Polymer Crosslinked Activated Carbon Pellets for Dye Adsorption
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Instrumentation
2.2. Experimental Method
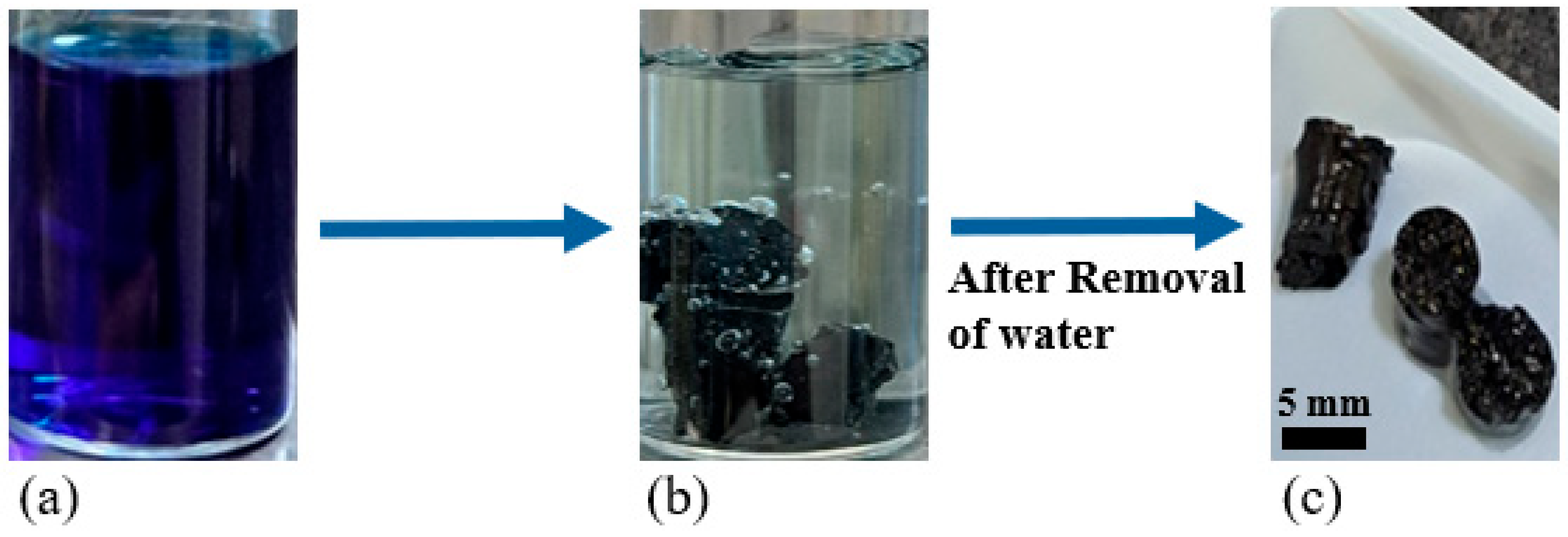
2.3. Encapsulation of Activated Carbon
- 80 °C for 8 h: Initiates gelation and network formation.
- 120 °C for 4 h: Converts the gel into a more continuous polymer network.
- 160 °C for 4 h: Promotes further crosslinking and network stabilization.
- 190 °C for 8 h: Completes the curing process, ensuring a fully crosslinked, mechanically robust, and solvent-free pellet structure as explained by Figures S19 and S20 and Table S4 of EDS analysis.
