Cytomegalovirus Anterior Uveitis: Clinical Manifestations, Diagnosis, Treatment, and Immunological Mechanisms
Abstract
1. Introduction
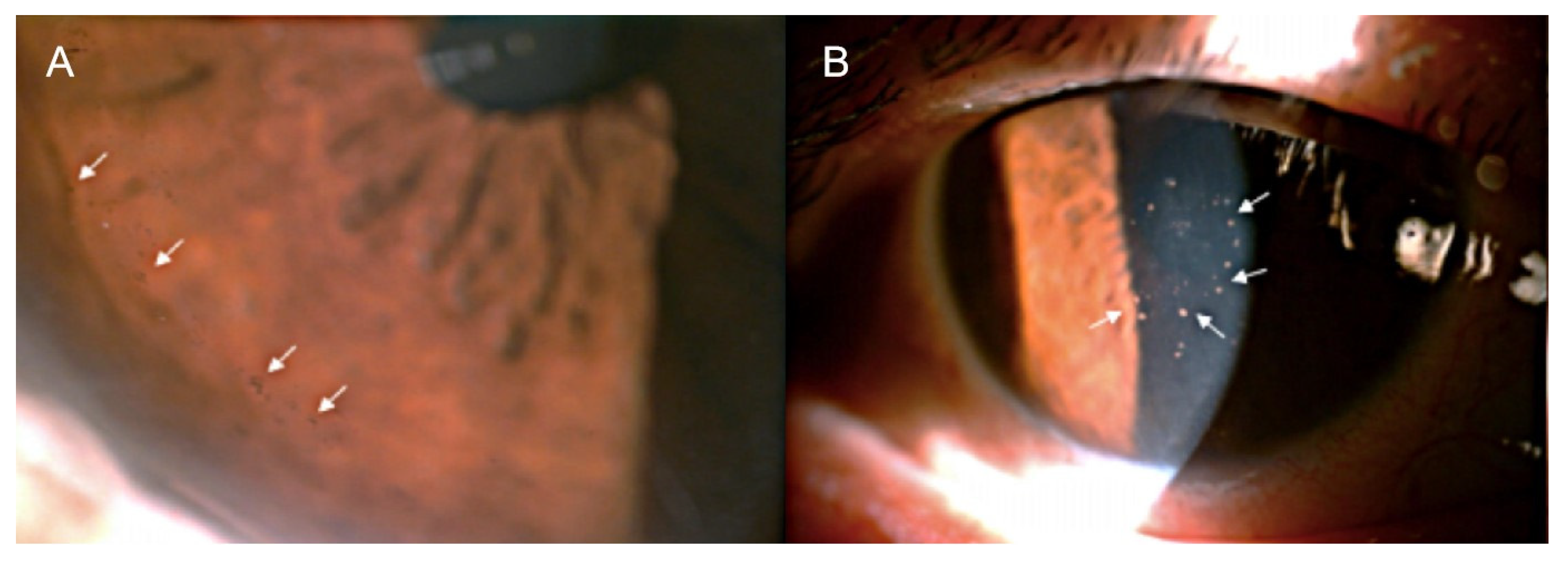
2. Clinical Features of CMV AU
3. Differential Diagnosis
4. Clinical Diagnostic Testing
5. Currently Used Antiviral Treatments and Updates
6. Currently Known Immune Mechanisms and Updates
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zuhair, M.; Smit, G.S.A.; Wallis, G.; Jabbar, F.; Smith, C.; Devleesschauwer, B.; Griffiths, P. Estimation of the worldwide seroprevalence of cytomegalovirus: A systematic review and meta-analysis. Rev. Med. Virol. 2019, 29, e2034. [Google Scholar] [CrossRef]
- Cannon, M.J.; Schmid, D.S.; Hyde, T.B. Review of cytomegalovirus seroprevalence and demographic characteristics associated with infection. Rev. Med. Virol. 2010, 20, 202–213. [Google Scholar] [CrossRef]
- Rawlinson, W.D.; Boppana, S.B.; Fowler, K.B.; Kimberlin, D.W.; Lazzarotto, T.; Alain, S.; Daly, K.; Doutre, S.; Gibson, L.; Giles, M.L.; et al. Congenital cytomegalovirus infection in pregnancy and the neonate: Consensus recommendations for prevention, diagnosis, and therapy. Lancet Infect. Dis. 2017, 17, e177–e188. [Google Scholar] [CrossRef] [PubMed]
- Gugliesi, F.; Coscia, A.; Griffante, G.; Galitska, G.; Pasquero, S.; Albano, C.; Biolatti, M. Where do we Stand after Decades of Studying Human Cytomegalovirus? Microorganisms 2020, 8, 685. [Google Scholar] [CrossRef] [PubMed]
- Munro, M.; Yadavalli, T.; Fonteh, C.; Arfeen, S.; Lobo-Chan, A.M. Cytomegalovirus Retinitis in HIV and Non-HIV Individuals. Microorganisms 2019, 8, 55. [Google Scholar] [CrossRef]
- Carmichael, A. Cytomegalovirus and the eye. Eye 2012, 26, 237–240. [Google Scholar] [CrossRef]
- de Schryver, I.; Rozenberg, F.; Cassoux, N.; Michelson, S.; Kestelyn, P.; Lehoang, P.; Davis, J.L.; Bodaghi, B. Diagnosis and treatment of cytomegalovirus iridocyclitis without retinal necrosis. Br. J. Ophthalmol. 2006, 90, 852–855. [Google Scholar] [CrossRef]
- Chan, N.S.; Chee, S.P.; Caspers, L.; Bodaghi, B. Clinical Features of CMV-Associated Anterior Uveitis. Ocul. Immunol. Inflamm. 2018, 26, 107–115. [Google Scholar] [CrossRef]
- Chee, S.P.; Bacsal, K.; Jap, A.; Se-Thoe, S.Y.; Cheng, C.L.; Tan, B.H. Clinical features of cytomegalovirus anterior uveitis in immunocompetent patients. Am. J. Ophthalmol. 2008, 145, 834–840. [Google Scholar] [CrossRef]
- Chee, S.P.; Jap, A. Presumed fuchs heterochromic iridocyclitis and Posner-Schlossman syndrome: Comparison of cytomegalovirus-positive and negative eyes. Am. J. Ophthalmol. 2008, 146, 883–889. [Google Scholar] [CrossRef]
