Abstract
Benzimidazole-based boranils were designed and synthesized in order to assess the influence of halogen substituents on their optoelectronic properties. All compounds are photoluminescent in solution and solid state. Compared to the free ligands, the new boranils emit at a lower wavelength, by elimination of the excited-state intramolecular proton transfer observed with the ligands. In the solid state, some of the boranils exhibit a deep blue emission, presenting Commission Internationale de l’Éclairage (CIE) coordinates with an x-component of less than 0.16 and a y-component smaller than 0.04, highly desired values for the development of blue emitting materials.
1. Introduction
Benzimidazole and other azole derivatives raise much interest due to their luminescence properties, and as promising candidates for the development of sensors and solar cells [1,2,3,4,5]. They are receiving increasing attention due to their non-linear optical properties [6,7] and excellent thermal stability [8,9]. Widely reported in the literature, these compounds are also of interest because of their intense emission via excited-state intramolecular proton transfer (ESIPT) [10,11,12]. Due to their optical properties, fully conjugated core, and possibility of functionalization of the aromatic ring, benzimidazole derivatives are privileged ligands to develop materials with bright blue emissions [7,13]. Another advantage of these compounds is that they have an excellent coordination ability with a wide range of transition metals, and may present interesting selectivity and sensitivity toward different metal cations [14].
Complexation with boron is widely used in the synthesis of luminescent dyes. Boron is a nontoxic alternative to transition metals, and boron complexes have been studied for their excellent photoluminescent properties [15,16,17,18,19]. The latter are cheaper than certain transition metals, and because of the strong covalent bonds with the ligand, they may allow the synthesis of more stable luminescent materials [20,21,22]. For example, difluoroboron complexation enabled thermally activated delayed fluorescence, which find interesting applications in organic light-emitting diodes [23]. Benzimidazoles bearing an ortho-hydroxy aromatic substituent are interesting fluorescent molecules (Figure 1) because of their large Stokes shifts due to their ESIPT [24,25]. This mechanism can be inhibited by binding a boron atom to the hydroxy group and the nitrogen atom of the azole unit: longer emission wavelengths are lost, resulting in a hypsochromic shift [26]. Derivatives of 2-(2-hydroxyphenyl)benzothiazole 1 and 2-(2-hydroxyphenyl)benzoxazole 2 are examples of compounds explored for blue emission [27,28,29]. Recently, boron complexes 3 incorporating imidazo[1,5-a]pyridine as a N,O-type ligand were reported [30], as well as tetraaryl substituted imidazole boron difluoride complexes 4 [31]. Examples of reported boron complexes based on bidentate 2-(2-pyridyl)imidazole 5, and N-alkylated 2-(2-hydroxyphenyl)benzimidazole 6, showed emissions in both solution and solid state [32,33].
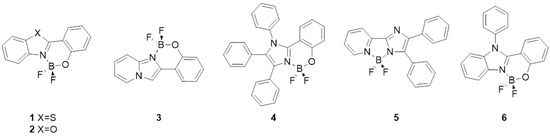
Figure 1.
Azole-based organoboron compounds suitable for optoelectronic applications.
Although imidazole-phenol complexes have been studied for application in electronic materials [34], the nitrogen atom not involved in the complexation usually bears a substituent, and if 2-(1H-benzo[d]imidazol-2-yl)phenol has been studied for boron complexation [35], to the best of our knowledge no studies of their photophysical properties have been performed.
The introduction of halogen atoms on the backbone of fluorophores is a strategy used to promote their phosphorescence. Indeed, the heavy atom effect can promote intersystem crossing between the excited singlet and triplet states. If a halogen bond is formed in the solid state, the effect can be even more pronounced, allowing the observation of phosphorescence in purely organic solids at room temperature [36]. This may find applications in the development of organic light emitting devices.
Here, we wish to report the synthesis and characterization of boron complexes 8 based on halogenated 2-(1H-benzo[d]imidazol-2-yl)phenol 7 (Figure 2), and the study of their optoelectronic properties. In the design of the fluorophores, the nitrogen not involved in the complexation was kept unsubstituted to minimize the conjugation of the backbone, in order to produce a blue emission. Halogen atoms have been introduced on one aromatic ring, in an attempt to promote intersystem crossing and therefore phosphorescent dyes.
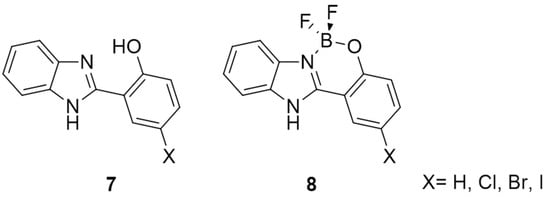
Figure 2.
Halogenated boron complexes 8 based on 2-(1H-benzo[d]imidazol-2-yl)phenol 7 studied in this work.
