Abstract
Herpes simplex viruses (HSV) are ubiquitously distributed with a seroprevalence ranging up to 95% in the adult population. Refractory viral infections with herpes simplex virus type 1 (HSV-1) and type 2 (HSV-2) represent a major global health issue. In particular, the increasing occurrence of resistance to conventional antiviral drugs make the therapy of such infections even more challenging. For instance, the frequent and long-term use of acyclovir and other nucleoside analogues targeting the viral DNA-polymerase enhance the development of resistant viruses. Particularly, the incidental increase of those strains in immunocompromised patients is alarming and represent a major health concern. Alternative treatment concepts are clearly needed. Natural products such as herbal medicines showed antiherpetic activity in vitro and in vivo and proved to be an excellent source for the discovery and isolation of novel antivirals. By this means, numerous plant-derived compounds with antiviral or antimicrobial activity could be isolated. Natural medicines and their ingredients are well-tolerated and could be a good alternative for treating herpes simplex virus infections. This review provides an overview of the recent status of natural sources such as plants, bacteria, fungi, and their ingredients with antiviral activity against herpes simplex viruses. Furthermore, we highlight the most potent herbal medicines and ingredients as promising candidates for clinical investigation and give an overview about the most important drug classes along with their potential antiviral mechanisms. The content of this review is based on articles that were published between 1996 and 2021.
1. Introduction
Herpes simplex virus infections are considered a major public health issue worldwide. These human pathogen DNA viruses belong to the family of Alphaherpesvirinae. Upon primary infection, the herpes simplex viruses type 1 and 2 (HSV-1 and HSV-2) persist lifelong in the autonomic and sensory ganglia of its host. Especially HSV-1 infections are ubiquitously distributed with a seroprevalence ranging up to 95% in the adult population [1]. After reactivation, HSV may cause symptoms ranging from painful, but self-limited infections of the oral or genital mucosa to severe infections of the eye or life-threatening infections in immunocompromised hosts or newborns [2,3]. Active HSV-1 infections are usually associated with oral or facial herpes, while HSV-2 predominately causes genital infections. Reactivated HSV-2 infections often exhibit painful genital lesions providing a higher risk for other sexually transmitted diseases and invasive cervical carcinoma [4].
Although numerous vaccine candidates have been investigated in clinical trials, there is no licensed vaccine available for the prevention of HSV infections. Over the last decades, many different antiviral drugs targeting the viral DNA-polymerase were approved for the treatment of acute HSV infections. The most widely used antiviral agents against HSV are acyclovir (ACV), valacyclovir, famciclovir, cidofovir, and foscarnet. ACV and the related nucleoside analogues can successfully inhibit viral replication and thereby mediate cure from HSV-related symptoms, but the emergence of drug resistance to ACV has created a barrier for the treatment of HSV infections [5]. Moreover, it has been demonstrated that ACV therapy in HIV/HSV-co-infected patients reduces HIV serum levels and may protract the necessity of an antiretroviral therapy [6,7,8]. However, ACV may interact directly with the HIV reverse transcriptase in HIV-infected cells, which may increase the occurrence of the reverse transcriptase mutants that are associated with a reduced sensitivity of the virus to antiretroviral therapy [9,10]. Furthermore, corneal morbidity and blindness are common issues of ACV-refractory HSV infections of the cornea in industrial nations [11]. ACV-resistant infections are frequently observed in immunocompromised patients. Due to a long-term prophylactic or pre-emptive antiviral treatment in these patients, the occurrence of ACV resistance is particularly high in this group [12]. In the average adult population, the frequency of ACV-resistant HSV was determined with 0.27% (n = 368) [12]. In contrast, the frequency of ACV-resistant HSV was assessed with 7.03% in immunocompromised patients [12]. The highest rates of ACV resistance were reported in patients undergoing hematopoietic stem cell transplantation (14.3%), followed by HIV-infected patients (3.92%) or patients suffering from various tumor diseases (3.85%) [12]. Cross-resistance to other nucleoside analogs targeting the viral HSV thymidine kinase (TK) are frequent, since a reduced sensitivity of HSV to ACV is mostly caused by mutations in the TK gene [5,13]. However, resistance not only emerges against drugs such as famciclovir or penciclovir that target HSV-TK but also against the viral DNA-polymerase inhibitors foscarnet and cidofovir [14], the latter severely in patients undergoing stem cell transplantation [12]. DNA-polymerase inhibitors can be used for the treatment of ACV-resistant HSV infections. However, their use is limited due to possible serious side effects, especially in patients with comorbidities [5].
Clearly, there is an urgent need to explore new effective and well-tolerated approaches for the treatment of HSV infections and reactivations. Traditional herbal medicines are an abundant source of antimicrobial or antiviral active substances. Plant extracts and other natural products have been used for hundreds of years for the treatment of infectious diseases. We screened the PubMed database to find relevant articles by using the keywords natural products, medicinal plants, medicinal herbs, herbal medicine, plant oils, herpes simplex virus, herpes labialis, and herpes genitalis. Articles included in our analysis were published from 1996 to 2021. We searched for studies that described compounds that were isolated from natural sources such as plants, fungi, and other sources. We focused on well-characterized compounds with already uncovered mechanisms of how these compounds interfere with the viral replication. This strategy allows for conclusions about the potential antiviral activity of these compounds against ACV-resistant viruses. The review gives an overview of the distinct compounds isolated from plants and other natural sources and summarizes the results from in vitro and in vivo studies conducted thus far.
3. Summary and Additional Comments
The current standard therapy for HSV primary infection and reactivation includes ACV and valacyclovir. However, the emergence of drug resistances limits the available treatment options [5]. Therefore, there is a clear need for the development of new effective antiviral drugs. Plants traditionally used for medical purposes are a promising source of new antiviral compounds.
