Experimental Research on the Ecological Recovery of Metals from Used Ni-MH Batteries
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
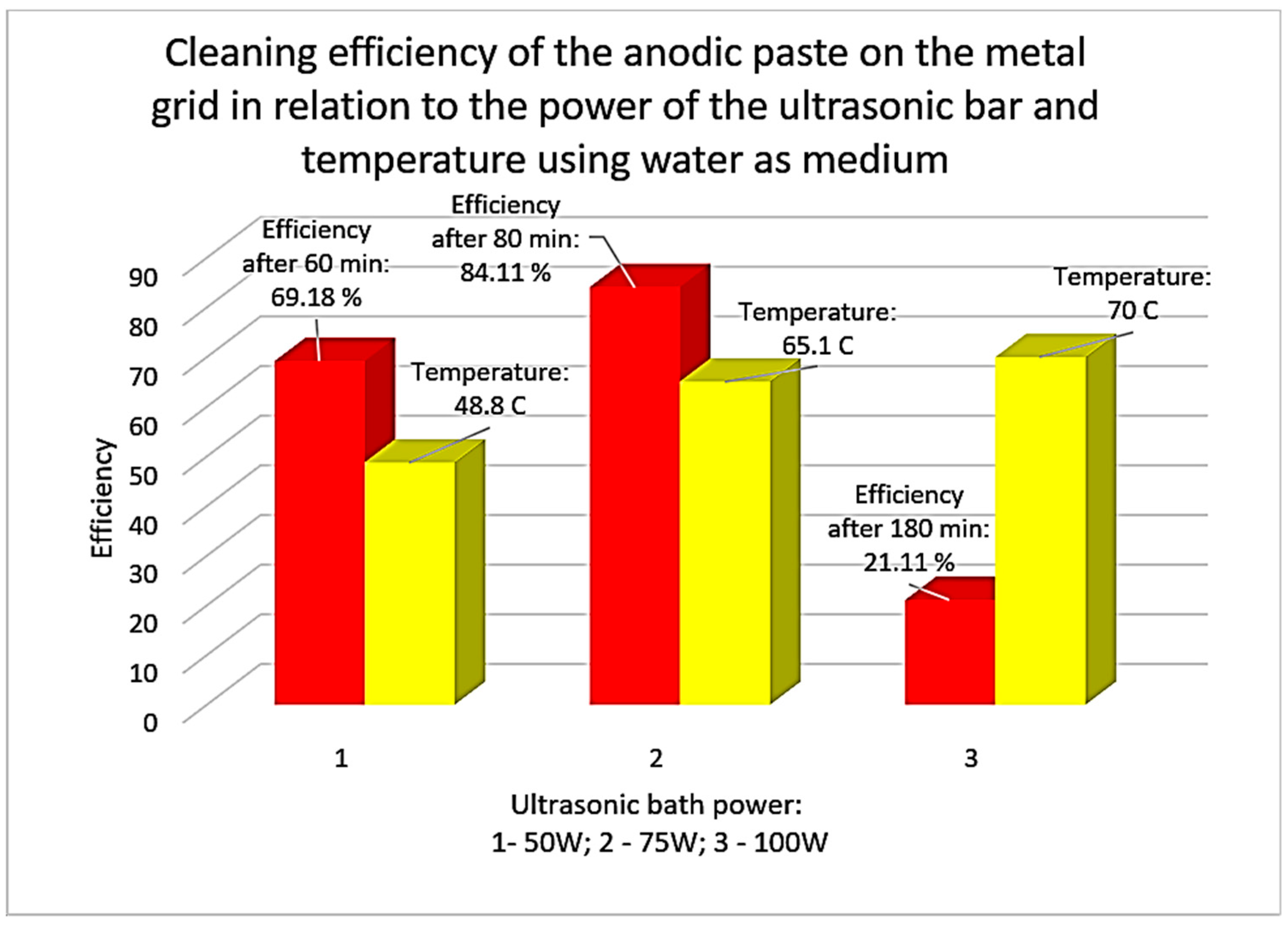
2.2. Laboratory Equipment Used and Working Method
- η—cleaning efficiency;
- mi—initial weight of anode (cathode) piece, g;
- mf—final weight of anode (cathode) piece, g.