2. Materials and Methods
2.1. Materials and Methods
All reagents were purchased from Sigma-Aldrich (St. Louis, MO, USA) and used without any further purification. The final compounds were purified by crystallization. 1H, 19F and 13C NMR spectra were recorded on Bruker 300 or 500 [300.13 MHz (1H), 282.41 (19F), 75.47 MHz (13C) or 500.13 MHz (1H)]. Unequivocal 13C assignments were made on the basis of 2D Heteronuclear Single Quantum Coherence (HSQC) (1H/13C) and Heteronuclear Multiple Bond Correlation (HMBC) experiments. DMSO-d6 was used as the solvent and tetramethylsilane (TMS) as the internal standard. Chemical shifts δ are reported in parts per million (ppm) relative to TMS (δ = 0), and the values of coupling constants (J) are given in Hertz (Hz). High-resolution mass spectra (HRMS-ESI+) were recorded on an LTQ Orbitrap™ XL hybrid mass spectrometer (Thermo Fischer Scientific, Bremen, Germany) controlled by LTQ Tune Plus 2.5.5 and Xcalibur 2.1.0. The capillary voltage of the electrospray ionization source (ESI) was set to 3.1 kV. Melting points were determined on a BUCHI Melting point apparatus (BÜCHI Labortechnik AG, Flawil, Switzerland) and are uncorrected. The ultraviolet-visible (UV-Vis) spectra in dimethyl sulfoxide solutions were obtained on a Shimadzu UV-2501 PC spectrophotometer (1 cm path length quartz cell, Shimatzu, Nakagyō-ku, Japan) and the UV-Vis absorption spectra of the solids were measured at room temperature on a JASCO V-560 instrument (JASCO Inc., Easton, MD, USA). The excitation and emission spectra, also in dimethyl sulfoxide solutions, were recorded on a Jobin Yvon FluoroMax-3 spectrofluorometer (Horiba, Lier, Belgium) and a JASCO spectrofluorometer (JASCO Inc., Easton, MD, USA). Fluorescence quantum yields φF were determined using fluorescein in 0.1 M NaOH water solution as a fluorescence standard. The absolute emission quantum yields in the solid state were measured at room temperature using a system (Quantaurus-QY Plus C13534, Hamamatsu, Shizuoka, Japan) with a 150 W xenon lamp coupled to a monochromator for wavelength discrimination, an integrating sphere as the sample chamber, and a multichannel analyzer for signal detection. The method is accurate to within 10%.
2.2. Synthesis
2.2.1. General Procedure for the Synthesis of Ligands 7a–d
The general procedure is described here for 7a: salicylaldehyde (1.0 equiv., 1.0 mL, 9.6 mmol) and o-phenylenediamine (1.5 equiv., 1.6 g, 14.4 mmol) were mixed in methanol (2 mL). After everything dissolved, a red solid immediately formed. Acetic acid (20 mL) was added and the reaction mixture was stirred at room temperature for 4 h. The reaction was quenched by adding water (60 mL), the solid was collected by filtration and washed with methanol (3 × 20 mL). The solid obtained was purified by flash column chromatography over silica gel, eluent hexane/ethyl acetate (2:1 v/v), to afford the product 7a as off-white crystals after crystallization in chloroform.
2-(1H-benzo[d]imidazol-2-yl)phenol 7a [37]
The compound 7a was obtained as colorless microcrystals (1.25 g, 5.94 mmol, 62%). M.P.: 242–243 °C; 1H NMR (300 MHz, DMSO-d6) δ 13.17 (br s, 2H, OH and NH), 8.05 (dd, 3JH-H 7.8, 4JH-H 1.4 Hz, 1H, Harom), 7.66 (br s, 2H, Harom), 7.38 (ddd, 3JH-H 8.6, 3JH-H 7.1, 4JH-H 1.6 Hz, 1H, Harom), 7.30–7.27 (m, 2H, Harom), 7.05–6.99 (m, 2H, Harom). HRMS-ESI+ m/z for [C13H10N2O + H]+ calcd 211.0866, found 221.0868.
2-(1H-benzo[d]imidazol-2-yl)-4-chlorophenol 7b [37]
The compound 7b was obtained as colorless microcrystals (90 mg, 0.36 mmol, 60%). M.P.: 306–307 °C; 1H NMR (500 MHz, DMSO-d6) δ 13.27 (br s, 2H, OH and NH), 8.17 (d, 4JH-H 2.6 Hz, 1H, Harom), 7.68 (br s, 2H, Harom), 7.42 (dd, 3JH-H 8.8, 4JH-H 2.6 Hz, 1H, Harom), 7.31 (br s, 2H, Harom), 7.08 (d, 3JH-H 8.8, 1H, Harom). HRMS-ESI+ m/z for [C13H9ClN2O + H]+ calcd 245.0476, found 245.0479.
2-(1H-benzo[d]imidazol-2-yl)-4-bromophenol 7c
The compound 7c was obtained as a brown solid (90 mg, 0.32 mmol, 63%). M.P.: 311–312 °C; 1H NMR (300 MHz, DMSO-d6) δ 13.30 (br s, 2H, OH and NH), 8.29 (d, 4JH-H 2.5 Hz, 1H, Harom), 7.68 (dd, 3JH-H 5.9 Hz, 4JH-H 3.2 Hz, 2H, Harom), 7.52 (dd, 3JH-H 8.8, 4JH-H 2.5 Hz, 1H, Harom), 7.30 (dd, 3JH-H 5.9 Hz, 4JH-H 3.2 Hz, 2H, Harom), 7.02 (d, 3JH-H 8.8, 1H, Harom). 13C NMR (75 MHz, DMSO-d6) δ 157.2 (1C, C-O), 150.3 (1C, C = N), 134.0 (1C, CHarom), 128.5 (1C, CHarom), 123.2 (4C, CHarom), 119.6 (1C, CHarom), 114.7 (1C, C-Br), 112.4 (2C, Carom) 110.2 (1C, Carom). HRMS-ESI+ m/z for [C13H9BrN2O + H]+ calcd 288.9971, found 288.9975.
2-(1H-benzo[d]imidazol-2-yl)-4-iodophenol 7d
The compound 7d was obtained as a brown solid (95 mg, 0.2 mmol, 68%). M.P.: 254–255 °C; 1H NMR (300 MHz, DMSO-d6) δ 13.28 (br s, 2H, OH and NH), 8.42 (d, 4JH-H 2.2 Hz, 1H, Harom), 7.67 (br s, 2H, Harom), 7.65 (dd, 3JH-H 8.7, 4JH-H 2.2 Hz, 1H, Harom), 7.30 (dd, 3JH-H 6.1 Hz, 4JH-H 2.4 Hz, 2H, Harom), 6.89 (d, 3JH-H 8.7 Hz, 1H, Harom). 13C NMR (75 MHz, DMSO -d6) δ 157.7 (1C, C-O), 150.2 (1C, C = N), 139.7 (1C, CHarom), 134.2 (1C, CHarom), 123.1 (4C, CHarom), 119.9 (1C, CHarom), 115.3 (1C, C-I), 110.4 (2C, Carom), 80.9 (1C, Carom). HRMS-ESI+ m/z for [C13H9IN2O + H]+ calcd 336.9832, found 336.9833.