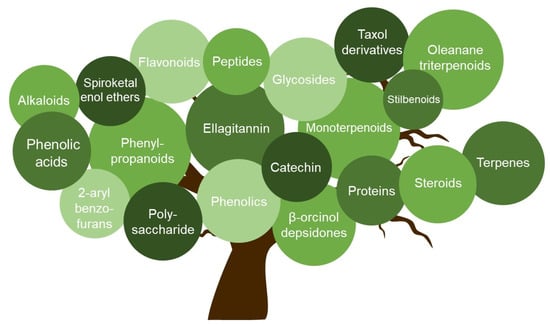
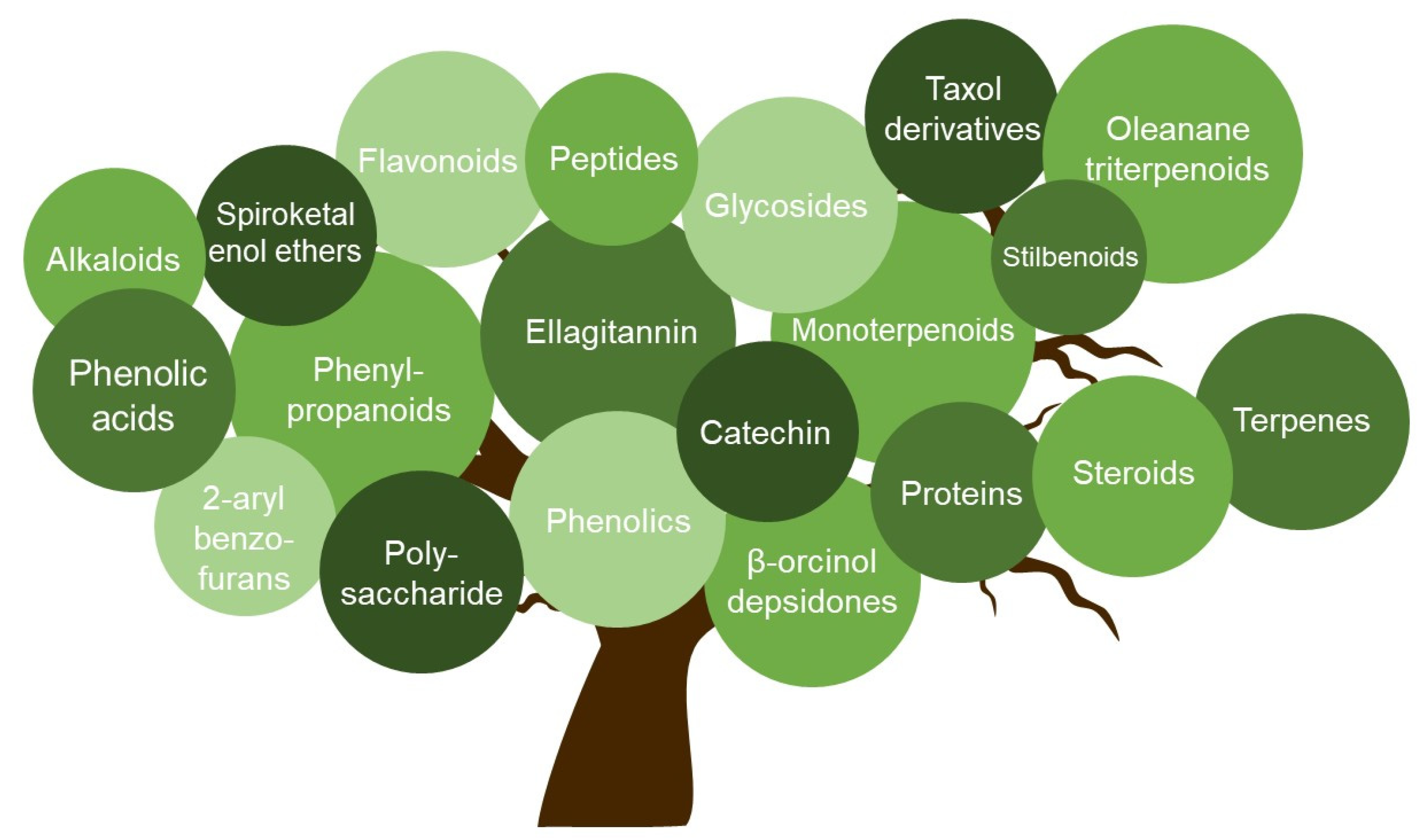
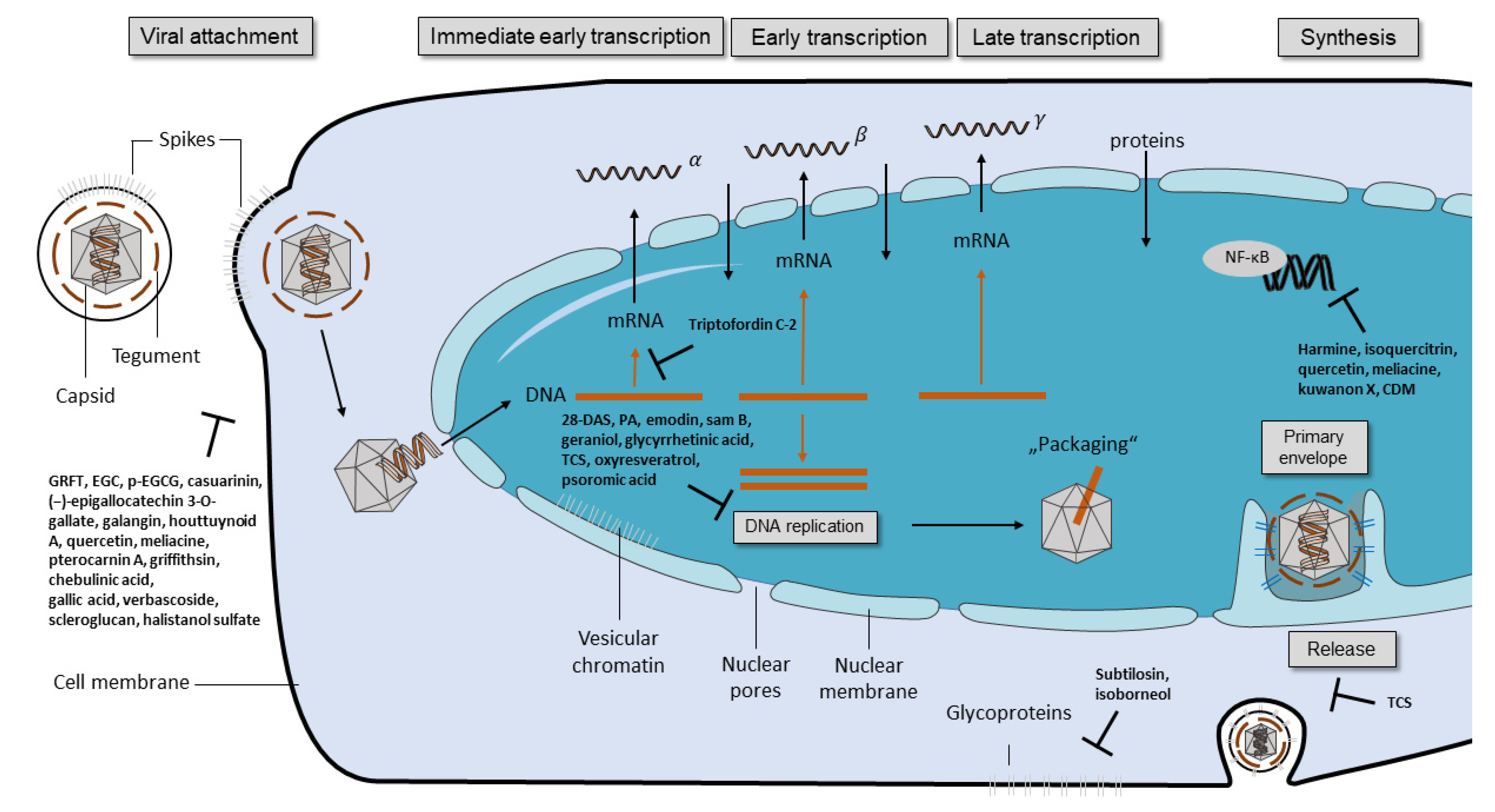
In the present review, we summarized compounds isolated from plants, bacteria, and fungi with different mechanisms inhibiting HSV, including the inhibition of viral attachment, penetration, and replication. The compounds belong to different groups such as catechins, flavonoids, phenolic acids, polysaccharides, terpenes, and peptides. With six compounds, flavonoids represent the largest group.
