Species, Climatypes, Climate Change, and Forest Health: A Conversion of Science to Practice for Inland Northwest (USA) Forests
Abstract
1. Introduction
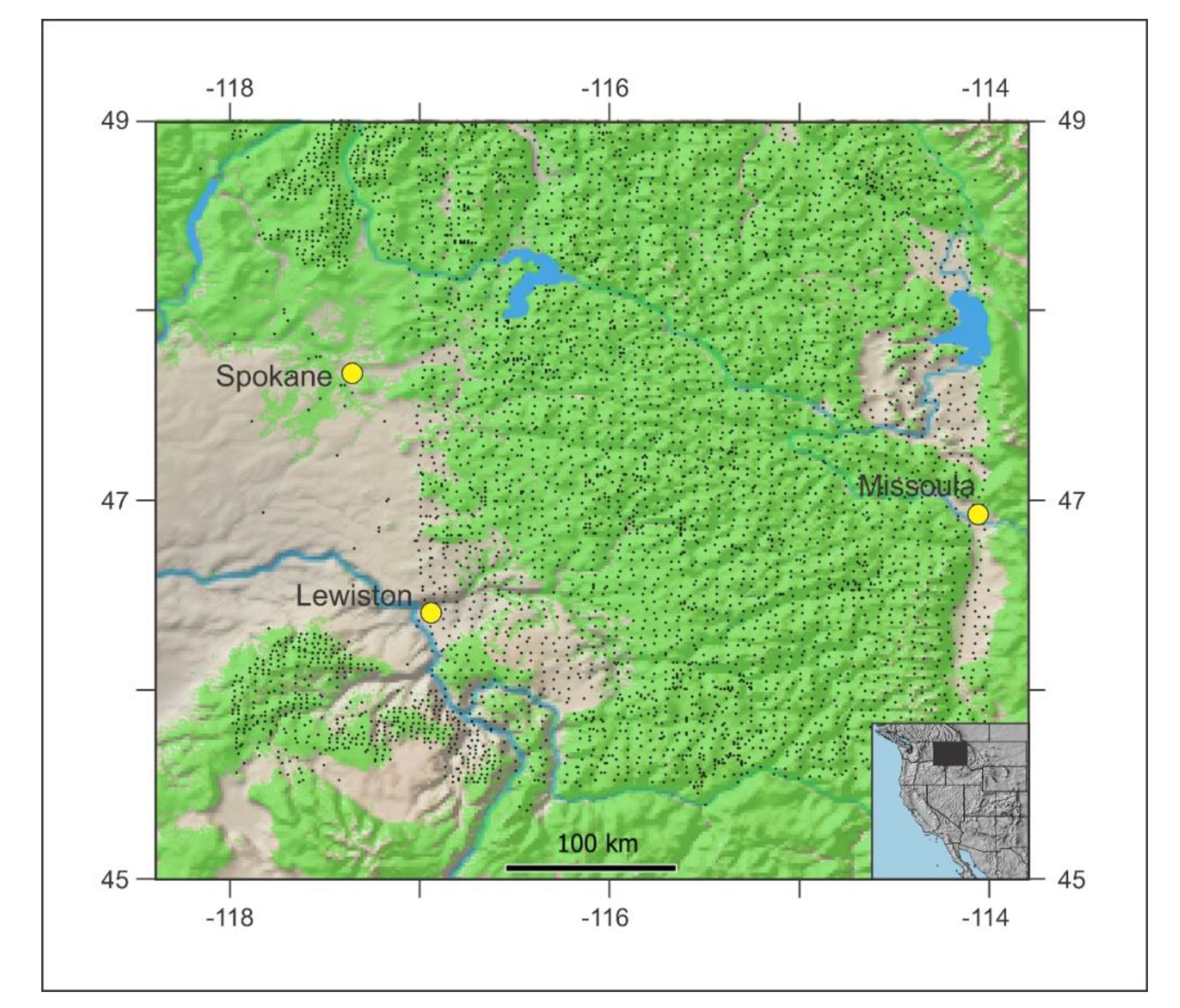
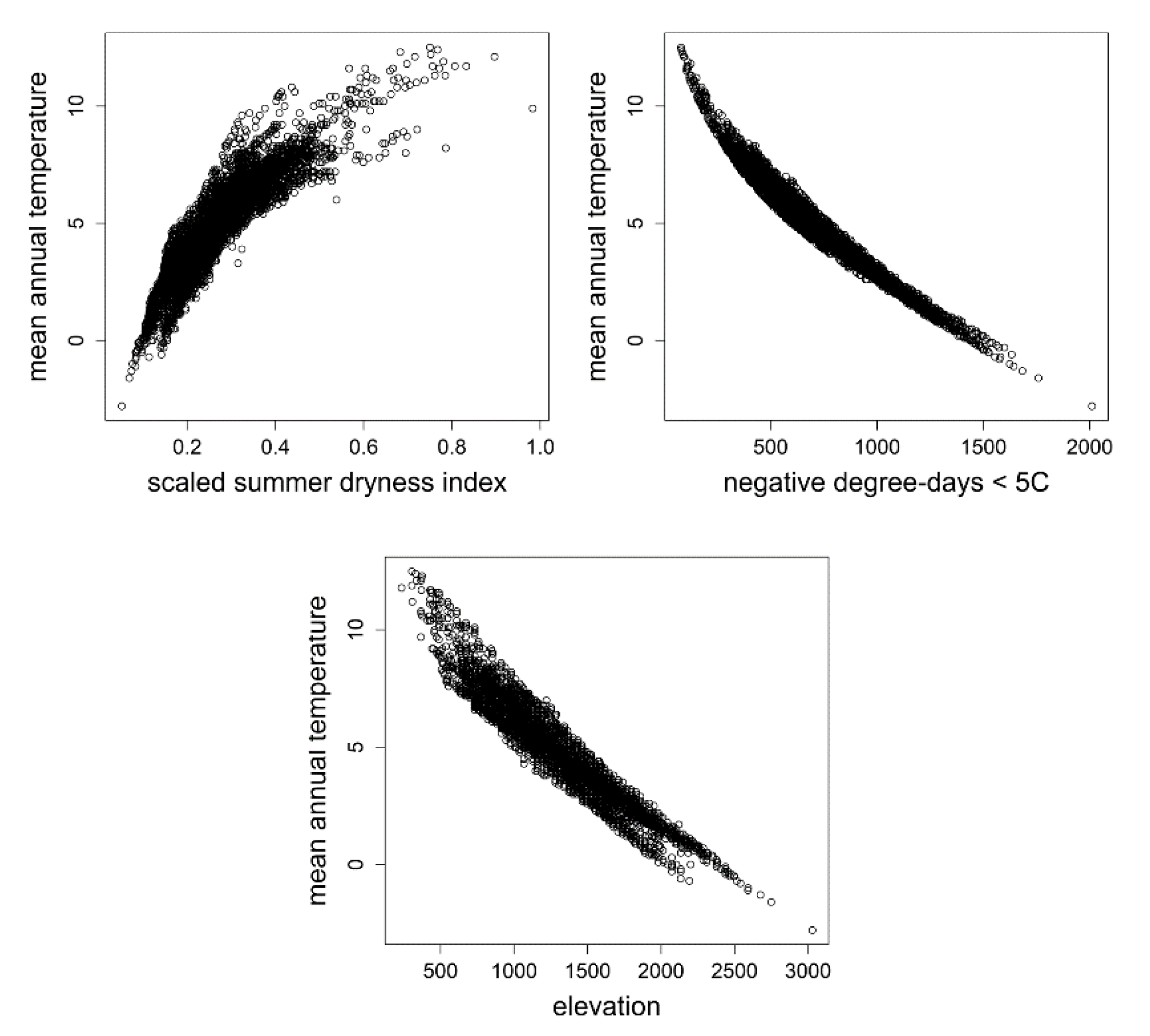
2. Methods
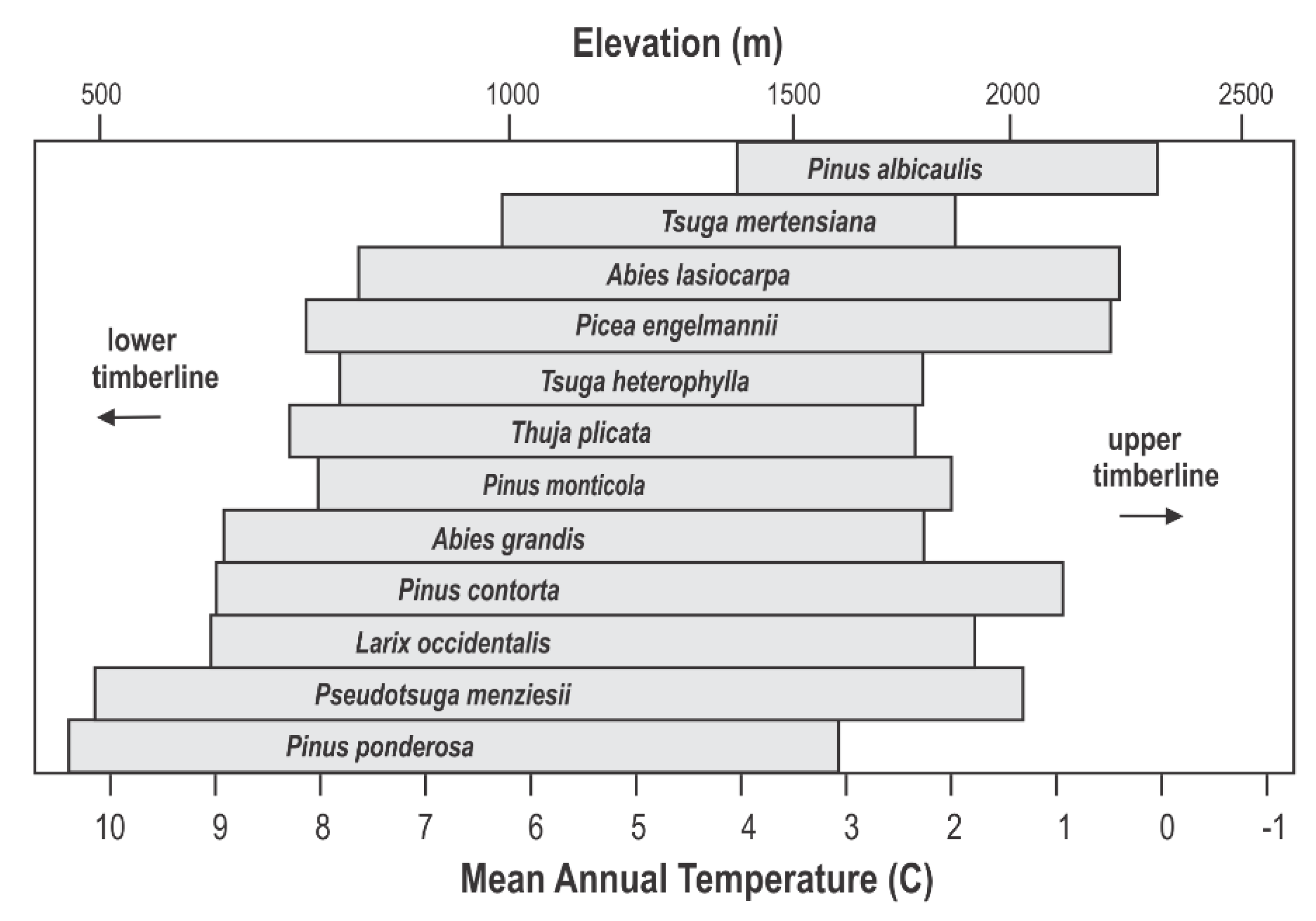
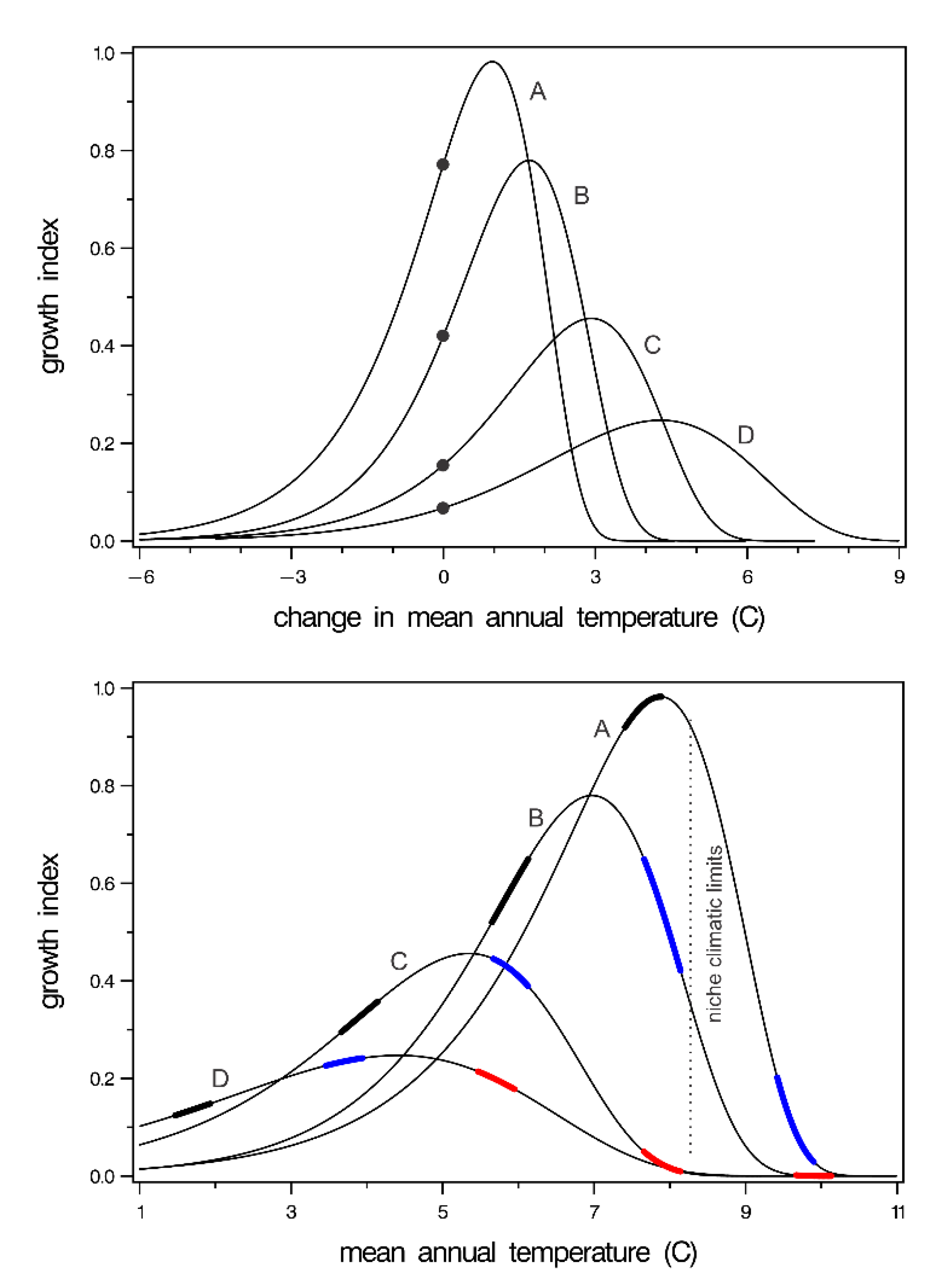
3. Results
4. Discussion
4.1. Managing the Current Generation
4.2. Considerations for Future Generations
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Schwartz, J. The Siberian Heat Wave? Yes, Climate Change Was a Big Factor. New York Times, 15 July 2020. [Google Scholar]
- Scheffers, B.R.; De Meester, L.; Bridge, T.C.L.; Hoffmann, A.A.; Pandolfi, J.M.; Corlett, R.T.; Butchart, S.H.M.; Pearce-kelly, P.; Kovacs, K.M.; Dudgeon, D.; et al. The broad footprint of climate change from genes to biomes to people. Science 2016, 354, aaf7671. [Google Scholar] [CrossRef] [PubMed]
- Sáenz-Romero, C.; Mendoza-Maya, E.; Gómez-Pineda, E.; Blanco-García, A.; Endara-Agramont, A.R.; Lindig-Cisneros, R.; López-Upton, J.; Trejo-Ramírez, O.; Wehenkel, C.; Cibrián-Tovar, D.; et al. Recent evidence of Mexican temperate forest decline and the need for ex situ conservation, assisted migration, and translocation of species ensembles as adaptive management to face projected climatic change impacts in a megadiverse country. Can. J. For. Res. 2020, 50, 843–854. [Google Scholar] [CrossRef]
- Bebber, D.P.; Ramotowski, M.A.T.; Gurr, S.J. Crop pests and pathogens move polewards in a warming world. Nat. Clim. Chang. 2013, 3, 985–988. [Google Scholar] [CrossRef]
- Anderegg, W.R.L.; Kane, J.M.; Anderegg, L.D.L. Consequences of widespread tree mortality triggered by drought and temperature stress. Nat. Clim. Chang. 2013, 3, 30–36. [Google Scholar] [CrossRef]