2.2.2. General Procedure for the Synthesis of Complexes 8a–d
The typical synthetic procedure of benzimidazole-based N,O-chelated boron complexes is described as following for 8a: BF3·OEt2 (3 equiv., 0.06 mL, 0.6 mmol) was added dropwise at room temperature to a stirred mixture of ligand 7a (0.2 mmol) in anhydrous tetrahydrofuran (2 mL), and the reaction mixture was stirred overnight. The solvent was removed under reduced pressure and the solid obtained was washed with anhydrous diethyl ether several times to obtain the complex.
6,6-difluoro-6,12-dihydrobenzo[e]benzo[4,5]imidazo[1,2-c][1,3,2]oxazaborinin-7-ium-6-uide 8a [35]
The compound 8a was obtained as a white solid (62 mg, 0.2 mmol, 100%). M.P.: 348–350 °C. 1H NMR (300 MHz, DMSO-d6) δ 14.65 (b s, 1H, NH), 8.05 (dd, 3JH-H 8.1 Hz, 4JH-H 1.5 Hz, 1H, Harom), 7.85 (dd, 3JH-H 6.2 Hz, 4JH-H 2.4 Hz, 2H, Harom), 7.59 (ddd, 3JH-H 7.8 Hz, 3JH-H 6.4 Hz, 4JH-H 1.3 Hz, 1H, Harom), 7.55 (dd, 3JH-H 6.2 Hz, 4JH-H 2.4 Hz, 2H, Harom), 7.21 (dd, 3JH-H 9.0 Hz, 4JH-H 0.7 Hz, 1H, Harom), 7.15 (ddd, 3JH-H 7.5 Hz, 3JH-H 6.2 Hz, 4JH-H 0.9 Hz, 1H, Harom). 13C NMR (75 MHz, DMSO d-6) δ 157.1 (1C, C-O), 146.8 (1C, C = N), 135.2 (1C, CHarom), 131.2 (2C, Carom), 129.3 (1C, CHarom), 125.9 (2C, CHarom), 120.0 (1C, CHarom), 117.3 (1C, CHarom), 114.1 (2C, CHarom), 109.2 (1C, Carom).19F NMR (282 MHz, DMSO-d6) δ -132.2 (dd, J 27.7 Hz, J 10.7 Hz, 2F). HRMS-ESI+ m/z for [C13H9BF2N2O + H] + calcd 259.0854, found 259.0853.
2-Chloro-6,6-difluoro-6,12-dihydrobenzo[e]benzo[4,5]imidazo[1,2-c][1,3,2]oxazaborinin-7-ium-6-uide 8b
The compound 8b was obtained as a white solid (29 mg, 0.099 mmol, 46%), M.P.: 308–309 °C. 1H NMR (300 MHz, DMSO-d6) δ 14.71 (s, 1H, NH), 8.21 (d, 4JH-H 2.6 Hz, 1H, Harom), 7.85–7.78 (m, 2H, Harom), 7.61 (dd, 3JH-H 8.9 Hz, 4JH-H 2.6 Hz, 1H, Harom), 7.49–7.57 (m, 2H, Harom), 7.16 (d, 3JH-H 8.9 Hz, 1H, Harom). 13C NMR (75 MHz, DMSO d-6) δ 155.3 (1C, C-O), 146.1 (1C, C = N), 134.5 (1C, CHarom), 132.2 (2C, Carom), 125.8 (1C, CHarom), 125.2 (1C, CHarom), 124.9 (1C, CHarom), 123.3 (1C, C-Cl), 121.2 (1C, CHarom), 115.1 (1C, CHarom), 113.4 (1C, CHarom), 110.0 (1C, Carom). 19F NMR (282 MHz, DMSO-d6) δ -132.1 (dd, J 25.4 Hz, J 8.7 Hz, 2F). HRMS-ESI+ m/z for [C13H8B37ClF2N2O + H] + calcd 293,0465, found 293,0466.
2-Bromo-6,6-difluoro-6,12-dihydrobenzo[e]benzo[4,5]imidazo[1,2-c][1,3,2]oxazaborinin-7-ium-6-uide 8c
The compound 8c was obtained as a white solid (34 mg, 0.10 mmol, 81%). M.P.: 395–396 °C. 1H NMR (300 MHz, DMSO-d6) δ 14.75 (s, 1H, NH), 8.34 (d, 4JH-H 2.5 Hz, 1H, Harom), 7.78–784 (m, 2H, Harom), 7.72 (dd, 3JH-H 8.9 Hz, 4JH-H 2.5 Hz, 1H, Harom), 7.49–7.58 (m, 2H, Harom), 7.10 (d, 3JH-H 8.9 Hz, 1H, Harom). 13C NMR (75 MHz, DMSO d-6) δ 155.6 (1C, C-O), 146.0 (1C, C = N), 137.2 (1C, CHarom), 132.3 (2C, Carom), 127.8 (1C, CHarom), 125.8 (1C, CHarom), 125.2 (1C, Charom), 123.3 (1C, C-Br), 121.6 (1C, CHarom), 115.1 (1C, CHarom), 113.4 (1C, CHarom), 110.7 (1C, Carom). 19F NMR (282 MHz, DMSO-d6) δ -132.1 (dd, J 25.4 Hz, J 8.7 Hz, 2F). MS-ESI+ m/z for [C13H8BBrFN2O] + 317.0 (79Br), 319.0 (81Br). HRMS-ESI- m/z for [C13H8BBrF2N2O-H]-calcd 334.9808, found 334.9827.