3. Results
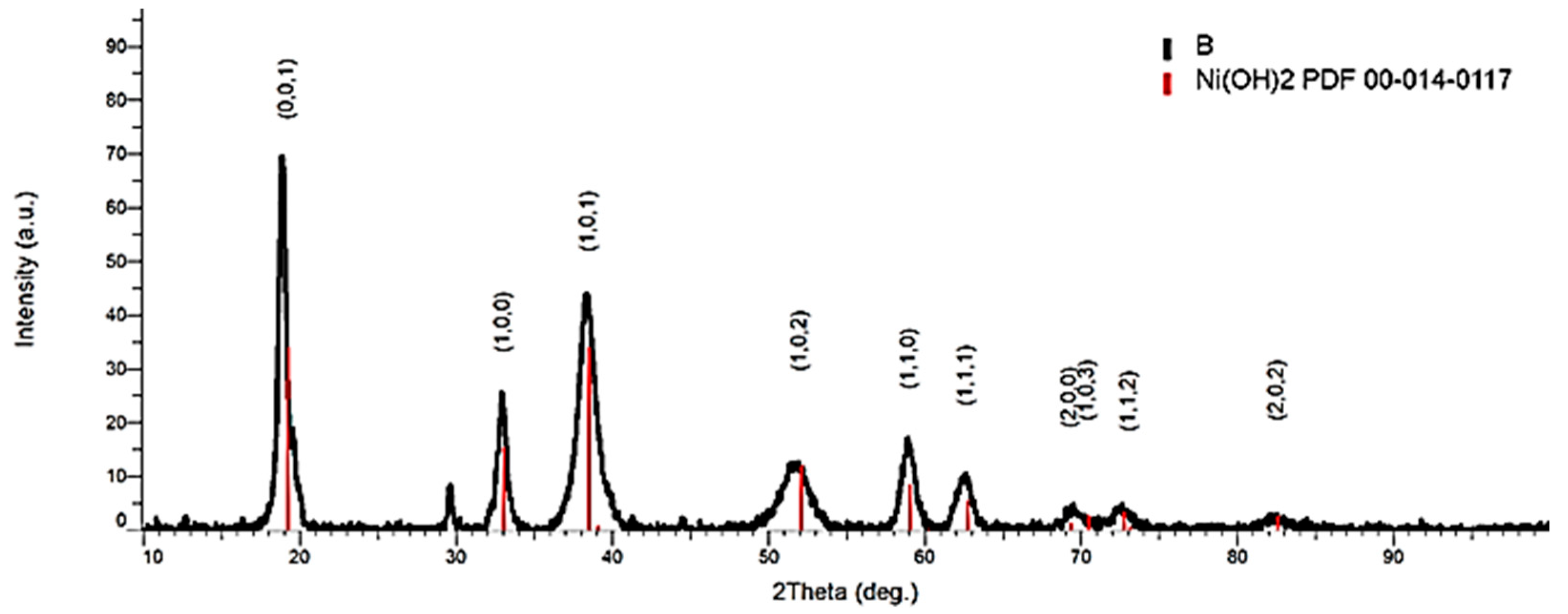
3.1. Separation/Recovery of Cathode Paste (Ni(OH)2) from the Support Mesh (Made of Ni)
3.2. Characterization of the Cathode Support Mesh After Ultrasonication
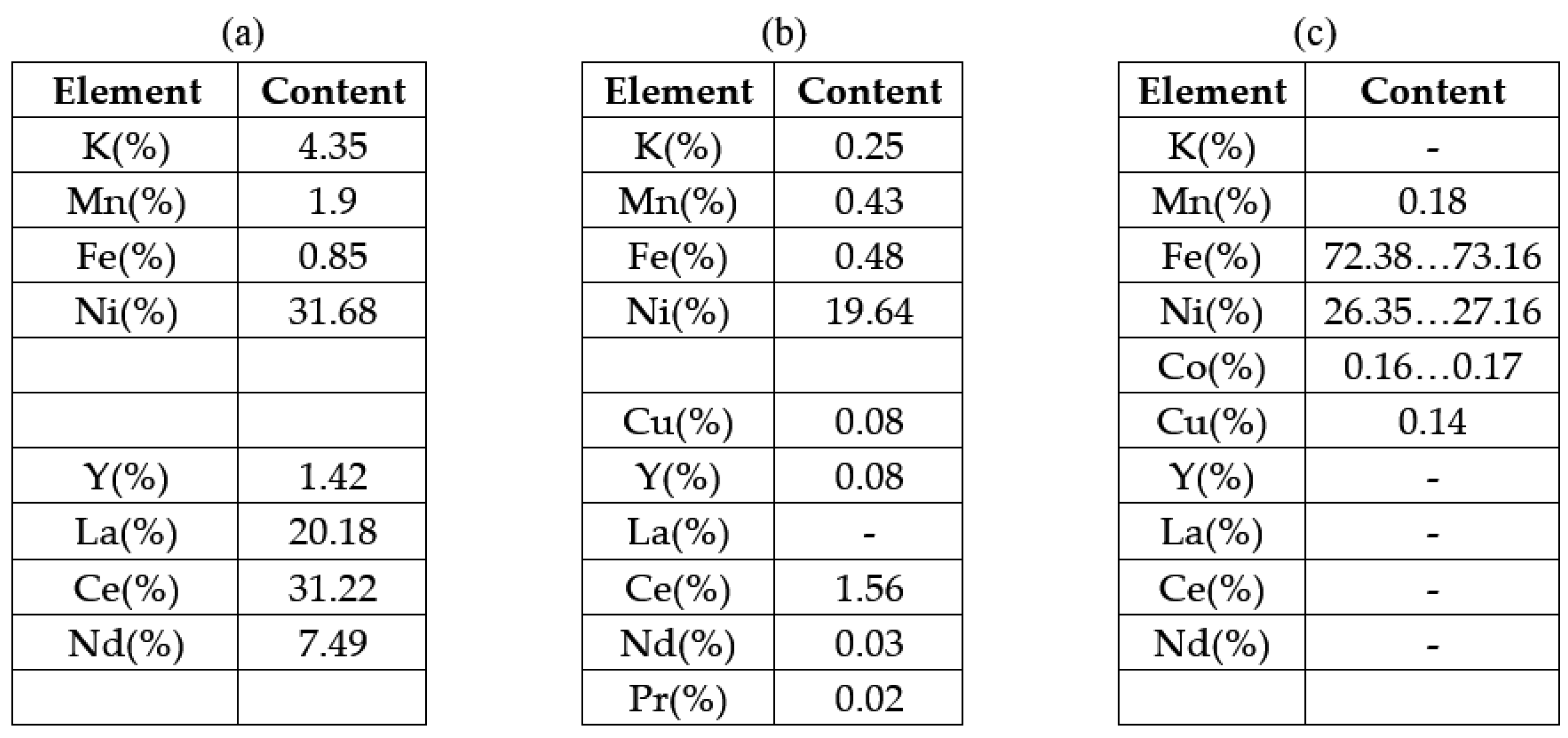
3.3. Characterization of Recovered Cathode Paste After Ultrasonication
- If Ni is to be recovered and melted in an electric arc furnace, the cathode support mesh made of Ni, (Tmelting = 1455 °C) together with the paste (Ni(OH)2), and Ni(OH)2 would decompose at 300–400 °C, releasing oxygen and transforming into NiO (temperature melting of NiO, Tmelting = 1995 °C) which would make the nickel impure.
- Being separated from the cathode mesh, Ni(OH)2 could be used as a precursor for the NiO product: Ni(OH)2 heated at 400 °C for 1 h in air leads to its decomposition into NiO and water.