3. Results and Discussion
3.1. Surface Morphology Examination
3.2. FTIR and Thermal Analysis
3.3. X-Ray Diffraction Analysis
3.4. Compressive Strength
3.5. Swelling and Water Absorbance Analysis
3.6. BET Analysis
3.7. Adsorption Analysis
3.8. Adsorption Mechanisms: Isotherms, Kinetics, and Thermodynamics Studies
3.8.1. Kinetic Studies
3.8.2. Adsorption Isotherm Studies
3.8.3. Thermodynamic Studies
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
AI Declaration
Conflicts of Interest
References
- Mechnou, I.; Meskini, S.; Elqars, E.; El Had, M.A.; Hlaibi, M. Efficient CO2 capture using a novel Zn-doped activated carbon developed from agricultural liquid biomass: Adsorption study, mechanism and transition state. Surf. Interfaces 2024, 52, 104846. [Google Scholar] [CrossRef]
- Serafin, J.; Dziejarski, B. Activated Carbons—Preparation, Characterization and Their Application in CO2 Capture: A Review; Springer: Berlin/Heidelberg, Germany, 2024; Volume 31. [Google Scholar] [CrossRef]
- Soonmin, H.; Kabbashi, N.A. Review on activated carbon: Synthesis, properties and applications. Int. J. Eng. Trends Technol. 2021, 69, 124–139. [Google Scholar] [CrossRef]
- Muniandy, L.; Adam, F.; Mohamed, A.R.; Ng, E.P. The synthesis and characterization of high purity mixed microporous/mesoporous activated carbon from rice husk using chemical activation with NaOH and KOH. Microporous Mesoporous Mater. 2014, 197, 316–323. [Google Scholar] [CrossRef]
- Mechnou, I.; Benabdallah, A.; Chham, A.-I.; Rachdi, Y.; Hlaibi, M.; El Kartouti, A.; Saleh, N. Activated carbons for effective pharmaceutical adsorption: Impact of feedstock origin, activation agents, adsorption conditions, and cost analysis. Results Eng. 2025, 27, 105966. [Google Scholar] [CrossRef]
- Krahnstöver, T.; Wintgens, T. Separating powdered activated carbon (PAC) from wastewater—Technical process options and assessment of removal efficiency. J. Environ. Chem. Eng. 2018, 6, 5744–5762. [Google Scholar] [CrossRef]
- Satyam, S.; Patra, S. Innovations and challenges in adsorption-based wastewater remediation: A comprehensive review. Heliyon 2024, 10, e29573. [Google Scholar] [CrossRef]
- Han, Z.; Sani, B.; Mrozik, W.; Obst, M.; Beckingham, B.; Karapanagioti, H.K.; Werner, D. Magnetite impregnation effects on the sorbent properties of activated carbons and biochars. Water Res. 2015, 70, 394–403. [Google Scholar] [CrossRef]
- Singh, B.; Kemell, M.L.; Repo, T. Porous Carbon Pellets for Physical Adsorption of CO2: Size and Shape Effect. Mater. Adv. 2024, 5, 7601–7608. [Google Scholar] [CrossRef]
- Kim, J.W.; Hong, S.M.; Kang, J.W.; Yang, J.H.; Jeon, S.C. Enhanced mechanical stability with hierarchical porosity of activated carbon pellets realized through the use of combined polymer binders. J. Korean Ceram. Soc. 2024, 62, 103–113. [Google Scholar] [CrossRef]
- Malik, M.I.; Rouabah, M.; Abatzoglou, N.; Achouri, I.E. Dry reforming of methane at high temperature and elevated pressure over nickel spinellized powder catalyst and pellets prepared from a metallurgical residue. Biofuels Bioprod. Biorefining 2024, 18, 1027–1046. [Google Scholar] [CrossRef]
