR at the N-terminus, dystrophia myotonica protein kinase (DMPK) at the ectodomain, and urokinase plasminogen activator receptor (uPAR) at the C-terminus was also shown to have antiviral activity similar to that of Tetherin, suggesting that this typical conformation or configuration acts to tether virion progeny at the cell surface [189].
Some viruses encode IFN antagonists to avoid or inhibit the innate immune response, such as arenavirus NP, EBOV VP35, and influenza virus NS1 [159,191,192]. In general, viral antagonists inhibit the innate immune signaling cascades. In addition to these IFN antagonists, some viruses encode specific viral proteins that antagonize the antiviral activity of Tetherin [178,193]. HIV-1 Vpu is the best characterized Tetherin antagonist. HIV-2 Env, SIV Env, SIV Nef, KSHV K5, and EBOV and MARV GP have also been reported as Tetherin antagonists, but their mechanisms of action seem to differ among these viral proteins [178,193,194,195].
Initially, we reported that Tetherin inhibits LASV Z-mediated VLP release, and this restriction was not overcome by LASV GP, but was overcome by HIV-1 Vpu expression [177]. In addition, we showed that the glycosylation of Tetherin at 65 and 92 asparagine (N) is dispensable for its activity [177]. Moreover, we showed that Tetherin dimerization through its C-C interaction had little effect on its inhibitory activity against Lassa VLP release, but was not essential for its antiviral activity [196]. It is likely that tethering of virions by Tetherin dimers is stronger than that by Tetherin monomers because of the stronger association with the membrane. Therefore, Tetherin appears to inhibit release of a wide variety of enveloped viruses from host cells by a similar mechanism, as Tetherin shows antiviral activities against many retroviruses, filoviruses, arenaviruses, and KSHV. However, there is controversy regarding the importance of glycosylation and dimerization of Tetherin for its antiviral activity.
Recent studies using mouse models have assessed the importance of Tetherin with regard to its antiviral activity against Mo-MLV [197], mouse mammary tumor virus (MMTV) [198], VSV, and influenza B virus [199]. In case of Mo-MLV and MMTV, knock out (KO) or knock down (KD) of Tetherin exhibited higher titer of these viruses in vivo, suggesting Tetherin exhibited antiviral activity against these viruses. In addition, HIV-1 Vpu, which antagonizes Tetherin, has been shown to contribute to the efficient spread of HIV-1 using the humanized mouse model transplanted human hematopoietic stem cells [200]. On the other hand, KO of Tetherin showed lower titer of VSV and influenza virus B [199]. These results indicated that the antiviral effect of Tetherin in vivo is complicated and showed different phenotype depending on the virus species.
In the case of arenaviruses, one study demonstrated that the infectious LASV was inhibited by human Tetherin [84]. In addition, this report indicated that LASV does not possess any Tetherin antagonist [84]. It is not yet clear whether Tetherin has any effect on arenavirus propagation and pathogenesis in vivo.
9. Concluding Remarks
This review described current knowledge regarding arenavirus assembly and budding. Although many groups have been worked in this field, many questions remain unanswered, including the role of E3 ligase in the budding process, identification of ISGs that contribute to (or inhibit) assembly and budding, as well as the precise molecular mechanisms by which these proteins facilitate virus budding.
Novel arenaviruses have been identified on average every three years. It is easy to imagine that a new highly pathogenic arenavirus may emerge, similar to Lujo virus [4]. Although individual arenaviruses use different strategies for assembly and budding, they can be classified into a few groups based on alignment of the Z C-terminus. Therefore, investigation of the molecular mechanism underlying this assembly and budding processes, and understanding of the common and unique mechanisms are necessary to develop and identify new antiviral therapies to combat pathogenic arenaviruses.
Acknowledgments
We thank all members of the Emerging Infectious Diseases Laboratory at the Institute of Tropical Medicine, Nagasaki University. J. Y. is supported by grants from the Ministry of Health, Labour and Welfare of Japan and the Japan Society for the Promotion of Science.
Conflict of Interest
The authors declare no conflict of interest.
References and Notes
- Homology Analysis (Lipman-Pearson Method), version 11.0.4, GENETYX Corporation: Tokyo, Japan, 2011.
- Buchmeier, M.J.; Peters, C.J.; de la Torre, J.C. Arenaviridae: The virus and their replication. Fields Virology 2007, 2, 1792–1827. [Google Scholar]
- Peters, C.J. Human infection with arenaviruses in the Americas. Curr. Top. Microbiol. Immunol. 2002, 262, 65–74. [Google Scholar] [CrossRef]
- Briese, T.; Paweska, J.T.; McMullan, L.K.; Hutchison, S.K.; Street, C.; Palacios, G.; Khristova, M.L.; Weyer, J.; Swanepoel, R.; Egholm, M.; et al. Genetic detection and characterization of Lujo virus, a new hemorrhagic fever-associated arenavirus from southern Africa. PLoS Pathog. 2009, 5, e1000455. [Google Scholar] [CrossRef]
- Delgado, S.; Erickson, B.R.; Agudo, R.; Blair, P.J.; Vallejo, E.; Albarino, C.G.; Vargas, J.; Comer, J.A.; Rollin, P.E.; Ksiazek, T.G.; et al. Chapare virus, a newly discovered arenavirus isolated from a fatal hemorrhagic fever case in Bolivia. PLoS Pathog. 2008, 4, e1000047. [Google Scholar] [CrossRef]
- Barton, L.L.; Mets, M.B.; Beauchamp, C.L. Lymphocytic choriomeningitis virus: Emerging fetal teratogen. Am. J. Obstet. Gynecol. 2002, 187, 1715–1716. [Google Scholar] [CrossRef]
- Fischer, S.A.; Graham, M.B.; Kuehnert, M.J.; Kotton, C.N.; Srinivasan, A.; Marty, F.M.; Comer, J.A.; Guarner, J.; Paddock, C.D.; DeMeo, D.L.; et al. Transmission of lymphocytic choriomeningitis virus by organ transplantation. N. Engl. J. Med. 2006, 354, 2235–2249. [Google Scholar]
- Palacios, G.; Druce, J.; Du, L.; Tran, T.; Birch, C.; Briese, T.; Conlan, S.; Quan, P.L.; Hui, J.; Marshall, J.; et al. A new arenavirus in a cluster of fatal transplant-associated diseases. N. Engl. J. Med. 2008, 358, 991–998. [Google Scholar]