6,6-difluoro-2-iodo-6,12-dihydrobenzo[e]benzo[4,5]imidazo[1,2-c][1,3,2]oxazaborinin-7-ium-6-uide 8d
The compound 8d was obtained as a brown solid (0.200 g, 0.5 mmol, 79%). M.P.: 379–380 °C. 1H NMR (300 MHz, DMSO-d6) δ 14.68 (s, 1H, NH), 8.48 (d, 4JH-H 2.2 Hz, 1H, Harom), 7.86 (dd, 3JH-H 8.7 Hz, 4JH-H 2.2 Hz, 1H, Harom), 7.78–7.81 (m, 2H, Harom), 7.49–7.58 (m, 2H, Harom), 6.97 (d, 3JH-H 8.7, 1H, Harom). 13C NMR (75 MHz, DMSO-d6) δ 156.1 (1C, C-O), 142.8 (1C, C = N), 142.8 (1C, CHarom), 133.6 (1C, CHarom), 123.2 (1C, Carom), 132.0 (1C, Carom), 121.7 (1C, CHarom), 125.8 (1C, CHarom), 125.2 (1C, CHarom), 115.1 (1C, CHarom), 113.3 (1C, CHarom), 108.2 (1C, Carom), 81.6 (1C, C-I). 19F NMR (282 MHz, DMSO-d6) δ -132.0 (dd, J 25.4 Hz, J 8.7 Hz, 2F). HRMS-ESI+ m/z for [C13H8BF2IN2O + Na]+, calcd 406,9640, found 406,9639.
3. Results and Discussion
3.1. Synthesis
The synthetic procedure for the synthesis of fluoroborates complexes 8a–d is described in Scheme 1.
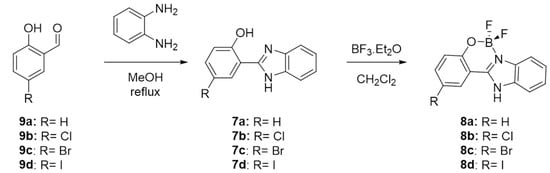
Scheme 1.
Synthetic procedure for the synthesis of fluoroborates complexes 8a–d.
Because benzimidazole rings are important building blocks for therapeutic drugs and optoelectronic materials, several strategies for their synthesis are reported in the literature [38]. The most common procedures call for the cyclocondensation of o-phenylenediamine derivatives with carboxylic acids under strongly acidic conditions, or with aldehydes under oxidative conditions. Although several oxidative and catalytic reagents have been employed in the synthesis of benzimidazoles from aldehydes [39], some of these methods have certain disadvantages, such as requiring large quantities of reagent, the high cost of the catalysts, prolonged reaction times, occurrence of side reactions, the harsh reaction conditions used, strong oxidizing nature of the reagents, or the use of toxic metal salts. For the synthesis of ligands 7a–d we have chosen the classical cyclocondensation of o-phenylenediamine with the corresponding salicylaldehydes 9a–d under oxidative conditions, using the atmospheric oxygen as the oxidant, and promoting the cyclization reaction by adding acetic acid. The reaction starts with the formation of a red solid (the intermediate Schiff base), which disappears after the addition of acetic acid. The reaction solution then became strongly photoluminescent, indicating the formation of the benzimidazole ring.
The synthesis of the fluoroborates complexes 8b–d followed the conditions described for 8a. Because this compound was obtained with a good yield, the conditions were maintained for the other complexes. Briefly, the ligands 7a–d were dissolved in THF and three equivalents of boron trifluoride diethyl etherate were added at room temperature. The complexes were obtained as off-white solids after evaporation of the reaction mixture under reduced pressure and after several wash cycles with diethyl ether. The instability of the complexes, which are sensitive to hydrolysis, made their isolation difficult, and their purification should be done as quickly as possible. Attempts to purify the complexes using silica gel column chromatography were unsuccessful, and a mixture of the complex and the ligand was systematically recovered. Additionally, the hydrolysis of the complexes was also observed during NMR characterization, if the spectrum was not recorded just after the preparation of the solution. All compounds were fully characterized (see Supplementary Materials).
3.2. Photophysical Properties
The absorption and emission spectra of benzimidazoles 7a–d were recorded in anhydrous DMSO solution and are shown in Figure 3. DMSO was selected to maximize the solubility of both the free ligands and the boron complexes. The solutions were freshly prepared and used immediately, to prevent the hydrolysis of the complexes. The photophysical properties of compounds 7a–d are summarized in Table 1.
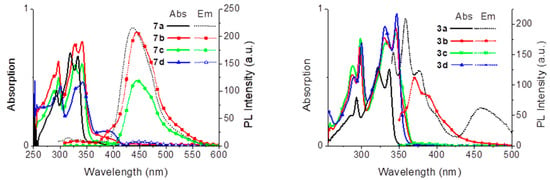
Figure 3.
Absorption (full lines) and emission spectra (dotted lines) of compounds 7a–d and compounds 8a–d, in DMSO (at 1 × 10−5 mol L−1).

Table 1.
Optical properties of compounds 7a–d and 8a–d.
The ligand 7a exhibits a maximum absorption peak at 333 nm, which is ascribed to the π → π * transition (Figure 3). For the halogenated derivatives 7b–d, the maximum absorption wavelength is slightly red shifted, by ca. 10 nm. This small shift can be related to the presence of electron withdrawing groups (–Cl, –Br and I) that decreases the Highest Occupied Molecular Orbital-Lowest Unoccupied Molecular Orbital (HOMO-LUMO) energy gap of substituted derivatives [40,41]. In these kind of compounds, ESIPT is strongly influenced by the nature and position of the substituents, which adjust the strength of the hydrogen bond [42,43,44].