- Weed, A.S.; Ayres, M.P.; Hicke, J.A. Consequences of climate change for biotic disturbances in North American forests. Ecol. Monogr. 2013, 83, 441–470. [Google Scholar] [CrossRef]
- Parmesan, C. Ecological and evolutionary responses to recent climate change. Annu. Rev. Ecol. Evol. Syst. 2006, 37, 637–669. [Google Scholar] [CrossRef]
- Root, T.L.; Price, J.T.; Hall, K.R.; Schneider, S.H.; Rosenzweig, C.; Pounds, J.A. Fingerprints of global warming on wild animals and plants. Nature 2003, 421, 57–60. [Google Scholar] [CrossRef]
- Reyes-Fox, M.; Steltzer, H.; Trlica, M.J.; McMaster, G.S.; Andales, A.A.; LeCain, D.R.; Morgan, J.A. Elevated CO2 further lengthens growing season under warming conditions. Nature 2014, 510, 259–262. [Google Scholar] [CrossRef]
- Allen, C.D.; Macalady, A.K.; Chenchouni, H.; Bachelet, D.; McDowell, N.; Vennetier, M.; Kitzberger, T.; Rigling, A.; Breshears, D.D.; Hogg, E.H.; et al. A global overview of drought and heat-induced tree mortality reveals emerging climate change risks for forests. For. Ecol. Manag. 2010, 259, 660–684. [Google Scholar] [CrossRef]
- Breshears, D.D.; Cobb, N.S.; Rich, P.M.; Price, K.P.; Allen, C.D.; Balice, R.G.; Romme, W.H.; Kastens, J.H.; Floyd, M.L.; Belnap, J.; et al. Regional vegetation die-off in response to global-change-type drought. Proc. Natl. Acad. Sci. USA 2005, 102, 15144–15148. [Google Scholar] [CrossRef]
- Gitlin, A.R.; Sthultz, C.M.; Bowker, M.A.; Stumpf, S.; Paxton, K.L.; Kennedy, K.; Muñoz, A.; Bailey, J.K.; Whitham, T.G. Mortality gradients within and among dominant plant populations as barometers of ecosystem change during extreme drought. Conserv. Biol. 2006, 20, 1477–1486. [Google Scholar] [CrossRef] [PubMed]