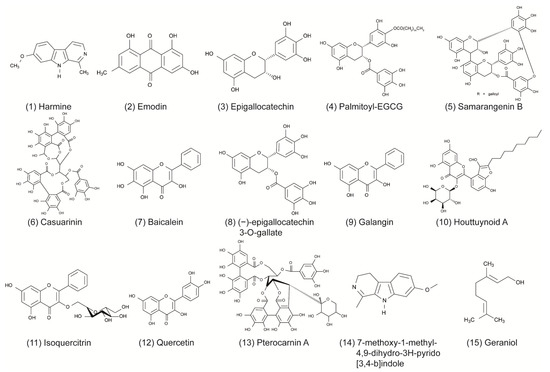
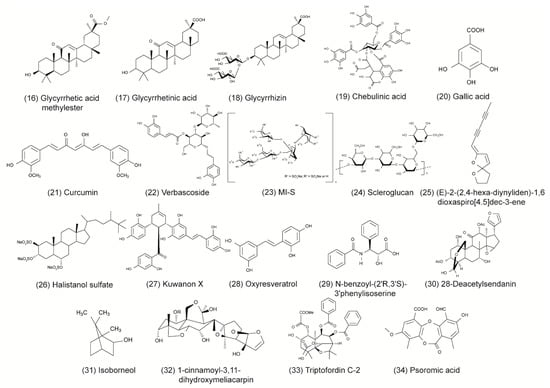
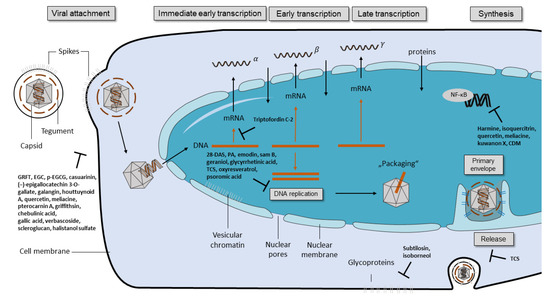
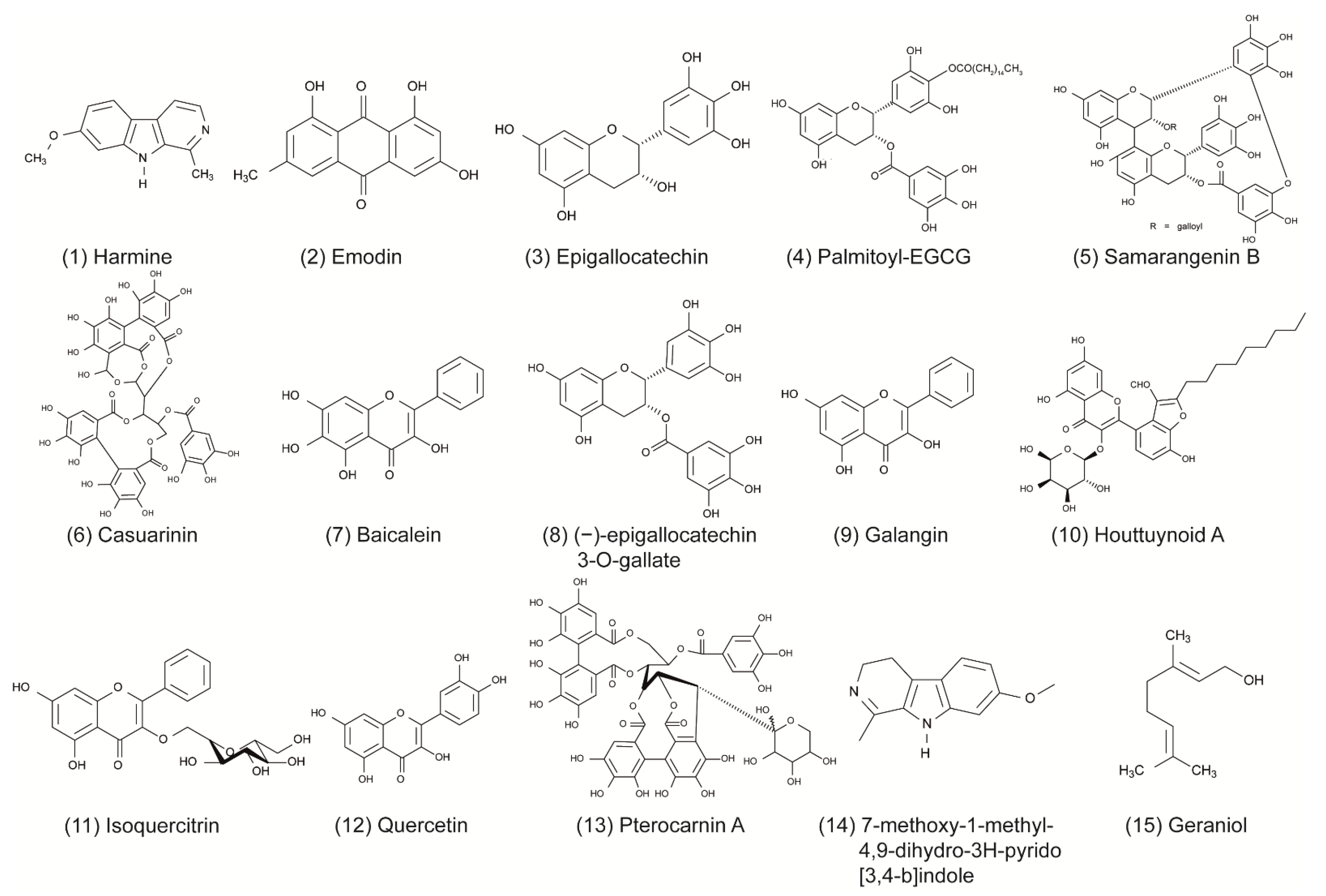
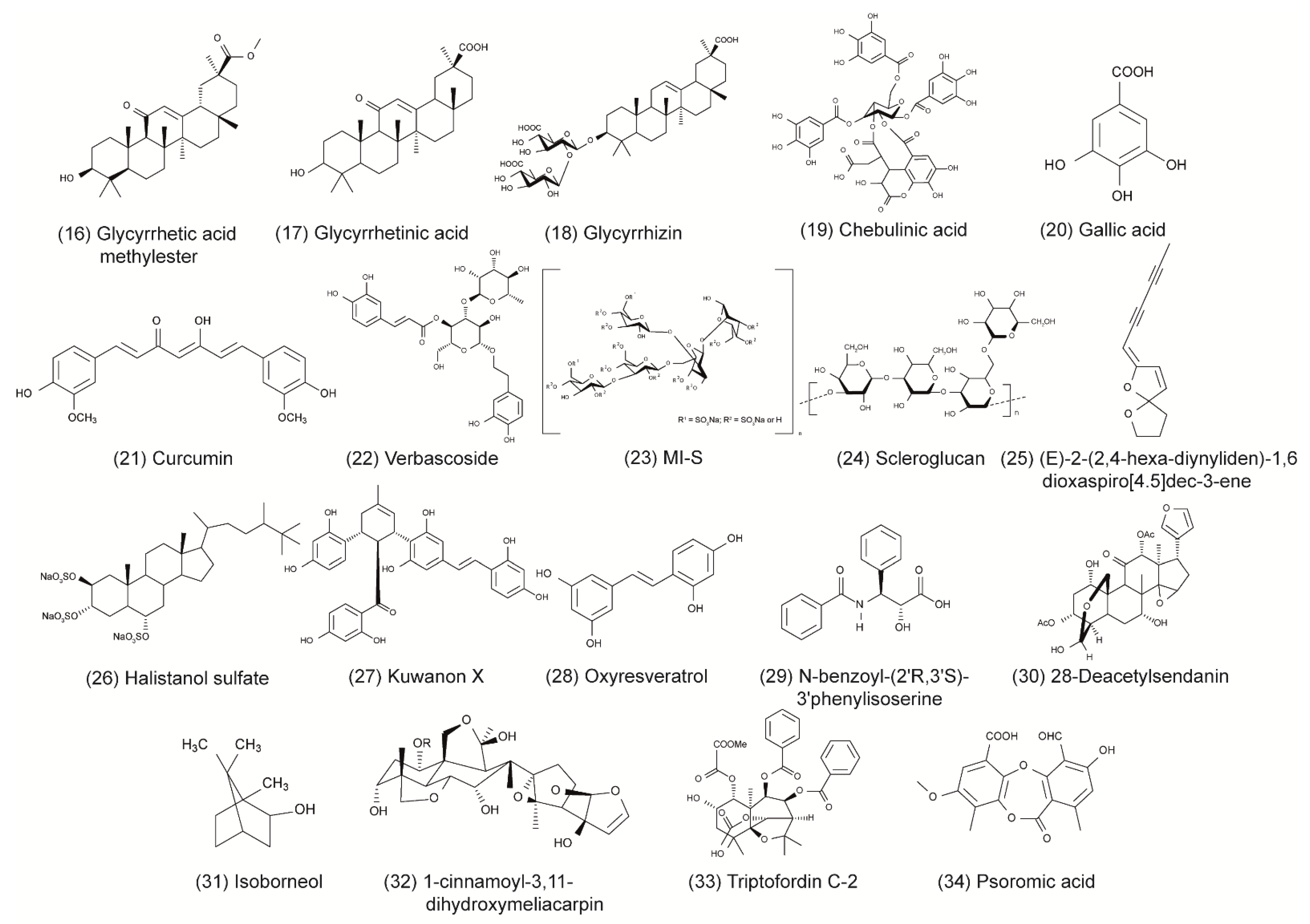
Some of the tested compounds showed a similar or even better antiviral activity against HSV-1 than ACV in vitro, including the anthraquinone emodin, the catechin epigallocatechin (EGC), the harmaline HM, and the β-orcinol depsidone PA as well as other promising compounds (Table 1 and Figure 1) [17,18,31,35,44,56]. Interestingly, when these compounds were supplemented with geraniol, they had a similar or better antiviral activity against HSV-2 [17,18,30,32,36,56]. These candidates might be good for further evaluation in the search for alternative treatments of ACV-resistant viruses. Additional studies are needed to investigate whether these compounds maintain their antiviral activity against HSV-1 and HSV-2 clinical isolates that are resistant to ACV. To date, only a few studies have included ACV-resistant strains in their experiments [4,47,69]. In the study of Luo et al. (2020), the antiviral effect of baicalein against an ACV-resistant HSV-1 strain (HSV-1 Blue) was comparable in its effect against the HSV-1 F strain without resistance [24]. As anticipated, ACV exhibited only a weak antiviral activity against the ACV-resistant virus. However, ACV showed a stronger inhibition against the HSV-1 F strain than baicalein. In a further in vitro study, the antiviral effect of MI-S was observed against HSV-1 KOS, an ACV-resistant HSV-1 strain (HSV-1 29R), and HSV-2 333 [69]. MI-S was similarly effective against HSV-1 KOS and HSV-1 29R when the compound was administered during or after infection. ACV showed no inhibitory activity against HSV-1 KOS when applied simultaneously with the virus, but it was more effective than MI-S under post-infection treatment conditions.
