HAS-Flow May Be an Adequate Method for Evaluating Human T-Cell Leukemia Virus Type 1 Infected Cells in Human T-Cell Leukemia Virus Type 1-Positive Rheumatoid Arthritis Patients Receiving Antirheumatic Therapies: A Retrospective Cross-Sectional Observation Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Participants
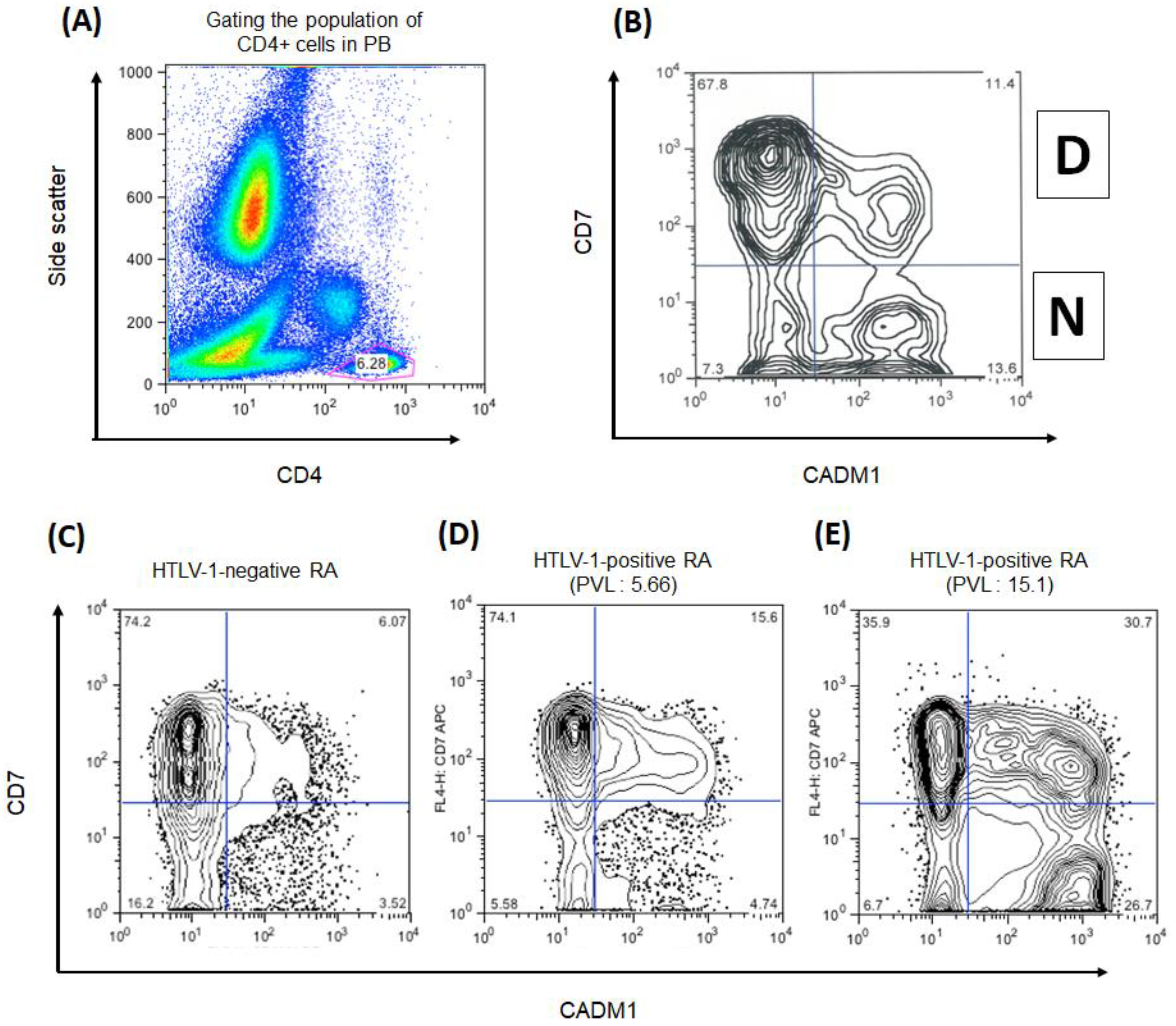
2.2. HAS-Flow
2.3. The Measurement of HTLV-1 PVL
2.4. Clinical Assessment of RA
2.5. Statistical Analysis
3. Results
3.1. Characteristics of HTLV-1-Negative and HTLV-1-Positive RA Participants
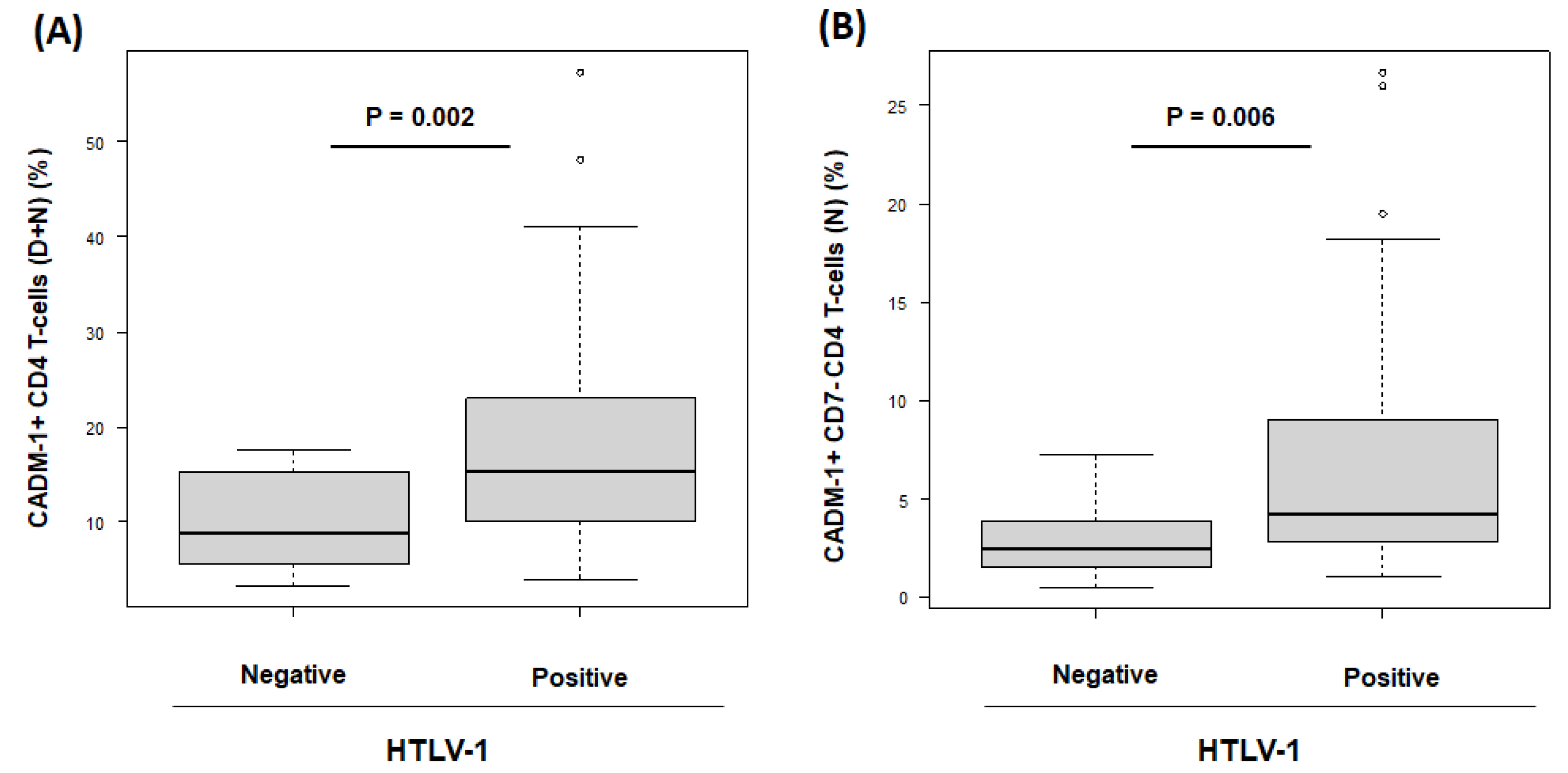
3.2. The Population of CADM1+ CD4+ Cells in HTLV-1-Negative and HTLV-1-Positive RA Participants
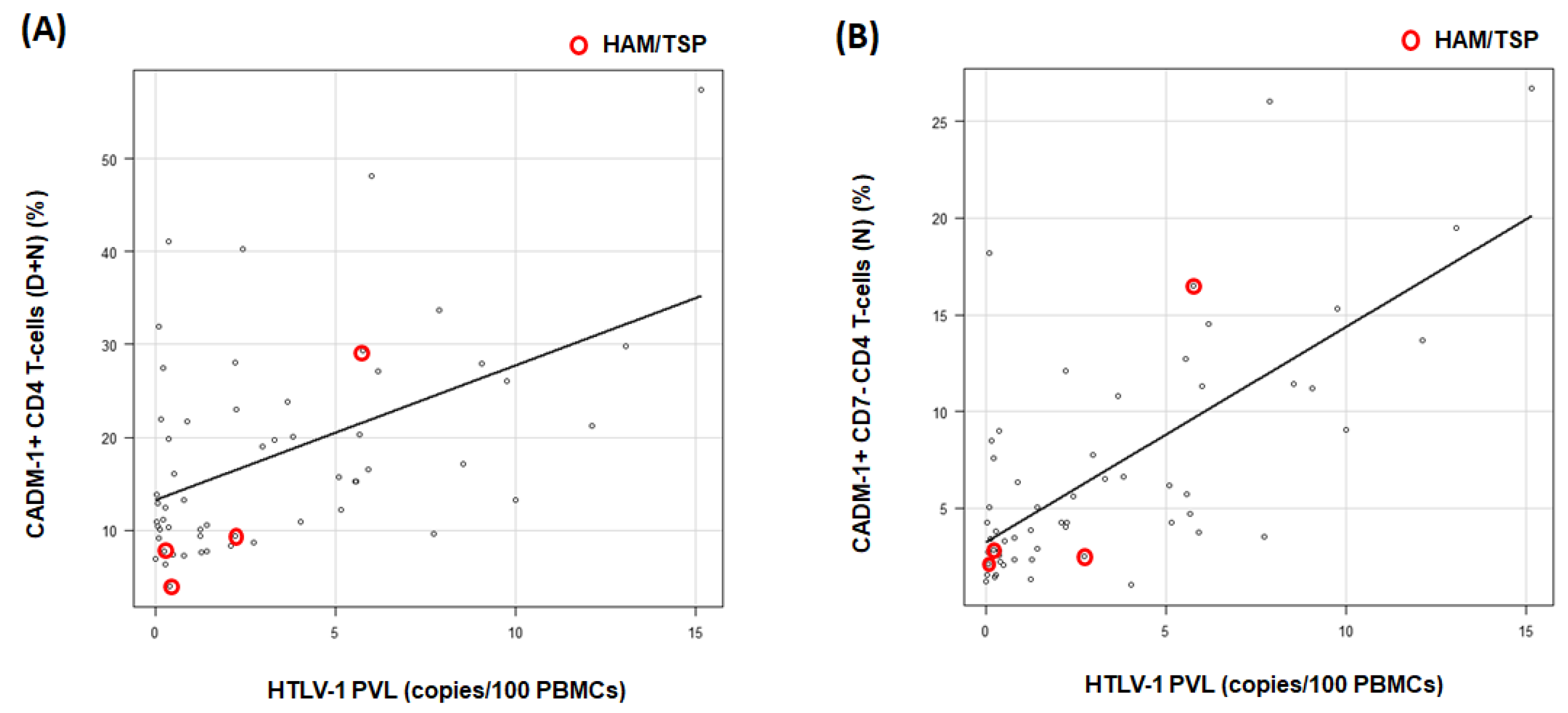
3.3. The Correlation between Population of CADM1+ CD4+ Cells and HTLV-1 PVL in HTLV-1-Positive RA Participants
3.4. Assessment of High-Risk RA Participants for ATL Development Based on HAS-Flow Analysis and HTLV-1 PVL Values
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Burmester, G.R.; Pope, J.E. Novel treatment strategies in rheumatoid arthritis. Lancet 2017, 389, 2338–2348. [Google Scholar] [CrossRef] [PubMed]
- Singh, J.A.; Cameron, C.; Noorbaloochi, S.; Cullis, T.; Tucker, M.; Christensen, R.; Ghogomu, E.T.; Coyle, D.; Clifford, T.; Tugwell, P.; et al. Risk of serious infection in biological treatment of patients with rheumatoid arthritis: A systematic review and meta-analysis. Lancet 2015, 386, 258–265. [Google Scholar] [CrossRef] [PubMed]
- Sepriano, A.; Kerschbaumer, A.; Smolen, J.S.; van der Heijde, D.; Dougados, M.; van Vollenhoven, R.; McInnes, I.B.; Bijlsma, J.W.; Burmester, G.R.; de Wit, M.; et al. Safety of synthetic and biological DMARDs: A systematic literature review informing the 2019 update of the EULAR recommendations for the management of rheumatoid arthritis. Ann. Rheum. Dis. 2020, 79, 760–770. [Google Scholar] [CrossRef] [PubMed]