- Jump, A.S.; Hunt, J.M.; Peñuelas, J. Rapid climate change-related growth decline at the southern range edge of Fagus sylvatica. Glob. Chang. Biol. 2006, 12, 2163–2174. [Google Scholar] [CrossRef]
- Michaelian, M.; Hogg, E.H.; Hall, R.J.; Arsenault, E. Massive mortality of aspen following severe drought along the southern edge of the Canadian boreal forest. Glob. Chang. Biol. 2011, 17, 2084–2094. [Google Scholar] [CrossRef]
- Shaw, J.D.; Steed, B.E.; DeBlander, L.T. Forest Inventory and Analysis (FIA) annual inventory answers the question: What is happening to pinyon-juniper woodlands? J. For. 2005, 103, 280–285. [Google Scholar]
- Woodall, C.W.; Oswalt, C.M.; Westfall, J.A.; Perry, C.H.; Nelson, M.D.; Finley, A.O. An indicator of tree migration in forests of the eastern United States. For. Ecol. Manag. 2009, 257, 1434–1444. [Google Scholar] [CrossRef]
- Lamprecht, A.; Semenchuk, P.R.; Steinbauer, K.; Winkler, M.; Pauli, H. Climate change leads to accelerated transformation of high-elevation vegetation in the central Alps. New Phytol. 2018, 220, 447–459. [Google Scholar] [CrossRef]
- Tchebakova, N.M.; Parfenova, E.I.; Soja, A.J. Potential climate-induced vegetation change in Siberia in the 21st century. In Environmental Change in Siberia; Baltzer, H., Ed.; Springer: Dordrecht, The Netherlands, 2010; pp. 67–82. [Google Scholar]
- Davis, M.B. Lags in vegetation response to greenhouse warming. Clim. Chang. 1989, 15, 75–82. [Google Scholar] [CrossRef]
- Davis, M.B.; Shaw, R.G. Range Shifts and Adaptive Responses to Quaternary Climate Change. Science 2001, 292, 673–679. [Google Scholar] [CrossRef]
- Aitken, S.N.; Yeaman, S.; Holliday, J.A.; Wang, T.; Curtis-McLane, S. Adaptation, migration or extirpation: Climate change outcomes for tree populations. Evol. Appl. 2008, 1, 95–111. [Google Scholar] [CrossRef]