Only a few of those compounds were evaluated for their efficacy against HSV infections in in vivo studies. Of these compounds, emodin exhibited promising results, as it strongly increased the survival rate of mice infected with HSV-1 and HSV-2 [17]. The compound was even more effective than ACV in increasing the survival rate and mean time to death. Furthermore, HM significantly increased the survival rate of mice infected with HSV-2 [30]. Although standard therapy with ACV was more effective, the increase of survival through the compound was still striking. The results of these studies indicate that emodin and HM are promising candidates for future clinical trials. Clearly, there is a need for more elaborate in vivo studies as well as clinical trials that compare the compounds to the standard therapy of HSV.
4. Conclusions
To date, numerous herbal medicines and their main ingredients were investigated for their antiviral efficacy against HSV-1 and HSV-2 in cell culture and animal models. For instance, the anthraquinone emodin derived from the plant Rheum tanguticum as well as the harmaline HM isolated from the herb Ophiorrhiza nicobarica Balkr exhibited a strong antiviral effect against HSV in vitro and in vivo, which were comparable to or even better than ACV. Clinical studies are needed to determine the efficacy of these compounds in humans. Furthermore, natural compounds might be an effective substitute for drugs such as ACV in the treatment of ACV-resistant HSV infections. Taken together, herbal medicines represent a promising source to isolate novel compounds with antiviral activity against HSV-1 and HSV-2. Numerous compounds could be isolated and pre-clinically characterized thus far. Further clinical evaluation of the agents that are most promising in animal trials may lead to the development of novel therapy options in humans.