- Tepe, M.; Avci, I.; Abukay, D. Effect of pelletization pressure on structural properties and critical current hysteresis of ceramic superconducting Bi1.7Pb0.3Sr2Ca2Cu3Oy. Phys. Status Solidi Appl. Res. 2003, 198, 420–426. [Google Scholar] [CrossRef]
- Topal, U. Influence of pelletization pressure on the structural and transport properties of TlCaBaCuO superconductors. Phys. Status Solidi Appl. Mater. Sci. 2007, 204, 4259–4265. [Google Scholar] [CrossRef]
- Herman, J.; Remon, J.P.; Lefebvre, R.; Bogaert, M.; Klinger, G.H.; Schwartz, J.B. The Dissolution Rate and Bioavailability of hydrochlorothiazide in Pellet Formulations. J. Pharm. Pharmacol. 1988, 40, 157–160. [Google Scholar] [CrossRef]
- Mehta, S.; De Beer, T.; Remon, J.P.; Vervaet, C. Effect of disintegrants on the properties of multiparticulate tablets comprising starch pellets and excipient granules. Int. J. Pharm. 2012, 422, 310–317. [Google Scholar] [CrossRef] [PubMed]
- Pai, R.; Kohli, K.; Shrivastava, B. Compression and evaluation of extended release matrix pellets prepared by the extrusion/spheronization process into disintegrating tablets. Braz. J. Pharm. Sci. 2012, 48, 117–129. [Google Scholar] [CrossRef]
- Wiwattanapatapee, R.; Pengnoo, A.; Kanjanamaneesathian, M.; Matchavanich, W.; Nilratana, L.; Jantharangsri, A. Floating pellets containing bacterial antagonist for control sheath blight of rice: Formulations, viability and bacterial release studies. J. Control. Release 2004, 95, 455–462. [Google Scholar] [CrossRef]
- Charoenthai, N.; Kleinebudde, P.; Puttipipatkhachorn, S. Influence of Chitosan type on the properties of extruded pellets with low amount of microcrystalline cellulose. AAPS PharmSciTech 2007, 8, E99–E109. [Google Scholar] [CrossRef]
- Hu, L.D.; Liu, Y.; Tang, X.; Zhang, Q. Preparation and in vitro/in vivo evaluation of sustained-release metformin hydrochloride pellets. Eur. J. Pharm. Biopharm. 2006, 64, 185–192. [Google Scholar] [CrossRef]
- Alamdari, N.S. Preparation and evaluation of sustained release pellets of Tramadol. African J. Pharm. Pharmacol. 2012, 6, 2123–2132. [Google Scholar] [CrossRef]
- Gawale, D.; Sonawane, R.O.; Pandey, V.V.; Pundlikarao, P. Formulation and In-vitro Characterization of Multiple Unit Sustained Release Matrix Pellets of Lornoxicam Using Natural Gums. Indian J. Nov. Drug Deliv. 2013, 5, 208–220. [Google Scholar]
- Kadajji, V.G.; Betageri, G.V. Water soluble polymers for pharmaceutical applications. Polymers 2011, 3, 1972–2009. [Google Scholar] [CrossRef]
- Zicari, T.J.; Scher, H.; Santa-Maria, M.; Strobel, S. Spray Dry Method for Encapsulation of Biological Moieties and Chemicals in Polymers Cross-Linked by Multivalent Ions for Controlled Release Applications. U.S. Patent 9,700,519, 11 July 2017. [Google Scholar]
- Mezreb, N.; Charrueau, C.; Boy, P.; Allain, P.; Chaumeil, J.C. Production of Carbopol® 974P and Carbopol® 971P pellets by extrusion-spheronization: Optimization of the processing parameters and water content. Drug Dev. Ind. Pharm. 2004, 30, 481–490. [Google Scholar] [CrossRef]
- Hussin, F.; Hazani, N.N.; Aroua, M.K. Effect of binder ratio on the physical properties of porous carbon pellet for CO2 capture. Mater. Today Proc. 2023. [Google Scholar] [CrossRef]
