Dimethyl Carbonate as a Promising Oxygenated Fuel for Combustion: A Review
Abstract
1. Introduction
2. DMC Production Process
2.1. The Conventional Process of DMC Production
2.1.1. DMC from Phosgene
2.1.2. DMC from Partial Carbonylation (Bayer Process)
2.1.3. DMC from Methyl Nitrite Process
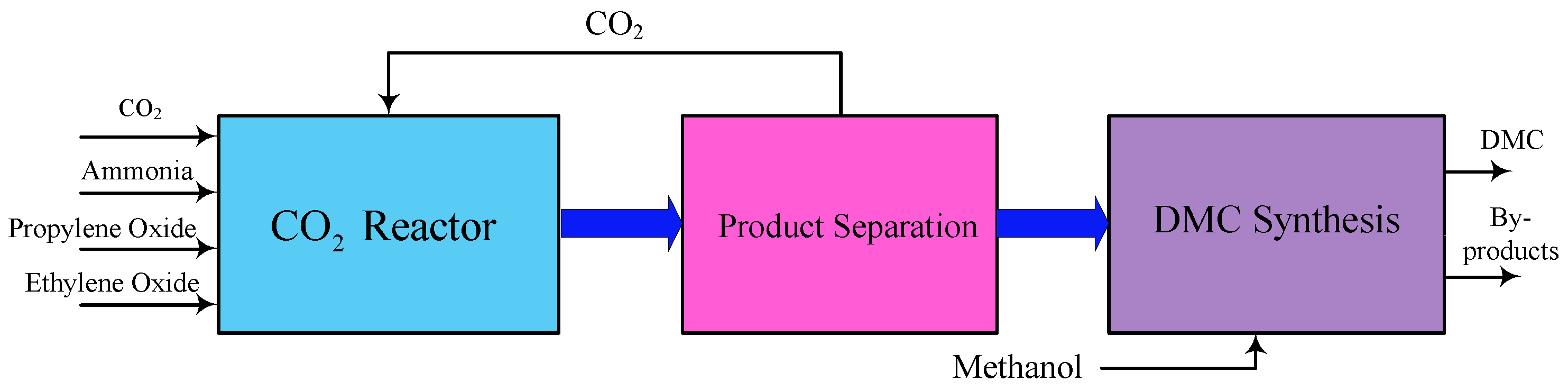
2.2. DMC Production from Carbon Dioxide
2.2.1. DMC from Direct Synthesis from and Alcohol
2.2.2. DMC Synthesis by Alcoholysis of Urea (Urea-Route)
2.2.3. DMC Creation from Propylene Carbonate (PC-Route)