- Miyazaki, D.; Shimizu, D.; Shimizu, Y.; Inoue, Y.; Inoue, T.; Higaki, S.; Ueta, M.; Sugita, S.; Real-Time PCR for Ocular Cytomegalovirus Infection Study Group. Diagnostic efficacy of real-time PCR for ocular cytomegalovirus infections. Graefes Arch. Clin. Exp. Ophthalmol. 2018, 256, 2413–2420. [Google Scholar] [CrossRef] [PubMed]
- Takase, H.; Kubono, R.; Terada, Y.; Imai, A.; Fukuda, S.; Tomita, M.; Miyanaga, M.; Kamoi, K.; Sugita, S.; Miyata, K.; et al. Comparison of the ocular characteristics of anterior uveitis caused by herpes simplex virus, varicella-zoster virus, and cytomegalovirus. Jpn. J. Ophthalmol. 2014, 58, 473–482. [Google Scholar] [CrossRef] [PubMed]
- Touhami, S.; Qu, L.; Angi, M.; Bojanova, M.; Touitou, V.; Lehoang, P.; Rozenberg, F.; Bodaghi, B. Cytomegalovirus Anterior Uveitis: Clinical Characteristics and Long-term Outcomes in a French Series. Am. J. Ophthalmol. 2018, 194, 134–142. [Google Scholar] [CrossRef]
- Hwang, Y.S.; Lin, K.K.; Lee, J.S.; Chang, S.H.; Chen, K.J.; Lai, C.C.; Huang, J.C.; Kuo, Y.H.; Hsiao, C.H. Intravitreal loading injection of ganciclovir with or without adjunctive oral valganciclovir for cytomegalovirus anterior uveitis. Graefes Arch. Clin. Exp. Ophthalmol. 2010, 248, 263–269. [Google Scholar] [CrossRef] [PubMed]
- Kongyai, N.; Sirirungsi, W.; Pathanapitoon, K.; Tananuvat, N.; Kunavisarut, P.; Leechanachai, P.; de Groot-Mijnes, J.D.; Rothova, A. Viral causes of unexplained anterior uveitis in Thailand. Eye 2012, 26, 529–534. [Google Scholar] [CrossRef] [PubMed]
- La Distia Nora, R.; Putera, I.; Mayasari, Y.D.; Hikmahwati, W.; Pertiwi, A.M.; Ridwan, A.S.; Sitompul, R.; Westcott, M.; Chee, S.P.; Pavesio, C.; et al. Clinical characteristics and treatment outcomes of cytomegalovirus anterior uveitis and endotheliitis: A systematic review and meta-analysis. Surv. Ophthalmol. 2022, 67, 1014–1030. [Google Scholar] [CrossRef] [PubMed]
- Woo, J.H.; Lim, W.K.; Ho, S.L.; Teoh, S.C. Characteristics of Cytomegalovirus Uveitis in Immunocompetent Patients. Ocul. Immunol. Inflamm. 2015, 23, 378–383. [Google Scholar] [CrossRef]
- Chee, S.P.; Bacsal, K.; Jap, A.; Se-Thoe, S.Y.; Cheng, C.L.; Tan, B.H. Corneal endotheliitis associated with evidence of cytomegalovirus infection. Ophthalmology 2007, 114, 798–803. [Google Scholar] [CrossRef]
- van Boxtel, L.A.; van der Lelij, A.; van der Meer, J.; Los, L.I. Cytomegalovirus as a cause of anterior uveitis in immunocompetent patients. Ophthalmology 2007, 114, 1358–1362. [Google Scholar] [CrossRef]
- Accorinti, M.; Gilardi, M.; Pirraglia, M.P.; Amorelli, G.M.; Nardella, C.; Abicca, I.; Pesci, F.R. Cytomegalovirus anterior uveitis: Long-term follow-up of immunocompetent patients. Graefes Arch. Clin. Exp. Ophthalmol. 2014, 252, 1817–1824. [Google Scholar] [CrossRef]