- Bavykina, A.V.; Rozhko, E.; Goesten, M.G.; Wezendonk, T.; Seoane, B.; Kapteijn, F.; Makkee, M.; Gascon, J. Shaping Covalent Triazine Frameworks for the Hydrogenation of Carbon Dioxide to Formic Acid. ChemCatChem 2016, 8, 2217–2221. [Google Scholar] [CrossRef]
- Haider, S.K.; Moon, S.; Park, H.; Yoon, H.; Yoon, S. Pelletizing highly porous covalent triazine framework under mild conditions and applying it as adsorbent for Ru3+ ions. Microporous Mesoporous Mater. 2024, 365, 112852. [Google Scholar] [CrossRef]
- Makarov, I.S.; Shambilova, G.K.; Vinogradov, M.I.; Anokhina, T.S.; Bukanova, A.S.; Kairliyeva, F.B.; Bukanova, S.K.; Levin, I.S. Membranes Based on Cellulose and Copolymers of Acrylonitrile Prepared from Joint Solutions. Membranes 2023, 13, 667. [Google Scholar] [CrossRef] [PubMed]
- Makarov, I.S.; Vinogradov, M.I.; Golova, L.K.; Arkharova, N.A.; Shambilova, G.K.; Makhatova, V.E.; Naukenov, M.Z. Design and Fabrication of Membranes Based on PAN Copolymer Obtained from Solutions in N-methylmorpholine-N-oxide. Polymers 2022, 14, 2861. [Google Scholar] [CrossRef]
- Brown, J.; Rhoney, I.; Pethrick, R.A. Epoxy resin based nanocomposites: 1. Diglycidylether of bisphenol A (DGEBA) with triethylenetetramine (TETA). Polym. Int. 2004, 53, 2130–2137. [Google Scholar] [CrossRef]
- Thomas, R.; Durix, S.; Sinturel, C.; Omonov, T.; Goossens, S.; Groeninckx, G.; Moldenaers, P.; Thomas, S. Cure kinetics, morphology and miscibility of modified DGEBA-based epoxy resin—Effects of a liquid rubber inclusion. Polymer 2007, 48, 1695–1710. [Google Scholar] [CrossRef]
- Long, J.; Nand, A.V.; Bunt, C.; Seyfoddin, A. Controlled release of dexamethasone from poly(vinyl alcohol) hydrogel. Pharm. Dev. Technol. 2019, 24, 839–848. [Google Scholar] [CrossRef] [PubMed]
- Rao, P.S.; Sridhar, S.; Krishnaiah, A. Dehydration of tetrahydrofuran by pervaporation using crosslinked PVA/PEI blend membranes. J. Appl. Polym. Sci. 2006, 102, 1152–1161. [Google Scholar] [CrossRef]
- Nataraj, D.; Reddy, R.; Reddy, N. Crosslinking electrospun poly (vinyl) alcohol fibers with citric acid to impart aqueous stability for medical applications. Eur. Polym. J. 2020, 124, 109484. [Google Scholar] [CrossRef]
- Sonker, A.K.; Rathore, K.; Nagarale, R.K.; Verma, V. Crosslinking of Polyvinyl Alcohol (PVA) and Effect of Crosslinker Shape (Aliphatic and Aromatic) Thereof. J. Polym. Environ. 2018, 26, 1782–1794. [Google Scholar] [CrossRef]
- Trung, N.V.; Thi, N.P.; Nguyen, T.H.; Nguyen, M.N.; Anh, D.T.; Trung, T.N.; Quang, T.T.; Van, H.T.; Thi, T.T. Tuning the thermal and mechanical properties of poly(vinyl alcohol) with 2,5-furandicarboxylic acid acting as a biobased crosslinking agent. Polym. J. 2022, 54, 335–343. [Google Scholar] [CrossRef]
- Nascimento, F.C.D.; de Aguiar, L.C.V.; Costa, L.A.T.; Fernandes, M.T.; Marassi, R.J.; Gomes, A.d.S.; de Castro, J.A. Formulation and characterization of crosslinked polyvinyl alcohol (PVA) membranes: Effects of the crosslinking agents. Polym. Bull. 2021, 78, 917–929. [Google Scholar] [CrossRef]
- Falath, W.; Sabir, A.; Jacob, K.I. Highly improved reverse osmosis performance of novel PVA/DGEBA cross-linked membranes by incorporation of Pluronic F-127 and MWCNTs for water desalination. Desalination 2016, 397, 53–66. [Google Scholar] [CrossRef]