- Rehfeldt, G.E.; Wykoff, W.R.; Ying, C.C. Physiologic Plasticity, Evolution, and Impacts of a Changing Climate on Pinus contorta. Clim. Chang. 2001, 50, 355–376. [Google Scholar] [CrossRef]
- Morgenstern, E.K. Geographic Variation in Forest Trees; UBC Press: Vancouver, BC, Canada, 1996. [Google Scholar]
- Grime, J.P. Plant Strategies and Vegetation Processes; John Wiley and Sons: Hoboken, NJ, USA, 1979. [Google Scholar]
- Loehle, C. Height Growth Rate Tradeoffs Determine Northern and Southern Range Limits for Trees. J. Biogeogr. 1998, 25, 735–742. [Google Scholar] [CrossRef]
- Rehfeldt, G.E. Microevolution of conifers in the northern Rocky Mountains: A view from common gardens. In Proceedings of the Eighth North American Forest Biology Workshop, Logan, UT, USA, 30 July–1 August 1984; pp. 32–46. [Google Scholar]
- Rehfeldt, G.E. Evolutionary genetics, the biological species and the ecology of the interior cedar-hemlock forests. In Proceedings of the Symposium: Interior Cedar-Hemlock-White Pine Forests: Ecology and Management, Spokane, WA, USA, 2–4 March 1993; Baumgartner, D.M., Lotan, J.E., Tonn, J.R., Eds.; Department of Natural Resources, Washington State University: Pullman, WA, USA, 1994; pp. 91–100. [Google Scholar]
- Leites, L.P.; Rehfeldt, G.E.; Steiner, K.C. Adaptation to climate in five eastern North America broadleaf deciduous species: Growth clines and evidence of the growth-cold tolerance trade-off. Perspect. Plant Ecol. Evol. Syst. 2019, 37, 64–72. [Google Scholar] [CrossRef]
- Rehfeldt, G.E.; Leites, L.P.; St. Clair, J.B.; Jaquish, B.C.; Sáenz-Romero, C.; López-Upton, J.; Joyce, D.G. Comparative genetic responses to climate in the varieties of Pinus ponderosa and Pseudotsuga menziesii: Clines in growth potential. For. Ecol. Manag. 2014, 324, 138–146. [Google Scholar] [CrossRef]