- Damonte, E.B.; Coto, C.E. Treatment of arenavirus infections: From basic studies to the challenge of antiviral therapy. Adv. Virus Res. 2002, 58, 125–155. [Google Scholar] [CrossRef]
- Lee, A.M.; Pasquato, A.; Kunz, S. Novel approaches in anti-arenaviral drug development. Virology 2011, 411, 163–169. [Google Scholar] [CrossRef]
- Nunberg, J.H.; York, J. The curious case of arenavirus entry, and its inhibition. Viruses 2012, 4, 83–101. [Google Scholar] [CrossRef]
- Lee, A.M.; Rojek, J.M.; Spiropoulou, C.F.; Gundersen, A.T.; Jin, W.; Shaginian, A.; York, J.; Nunberg, J.H.; Boger, D.L.; Oldstone, M.B.; et al. Unique small molecule entry inhibitors of hemorrhagic fever arenaviruses. J. Biol. Chem. 2008, 283, 18734–18742. [Google Scholar]
- Bolken, T.C.; Laquerre, S.; Zhang, Y.; Bailey, T.R.; Pevear, D.C.; Kickner, S.S.; Sperzel, L.E.; Jones, K.F.; Warren, T.K.; Amanda Lund, S.; et al. Identification and characterization of potent small molecule inhibitor of hemorrhagic fever New World arenaviruses. Antivir. Res. 2006, 69, 86–97. [Google Scholar] [CrossRef]
- Larson, R.A.; Dai, D.; Hosack, V.T.; Tan, Y.; Bolken, T.C.; Hruby, D.E.; Amberg, S.M. Identification of a broad-spectrum arenavirus entry inhibitor. J. Virol. 2008, 82, 10768–10775. [Google Scholar]
- Cashman, K.A.; Smith, M.A.; Twenhafel, N.A.; Larson, R.A.; Jones, K.F.; Allen, R.D., 3rd; Dai, D.; Chinsangaram, J.; Bolken, T.C.; Hruby, D.E.; et al. Evaluation of Lassa antiviral compound ST-193 in a guinea pig model. Antivir. Res. 2011, 90, 70–79. [Google Scholar]
- York, J.; Dai, D.; Amberg, S.M.; Nunberg, J.H. pH-induced activation of arenavirus membrane fusion is antagonized by small-molecule inhibitors. J. Virol. 2008, 82, 10932–10939. [Google Scholar] [CrossRef]
- Thomas, C.J.; Casquilho-Gray, H.E.; York, J.; DeCamp, D.L.; Dai, D.; Petrilli, E.B.; Boger, D.L.; Slayden, R.A.; Amberg, S.M.; Sprang, S.R.; et al. A specific interaction of small molecule entry inhibitors with the envelope glycoprotein complex of the Junin hemorrhagic fever arenavirus. J. Biol. Chem. 2011, 286, 6192–6200. [Google Scholar]
- Urata, S.; de la Torre, J.C. Arenavirus budding. Adv. Virol. 2011, 2011, 180326. [Google Scholar]
- Capul, A.A.; de la Torre, J.C. A cell-based luciferase assay amenable to high-throughput screening of inhibitors of arenavirus budding. Virology 2008, 382, 107–114. [Google Scholar] [CrossRef]
- Cao, W.; Henry, M.D.; Borrow, P.; Yamada, H.; Elder, J.H.; Ravkov, E.V.; Nichol, S.T.; Compans, R.W.; Campbell, K.P.; Oldstone, M.B. Identification of alpha-dystroglycan as a receptor for lymphocytic choriomeningitis virus and Lassa fever virus. Science 1998, 282, 2079–2081. [Google Scholar] [CrossRef]
- Radoshitzky, S.R.; Abraham, J.; Spiropoulou, C.F.; Kuhn, J.H.; Nguyen, D.; Li, W.; Nagel, J.; Schmidt, P.J.; Nunberg, J.H.; Andrews, N.C.; et al. Transferrin receptor 1 is a cellular receptor for New World haemorrhagic fever arenaviruses. Nature 2007, 446, 92–96. [Google Scholar] [CrossRef]
- Shimojima, M.; Stroher, U.; Ebihara, H.; Feldmann, H.; Kawaoka, Y. Identification of cell surface molecules involved in dystroglycan-independent Lassa virus cell entry. J. Virol. 2012, 86, 2067–2078. [Google Scholar] [CrossRef]
- Kunz, S.; Edelmann, K.H.; de la Torre, J.C.; Gorney, R.; Oldstone, M.B. Mechanisms for lymphocytic choriomeningitis virus glycoprotein cleavage, transport, and incorporation into virion. Virology 2003, 314, 168–178. [Google Scholar] [CrossRef]
- Kunz, S.; Borrow, P.; Oldstone, M.B. Receptor structure, binding, and cell entry of arenaviruse. Curr. Top. Microbiol. Immunol. 2002, 262, 111–137. [Google Scholar]
- Lee, K.J.; Novella, I.S.; Teng, M.N.; Oldstone, M.B.; de La Torre, J.C. NP and L proteins of lymphocytic choriomeningitis virus (LCMV) are sufficient for efficient transcription and replication of LCMV genomic RNA analogs. J. Virol. 2000, 74, 3470–3477. [Google Scholar] [CrossRef]
- Cornu, T.I.; de la Torre, J.C. RING finger Z protein of lymphocytic choriomeningitis virus (LCMV) inhibits transcription and RNA replication of an LCMV S-segment minigenome. J. Virol. 2001, 75, 9415–9426. [Google Scholar] [CrossRef]
- Cornu, T.I.; de la Torre, J.C. Characterization of the arenavirus RING finger Z protein regions required for Z-mediated inhibition of viral RNA synthesis. J. Virol. 2002, 76, 6678–6688. [Google Scholar]
- Hass, M.; Golnitz, U.; Muller, S.; Becker-Ziaja, B.; Gunther, S. Replicon system for Lassa virus. J. Virol. 2004, 78, 13793–13803. [Google Scholar]
- Lan, S.; McLay Schelde, L.; Wang, J.; Kumar, N.; Ly, H.; Liang, Y. Development of infectious clones for virulent and avirulent pichinde viruses: A model virus to study arenavirus-induced hemorrhagic fevers. J. Virol. 2009, 83, 6357–6362. [Google Scholar]
- Lopez, N.; Jacamo, R.; Franze-Fernandez, M.T. Transcription and RNA replication of tacaribe virus genome and antigenome analogs require N and L proteins: Z protein is an inhibitor of these processes. J. Virol. 2001, 75, 12241–12251. [Google Scholar] [CrossRef]
- Cornu, T.I.; Feldmann, H.; de la Torre, J.C. Cells expressing the RING finger Z protein are resistant to arenavirus infection. J. Virol. 2004, 78, 2979–2983. [Google Scholar]
- Emonet, S.E.; Urata, S.; de la Torre, J.C. Arenavirus reverse genetics: New approaches for the investigation of arenavirus biology and development of antiviral strategies. Virology 2011, 411, 416–425. [Google Scholar] [CrossRef]
- Volpon, L.; Osborne, M.J.; Capul, A.A.; de la Torre, J.C.; Borden, K.L. Structural characterization of the Z RING-eIF4E complex reveals a distinct mode of control for eIF4E. Proc. Natl. Acad. Sci. U. S. A. 2010, 107, 5441–5446. [Google Scholar]
- Asper, M.; Sternsdorf, T.; Hass, M.; Drosten, C.; Rhode, A.; Schmitz, H.; Gunther, S. Inhibition of different Lassa virus strains by alpha and gamma interferons and comparison with a less pathogenic arenavirus. J. Virol. 2004, 78, 3162–3169. [Google Scholar]
- Kranzusch, P.J.; Whelan, S.P. Arenavirus Z protein controls viral RNA synthesis by locking a polymerase-promoter complex. Proc. Natl. Acad. Sci. U. S. A. 2011, 108, 19743–19748. [Google Scholar] [CrossRef]