All compounds are fluorescent in DMSO solution and show large Stokes’ shifts of ca. 125 nm, characteristic of fluorophores presenting ESIPT [43,45,46]. The maximum emission wavelength of 7b–d is also slightly red shifted (5–10 nm) moving down the halogen series (Figure 3). The quantum yields are, however, very different, and for 7a and 7b they are very good, 93% and 95%, respectively. The quenching observed for 7c (quantum yield 33%) and 7d (quantum yield 4%) results from the expected internal heavy atom effect [47], which is stronger with the iodine than with the bromine substituents. All ligands 7a–d exhibit a bright blue fluorescence under irradiation at 254 or 365 nm (Table 2).

Table 2.
Photographs of the ligands 7a–d and fluoroborates complexes 8a–d in solid state under UV lamp.
The absorption and emission spectra of boron complexes 8a–d in anhydrous DMSO are displayed in Figure 3. Their maximum absorption energy is also ascribed to the π → π * transition [48]. The emission and absorption spectra of the complexes are mirror images and display a small Stokes shift between 16 and 26 nm, indicating the absence of major internal conversions.
The ESIPT (Figure 4) witnesses the transfer of the proton from the hydroxy to the imidazole-N moiety through a pre-existing hydrogen bond, and is responsible for the large Stokes’ shift observed for the ligands. The complexation of boron supresses this phenomenon, as the hydrogen atom is no longer present and, as a consequence, the emission profile is dramatically altered. Here, the halogen atoms have a more significant effect on the emission properties: the quantum yields of 8b–d decrease significantly relatively to 8a. The small Stokes’ shifts and the spectral overlap may lead to the self-quenching of fluorescence for compounds 8a–d, and the halogen atoms may promote the intersystem crossing, leading to a quenching of the fluorescence instead of the expected promotion of phosphorescence.
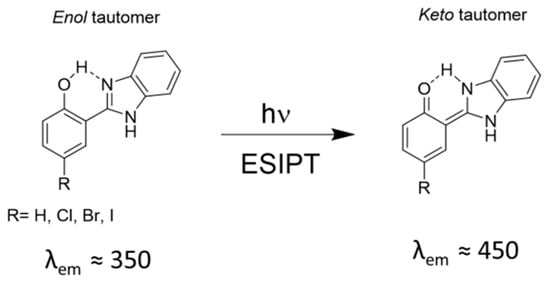
Figure 4.
ESIPT fluorescence of benzimidazol derivatives.
Figure 5 depicts the emission spectra of complexes and ligands in the solid state and in DMSO solution. The emissions of the complexes are mirror images of their absorption, with structured emission bands, which is not the case for the ligands. This has been observed before for similar fluorophores [32]. While the solid state and solution emission spectra of the ligands present the same profile, the complexes do not. The emission maxima of the latter are red shifted in the solid state relatively to solution. This indicates possible intermolecular interactions in the solid state, such as π···π stacking, which may be responsible for both the red shift and the quenching observed.
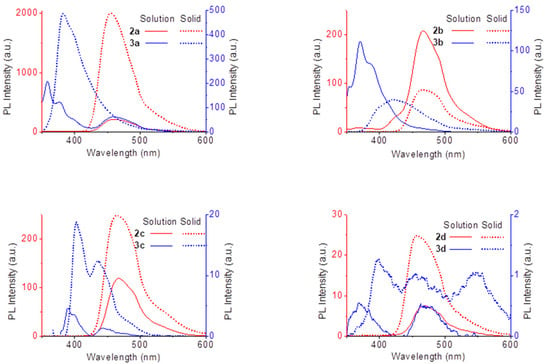
Figure 5.
Emission spectra of ligands (red) and complexes (blue), in DMSO solutions (1.0 × 10−5 M, full line) and solid state (dotted line).
All compounds exhibit a blue emission in solution and solid state (Figure 6 and Figure 7). In solution, 8b, with a moderate quantum yield of 26%, exhibits a deep blue emission with CIE coordinates of (0.16, 0.02) with a y-component smaller than 0.04, that is highly desirable and follows the specifications of the European Broadcast Union (EBU) television (0.15, 0.06) [49,50]. In the solid state, all complexes emit a blue color under a UV lamp (Table 2). Both 8a (0.16, 0.04) and 8b (0.16, 0.08) present a deep blue color in the solid state.
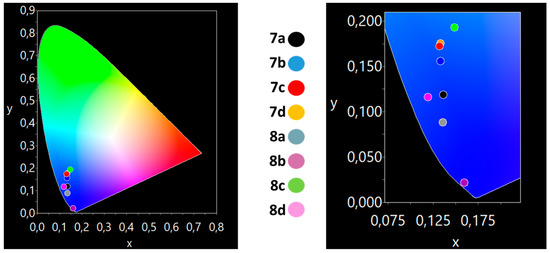
Figure 6.
The CIE coordinates for the compounds 7a–d and 8a–d in DMSO solution, showing the deep-blue color of 8b.
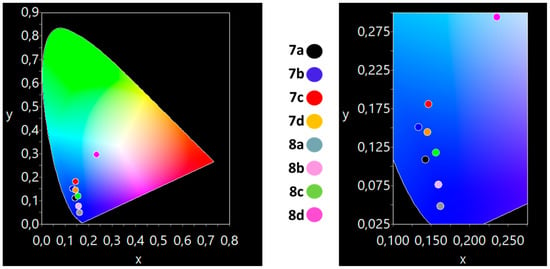
Figure 7.
The CIE coordinates for the compounds 7a–d and 8a–d in solid state, showing the deep-blue color of 8a–b.
4. Conclusions
Boron complexes based on halogenated 2-(1H-benzo[d]imidazol-2-yl)phenol were synthesized and characterized. All the halogenated benzimidazole complexes exhibit a blue emission in both DMSO solution and solid state, with moderate quantum yields, demonstrating that keeping the conjugation minimal is an adequate strategy to tune the emission color. On the other hand, the introduction of halogen substituents lowered their quantum yields, and did not promote their phosphorescence in the solid state, making these compounds less promising for electroluminescence. Unfortunately, the instability of the complexes, which are sensitive to hydrolysis, may prevent their applications as emissive materials. Nevertheless, some of them present a deep blue emission, making them interesting starting points for the development of emissive materials.