- Tchebakova, N.M.; Rehfeldt, G.E.; Parfenova, E.I. Impacts of climate change on the distribution of Larix spp. and Pinus sylvestris and their climatypes in Siberia. Mitig. Adapt. Strateg. Glob. Chang. 2006, 11, 861–882. [Google Scholar] [CrossRef]
- Rehfeldt, G.E.; Tchebakova, N.M.; Parfenova, E.I.; Pandalai, S.G. Genetic responses to climate and climate-change in conifers of the temperate and boreal forests. Recent Res. Dev. Genet Breed 2004, 1, 113–130. [Google Scholar]
- Turesson, G. The genotypical response of the plant species to the Habitat. Hereditas 1922, 3, 211–350. [Google Scholar] [CrossRef]
- Mátyás, C. Adaptation lag: A general feature of natural populations. (Invited lecture). In Proceedings of the WFGA-IUFRO Symposium, Olympia, WA, USA, 20–25 August 1990; No. 2.226. 10p. [Google Scholar]
- Rehfeldt, G.E.; Ying, C.C.; Spittlehouse, D.L.; Hamilton, D.A. Genetic responses to climate in Pinus contorta: Niche breadth, climate change, and reforestation. Ecol. Monogr. 1999, 69, 375–407. [Google Scholar] [CrossRef]
- Rehfeldt, G.E.; Tchebakova, N.M.; Parfenova, Y.I.; Wykoff, W.R.; Kuzmina, N.A.; Milyutin, L.I. Intraspecific responses to climate in Pinus sylvestris. Glob. Chang. Biol. 2002, 8, 912–929. [Google Scholar] [CrossRef]
- Betancourt, J.L. Late Quaternary Biogeography of the Colorado Plateau. In Packrat Middens, the Last 40,000 Years of Biotic Change; Betancourt, J.L., Van Devender, T.R., Martin, P.S., Eds.; Univ. Arizona Press: Tucson, AZ, USA, 1990; pp. 259–302. [Google Scholar]
- Millar, C.I.; Stephenson, N.L.; Stephens, S.L. Climate change and forests of the future: Managing in the face of uncertainty. Ecol. Appl. 2007, 17, 2145–2151. [Google Scholar] [CrossRef]