Author Contributions
Conceptualization, L.v.d.S., M.B., O.W., and A.K.; writing—original draft preparation, L.v.d.S., M.B., Y.S., C.S.H., O.W. and A.K.; writing—review and editing, C.S.H.; visualization, L.v.d.S. and M.B.; supervision, A.K.; funding acquisition, L.v.d.S., O.W. and A.K. All authors have read and agreed to the published version of the manuscript.
Funding
This study was supported by the Stiftung Universitätsmedizin Essen (awarded to A. Krawczyk), the Else-Kröner Promotionskolleg ELAN (awarded to L. van de Sand), and the Rudolf Ackermann Foundation (awarded to O. Witzke).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Acknowledgments
The authors thank Delia Cosgrove for proofreading of the manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Pebody, R.; Andrews, N.; Brown, D.; Gopal, R.; Melker, H.; François, G.; Gatcheva, N.; Hellenbrand, W.; Jokinen, S.; Klavs, I.; et al. The seroepidemiology of herpes simplex virus type 1 and 2 in Europe. Sex. Transm. Infect. 2004, 80, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Berrington, W.R.; Jerome, K.R.; Cook, L.; Wald, A.; Corey, L.; Casper, C. Clinical correlates of herpes simplex virus viremia among hospitalized adults. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2009, 49, 1295–1301. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kimberlin, D.W.; Lin, C.Y.; Jacobs, R.F.; Powell, D.A.; Corey, L.; Gruber, W.C.; Rathore, M.; Bradley, J.S.; Diaz, P.S.; Kumar, M.; et al. Safety and efficacy of high-dose intravenous acyclovir in the management of neonatal herpes simplex virus infections. Pediatrics 2001, 108, 230–238. [Google Scholar] [CrossRef]
- Akinyi, B.; Odhiambo, C.; Otieno, F.; Inzaule, S.; Oswago, S.; Kerubo, E.; Ndivo, R.; Zeh, C. Prevalence, incidence and correlates of HSV-2 infection in an HIV incidence adolescent and adult cohort study in western Kenya. PLoS ONE 2017, 12, e0178907. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jiang, Y.-C.; Feng, H.; Lin, Y.-C.; Guo, X.-R. New strategies against drug resistance to herpes simplex virus. Int. J. Oral Sci. 2016, 8, 1–6. [Google Scholar] [CrossRef]
- Baeten, J.M.; Strick, L.B.; Lucchetti, A.; Whittington, W.L.; Sanchez, J.; Coombs, R.W.; Magaret, A.; Wald, A.; Corey, L.; Celum, C. Herpes simplex virus (HSV)-suppressive therapy decreases plasma and genital HIV-1 levels in HSV-2/HIV-1 coinfected women: A randomized, placebo-controlled, cross-over trial. J. Infect. Dis. 2008, 198, 1804–1808. [Google Scholar] [CrossRef] [Green Version]
- Delany, S.; Mlaba, N.; Clayton, T.; Akpomiemie, G.; Capovilla, A.; Legoff, J.; Belec, L.; Stevens, W.; Rees, H.; Mayaud, P. Impact of aciclovir on genital and plasma HIV-1 RNA in HSV-2/HIV-1 co-infected women: A randomized placebo-controlled trial in South Africa. AIDS 2009, 23, 461–469. [Google Scholar] [CrossRef] [Green Version]
- Nagot, N.; Ouédraogo, A.; Foulongne, V.; Konaté, I.; Weiss, H.A.; Vergne, L.; Defer, M.C.; Djagbaré, D.; Sanon, A.; Andonaba, J.B.; et al. Reduction of HIV-1 RNA levels with therapy to suppress herpes simplex virus. N. Engl. J. Med. 2007, 356, 790–799. [Google Scholar] [CrossRef] [Green Version]
- McMahon, M.A.; Siliciano, J.D.; Lai, J.; Liu, J.O.; Stivers, J.T.; Siliciano, R.F.; Kohli, R.M. The antiherpetic drug acyclovir inhibits HIV replication and selects the V75I reverse transcriptase multidrug resistance mutation. J. Biol. Chem. 2008, 283, 31289–31293. [Google Scholar] [CrossRef] [Green Version]
- Matamoros, T.; Kim, B.; Menéndez-Arias, L. Mechanistic insights into the role of Val75 of HIV-1 reverse transcriptase in misinsertion and mispair extension fidelity of DNA synthesis. J. Mol. Biol. 2008, 375, 1234–1248. [Google Scholar] [CrossRef] [Green Version]
- Farooq, A.V.; Shukla, D. Herpes simplex epithelial and stromal keratitis: An epidemiologic update. Surv. Ophthalmol. 2012, 57, 448–462. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stránská, R.; Schuurman, R.; Nienhuis, E.; Goedegebuure, I.W.; Polman, M.; Weel, J.F.; Wertheim-Van Dillen, P.M.; Berkhout, R.J.; van Loon, A.M. Survey of acyclovir-resistant herpes simplex virus in the Netherlands: Prevalence and characterization. J. Clin. Virol. 2005, 32, 7–18. [Google Scholar] [CrossRef] [PubMed]
- Sauerbrei, A.; Bohn, K.; Heim, A.; Hofmann, J.; Weissbrich, B.; Schnitzler, P.; Hoffmann, D.; Zell, R.; Jahn, G.; Wutzler, P.H.; et al. Novel resistance-associated mutations of thymidine kinase and DNA polymerase genes of herpes simplex virus type 1 and type 2. Antivir. Ther. 2011, 16, 1297–1308. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Blot, N.; Schneider, P.; Young, P.; Janvresse, C.; Dehesdin, D.; Tron, P.; Vannier, J.P. Treatment of an acyclovir and foscarnet-resistant herpes simplex virus infection with cidofovir in a child after an unrelated bone marrow transplant. Bone Marrow Transpl. 2000, 26, 903–905. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, D.; Su, A.; Fu, Y.; Wang, X.; Lv, X.; Xu, W.; Xu, S.; Wang, H.; Wu, Z. Harmine blocks herpes simplex virus infection through downregulating cellular NF-κB and MAPK pathways induced by oxidative stress. Antivir. Res. 2015, 123, 27–38. [Google Scholar] [CrossRef]