Supplementary Materials
The following are available online at https://www.mdpi.com/article/10.3390/ma14154298/s1: NMR spectra of all new compounds; absorption, emission and excitation spectra in solution; excitation and emission spectra in solid state.
Author Contributions
Conceptualization, S.G. and P.A.A.M.V.; investigation, P.A.A.M.V.; writing—original draft preparation, P.A.A.M.V.; writing—review and editing, S.G., J.R. and A.M.S.S.; supervision, S.G.; funding acquisition, J.R. and A.M.S.S. All authors have read and agreed to the published version of the manuscript.
Funding
Thanks are due to University of Aveiro, FCT/MEC, Centro 2020 and Portugal 2020, the COMPETE program, and the European Union (FEDER program) via the financial support to the LAQV-REQUIMTE (UIDB/50006/2020), to the CICECO-Aveiro Institute of Materials (UID/CTM/50011/2019, UIDB/50011/2020 & UIDP/50011/2020), financed by national funds through the FCT/MCTES, to the Portuguese NMR Network, and to the PAGE project “Protein aggregation across the lifespan” (CENTRO-01-0145-FEDER-000003). SG is supported by national funds (OE), through FCT, I.P., in the scope of the framework contract foreseen in the numbers 4, 5, and 6 of the article 23, of the Decree-Law 57/2016, of August 29, changed by Law 57/2017, of July 19. PV thanks the FCT for a doctoral grant (SFRH/BD/99809/2014).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.
Acknowledgments
Rute AS Ferreira (phantom-g, CICECO, Department of Physics, University of Aveiro) is acknowledged for the solid state quantum yield measurements.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
References
- Cansu-ergun, E.G. Chemical insight into benzimidazole containing donor-acceptor-donor type π-conjugated polymers: Benzimidazole as an acceptor. Polym. Rev. 2018, 58, 42–62. [Google Scholar] [CrossRef]
- Burganov, T.I.; Zhukova, N.A.; Mamedov, V.A.; Bannwarth, C.; Grimme, S.; Katsyuba, S.A. Benzimidazolylquinoxalines: Novel fluorophores with tuneable sensitivity to solvent effects. Phys. Chem. Chem. Phys. 2017, 19, 6095–6104. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, B.; Li, Y.; Song, P.; Ma, F.; Sun, M. Photoactive layer based on T-shaped benzimidazole dyes used for solar cell: From photoelectric properties to molecular design. Nat. Sci. Rep. 2017, 7, 45688. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reddy, S.S.; Cho, W.; Sree, V.G.; Jin, S. Multi-functional highly efficient bipolar 9,9-dimethyl-9,10-dihydroacridine/imidazole-based materials for solution-processed organic light-emitting diode applications. Dyes Pigment. 2016, 134, 315–324. [Google Scholar] [CrossRef]
- Bodedla, G.B.; Thomas, K.R.J.; Fan, M.; Ho, K. Benzimidazole-Branched Isomeric Dyes: Effect of Molecular Constitution on Photophysical, Electrochemical, and Photovoltaic Properties. J. Org. Chem. 2016, 81, 640–653. [Google Scholar] [CrossRef] [Green Version]
- Kulhánek, J.; Bures, F.; Pytela, O.; Mikysek, T.; Ludvík, J. Imidazole as a donor/acceptor unit in charge-transfer chromophores with extended π-linkers. Chem. Asian J. 2011, 6, 1604–1612. [Google Scholar] [CrossRef]
- Lai, M.; Chen, C.; Huang, W.; Lin, J.T.; Ke, T.; Chen, L.; Tsai, M.; Wu, C. Benzimidazole/Amine-based compounds capable of ambipolar transport for application in single-layer blue-emitting oleds and as hosts for phosphorescent emitters. Angew. Chem. Int. Ed. 2008, 47, 581–585. [Google Scholar] [CrossRef]
- Sim, B.R.; Kim, B.-G.; Lee, J.K.; Do, J.Y. Photovoltaic properties of benzimidazole-derived perylene imides as an n-type material. Thin Solid Films 2011, 519, 8091–8094. [Google Scholar] [CrossRef]
- Liu, Y.; Gao, Z.; Wang, Z.; Feng, C.; Shen, F.; Lu, P.; Ma, Y. Synthesis and characterization of an imidazole-containing pyrene π-system. Eur. J. Org. Chem. 2013, 32, 7267–7371. [Google Scholar] [CrossRef]
- Chen, L.; Yin, S.-Y.; Pan, M.; Wu, K.; Wang, H.-P.; Fan, Y.-N.; Su, C.-Y. A naked eye colorimetric sensor for alcohol vapor discrimination and amplified spontaneous emission (ASE) from a highly fluorescent excited-state intramolecular proton transfer (ESIPT) molecule. J. Mater. Chem. C 2016, 4, 6962–6966. [Google Scholar] [CrossRef]
- Nieto, C.I.; Cabildo, P.; García, M.Á.; Claramunt, R.M.; Alkorta, I.; Elguero, J. An experimental and theoretical NMR study of NH-benzimidazoles in solution and in the solid state: Proton transfer and tautomerism. Beilstein J. Org. Chem. 2014, 10, 1620–1629. [Google Scholar] [CrossRef]