- Keenan, R.J. Climate change impacts and adaptation in forest management: A review. Ann. For. Sci. 2015, 72, 145–167. [Google Scholar] [CrossRef]
- Yousefpour, R.; Jacobsen, J.B.; Thorsen, B.J.; Meilby, H.; Hanewinkel, M.; Oehler, K. A review of decision-making approaches to handle uncertainty and risk in adaptive forest management under climate change. Ann. For. Sci. 2012, 69, 1–15. [Google Scholar] [CrossRef]
- Wang, T.; Campbell, E.M.; O’Neill, G.A.; Aitken, S.N. Projecting future distributions of ecosystem climate niches: Uncertainties and management applications. For. Ecol. Manag. 2012, 279, 128–140. [Google Scholar] [CrossRef]
- Elith, J.; Leathwick, J.R. Species distribution models: Ecological explanation and prediction across space and time. Annu. Rev. Ecol. Evol. Syst. 2009, 40, 677–697. [Google Scholar] [CrossRef]
- Iverson, L.; Prasad, A.; Matthews, S. Modeling potential climate change impacts on the trees of the northeastern United States. Mitig. Adapt. Strateg. Glob. Chang. 2008, 13, 487–516. [Google Scholar] [CrossRef]
- Gómez-Pineda, E.; Sáenz-Romero, C.; Ortega-Rodríguez, J.M.; Blanco-García, A.; Madrigal-Sánchez, X.; Lindig-Cisneros, R.; Lopez-Toledo, L.; Pedraza-Santos, M.E.; Rehfeldt, G.E. Suitable climatic habitat changes for Mexican conifers along altitudinal gradients under climatic change scenarios. Ecol. Appl. 2020, 30, e02041. [Google Scholar] [CrossRef]
- Aubry, C.; Devine, W.; Shoal, R.; Bower, A.; Miller, J.; Maggiulli, N. Climate Change and Forest Biodiversity: A Vulnerability Assessment and Action Plan for National Forests in Western Washington; US Department of Agriculture, Forest Service, Pacific Northwest Region: Olympia, WA, USA, 2011.