- Jap, A.; Sivakumar, M.; Chee, S.P. Is Posner Schlossman syndrome benign? Ophthalmology 2001, 108, 913–918. [Google Scholar] [CrossRef] [PubMed]
- Hwang, Y.S.; Shen, C.R.; Chang, S.H.; Lai, C.C.; Liu, C.L.; Chen, K.J.; Lin, K.K.; Chen, T.L.; Hsiao, C.H. The validity of clinical feature profiles for cytomegaloviral anterior segment infection. Graefes Arch. Clin. Exp. Ophthalmol. 2011, 249, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Chan, N.S.; Chee, S.P. Demystifying viral anterior uveitis: A review. Clin. Exp. Ophthalmol. 2019, 47, 320–333. [Google Scholar] [CrossRef] [PubMed]
- Watkins, J. Rubella: An overview of the symptoms and complications. Br. J. Sch. Nurs. 2014, 9, 284–286. [Google Scholar] [CrossRef]
- Yamamoto, S.; Shimomura, Y.; Kinoshita, S.; Tano, Y. Differentiating zosteriform herpes simplex from ophthalmic zoster. Arch. Ophthalmol. 1994, 112, 1515–1516. [Google Scholar] [CrossRef]
- Van der Lelij, A.; Ooijman, F.M.; Kijlstra, A.; Rothova, A. Anterior uveitis with sectoral iris atrophy in the absence of keratitis: A distinct clinical entity among herpetic eye diseases. Ophthalmology 2000, 107, 1164–1170. [Google Scholar] [CrossRef]
- Relvas, L.J.; Caspers, L.; Chee, S.P.; Zierhut, M.; Willermain, F. Differential Diagnosis of Viral-Induced Anterior Uveitis. Ocul. Immunol. Inflamm. 2018, 26, 726–731. [Google Scholar] [CrossRef]
- Tugal-Tutkun, I.; Otuk-Yasar, B.; Altinkurt, E. Clinical features and prognosis of herpetic anterior uveitis: A retrospective study of 111 cases. Int. Ophthalmol. 2010, 30, 559–565. [Google Scholar] [CrossRef]
- Wensing, B.; Relvas, L.M.; Caspers, L.E.; Valentincic, N.V.; Stunf, S.; de Groot-Mijnes, J.D.; Rothova, A. Comparison of rubella virus- and herpes virus-associated anterior uveitis: Clinical manifestations and visual prognosis. Ophthalmology 2011, 118, 1905–1910. [Google Scholar] [CrossRef]
- Kuo, Y.W.; Chang, E.C.; Lee, C.Y.; Lee, S.H.; Liang, I.C.; Chen, Y.C.; Hou, Y.C. Effectiveness of Topical Ganciclovir 2% Monotherapy Versus Combined Steroid Therapy in Cytomegalovirus Endotheliitis. J. Clin. Med. 2022, 11, 5811. [Google Scholar] [CrossRef]
- de-la-Torre, A.; Valdes-Camacho, J.; Foster, C.S. Bilateral Herpes Simplex Uveitis: Review of the Literature and Own Reports. Ocul. Immunol. Inflamm. 2017, 25, 497–502. [Google Scholar] [CrossRef] [PubMed]
- Kam, K.W.; Wong, C.H.; Ho, M.; Sze, R.K.H.; Chan, P.K.S.; Young, A.L. Iris Depigmentation in the Prediction of Cytomegalovirus Anterior Uveitis. Ocul. Immunol. Inflamm. 2021, 30, 1–6. [Google Scholar] [CrossRef]
- Pleyer, U.; Chee, S.P. Current aspects on the management of viral uveitis in immunocompetent individuals. Clin. Ophthalmol. 2015, 9, 1017–1028. [Google Scholar] [CrossRef] [PubMed]