- Chipem, F.A.S.; Behera, S.K.; Krishnamoorthy, G. Excited state proton transfer of 2-(2′-hydroxyphenyl)benzimidazole and its nitrogen substituted analogues in bovine serum albumin. Photochem. Photobiol. Sci. 2014, 13, 1297–1304. [Google Scholar] [CrossRef]
- Forés, M.; Duran, M.; Solà, M.; Orozco, M.; Luque, F.J. Theoretical evaluation of solvent effects on the conformational and tautomeric equilibriaof 2-(2′-hydroxyphenyl)benzimidazole and on its absorption and fluorescence spectra. J. Phys. Chem. A 1999, 103, 4525–4532. [Google Scholar] [CrossRef]
- Qin, M.; Jin, D.; Che, W.; Jiang, Y.; Zhang, L.; Zhu, D.; Su, Z. Fluorescence response and detection of Cu2+ with 2-(2-hydroxyphenyl)benzimidazole in aqueous medium. Inorg. Chem. Commun. 2017, 75, 25–28. [Google Scholar] [CrossRef]
- Vaz, P.A.A.M.; Rocha, J.; Silva, A.M.S.; Guieu, S. Difluoroborate complexes based on 2′-hydroxyphenones as solid-state fluorophores. Dyes Pigment. 2021, 184, 108720. [Google Scholar] [CrossRef]
- Vaz, P.A.A.M.; Rocha, J.; Silva, A.M.S.; Guieu, S. Aggregation-induced emission enhancement of chiral boranils. New J. Chem. 2018, 42, 18166–18171. [Google Scholar] [CrossRef]
- Guieu, S.; Pinto, J.; Silva, V.L.M.; Rocha, J.; Silva, A.M.S. Synthesis, post-modification and fluorescence properties of boron diketonate complexes. Eur. J. Org. Chem. 2015, 2015, 3423–3426. [Google Scholar] [CrossRef]
- Cardona, F.; Rocha, J.; Silva, A.M.S.; Guieu, S. Δ1-pyrroline based boranyls: Synthesis, crystal structures and luminescent properties. Dyes Pigment. 2014, 111, 16–20. [Google Scholar] [CrossRef]
- Costa, L.D.; Guieu, S.; Rocha, J.; Silva, A.M.S.; Tomé, A.C. Porphyrin–boron diketonate dyads. New J. Chem. 2017, 41, 2186–2192. [Google Scholar] [CrossRef]
- Cui, Y.; Wang, S. Diboron and triboron compounds based on linear and star-shaped conjugated ligands with 8-hydroxyquinolate functionality: impact of intermolecular interaction and boron coordination on luminescence. J. Org. Chem. 2006, 71, 6485–6496. [Google Scholar] [CrossRef]
- Dhanunjayarao, K.; Mukundam, V.; Ramesh, M.; Venkatasubbaiah, K. Synthesis and optical properties of salicylaldimine-based diboron complexes. Eur. J. Inorg. Chem. 2014, 2014, 539–545. [Google Scholar] [CrossRef]
- Guieu, S.; Cardona, F.; Rocha, J.; Silva, A.M.S. Luminescent bi-metallic fluoroborate derivatives of bulky salen ligands. New J. Chem. 2014, 38, 5411–5414. [Google Scholar] [CrossRef]
- Li, G.; Lou, W.; Wang, D.; Deng, C.; Zhang, Q. Difluoroboron-enabled thermally activated delayed fluorescence. ACS Appl. Mater. Interfaces 2019, 11, 32209–32217. [Google Scholar] [CrossRef]
- Azarias, C.; Budzák, Š.; Laurent, A.D.; Ulrich, G.; Jacquemin, D. Tuning ESIPT fluorophores into dual emitters. Chem. Sci. 2016, 7, 3763–3774. [Google Scholar] [CrossRef] [Green Version]
- Sedgwick, A.C.; Wu, L.; Han, H.H.; Bull, S.D.; He, X.P.; James, T.D.; Sessler, J.L.; Tang, B.Z.; Tian, H.; Yoon, J. Excited-state intramolecular proton-transfer (ESIPT) based fluorescence sensors and imaging agents. Chem. Soc. Rev. 2018, 47, 8842–8880. [Google Scholar] [CrossRef] [Green Version]
- Kim, T.-I.; Kang, H.J.; Han, G.; Chung, S.J.; Kim, Y. A highly selective fluorescent ESIPT probe for the dual specificity phosphatase MKP-6. Chem. Commun. 2009, 2, 5895–5897. [Google Scholar] [CrossRef]
- Li, X.; Son, Y.-A. Efficient luminescence from easily prepared fluorine–boron core complexes based on benzothiazole and benzoxazole. Dyes Pigment. 2014, 107, 182–187. [Google Scholar] [CrossRef]
- Massue, J.; Ulrich, G.; Ziessel, R. Effect of 3,5-disubstitution on the optical properties of luminescent 2-(2′-hydroxyphenyl)benzoxazoles and their borate complexes. Eur. J. Org. Chem. 2013, 2013, 5701–5709. [Google Scholar] [CrossRef]
- Massue, J.; Frath, D.; Ulrich, G.; Retailleau, P.; Ziessel, R. Synthesis of luminescent 2-(2′-hydroxyphenyl)benzoxazole (hbo) borate complexes. Org. Lett. 2012, 14, 230–233. [Google Scholar] [CrossRef]
- Frath, D.; Massue, J.; Ulrich, G.; Ziessel, R. Luminescent materials: Locking π-conjugated and heterocyclic ligands with boron(III). Angew. Chem. Int. Ed. 2014, 53, 2290–2310. [Google Scholar] [CrossRef] [PubMed]
- Mukundam, V.; Dhanunjayarao, K.; Samal, S.; Venkatasubbaiah, K. Variation of para substituent on 2-phenol of tetraaryl-substituted imidazole-boron difluoride complexes: Synthesis, characterization, and photophysical properties. Asian J. Org. Chem. 2017, 6, 1054–1062. [Google Scholar] [CrossRef]
- Benelhadj, K.; Massue, J.; Retailleau, P.; Ulrich, G.; Ziessel, R. 2-(2′-Hydroxyphenyl)benzimidazole and 9,10-phenanthroimidazole chelates and borate complexes: Solution- and solid-state emitters. Org. Lett. 2013, 15, 2918–2921. [Google Scholar] [CrossRef]
