Abstract
Epigeic bryophytes represent an important but often overlooked component of forest biodiversity, closely linked to fine-scale habitat heterogeneity. The conducted research revealed clear differences in both species richness and composition between phytocoenoses and substrates. Mineral soil was the most species-rich substrate, hosting 90 taxa, whereas litter supported the lowest diversity, with only 33 species, emphasising the role of substrate stability and microhabitat availability in shaping bryophyte assemblages. Although forest ecosystems and forest roads exhibited comparable overall species richness, their bryophyte floras differed markedly in species composition, indicating that non-forest habitats provide distinct ecological niches and function as refugia for rare and restricted taxa. Analyses demonstrated that substrates with similar properties often formed coherent species groups across different phytocoenoses and may play a more important role than forest type in structuring epigeic bryophyte communities. On the other hand, species with broad ecological amplitudes were widespread and structurally dominant, whereas taxa restricted to single phytocoenoses showed high bioindicative value. These restricted species proved particularly useful for differentiating phytocoenoses despite their limited spatial extent. Overall, the results highlight bryophytes’ sensitivity to microhabitat variation and underscore their value as effective indicators of habitat differentiation in forest ecosystems.
1. Introduction
Bryophytes are among the least-studied groups of plants; consequently, our knowledge of their biodiversity, distribution, and ecological preferences remains limited and often relies on historical data. This is especially evident in Central Poland, which remains one of the least researched regions in the country in this regard, despite a lengthy history of bryological research [1,2].
Mosses and liverworts are common components of most phytocoenoses in which individual plant communities differ not only in species richness and frequency of particular taxa, but also in the degree of development and cover of the bryophyte layer [3,4,5,6,7]. The variations arise not only from the extent of preservation and naturalness of specific ecosystems but, more importantly, from the species composition of vascular plants that constitute a particular phytocoenosis. In forest ecosystems, this is predominantly evident in the species composition of the tree stand. Consequently, it is widely accepted that different pools of moss and liverwort species are recorded in deciduous forests than in coniferous or mixed forests (e.g., refs. [4,8,9]).
In the lowland forest ecosystems of Europe, mosses and liverworts, unlike vascular plants, colonise not only soil but also all available substrates. As a result, several ecological groups can be distinguished, e.g., epigeic, epixylic, epiphytic, or, much rarer in lowland areas, epilithic bryophytes. All of these substrates and the species growing on them have been the subject of detailed studies for many years [8,10,11,12]. However, the most extensive information concerns epigeic species, as they not only constitute a component of virtually all bryological studies but also represent an important element of phytosociological research widely applied in Europe [13,14].
The studies referenced [13,14] indicate that, in addition to lichens and vascular plants, bryophytes were identified as a key element in every examined plant community. Nevertheless, for many years, their significance in the categorization of specific syntaxa and, consequently, their value as bioindicators have been overlooked [15,16,17]. This situation has begun to change in recent years, as classical ecological and phytosociological studies have been supported by machine learning approaches, thereby revealing a markedly different view of nature and the real bioindicative role of bryophytes in the ecosystems surrounding us (e.g., refs. [6,7,18]), however these cited studies refer primarily to the lowest syntaxonomic units. On the other hand, in European syntaxonomic frameworks, bryophytes are relatively rarely listed as indicative of higher-ranking units, such as classes, orders, or alliances [15,19,20,21,22,23,24,25,26,27].
Taking the above facts into account, this study hypothesised that the pools of epigeic bryophyte species inhabiting individual forest phytocoenoses differ from one another both in species composition and in quantitative structure. At the same time, it was assumed that the epigeic bryophyte flora of deciduous forests differs unambiguously from that of coniferous forests. Furthermore, it was hypothesised that within the studied forest alliances it is possible to distinguish epigeic bryophyte species restricted to specific phytocoenoses, which may function as indicators and differentiate a given syntaxon from the others.
Accordingly, the aims of the study were to: (i) investigate the species richness of the epigeic bryophyte flora of the studied phytocoenoses; (ii) analyse the similarity of epigeic substrates among individual alliances; (iii) examine the substrate and phytocoenotic preferences of individual taxa; and (iv) assess their bioindicative potential.
2. Materials and Methods
2.1. Sampling Design
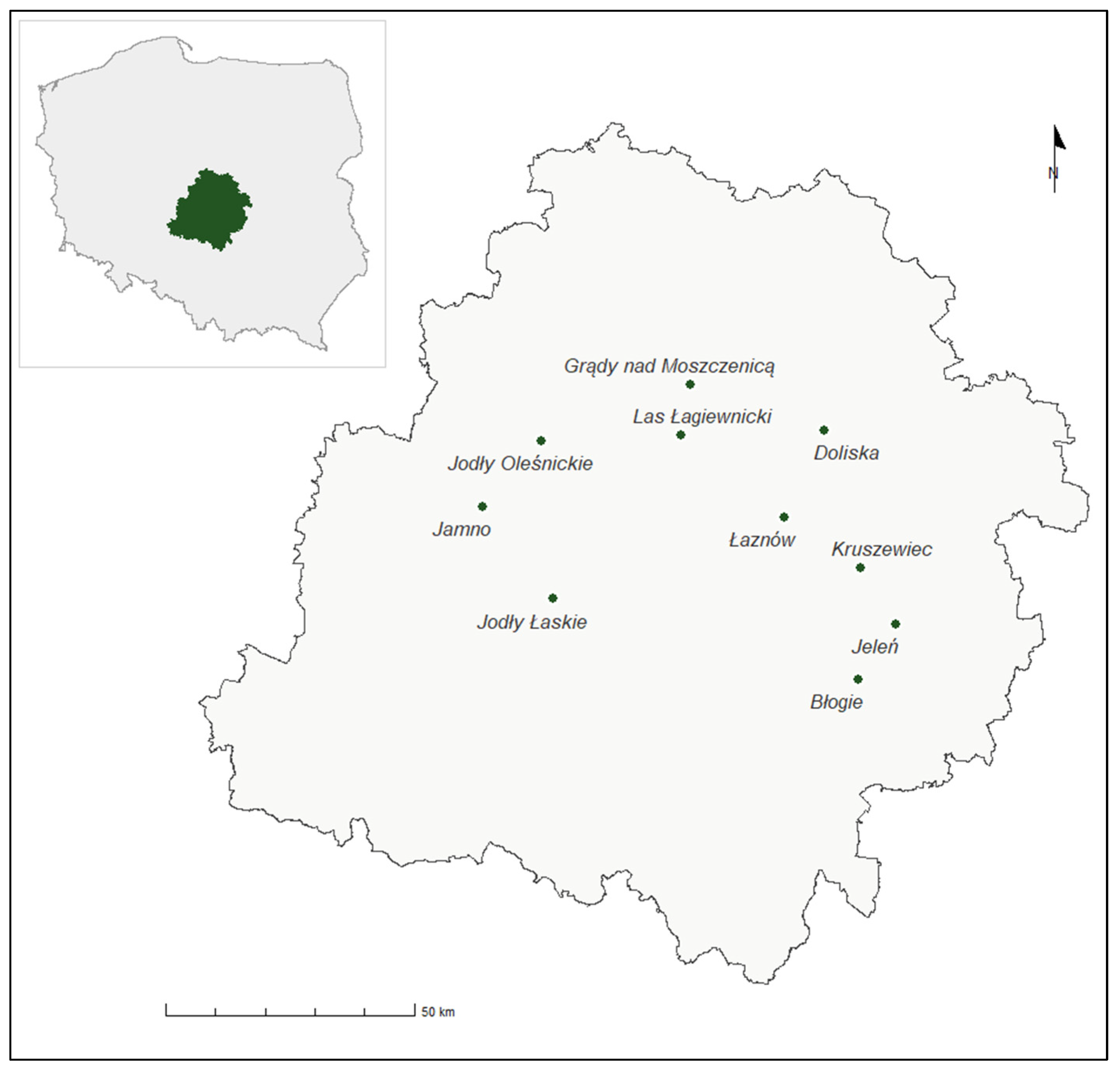
Field studies were carried out in Central Poland, which, according to Jakubowska-Gabara et al. [28], is recognised as part of the Łódzkie Voivodeship. The research was conducted in nature reserves, which are regarded as the best-preserved forest fragments in this region. Based on a review of the literature and numerous preliminary field surveys, ten nature reserves were selected for detailed investigations. These sites not only represent a range of reserves differing in area but also encompass various types of phytocoenoses characteristic of this part of Poland.