- Relvas, L.J.M.; Antoun, J.; de Groot-Mijnes, J.D.F.; Motulsky, E.; Ten Dam-Van Loon, N.H.; Makhoul, D.; Willermain, F.; Caspers, L. Diagnosis of Cytomegalovirus Anterior Uveitis in Two European Referral Centers. Ocul. Immunol. Inflamm. 2018, 26, 116–121. [Google Scholar] [CrossRef] [PubMed]
- Mochizuki, M.; Sugita, S.; Kamoi, K.; Takase, H. A new era of uveitis: Impact of polymerase chain reaction in intraocular inflammatory diseases. Jpn. J. Ophthalmol. 2017, 61, 1–20. [Google Scholar] [CrossRef]
- Kandori, M.; Miyazaki, D.; Yakura, K.; Komatsu, N.; Touge, C.; Ishikura, R.; Inoue, Y. Relationship between the number of cytomegalovirus in anterior chamber and severity of anterior segment inflammation. Jpn. J. Ophthalmol. 2013, 57, 497–502. [Google Scholar] [CrossRef] [PubMed]
- Kalogeropoulos, D.; Sakkas, H.; Mohammed, B.; Vartholomatos, G.; Malamos, K.; Sreekantam, S.; Kanavaros, P.; Kalogeropoulos, C. Ocular toxoplasmosis: A review of the current diagnostic and therapeutic approaches. Int. Ophthalmol. 2022, 42, 295–321. [Google Scholar] [CrossRef]
- Wollanke, B.; Gerhards, H.; Ackermann, K. Infectious Uveitis in Horses and New Insights in Its Leptospiral Biofilm-Related Pathogenesis. Microorganisms 2022, 10, 387. [Google Scholar] [CrossRef]
- Cimino, L.; Aldigeri, R.; Parmeggiani, M.; Belloni, L.; Zotti, C.A.; Fontana, L.; Invernizzi, A.; Salvarani, C.; Cappuccini, L. Searching for viral antibodies and genome in intraocular fluids of patients with Fuchs uveitis and non-infectious uveitis. Graefes Arch. Clin. Exp. Ophthalmol. 2013, 251, 1607–1612. [Google Scholar] [CrossRef]
- Wu, W.; Jiang, H.; Zhang, Y.; Zhou, Y.; Bai, G.; Shen, L.; Zhou, H.; Chen, X.; Hu, L. Clinical Metagenomic Next-Generation Sequencing for Diagnosis of Secondary Glaucoma in Patients With Cytomegalovirus-Induced Corneal Endotheliitis. Front. Microbiol. 2022, 13, 940818. [Google Scholar] [CrossRef]
- Crumpacker, C.S. Ganciclovir. N. Engl. J. Med. 1996, 335, 721–729. [Google Scholar] [CrossRef]
- Chee, S.P.; Jap, A. Cytomegalovirus anterior uveitis: Outcome of treatment. Br. J. Ophthalmol. 2010, 94, 1648–1652. [Google Scholar] [CrossRef]
- Sobolewska, B.; Deuter, C.; Doycheva, D.; Zierhut, M. Long-term oral therapy with valganciclovir in patients with Posner-Schlossman syndrome. Graefes Arch. Clin. Exp. Ophthalmol. 2014, 252, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Morlet, N.; Young, S.; Naidoo, D.; Graham, G.; Coroneo, M.T. High dose intravitreal ganciclovir injection provides a prolonged therapeutic intraocular concentration. Br. J. Ophthalmol. 1996, 80, 214–216. [Google Scholar] [CrossRef]
- Sakai, J.I.; Usui, Y.; Suzuki, J.; Kezuka, T.; Goto, H. Clinical features of anterior uveitis caused by three different herpes viruses. Int. Ophthalmol. 2019, 39, 2785–2795. [Google Scholar] [CrossRef] [PubMed]