- Fan, L.; Briese, T.; Lipkin, W.I. Z proteins of New World arenaviruses bind RIG-I and interfere with type I interferon induction. J. Virol. 2010, 84, 1785–1791. [Google Scholar] [CrossRef]
- Colina, R.; Costa-Mattioli, M.; Dowling, R.J.; Jaramillo, M.; Tai, L.H.; Breitbach, C.J.; Martineau, Y.; Larsson, O.; Rong, L.; Svitkin, Y.V.; et al. Translational control of the innate immune response through IRF-7. Nature 2008, 452, 323–328. [Google Scholar]
- Gamsjaeger, R.; Liew, C.K.; Loughlin, F.E.; Crossley, M.; Mackay, J.P. Sticky fingers: Zinc-fingers as protein-recognition motifs. Trends Biochem. Sci. 2007, 32, 63–70. [Google Scholar] [CrossRef]
- Deshaies, R.J.; Joazeiro, C.A. RING domain E3 ubiquitin ligases. Annu. Rev. Biochem. 2009, 78, 399–434. [Google Scholar] [CrossRef]
- Shtanko, O.; Imai, M.; Goto, H.; Lukashevich, I.S.; Neumann, G.; Watanabe, T.; Kawaoka, Y. A role for the C terminus of Mopeia virus nucleoprotein in its incorporation into Z protein-induced virus-like particles. J. Virol. 2010, 84, 5415–5422. [Google Scholar]
- Shtanko, O.; Watanabe, S.; Jasenosky, L.D.; Watanabe, T.; Kawaoka, Y. ALIX/AIP1 is required for NP incorporation into Mopeia virus Z-induced virus-like particles. J. Virol. 2011, 85, 3631–3641. [Google Scholar]
- Bieniasz, P.D. Late budding domains and host proteins in enveloped virus release. Virology 2006, 344, 55–63. [Google Scholar] [CrossRef]
- Schmitt, A.P.; Leser, G.P.; Morita, E.; Sundquist, W.I.; Lamb, R.A. Evidence for a new viral late-domain core sequence, FPIV, necessary for budding of a paramyxovirus. J. Virol. 2005, 79, 2988–2997. [Google Scholar] [CrossRef]
- Morita, E.; Sundquist, W.I. Retrovirus budding. Annu. Rev. Cell Dev. Biol. 2004, 20, 395–425. [Google Scholar] [CrossRef]
- Martin-Serrano, J.; Neil, S.J. Host factors involved in retroviral budding and release. Nat. Rev. Microbiol. 2011, 9, 519–531. [Google Scholar]
- Rusten, T.E.; Vaccari, T.; Stenmark, H. Shaping development with ESCRTs. Nat. Cell Biol. 2012, 14, 38–45. [Google Scholar]
- Harty, R.N.; Brown, M.E.; Wang, G.; Huibregtse, J.; Hayes, F.P. A PPxY motif within the VP40 protein of Ebola virus interacts physically and functionally with a ubiquitin ligase: Implications for filovirus budding. Proc. Natl. Acad. Sci. U. S. A. 2000, 97, 13871–13876. [Google Scholar] [CrossRef]
- Urata, S.; Yasuda, J.; de la Torre, J.C. The z protein of the new world arenavirus tacaribe virus has bona fide budding activity that does not depend on known late domain motifs. J. Virol. 2009, 83, 12651–12655. [Google Scholar] [CrossRef]
- Groseth, A.; Wolff, S.; Strecker, T.; Hoenen, T.; Becker, S. Efficient budding of the tacaribe virus matrix protein z requires the nucleoprotein. J. Virol. 2010, 84, 3603–3611. [Google Scholar]
- Gottlinger, H.G.; Dorfman, T.; Sodroski, J.G.; Haseltine, W.A. Effect of mutations affecting the p6 gag protein on human immunodeficiency virus particle release. Proc. Natl. Acad. Sci. U. S. A. 1991, 88, 3195–3199. [Google Scholar]
- Huang, M.; Orenstein, J.M.; Martin, M.A.; Freed, E.O. p6Gag is required for particle production from full-length human immunodeficiency virus type 1 molecular clones expressing protease. J. Virol. 1995, 69, 6810–6818. [Google Scholar]
- Yu, X.F.; Matsuda, Z.; Yu, Q.C.; Lee, T.H.; Essex, M. Role of the C terminus Gag protein in human immunodeficiency virus type 1 virion assembly and maturation. J. Gen. Virol. 1995, 76, 3171–3179. [Google Scholar] [CrossRef]
- Puffer, B.A.; Parent, L.J.; Wills, J.W.; Montelaro, R.C. Equine infectious anemia virus utilizes a YXXL motif within the late assembly domain of the Gag p9 protein. J. Virol. 1997, 71, 6541–6546. [Google Scholar]
- Yuan, B.; Li, X.; Goff, S.P. Mutations altering the moloney murine leukemia virus p12 Gag protein affect virion production and early events of the virus life cycle. EMBO J. 1999, 18, 4700–4710. [Google Scholar] [CrossRef]
- Yasuda, J.; Hunter, E. A proline-rich motif (PPPY) in the Gag polyprotein of Mason-Pfizer monkey virus plays a maturation-independent role in virion release. J. Virol. 1998, 72, 4095–4103. [Google Scholar]
- Wills, J.W.; Cameron, C.E.; Wilson, C.B.; Xiang, Y.; Bennett, R.P.; Leis, J. An assembly domain of the Rous sarcoma virus Gag protein required late in budding. J. Virol. 1994, 68, 6605–6618. [Google Scholar]
- Xiang, Y.; Cameron, C.E.; Wills, J.W.; Leis, J. Fine mapping and characterization of the Rous sarcoma virus Pr76gag late assembly domain. J. Virol. 1996, 70, 5695–5700. [Google Scholar]
- Stange, A.; Mannigel, I.; Peters, K.; Heinkelein, M.; Stanke, N.; Cartellieri, M.; Gottlinger, H.; Rethwilm, A.; Zentgraf, H.; Lindemann, D. Characterization of prototype foamy virus gag late assembly domain motifs and their role in particle egress and infectivity. J. Virol. 2005, 79, 5466–5476. [Google Scholar] [CrossRef]
- Le Blanc, I.; Prevost, M.C.; Dokhelar, M.C.; Rosenberg, A.R. The PPPY motif of human T-cell leukemia virus type 1 Gag protein is required early in the budding process. J. Virol. 2002, 76, 10024–10029. [Google Scholar]
- Jayakar, H.R.; Jeetendra, E.; Whitt, M.A. Rhabdovirus assembly and budding. Virus Res. 2004, 106, 117–132. [Google Scholar] [CrossRef]
- Jayakar, H.R.; Murti, K.G.; Whitt, M.A. Mutations in the PPPY motif of vesicular stomatitis virus matrix protein reduce virus budding by inhibiting a late step in virion release. J. Virol. 2000, 74, 9818–9827. [Google Scholar] [CrossRef]
- Noda, T.; Sagara, H.; Suzuki, E.; Takada, A.; Kida, H.; Kawaoka, Y. Ebola virus VP40 drives the formation of virus-like filamentous particles along with GP. J. Virol. 2002, 76, 4855–4865. [Google Scholar] [CrossRef]
- Craven, R.C.; Harty, R.N.; Paragas, J.; Palese, P.; Wills, J.W. Late domain function identified in the vesicular stomatitis virus M protein by use of rhabdovirus-retrovirus chimeras. J. Virol. 1999, 73, 3359–3365. [Google Scholar]
- Justice, P.A.; Sun, W.; Li, Y.; Ye, Z.; Grigera, P.R.; Wagner, R.R. Membrane vesiculation function and exocytosis of wild-type and mutant matrix proteins of vesicular stomatitis virus. J. Virol. 1995, 69, 3156–3160. [Google Scholar]