- Satake, M.; Yamaguchi, K.; Tadokoro, K. Current prevalence of HTLV-1 in Japan as determined by screening of blood donors. J. Med. Virol. 2012, 84, 327–335. [Google Scholar] [CrossRef] [PubMed]
- Satake, M.; Iwanaga, M.; Sagara, Y.; Watanabe, T.; Okuma, K.; Hamaguchi, I. Incidence of human T-lymphotropic virus 1 infection in adolescent and adult blood donors in Japan: A nationwide retrospective cohort analysis. Lancet Infect. Dis. 2016, 16, 1246–1254. [Google Scholar] [CrossRef]
- Iwanaga, M.; Watanabe, T.; Utsunomiya, A.; Okayama, A.; Uchimaru, K.; Koh, K.R.; Ogata, M.; Kikuchi, H.; Sagara, Y.; Uozumi, K.; et al. Joint Study on Predisposing Factors of ATL Development investigators. Human T-cell leukemia virus type I (HTLV-1) proviral load and disease progression in asymptomatic HTLV-1 carriers: A nationwide prospective study in Japan. Blood 2010, 116, 1211–1219. [Google Scholar] [CrossRef]
- Firouzi, S.; Farmanbar, A.; Nakai, K.; Iwanaga, M.; Uchimaru, K.; Utsunomiya, A.; Suzuki, Y.; Watanabe, T. Clonality of HTLV-1-infected T cells as a risk indicator for development and progression of adult T-cell leukemia. Blood Adv. 2017, 1, 1195–1205. [Google Scholar] [CrossRef]
- Arisaw, K.; Katamine, S.; Kamihira, S.; Kurokawa, K.; Sawada, T.; Soda, M.; Doi, H.; Saito, H.; Shirahama, S. A nested case-control study of risk factors for adult T-cell leukemia/lymphoma among human T-cell lymphotropic virus type-I carriers in Japan. Cancer Causes Control 2002, 13, 657–663. [Google Scholar] [CrossRef]
- Kobayashi, S.; Nakano, K.; Watanabe, E.; Ishigaki, T.; Ohno, N.; Yuji, K.; Oyaizu, N.; Asanuma, S.; Yamagishi, M.; Yamochi, T.; et al. CADM1 expression and stepwise downregulation of CD7 are closely associated with clonal expansion of HTLV-I-infected cells in adult T-cell leukemia/lymphoma. Clin. Cancer Res. 2014, 20, 2851–2861. [Google Scholar] [CrossRef]