- Waduthantri, S.; Zhou, L.; Chee, S.P. Intra-cameral level of ganciclovir gel, 0.15% following topical application for cytomegalovirus anterior segment infection: A pilot study. PLoS ONE 2018, 13, e0191850. [Google Scholar] [CrossRef] [PubMed]
- Takhar, J.S.; Joye, A.S.; Somkijrungroj, T.; Laovirojjanakul, W.; Lin, C.P.; Lietman, T.M.; Porco, T.C.; Keenan, J.D.; Gebreegziabher, E.A.; Seitzman, G.D.; et al. A double masked randomised 4-week, placebo-controlled study in the USA, Thailand and Taiwan to compare the efficacy of oral valganciclovir and topical 2% ganciclovir in the treatment of cytomegalovirus anterior uveitis: Study protocol. BMJ Open 2019, 9, e033175. [Google Scholar] [CrossRef] [PubMed]
- Pavan-Langston, D.; Welch, C.L.; Zegans, M.E. Ganciclovir gel for cytomegalovirus keratouveitis. Ophthalmology 2012, 119, 2411. [Google Scholar] [CrossRef] [PubMed]
- Antoun, J.; Willermain, F.; Makhoul, D.; Motulsky, E.; Caspers, L.; Relvas, L.J. Topical Ganciclovir in Cytomegalovirus Anterior Uveitis. J. Ocul. Pharmacol. Ther. 2017, 33, 313–318. [Google Scholar] [CrossRef]
- Wong, M.O.M.; Yu, A.H.Y.; Chan, C.K.M. Efficacy and safety of oral valganciclovir in cytomegalovirus anterior uveitis with uncontrolled intraocular pressure. Br. J. Ophthalmol. 2021, 105, 1666–1671. [Google Scholar] [CrossRef]
- Bhoopat, T.; Takhar, J.S.; Oldenburg, C.E.; Keenan, J.D.; Gonzales, J.A.; Margolis, T.P. Treatment of Cytomegalovirus Anterior Uveitis at a North American Tertiary Center With Oral Valganciclovir. Cornea 2020, 39, 584–589. [Google Scholar] [CrossRef] [PubMed]
- Markan, A.; Gupta, N.; Dogra, M.; Sharma, A.; Singh, R. Oral valganciclovir in human immunodeficiency virus-positive patients suffering from cytomegalovirus retinitis at a tertiary care hospital in North India. Indian J. Ophthalmol. 2022, 70, 2472–2475. [Google Scholar] [CrossRef] [PubMed]
- Ligat, G.; Cazal, R.; Hantz, S.; Alain, S. The human cytomegalovirus terminase complex as an antiviral target: A close-up view. FEMS Microbiol. Rev. 2018, 42, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Tsui, E.; Gonzales, J.A.; Shantha, J.G.; Acharya, N.; Doan, T. Letermovir for the Management of Cytomegalovirus-associated Uveitis. Ocul. Immunol. Inflamm. 2021, 29, 169–174. [Google Scholar] [CrossRef]
- Chou, S.; Satterwhite, L.E.; Ercolani, R.J. New Locus of Drug Resistance in the Human Cytomegalovirus UL56 Gene Revealed by In Vitro Exposure to Letermovir and Ganciclovir. Antimicrob. Agents Chemother. 2018, 62, e00922-18. [Google Scholar] [CrossRef]