2.2.4. DMC Creation from Ethylene Carbonate (EC-Route)
3. The Use of DMC to Fuel IC Engines
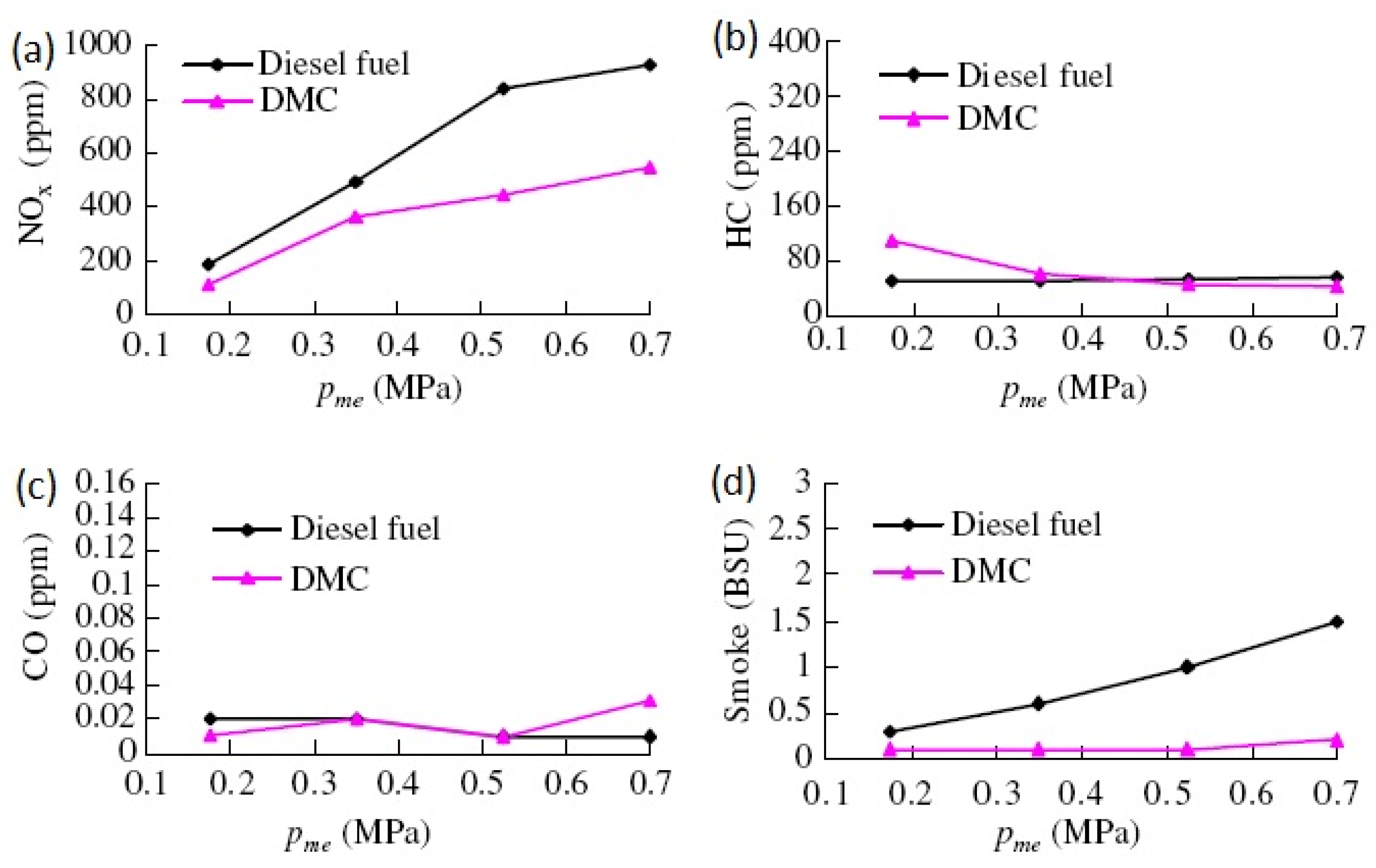
3.1. The Use of Pure DMC to Fuel Diesel Engines
3.2. The Use of DMC/Diesel Blends to Fuel Diesel Engines
3.3. The Use of DMC/Gasoline Blends to Fuel Gasoline Engines
4. The Oxidation and Combustion Kinetics of DMC
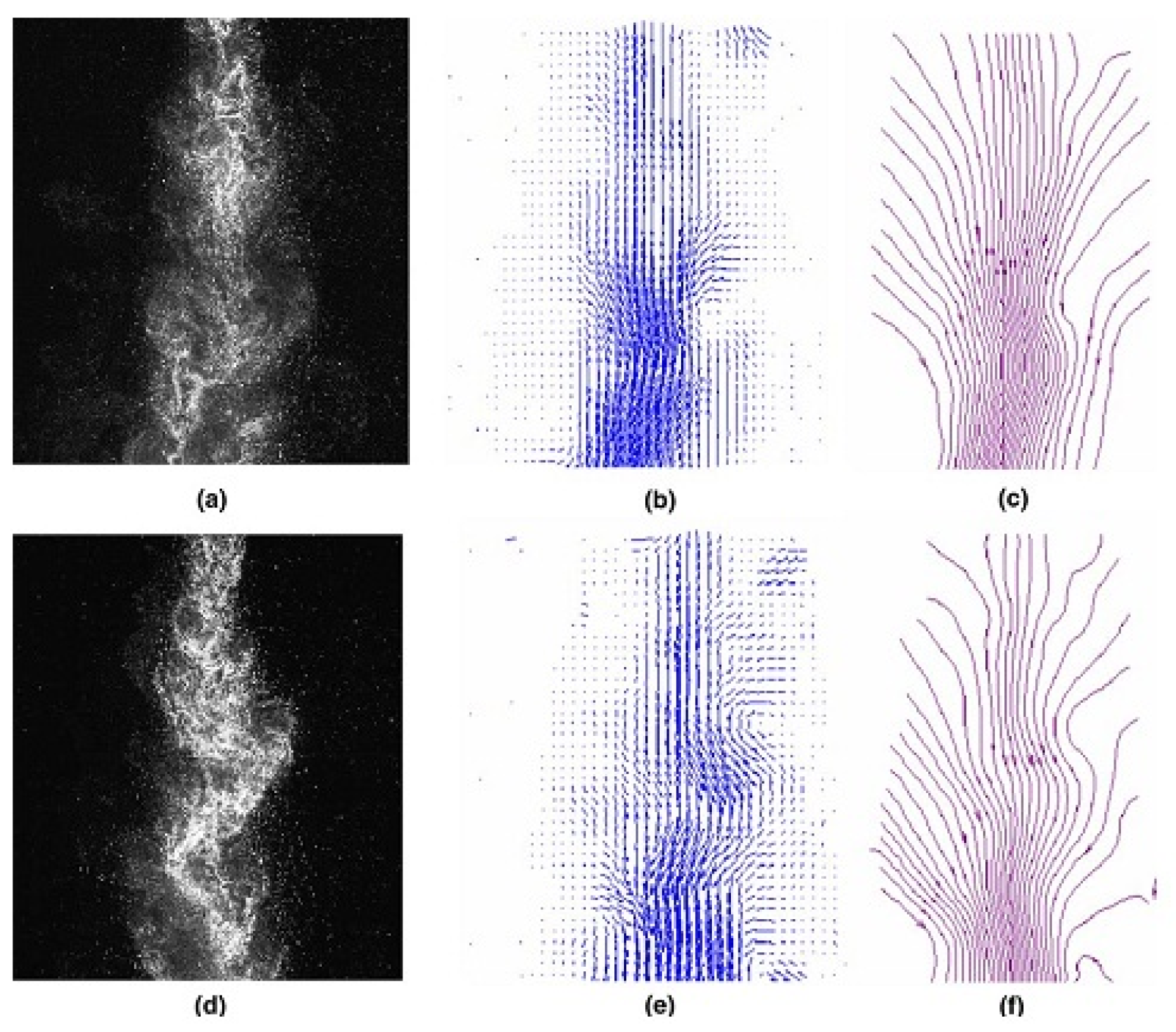
4.1. Laminar Burning Characteristics of DMC
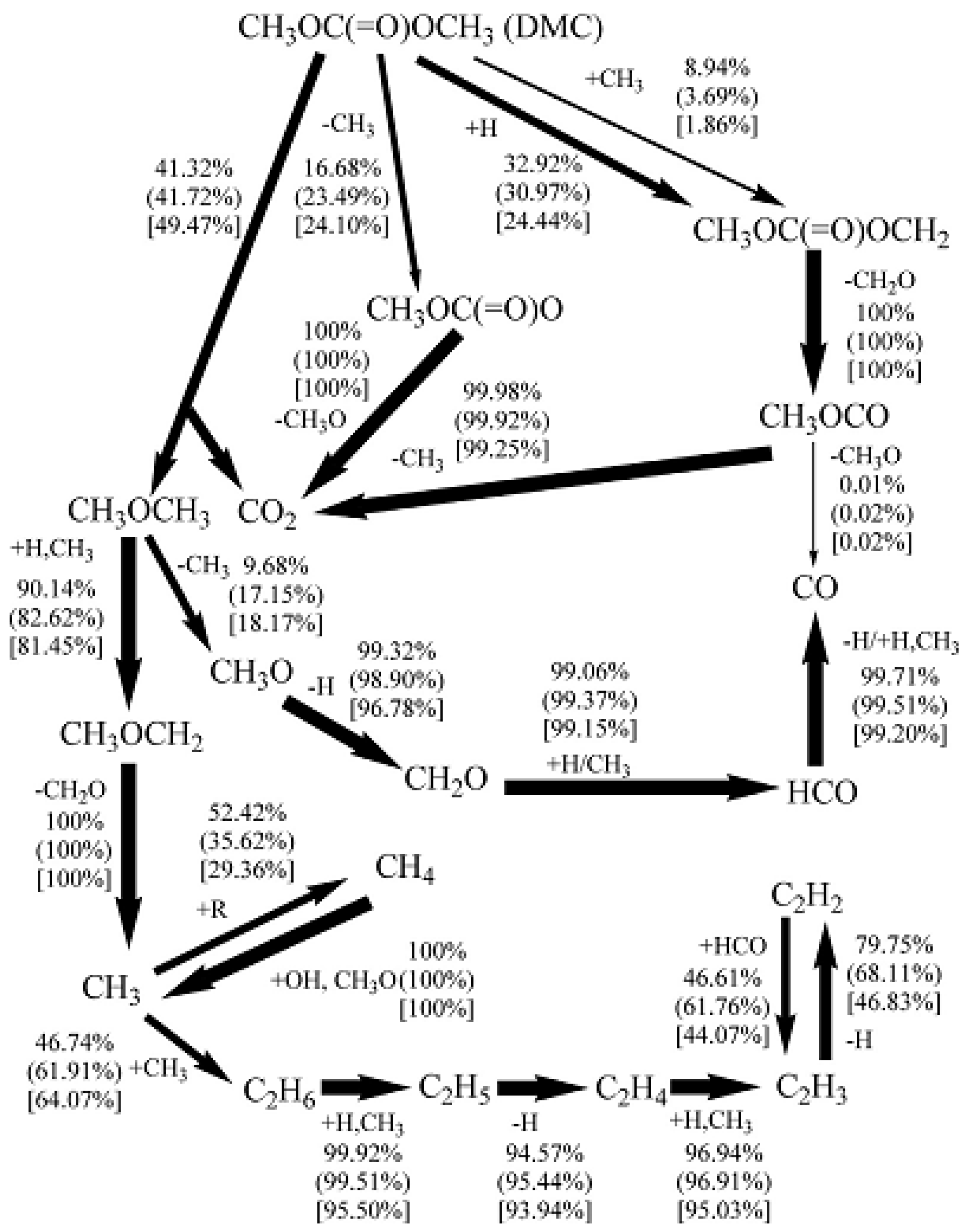
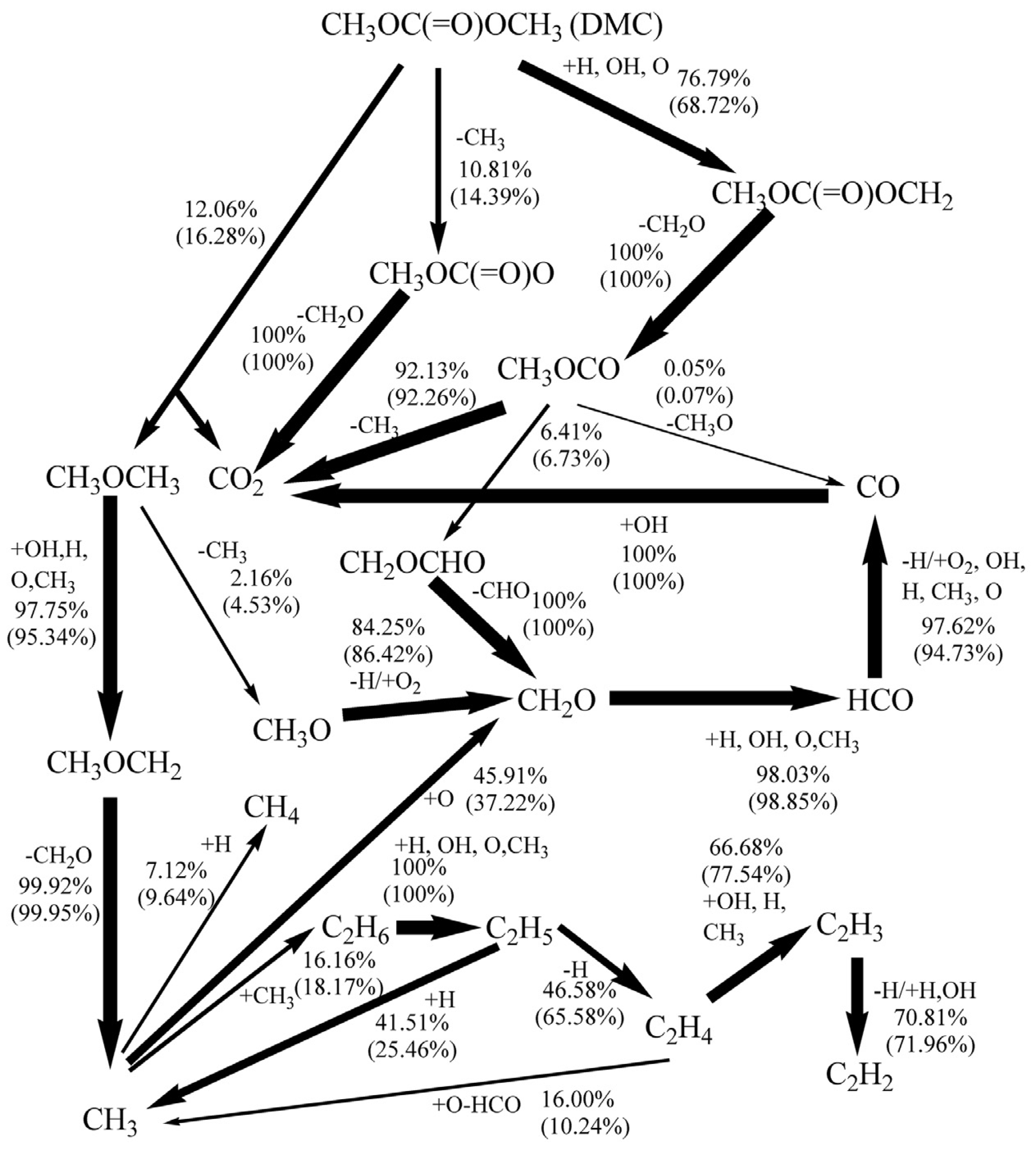
4.2. DMC Pyrolysis Mechanism
4.3. DMC Oxidation Mechanism
- DMC was consumed slower than alcohols such as methanol and ethanol.
- Increasing the DMC concentration can only affect slightly the yield of soot without remarkable influence on the gas yield, while the pyrolysis temperature affects soot formation, yields and gas yields.
- Effects of the equivalence ratio on ignition delay time depend on the initial pressure of DMC while increasing in the DMC pressure and concentration decreases the ignition delay time in terms of DMC oxidation and combustion mechanism.
