Seoul Virus in Pet and Feeder Rats in The Netherlands
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Dissection
2.3. Molecular Diagnostics
2.4. Serology
2.5. Comparison of RT-qPCR Detection in Fresh and Frozen Tissue
2.6. Ethics Statement
3. Results
3.1. Rats
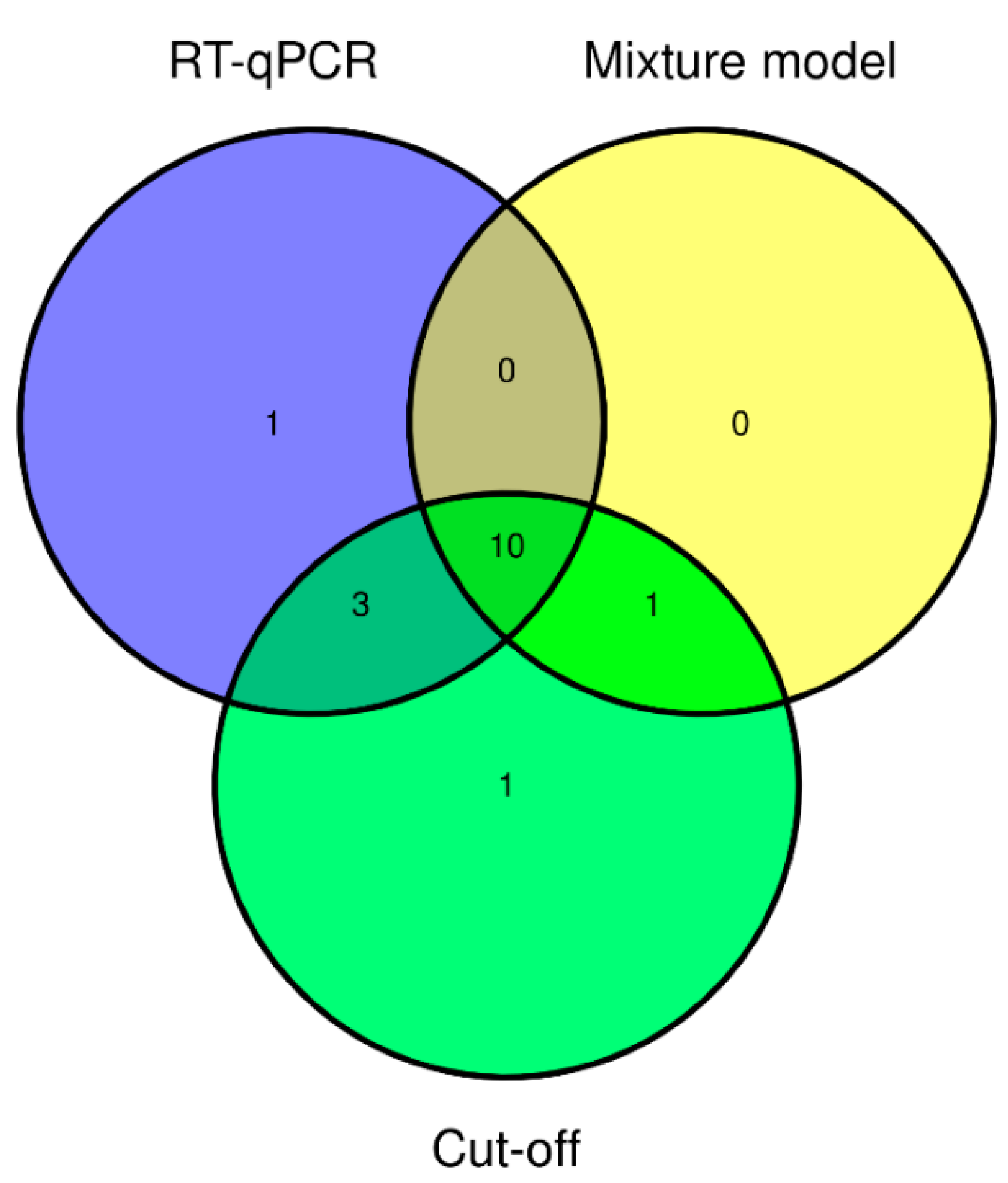
3.2. SEOV Infections
3.3. Questionnaires
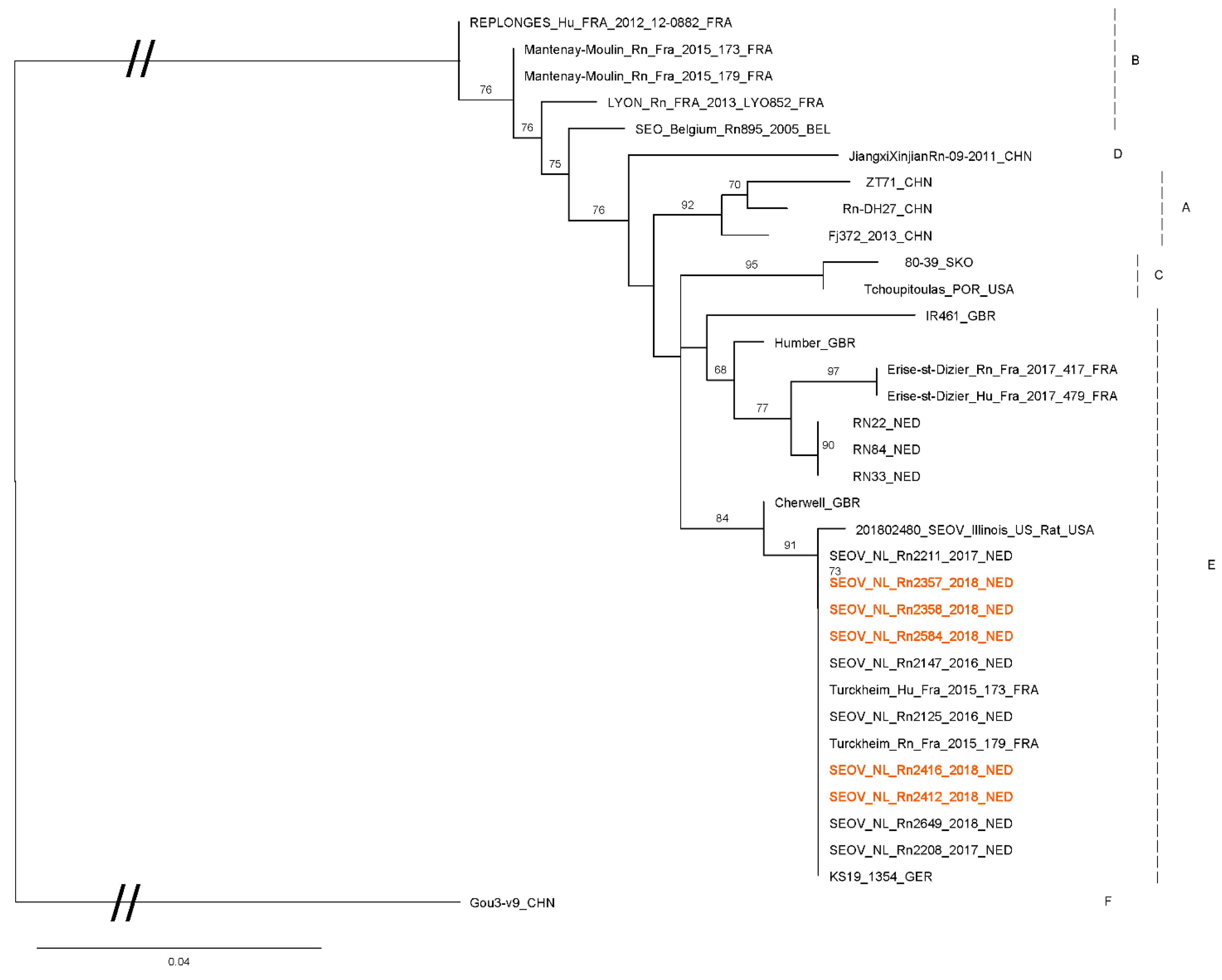
3.4. Phylogenetics
3.5. Comparison of RT-qPCR Detection in Fresh and Frozen Material
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Clement, J.; LeDuc, J.W.; McElhinney, L.M.; Reynes, J.-M.; Van Ranst, M.; Calisher, C.H. Clinical Characteristics of Ratborne Seoul Hantavirus Disease. Emerg. Infect. Dis. 2019, 25, 387–388. [Google Scholar] [CrossRef]
- Jonsson, C.B.; Figueiredo, L.T.M.; Vapalahti, O. A Global Perspective on Hantavirus Ecology, Epidemiology, and Disease. Clin. Microbiol. Rev. 2010, 23, 412–441. [Google Scholar] [CrossRef]
- Kim, Y.S.; Ahn, C.; Han, J.S.; Kim, S.; Lee, J.S.; Lee, P.W. Hemorrhagic Fever with Renal Syndrome Caused by the Seoul Virus. Nephron 1995, 71, 419–427. [Google Scholar] [CrossRef]
- Clement, J.; LeDuc, J.W.; Lloyd, G.; Reynes, J.M.; McElhinney, L.; Van Ranst, M.; Lee, H.W. Wild Rats, Laboratory Rats, Pet Rats: Global Seoul Hantavirus Disease Revisited. Viruses 2019, 11, 652. [Google Scholar] [CrossRef]
- Swanink, C.; Reimerink, J.; Gisolf, J.; De Vries, A.; Claassen, M.; Martens, L.; Waegemaekers, T.; Rozendaal, H.; Valkenburgh, S.; Hoornweg, T.; et al. Autochthonous Human Case of Seoul Virus Infection, the Netherlands. Emerg. Infect. Dis. 2018, 24, 2158–2163. [Google Scholar] [CrossRef] [PubMed]
- Kerins, J.L.; Koske, S.E.; Kazmierczak, J.; Austin, C.; Gowdy, K.; Dibernardo, A. Outbreak of Seoul virus among rats and rat owners—United States and Canada. Can. Commun. Dis. Rep. 2018, 44, 71–74. [Google Scholar] [CrossRef] [PubMed]
- Reynes, J.M.; Carli, D.; Bour, J.B.; Boudjeltia, S.; Dewilde, A.; Gerbier, G.; Nussbaumer, T.; Jacomo, V.; Rapt, M.P.; Rollin, P.E.; et al. Seoul Virus Infection in Humans, France, 2014. Emerg. Infect. Dis. 2017, 23, 973–977. [Google Scholar] [CrossRef]