- Daubenmire, R.; Daubenmire, J.B. Forest Vegetation of Eastern Washington and Northern Idaho; Technical Bulletin 6; Washington State University: Pullman, WA, USA, 1968; p. 104. [Google Scholar]
- Daubenmire, R. Vegetation: Identification of typal communities. Science 1966, 151, 291–298. [Google Scholar] [CrossRef]
- Rehfeldt, G.E.; Ferguson, D.E.; Crookston, N.L. Quantifying the abundance of co-occurring conifers along inland northwest (USA) climate gradients. Ecology 2008, 89, 2127–2139. [Google Scholar] [CrossRef]
- Rehfeldt, G.E. Ecological adaptations in Douglas-Fir (Pseudotsuga menziesii var. glauca): A Synthesis. For. Ecol. Manag. 1989, 28, 203–215. [Google Scholar] [CrossRef]
- Rehfeldt, G.E. Ecological Genetics of Pinus contorta: A synthesis. Silvae Genet. 1988, 37, 131–135. [Google Scholar]
- Rehfeldt, G.E. A model of genetic variation for Pinus ponderosa in the Inland Northwest (USA): Applications in gene resource management. Can. J. For. Res. 1991, 21, 1491–1500. [Google Scholar] [CrossRef]
- Rehfeldt, G.E. Adaptation of Picea engelmannii populations to the heterogeneous environments of the Intermountain West. Can. J. Bot. 1994, 72, 1197–1208. [Google Scholar] [CrossRef]
- Rehfeldt, G.E. Genetic variation, climate models and the ecological genetics of Larix occidentalis. For. Ecol. Manag. 1995, 78, 21–37. [Google Scholar] [CrossRef]
- Warwell, M.V.; Shaw, R.G. Climate-related genetic variation in a threatened tree species, Pinus albicaulis. Am. J. Bot. 2017, 104, 1205–1218. [Google Scholar] [CrossRef] [PubMed]
- Bower, A.D.; Aitken, S.N. Ecological genetics and seed transfer guidelines for Pinus albicaulis (Pinaceae). Am. J. Bot. 2008, 95, 66–76. [Google Scholar] [CrossRef]
- Rehfeldt, G.E. Genetic structure of western red cedar populations in the Interior West. Can. J. For. Res. 1994, 24, 670–680. [Google Scholar] [CrossRef]
- Rehfeldt, G.E. Ecotypic differentiation in populations of Pinus monticola in north Idaho—Myth or reality? Am. Nat. 1979, 114, 627–636. [Google Scholar] [CrossRef]
- Mátyás, C. Modeling climate change effects with provenance test data. Tree Physiol. 1994, 14, 797–804. [Google Scholar] [CrossRef]
- Rehfeldt, G.E.; Leites, L.P.; Joyce, D.G.; Weiskittel, A.R. Role of population genetics in guiding ecological responses to climate. Glob. Chang. Biol. 2018, 24, 858–868. [Google Scholar] [CrossRef]
- Mátýas, C.; Yeatman, C.W. Effect of geographical transfer on growth and survival of jack pine (Pinus banksiana Lamb.) populations. Silvae Genet. 1992, 41, 370–376. [Google Scholar]
- Rehfeldt, G.E.; Tchebakova, N.M.; Milyutin, L.I.; Parfenova, E.I.; Wykoff, W.R.; Kouzmina, N.A. Assessing population responses to climate in Pinus sylvestris and Larix spp. of Eurasia with climate-transfer models. Eurasian J. For. Res. 2003, 6, 83–98. [Google Scholar]
- Namkoong, G. Nonoptimality of local races. In Proceedings of the 10th Southern Forest Tree Improvement Conference, College Station, TX, USA, 17–19 June 1969; pp. 149–153. [Google Scholar]
- Schmidtling, R.C. Use of provenance tests to predict response to climatic change: Loblolly pine and Norway spruce. Tree Physiol. 1994, 14, 805–817. [Google Scholar] [CrossRef] [PubMed]
- Carter, K.K. Provenance tests as Indicators of growth response to climate change in 10 north temperate tree species. Can. J. For. Res. 1996, 26, 1089–1095. [Google Scholar] [CrossRef]
- Jackson, S.T.; Overpeck, J.T. Responses of plant populations and communities to environmental changes of the late Quaternary. Paleobiology 2000, 26, 194–220. [Google Scholar] [CrossRef]
- Williams, J.W.; Jackson, S.T. Novel climates, no-analog communities, and ecological surprises. Paleoecology 2007, 5, 475–482. [Google Scholar] [CrossRef]
- Rehfeldt, G.E.; Crookston, N.L.; Warwell, M.V.; Evans, J.S. Empirical analyses of plant-climate relationships for the western United States. Int. J. Plant Sci. 2006, 167, 1123–1150. [Google Scholar] [CrossRef]