Consequently, seven alliances and all forest associations available within the study area were included, namely: Alnion glutinosae (Ribeso nigri-Alnetum); Alno-Ulmion (Fraxino-Alnetum); Carpinion betuli (Tilio-Carpinetum); Dicrano-Pinion (Cladonio-Pinetum, Leucobryo-Pinetum, Vaccinio uliginosi-Pinetum, Querco roboris-Pinetum); Piceion abietis (Abietetum albae); Potentillo albae-Quercion petraeae (Potentillo albae-Quercetum); and Quercion robori-petraeae (Calamagrostio arundinaceae-Quercetum petraeae) as well as the non-forest areas within these reserves (Table 1). The studied phytocoenoses were selected due to their widespread occurrence within the investigated area (Figure 1).

Table 1.
Location, area and associations of the studied nature reserves.
Figure 1.
Location of the nature reserves within the borders of Central Poland (design, and creation of the graphic by Mikołaj Latoszewski).
The units of analysis in this study were identified forest associations (syntaxa), treated as distinct phytosociological units within the boundaries of the investigated nature reserves. The research was based on a comprehensive inventory of phytocoenoses rather than randomly selected sampling plots. The conclusions pertain to the diversity and diagnostic role of epigeic bryophytes within these syntaxonomic units.
The herbarium material documenting these studies was deposited in the Herbarium LOD and KRAM-B. The names of vascular plants followed Mirek et al. [29], and the names of bryophytes were adopted according to Hodgetts et al. [30].
2.2. Qualitative and Quantitative Analysis
The research was conducted across the entire area of all investigated reserves, rather than being limited to selected plots or sub-areas. Consequently, the issues of sampling design or plot selection are not applicable, as the terrestrial bryophyte flora was analysed within individual phytocoenoses throughout the complete territory of the selected nature reserves. The investigations yielded both qualitative and quantitative information regarding the epigeic bryophytes found in the area (Supplementary Material S1). The initial application of this data was to offer a comprehensive overview of the vegetation within the study area, in addition to detailing the terrestrial flora of specific phytocoenoses, encompassing both forested and non-forested regions (Supplementary Material S1).
In order to analyse differences in species pools among individual ecosystems, and due to the lack of detailed data on the exact area of particular phytocoenoses, the primary qualitative dataset (Supplementary Material S1) was transformed into a 0–1 dataset (Supplementary Material S2). These data were subsequently used for statistical analyses, encompassing all bryophyte species, including those occurring sporadically. It was necessary to analyse and compare phytocenoses of different areas to ensure that the results were reliable.
To assess the similarity among the studied phytocoenoses, hierarchical clustering was performed using the Jaccard similarity coefficient and Ward’s linkage method (HCA dendrogram). In order to examine the relationships among the studied phytocoenoses in greater detail, a principal coordinates analysis (PCoA) was also conducted. All calculations were performed using the PAST software package, version 4.09.
3. Results
3.1. General Species Richness of the Studied Phytocoenoses
During the study, a total of 102 bryophyte species were recorded on epigeic habitats within seven alliances (Alnion glutinosae, Alno-Ulmion, Carpinion betuli, Dicrano-Pinion, Piceion abietis, Potentillo albae-Quercion petraeae, Quercion robori-petraeae), as well as along forest roads. Among the recorded taxa, the rarest were, e.g., Conocephalum salebrosum, Eurhynchium striatum, Hylocomiadelphus triquetrus, Riccia rhenana, Scapania curta, and Solenostoma gracillimum, which were noted only sporadically. In contrast, the most frequent species, commonly recorded throughout the entire study area, included, e.g., Atrichum undulatum, Plagiomnium affine, Plagiothecium curvifolium, Pleurozium schreberi, Pohlia nutans, and Polytrichum formosum (Supplementary Material S1).
The recorded bryoflora revealed differences between the studied forest ecosystems and the analysed forest tracks. While both forest and non-forest ecosystems exhibited a comparable diversity of species, with 88 and 70 taxa, respectively, qualitative distinctions between the two were clearly observable.
Among forest phytocoenoses, the highest number of taxa (69 species, representing 68% of the total recorded bryoflora) was recorded within Carpinion betuli (Figure 2). On the epigeic habitats of this alliance, a number of rare species were noted, e.g., Calypogeia azurea, Eurhynchium striatum, and Sciuro-hypnum starkei. In contrast, the most frequent taxa included common forest species, such as Eurhynchium angustirete, Pohlia nutans, and Polytrichum formosum (Figure 3 and Supplementary Material S1). Fewer taxa (55 species, 53%) were recorded within Dicrano-Pinion (Figure 2), where rare species such as Buxbaumia aphylla, Sphagnum squarrosum, and Riccardia palmata were included. The most frequent taxa comprised species typical of pine coniferous forests, including Dicranum scoparium, D. polysetum, and Pleurozium schreberi (Supplementary Material S1).

Figure 2.
Examples of phytocoenoses of the studied alliances. (A)—Tilio-Carpinetum belonging to Carpinion betuli, Grądy nad Moszczenicą nature reserve; (B)—Leucobryo-Pinetum from Dicrano-Pinion, Jeleń nature reserve; (C)—Abietetum albae from the Piceion abietis alliance, Łaznów nature reserve; (D)—Fraxino-Alnetum from Alno-Ulmion, Grądy nad Moszczenicą nature reserve (photos by Grzegorz J. Wolski, August 2025).

Figure 3.
Examples of common and very common species of the studied phytocoenoses. (A)—Eurhynchium angustirete from Carpinion betuli, Grądy nad Moszczenicą nature reserve; (B)—Pleurozium schreberi from Dicrano-Pinion, Jeleń nature reserve; (C)—Thuidium tamariscinum from Piceion abietis, Łaznów nature reserve; (D)—Plagiomnium undulatum from Alno-Ulmion, Grądy nad Moszczenicą nature reserve (photos by Grzegorz J. Wolski, August 2025).
Forty eight species (47% of the total bryoflora) were recorded within Alno-Ulmion. In this alliance, rare taxa such as Pellia epiphylla, Conocephalum conicum, and Chiloscyphus polyanthos were included, whereas the most frequent species were those considered common in deciduous forests, such as Plagiomnium affine, Polytrichum formosum, and Atrichum undulatum (Supplementary Material S1). An identical number of species (36, representing 35% of the total bryoflora) were recorded within Alnion glutinosae and Piceion abietis; however, the two alliances differed markedly in terms of the bryophyte species observation. Within Alnion glutinosae (Figure 4), rare species included Hylocomiadelphus triquetrus, Sphagnum palustre (Figure 5), and Plagiochila asplenioides, whereas the most frequent taxa were, e.g., Brachythecium rutabulum, Sciuro-hypnum oedipodium, and Polytrichum formosum (Supplementary Material S1). In Piceion abietis, rare taxa included Sphagnum fimbriatum, S. russowi, and Bazzania trilobata, while frequent species occurring also in other forest types included Thuidium tamariscinum, Polytrichum formosum and Plagiomnium affine (Supplementary Material S1).

Figure 4.
Examples of phytocoenoses of the studied alliances. (A)—Ribeso nigri-Alnetum belonging to the Alnion glutinosae alliance, Jeleń nature reserve; (B)—forest track in the Kruszewiec nature reserve; (C)—Potentillo albae-Quercetum from the Potentillo albae-Quercion petraeae alliance, Las Łagiewnicki nature reserve; (D)—Calamagrostio arundinaceae-Quercetum petraeae belonging to Quercion robori-petraeae, Las Łagiewnicki nature reserve (photos by Grzegorz J. Wolski, August 2025).

Figure 5.
Examples of common and very common species of the studied phytocoenoses. (A)—Sphagnum palustre from Alnion glutinosae, Jeleń nature reserve; (B)—Brachythecium albicans recorded on forest tracks, Jodły Łaskie nature reserve; (C)—Polytrichum formosum from Potentillo albae-Quercion petraeae, Las Łagiewnicki nature reserve; (D)—Pleurozium schreberi recorded in Quercion robori-petraeae, Las Łagiewnicki nature reserve (photos by Grzegorz J. Wolski, August 2025).