3.4. Separation/Recovery of Rare-Earth-Containing Anode Paste from the Support Grid (Made of Ni Alloy)
3.5. Ultrasonication in Citric Acid Medium of an Anode Grid Section
4. Conclusions
- Finding a solution for mechanized dismantling and sorting of used Ni-MH batteries into components;
- Optimizing the process of recovering useful metals from used Ni-MH batteries by ultrasonication in a citric acid environment, because the process is cheap and environmentally friendly;
- Advantageous utilization of expensive and scarce non-ferrous metals and rare earths contained in used Ni-MH batteries.
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Alluraiah, C.N.; Nandogopal, V.; Veeramanikandan, P.; Godfrey, D.; Meena, S.; Bridha, G. Comparison of SoC in Ni-MH and Lithium-Ion Battery for E Vehicle. Int. J. Electr. Electron. Res. (IJEER) 2024, 12, 1258–1263. [Google Scholar] [CrossRef]
- Ourici, A. Battery Technologies Comparison for Electric Vehicles. Indian J. Sci. Technol. 2023, 16, 1461–1468. [Google Scholar] [CrossRef]
- Mars, N.; Krouz, F.; Louar, F.; Sbita, L. Comparison Study of Different Dynamic Battery Models. In Proceedings of the 2017 International Conference on Green Energy Conversion Systems (GECS), Hammamet, Tunisia, 23–25 March 2017; IEEE: New York, NY, USA, 2017; pp. 1–6. [Google Scholar] [CrossRef]
- Georgi-Maschler, T.; Friedrich, B.; Weyhe, R.; Heegn, H.; Rutz, M. Development of a Recycling Process for Li-Ion Batteries. J. Power Sources 2012, 207, 173–182. [Google Scholar] [CrossRef]
- Hu, J.; Zhang, J.; Li, H.; Chen, Y.; Wang, C. A Promising Approach for the Recovery of High Value-Added Metals from Spent Lithium-Ion Batteries. J. Power Sources 2017, 351, 192–199. [Google Scholar] [CrossRef]
- Müller, T.; Friedrich, B. Development of a Recycling Process for Nickel-Metal Hydride Batteries. J. Power Sources 2006, 158, 1498–1509. [Google Scholar] [CrossRef]
- Innocenzi, V.; Veglio, F. Separation of Manganese, Zinc and Nickel from Leaching Solution of Nickel-Metal Hydride Spent Batteries by Solvent Extraction. Hydrometallurgy 2012, 129–130, 50–58. [Google Scholar] [CrossRef]
- Innocenzi, V.; Ippolito, N.A.; De Michelis, I.; Prisciandaro, M.; Medici, F.; Veglio, F. A Review of the Processes and Lab-Scale Techniques for the Treatment of Spent Rechargeable NiMH Batteries. J. Power Sources 2017, 362, 202–218. [Google Scholar] [CrossRef]
- Rueda, H.; Arenas, M.; Delvasto, P. Production of a Nickel-Based Catalyst for Urea Electrooxidation Using Spent Batteries as Raw Material: Electrochemical Synthesis and Implications from a Circular Economy Standpoint. Sustain. Mater. Technol. 2021, 29, e00296. [Google Scholar] [CrossRef]
- Nan, J.; Hou, X.; Yang, M.; Han, D.; Li, W. Effect of Rare-Earth Elements on the Temperature Performances of Power Nickel-Metal Hydride Batteries for Electric Tools. Electrochem. Soc. J. Electrochem. Soc. 2006, 153, A1159. [Google Scholar] [CrossRef]
- Qingxue, Z.; Joubert, J.-M.; Latroche, M.; Jun, D.; Percheron-Guégan, A. Influence of the rare earth composition on the properties of Ni–MH electrodes. J. Alloys Compd. 2003, 360, 290–293. [Google Scholar] [CrossRef]
- Panasonic Energy Co. Technical Downloads. Available online: https://energy.panasonic.com/na/business/downloads/psds (accessed on 14 October 2025).