5. Conclusions and Future Perspectives
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Fernando, S.; Hall, C.; Jha, S. NOx Reduction from Biodiesel Fuels. Energy Fuels 2006, 20, 376–382. [Google Scholar] [CrossRef]
- Beatrice, C.; Di Blasio, G.; Lazzaro, M.; Cannilla, C.; Bonura, G.; Frusteri, F.; Asdrubali, F.; Baldinelli, G.; Presciutti, A.; Fantozzi, F.; et al. Technologies for energetic exploitation of biodiesel chain derived glycerol: Oxy-fuels production by catalytic conversion. Appl. Energy 2013, 102, 63–71. [Google Scholar] [CrossRef]
- Fantozzi, F.; Frassoldati, A.; Bartocci, P.; Cinti, G.; Quagliarini, F.; Bidini, G.; Ranzi, E. An experimental and kinetic modeling study of glycerol pyrolysis. Appl. Energy 2016, 184, 68–76. [Google Scholar] [CrossRef]
- Xiaolu, L.; Hongyan, C.; Zhiyong, Z.; Zhen, H. Study of combustion and emission characteristics of a diesel engine operated with dimethyl carbonate. Energy Convers. Manag. 2006, 47, 1438–1448. [Google Scholar] [CrossRef]
- Huang, Z.; Jiang, D.; Zeng, K.; Liu, B.; Yang, Z. Combustion characteristics and heat release analysis of a direct injection compression ignition engine fuelled with diesel dimethyl carbonate blends. Proc. Inst. Mech. Eng. Part D J. Automob. Eng. 2003, 217, 595–605. [Google Scholar] [CrossRef]
- Rounce, P.; Tsolakis, A.; Leung, P.; York, A. A comparison of diesel and biodiesel emissions using dimethyl carbonate as an oxygenated additive. Energy Fuels 2010, 24, 4812–4819. [Google Scholar] [CrossRef]
- Schifter, I.; Gonzlez, U.; Gonzlez-Macas, C. Effects of ethanol, ethyl- tert-butyl ether and dimethyl-carbonate blends with gasoline on SI engine. Fuel 2016, 183, 253–261. [Google Scholar] [CrossRef]
- Wen, L.b.; Xin, C.Y.; Yang, S.C. The effect of adding dimethyl carbonate (DMC) and ethanol to unleaded gasoline on exhaust emission. Appl. Energy 2010, 87, 115–121. [Google Scholar] [CrossRef]
- Tundo, P.; Selva, M. The chemistry of dimethyl carbonate. Acc. Chem. Res. 2002, 35, 706–716. [Google Scholar] [CrossRef] [PubMed]
- Kumar, B.R.; Saravanan, S. Partially premixed low temperature combustion using dimethyl carbonate (DMC) in a DI diesel engine for favorable smoke/NOx emissions. Fuel 2016, 180, 396–406. [Google Scholar] [CrossRef]
- Alexandrino, K.; Alzueta, M.U.; Curran, H.J. An experimental and modeling study of the ignition of dimethyl carbonate in shock tubes and rapid compression machine. Combust. Flame 2018, 188, 212–226. [Google Scholar] [CrossRef]
- Alexandrino, K.; Salinas, J.; Millera, Á.; Bilbao, R.; Alzueta, M.U. Sooting propensity of dimethyl carbonate, soot reactivity and characterization. Fuel 2016, 183, 64–72. [Google Scholar] [CrossRef]
- Mei, D.; Hielscher, K.; Baar, R. Study on Combustion Process and Emissions of a Single Cylinder Diesel Engine Fueled with DMC/Diesel Blend. J. Energy Eng. 2014, 140, 04013004. [Google Scholar] [CrossRef]
- Glaude, P.A.; Pitz, W.J.; Thomson, M.J. Chemical kinetic modeling of dimethyl carbonate in an opposed flow diffusion flame. Proc. Combust. Inst. 2005, 30, 1111–1118. [Google Scholar] [CrossRef]
- Dias, V.; Vandooren, J. Experimental and modeling studies of C2H4/O2/Ar, C2H4/methylal/O2/Ar and C2H4/ethylal/O2/Ar rich flames and the effect of oxygenated additives. Combust. Flame 2011, 158, 848–859. [Google Scholar] [CrossRef]
- Noorman, M.T. The Effect of MTBE, DIPE and TAME on Vehicle Emissions. Exhaust Emiss. 1993. [Google Scholar] [CrossRef]
- Kivi, J.; Niemi, A.; Nylund, N.O.; Kyt, M.; Orre, K. Use of MTBE and ETBE as Gasoline Reformulation Components. SAE Tech. Pap. 1992, 22, 209–226. [Google Scholar]
- Mennear, J.H. Carcinogenicity Studies on MTBE: Critical Review and Interpretation. Risk Anal. 1997, 17, 673. [Google Scholar] [CrossRef] [PubMed]
- Keller, A.A.; Sandall, O.C.; Rinker, R.G.; Mitani, M.M.; Bierwagen, B.; Snodgrass, M.J. Cost and Performance Evaluation of Treatment Technologies for MTBE-Contaminated Water. UC TSR&TP Report to the Governor of California. 1998. Available online: https://www.researchgate.net/publication/243778975_Cost_and_Performance_Evaluation_of_Treatment_Technologies_for_MTBE-Contaminated_Water (accessed on 5 June 2018).