The species-poorest phytocoenoses were Quercion robori-petraeae and Potentillo albae-Quercion petraeae (Figure 4 and Figure 5), where 23 (22%) and 20 taxa (19% of the total bryoflora) were recorded, respectively. These alliances differed in their rare species: for Quercion robori-petraeae, rare taxa included Brachytheciastrum velutinum, Calypogeia azurea, and Chiloscyphus polyanthos, whereas for Potentillo albae-Quercion petraeae these included Brachythecium salebrosum, Leucobryum glaucum, and Lophocolea heterophylla. However, the same relatively widespread moss species were considered frequent in both phytocoenoses, e.g., Dicranella heteromalla, Polytrichum formosum, and Atrichum undulatum (Figure 2, Figure 3, Figure 4 and Figure 5 and Supplementary Material S1 and S2).
When comparing forests to forest trails, both showed similar species richness; however, the bryoflora composition along the trails was distinctly different from that in the aforementioned forest ecosystems. In the epigeic habitats of forest tracks, several uncommon species that are infrequent throughout the entire study area were documented, including Apopellia endiviifolia, Sciuro-hypnum starkei, and Scapania curta. Conversely, the taxa that appeared most frequently comprised common species like Oxyrrhynchium hians, Plagiomnium affine, and Polytrichum formosum (Supplementary Material S1).
Considering the data presented, there is sufficient justification to accept the hypothesis that “the species pools colonising epigeic habitats of individual phytocoenoses differ,” as they exhibit both qualitative and quantitative variations from one another.
3.2. Similarity of Epigeic Substrates Among Individual Phytocoenoses
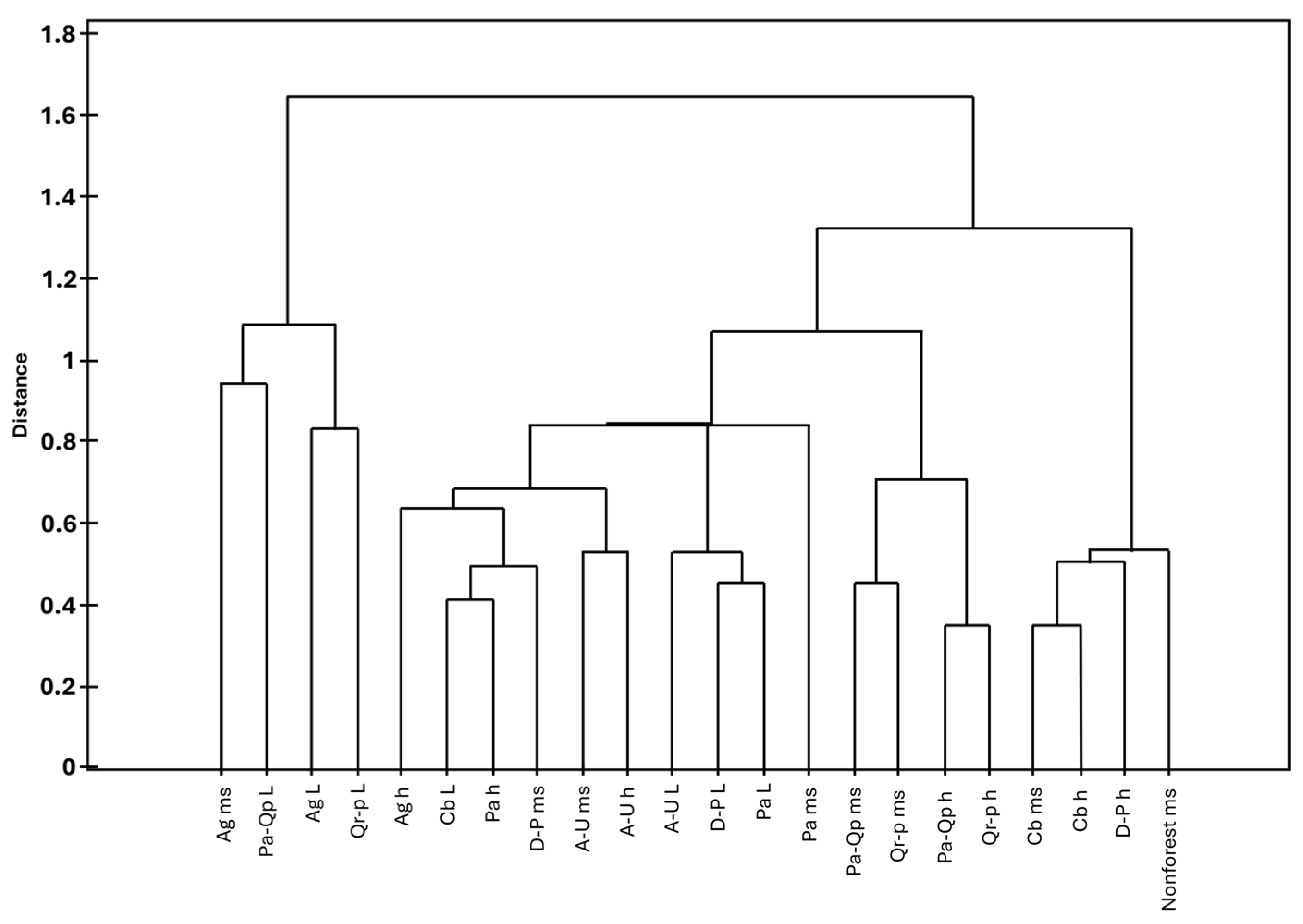
The analysis of species pools colonising individual substrates of the studied phytocoenoses showed that they are arranged into six distinct groups. However, these groups consist of different substrates from different phytocoenoses, and the grouping of particular substrates is influenced by both qualitative and quantitative characteristics of their bryoflora (Figure 6 and Figure 7, and Supplementary Material S1).
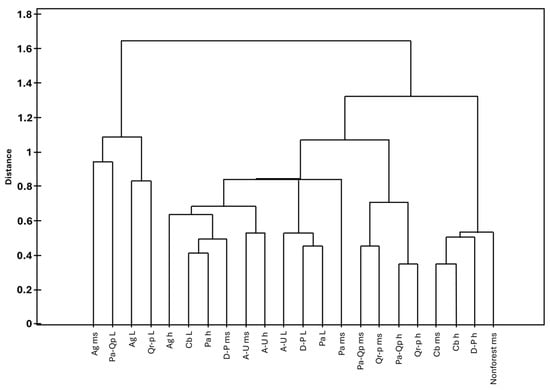
Figure 6.
Similarity analysis of substrates in the studied phytocoenoses. Explanation of abbreviations: substrates—ms, mineral soil; h, humus; L, litter; phytocoenoses—Ag, Alnion glutinosae; A-U, Alno-Ulmion; Cb, Carpinion betuli; D-P, Dicrano-Pinion; Pa, Piceion abietis; Pa-Qp, Potentillo albae-Quercion petraeae; Qr-p, Quercion robori-petraeae, Jaccard index, Warda linkage.