- Hutterer, C.; Milbradt, J.; Hamilton, S.; Zaja, M.; Leban, J.; Henry, C.; Vitt, D.; Steingruber, M.; Sonntag, E.; Zeitträger, I.; et al. Inhibitors of dual-specificity tyrosine phosphorylation-regulated kinases (DYRK) exert a strong anti-herpesviral activity. Antivir. Res. 2017, 143, 113–121. [Google Scholar] [CrossRef]
- Xiong, H.R.; Luo, J.; Hou, W.; Xiao, H.; Yang, Z.Q. The effect of emodin, an anthraquinone derivative extracted from the roots of Rheum tanguticum, against herpes simplex virus in vitro and in vivo. J. Ethnopharmacol. 2011, 133, 718–723. [Google Scholar] [CrossRef]
- Lyu, S.Y.; Rhim, J.Y.; Park, W.B. Antiherpetic activities of flavonoids against herpes simplex virus type 1 (HSV-1) and type 2 (HSV-2) in vitro. Arch. Pharm. Res. 2005, 28, 1293–1301. [Google Scholar] [CrossRef]
- Isaacs, C.E.; Wen, G.Y.; Xu, W.; Jia, J.H.; Rohan, L.; Corbo, C.; Di Maggio, V.; Jenkins, E.C., Jr.; Hillier, S. Epigallocatechin gallate inactivates clinical isolates of herpes simplex virus. Antimicrob. Agents Chemother. 2008, 52, 962–970. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- de Oliveira, A.; Adams, S.D.; Lee, L.H.; Murray, S.R.; Hsu, S.D.; Hammond, J.R.; Dickinson, D.; Chen, P.; Chu, T.C. Inhibition of herpes simplex virus type 1 with the modified green tea polyphenol palmitoyl-epigallocatechin gallate. Food Chem. Toxicol. 2013, 52, 207–215. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kuo, Y.C.; Lin, L.C.; Tsai, W.J.; Chou, C.J.; Kung, S.H.; Ho, Y.H. Samarangenin B from Limonium sinense suppresses herpes simplex virus type 1 replication in Vero cells by regulation of viral macromolecular synthesis. Antimicrob. Agents Chemother. 2002, 46, 2854–2864. [Google Scholar] [CrossRef] [Green Version]
- Lin, L.C.; Kuo, Y.C.; Chou, C.J. Anti-herpes simplex virus type-1 flavonoids and a new flavanone from the root of Limonium sinense. Planta Med. 2000, 66, 333–336. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.Y.; Lin, C.C.; Lin, T.C. Antiherpes simplex virus type 2 activity of casuarinin from the bark of Terminalia arjuna Linn. Antivir. Res. 2002, 55, 447–455. [Google Scholar] [CrossRef]
- Luo, Z.; Kuang, X.P.; Zhou, Q.Q.; Yan, C.Y.; Li, W.; Gong, H.B.; Kurihara, H.; Li, W.X.; Li, Y.F.; He, R.R. Inhibitory effects of baicalein against herpes simplex virus type 1. Acta Pharm. Sin. B 2020, 10, 2323–2338. [Google Scholar] [CrossRef]
- Isaacs, C.E.; Xu, W.; Merz, G.; Hillier, S.; Rohan, L.; Wen, G.Y. Digallate dimers of (-)-epigallocatechin gallate inactivate herpes simplex virus. Antimicrob. Agents Chemother. 2011, 55, 5646–5653. [Google Scholar] [CrossRef] [Green Version]
- Li, T.; Liu, L.; Wu, H.; Chen, S.; Zhu, Q.; Gao, H.; Yu, X.; Wang, Y.; Su, W.; Yao, X.; et al. Anti-herpes simplex virus type 1 activity of Houttuynoid A, a flavonoid from Houttuynia cordata Thunb. Antivir. Res. 2017, 144, 273–280. [Google Scholar] [CrossRef]
- Hung, P.Y.; Ho, B.C.; Lee, S.Y.; Chang, S.Y.; Kao, C.L.; Lee, S.S.; Lee, C.N. Houttuynia cordata targets the beginning stage of herpes simplex virus infection. PLoS ONE 2015, 10, e0115475. [Google Scholar] [CrossRef] [Green Version]
- Chiang, L.C.; Chiang, W.; Liu, M.C.; Lin, C.C. In vitro antiviral activities of Caesalpinia pulcherrima and its related flavonoids. J. Antimicrob. Chemother. 2003, 52, 194–198. [Google Scholar] [CrossRef] [Green Version]
- Cheng, H.Y.; Lin, T.C.; Yang, C.M.; Wang, K.C.; Lin, C.C. Mechanism of action of the suppression of herpes simplex virus type 2 replication by pterocarnin A. Microbes Infect. 2004, 6, 738–744. [Google Scholar] [CrossRef] [PubMed]
- Bag, P.; Ojha, D.; Mukherjee, H.; Halder, U.C.; Mondal, S.; Chandra, N.S.; Nandi, S.; Sharon, A.; Sarkar, M.C.; Chakrabarti, S.; et al. An Indole Alkaloid from a Tribal Folklore Inhibits Immediate Early Event in HSV-2 Infected Cells with Therapeutic Efficacy in Vaginally Infected Mice. PLoS ONE 2013, 8, e77937. [Google Scholar] [CrossRef]
- Bag, P.; Ojha, D.; Mukherjee, H.; Halder, U.C.; Mondal, S.; Biswas, A.; Sharon, A.; Van Kaer, L.; Chakrabarty, S.; Das, G.; et al. A dihydro-pyrido-indole potently inhibits HSV-1 infection by interfering the viral immediate early transcriptional events. Antivir. Res. 2014, 105, 126–134. [Google Scholar] [CrossRef]
- Hassan, S.T.S.; Berchová-Bímová, K.; Šudomová, M.; Malaník, M.; Šmejkal, K.; Rengasamy, K.R.R. In Vitro Study of Multi-Therapeutic Properties of Thymus bovei Benth. Essential Oil and Its Main Component for Promoting Their Use in Clinical Practice. J. Clin. Med. 2018, 7, 283. [Google Scholar] [CrossRef] [Green Version]
- Ikeda, T.; Yokomizo, K.; Okawa, M.; Tsuchihashi, R.; Kinjo, J.; Nohara, T.; Uyeda, M. Anti-herpes virus type 1 activity of oleanane-type triterpenoids. Biol. Pharm. Bull. 2005, 28, 1779–1781. [Google Scholar] [CrossRef] [Green Version]