- Jameson, L.J.; Taori, S.K.; Atkinson, B.; Levick, P.; Featherstone, C.A.; Van Der Burgt, G.; McCarthy, N.; Hart, J.; Osborne, J.C.; Walsh, A.L.; et al. Pet rats as a source of hantavirus in England and Wales. Eurosurveillance 2013, 18, 415. [Google Scholar]
- McElhinney, L.M.; Marston, D.A.; Pounder, K.C.; Goharriz, H.; Wise, E.L.; Carlsson, V.J.; Jennings, D.; Johnson, N.; Civello, A.; Nunez, A.; et al. High prevalence of Seoul hantavirus in a breeding colony of pet rats. Epidemiol. Infect. 2017, 145, 3115–3124. [Google Scholar] [CrossRef] [PubMed]
- Murphy, E.G.; Williams, N.J.; Bennett, M.; Jennings, D.; Chantrey, J.; McElhinney, L.M. Detection of Seoul virus in wild brown rats (Rattus norvegicus) from pig farms in Northern England. Veter. Rec. 2019, 184, 525. [Google Scholar] [CrossRef]
- Maas, M.; De Vries, A.; Reusken, C.; Buijs, J.; Goris, M.; Hartskeerl, R.; Ahmed, A.; Van Tulden, P.; Swart, A.; Pijnacker, R.; et al. Prevalence of Leptospira spp. and Seoul hantavirus in brown rats (Rattus norvegicus) in four regions in the Netherlands, 2011. Infect. Ecol. Epidemiol. 2018, 8, 135. [Google Scholar] [CrossRef]
- Carlsson, V.J.; Lõhmus, M.; Sundström, K.; Strand, T.M.; Verkerk, M.; Reusken, C.; Yoshimatsu, K.; Arikawa, J.; Van De Goot, F.; Lundkvist, Å. First evidence of Seoul hantavirus in the wild rat population in the Netherlands. Infect. Ecol. Epidemiol. 2015, 5, 7215. [Google Scholar] [CrossRef][Green Version]
- Dupinay, T.; Pounder, K.C.; Ayral, F.; Laaberki, M.H.; Marston, D.A.; Lacote, S.; Rey, C.; Barbet, F.; Voller, K.; Nazaret, N.; et al. Detection and genetic characterization of Seoul Virus from commensal brown rats in France. Virol. J. 2014, 11, 32. [Google Scholar] [CrossRef] [PubMed]
- Meyer, B.J.; Schmaljohn, C.S. Persistent hantavirus infections: Characteristics and mechanisms. Trends Microbiol. 2000, 8, 61–67. [Google Scholar] [CrossRef]