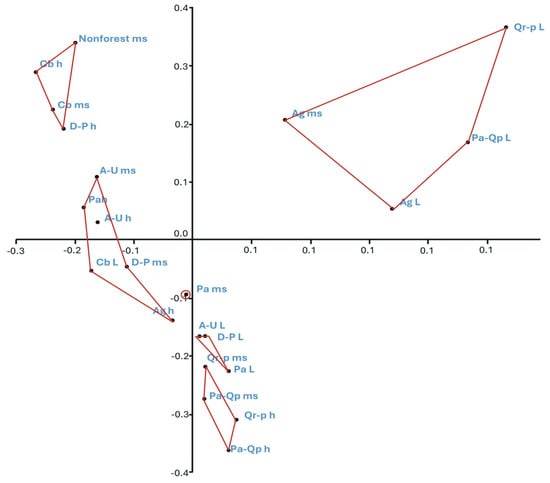
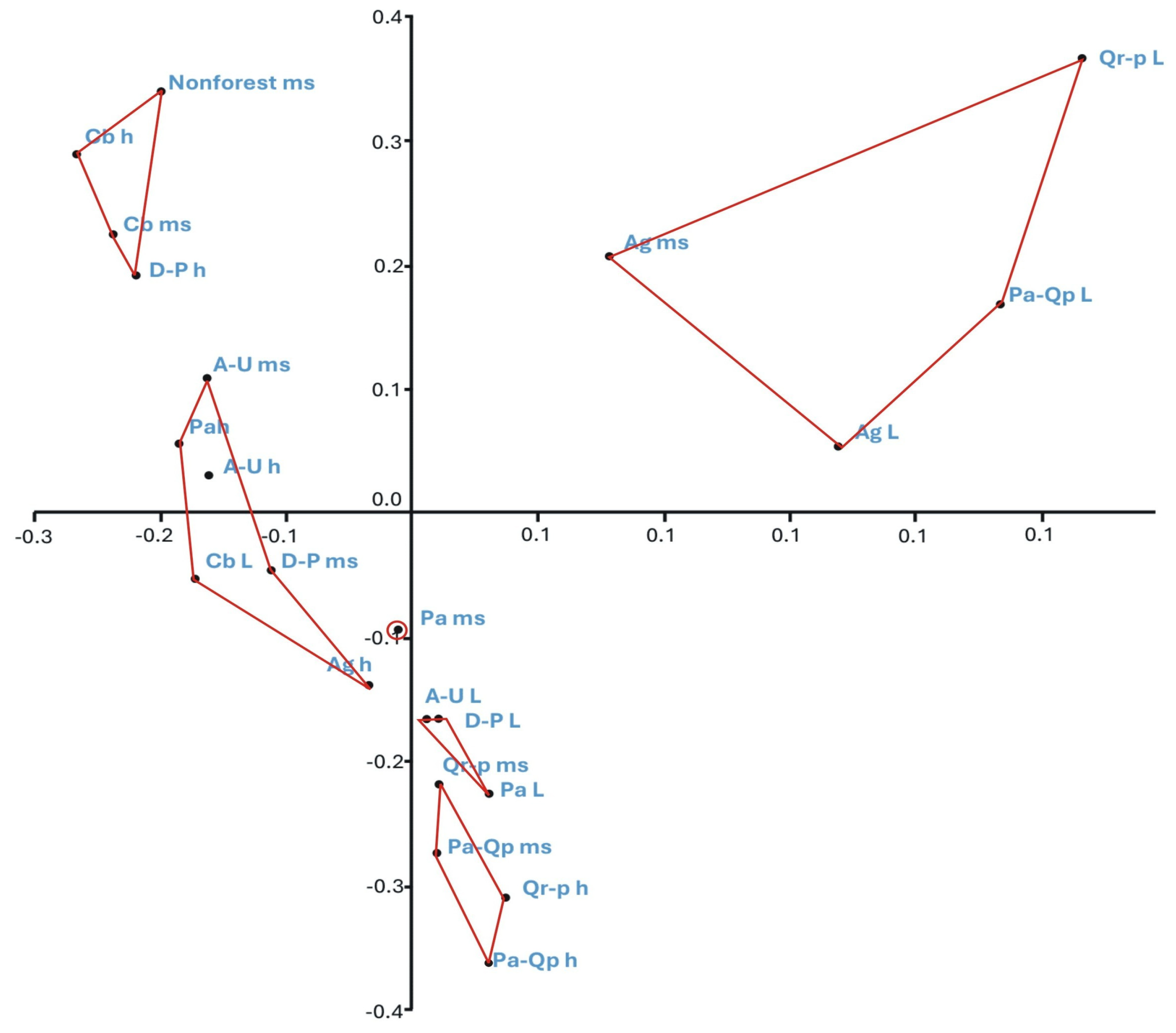
Figure 7.
PCoA ordination of the bryophyte flora of substrates in the studied phytocoenoses. Explanation of abbreviations: substrates—ms, mineral soil; h, humus; L, litter; phytocoenoses—Ag, Alnion glutinosae; A-U, Alno-Ulmion; Cb, Carpinion betuli; D-P, Dicrano-Pinion; Pa, Piceion abietis; Pa-Qp, Potentillo albae-Quercion petraeae; Qr-p, Quercion robori-petraeae. Axis I and II explains 34% of the variation in site similarity, Jaccard index, UPGMA linkage.
The first group, the most distinct from the others, is formed by soil and litter in Alnion glutinosae as well as litter in Potentillo albae-Quercion petraeae and Quercion robori-petraeae; it is characterised by the lowest species diversity. On average, only six taxa were recorded, all representing species with a broad ecological amplitude, such as Brachythecium rutabulum, Sciuro-hypnum oedipodium, and Dicranum scoparium.
The second group, also clearly distinct from the remaining ones, is characterised by the highest biodiversity, with an average of 59 taxa recorded. This group is formed by mineral soil in Carpinion betuli and in non-forest ecosystems, as well as by humus substrates in Carpinion betuli and Dicrano-Pinion. It differs from the other groups by the presence of numerous taxa, e.g., Sphagnum squarrosum, Rhytidiadelphus squarrosus, and Pohlia wahlenbergii, which were recorded exclusively on these substrates and within these phytocoenoses. Other species common in this group were recorded elsewhere only sporadically, such as Cirriphyllum piliferum, Calypogeia muelleriana, and Sciuro-hypnum starkei (Figure 6 and Figure 7, and Supplementary Material S1).
The next group is formed by humus and mineral soil in Potentillo albae-Quercion petraeae and Quercion robori-petraeae and is characterised not only by a relatively low number of species—on average 15 taxa—but also by the presence of numerous taxa with a broad ecological amplitude, such as Polytrichum formosum, Brachythecium rutabulum and Dicranella heteromalla.
The final group is further divided into three subgroups, one of which is formed by a single element—mineral soil in Piceion abietis. This subgroup is characterised by a relatively low number of species (14 taxa), the presence of restricted taxa (e.g., Sphagnum girgensohnii), as well as a set of species commonly recorded there but only sporadically elsewhere (e.g., Sphagnum russowi), and taxa with a broad ecological amplitude, such as Pohlia nutans, Dicranella heteromalla, and Plagiomnium affine. The last two groups are the most similar to each other. The first is formed by litter in Alno-Ulmion, Dicrano-Pinion, and Piceion abietis, whereas the second comprises mineral soil and humus in Alno-Ulmion, humus in Alnion glutinosae and Piceion abietis, mineral soil in Dicrano-Pinion, and litter in Carpinion betuli. They differ in the mean number of recorded taxa, with the first being poorer (17 species) than the second (30 species), as well as in the presence or absence of certain taxa, including Rhizomnium punctatum, Pellia epiphylla, and Brachythecium rivulare (Figure 6 and Figure 7, and Supplementary Material S1).
Thus, it can be concluded that the substrates that are most similar in terms of bryophyte composition are: humus and mineral soil in Carpinion betuli; humus and mineral soil in Quercion robori-petraeae and Potentillo albae-Quercion petraeae; litter in Piceion abietis and Dicrano-Pinion; mineral soil and humus in Alno-Ulmion; and litter in Potentillo albae-Quercion petraeae, Alnion glutinosae, and Quercion robori-petraeae. The conducted analysis shows that, despite certain ambiguities, most of the analysed substrates form relatively homogeneous groups.
However, given the results and the fact that epigeic substrates in coniferous and deciduous forests do not form homogeneous groups, the hypothesis that “the epigeic flora of deciduous forests differs from that of coniferous forests” should be rejected.
3.3. Ecological Amplitude of Bryophytes and Their Discriminatory Potential
The occurrence of individual taxa is directly related to their ecological amplitude, understood in both phytocoenotic and substrate-related terms. By considering the presence of bryophytes within the studied phytocoenoses and on particular types of substrates, it is possible to distinguish restricted and shared taxa, characteristic both of specific substrates and of the phytocoenoses in which they were recorded.
In the study area, mineral soil proved to be the most species-rich substrate, with a total of 90 taxa recorded (88% of the total bryoflora noted). The overall richness of this substrate was undoubtedly influenced by the inclusion of forest tracks in the study, on which as many as 70 taxa were recorded (Supplementary Material S1 and S2). The most frequent species on this substrate were those recorded in all studied phytocoenoses; only two taxa met this criterion, Dicranella heteromalla and Polytrichum formosum (Supplementary Material S1). In contrast, the rarest species were those recorded restricted within a single phytocoenosis, e.g., Campylopus introflexus (within Dicrano-Pinion), Riccia rhenana (within Carpinion betuli), and Barbula unguiculata (recorded on forest tracks).
A total of 75 taxa (73% of the total recorded bryoflora) were noted on humus. The most frequent species were those recorded in all studied phytocoenoses, e.g., Brachythecium rutabulum, Lophocolea bidentata, and Plagiothecium curvifolium (Supplementary Material S1). In contrast, the rarest taxa were those recorded in only a single phytocoenosis, e.g., Aulacomnium palustre (within Dicrano-Pinion) and Cephaloziella divaricata (within Carpinion betuli) (Supplementary Material S1).