- Nagasaka, M.; Yamagishi, M.; Yagishita, N.; Araya, N.; Kobayashi, S.; Makiyama, J.; Kubokawa, M.; Yamauchi, J.; Hasegawa, D.; Coler-Reilly, A.L.G.; et al. Mortality and risk of progression to adult T cell leukemia/lymphoma in HTLV-1-associated myelopathy/tropical spastic paraparesis. Proc. Natl. Acad. Sci. USA 2020, 117, 11685–11691. [Google Scholar] [CrossRef]
- Kobayashi, S.; Watanabe, E.; Ishigaki, T.; Ohno, N.; Yuji, K.; Nakano, K.; Yamochi, T.; Watanabe, N.; Tojo, A.; Watanabe, T.; et al. Advanced human T-cell leukemia virus type 1 carriers and early-stage indolent adult T-cell leukemia-lymphoma are indistinguishable based on CADM1 positivity in flow cytometry. Cancer Sci. 2015, 106, 598–603. [Google Scholar] [CrossRef]
- Umekita, K.; Hashiba, Y.; Kariya, Y.; Kubo, K.; Miyauchi, S.; Aizawa, A.; Umeki, K.; Nomura, H.; Kawaguchi, T.; Matsuda, M.; et al. The time-sequential changes of risk factors for adult T-cell leukemia development in human T-cell leukemia virus-positive patients with rheumatoid arthritis: A retrospective cohort study. Mod. Rheumatol. 2019, 29, 795–801. [Google Scholar] [CrossRef]
- Arnett, F.C.; Edworthy, S.M.; Bloch, D.A.; McShane, D.J.; Fries, J.F.; Cooper, N.S.; Healey, L.A.; Kaplan, S.R.; Liang, M.H.; Luthra, H.S.; et al. The American Rheumatism Association 1987 revised criteria for the classification of rheumatoid arthritis. Arthritis Rheum. Am. Rheum. Assoc. 1988, 31, 315–324. [Google Scholar] [CrossRef]
- Aletaha, D.; Neogi, T.; Silman, A.J.; Funovits, J.; Felson, D.T.; Bingham, O.C., 3rd; Birnbaum, N.S.; Burmester, G.R.; Bykerk, V.P.; Cohen, M.D.; et al. 2010 Rheumatoid arthritis classification criteria: An American College of Rheumatology/European League Against Rheumatism collaborative initiative. Arthritis Rheum. 2010, 62, 2569–2581. [Google Scholar] [CrossRef]
- Singh, J.A.; Saag, K.G.; Bridges, S.L.J.r.; Akl, E.A.; Bannuru, R.R.; Sullivan, M.C.; Vaysbrot, E.; McNaughton, C.; Osani, M.; Shmerling, R.H.; et al. 2015 American College of Rheumatology guideline for the Treatment of rheumatoid arthritis. Arthritis Care Res. 2016, 68, 1–25. [Google Scholar] [CrossRef]