- Innocenzi, V.; Veglio, F. Recovery of Rare Earths and Base Metals from Spent Nickel-Metal Hydride Batteries by Sequential Sulphuric Acid Leaching and Selective Precipitations. J. Power Sources 2012, 211, 184–191. [Google Scholar] [CrossRef]
- Constantine, J.; Lie, J.; Liu, J.C. Recovery of Rare Earth Elements from Spent NiMH Batteries Using Subcritical Water Extraction with Citric Acid. J. Environ. Chem. Eng. 2022, 10, 108000. [Google Scholar] [CrossRef]
- Lie, J.; Liu, J.C. Selective Recovery of Rare Earth Elements (REEs) from Spent NiMH Batteries by Two-Stage Acid Leaching. J. Environ. Chem. Eng. 2021, 9, 106084. [Google Scholar] [CrossRef]
- Liu, H.; Zhang, S.; Pan, D.; Tian, J.; Yang, M.; Wu, M. Rare Earth Elements: Recycling from Waste Phosphor by Dual Hydrochloric Acid Dissolution. J. Hazard. Mater. 2014, 272, 96–101. [Google Scholar] [CrossRef] [PubMed]
- Badanoiu, G.; Buzatu, T.; Ghica, V.G.; Buzatu, M.; Iacob, G.; Petrescu, M.I. Study of PbSO4 Solubilisation in NaOH Solution for Treatment of Oxide–Sulphate Pastes Obtained from Dismembered Lead–Acid Batteries. UPB Sci. Bull. Ser. B Chem. Mater. Sci. 2014, 76, 209–214. [Google Scholar]
- Ghica, V.G.; Toma, C.M.; Buzatu, M.; Petrescu, M.I.; Iacob, G.; Antoniac, I.V.; Vasile, E.; Veglio, F. Recovery of Active Cathode Material Containing Co and Li from Waste Li-Ion Batteries. UPB Sci. Bull. Ser. B Chem. Mater. Sci. 2017, 79, 75–86. [Google Scholar]
- Vasile, A.; Ghica, V.G.; Iacob, G.; Petrescu, M.I.; Mihailov, E.; Patroi, D. Recycling Ni-MH Batteries Used in Medical Devices. Part I—Investigations on the Contents of Ni-MH Batteries. UPB Sci. Bull. Ser. B Chem. Mater. Sci. 2024, 86, 223–234. [Google Scholar]
- Li, T.; Okada, T.; Ichimura, M. Drop-Dry Deposition of Ni(OH)2 Precursor for Fabrication of NiO Thin Films. Materials 2022, 15, 4513. [Google Scholar] [CrossRef]
- Li, L.; Xu, S.; Ju, Z.; Wu, F. Recovery of Ni, Co and Rare Earths from Spent Ni–Metal Hydride Batteries and Preparation of Spherical Ni(OH)2. Hydrometallurgy 2009, 100, 41–46. [Google Scholar] [CrossRef]
- Díaz-López, J.C.; Angarita, J.; Vargas-Angarita, C.Y.; Blanco, S.; Delvasto, P. Electrolytic Recovery of Nickel and Cobalt as Multi-Elemental Coatings: An Option for the Recycling of Spent Ni-MH Batteries. J. Phys. Conf. Ser. 2018, 1119, 012003. [Google Scholar] [CrossRef]
- Assefi, M.; Maroufi, S.; Yamauchi, Y.; Sahajwalla, V. Pyrometallurgical Recycling of Li-Ion, Ni–Cd and Ni–MH Batteries: A Minireview. Curr. Opin. Green Sustain. Chem. 2020, 24, 26–31. [Google Scholar] [CrossRef]
- Pham, H.D.; Krishnan, S.G.; Wang, T.; Fernando, J.F.S.; Padwal, C.; Golberg, D.V.; Dubal, D.P. Upcycling of Nickel Oxide from Spent Ni-MH Batteries as Ultra-High Capacity and Stable Li-Based Energy Storage Devices. Sustain. Mater. Technol. 2023, 36, e00602. [Google Scholar] [CrossRef]