Among the examined substrates, litter exhibited the least diversity, with merely 33 taxa documented, representing 32% of the overall bryoflora. Within Potentillo albae-Quercion petraeae and Quercion robori-petraeae, only a single taxon was recorded on this substrate, and the remaining phytocoenoses were not markedly richer in this respect. During the study, no species common to all investigated alliances were recorded on litter; nevertheless, the most frequent species, noted in six of them, was Sciuro-hypnum oedipodium. In contrast, the most infrequent or uncommon taxa were those recorded in only one phytocoenosis; in the case of litter, such taxa were recorded exclusively within Carpinion betuli and included, e.g., Kindbergia praelonga, Brachytheciastrum velutinum, and Dicranum montanum (Supplementary Material S1).
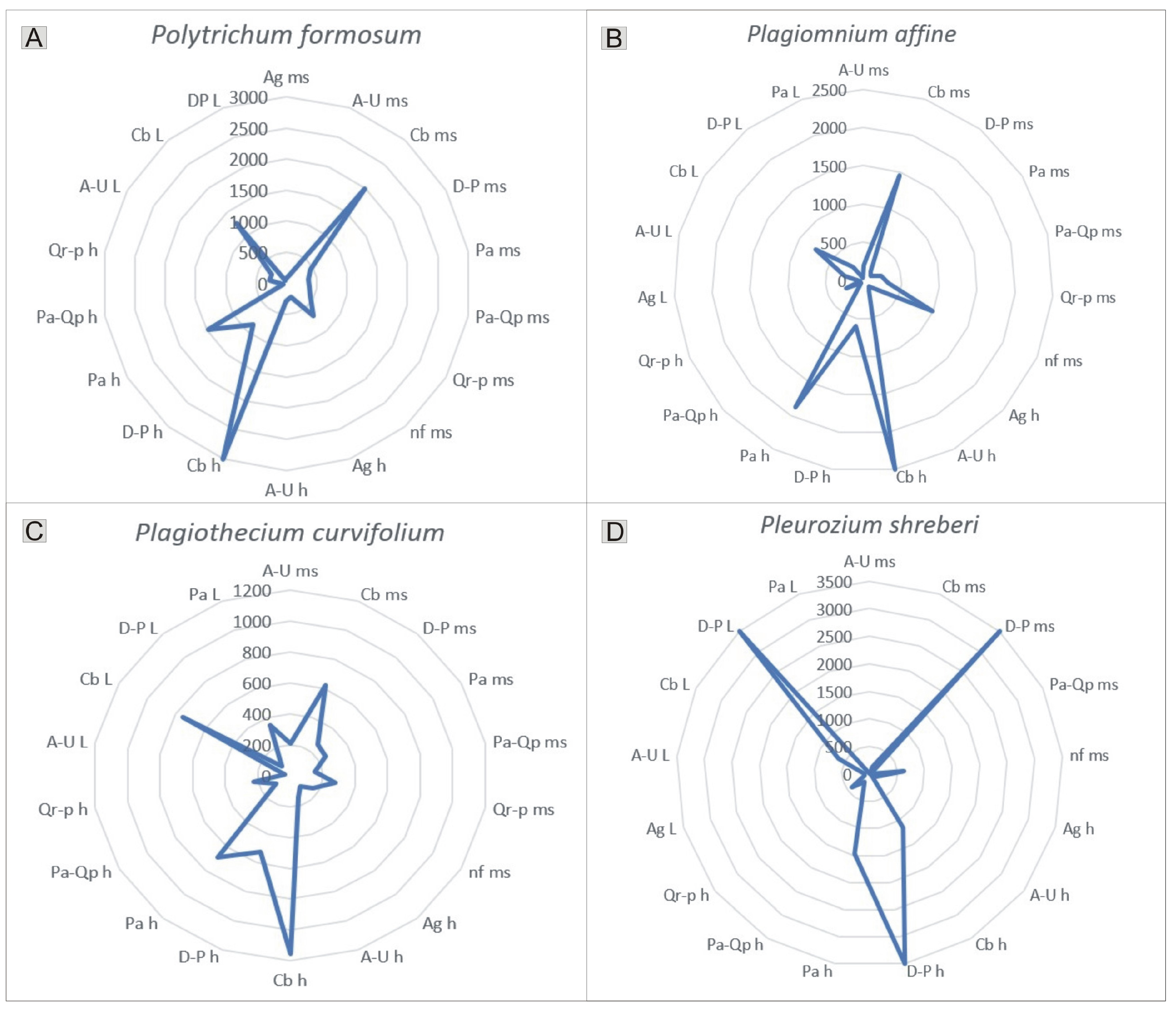
The occurrence of the species studied on particular substrates clearly reflects their distinct substrate preferences. In the study area, Polytrichum formosum, Plagiothecium curvifolium, and Plagiomnium affine were recorded on all substrates within the Carpinion betuli alliance; however, they were also noted, for example, on humus within the Piceion abietis alliance. In contrast, Pleurozium schreberi occurred mainly on all investigated substrates of Dicrano-Pinion (Figure 8).
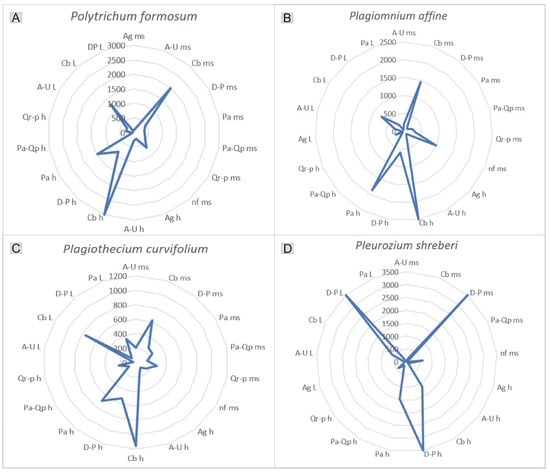
Figure 8.
Substrate preferences of selected, widely distributed species. (A)—Polytrichum formosum, (B)—Plagiomnium affine, (C)—Plagiothecium curvifolium, (D)—Pleurozium schreberi. Explanation of abbreviations: substrates—ms, mineral soil; h, humus; L, litter; phytocoenoses—Ag, Alnion glutinosae; A-U, Alno-Ulmion; Cb, Carpinion betuli; D-P, Dicrano-Pinion; Pa, Piceion abietis; Pa-Qp, Potentillo albae-Quercion petraeae; Qr-p, Quercion robori-petraeae, nf, non-forest.
With respect to individual phytocoenoses, it is also possible to distinguish both shared and restricted species. Shared species were defined as taxa with a wide phytocoenotic amplitude, understood as those recorded in many of the studied phytocoenoses. The most widespread among them were taxa noted in all investigated phytocoenoses, including, for example, Dicranella heteromalla, Lophocolea heterophylla, and Pleurozium schreberi (Supplementary Material S2).
Despite the widespread occurrence of shared species, their frequency of occurrence allows the determination of their phytocoenotic optimum, defined as the phytocoenosis in which they were recorded most frequently. For example, the phytocoenotic optimum for Polytrichum formosum, Plagiothecium curvifolium, and Plagiomnium affine was the Carpinion betuli alliance, whereas for Pleurozium schreberi it was Dicrano-Pinion (Figure 8, Supplementary Material S1 and S2).
A substantial amount of information on the role of bryophytes is also provided by the group of restricted species, i.e., taxa with a very narrow phytocoenotic amplitude, recorded within only a single phytocoenosis. The highest number of restricted species (13; 12% of the total bryoflora) was recorded on forest tracks, including, for example, Bryum argenteum, Campylium stellatum, Campylophyllopsis calcarea, Ptychostomum caespiticium, Pohlia cruda, and Streblotrichum convolutum. A lower number (seven; 7%) was noted within Dicrano-Pinion, including, for example, Aulacomnium palustre, Campylopus introflexus, Dicranum spurium, Polytrichum piliferum, and Riccardia palmata. Within Carpinion betuli, six restricted species were recorded (6% of the total bryoflora), such as Eurhynchium striatum, Oxyrrhynchium schleicheri, and Ptychostomum capillare. For Alnion glutinosae, two such taxa were noted—Hylocomiadelphus triquetrus and Straminergon stramineum—whereas only a single restricted species, Sphagnum fimbriatum, was recorded for Piceion abietis (Supplementary Material S2). No restricted species were recorded within Alno-Ulmion, Potentillo albae–Quercion petraeae, or Quercion robori-petraeae in the studied area (Supplementary Material S2).