- Rehfeldt, G.E.; Crookston, N.L.; Sáenz-Romero, C.; Campbell, E.M. North American vegetation model for land-use planning in a changing climate: A solution to large classification problems. Ecol. Appl. 2012, 22, 119–141. [Google Scholar] [CrossRef]
- Kirtman, B.; Power, S.B.; Adedoyin, A.J.; Boer, G.J.; Bojariu, R.; Camilloni, I.; Doblas-Reyes, F.; Fiore, A.M.; Kimoto, M.; Meehl, G.; et al. Near-term climate change: Projections and predictability. In Climate Change 2013: The Physical Science Basis. The Working Group I Contribution to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Cambridge University Press: Cambridge, UK; Cambridge, MA, USA, 2013; Volume 9781107057, pp. 953–1028. [Google Scholar]
- Jackson, S.T.; Betancourt, J.L.; Booth, R.K.; Gray, S.T. Ecology and the ratchet of events: Climate variability, niche dimensions, and species distributions. Proc. Natl. Acad. Sci. USA 2009, 106, 19685–19692. [Google Scholar] [CrossRef]
- Körner, C. Alpine Treelines. Functional Ecology of the Global High Elevation Tree Limits, 1st ed.; Springer: Basel, Switzerland, 2012; ISBN 978-3-0348-0395-3. [Google Scholar]
- Matteodo, M.; Wipf, S.; Stöckli, V.; Rixen, C.; Vittoz, P. Elevation gradient of successful plant traits for colonizing alpine summits under climate change. Environ. Res. Lett. 2013, 8, 024043. [Google Scholar] [CrossRef]
- Stöckli, V.; Wipf, S.; Nilsson, C.; Rixen, C. Using historical plant surveys to track biodiversity on mountain summits. Plant Ecol. Divers. 2012, 4, 415–425. [Google Scholar] [CrossRef]
- Grabherr, G.; Gottfried, M.; Pauli, H. Climate effects on mountain plants. Nature 1994, 369, 448. [Google Scholar] [CrossRef]
- Rehfeldt, G.E.; Jaquish, B.C.; Sáenz-Romero, C.; Joyce, D.G.; Leites, L.P.; St. Clair, J.B.; López-Upton, J. Comparative genetic responses to climate in the varieties of Pinus ponderosa and Pseudotsuga menziesii: Reforestation. For. Ecol. Manag. 2014, 324, 147–157. [Google Scholar] [CrossRef]
- Keiter, R.; Boling, T.; Milkman, L. Legal perspectives on ecosystem management: Legitimizing a new federal land management policy. In Ecological Stewardship, A Common Reference for Ecosystem Management, Volume III; Sexton, W.T., Malk, A.J., Szaro, R.C., Johnson, N.C., Eds.; Elsevier Science Ltd.: Kidlington, UK, 1999; pp. 9–41. [Google Scholar]


| Species | Climatype Breadth (m Elevation) | Reference |
|---|---|---|
| Pseudotsuga menziesii | Nonlinear: ca. 240 at low elevation, 350 at mid elevation, 1 climatype for elevation > 2000 | [48] |
| Pinus contorta | 300 | [49] |
| Pinus ponderosa | 400 | [50] |
| Picea engelmannii | 420 | [51] |
| Larix occidentalis | 450 | [52] |
| Pinus albicaulis | 450 | [53,54] |
| Thuja plicata | 600 | [55] |
| Pinus monticola | no detectable climatypes | [56] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rehfeldt, G.E.; Warwell, M.V.; Monserud, R.A. Species, Climatypes, Climate Change, and Forest Health: A Conversion of Science to Practice for Inland Northwest (USA) Forests. Forests 2020, 11, 1237. https://doi.org/10.3390/f11121237
Rehfeldt GE, Warwell MV, Monserud RA. Species, Climatypes, Climate Change, and Forest Health: A Conversion of Science to Practice for Inland Northwest (USA) Forests. Forests. 2020; 11(12):1237. https://doi.org/10.3390/f11121237
Chicago/Turabian StyleRehfeldt, Gerald E., Marcus V. Warwell, and Robert A. Monserud. 2020. "Species, Climatypes, Climate Change, and Forest Health: A Conversion of Science to Practice for Inland Northwest (USA) Forests" Forests 11, no. 12: 1237. https://doi.org/10.3390/f11121237
APA StyleRehfeldt, G. E., Warwell, M. V., & Monserud, R. A. (2020). Species, Climatypes, Climate Change, and Forest Health: A Conversion of Science to Practice for Inland Northwest (USA) Forests. Forests, 11(12), 1237. https://doi.org/10.3390/f11121237