- Keller, A.A.; Bierwagen, B.; Sirivithayapakorn, S.; Kram, M. Advances In Groundwater Treatment To Remove Mtbe. In Hazardous and Industrial Waste Proceedings, 31st Mid-Atlantic Conference; CRC Press: Boca Raton, FL, USA, 2007. [Google Scholar]
- Kampbell, D.H.; An, Y.J.; Williams, V.R. Influence of Methyl tert -Butyl Ether on Lake Water Algae. Bull. Environ. Contam. Toxicol. 2001, 67, 574–579. [Google Scholar] [CrossRef] [PubMed]
- Bretherick, L. Gasoline Motor Fuels Containing Dialkyl Carbonate—As Octane Improver. EP Patent 82688, 29 June 1983. [Google Scholar]
- Short, G.D.; Spencer, M.S. Gasoline Compositions Foric Engine—Comprises Gasoline Hydrocarbon and Dimethyl Carbonate. EP Patent 98691, 18 April 1984. [Google Scholar]
- Jessup, P.J.; Brass, S.G.; Croudace, M.C. Gasoline Compositions Containing Carbonates. U.S. Patent 06728244, 29 April 1986. [Google Scholar]
- Smith, H.A. Dialkyl Carbonates as Phase Separation Inhibitors in Liquid Hydrocarbon Fuel and Ethanol Mixtures. U.S. Patent 06353691, 1 March 1983. [Google Scholar]
- Jiang, C.; Guo, Y.; Wang, C.; Hu, C.; Wu, Y.; Wang, E. Synthesis of dimethyl carbonate from methanol and carbon dioxide in the presence of polyoxometalates under mild conditions. Appl. Catal. A Gen. 2003, 256, 203–212. [Google Scholar] [CrossRef]
- Liotta, F.J.; Montalvo, D.M. The effect of oxygenated fuels on emissions from a modern heavy duty diesel engine. SAE Tech. Pap. 1993. [Google Scholar] [CrossRef]
- Westbrook, C.K.; Pitz, W.J.; Curran, H.J. Chemical Kinetic Modeling Study of the Effects of Oxygenated Hydrocarbons on Soot Emissions from Diesel Engines. J. Phys. Chem. A 2006, 110, 6912–6922. [Google Scholar] [CrossRef] [PubMed]
- Miyamoto, N.; Ogawa, H.; Arima, T.; Miyakawa, K. Improvement of Diesel Combustion and Emissions with Addition of Various Oxygenated Agents to Diesel Fuels. SAE Tech. Pap. 1996, 11–26. [Google Scholar] [CrossRef]
- Renard, C.; Tiggelen, P.J.V.; Vandooren, J. Effect of dimethoxymethane addition on the experimental structure of a rich ethylene/oxygen/argon flame. Proc. Combust. Inst. 2002, 29, 1277–1284. [Google Scholar] [CrossRef]
- Dias, V.; Katshiatshia, H.M.; Jeanmart, H. The influence of ethanol addition on a rich premixed benzene flame at low pressure. Combust. Flame 2014, 161, 2297–2304. [Google Scholar] [CrossRef]
- Pacheco, M.A.; Marshall, C.L. Review of Dimethyl Carbonate (DMC) Manufacture and Its Characteristics as a Fuel Additive. Energy Fuels 1997, 11, 2–29. [Google Scholar] [CrossRef]
- Huang, S.; Yan, B.; Wang, S.; Ma, X. ChemInform Abstract: Recent Advances in Dialkyl Carbonates Synthesis and Applications. Chem. Soc. Rev. 2015, 44, 3079. [Google Scholar] [CrossRef] [PubMed]
- Pyo, S.H.; Ji, H.P.; Chang, T.S.; Hatti-Kaul, R. Dimethyl carbonate as a green chemical. Curr. Opin. Green Sustain. Chem. 2017, 5, 61–66. [Google Scholar] [CrossRef]
- Qian, Y.; Zhu, L.; Wang, Y.; Lu, X. Recent progress in the development of biofuel 2,5-dimethylfuran. Renew. Sustain. Energy Rev. 2015, 41, 633–646. [Google Scholar] [CrossRef]
- Arteconi, A.; Mazzarini, A.; Di Nicola, G. Emissions from ethers and organic carbonate fuel additives: A review. Water Air Soil Pollut. 2011, 221, 405. [Google Scholar] [CrossRef]
- Bhanage, B.M.; Fujita, S.I.; Ikushima, Y.; Arai, M. Synthesis of dimethyl carbonate and glycols from carbon dioxide, epoxides, and methanol using heterogeneous basic metal oxide catalysts with high activity and selectivity. Appl. Catal. A Gen. 2001, 219, 259–266. [Google Scholar] [CrossRef]