- Kolesnikova, L.; Bugany, H.; Klenk, H.D.; Becker, S. VP40, the matrix protein of Marburg virus, is associated with membranes of the late endosomal compartment. J. Virol. 2002, 76, 1825–1838. [Google Scholar] [CrossRef]
- Urata, S.; Noda, T.; Kawaoka, Y.; Morikawa, S.; Yokosawa, H.; Yasuda, J. Interaction of Tsg101 with Marburg virus VP40 depends on the PPPY motif, but not the PT/SAP motif as in the case of Ebola virus, and Tsg101 plays a critical role in the budding of Marburg virus-like particles induced by VP40, NP, and GP. J. Virol. 2007, 81, 4895–4899. [Google Scholar] [CrossRef]
- Jasenosky, L.D.; Neumann, G.; Lukashevich, I.; Kawaoka, Y. Ebola virus VP40-induced particle formation and association with the lipid bilayer. J. Virol. 2001, 75, 5205–5214. [Google Scholar] [CrossRef]
- Timmins, J.; Scianimanico, S.; Schoehn, G.; Weissenhorn, W. Vesicular release of ebola virus matrix protein VP40. Virology 2001, 283, 1–6. [Google Scholar] [CrossRef]
- Harty, R.N.; Paragas, J.; Sudol, M.; Palese, P. A proline-rich motif within the matrix protein of vesicular stomatitis virus and rabies virus interacts with WW domains of cellular proteins: Implications for viral budding. J. Virol. 1999, 73, 2921–2929. [Google Scholar]
- Pohl, C.; Duprex, W.P.; Krohne, G.; Rima, B.K.; Schneider-Schaulies, S. Measles virus M and F proteins associate with detergent-resistant membrane fractions and promote formation of virus-like particles. J. Gen. Virol. 2007, 88, 1243–1250. [Google Scholar] [CrossRef]
- Runkler, N.; Pohl, C.; Schneider-Schaulies, S.; Klenk, H.D.; Maisner, A. Measles virus nucleocapsid transport to the plasma membrane requires stable expression and surface accumulation of the viral matrix protein. Cell Microbiol. 2007, 9, 1203–1214. [Google Scholar] [CrossRef]
- Pantua, H.D.; McGinnes, L.W.; Peeples, M.E.; Morrison, T.G. Requirements for the assembly and release of Newcastle disease virus-like particles. J. Virol. 2006, 80, 11062–11073. [Google Scholar] [CrossRef]
- Coronel, E.C.; Murti, K.G.; Takimoto, T.; Portner, A. Human parainfluenza virus type 1 matrix and nucleoprotein genes transiently expressed in mammalian cells induce the release of virus-like particles containing nucleocapsid-like structures. J. Virol. 1999, 73, 7035–7038. [Google Scholar]
- Takimoto, T.; Murti, K.G.; Bousse, T.; Scroggs, R.A.; Portner, A. Role of matrix and fusion proteins in budding of Sendai virus. J. Virol. 2001, 75, 11384–11391. [Google Scholar] [CrossRef]
- Patch, J.R.; Crameri, G.; Wang, L.F.; Eaton, B.T.; Broder, C.C. Quantitative analysis of Nipah virus proteins released as virus-like particles reveals central role for the matrix protein. Virol. J. 2007, 4, 1. [Google Scholar] [CrossRef]
- Patch, J.R.; Han, Z.; McCarthy, S.E.; Yan, L.; Wang, L.F.; Harty, R.N.; Broder, C.C. The YPLGVG sequence of the Nipah virus matrix protein is required for budding. Virol. J. 2008, 5, 137. [Google Scholar] [CrossRef]
- Ciancanelli, M.J.; Basler, C.F. Mutation of YMYL in the Nipah virus matrix protein abrogates budding and alters subcellular localization. J. Virol. 2006, 80, 12070–12078. [Google Scholar] [CrossRef]
- Takimoto, T.; Portner, A. Molecular mechanism of paramyxovirus budding. Virus Res. 2004, 106, 133–145. [Google Scholar] [CrossRef]
- Harrison, M.S.; Sakaguchi, T.; Schmitt, A.P. Paramyxovirus assembly and budding: Building particles that transmit infections. Int. J. Biochem. Cell Biol. 2010, 42, 1416–1429. [Google Scholar] [CrossRef]
- Perez, M.; Craven, R.C.; de la Torre, J.C. The small RING finger protein Z drives arenavirus budding: Implications for antiviral strategies. Proc. Natl. Acad. Sci. U. S. A. 2003, 100, 12978–12983. [Google Scholar]
- Strecker, T.; Eichler, R.; Meulen, J.; Weissenhorn, W.; Dieter Klenk, H.; Garten, W.; Lenz, O. Lassa virus Z protein is a matrix protein and sufficient for the release of virus-like particles [corrected]. J. Virol. 2003, 77, 10700–10705. [Google Scholar]
- Urata, S.; Noda, T.; Kawaoka, Y.; Yokosawa, H.; Yasuda, J. Cellular factors required for Lassa virus budding. J. Virol. 2006, 80, 4191–4195. [Google Scholar] [CrossRef]
- Eichler, R.; Strecker, T.; Kolesnikova, L.; ter Meulen, J.; Weissenhorn, W.; Becker, S.; Klenk, H.D.; Garten, W.; Lenz, O. Characterization of the Lassa virus matrix protein Z: Electron microscopic study of virus-like particles and interaction with the nucleoprotein (NP). Virus Res. 2004, 100, 249–255. [Google Scholar] [CrossRef]
- Radoshitzky, S.R.; Dong, L.; Chi, X.; Clester, J.C.; Retterer, C.; Spurgers, K.; Kuhn, J.H.; Sandwick, S.; Ruthel, G.; Kota, K.; et al. Infectious Lassa virus, but not filoviruses, is restricted by BST-2/tetherin. J. Virol. 2010, 84, 10569–10580. [Google Scholar]
- Casabona, J.C.; Levingston Macleod, J.M.; Loureiro, M.E.; Gomez, G.A.; Lopez, N. The RING domain and the L79 residue of Z protein are involved in both the rescue of nucleocapsids and the incorporation of glycoproteins into infectious chimeric arenavirus-like particles. J. Virol. 2009, 83, 7029–7039. [Google Scholar] [CrossRef]
- Ghanam, R.H.; Samal, A.B.; Fernandez, T.F.; Saad, J.S. Role of the HIV-1 matrix protein in gag intracellular trafficking and targeting to the plasma membrane for virus assembly. Front. Microbiol. 2012, 3, 55. [Google Scholar]
- Chukkapalli, V.; Ono, A. Molecular determinants that regulate plasma membrane association of HIV-1 Gag. J. Mol. Biol. 2011, 410, 512–524. [Google Scholar] [CrossRef]
- Capul, A.A.; de la Torre, J.C.; Buchmeier, M.J. Conserved residues in Lassa fever virus Z protein modulate viral infectivity at the level of the ribonucleoprotein. J. Virol. 2011, 85, 3172–3178. [Google Scholar] [CrossRef]
- Levingston Macleod, J.M.; D'Antuono, A.; Loureiro, M.E.; Casabona, J.C.; Gomez, G.A.; Lopez, N. Identification of two functional domains within the arenavirus nucleoprotein. J. Virol. 2011, 85, 2012–2023. [Google Scholar]
- Loureiro, M.E.; Wilda, M.; Levingston Macleod, J.M.; D'Antuono, A.; Foscaldi, S.; Marino Buslje, C.; Lopez, N. Molecular determinants of arenavirus Z protein homo-oligomerization and L polymerase binding. J. Virol. 2011, 85, 12304–12314. [Google Scholar] [CrossRef]