- Chaouat, M.; Le Visage, C.; Baille, W.E.; Escoubet, B.; Chaubet, F.; Mateescu, M.A.; Letourneur, D. A novel cross-linked poly(vinyl alcohol) (PVA) for vascular grafts. Adv. Funct. Mater. 2008, 18, 2855–2861. [Google Scholar] [CrossRef]
- Zhao, H.; Li, X.; Zhang, L.; Hu, Z.; Zhong, L.; Xue, J. Preparation and bacteriostatic research of porous polyvinyl alcohol/biochar/nanosilver polymer gel for drinking water treatment. Sci. Rep. 2021, 11, 12205. [Google Scholar] [CrossRef]
- Ismail, A.S.; Jawaid, M.; Hamid, N.H.; Yahaya, R.; Hassan, A.; Asim, M.; Supian, A.B.M. Effect of Curing Temperature on Mechanical Properties of Bio-phenolic/Epoxy Polymer Blends. J. Polym. Environ. 2022, 30, 878–885. [Google Scholar] [CrossRef]
- Rynkowska, E.; Fatyeyeva, K.; Kujawa, J. Chemically and Thermally Crosslinked PVA-Based Membranes: Effect on Swelling and Transport Behavior. Polymers 2019, 11, 1799. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Y.; Liu, J.; Li, C.; Wang, S. Preparation and Properties of Phosphorylated and Crosslinked Poly(vinyl alcohol)/Bisphenol A–Epoxy Resin Membranes for Fuel Cell Applications. J. Appl. Polym. Sci. 2011, 122, 3071–3079. [Google Scholar] [CrossRef]
- González, M.G.; Cabanelas, J.C.; Baselga, J. Applications of FTIR on Epoxy Resins—Identification, Monitoring the Curing Process, Phase Separation and Water Uptake. Infrared Spectrosc. Mater. Sci. Eng. Technol. 2012, 2, 261–284. [Google Scholar] [CrossRef]
- Sawasdee, S.; Watcharabundit, P. Characterization and Adsorption Mechanism of Methylene Blue Dye by Mesoporous Activated Carbon Prepared from Rice Husks. Environ. Nat. Resour. J. 2023, 21, 458–470. [Google Scholar] [CrossRef]
- Jiang, R.; Ren, F. Adsorption of Methylene Blue Dye by Modified Reed Activated Carbon: Adsorption Optimization and Adsorption Performance. Pol. J. Environ. Stud. 2025, 34, 2223–2232. [Google Scholar] [CrossRef]
- Zhang, P.; An, L.; Zhao, C.; Chang, Q.; Wang, F.; Zhang, C.; Zhu, Y.; Yang, Y.; Li, Y. ‘Honeycomb catalytic strategy’ for carbonylation reaction based on the structural evolution of cobalt species. Catal. Sci. Technol. 2024, 14, 2235–2243. [Google Scholar] [CrossRef]
- Jin, X.; Li, L.; Xu, R.; Liu, Q.; Ding, L.; Pan, Y.; Wang, C.; Hung, W.; Lee, K.; Wang, T. Effects of thermal cross-linking on the structure and property of asymmetric membrane prepared from the polyacrylonitrile. Polymers 2018, 10, 539. [Google Scholar] [CrossRef] [PubMed]
- Matatov-Meytal, Y.I.; Sheintuch, M.; Shter, G.E.; Grader, G.S. Optimal temperatures for catalytic regeneration of activated carbon. Carbon 1997, 35, 1527–1531. [Google Scholar] [CrossRef]
- De Oliveira, G.F.; De Andrade, R.C.; Trindade, M.A.G.; Andrade, H.M.C.; De Carvalho, C.T. Thermogravimetric and spectroscopic study (Tg-DTA/FT-IR) of activated carbon from the renewable biomass source babassu. Quim. Nova 2017, 40, 284–292. [Google Scholar] [CrossRef]
- Ali, M.; Mir, S.; Atanase, L.I.; Abid, O.U.R.; Kazi, M.; Atanase, L.I. Chitosan-PVA-PVP/nano-clay composite: A promising tool for controlled drug delivery. RSC Adv. 2024, 14, 15777–15790. [Google Scholar] [CrossRef] [PubMed]