- Huang, W.; Chen, X.; Li, Q.; Li, P.; Zhao, G.; Xu, M.; Xie, P. Inhibition of intercellular adhesion in herpex simplex virus infection by glycyrrhizin. Cell Biochem. Biophys. 2012, 62, 137–140. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.T.; Chen, T.Y.; Chung, C.Y.; Noyce, R.S.; Grindley, T.B.; McCormick, C.; Lin, T.C.; Wang, G.H.; Lin, C.C.; Richardson, C.D. Hydrolyzable tannins (chebulagic acid and punicalagin) target viral glycoprotein-glycosaminoglycan interactions to inhibit herpes simplex virus 1 entry and cell-to-cell spread. J. Virol. 2011, 85, 4386–4398. [Google Scholar] [CrossRef] [Green Version]
- Kesharwani, A.; Polachira, S.K.; Nair, R.; Agarwal, A.; Mishra, N.N.; Gupta, S.K. Anti-HSV-2 activity of Terminalia chebula Retz extract and its constituents, chebulagic and chebulinic acids. BMC Complement. Altern. Med. 2017, 17, 110. [Google Scholar] [CrossRef] [Green Version]
- Kratz, J.M.; Andrighetti-Fröhner, C.R.; Kolling, D.J.; Leal, P.C.; Cirne-Santos, C.C.; Yunes, R.A.; Nunes, R.J.; Trybala, E.; Bergström, T.; Frugulhetti, I.C.; et al. Anti-HSV-1 and anti-HIV-1 activity of gallic acid and pentyl gallate. Mem. Inst. Oswaldo Cruz. 2008, 103, 437–442. [Google Scholar] [CrossRef] [Green Version]
- Kratz, J.M.; Andrighetti-Fröhner, C.R.; Leal, P.C.; Nunes, R.J.; Yunes, R.A.; Trybala, E.; Bergström, T.; Barardi, C.R.; Simões, C.M. Evaluation of anti-HSV-2 activity of gallic acid and pentyl gallate. Biol. Pharm. Bull. 2008, 31, 903–907. [Google Scholar] [CrossRef] [Green Version]
- El-Toumy, S.; Salib, J.; El-Kashak, W.; Marty, C.; Bedoux, G.; Bourgougnon, N. Antiviral effect of polyphenol rich plant extracts on herpes simplex virus type 1. Food Sci. Hum. Wellness 2018, 7, 91–101. [Google Scholar] [CrossRef]
- Kutluay, S.B.; Doroghazi, J.; Roemer, M.E.; Triezenberg, S.J. Curcumin inhibits herpes simplex virus immediate-early gene expression by a mechanism independent of p300/CBP histone acetyltransferase activity. Virology 2008, 373, 239–247. [Google Scholar] [CrossRef] [Green Version]
- Martins, F.O.; Esteves, P.F.; Mendes, G.S.; Barbi, N.S.; Menezes, F.S.; Romanos, M.T. Verbascoside isolated from Lepechinia speciosa has inhibitory activity against HSV-1 and HSV-2 in vitro. Nat. Prod. Commun. 2009, 4, 1693–1696. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- de Sousa Cardozo, F.T.G.; Camelini, C.M.; Mascarello, A.; José Rossi, M.; José Nunes, R.; Monte Barardi, C.R.; de Mendonça, M.M.; Simões, C.M.O. Antiherpetic activity of a sulfated polysaccharide from Agaricus brasiliensis mycelia. Antivir. Res. 2011, 92, 108–114. [Google Scholar] [CrossRef] [Green Version]
- Marchetti, M.; Pisani, S.; Pietropaolo, V.; Seganti, L.; Nicoletti, R.; Degener, A.; Orsi, N. Antiviral effect of a polysaccharide from Sclerotium glucanicum towards herpes simplex virus type 1 infection. Planta Med. 1996, 62, 303–307. [Google Scholar] [CrossRef] [PubMed]
- Álvarez, Á.; Habtemariam, S.L.; Abdel Moneim, A.E.; Melón, S.; Dalton, K.P.; Parra, F. A spiroketal-enol ether derivative from Tanacetum vulgare selectively inhibits HSV-1 and HSV-2 glycoprotein accumulation in Vero cells. Antivir. Res. 2015, 119, 8–18. [Google Scholar] [CrossRef]
- da Rosa Guimarães, T.; Quiroz, C.G.; Borges, C.R.; de Oliveira, S.Q.; de Almeida, M.T.; Bianco, É.M.; Moritz, M.I.; Carraro, J.L.; Palermo, J.A.; Cabrera, G.; et al. Anti HSV-1 activity of halistanol sulfate and halistanol sulfate C isolated from Brazilian marine sponge Petromica citrina (Demospongiae). Mar. Drugs 2013, 11, 4176–4192. [Google Scholar] [CrossRef]
- Ma, F.; Shen, W.; Zhang, X.; Li, M.; Wang, Y.; Zou, Y.; Li, Y.; Wang, H. Anti-HSV Activity of Kuwanon X from Mulberry Leaves with Genes Expression Inhibitory and HSV-1 Induced NF-κB Deactivated Properties. Biol. Pharm. Bull. 2016, 39, 1667–1674. [Google Scholar] [CrossRef] [Green Version]
- Chuanasa, T.; Phromjai, J.; Lipipun, V.; Likhitwitayawuid, K.; Suzuki, M.; Pramyothin, P.; Hattori, M.; Shiraki, K. Anti-herpes simplex virus (HSV-1) activity of oxyresveratrol derived from Thai medicinal plant: Mechanism of action and therapeutic efficacy on cutaneous HSV-1 infection in mice. Antivir. Res. 2008, 80, 62–70. [Google Scholar] [CrossRef]
- Likhitwitayawuid, K.; Sritularak, B.; Benchanak, K.; Lipipun, V.; Mathew, J.; Schinazi, R.F. Phenolics with antiviral activity from Millettia erythrocalyx and Artocarpus lakoocha. Nat. Prod. Res. 2005, 19, 177–182. [Google Scholar] [CrossRef]
- Krawczyk, E.; Luczak, M.; Kniotek, M.; Nowaczyk, M. Cytotoxic, antiviral (in-vitro and in-vivo), immunomodulatory activity and influence on mitotic divisions of three taxol derivatives: 10-deacetyl-baccatin III, methyl (N-benzoyl-(2′R,3′S)-3′-phenylisoserinate) and N-benzoyl-(2′R,3′S)-3′-phenylisoserine. J. Pharm. Pharm. 2005, 57, 791–797. [Google Scholar] [CrossRef]
- Kim, M.; Kim, S.K.; Park, B.N.; Lee, K.H.; Min, G.H.; Seoh, J.Y.; Park, C.G.; Hwang, E.S.; Cha, C.Y.; Kook, Y.H. Antiviral effects of 28-deacetylsendanin on herpes simplex virus-1 replication. Antivir. Res. 1999, 43, 103–112. [Google Scholar] [CrossRef]
- Armaka, M.; Papanikolaou, E.; Sivropoulou, A.; Arsenakis, M. Antiviral properties of isoborneol, a potent inhibitor of herpes simplex virus type 1. Antivir. Res. 1999, 43, 79–92. [Google Scholar] [CrossRef]