- Kramski, M.; Meisel, H.; Klempa, B.; Krüger, D.H.; Pauli, G.; Nitsche, A. Detection and Typing of Human Pathogenic Hantaviruses by Real-Time Reverse Transcription-PCR and Pyrosequencing. Clin. Chem. 2007, 53, 1899–1905. [Google Scholar] [CrossRef] [PubMed]
- Kariwa, H.; Kimura, M.; Yoshizumi, S.; Arikawa, J.; Yoshimatsu, K.; Takashima, I.; Hashimoto, N. Modes of Seoul virus infections: Persistency in newborn rats and transiency in adult rats. Arch. Virol. 1996, 141, 2327–2338. [Google Scholar] [CrossRef]
- Nguyen, L.T.; Schmidt, H.A.; von Haeseler, A.; Minh, B.Q. IQ-TREE: A fast and effective stochastic algorithm for estimating maximum-likelihood phylogenies. Mol. Biol. Evol. 2015, 32, 268–274. [Google Scholar] [CrossRef]
- Minh, B.Q.; Nguyen, M.A.T.; Von Haeseler, A. Ultrafast Approximation for Phylogenetic Bootstrap. Mol. Biol. Evol. 2013, 30, 1188–1195. [Google Scholar] [CrossRef] [PubMed]
- Rambaut, A. FigTree: Tree Figure Drawing Tool, v.1.4.4. 2009. Available online: http://tree.bio.ed.ac.uk/software/figtree (accessed on 4 March 2021).
- Maas, M.; Van Heteren, M.; De Vries, A.; Kuiken, T.; Hoornweg, T.; Kroeze, E.V.; Rockx, B. Seoul Virus Tropism and Pathology in Naturally Infected Feeder Rats. Viruses 2019, 11, 531. [Google Scholar] [CrossRef]
- Swart, A.; Maas, M.; de Vries, A.; Cuperus, T.; Opsteegh, M. Bayesian Binary Mixture Models as a Flexible Alternative to Cut-off Analysis of ELISA Results, a Case Study of Seoul Orthohantavirus. 2021; under review. [Google Scholar]
- Hoornweg, T.E.; Zutt, I.; De Vries, A.; Maas, M.; Hoogerwerf, M.N.; Županc, A.T.; Korva, M.; Reimerink, J.H.J.; Reusken, C.B.E.M. Development of a Comparative European Orthohantavirus Microneutralization Assay with Multi-Species Validation and Evaluation in a Human Diagnostic Cohort. Front. Cell. Infect. Microbiol. 2020, 10. [Google Scholar] [CrossRef]
- Kim, W.K.; No, J.S.; Lee, S.H.; Song, D.H.; Lee, D.; Kim, J.A.; Gu, S.H.; Park, S.; Jeong, S.T.; Kim, H.C.; et al. Multiplex PCR−Based Next-Generation Sequencing and Global Diversity of Seoul Virus in Humans and Rats. Emerg. Infect. Dis. 2018, 24, 249–257. [Google Scholar] [CrossRef]