- Vapor, W.; Packaging, C. Crosslinked PVA/Nanoclay Hydrogel Coating for Improving. Coatings 2022, 12, 1562. [Google Scholar] [CrossRef]
- Manoj, B.; Kunjomana, A.G. Study of stacking structure of amorphous carbon by X-ray diffraction technique. Int. J. Electrochem. Sci. 2012, 7, 3127–3134. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, J.; Liu, Z. Tubular composite of doped polyaniline with multi-walled carbon nanotubes. Appl. Phys. A Mater. Sci. Process. 2005, 80, 1813–1817. [Google Scholar] [CrossRef]
- Islam, M.S.; Ang, B.C.; Gharehkhani, S.; Afifi, A.B.M. Adsorption capability of activated carbon synthesized from coconut shell. Carbon Lett. 2016, 20, 1–9. [Google Scholar] [CrossRef]
- Bardestani, R.; Patience, G.S.; Kaliaguine, S. Experimental methods in chemical engineering: Specific surface area and pore size distribution measurements—BET, BJH, and DFT. Can. J. Chem. Eng. 2019, 97, 2781–2791. [Google Scholar] [CrossRef]
- Tang, S.H.; Zaini, M.A.A. Development of activated carbon pellets using a facile low-cost binder for effective malachite green dye removal. J. Clean. Prod. 2020, 253, 119970. [Google Scholar] [CrossRef]
- Li, S.; Cui, Y.; Wen, M.; Ji, G. Toxic Effects of Methylene Blue on the Growth, Reproduction and Physiology of Daphnia magna. Toxics 2023, 11, 594. [Google Scholar] [CrossRef]
- Oladoye, P.O.; Ajiboye, T.O.; Omotola, E.O.; Oyewola, O.J. Methylene blue dye: Toxicity and potential elimination technology from wastewater. Results Eng. 2022, 16, 100678. [Google Scholar] [CrossRef]
- Ren, Y.; Chen, F.; Pan, K.; Zhao, Y.; Ma, L.; Wei, S. Studies on kinetics, isotherms, thermodynamics and adsorption mechanism of methylene blue by N and S co-doped porous carbon spheres. Nanomaterials 2021, 11, 1819. [Google Scholar] [CrossRef] [PubMed]
- Ghaffar, A.; Younis, M.N. Interaction and thermodynamics of methylene blue adsorption on oxidized multi-walled carbon nanotubes. Green Process. Synth. 2015, 4, 209–217. [Google Scholar] [CrossRef]
- Xue, H.; Wang, X.; Xu, Q.; Dhaouadi, F.; Sellaoui, L.; Seliem, M.K.; Ben Lamine, A.; Belmabrouk, H.; Bajahzar, A.; Bonilla-Petriciolet, A.; et al. Adsorption of methylene blue from aqueous solution on activated carbons and composite prepared from an agricultural waste biomass: A comparative study by experimental and advanced modeling analysis. Chem. Eng. J. 2022, 430, 132801. [Google Scholar] [CrossRef]
- Pathania, D.; Sharma, S.; Singh, P. Removal of methylene blue by adsorption onto activated carbon developed from Ficus carica bast. Arab. J. Chem. 2017, 10, S1445–S1451. [Google Scholar] [CrossRef]
- Thang, N.H.; Khang, D.S.; Hai, T.D.; Nga, D.T.; Tuan, P.D. Methylene blue adsorption mechanism of activated carbon synthesised from cashew nut shells. RSC Adv. 2021, 11, 26563–26570. [Google Scholar] [CrossRef] [PubMed]
- El Qada, E.N.; Allen, S.J.; Walker, G.M. Adsorption of basic dyes from aqueous solution onto activated carbons. Chem. Eng. J. 2008, 135, 174–184. [Google Scholar] [CrossRef]