- Ortiz-Riano, E.; Cheng, B.Y.; de la Torre, J.C.; Martinez-Sobrido, L. The C-terminal region of lymphocytic choriomeningitis virus nucleoprotein contains distinct and segregable functional domains involved in NP-Z interaction and counteraction of the type I interferon response. J. Virol. 2011, 85, 13038–13048. [Google Scholar] [CrossRef]
- Ortiz-Riano, E.; Cheng, B.Y.; de la Torre, J.C.; Martinez-Sobrido, L. Self-association of lymphocytic choriomeningitis virus nucleoprotein is mediated by its N-terminal region and is not required for its anti-interferon function. J. Virol. 2012, 86, 3307–3317. [Google Scholar]
- Ono, A.; Freed, E.O. Cell-type-dependent targeting of human immunodeficiency virus type 1 assembly to the plasma membrane and the multivesicular body. J. Virol. 2004, 78, 1552–1563. [Google Scholar] [CrossRef]
- Nydegger, S.; Foti, M.; Derdowski, A.; Spearman, P.; Thali, M. HIV-1 egress is gated through late endosomal membranes. Traffic 2003, 4, 902–910. [Google Scholar] [CrossRef]
- Sherer, N.M.; Lehmann, M.J.; Jimenez-Soto, L.F.; Ingmundson, A.; Horner, S.M.; Cicchetti, G.; Allen, P.G.; Pypaert, M.; Cunningham, J.M.; Mothes, W. Visualization of retroviral replication in living cells reveals budding into multivesicular bodies. Traffic 2003, 4, 785–801. [Google Scholar] [CrossRef]
- Perlman, M.; Resh, M.D. Identification of an intracellular trafficking and assembly pathway for HIV-1 gag. Traffic 2006, 7, 731–745. [Google Scholar] [CrossRef]
- Wirblich, C.; Tan, G.S.; Papaneri, A.; Godlewski, P.J.; Orenstein, J.M.; Harty, R.N.; Schnell, M.J. PPEY motif within the rabies virus (RV) matrix protein is essential for efficient virion release and RV pathogenicity. J. Virol. 2008, 82, 9730–9738. [Google Scholar] [CrossRef]
- Irie, T.; Licata, J.M.; Jayakar, H.R.; Whitt, M.A.; Bell, P.; Harty, R.N. Functional analysis of late-budding domain activity associated with the PSAP motif within the vesicular stomatitis virus M protein. J. Virol. 2004, 78, 7823–7827. [Google Scholar] [CrossRef]
- Taylor, G.M.; Hanson, P.I.; Kielian, M. Ubiquitin depletion and dominant-negative VPS4 inhibit rhabdovirus budding without affecting alphavirus budding. J. Virol. 2007, 81, 13631–13639. [Google Scholar] [CrossRef]
- Neumann, G.; Ebihara, H.; Takada, A.; Noda, T.; Kobasa, D.; Jasenosky, L.D.; Watanabe, S.; Kim, J.H.; Feldmann, H.; Kawaoka, Y. Ebola virus VP40 late domains are not essential for viral replication in cell culture. J. Virol. 2005, 79, 10300–10307. [Google Scholar]
- Pinschewer, D.D.; Perez, M.; de la Torre, J.C. Dual role of the lymphocytic choriomeningitis virus intergenic region in transcription termination and virus propagation. J. Virol. 2005, 79, 4519–4526. [Google Scholar] [CrossRef]
- Capul, A.A.; Perez, M.; Burke, E.; Kunz, S.; Buchmeier, M.J.; de la Torre, J.C. Arenavirus Z-glycoprotein association requires Z myristoylation but not functional RING or late domains. J. Virol. 2007, 81, 9451–9460. [Google Scholar] [CrossRef]
- Kolesnikova, L.; Strecker, T.; Morita, E.; Zielecki, F.; Mittler, E.; Crump, C.; Becker, S. Vacuolar protein sorting pathway contributes to the release of Marburg virus. J. Virol. 2009, 83, 2327–2337. [Google Scholar] [CrossRef]
- Dolnik, O.; Kolesnikova, L.; Stevermann, L.; Becker, S. Tsg101 is recruited by a late domain of the nucleocapsid protein to support budding of Marburg virus-like particles. J. Virol. 2010, 84, 7847–7856. [Google Scholar]
- Spiropoulou, C.F.; Kunz, S.; Rollin, P.E.; Campbell, K.P.; Oldstone, M.B. New World arenavirus clade C, but not clade A and B viruses, utilizes alpha-dystroglycan as its major receptor. J. Virol. 2002, 76, 5140–5146. [Google Scholar] [CrossRef]
- Radoshitzky, S.R.; Kuhn, J.H.; Spiropoulou, C.F.; Albarino, C.G.; Nguyen, D.P.; Salazar-Bravo, J.; Dorfman, T.; Lee, A.S.; Wang, E.; Ross, S.R.; et al. Receptor determinants of zoonotic transmission of New World hemorrhagic fever arenaviruses. Proc. Natl. Acad. Sci. U. S. A. 2008, 105, 2664–2669. [Google Scholar]
- Beyer, W.R.; Popplau, D.; Garten, W.; von Laer, D.; Lenz, O. Endoproteolytic processing of the lymphocytic choriomeningitis virus glycoprotein by the subtilase SKI-1/S1P. J. Virol. 2003, 77, 2866–2872. [Google Scholar] [CrossRef]
- Lenz, O.; ter Meulen, J.; Klenk, H.D.; Seidah, N.G.; Garten, W. The Lassa virus glycoprotein precursor GP-C is proteolytically processed by subtilase SKI-1/S1P. Proc. Natl. Acad. Sci. U. S. A. 2001, 98, 12701–12705. [Google Scholar]
- Wright, K.E.; Spiro, R.C.; Burns, J.W.; Buchmeier, M.J. Post-translational processing of the glycoproteins of lymphocytic choriomeningitis virus. Virology 1990, 177, 175–183. [Google Scholar] [CrossRef]
- Burri, D.J.; Pasqual, G.; Rochat, C.; Seidah, N.G.; Pasquato, A.; Kunz, S. Molecular characterization of the processing of arenavirus envelope glycoprotein precursors by subtilisin kexin isozyme-1/site-1 protease. J. Virol. 2012, 86, 4935–4946. [Google Scholar] [CrossRef]
- Rojek, J.M.; Lee, A.M.; Nguyen, N.; Spiropoulou, C.F.; Kunz, S. Site 1 protease is required for proteolytic processing of the glycoproteins of the South American hemorrhagic fever viruses Junin, Machupo, and Guanarito. J. Virol. 2008, 82, 6045–6051. [Google Scholar]
- Schlie, K.; Maisa, A.; Lennartz, F.; Stroher, U.; Garten, W.; Strecker, T. Characterization of Lassa virus glycoprotein oligomerization and influence of cholesterol on virus replication. J. Virol. 2010, 84, 983–992. [Google Scholar]
- Rojek, J.M.; Pasqual, G.; Sanchez, A.B.; Nguyen, N.T.; de la Torre, J.C.; Kunz, S. Targeting the proteolytic processing of the viral glycoprotein precursor is a promising novel antiviral strategy against arenaviruses. J. Virol. 2010, 84, 573–584. [Google Scholar]
- Urata, S.; Yun, N.; Pasquato, A.; Paessler, S.; Kunz, S.; de la Torre, J.C. Antiviral activity of a small-molecule inhibitor of arenavirus glycoprotein processing by the cellular site 1 protease. J. Virol. 2011, 85, 795–803. [Google Scholar] [CrossRef]
- Pasquato, A.; Rochat, C.; Burri, D.J.; Pasqual, G.; de la Torre, J.C.; Kunz, S. Evaluation of the anti-arenaviral activity of the subtilisin kexin isozyme-1/site-1 protease inhibitor PF-429242. Virology 2012, 423, 14–22. [Google Scholar] [CrossRef]