- Matsuzaki, T.; Nakamura, A. Dimethyl carbonate synthesis and other oxidative reactions using alkyl nitrites. Catal. Surv. Asia 1997, 1, 77–88. [Google Scholar] [CrossRef]
- Aricò, F.; Tundo, P. Dimethyl carbonate: A modern green reagent and solvent. Russ. Chem. Rev. 2010, 79, 479–489. [Google Scholar] [CrossRef]
- Han, M.S.; Lee, B.G.; Suh, I.; Kim, H.S.; Ahn, B.S.; Hong, S.I. Synthesis of dimethyl carbonate by vapor phase oxidative carbonylation of methanol over Cu based catalysts. J. Mol. Catal. A Chem. 2001, 170, 225–234. [Google Scholar] [CrossRef]
- Sato, Y.; Kagotani, M.; Souma, Y. A new type of support ‘bipyridine containing aromatic polyamide’ to CuCl2 for synthesis of dimethyl carbonate (DMC) by oxidative carbonylation of methanol. J. Mol. Catal. A Chem. 2000, 1–2, 79–85. [Google Scholar] [CrossRef]
- Itoh, H.; Watanabe, Y.; Mori, K.; Umino, H. Synthesis of dimethyl carbonate by vapor phase oxidative carbonylation of methanol. Green Chem. 2003, 5, 558–562. [Google Scholar] [CrossRef]
- Yamamoto, Y. Vapor Phase Carbonylation Reactions Using Methyl Nitrite Over Pd Catalysts. Catal. Surv. Asia 2010, 14, 103–110. [Google Scholar] [CrossRef]
- Delledonne, D.; Rivetti, F.; Romano, U. Developments in the production and application of dimethylcarbonate. Appl. Catal. A Gen. 2001, 221, 241–251. [Google Scholar] [CrossRef]
- Wang, W.; Wang, S.; Ma, X.; Gong, J. Recent advances in catalytic hydrogenation of carbon dioxide. Chem. Soc. Rev. 2011, 40, 3703–3727. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.C.; Sakakura, T.; Sako, T. Reaction of Dialkyltin Methoxide with Carbon Dioxide Relevant to the Mechanism of Catalytic Carbonate Synthesis. J. Am. Chem. Soc. 1999, 121, 3793–3794. [Google Scholar] [CrossRef]
- Zhao, T.; Han, Y.; Sun, Y. Novel reaction route for dimethyl carbonate synthesis from CO2 and methanol. Fuel Proc. Technol. 2000, 62, 187–194. [Google Scholar] [CrossRef]
- Yu, K.M.; Curcic, I.; Gabriel, J.; Tsang, S.C. Recent advances in CO2 capture and utilization. Chemsuschem 2008, 1, 893–899. [Google Scholar] [CrossRef] [PubMed]
- Kongpanna, P.; Pavarajarn, V.; Gani, R.; Assabumrungrat, S. Techno-economic evaluation of different CO2 based processes for dimethyl carbonate production. Chem. Eng. Res. Des. 2015, 93, 496–510. [Google Scholar] [CrossRef]
- Hoffman, W.A., III. A convenient preparation of carbonates from alcohols and carbon dioxide. J. Org. Chem. 1982, 47, 5209–5210. [Google Scholar] [CrossRef]
- Eta, V.; Mäki-Arvela, P.; Wärnå, J.; Salmi, T.; Mikkola, J.P.; Murzin, D.Y. Kinetics of dimethyl carbonate synthesis from methanol and carbon dioxide over ZrO2 MgO catalyst in the presence of butylene oxide as additive. Appl. Catal. A Gen. 2011, 404, 39–46. [Google Scholar] [CrossRef]
- Aresta, M. Perspectives in the use of carbon dioxide. Química Nova 1999, 22, 269–272. [Google Scholar] [CrossRef]
- Guo, X.C.; Qin, Z.F.; Wang, G.F.; Wang, J.G. Critical temperatures and pressures of reacting mixture in synthesis of dimethyl carbonate with methanol and carbon dioxide. Chin. Chem. Lett. 2008, 19, 249–252. [Google Scholar] [CrossRef]
- Shukla, K.; Srivastava, V.C. Synthesis of organic carbonates from alcoholysis of urea: A review. Catal. Rev. 2017, 59, 1–43. [Google Scholar] [CrossRef]
- Wang, M.; Wang, H.; Zhao, N.; Wei, W.; Sun, Y. Synthesis of dimethyl carbonate from urea and methanol over solid base catalysts. Catal. Commun. 2006, 7, 6–10. [Google Scholar] [CrossRef]