- Khan, S.A.; Arshad, Z.; Shahid, S.; Arshad, I.; Rizwan, K.; Sher, M.; Fatima, U. Synthesis of TiO2/Graphene oxide nanocomposites for their enhanced photocatalytic activity against methylene blue dye and ciprofloxacin. Compos. Part B Eng. 2019, 175, 107120. [Google Scholar] [CrossRef]
- Fito, J.; Abewaa, M.; Mengistu, A.; Angassa, K.; Ambaye, A.D.; Moyo, W.; Nkambule, T. Adsorption of methylene blue from textile industrial wastewater using activated carbon developed from Rumex abyssinicus plant. Sci. Rep. 2023, 13, 5427. [Google Scholar] [CrossRef]
- Alharby, N.F.; Almutairi, R.S.; Mohamed, N.A. Adsorption behavior of methylene blue dye by novel crosslinked o-cm-chitosan hydrogel in aqueous solution: Kinetics, isotherm and thermodynamics. Polymers 2021, 13, 3659. [Google Scholar] [CrossRef]
- El Jery, A.; Alawamleh, H.S.K.; Sami, M.H.; Abbas, H.A.; Sammen, S.S.; Ahsan, A.; Imteaz, M.A.; Shanableh, A.; Shafiquzzaman, M.; Osman, H.; et al. Isotherms, kinetics and thermodynamic mechanism of methylene blue dye adsorption on synthesized activated carbon. Sci. Rep. 2024, 14, 970. [Google Scholar] [CrossRef]
- Inthapat, P.; Worasuwannarak, N.; Li, X.; Yao, H.; Sutthasupa, S.; Prachakittikul, P.; Koo-amornpattana, W.; Klaitong, P.; Chaiwat, W. Adsorption of Methylene Blue Using As-Developed Binderless Hot-Pressed Granular Activated Carbon Derived from Sugarcane Bagasse Residues. ACS Omega 2025, 10, 34618–34632. [Google Scholar] [CrossRef]







| Compositions | Compressive Strength | ESR a | EWC b |
|---|---|---|---|
| 30 wt.% Binder’s Pellets | 3.37 ± 0.46 MPa | 64% | 39% |
| 25 wt.% Binder’s Pellets | 2.13 ± 0.60 MPa | 62% | 38% |
| 18 wt.% Binder’s Pellets | 1.71 ± 0.69 MPa | 56% | 35% |
| Properties | Grinded Activated Carbon | 30 wt.% Binder’s AC Pellet |
|---|---|---|
| BET Surface Area (m2/g) | 590.5 | 154.2 |
| Total Pore Volume (cm3/g) | 0.698 | 0.403 |
| Average Pore Diameter (nm) | 2.36 | 5.23 |
| Volume of N2 adsorbed (Vm cm3(STP) g−1) | 135.6 | 35.44 |
| Model | Equation | Parameters | Value | R2 |
|---|---|---|---|---|
| Pseudo-first order | ln (qe − qt) = ln qe − k1 t | (qe) (exp) | 3.24 mg·g−1 | 0.99 |
| (k1) | 0.077 min−1 | |||
| Pseudo- second order | t/qt = 1/(k2 qe2) +t/qe | (qe) (model) | 5.10 mg·g−1 | 0.96 |
| (k2) | 5.1 × 10−3 g·mg−1·min−1 | |||
| Intraparticle diffusion | qt = kid t1/2 + C | kid | 0.45 mg·g−1·min−1/2 | 0.95 |
| C | −0.22 mg·g−1 |
| Model | Equation | Parameter | Value | R2 |
|---|---|---|---|---|
| Langmuir | (mg g−1) | 9.68 | 0.85 | |
| (L mg−1) | 0.41 | |||
| Freundlich | (mg g−1) (L mg−1)1/n | 2.98 | 0.82 | |
| 2.13 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Hadi, M.; Yoon, S. Polymer Crosslinked Activated Carbon Pellets for Dye Adsorption. Materials 2026, 19, 155. https://doi.org/10.3390/ma19010155
Hadi M, Yoon S. Polymer Crosslinked Activated Carbon Pellets for Dye Adsorption. Materials. 2026; 19(1):155. https://doi.org/10.3390/ma19010155
Chicago/Turabian StyleHadi, Muhammad, and Sungho Yoon. 2026. "Polymer Crosslinked Activated Carbon Pellets for Dye Adsorption" Materials 19, no. 1: 155. https://doi.org/10.3390/ma19010155
APA StyleHadi, M., & Yoon, S. (2026). Polymer Crosslinked Activated Carbon Pellets for Dye Adsorption. Materials, 19(1), 155. https://doi.org/10.3390/ma19010155