- Popkin, D.L.; Teijaro, J.R.; Sullivan, B.M.; Urata, S.; Rutschmann, S.; de la Torre, J.C.; Kunz, S.; Beutler, B.; Oldstone, M. Hypomorphic mutation in the site-1 protease Mbtps1 endows resistance to persistent viral infection in a cell-specific manner. Cell Host Microbe 2011, 9, 212–222. [Google Scholar] [CrossRef]
- Maisa, A.; Stroher, U.; Klenk, H.D.; Garten, W.; Strecker, T. Inhibition of Lassa virus glycoprotein cleavage and multicycle replication by site 1 protease-adapted alpha(1)-antitrypsin variants. PLoS Negl. Trop. Dis. 2009, 3, e446. [Google Scholar] [CrossRef]
- Urata, S.; Ngo, N.; de la Torre, J.C. The PI3K/Akt pathway contributes to arenavirus budding. J. Virol. 2012, 86, 4578–4585. [Google Scholar] [CrossRef]
- Saunders, A.A.; Ting, J.P.; Meisner, J.; Neuman, B.W.; Perez, M.; de la Torre, J.C.; Buchmeier, M.J. Mapping the landscape of the lymphocytic choriomeningitis virus stable signal peptide reveals novel functional domains. J. Virol. 2007, 81, 5649–5657. [Google Scholar]
- Messina, E.L.; York, J.; Nunberg, J.H. Dissection of the role of the stable signal peptide of the arenavirus envelope glycoprotein in membrane fusion. J. Virol. 2012. [Google Scholar]
- Eichler, R.; Lenz, O.; Strecker, T.; Eickmann, M.; Klenk, H.D.; Garten, W. Identification of Lassa virus glycoprotein signal peptide as a trans-acting maturation factor. EMBO Rep. 2003, 4, 1084–1088. [Google Scholar] [CrossRef]
- Messina, E.L.; York, J.; Nunberg, J.H. Dissection of the role of the stable signal Peptide of the arenavirus envelope glycoprotein in membrane fusion. J. Virol. 2012, 86, 6138–6145. [Google Scholar] [CrossRef]
- Bonhomme, C.J.; Capul, A.A.; Lauron, E.J.; Bederka, L.H.; Knopp, K.A.; Buchmeier, M.J. Glycosylation modulates arenavirus glycoprotein expression and function. Virology 2011, 409, 223–233. [Google Scholar] [CrossRef]
- Eichler, R.; Lenz, O.; Garten, W.; Strecker, T. The role of single N-glycans in proteolytic processing and cell surface transport of the Lassa virus glycoprotein GP-C. Virol. J. 2006, 3, 41. [Google Scholar] [CrossRef]
- Inlora, J.; Chukkapalli, V.; Derse, D.; Ono, A. Gag localization and virus-like particle release mediated by the matrix domain of human T-lymphotropic virus type 1 Gag are less dependent on phosphatidylinositol-(4,5)-bisphosphate than those mediated by the matrix domain of HIV-1 Gag. J. Virol. 2011, 85, 3802–3810. [Google Scholar] [CrossRef]
- Hogue, I.B.; Grover, J.R.; Soheilian, F.; Nagashima, K.; Ono, A. Gag induces the coalescence of clustered lipid rafts and tetraspanin-enriched microdomains at HIV-1 assembly sites on the plasma membrane. J. Virol. 2011, 85, 9749–9766. [Google Scholar] [CrossRef]
- Saad, J.S.; Miller, J.; Tai, J.; Kim, A.; Ghanam, R.H.; Summers, M.F. Structural basis for targeting HIV-1 Gag proteins to the plasma membrane for virus assembly. Proc. Natl. Acad. Sci. U. S. A. 2006, 103, 11364–11369. [Google Scholar]
- Saad, J.S.; Loeliger, E.; Luncsford, P.; Liriano, M.; Tai, J.; Kim, A.; Miller, J.; Joshi, A.; Freed, E.O.; Summers, M.F. Point mutations in the HIV-1 matrix protein turn off the myristyl switch. J. Mol. Biol. 2007, 366, 574–585. [Google Scholar]
- Provitera, P.; El-Maghrabi, R.; Scarlata, S. The effect of HIV-1 Gag myristoylation on membrane binding. Biophys. Chem. 2006, 119, 23–32. [Google Scholar] [CrossRef]
- Seaton, K.E.; Smith, C.D. N-Myristoyltransferase isozymes exhibit differential specificity for human immunodeficiency virus type 1 Gag and Nef. J. Gen. Virol. 2008, 89, 288–296. [Google Scholar] [CrossRef]
- Fledderman, E.L.; Fujii, K.; Ghanam, R.H.; Waki, K.; Prevelige, P.E.; Freed, E.O.; Saad, J.S. Myristate exposure in the human immunodeficiency virus type 1 matrix protein is modulated by pH. Biochemistry 2010, 49, 9551–9562. [Google Scholar]
- Ghanam, R.H.; Fernandez, T.F.; Fledderman, E.L.; Saad, J.S. Binding of calmodulin to the HIV-1 matrix protein triggers myristate exposure. J. Biol. Chem. 2010, 285, 41911–41920. [Google Scholar]
- Samal, A.B.; Ghanam, R.H.; Fernandez, T.F.; Monroe, E.B.; Saad, J.S. NMR, biophysical, and biochemical studies reveal the minimal Calmodulin binding domain of the HIV-1 matrix protein. J. Biol. Chem. 2011, 286, 33533–33543. [Google Scholar]
- Alfadhli, A.; Still, A.; Barklis, E. Analysis of human immunodeficiency virus type 1 matrix binding to membranes and nucleic acids. J. Virol. 2009, 83, 12196–12203. [Google Scholar]
- Li, H.; Dou, J.; Ding, L.; Spearman, P. Myristoylation is required for human immunodeficiency virus type 1 Gag-Gag multimerization in mammalian cells. J. Virol. 2007, 81, 12899–12910. [Google Scholar] [CrossRef]
- Dou, J.; Wang, J.J.; Chen, X.; Li, H.; Ding, L.; Spearman, P. Characterization of a myristoylated, monomeric HIV Gag protein. Virology 2009, 387, 341–352. [Google Scholar] [CrossRef]
- Strecker, T.; Maisa, A.; Daffis, S.; Eichler, R.; Lenz, O.; Garten, W. The role of myristoylation in the membrane association of the Lassa virus matrix protein Z. Virol. J. 2006, 3, 93. [Google Scholar] [CrossRef]
- Perez, M.; Greenwald, D.L.; de la Torre, J.C. Myristoylation of the RING finger Z protein is essential for arenavirus budding. J. Virol. 2004, 78, 11443–11448. [Google Scholar] [CrossRef]
- Liu, P.; Cheng, H.; Roberts, T.M.; Zhao, J.J. Targeting the phosphoinositide 3-kinase pathway in cancer. Nat. Rev. Drug Discov. 2009, 8, 627–644. [Google Scholar] [CrossRef]
- Morita, E. Differential requirements of mammalian ESCRTs in multivesicular body formation, virus budding and cell division. FEBS J. 2012, 279, 1399–1406. [Google Scholar] [CrossRef]
- Chen, B.J.; Lamb, R.A. Mechanisms for enveloped virus budding: Can some viruses do without an ESCRT? Virology 2008, 372, 221–232. [Google Scholar] [CrossRef]
- Irie, T.; Licata, J.M.; McGettigan, J.P.; Schnell, M.J.; Harty, R.N. Budding of PPxY-containing rhabdoviruses is not dependent on host proteins TGS101 and VPS4A. J. Virol. 2004, 78, 2657–2665. [Google Scholar] [CrossRef]
- Langelier, C.; von Schwedler, U.K.; Fisher, R.D.; De Domenico, I.; White, P.L.; Hill, C.P.; Kaplan, J.; Ward, D.; Sundquist, W.I. Human ESCRT-II complex and its role in human immunodeficiency virus type 1 release. J. Virol. 2006, 80, 9465–9480. [Google Scholar]