- Schulz, U.; Solidoro, P.; Muller, V.; Szabo, A.; Gottlieb, J.; Wilkens, H.; Enseleit, F. CMV Immunoglobulins for the Treatment of CMV Infections in Thoracic Transplant Recipients. Transplantation 2016, 100 (Suppl. 3), S5–S10. [Google Scholar] [CrossRef]
- Revello, M.G.; Lazzarotto, T.; Guerra, B.; Spinillo, A.; Ferrazzi, E.; Kustermann, A.; Guaschino, S.; Vergani, P.; Todros, T.; Frusca, T.; et al. A randomized trial of hyperimmune globulin to prevent congenital cytomegalovirus. N. Engl. J. Med. 2014, 370, 1316–1326. [Google Scholar] [CrossRef]
- Nigro, G.; Adler, S.P.; La Torre, R.; Best, A.M.; Congenital Cytomegalovirus Collaborating Group. Passive immunization during pregnancy for congenital cytomegalovirus infection. N. Engl. J. Med. 2005, 353, 1350–1362. [Google Scholar] [CrossRef]
- Maidji, E.; Nigro, G.; Tabata, T.; McDonagh, S.; Nozawa, N.; Shiboski, S.; Muci, S.; Anceschi, M.M.; Aziz, N.; Adler, S.P.; et al. Antibody treatment promotes compensation for human cytomegalovirus-induced pathogenesis and a hypoxia-like condition in placentas with congenital infection. Am. J. Pathol. 2010, 177, 1298–1310. [Google Scholar] [CrossRef]
- Visentin, S.; Manara, R.; Milanese, L.; Da Roit, A.; Forner, G.; Salviato, E.; Citton, V.; Magno, F.M.; Orzan, E.; Morando, C.; et al. Early primary cytomegalovirus infection in pregnancy: Maternal hyperimmunoglobulin therapy improves outcomes among infants at 1 year of age. Clin. Infect. Dis. 2012, 55, 497–503. [Google Scholar] [CrossRef]
- Kagan, K.O.; Enders, M.; Schampera, M.S.; Baeumel, E.; Hoopmann, M.; Geipel, A.; Berg, C.; Goelz, R.; De Catte, L.; Wallwiener, D.; et al. Prevention of maternal-fetal transmission of cytomegalovirus after primary maternal infection in the first trimester by biweekly hyperimmunoglobulin administration. Ultrasound Obstet. Gynecol. 2019, 53, 383–389. [Google Scholar] [CrossRef] [PubMed]
- Varani, S.; Landini, M.P. Cytomegalovirus-induced immunopathology and its clinical consequences. Herpesviridae 2011, 2, 6. [Google Scholar] [CrossRef] [PubMed]
- La Rosa, C.; Diamond, D.J. The immune response to human CMV. Future Virol. 2012, 7, 279–293. [Google Scholar] [CrossRef] [PubMed]
- Sester, M.; Sester, U.; Gartner, B.C.; Girndt, M.; Meyerhans, A.; Kohler, H. Dominance of virus-specific CD8 T cells in human primary cytomegalovirus infection. J. Am. Soc. Nephrol. 2002, 13, 2577–2584. [Google Scholar] [CrossRef] [PubMed]
- Lim, E.Y.; Jackson, S.E.; Wills, M.R. The CD4+ T Cell Response to Human Cytomegalovirus in Healthy and Immunocompromised People. Front. Cell. Infect. Microbiol. 2020, 10, 202. [Google Scholar] [CrossRef] [PubMed]
- Juno, J.A.; van Bockel, D.; Kent, S.J.; Kelleher, A.D.; Zaunders, J.J.; Munier, C.M. Cytotoxic CD4 T Cells-Friend or Foe during Viral Infection? Front. Immunol. 2017, 8, 19. [Google Scholar] [CrossRef]