Although a much higher number of species were recorded within these ecosystems, it is precisely the group of restricted taxa that may prove useful as differentiating taxa. Conversely, the absence of restricted species in some phytocoenoses is most likely a consequence of their small area covered by the study. Consequently, based on the results obtained, the hypothesis asserting that “for the studied alliances, it is feasible to propose restricted species that differentiate a specific syntaxon from others” is affirmed.
4. Discussion
Mosses and liverworts, unlike other plant groups, demonstrate the capacity to colonise a diverse range of substrate types. Consequently, as highlighted by various researchers, the existence of different habitat types is evident in the species composition of the bryoflora, which in turn influences both the quantitative and qualitative relationships within the local moss and liverwort flora [11,12,31,32]. The degree of preservation of individual phytocoenoses is also of considerable importance [9,11,33,34,35], as their condition and level of development directly affect the overall biodiversity of the studied plant group. On the other hand, many species—particularly restricted ones—occur on a single type of substrate or habitat [6,7,11,36]. The stenotopic character of these organisms results in their persistence within forest ecosystems being dependent on the presence and cyclical availability of specific substrate types [37].
Mosses and liverworts are well-established bioindicators, not only of heavy metals but also of substrates and phytocoenoses [6,7,18,38,39,40]. However, from a phytosociological perspective, their role has been underestimated and is very often overlooked (e.g., refs. [15,16,17]). Conversely, this very fact has become a premise for renewed investigations into their indicative potential [6,7,18].
A total of 102 species were recorded during the study. This is a very high number, considering not only the relatively limited study area but also the fact that only a single habitat type was analysed. This number represents nearly 30% of all bryophyte species recorded to date in the entire region of Central Poland, where all types of phytocoenoses were taken into account; the literature also emphasises that bryological studies in this area have been conducted since the nineteenth century (e.g., refs. [1,8]).
Thus, it can be clearly stated that epigeic habitats of nature reserves in Central Poland constitute important refugia of bryophyte biodiversity in this part of the country; in this respect, both forest and non-forest habitats within the studied reserves are of high conservation value.
The relationship between the quantitative and qualitative composition of the moss layer and the type of phytocoenosis in which it develops has been analysed repeatedly in the literature [8,11,32,41]. However, existing studies (e.g., refs. [6,7,11,15,18]) most often refer to plant associations rather than to higher syntaxonomic units. Taxonomic categories such as classes and orders are infrequently examined, and the quantity of accessible bryological data for these classifications is exceedingly restricted. Thus, the analyses herein offer a significant collection of novel data regarding the biodiversity of the examined plant group and its indicative potential, at a markedly higher level of syntactic resolution.
Among forest phytocoenoses, the highest number of taxa was recorded in the Carpinion betuli alliance, where as many as 69 species were recorded in epigeic habitats. This finding is consistent with the literature, which indicates that hornbeam forests are phytocoenoses particularly rich in ground-dwelling bryophyte species [7]. A high number of taxa was also recorded in Dicrano-Pinion, where 55 bryophyte species were noted, a pattern reflecting the high diversity of phytocoenoses within this alliance. These comprise both very dry lichen pine forests (Cladonio-Pinetum) and wet boggy pine forests (Vaccinio uliginosi-Pinetum), as well as numerous intermediate phytocoenoses, such as Leucobryo-Pinetum [15]. This diversity of ecological conditions such as soil fertility, moisture, and light availability—clearly contributes to the species richness of this alliance. A relatively high number of species (36) was also recorded within Piceion abietis, which is somewhat surprising, as fir forests (Abietetum albae) belonging to this alliance are not generally characterised by particularly high biodiversity [15].
On the other hand, relatively few species were recorded within Alnion glutinosae (36 species), Quercion robori-petraeae (23 species), and Potentillo albae–Quercion petraeae (20 taxa). This is particularly surprising in the case of Alnion glutinosae, as phytocoenoses of this alliance—i.e., alder forests—are typically reported in the literature as being characterised by high bryophyte diversity (e.g., refs. [12,41,42]). The low number of species recorded within these alliances is undoubtedly influenced by the small area of the studied phytocoenoses and their transformed character. However, direct comparison of these results with the literature data is difficult, as alliances are rarely the focus of bryological studies.
The conducted studies showed that among all analysed substrates, mineral soil was the most species-rich, supporting 90 taxa. This high number of species was undoubtedly influenced by the inclusion of forest tracks in the study, which are known to increase the overall biodiversity of the investigated area [43,44]. In contrast, the lowest number of taxa was recorded on litter, which is generally much less frequently colonised by bryophytes; nevertheless, in forest ecosystems it constitutes an important substrate for these plants [4,32].
The species composition and frequency of individual taxa translate into the degree of similarity among the analysed alliances. Thus, similarity between them is influenced both by restricted species and by the presence of taxa with a broader ecological amplitude (e.g., Dicranella heteromalla, Polytrichum formosum, Plagiomnium affine). These taxa are among the most frequent general forest moss species [45]. On the other hand, the presence of restricted species determines the distinctiveness of the studied alliances and their differentiation among individual phytocoenoses [6,7,18,35].
In classical phytosociological approaches, bryophytes as bioindicators are marginalised or entirely omitted. In contrast, the recent literature presents a different picture of the surrounding natural environment and highlights the significant role of bryophytes as indicators of plant communities [6,7,18]. One of the most striking examples of the marginalisation of their bioindicative role is their omission in studies of coniferous forests, particularly pine forests. Pine forests are not only habitats of the best-known and most frequently recognised species, such as Pleurozium schreberi, Dicranum scoparium, D. polysetum, or Hylocomium splendens. They also represent highly diverse communities characterised by unique ecological conditions which, contrary to common opinion, support numerous restricted species [4,6,7,18,33,34].
Species regarded as restricted to Carpinion betuli included, for example, Eurhynchium striatum, Oxyrrhynchium schleicheri and Ptychostomum capillare, which in the study area distinguished Carpinion betuli from the other analysed alliances. Among them, Eurhynchium striatum is described in the literature as a species associated with fertile, moderately moist and shaded deciduous forests, especially hornbeam forests, with its occurrence largely restricted to stable and weakly transformed habitats [46,47,48].
Species regarded as restricted to Dicrano-Pinion included, Aulacomnium palustre, Campylopus introflexus, Dicranum spurium, Polytrichum piliferum and Riccardia palmata. Some of these taxa (Aulacomnium palustre, Dicranum spurium) are recognised bioindicators of plant communities belonging to this alliance [15]; therefore, their indicator value for Dicrano-Pinion in the present study is not surprising. Others, such as Campylopus introflexus, have not previously been analysed in this context; however, they are known to be recorded mainly in pine forests [41,49], and thus their indicative character with respect to pine forests is not unexpected. For Alnion glutinosae, the species regarded as restricted were Hylocomiadelphus triquetrus and Straminergon stramineum, whereas for Piceion abietis only Sphagnum fimbriatum was indicated. However, these taxa are characterised by a much broader ecological tolerance [45]. Therefore, the present study and the conclusions drawn from it should be interpreted not in the context of the overall body of knowledge on the ecological preferences of bryophytes, but rather in relation to the specific region in which the research was conducted.
On the other hand, the highest number of restricted species was recorded on forest tracks, including, e.g., Bryum argenteum, Campylium stellatum, Campylophyllopsis calcarea, Ptychostomum caespiticium and Streblotrichum convolutum. These are typical taxa of open habitats [45], and their occurrence on forest tracks in the studied area, as well as their distinguishing character, is therefore not surprising, as they are usually not recorded within forest interiors [43,44,50]. The present results, together with the cited literature, confirm that forest tracks are characterised by high natural value and may serve as refugia for locally rare and valuable bryophyte species.
In contrast, the lack of differential species, i.e., restricted species, for Alno-Ulmion, Quercion robori-petraeae and Potentillo albae-Quercion petraeae points to several potential causes. These include either the very small area of the studied phytocoenoses or their strong transformation, leading to the dominance of common species with broad ecological amplitudes. Both hypotheses are highly plausible in the case of these phytocoenoses. Similar factors may also underline the observed similarity of species pools among individual substrates in coniferous and deciduous forests. These data challenge the generally accepted paradigm and may indicate local disturbances of vegetation in the study area and the transformed character of its plant cover [51].