- Wang, H.; Lu, B.; Wang, X.; Zhang, J.; Cai, Q. Highly selective synthesis of dimethyl carbonate from urea and methanol catalyzed by ionic liquids. Fuel Proc. Technol. 2009, 90, 1198–1201. [Google Scholar] [CrossRef]
- Haggin, J. Catalytic cosynthesis method developed. Chem. Eng. News 1992, 70, 25–26. [Google Scholar] [CrossRef]
- Knifton, J.F.; Duranleau, R.G. Ethylene glycol dimethyl carbonate cogeneration. J. Mol. Catal. 1991, 67, 389–399. [Google Scholar] [CrossRef]
- Santos, B.A.; Silva, V.M.; Loureiro, J.M.; Rodrigues, A.E. Review for the direct synthesis of dimethyl carbonate. ChemBioEng Rev. 2014, 1, 214–229. [Google Scholar] [CrossRef]
- Murugan, C.; Bajaj, H. Transesterification of Propylene Carbonate With Methanol Using Mg-Al-CO3 Hydrotalcite as Solid Base Catalyst. Catal. Lett. 2010, 137, 224–231. [Google Scholar] [CrossRef]
- Zhang, G.; Liu, H.; Xia, X.; Zhang, W.; Fang, J. Effects of dimethyl carbonate fuel additive on diesel engine performances. Proc. Inst. Mech. Eng. Part D J. Automob. Eng. 2005, 219, 897–903. [Google Scholar] [CrossRef]
- Cheung, C.; Zhu, R.; Huang, Z. Investigation on the gaseous and particulate emissions of a compression ignition engine fueled with diesel dimethyl carbonate blends. Sci. Total Environ. 2011, 409, 523–529. [Google Scholar] [CrossRef] [PubMed]
- Mehta, B.H.; Mandalia, H.V.; Mistry, A.B. A review on effect of oxygenated fuel additive on the performance and emission characteristics of diesel engine. In Proceedings of the National Conference on Recent Trends in Engineering & Technology, Anand, India, 13–14 May 2011; pp. 13–14. [Google Scholar]
- Wang, H.; Huang, Z.; Zhou, L.; Jiang, D.; Yang, Z. Investigation on emission characteristics of a compression ignition engine with oxygenated fuels and exhaust gas recirculation. Proc. Inst. Mech. Eng. Part D J. Automob. Eng. 2000, 214, 503–508. [Google Scholar] [CrossRef]
- Mei, D.; Yue, S.; Zhao, X.; Hielscher, K.; Baar, R. Effects of center of heat release on combustion and emissions in a PCCI diesel engine fuelled by DMC diesel blend. Appl. Therm. Eng. 2017, 114, 969–976. [Google Scholar] [CrossRef]
- Schifter, I.; Gonzalez, U.; Diaz, L.; Sanchez Reyna, G.; Mejia Centeno, I.; Gonzalez Macias, C. Comparison of performance and emissions for gasoline oxygenated blends up to 20 percent oxygen and implications for combustion on a spark ignited engine. Fuel 2017, 208, 673–681. [Google Scholar] [CrossRef]
- Gopinath, D.; Sundaram, E.G. Experimental Investigation on the Effect of Adding Di Methyl Carbonate to Gasoline in a SI Engine Performance. Int. J. Sci. Eng. Res. 2012, 3, 1–5. [Google Scholar]
- Xu, C.; Zhong, A.; Li, X.; Wang, C.; Sahu, A.; Xu, H.; Lattimore, T.; Zhou, K.; Huang, Y. Laminar burning characteristics of upgraded biomass pyrolysis fuel derived from rice husk at elevated pressures and temperatures. Fuel 2017, 210, 249–261. [Google Scholar] [CrossRef]
- Wu, X.; Huang, Z.; Jin, C.; Wang, X.; Wei, L. Laminar burning velocities and Markstein lengths of 2, 5 dimethylfuran-air premixed flames at elevated temperatures. Combust. Sci. Technol. 2010, 183, 220–237. [Google Scholar] [CrossRef]
- Kitagawa, T. Effects of pressure on burning velocity and instabilities of propane air premixed flames. JSME Int. J. Ser. B Fluids Therm. Eng. 2005, 48, 2–8. [Google Scholar] [CrossRef]
- Gu, X.J.; Haq, M.Z.; Lawes, M.; Woolley, R. Laminar burning velocity and Markstein lengths of methane air mixtures. Combust. Flame 2000, 121, 41–58. [Google Scholar] [CrossRef]