- Smolen, J.S.; Landewé, R.; Bijlsma, J.; Burmester, G.; Chatzidionysiou, K.; Dougados, M.; Nam, J.; Ramiro, S.; Voshaar, M.; van Vollenhoven, R.; et al. EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifyingantirheumatic drugs: 2016 update. Ann. Rheum. Dis. 2017, 76, 960–977. [Google Scholar] [CrossRef]
- Smolen, J.S.; Landewé, R.B.M.; Bijlsma, J.W.J.; Burmester, G.R.; Dougados, M.; Kerschbaumer, A.; McInnes, I.B.; Sepriano, A.; van Vollenhoven, R.F.; de Wit, M.; et al. EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifyingantirheumatic drugs: 2019 update. Ann. Rheum. Dis. 2020, 79, 685–699. [Google Scholar] [CrossRef]
- Tanaka, G.; Okayama, A.; Watanabe, T.; Aizawa, S.; Stuver, S.; Mueller, N.; Hsieh, C.C.; Tsubouchi, H. The clonal expansion of human T lymphotropic virus type 1-infected T cells: A comparison between seroconverters and long-term carriers. J. Infect. Dis. 2005, 191, 1140–1147. [Google Scholar] [CrossRef]
- Anderson, J.K.; Zimmerman, L.; Caplan, L.; Michaud, K. Measures of rheumatoid arthritis disease activity: Patient (PtGA) and Provider (PrGA) Global Assessment of Disease Activity, Disease Activity Score (DAS) and Disease Activity Score with 28-Joint Counts (DAS28), simplified disease activity index (SDAI), Clinical Disease Activity Index (CDAI), Patient Activity Score (PAS) and Patient Activity Score-II (PASII), Routine Assessment of Patient Index Data (RAPID), Rheumatoid Arthritis Disease Activity Index (RADAI) and Rheumatoid Arthritis Disease Activity Index-5 (RADAI-5), Chronic Arthritis Systemic Index (CASI), Patient-Based Disease Activity Score with ESR (PDAS1) and Patient-Based Disease Activity Score without ESR (PDAS2), and Mean Overall Index for Rheumatoid Arthritis (MOI-RA). Arthritis Care Res. 2011, 63 (Suppl. S11), S14–S36. [Google Scholar]
- Kanda, Y. Investigation of the freely available easy-to-use software ‘EZR’ for medical statistics. Bone Marrow Transplant. 2013, 48, 452–458. [Google Scholar] [CrossRef]