- Dhanunjayarao, K.; Mukundam, V.; Venkatasubbaiah, K. tetracoordinate imidazole-based boron complexes for the selective detection of picric acid. Inorg. Chem. 2016, 55, 11153–11159. [Google Scholar] [CrossRef]
- Xu, H.; Xu, Z.-F.; Yue, Z.-Y.; Yan, P.-F.; Wang, B.; Jia, L.-W.; Li, G.-M.; Sun, W.-B.; Zhang, J.-W. A novel deep blue-emitting znii complex based on carbazole-modified 2-(2-hydroxyphenyl)benzimidazole: Synthesis, bright electroluminescence, and substitution effect on photoluminescent, thermal, and electrochemical properties. J. Phys. Chem. C 2008, 112, 15517–15525. [Google Scholar] [CrossRef]
- Esparza-Ruiz, A.; Peña-Hueso, A.; Nöth, H.; Flores-Parra, A.; Contreras, R. Boron coordination compounds derived from 2-phenyl-benzimidazole and 2-phenyl-benzotriazole bidentate ligands. J. Organomet. Chem. 2009, 694, 3814–3822. [Google Scholar] [CrossRef]
- Bolton, O.; Lee, K.; Kim, H.-J.; Lin, K.Y.; Kim, J. Activating efficient phosphorescence from purely organic materials by crystal design. Nat. Chem. 2011, 3, 205–210. [Google Scholar] [CrossRef]
- Ouyang, J.; Ouyang, C.; Fujii, Y.; Nakano, Y.; Shoda, T.; Nagano, T. Synthesis and fluorescent properties of 2-(1H-benzimidazol-2-yl)-phenol derivatives. J. Heterocycl. Chem. 2004, 41, 359–365. [Google Scholar] [CrossRef]
- Alaqeel, S.I. Synthetic approaches to benzimidazoles from o-phenylenediamine: A literature review. J. Saudi Chem. Soc. 2017, 21, 229–237. [Google Scholar] [CrossRef] [Green Version]
- Resch-Genger, U.; Rurack, K. Determination of the photoluminescence quantum yield of dilute dye solutions (IUPAC Technical Report). Pure Appl. Chem. 2013, 85, 2005–2013. [Google Scholar] [CrossRef]
- Manojai, N.; Daengngern, R.; Kerdpol, K.; Ngaojampa, C.; Kungwan, N. Heteroatom effect on photophysical properties of 2-(2′-hydroxyphenyl)benzimidazole and its derivatives as fluorescent dyes: A TD-DFT study. J. Lumin. 2017, 188, 275–282. [Google Scholar] [CrossRef]
- Akutsu, K.; Mori, S.; Shinmei, K.; Iwase, H.; Nakano, Y.; Fujii, Y. Investigation of substitution effect on fluorescence properties of Zn2+-selective ratiometric fluorescent compounds: 2-(2-Hydroxyphenyl)benzimidazole derivatives. Talanta 2016, 146, 575–584. [Google Scholar] [CrossRef] [PubMed]
- Padalkar, V.S.; Ramasami, P.; Sekar, N. A comprehensive spectroscopic and computational investigation of intramolecular proton transfer in the excited states of 2-(2-hydroxyphenyl) benzoxazole and its derivatives. J. Lumin. 2014, 146, 527–538. [Google Scholar] [CrossRef]
- Chipem, F.A.S.; Dash, N.; Krishnamoorthy, G. Role of nitrogen substitution in phenyl ring on excited state intramolecular proton transfer and rotamerism of 2-(2-hydroxyphenyl)benzimidazole: A theoretical study. J. Chem. Phys. 2011, 134, 104308. [Google Scholar] [CrossRef]
- Wang, R.; Liu, D.; Xu, K.; Li, J. Substituent and solvent effects on excited state intramolecular proton transfer in novel 2-(2′-hydroxyphenyl)benzothiazole derivatives. J. Photochem. Photobiol. A 2009, 205, 61–69. [Google Scholar] [CrossRef]
- Fontes, L.F.B.; Nunes da Silva, R.; Silva, A.M.S.; Guieu, S. Unsymmetrical 2,4,6-triarylpyridines as versatile scaffolds for deep-blue and dual emission fluorophores. ChemPhotoChem 2020, 4, 5312–5317. [Google Scholar] [CrossRef]
- Chen, K.-Y.; Hsieh, C.-C.; Cheng, Y.-M.; Lai, C.-H.; Chou, P.-T. Extensive spectral tuning of the proton transfer emission from 550 to 675 nm via a rational derivatization of 10-hydroxybenzo[h]quinoline. Chem. Commun. 2006, 1, 4395–4397. [Google Scholar] [CrossRef]
- Foley, S.; Berberan-Santos, M.N.; Fedorov, A.; Bensasson, R.V.; Leach, S.; Gigante, B. Effect of halogenated compounds on the photophysics of C70 and a monoadduct of C70: Some implications on optical limiting behaviour. Chem. Phys. 2001, 263, 437–447. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhang, H.; Jiao, C.; Ye, K.; Zhang, H.; Zhang, J.; Wang, Y. 2-(2-hydroxyphenyl)benzimidazole-based four-coordinate boron-containing materials with highly efficient deep-blue photoluminescence and electroluminescence. Inorg. Chem. 2015, 54, 2652–2659. [Google Scholar] [CrossRef]
- Yang, X.; Xu, X.; Zhou, G. Recent advances of the emitters for high performance deep-blue organic light-emitting diodes. J. Mater. Chem. C 2015, 3, 913–944. [Google Scholar] [CrossRef]
- ITU-R Recommendation BT.1700, Characteristics of Composite Video Signals for Conventional Analogue Television Systems. 2004. Available online: https://www.itu.int/rec/R-REC-BT.1700-0-200502-I/en (accessed on 1 March 2021).
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).