- Strack, B.; Calistri, A.; Craig, S.; Popova, E.; Gottlinger, H.G. AIP1/ALIX is a binding partner for HIV-1 p6 and EIAV p9 functioning in virus budding. Cell 2003, 114, 689–699. [Google Scholar] [CrossRef]
- Fujii, K.; Hurley, J.H.; Freed, E.O. Beyond Tsg101: The role of Alix in 'ESCRTing' HIV-1. Nat. Rev. Microbiol. 2007, 5, 912–916. [Google Scholar] [CrossRef]
- Morita, E.; Sandrin, V.; McCullough, J.; Katsuyama, A.; Baci Hamilton, I.; Sundquist, W.I. ESCRT-III protein requirements for HIV-1 budding. Cell Host Microbe 2011, 9, 235–242. [Google Scholar] [CrossRef]
- Urata, S.; Yasuda, J. Regulation of Marburg virus (MARV) budding by Nedd4.1: A different WW domain of Nedd4.1 is critical for binding to MARV and Ebola virus VP40. J. Gen. Virol. 2010, 91, 228–234. [Google Scholar] [CrossRef]
- Yasuda, J.; Nakao, M.; Kawaoka, Y.; Shida, H. Nedd4 regulates egress of Ebola virus-like particles from host cells. J. Virol. 2003, 77, 9987–9992. [Google Scholar] [CrossRef]
- Sakurai, A.; Yasuda, J.; Takano, H.; Tanaka, Y.; Hatakeyama, M.; Shida, H. Regulation of human T-cell leukemia virus type 1 (HTLV-1) budding by ubiquitin ligase Nedd4. Microb. Infect. 2004, 6, 150–156. [Google Scholar] [CrossRef]
- Heidecker, G.; Lloyd, P.A.; Fox, K.; Nagashima, K.; Derse, D. Late assembly motifs of human T-cell leukemia virus type 1 and their relative roles in particle release. J. Virol. 2004, 78, 6636–6648. [Google Scholar] [CrossRef]
- Heidecker, G.; Lloyd, P.A.; Soheilian, F.; Nagashima, K.; Derse, D. The role of WWP1-Gag interaction and Gag ubiquitination in assembly and release of human T-cell leukemia virus type 1. J. Virol. 2007, 81, 9769–9777. [Google Scholar]
- Dorjbal, B.; Derse, D.; Lloyd, P.; Soheilian, F.; Nagashima, K.; Heidecker, G. The role of ITCH protein in human T-cell leukemia virus type 1 release. J. Biol. Chem. 2011, 286, 31092–31104. [Google Scholar]
- Kikonyogo, A.; Bouamr, F.; Vana, M.L.; Xiang, Y.; Aiyar, A.; Carter, C.; Leis, J. Proteins related to the Nedd4 family of ubiquitin protein ligases interact with the L domain of Rous sarcoma virus and are required for gag budding from cells. Proc. Natl. Acad. Sci. U. S. A. 2001, 98, 11199–11204. [Google Scholar]
- Yasuda, J.; Hunter, E.; Nakao, M.; Shida, H. Functional involvement of a novel Nedd4-like ubiquitin ligase on retrovirus budding. EMBO Rep. 2002, 3, 636–640. [Google Scholar] [CrossRef]
- Martin-Serrano, J.; Eastman, S.W.; Chung, W.; Bieniasz, P.D. HECT ubiquitin ligases link viral and cellular PPXY motifs to the vacuolar protein-sorting pathway. J. Cell Biol. 2005, 168, 89–101. [Google Scholar]
- Usami, Y.; Popov, S.; Popova, E.; Gottlinger, H.G. Efficient and specific rescue of human immunodeficiency virus type 1 budding defects by a Nedd4-like ubiquitin ligase. J. Virol. 2008, 82, 4898–4907. [Google Scholar]
- Chung, H.Y.; Morita, E.; von Schwedler, U.; Muller, B.; Krausslich, H.G.; Sundquist, W.I. NEDD4L overexpression rescues the release and infectivity of human immunodeficiency virus type 1 constructs lacking PTAP and YPXL late domains. J. Virol. 2008, 82, 4884–4897. [Google Scholar] [CrossRef]
- Rauch, S.; Martin-Serrano, J. Multiple interactions between the ESCRT machinery and arrestin-related proteins: Implications for PPXY-dependent budding. J. Virol. 2011, 85, 3546–3556. [Google Scholar] [CrossRef]
- Borrow, P.; Martinez-Sobrido, L.; de la Torre, J.C. Inhibition of the type I interferon antiviral response during arenavirus infection. Viruses 2010, 2, 2443–2480. [Google Scholar] [CrossRef]
- Versteeg, G.A.; Garcia-Sastre, A. Viral tricks to grid-lock the type I interferon system. Curr. Opin. Microbiol. 2010, 13, 508–516. [Google Scholar] [CrossRef]
- Malim, M.H.; Bieniasz, P.D. HIV Restriction factors and mechanisms of evasion. Cold Spring Harb. Perspect. Med. 2012, 2, a006940. [Google Scholar]
- Nakayama, E.E.; Shioda, T. TRIM5alpha and species tropism of HIV/SIV. Front. Microbiol. 2012, 3, 13. [Google Scholar]
- Stremlau, M.; Owens, C.M.; Perron, M.J.; Kiessling, M.; Autissier, P.; Sodroski, J. The cytoplasmic body component TRIM5alpha restricts HIV-1 infection in Old World monkeys. Nature 2004, 427, 848–853. [Google Scholar] [CrossRef]
- Sakuma, R.; Noser, J.A.; Ohmine, S.; Ikeda, Y. Rhesus monkey TRIM5alpha restricts HIV-1 production through rapid degradation of viral Gag polyproteins. Nat. Med. 2007, 13, 631–635. [Google Scholar] [CrossRef]
- Sakuma, R.; Ohmine, S.; Ikeda, Y. Determinants for the rhesus monkey TRIM5alpha-mediated block of the late phase of HIV-1 replication. J. Biol. Chem. 2010, 285, 3784–3793. [Google Scholar] [CrossRef]
- Schoggins, J.W.; Wilson, S.J.; Panis, M.; Murphy, M.Y.; Jones, C.T.; Bieniasz, P.; Rice, C.M. A diverse range of gene products are effectors of the type I interferon antiviral response. Nature 2011, 472, 481–485. [Google Scholar]
- Liu, S.Y.; Sanchez, D.J.; Aliyari, R.; Lu, S.; Cheng, G. Systematic identification of type I and type II interferon-induced antiviral factors. Proc. Natl. Acad. Sci. U. S. A. 2012, 109, 4239–4244. [Google Scholar]
- Okumura, A.; Lu, G.; Pitha-Rowe, I.; Pitha, P.M. Innate antiviral response targets HIV-1 release by the induction of ubiquitin-like protein ISG15. Proc. Natl. Acad. Sci. U. S. A. 2006, 103, 1440–1445. [Google Scholar]
- Okumura, A.; Pitha, P.M.; Harty, R.N. ISG15 inhibits Ebola VP40 VLP budding in an L-domain-dependent manner by blocking Nedd4 ligase activity. Proc. Natl. Acad. Sci. U. S. A. 2008, 105, 3974–3979. [Google Scholar]
- Seo, E.J.; Leis, J. Budding of enveloped viruses: Interferon-induced ISG15-antivirus mechanisms targeting the release process. Adv. Virol. 2012, 2012, 532723. [Google Scholar]
- Kuang, Z.; Seo, E.J.; Leis, J. Mechanism of inhibition of retrovirus release from cells by interferon-induced gene ISG15. J. Virol. 2011, 85, 7153–7161. [Google Scholar] [CrossRef]
- Pincetic, A.; Kuang, Z.; Seo, E.J.; Leis, J. The interferon-induced gene ISG15 blocks retrovirus release from cells late in the budding process. J. Virol. 2010, 84, 4725–4736. [Google Scholar] [CrossRef]