- Kuramochi, M.; Fukuhara, H.; Nobukuni, T.; Kanbe, T.; Maruyama, T.; Ghosh, H.P.; Pletcher, M.; Isomura, M.; Onizuka, M.; Kitamura, T.; et al. TSLC1 is a tumor-suppressor gene in human non-small-cell lung cancer. Nat. Genet. 2001, 27, 427–430. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, H.; Nishikata, I.; Shiraga, T.; Akamatsu, E.; Fukami, T.; Hidaka, T.; Kubuki, Y.; Okayama, A.; Hamada, K.; Okabe, H.; et al. Overexpression of a cell adhesion molecule, TSLC1, as a possible molecular marker for acute-type adult T-cell leukemia. Blood 2005, 105, 1204–1213. [Google Scholar] [CrossRef] [PubMed]
- Nakahata, S.; Saito, Y.; Marutsuka, K.; Hidaka, T.; Maeda, K.; Hatakeyama, K.; Shiraga, T.; Goto, A.; Takamatsu, N.; Asada, Y.; et al. Clinical significance of CADM1/TSLC1/IgSF4 expression in adult T-cell leukemia/lymphoma. Leukemia 2012, 26, 1238–1246. [Google Scholar] [CrossRef] [PubMed]
- Makiyama, J.; Kobayashi, S.; Watanabe, E.; Ishigaki, T.; Kawamata, T.; Nakashima, M.; Yamagishi, M.; Nakano, K.; Tojo, A.; Watanabe, T.; et al. CD4+ CADM1+ cell percentage predicts disease progression in HTLV-1 carriers and indolent adult T-cell leukemia/lymphoma. Cancer Sci. 2019, 110, 3746–3753. [Google Scholar] [CrossRef]
- Manivannan, K.; Rowan, A.G.; Tanaka, Y.; Taylor, G.P.; Bangham, C.R. CADM1/TSLC1 Identifies HTLV-1-Infected cells and determines their susceptibility to CTL-Mediated lysis. PLoS Pathog. 2016, 12, e1005560. [Google Scholar] [CrossRef]
- Sarkar, B.; Nishikata, I.; Nakahata, S.; Ichikawa, T.; Shiraga, T.; Saha, H.R.; Fujii, M.; Tanaka, Y.; Shimoda, K.; Morishita, K. Degradation of p47 by autophagy contributes to CADM1 overexpression in ATLL cells through the activation of NF-κB. Sci. Rep. 2019, 9, 3491. [Google Scholar] [CrossRef]