- Knust, B.; Brown, S.; de Maurice, S.A.; Whitmer, S.; Koske, S.E.; Ervin, E.; Patel, K.; Graziano, J.; Betoulle, M.M.E.; House, J.; et al. Seoul virus infection and spread in US home-based ratteries—Rat and human testing results from a multistate outbreak investigation. J. Infect. Dis. 2020, 222, 1311–1319. [Google Scholar] [CrossRef]
- Osterhaus, A.D.; Spijkers, I.; Van Steenis, G.; Van Der Groen, G. Hantavirus infections in the Netherlands. Ned. Tijdschr. Geneeskd. 1984, 128, 2461–2462. [Google Scholar]
- Lloyd, G.; Bowen, E.; Jones, N.; Pendry, A. HFRS Outbreak Associated with Laboratory Rats in UK. Lancet 1984, 323, 1175–1176. [Google Scholar] [CrossRef]
- Desmyter, J.; LeDuc, J.W.; Johnson, K.M.; Brasseur, F.; Deckers, C.; van de Strihou, Y.C. Laboratory rat associ-ated outbreak of haemorrhagic fever with renal syndrome due to Hantaan-like virus in Belgium. Lancet 1983, 2, 1445–1448. [Google Scholar] [CrossRef]
- Convenor, M.M.; Berard, M.; Feinstein, R.; Gallagher, A.; Wilcke, I.B.; Corning, P.K.; Raspa, M. FELASA recommendations for the health monitoring of mouse, rat, hamster, guinea pig and rabbit colonies in breeding and experimental units. Lab. Anim. 2014, 48, 178–192. [Google Scholar]
- Ling, J.; Carlsson, V.J.; Eriksson, P.; Plyusnina, A.; Löhmus, M.; Järhult, J.D.; Van De Goot, F.; Plyusnin, A.; Lundkvist, Å.; Sironen, T. Genetic analyses of Seoul hantavirus genome recovered from rats (Rattus norvegicus) in the Netherlands unveils diverse routes of spread into Europe. J. Med Virol. 2019, 91, 724–730. [Google Scholar] [CrossRef]
- Compton, S.R.; Jacoby, R.O.; Paturzo, F.X.; Smith, A.L. Persistent Seoul virus infection in Lewis rats. Arch. Virol. 2004, 149, 1325–1339. [Google Scholar] [CrossRef] [PubMed]
- Robin, C.; Perkins, E.; Watkins, F.; Christley, R. Pets, Purity and Pollution: Why Conventional Models of Disease Trans-mission Do Not Work for Pet Rat Owners. Int. J. Environ. Res. Public Health 2017, 14, 1526. [Google Scholar] [CrossRef]