- Marins, A.A.L.; Boasquevisque, L.M.; Muri, E.J.B.; Freitas, M.B.J.G. Environmentally Friendly Recycling of Spent Ni–MH Battery Anodes and Electrochemical Characterization of Nickel and Rare Earth Oxides Obtained by Sol–Gel Synthesis. Mater. Chem. Phys. 2022, 280, 125821. [Google Scholar] [CrossRef]














| Lead–Acid | Ni/MH | Li-Ion | |
|---|---|---|---|
| Voltage | 2.1 V | 1.3 V | 3.8 V Volume energy |
| Weight energy | 30–50 Wh/kg | 70–80 Wh/kg (2001) 100–500 Wh/kg (2023) | 120 Wh/kg (2001) 300 Wh/kg (2001) |
| Volume energy | 80–90 Wh/dm3 (2001) 400 Wh/dm3 (2023) | 150–200 Wh/dm3 (2001) 300 Wh/dm3 (2023) | 150 Wh/dm3 (2001) 700 Wh/dm3 (2023) |
| Power | 180 W/kg (2001) <1000 W/kg (2001) | 200–300 Wh/kg (2021) 100–500 Wh/kg (2023) | 500 Wh/kg (2021) 500–700 Wh/kg (2023) |
| Cycle life | <350 | 1000 (2001) 2000 (2023) | 1500 (2001) 3000 (2023) |
| Cost | 50–150 $/KWh | 330 $/KWh (2001) 139 $/KWh (2023) | 800 $/KWh (2001) 75–259 $/KWh (2023) |
| Common Chemical Name | CAS Number | Concentration/ Percentage Range |
|---|---|---|
| Nickel Hydroxide Cobalt Hydroxide | 12054-48-7 21041-93-0 | 15–25% (15–30%) * 1–5% |
| Hydrogen-absorbing alloy | 7440-02-0(Ni) 7440-48-4(Co) 7439-96-5(Mn) 7429-90-5(Al) | 20–35% (20–40%) * |
| Nickel | 7440-02-0(Ni) | 3–10% (3–10%) * |
| Iron | 7439-89-6(Fe) | 10–25% (15–40%) * |
| Potassium Hydroxide Sodium Hydroxide Lithium Hydroxide | 1310-58-3 1310-73-2 1310-65-2 | 0–15% (0–15%) * |
| 2θ | D (Å) | I | h | k | l |
|---|---|---|---|---|---|
| 19.258 | 4.605 | 100 | 0 | 0 | 1 |
| 33.064 | 2.707 | 45 | 1 | 0 | 0 |
| 38.541 | 2.334 | 100 | 1 | 0 | 1 |
| 39.098 | 2.302 | 2 | 0 | 0 | 2 |
| 52.100 | 1.754 | 35 | 1 | 0 | 2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ghica, V.G.; Miculescu, F.; Vasile, A.; Saftere, N.D.; Markopoulos, A.P.; Karabulut, Ș.; Petrescu, M.I.; Tanasă, E.; Icleanu, A. Experimental Research on the Ecological Recovery of Metals from Used Ni-MH Batteries. Materials 2025, 18, 5549. https://doi.org/10.3390/ma18245549
Ghica VG, Miculescu F, Vasile A, Saftere ND, Markopoulos AP, Karabulut Ș, Petrescu MI, Tanasă E, Icleanu A. Experimental Research on the Ecological Recovery of Metals from Used Ni-MH Batteries. Materials. 2025; 18(24):5549. https://doi.org/10.3390/ma18245549
Chicago/Turabian StyleGhica, Valeriu Gabriel, Florin Miculescu, Ana Vasile, Narcis Daniel Saftere, Angelos P. Markopoulos, Șener Karabulut, Mircea Ionuț Petrescu, Eugenia Tanasă, and Anca Icleanu. 2025. "Experimental Research on the Ecological Recovery of Metals from Used Ni-MH Batteries" Materials 18, no. 24: 5549. https://doi.org/10.3390/ma18245549
APA StyleGhica, V. G., Miculescu, F., Vasile, A., Saftere, N. D., Markopoulos, A. P., Karabulut, Ș., Petrescu, M. I., Tanasă, E., & Icleanu, A. (2025). Experimental Research on the Ecological Recovery of Metals from Used Ni-MH Batteries. Materials, 18(24), 5549. https://doi.org/10.3390/ma18245549