- Reus, B.; Caserta, S.; Larsen, M.; Morrow, G.; Bano, A.; Hallensleben, M.; Rajkumar, C.; Pera, A.; Kern, F. In-Depth Profiling of T-Cell Responsiveness to Commonly Recognized CMV Antigens in Older People Reveals Important Sex Differences. Front. Immunol. 2021, 12, 707830. [Google Scholar] [CrossRef]
- Griessl, M.; Renzaho, A.; Freitag, K.; Seckert, C.K.; Reddehase, M.J.; Lemmermann, N.A.W. Stochastic Episodes of Latent Cytomegalovirus Transcription Drive CD8 T-Cell “Memory Inflation” and Avoid Immune Evasion. Front. Immunol. 2021, 12, 668885. [Google Scholar] [CrossRef]
- Wills, M.R.; Poole, E.; Lau, B.; Krishna, B.; Sinclair, J.H. The immunology of human cytomegalovirus latency: Could latent infection be cleared by novel immunotherapeutic strategies? Cell. Mol. Immunol. 2015, 12, 128–138. [Google Scholar] [CrossRef]
- Sugar, E.A.; Jabs, D.A.; Ahuja, A.; Thorne, J.E.; Danis, R.P.; Meinert, C.L.; Studies of the Ocular Complications of AIDS Research Group. Incidence of cytomegalovirus retinitis in the era of highly active antiretroviral therapy. Am. J. Ophthalmol. 2012, 153, 1016–1024. [Google Scholar] [CrossRef]
- Jackson, S.E.; Chen, K.C.; Groves, I.J.; Sedikides, G.X.; Gandhi, A.; Houldcroft, C.J.; Poole, E.L.; Montanuy, I.; Mason, G.M.; Okecha, G.; et al. Latent Cytomegalovirus-Driven Recruitment of Activated CD4+ T Cells Promotes Virus Reactivation. Front. Immunol. 2021, 12, 657945. [Google Scholar] [CrossRef] [PubMed]
- Pei, X.Y.; Liu, X.F.; Zhao, X.Y.; Lv, M.; Mo, X.D.; Chang, Y.J.; Shang, Q.N.; Sun, Y.Q.; Chen, Y.H.; Xu, L.P.; et al. Comparable anti-CMV responses of transplant donor and third-party CMV-specific T cells for treatment of CMV infection after allogeneic stem cell transplantation. Cell. Mol. Immunol. 2022, 19, 482–491. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Rios, E.; Nuevalos, M.; Mancebo, F.J.; Perez-Romero, P. Is It Feasible to Use CMV-Specific T-Cell Adoptive Transfer as Treatment Against Infection in SOT Recipients? Front. Immunol. 2021, 12, 657144. [Google Scholar] [CrossRef]
- Mo, W.; Liang, Y.; Wang, C.; Zhang, Y. A Simplified CMV-specific T Cell Transfer Targeting Drug-resistant CMV Retinitis. Ocul. Immunol. Inflamm. 2022, 30, 1541–1543. [Google Scholar] [CrossRef] [PubMed]
- Voigt, V.; Andoniou, C.E.; Schuster, I.S.; Oszmiana, A.; Ong, M.L.; Fleming, P.; Forrester, J.V.; Degli-Esposti, M.A. Cytomegalovirus establishes a latent reservoir and triggers long-lasting inflammation in the eye. PLoS Pathog. 2018, 14, e1007040. [Google Scholar] [CrossRef] [PubMed]
| Variable 1 | HSV | VZV | CMV | RV |
|---|---|---|---|---|
| Age (years) | <50 | >60 | Acute, >30 chronic, >50 | 20–40 |