- Garrus, J.E.; von Schwedler, U.K.; Pornillos, O.W.; Morham, S.G.; Zavitz, K.H.; Wang, H.E.; Wettstein, D.A.; Stray, K.M.; Cote, M.; Rich, R.L.; et al. Tsg101 and the vacuolar protein sorting pathway are essential for HIV-1 budding. Cell 2001, 107, 55–65. [Google Scholar] [CrossRef]
- Martin-Serrano, J. The role of ubiquitin in retroviral egress. Traffic 2007, 8, 1297–1303. [Google Scholar] [CrossRef]
- Van Damme, N.; Goff, D.; Katsura, C.; Jorgenson, R.L.; Mitchell, R.; Johnson, M.C.; Stephens, E.B.; Guatelli, J. The interferon-induced protein BST-2 restricts HIV-1 release and is downregulated from the cell surface by the viral Vpu protein. Cell Host Microbe 2008, 3, 245–252. [Google Scholar] [CrossRef]
- Neil, S.J.; Zang, T.; Bieniasz, P.D. Tetherin inhibits retrovirus release and is antagonized by HIV-1 Vpu. Nature 2008, 451, 425–430. [Google Scholar] [CrossRef]
- Sakuma, T.; Noda, T.; Urata, S.; Kawaoka, Y.; Yasuda, J. Inhibition of Lassa and Marburg virus production by tetherin. J. Virol. 2009, 83, 2382–2385. [Google Scholar] [CrossRef]
- Le Tortorec, A.; Willey, S.; Neil, S.J. Antiviral inhibition of enveloped virus release by tetherin/BST-2: Action and counteraction. Viruses 2011, 3, 520–540. [Google Scholar]
- Ohtomo, T.; Sugamata, Y.; Ozaki, Y.; Ono, K.; Yoshimura, Y.; Kawai, S.; Koishihara, Y.; Ozaki, S.; Kosaka, M.; Hirano, T.; et al. Molecular cloning and characterization of a surface antigen preferentially overexpressed on multiple myeloma cells. Biochem. Biophys. Res. Commun. 1999, 258, 583–591. [Google Scholar] [CrossRef]
- Wong, Y.F.; Cheung, T.H.; Lo, K.W.; Yim, S.F.; Siu, N.S.; Chan, S.C.; Ho, T.W.; Wong, K.W.; Yu, M.Y.; Wang, V.W.; et al. Identification of molecular markers and signaling pathway in endometrial cancer in Hong Kong Chinese women by genome-wide gene expression profiling. Oncogene 2007, 26, 1971–1982. [Google Scholar] [CrossRef]
- Wang, W.; Nishioka, Y.; Ozaki, S.; Jalili, A.; Abe, S.; Kakiuchi, S.; Kishuku, M.; Minakuchi, K.; Matsumoto, T.; Sone, S. HM1.24 (CD317) is a novel target against lung cancer for immunotherapy using anti-HM1.24 antibody. Cancer Immunol. Immunother. 2009, 58, 967–976. [Google Scholar] [CrossRef]
- Wainwright, D.A.; Balyasnikova, I.V.; Han, Y.; Lesniak, M.S. The expression of BST2 in human and experimental mouse brain tumors. Exp. Mol. Pathol. 2011, 91, 440–446. [Google Scholar] [CrossRef]
- Wang, W.; Nishioka, Y.; Ozaki, S.; Jalili, A.; Verma, V.K.; Hanibuchi, M.; Abe, S.; Minakuchi, K.; Matsumoto, T.; Sone, S. Chimeric and humanized anti-HM1.24 antibodies mediate antibody-dependent cellular cytotoxicity against lung cancer cells. Lung Cancer 2009, 63, 23–31. [Google Scholar] [CrossRef]
- Swiecki, M.; Scheaffer, S.M.; Allaire, M.; Fremont, D.H.; Colonna, M.; Brett, T.J. Structural and biophysical analysis of BST-2/tetherin ectodomains reveals an evolutionary conserved design to inhibit virus release. J. Biol. Chem. 2011, 286, 2987–2997. [Google Scholar]
- Schubert, H.L.; Zhai, Q.; Sandrin, V.; Eckert, D.M.; Garcia-Maya, M.; Saul, L.; Sundquist, W.I.; Steiner, R.A.; Hill, C.P. Structural and functional studies on the extracellular domain of BST2/tetherin in reduced and oxidized conformations. Proc. Natl. Acad. Sci. U. S. A. 2010, 107, 17951–17956. [Google Scholar]
- Hinz, A.; Miguet, N.; Natrajan, G.; Usami, Y.; Yamanaka, H.; Renesto, P.; Hartlieb, B.; McCarthy, A.A.; Simorre, J.P.; Gottlinger, H.; et al. Structural basis of HIV-1 tethering to membranes by the BST-2/tetherin ectodomain. Cell Host Microbe 2010, 7, 314–323. [Google Scholar] [CrossRef]
- Yang, H.; Wang, J.; Jia, X.; McNatt, M.W.; Zang, T.; Pan, B.; Meng, W.; Wang, H.W.; Bieniasz, P.D.; Xiong, Y. Structural insight into the mechanisms of enveloped virus tethering by tetherin. Proc. Natl. Acad. Sci. U. S. A. 2010, 107, 18428–18432. [Google Scholar]
- Arias, J.F.; Iwabu, Y.; Tokunaga, K. Structural basis for the antiviral activity of BST-2/Tetherin and its viral antagonism. Front. Microbiol. 2011, 2, 250. [Google Scholar]
- Perez-Caballero, D.; Zang, T.; Ebrahimi, A.; McNatt, M.W.; Gregory, D.A.; Johnson, M.C.; Bieniasz, P.D. Tetherin inhibits HIV-1 release by directly tethering virions to cells. Cell 2009, 139, 499–511. [Google Scholar] [CrossRef]
- Hammonds, J.; Wang, J.J.; Yi, H.; Spearman, P. Immunoelectron microscopic evidence for Tetherin/BST2 as the physical bridge between HIV-1 virions and the plasma membrane. PLoS Pathog. 2010, 6, e1000749. [Google Scholar] [CrossRef]
- Basler, C.F.; Amarasinghe, G.K. Evasion of interferon responses by Ebola and Marburg viruses. J. Interferon Cytokine Res. 2009, 29, 511–520. [Google Scholar] [CrossRef]
- Garcia-Sastre, A. Induction and evasion of type I interferon responses by influenza viruses. Virus Res. 2011, 162, 12–18. [Google Scholar] [CrossRef]
- Kuhl, B.D.; Cheng, V.; Wainberg, M.A.; Liang, C. Tetherin and its viral antagonists. J. Neuroimmune Pharmacol. 2011, 6, 188–201. [Google Scholar] [CrossRef]
- Yasuda, J. Ebolavirus Replication and Tetherin/BST-2. Front. Microbiol. 2012, 3, 111. [Google Scholar]
- Sato, K.; Gee, P.; Koyanagi, Y. Vpu and BST2: Still not there yet? Front. Microbiol. 2012, 3, 131. [Google Scholar]
- Sakuma, T.; Sakurai, A.; Yasuda, J. Dimerization of tetherin is not essential for its antiviral activity against Lassa and Marburg viruses. PLoS One 2009, 4, e6934. [Google Scholar]
- Liberatore, R.A.; Bieniasz, P.D. Tetherin is a key effector of the antiretroviral activity of type I interferon in vitro and in vivo. Proc. Natl. Acad. Sci. U. S. A. 2011, 108, 18097–18101. [Google Scholar] [CrossRef]
- Jones, P.H.; Mehta, H.V.; Maric, M.; Roller, R.J.; Okeoma, C.M. Bone marrow stromal cell antigen 2 (BST-2) restricts mouse mammary tumor virus (MMTV) replication in vivo. Retrovirology 2012, 9, 10. [Google Scholar] [CrossRef]
- Swiecki, M.; Wang, Y.; Gilfillan, S.; Lenschow, D.J.; Colonna, M. Cutting edge: Paradoxical roles of BST2/tetherin in promoting type I IFN response and viral infection. J. Immunol. 2012, 188, 2488–2492. [Google Scholar] [CrossRef]
- Sato, K.; Misawa, N.; Fukuhara, M.; Iwami, S.; An, D.S.; Ito, M.; Koyanagi, Y. Vpu augments the initial burst phase of HIV-1 propagation and downregulates BST2 and CD4 in humanized mice. J. Virol. 2012, 86, 5000–5013. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).