- Wolf, S.N.; Haddow, J.; Greiller, C.; Taylor, G.P.; Cook, L.B.M.; Rowan, A.G. Quantification of T cell clonality in human T cell leukaemia virus type-1 carriers can detect the development of adult T cell leukaemia early. Blood Cancer J. 2021, 11, 66. [Google Scholar] [CrossRef]
- Wada, Y.; Sato, T.; Hasegawa, H.; Matsudaira, T.; Nao, N.; Coler-Reilly, A.L.G.; Tasaka, T.; Yamauchi, S.; Okagawa, T.; Momose, H.; et al. RAISING is a high-performance method for identifying random transgene integration sites. Commun. Biol. 2022, 5, 535. [Google Scholar] [CrossRef]
| HTLV-1 Negative | HTLV-1 Positive | ||
|---|---|---|---|
| (n = 13) | (n = 57) | p-Value | |
| Age, year (IQR) | 67 (19) | 70 (12.8) | 0.57 |
| Female, no. (%) | 10 (76.9) | 45 (77.6) | 0.99 |
| RA duration, year (IQR) a | 13 (19) | 11 (15) | 0.93 |
| Positive for RF, no. (%) b | 61.5 | 68.1 | 0.74 |
| Positive for ACPA, no. (%) c | 66.7 | 66.7 | 0.99 |
| Steinbrocker’s classification, (%) | |||
| Stage Ⅰ/Ⅱ/Ⅲ/Ⅳ | 3/4/3/3 | 5/27/6/18 | 0.24 |
| Class 1/2/3/4 | 6/6/1/0 | 31/10//9/6 | 0.16 |
| CRP (mg/dl) (IQR) | 0.09 (0.2) | 0.11 (0.3) | 0.57 |
| ESR (mm/60 min) (IQR) | 18 (30) | 14 (19) | 0.68 |
| CDAI (IQR) d | 5.00 (5) | 2.9 (7.5) | 0.98 |
| SDAI (IQR) d | 5.56 (5) | 3.25 (8.5) | 0.99 |
| DAS28 (IQR) d | 2.74 (2) | 2.27 (1.4) | 0.88 |
| HAQ-DI (IQR) d | 0 (0) | 0.19 (1.6) | 0.39 |
| Corticosteroid in use, no. (%) | 6 (46) | 29 (51) | 0.99 |
| Dose of corticosteroid mg/day (IQR) e | 4.0 (5) | 4.0 (4) | 0.94 |
| MTX current user, no. (%) | 9 (69) | 27 (47) | 0.22 |
| Dose of MTX mg/week (IQR) | 6.0 (2) | 8.0 (4) | 0.41 |
| TAC current user, no. (%) | 1 (7.7) | 13 (23) | 0.44 |
| Biologics current user, no. (%) | 6 (46) | 13 (23) | 0.16 |
| HAS-Flow Analysis | ||
|---|---|---|
| No. of samples | percentage, % | |
| No. of HTLV-1-positive RA patients | 57 | 100 |
| CADM1 ≥ 25% | 13 | 22.8 |
| (a) CADM1 + CD7dim > CADM1 + CD7neg | 6 | 10.5 |
| (b) CADM1 + CD7dim < CADM1 + CD7neg | 7 | 12.3 |
| HTLV-1 PVL > 4.0 copies/100PBMCs | 19 | 33.3 |
| with (a) | 2 | 3.5 |