5. Conclusions
The epigeic bryophyte flora of the studied phytocoenoses exhibits pronounced qualitative and quantitative differentiation, with species composition being shaped primarily by the combined effects of phytocoenosis type and substrate characteristics, rather than by forest type alone.
Mineral soil was the most species-rich epigeic substrate, whereas litter supported the lowest bryophyte diversity, highlighting the importance of substrate stability and microhabitat availability for bryophyte establishment and persistence.
Although forests and forest roads hosted comparable numbers of bryophyte species, their species composition differed markedly, indicating that non-forest habitats serve as important refugia for rare and habitat-specific taxa.
Similarity analyses revealed that substrates with comparable properties may host analogous bryophyte assemblages across different phytocoenoses, thereby challenging a strict separation of epigeic bryophyte floras between deciduous and coniferous forests.
Widespread species with broad ecological amplitudes contribute to the structural consistency of bryophyte assemblages; however, restricted taxa show the highest distinguishing potential.
Overall, the results confirm that bryophytes are sensitive and effective indicators of fine-scale habitat heterogeneity in forest ecosystems and underscore the need to incorporate substrate diversity into ecological research and conservation strategies.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/f17030321/s1.
Author Contributions
Conceptualization, G.J.W.; investigation, G.J.W.; data curation, G.J.W.; writing—original draft preparation, G.J.W., A.S.K., Z.S. and M.S.; writing—review and editing, G.J.W., A.S.K., Z.S. and M.S.; visualisation, G.J.W.; supervision, G.J.W.; project administration, G.J.W. All authors have read and agreed to the published version of the manuscript.
Funding
Co-financed by the Minister of Science under the “Regional Excellence Initiative” Program for 2024-2027 (RID/SP/0045/2024/01).
Data Availability Statement
All data generated or analysed during this study are included in this published article and its Supplementary Materials.
Acknowledgments
The authors wish to express their sincere gratitude to Ewa Fudali, the supervisor and mentor of the first author’s doctoral dissertation, within the framework of which the research was conducted and part of the results have been presented in this publication.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Staniaszek-Kik, M.; Wolski, G.J. Mszaki—Zróżnicowanie, zmiany, zagrożenia. In Szata Roślinna Polski Środkowej; Kurowski, J.K., Ed.; Towarzystwo Ochrony Krajobrazu Wydawnictwo EKO-GRAF: Łódź, Poland, 2009; pp. 48–56. [Google Scholar]
- Wolski, G.J.; Cienkowska, A.; Plášek, V. Habitat preferences and ecological relationships of bark-inhabiting bryophytes in Central Polish forests. Forests 2026, 17, 66. [Google Scholar] [CrossRef]
- Rola, K.; Plášek, V.; Rożek, K.; Zubek, S. Effect of tree species identity and related habitat parameters on understorey bryophytes—Interrelationships between bryophyte, soil and tree factors in a 50-year-old experimental forest. Plant Soil 2021, 466, 613–630. [Google Scholar] [CrossRef]
- Stefańska-Krzaczek, E.; Swacha, G.; Żarnowiec, J.; Raduła, M.W.; Kącki, Z.; Staniaszek-Kik, M. Central European forest floor bryophytes: Richness, species composition, coexistence and diagnostic significance across environmental gradients of forest habitats. Ecol. Indic. 2022, 139, 108954. [Google Scholar] [CrossRef]
- Kutnar, L.; Kermavnar, J.; Sabovljević, M.S. Bryophyte diversity, composition and functional traits in relation to bedrock and tree species composition in close-to-nature managed forests. Eur. J. For. Res. 2023, 142, 865–882. [Google Scholar] [CrossRef]
- Wolski, G.J.; Sobisz, Z.; Mitka, J.; Kruk, A.; Jukoniené, I.; Popiela, A. Vascular plants and mosses as bioindicators of variability of the coastal pine forest (Empetro nigri–Pinetum). Sci. Rep. 2024, 14, 76. [Google Scholar] [CrossRef]
- Wolski, G.J.; Celka, Z.; Marciniuk, J.; Marciniuk, P.; Mitka, J.; Nowak, S.; Afrontowicz-Cieślak, R.; Kruk, A.; Łysko, A.; Popiela, A. New indicator species for associations within mesotrophic oak-hornbeam forests in Poland. Ecol. Indic. 2025, 173, 113339. [Google Scholar] [CrossRef]
- Urbanek-Rutowicz, H. Udział i Rola Diagnostyczna Mszaków oraz Stosunki Florystyczno-Fitosocjologiczne w Przewodnich Zespołach Roślinnych Regionu Łódzkiego i Jego Pobrzeży; Wyd. UŁ: Łódź, Poland, 1969; pp. 1–253. [Google Scholar]
- Mišíková, K.; Mišík, M. Bryophytes in managed lowland forests of Slovakia (Central Europe): Looking into species diversity across different forest types. Biodivers. Conserv. 2024, 33, 4195–4213. [Google Scholar] [CrossRef]
- Söderström, L. Substrate preference in some forest bryophytes: A quantitative study. Lindbergia 1993, 18, 98–103. [Google Scholar]
- Klama, H. Distribution Patterns of Liverworts (Marchantiopsida) in Natural Forest Communities (Białowieża Primeval Forest, NE Poland); University of Bielsko-Biała: Bielsko-Biała, Poland, 2002; pp. 1–278. [Google Scholar]
- Stebel, A. The Mosses of the Beskidy Zachodnie as a Paradigm of Biological and Environmental Changes in the Flora of the Polish Western Carpatians. Ph.D. Thesis, Medical University of Silesia in Katowice, Katowice–Poznań, Poland, 2006; pp. 1–348. [Google Scholar]
- Braun-Blanquet, J. Plant Sociology. The Study of Plant Communities; XVII; McGraw-Hill Book Co., Inc.: New York, NY, USA, 1932; pp. 1–439. [Google Scholar]
- Braun-Blanquet, J. Pfanzensoziologie, Grundzüge der Vegetationskunde, 3rd ed.; Springer: Wien, Austria; New York, NY, USA, 1964; pp. 1–865. [Google Scholar] [CrossRef]
- Matuszkiewicz, W. Przewodnik do oznaczania zbiorowisk roślinnych Polski. In Vademecum Geobotanicum; Faliński, J.B., Ed.; Wydawnictwo Naukowe PWN: Warszawa, Poland, 2019; Volume 3, pp. 1–540. [Google Scholar]
- Chytrý, M.; Tichý, L. National vegetation classification of the Czech Republic: A summary of the approach. Phytocoenologia 2018, 48, 121–131. [Google Scholar] [CrossRef]
- Chytrý, M.; Horsák, M.; Danihelka, J.; Ermakov, N.; German, D.A.; Hájek, M.; Hájková, P.; Kočí, M.; Kubešová, S.; Lustyk, P.; et al. A modern analogue of the Pleistocene steppe-tundra ecosystem in southern Siberia. Boreas 2019, 48, 36–56. [Google Scholar] [CrossRef]
- Wolski, G.J.; Kruk, A. Determination of plant communities based on bryophytes: The combined use of Kohonen artificial neural network and indicator species analysis. Ecol. Indic. 2020, 113, 106160. [Google Scholar] [CrossRef]
- Rodwell, J.S. Woodlands and Scrub. In British Plant Communities; Cambridge University Press: Cambridge, UK, 1991; Volume 1, pp. 1–400. [Google Scholar]
- Rodwell, J.S. Mires and Heaths. In British Plant Communities; Cambridge University Press: Cambridge, UK, 1991; Volume 2, pp. 1–640. [Google Scholar]