| Sex | No predilection | No predilection | Male predilection | No predilection |
| Race | All | All | Asian | Western |
| Symptoms | Severe | Severe | Mild or absent | Mild or absent |
| Intraocular pressure | Acute spikes | Acute spikes | Very high (Up to 50 mmHg) | Persistent elevation |
| Keratic precipitates | ||||
| Distribution | Arlt’s triangle | Arlt’s triangle | Diffusely distributed | Diffusely distributed |
| Morphology | Small to medium | Small to medium | Medium to large, coin shaped | Fine stellate and non-stellate |
| Color | Pigmented | Pigmented | White or pigmented | White |
| Dermal manifestation | Vesicular rash | Dermatomal rash | None | Maculopapular rash |
| Corneal | Dendritic ulcers | Pseudodendritic ulcers | Endotheliitis, endothelial cell loss | Normal |
| Iris atrophy | Sectoral iris atrophy | Spiral atrophy | Iris stromal atrophy | Diffuse atrophy |
| Pupil | Eccentric dilated | Eccentric dilated | Round | Round |
| Cataract | Late at presentation | Late at presentation | Late at presentation | Early at presentation |
| Vitritis | Always | Sometimes | Rarely | Always |
| Technique * | Sensitivity | Specificity | Feasibility | Rapidness | Application |
|---|---|---|---|---|---|
| PCR | ++ | +++ | +++ | +++ | Early-stage diagnosis |
| Qualitative multiplex PCR | ++ | +++ | +++ | +++ | Virus screening |
| Real-time PCR | ++ | +++ | +++ | +++ | Detection of viral loads |
| GWC | +++ | + | +++ | +++ | Late-stage diagnosis |
| Viral culture | + | +++ | + | + | Accurate diagnosis |
| Isolated serological tests | + | + | ++ | +++ | Limited value |
| Metagenomic deep sequencing | +++ | +++ | + | +++ | Detection of fungi, parasites, and DNA and RNA viruses simultaneously |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, J.; Kamoi, K.; Zong, Y.; Yang, M.; Ohno-Matsui, K. Cytomegalovirus Anterior Uveitis: Clinical Manifestations, Diagnosis, Treatment, and Immunological Mechanisms. Viruses 2023, 15, 185. https://doi.org/10.3390/v15010185
Zhang J, Kamoi K, Zong Y, Yang M, Ohno-Matsui K. Cytomegalovirus Anterior Uveitis: Clinical Manifestations, Diagnosis, Treatment, and Immunological Mechanisms. Viruses. 2023; 15(1):185. https://doi.org/10.3390/v15010185
Chicago/Turabian StyleZhang, Jing, Koju Kamoi, Yuan Zong, Mingming Yang, and Kyoko Ohno-Matsui. 2023. "Cytomegalovirus Anterior Uveitis: Clinical Manifestations, Diagnosis, Treatment, and Immunological Mechanisms" Viruses 15, no. 1: 185. https://doi.org/10.3390/v15010185
APA StyleZhang, J., Kamoi, K., Zong, Y., Yang, M., & Ohno-Matsui, K. (2023). Cytomegalovirus Anterior Uveitis: Clinical Manifestations, Diagnosis, Treatment, and Immunological Mechanisms. Viruses, 15(1), 185. https://doi.org/10.3390/v15010185