- Barquero, A.A.; Michelini, F.M.; Alché, L.E. 1-Cinnamoyl-3,11-dihydroxymeliacarpin is a natural bioactive compound with antiviral and nuclear factor-kappaB modulating properties. Biochem. Biophys. Res. Commun. 2006, 344, 955–962. [Google Scholar] [CrossRef]
- Bueno, C.A.; Barquero, A.A.; Di Cónsoli, H.; Maier, M.S.; Alché, L.E. A natural tetranortriterpenoid with immunomodulating properties as a potential anti-HSV agent. Virus Res. 2009, 141, 47–54. [Google Scholar] [CrossRef]
- Alché, L.E.; Assad Ferek, G.; Meo, M.; Coto, C.E.; Maier, M.S. An Antiviral Meliacarpin from Leaves of Melia azedarach L. Z. Für. Nat. C 2003, 58, 215–219. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, K.; Hayashi, T.; Ujita, K.; Takaishi, Y. Characterization of antiviral activity of a sesquiterpene, triptofordin C-2. J. Antimicrob. Chemother. 1996, 37, 759–768. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hassan, S.T.S.; Šudomová, M.; Berchová-Bímová, K.; Šmejkal, K.; Echeverría, J. Psoromic Acid, a Lichen-Derived Molecule, Inhibits the Replication of HSV-1 and HSV-2, and Inactivates HSV-1 DNA Polymerase: Shedding Light on Antiherpetic Properties. Molecules 2019, 24, 2912. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nixon, B.; Stefanidou, M.; Mesquita, P.M.; Fakioglu, E.; Segarra, T.; Rohan, L.; Halford, W.; Palmer, K.E.; Herold, B.C. Griffithsin protects mice from genital herpes by preventing cell-to-cell spread. J. Virol. 2013, 87, 6257–6269. [Google Scholar] [CrossRef] [Green Version]
- Torres, N.I.; Noll, K.S.; Xu, S.; Li, J.; Huang, Q.; Sinko, P.J.; Wachsman, M.B.; Chikindas, M.L. Safety, formulation, and in vitro antiviral activity of the antimicrobial peptide subtilosin against herpes simplex virus type 1. Probiotics Antimicrob. Proteins 2013, 5, 26–35. [Google Scholar] [CrossRef] [Green Version]
- Quintana, V.M.; Torres, N.I.; Wachsman, M.B.; Sinko, P.J.; Castilla, V.; Chikindas, M. Antiherpes simplex virus type 2 activity of the antimicrobial peptide subtilosin. J. Appl. Microbiol. 2014, 117, 1253–1259. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, H.; Chan, H.; Wang, Y.Y.; Ouyang, D.Y.; Zheng, Y.T.; Tam, S.C. Trichosanthin suppresses the elevation of p38 MAPK, and Bcl-2 induced by HSV-1 infection in Vero cells. Life Sci. 2006, 79, 1287–1292. [Google Scholar] [CrossRef]
- He, D.X.; Tam, S.C. Trichosanthin affects HSV-1 replication in Hep-2 cells. Biochem. Biophys. Res. Commun. 2010, 402, 670–675. [Google Scholar] [CrossRef]
- Zheng, Y.T.; Chan, W.L.; Chan, P.; Huang, H.; Tam, S.C. Enhancement of the anti-herpetic effect of trichosanthin by acyclovir and interferon. FEBS Lett. 2001, 496, 139–142. [Google Scholar] [CrossRef] [Green Version]
- Spear, P.G. Herpes simplex virus: Receptors and ligands for cell entry. Cell Microbiol. 2004, 6, 401–410. [Google Scholar] [CrossRef] [PubMed]
- Däumer, M.P.; Schneider, B.; Giesen, D.M.; Aziz, S.; Kaiser, R.; Kupfer, B.; Schneweis, K.E.; Schneider-Mergener, J.; Reineke, U.; Matz, B.; et al. Characterisation of the epitope for a herpes simplex virus glycoprotein B-specific monoclonal antibody with high protective capacity. Med. Microbiol. Immunol. 2011, 200, 85–97. [Google Scholar] [CrossRef] [PubMed]
- Patel, A.; Hanson, J.; McLean, T.I.; Olgiate, J.; Hilton, M.; Miller, W.E.; Bachenheimer, S.L. Herpes Simplex Virus Type 1 Induction of Persistent NF-κB Nuclear Translocation Increases the Efficiency of Virus Replication. Virology 1998, 247, 212–222. [Google Scholar] [CrossRef] [Green Version]
- Goodkin, M.L.; Ting, A.T.; Blaho, J.A. NF-κB Is Required for Apoptosis Prevention during Herpes Simplex Virus Type 1 Infection. J. Virol. 2003, 77, 7261–7280. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chiang, L.C.; Cheng, H.Y.; Liu, M.C.; Chiang, W.; Lin, C.C. In vitro anti-herpes simplex viruses and anti-adenoviruses activity of twelve traditionally used medicinal plants in Taiwan. Biol. Pharm. Bull. 2003, 26, 1600–1604. [Google Scholar] [CrossRef] [Green Version]
- Vitali, D.; Bagri, P.; Wessels, J.M.; Arora, M.; Ganugula, R.; Parikh, A.; Mandur, T.; Felker, A.; Garg, S.; Kumar, M.N.V.R.; et al. Curcumin Can Decrease Tissue Inflammation and the Severity of HSV-2 Infection in the Female Reproductive Mucosa. Int. J. Mol. Sci. 2020, 21, 337. [Google Scholar] [CrossRef] [Green Version]
- Cardozo, F.T.G.S.; Larsen, I.V.; Carballo, E.V.; Jose, G.; Stern, R.A.; Brummel, R.C.; Camelini, C.M.; Rossi, M.J.; Simões, C.M.O.; Brandt, C.R. In VivoAnti-Herpes Simplex Virus Activity of a Sulfated Derivative of Agaricus brasiliensis Mycelial Polysaccharide. Antimicrob. Agents Chemother. 2013, 57, 2541–2549. [Google Scholar] [CrossRef] [Green Version]
- Petrera, E.; Coto, C.E. Therapeutic effect of meliacine, an antiviral derived from Melia azedarach L.; in mice genital herpetic infection. Phytother. Res. 2009, 23, 1771–1777. [Google Scholar] [CrossRef]
- Alché, L.E.; Berra, A.; Veloso, M.J.; Coto, C.E. Treatment with meliacine, a plant derived antiviral, prevents the development of herpetic stromal keratitis in mice. J. Med. Virol. 2000, 61, 474–480. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).