- Rodwell, J.S. Grasslands and Montane Communities. In British Plant Communities; Cambridge University Press: Cambridge, UK, 1992; Volume 3, pp. 1–540. [Google Scholar]
- Rodwell, J.S. Aquatic Communities, Swamps and Tall-Herb Fens. In British Plant Communities; Cambridge University Press: Cambridge, UK, 1995; Volume 4, pp. 1–296. [Google Scholar]
- Rodwell, J.S. Maritime communities and vegetation of open habitats. In British Plant Communities; Cambridge University Press: Cambridge, UK, 2000; Volume 5, pp. 1–528. [Google Scholar]
- Košir, P.; Casavecchia, S.; Čarni, A.; Škvorc, Ž.; Zivkovic, L.; Biondi, E. Ecological and phytogeographical differentiation of oak-hornbeam forests in southeastern Europe. Plant Biosyst. 2013, 147, 84–98. [Google Scholar] [CrossRef]
- Novák, P.; Willner, W.; Biurrun, I.; Gholizadeh, H.; Heinken, T.; Jandt, U.; Kollár, J.; Kozhevnikova, M.; Naqinezhad, A.; Onyshchenko, V.; et al. Classification of European oak–hornbeam forests and related vegetation types. App. Veg. Sci. 2023, 26, e12712. [Google Scholar] [CrossRef]
- Novák, P.; Willner, W.; Zukal, D.; Kollár, J.; Roleček, J.; Świerkosz, K.; Ewald, J.; Wohlgemuth, T.; Csiky, J.; Onyshchenko, V.; et al. Oak-hornbeam forests of central Europe: A formalized classification and syntaxonomic revision. Preslia 2020, 92, 1–34. [Google Scholar] [CrossRef]
- Novák, P.; Zukal, D.; Večeřa, M.; Píšťková, K. Vegetation of oak-hornbeam, scree and ravine forests at lower altitudes in Transcarpathia, Western Ukraine. Tuexenia 2017, 37, 47–63. [Google Scholar] [CrossRef]
- Jakubowska-Gabara, J.; Kucharski, L.; Zielińska, K.; Kołodziejek, J.; Witosławski, P.; Popkiewicz, P. Atlas Rozmieszczenia Roślin Naczyniowych w Polsce Środkowej. Gatunki Chronione, Rzadkie, Ginące i Zagrożone; Wyd. UŁ: Łódź, Poland, 2011; pp. 1–283. [Google Scholar]
- Mirek, Z.; Piękoś-Mirkowa, H.; Zając, A.; Zając, M. (Eds.) Vascular plants of Poland. An annotated checklist. In Biodiversity of Poland; W. Szafer Institute of Botany, Polish Academy of Sciences: Kraków, Poland, 2020; pp. 1–526. [Google Scholar]
- Hodgetts, N.G.; Söderström, L.; Blockeel, L.; Caspari, S.; Ignatov, M.S.; Konstantinova, N.A.; Lockhart, N.; Papp, B.; Schröck, C.; Sim-Sim, M.; et al. An annotated checklist of bryophytes of Europe, Macaronesia and Cyprus. J. Bryol. 2020, 42, 1–116. [Google Scholar] [CrossRef]
- Fojcik, B. Mchy Wyżyny Krakowsko-Częstochowskiej w Obliczu Antropogenicznych Przemian Szaty Roślinnej; Wyd. Uniwersytetu Śląskiego: Katowice, Poland, 2011; pp. 1–231. [Google Scholar]
- Evans, S.A.; Halpern, C.B.; McKenzie, D. The Contributions of Forest Structure and Substrate to Bryophyte Diversity and Abundance in Mature Coniferous forests of the Pacific Northwest. Bryologist 2012, 115, 278–294. [Google Scholar] [CrossRef]
- Stefańska-Krzaczek, E.; Staniaszek-Kik, M.; Fałtynowicz, W. Positive aspects of clear-cut logging? Ground bryophyte diversity along the age gradient of managed Pinus sylvestris stands. Cryptogam. Bryol. 2016, 37, 181–197. [Google Scholar] [CrossRef]
- Stefańska-Krzaczek, E.; Staniaszek-Kik, M.; Szczepańska, K.; Szymura, T.H. Species diversity patterns in managed Scots pine stands in ancient forest sites. PLoS ONE 2019, 14, e0219620. [Google Scholar] [CrossRef]
- Wolski, G.J. Habitat Conditions for the Occurrence of Bryophytes in Nature Reserves Protecting Silver Fir in Central Poland. Ph.D. Thesis, University of Lodz, Łódź, Poland, 2013. [Google Scholar]
- Cole, H.A.; Newmaster, S.G.; Bell, F.W.; Pitt, D.; Stinson, A. Influence of microhabitat on bryophyte diversity in Ontario mixedwood boreal forest. Can. J. For. Res. 2008, 38, 1867–1876. [Google Scholar] [CrossRef]
- Söderström, L. Dispersal as a limiting factors for distributions among epixylic bryophytes. Symp. Biol. Hung. 1987, 35, 475–483. [Google Scholar]
- Rajfur, M.; Plášek, V.; Zinicovscaia, I.; Świsłowski, P. Green sentinels in a changing climate: Moss biomonitoring reveals air pollution dynamics in New Zealand. Sci. Total Environ. 2026, 1011, 181185. [Google Scholar] [CrossRef] [PubMed]
- Rajfur, M.; Świsłowski, P.; Turlej, T.; Isinkaralar, O.; Isinkaralar, K.; Almasi, S.; Callegari, A.; Stoica, A.-I. Comparative (Bio)monitoring of Airborne PAHs Using Mosses and Filters. Molecules 2025, 30, 4009. [Google Scholar] [CrossRef]
- Isinkaralar, K.; Świsłowski, P.; Isinkaralar, O.; Rajfur, M.; Nguyen, T.N.T.; Di Palma, A.; Gautam, S. Assessing heavy metal pollution in urban children’s playgrounds: Comparative analysis of soil, dust, and moss bioaccumulation. J. Environ. Sci. 2026, 160, 700–712. [Google Scholar] [CrossRef]
- Klama, H.; Żarnowiec, J.; Jędrzejko, K. Mszaki Naziemne w Strukturze Zbiorowisk Roślinnych Rezerwatów Przyrody Makroregionu Południowego Polski; Politechnika Łódzka Filia w Bielsku-Białej: Bielsko-Biała, Poland, 1999; pp. 1–236. [Google Scholar]
- Darell, P.; Cronberg, N. Bryophytes in black alder swamps in south Sweden: Habitat classification, environmental factors and life-strategies. Lindbergia 2011, 34, 9–29. [Google Scholar]
- Staniaszek-Kik, M.; Zielińska, K.M.; Misztal, M. How do ditches contribute to bryophyte diversity in managed forests in East-Central Europe? Eur. J. Res. 2016, 135, 621–632. [Google Scholar] [CrossRef]
- Zielińska, K.M.; Staniaszek-Kik, M.; Misztal, M. Vascular plants and bryophytes in managed forests—Analysis of the impact of the old ditches on the species diversity (Central European Plain). Appl. Ecol. Environ. Res. 2017, 15, 1375–1392. [Google Scholar] [CrossRef]
- Dierssen, K. Distribution, Ecological Amplitude and Phytosociological Characterization of European Bryophytes; Bryophytorum Bibliotheca 56. J; Cramer: Stuttgart, Germany, 2001; pp. 1–289. [Google Scholar]
- Düll, R.; Meinunger, L. Deutschlands Moose. Die Verbreitung der Deutschen Moose in der BR Deutschland und in dre DDR, Ihre Höhen verbreitung, Ihre Arealtypen, Sowie Angaben zum Rückgang der Arten; Teil 1; IDH: Bad Münstereifl-Ohlerath, Germany, 1989; pp. 1–368. [Google Scholar]
- Hill, M.O.; Preston, C.D.; Bosanquet, S.D.S.; Roy, D.B. BRYOATT: Attributes of British and Irish Mosses, Liverworts and Hornworts with Information on Native Status, Size, Life Form, Life History, Geography and Habitat; Centre for Ecology end Hydrology (CEH) Publication: Huntingdon, UK, 2007; pp. 1–88. [Google Scholar]
- Smith, A.J.E. The Moss Flora of Britain and Ireland, 2nd ed.; Cambridge University Press: Cambridge, UK, 2004; pp. 1–1026. [Google Scholar]
- Mikulášková, E.; Fajmonová, Z.; Hájek, M. Invasion of central-European habitats by the moss Campylopus introflexus. Preslia 2012, 84, 863–886. [Google Scholar]
- Wolski, G.J.; Fudali, E. Species and ecological diversity of bryophytes occurring on midforest roads in some forest nature reserves in Central Poland. Bot. Steciana 2013, 17, 141–148. [Google Scholar]
- Olaczek, R. Formy Antropogenicznej Degeneracji Leśnych Zbiorowisk Roślinnych w Krajobrazie Rolniczym Polski Niżowej; Wyd. UŁ: Łódź, Poland, 1972; pp. 1–170. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.