| with (b) | 6 | 10.5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Umekita, K.; Hashikura, Y.; Takaki, A.; Kimura, M.; Kawano, K.; Iwao, C.; Miyauchi, S.; Kawaguchi, T.; Matsuda, M.; Hashiba, Y.; et al. HAS-Flow May Be an Adequate Method for Evaluating Human T-Cell Leukemia Virus Type 1 Infected Cells in Human T-Cell Leukemia Virus Type 1-Positive Rheumatoid Arthritis Patients Receiving Antirheumatic Therapies: A Retrospective Cross-Sectional Observation Study. Viruses 2023, 15, 468. https://doi.org/10.3390/v15020468
Umekita K, Hashikura Y, Takaki A, Kimura M, Kawano K, Iwao C, Miyauchi S, Kawaguchi T, Matsuda M, Hashiba Y, et al. HAS-Flow May Be an Adequate Method for Evaluating Human T-Cell Leukemia Virus Type 1 Infected Cells in Human T-Cell Leukemia Virus Type 1-Positive Rheumatoid Arthritis Patients Receiving Antirheumatic Therapies: A Retrospective Cross-Sectional Observation Study. Viruses. 2023; 15(2):468. https://doi.org/10.3390/v15020468
Chicago/Turabian StyleUmekita, Kunihiko, Yuki Hashikura, Akira Takaki, Masatoshi Kimura, Katsumi Kawano, Chihiro Iwao, Shunichi Miyauchi, Takeshi Kawaguchi, Motohiro Matsuda, Yayoi Hashiba, and et al. 2023. "HAS-Flow May Be an Adequate Method for Evaluating Human T-Cell Leukemia Virus Type 1 Infected Cells in Human T-Cell Leukemia Virus Type 1-Positive Rheumatoid Arthritis Patients Receiving Antirheumatic Therapies: A Retrospective Cross-Sectional Observation Study" Viruses 15, no. 2: 468. https://doi.org/10.3390/v15020468
APA StyleUmekita, K., Hashikura, Y., Takaki, A., Kimura, M., Kawano, K., Iwao, C., Miyauchi, S., Kawaguchi, T., Matsuda, M., Hashiba, Y., & Hidaka, T. (2023). HAS-Flow May Be an Adequate Method for Evaluating Human T-Cell Leukemia Virus Type 1 Infected Cells in Human T-Cell Leukemia Virus Type 1-Positive Rheumatoid Arthritis Patients Receiving Antirheumatic Therapies: A Retrospective Cross-Sectional Observation Study. Viruses, 15(2), 468. https://doi.org/10.3390/v15020468