| Source | Number of Participants | Total Number of Rats Collected | Number of Rats per Participant (Range) |
|---|---|---|---|
| Private owners via veterinarians | 8 veterinarians 1 | 29 | 2–11 |
| Ratteries and rat shelters | 5 ratteries, 2 shelters | 56 | 5–10 |
| Commercial breeders and traders | 9 | 90 | 10 |
| Source | Total Number of Rats | Nr. of Rats RT-qPCR+ (Percentage) | Nr. of Rats ELISA+ Based on Cut-Off 1 (Percentage) | Nr. of Rats ELISA+ Based on Mixture Model (Percentage) |
|---|---|---|---|---|
| Private owners via veterinarians | 29 | 1 (3.4%) | 1 (3.4%) | 1 (3.4%) |
| Ratteries and rat shelters | 56 | 2 (3.6%) | 3 (5.4%) | 1 (1.8%) |
| Commercial breeders and traders | 90 | 11 (12.2%) | 11 (12.2%) | 9 (10.0%) |
| Rat ID | RT-qPCR Result on Fresh Tissue (Cq Value) | RT-qPCR Result on Frozen Tissue (Cq Value) |
|---|---|---|
| RN2352 | - | - |
| RN2353 | +(27.83) | - |
| RN2354 | +(30.11) | +(26.53) |
| RN2355 | - | - |
| RN2356 | - | +(28.38) |
| RN2357 | +(25.61) | +(25.72) |
| RN2358 | +(23.68) | +(22.61) |
| RN2359 | +(26.67) | +(27.91) |
| RN2360 | - | - |
| RN2361 | +(29.84) | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cuperus, T.; de Vries, A.; Hoornweg, T.E.; Fonville, M.; Jaarsma, R.I.; Opsteegh, M.; Maas, M. Seoul Virus in Pet and Feeder Rats in The Netherlands. Viruses 2021, 13, 443. https://doi.org/10.3390/v13030443
Cuperus T, de Vries A, Hoornweg TE, Fonville M, Jaarsma RI, Opsteegh M, Maas M. Seoul Virus in Pet and Feeder Rats in The Netherlands. Viruses. 2021; 13(3):443. https://doi.org/10.3390/v13030443
Chicago/Turabian StyleCuperus, Tryntsje, Ankje de Vries, Tabitha E. Hoornweg, Manoj Fonville, Ryanne I. Jaarsma, Marieke Opsteegh, and Miriam Maas. 2021. "Seoul Virus in Pet and Feeder Rats in The Netherlands" Viruses 13, no. 3: 443. https://doi.org/10.3390/v13030443
APA StyleCuperus, T., de Vries, A., Hoornweg, T. E., Fonville, M., Jaarsma, R. I., Opsteegh, M., & Maas, M. (2021). Seoul Virus in Pet and Feeder Rats in The Netherlands. Viruses, 13(3), 443. https://doi.org/10.3390/v13030443