- Xie, Y.; Wang, J.; Cai, X.; Huang, Z. Self-acceleration of cellular flames and laminar flame speed of syngas/air mixtures at elevated pressures. Int. J. Hydrogen Energy 2016, 41, 18250–18258. [Google Scholar] [CrossRef]
- Bardin, M.E.; Ivanov, E.V.; Nilsson, E.J.; Vinokurov, V.A.; Konnov, A.A. Laminar burning velocities of dimethyl carbonate with air. Energy Fuels 2013, 27, 5513–5517. [Google Scholar] [CrossRef]
- Sun, W.; Yang, B.; Hansen, N.; Westbrook, C.K.; Zhang, F.; Wang, G.; Moshammer, K.; Law, C.K. An experimental and kinetic modeling study on dimethyl carbonate (DMC) pyrolysis and combustion. Combust. Flame 2016, 164, 224–238. [Google Scholar] [CrossRef]
- Yu, H.; Hu, E.; Yang, K.; Huang, Z. Experimental and Numerical Research on Laminar Burning Characteristics of Premixed Dimethyl Carbonate/Air Flames. J. Xi’an Jiaotong Univ. 2014, 48, 25–31. [Google Scholar]
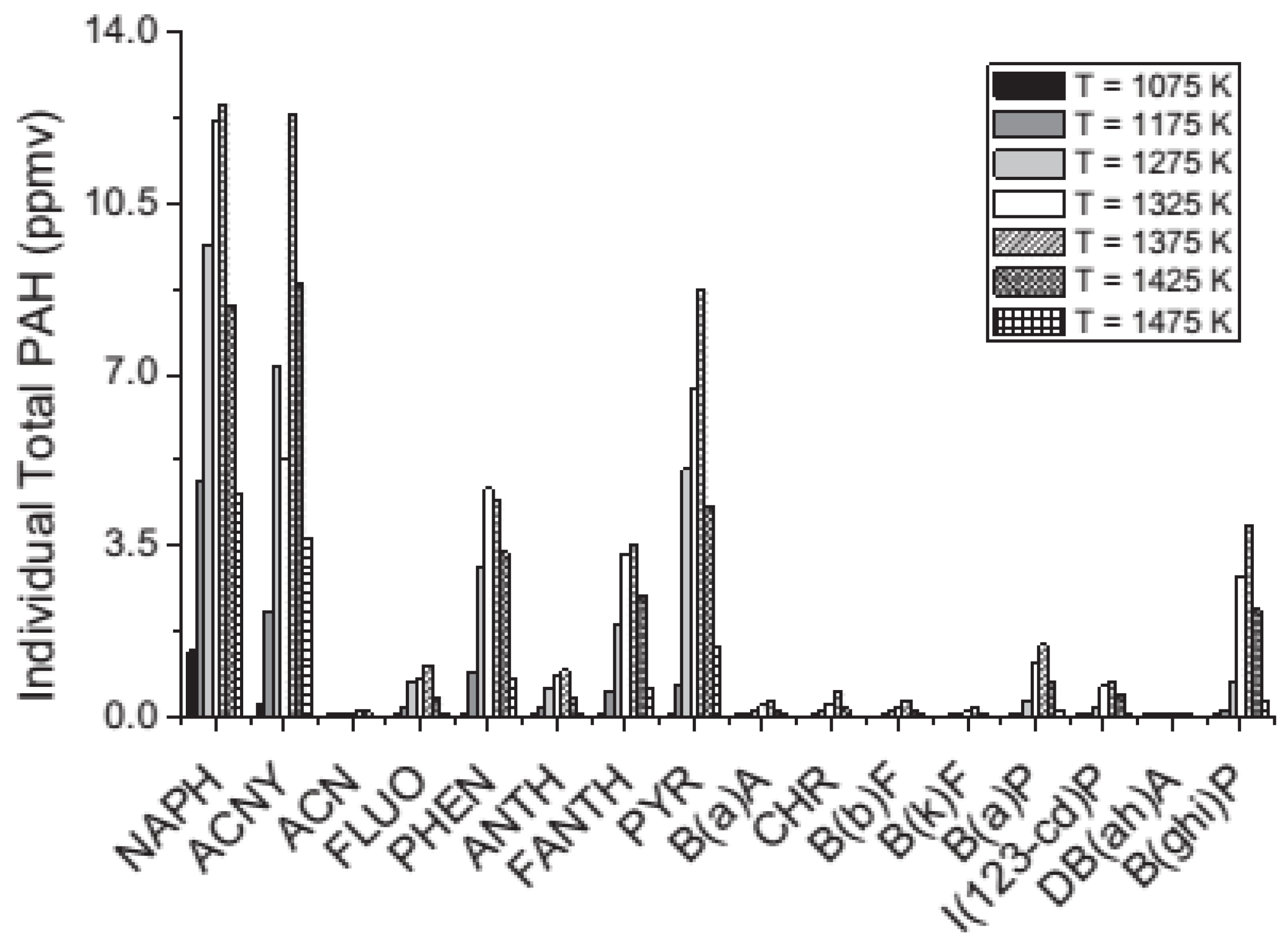
- Viteri, F.; Salinas, J.; Millera, Á.; Bilbao, R.; Alzueta, M. Pyrolysis of dimethyl carbonate: PAH formation. J. Anal. Appl. Pyrolysis 2016, 122, 524–530. [Google Scholar] [CrossRef]
- Alzueta, M.U.; Salinas, P.; Millera, Á.; Bilbao, R.; Abián, M. A study of dimethyl carbonate conversion and its impact to minimize soot and NO emissions. Proc. Combust. Inst. 2017, 36, 3985–3993. [Google Scholar] [CrossRef]
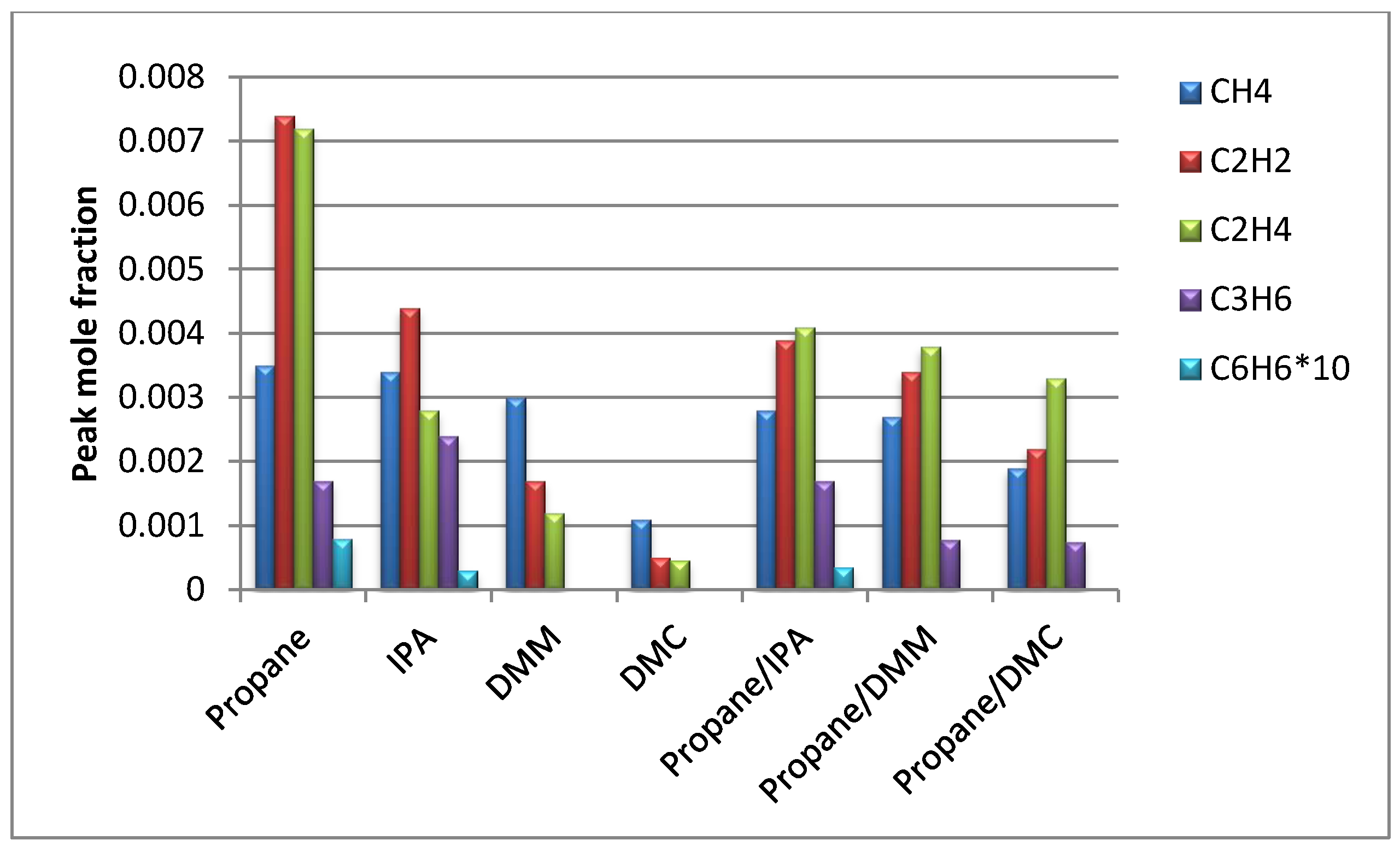
- Sinha, A.; Thomson, M. The chemical structures of opposed flow diffusion flames of C3 oxygenated hydrocarbons (isopropanol, dimethoxy methane, and dimethyl carbonate) and their mixtures. Combust. Flame 2004, 136, 548–556. [Google Scholar] [CrossRef]
- Chen, G.; Yu, W.; Fu, J.; Mo, J.; Huang, Z.; Yang, J.; Wang, Z.; Jin, H.; Qi, F. Experimental and modeling study of the effects of adding oxygenated fuels to premixed n-heptane flames. Combust. Flame 2012, 159, 2324–2335. [Google Scholar] [CrossRef]
- Sun, W.; Yang, B.; Hansen, N.; Moshammer, K. The influence of dimethoxy methane (DMM)/dimethyl carbonate (DMC) addition on a premixed ethane/oxygen/argon flame. Proc. Combust. Inst. 2017, 36, 449–457. [Google Scholar] [CrossRef]
- Hu, E.; Chen, Y.; Zhang, Z.; Pan, L.; Li, Q.; Cheng, Y.; Huang, Z. Experimental and kinetic study on ignition delay times of dimethyl carbonate at high temperature. Fuel 2015, 140, 626–632. [Google Scholar] [CrossRef]



| Properties | DMC [32,33,34] | Ethanol [35] | MTBE [36] | DMM [36] | Gasoline [35] | Diesel [35] |
|---|---|---|---|---|---|---|
| Chemical formula | – | – | ||||
| Molar mass (g/mol) | 90.08 | 46.07 | 88.15 | 76.1 | 100–105 | 180–200 |
| Carbon content (%) | 40 | 52.14 | 68.13 | 47.35 | – | 86 |
| Hydrogen content (%) | 6.71 | 13.13 | 13.72 | 10.6 | – | 14 |
| Oxygen content (%) | 53.28 | 34.73 | 18.15 | 42.05 | 0 | 0 |
| Carbon to oxygen ratio (by mass) | 0.75 | 1.5 | 3.75 | 1.13 | 0 | 0 |
| Stoichiometric air-fuel ratio | 4.57 | 8.94 | 11.68 | 7.22 | 14.7 | 14.3 |
| Melting point (C) | 4.6 | −114.14 | – | – | ||
| Flash point (C) | 21.7 | – | 70 | |||
| Boiling point (C) | 90.1 | 78.24 | 55.2 | 42 | 27–225 | 180–370 |
| Auto–ignition temperature (C) | 195 | 434 | 460 | 237 | 420 | 246 |
| Density (kg/m at 20 C) | 1069.4 | 790.9 | 740.4 | 859.3 | 744.6 | 820 |
| Kinematic viscosity (cSt, 20 C) | 0.63 | 1.5 | – | – | 0.37–0.44 | 2.77 |
| Net lower-heat value (MJ/kg) | 15.78 | 26.9 | 35.2 | 22.4 | 42.9 | 42.5 |
| Latent heat of evaporation (kJ/kg) | 369 | 919.6 | – | – | 351 | 270–301 |
| Research octane number (RON) | 101–116 | 108.7 | 115 | – | 90–100 | 25 |
| Cetane number | 35 | 8 | – | 30 | 10–15 | 40–45 |
| Water solubility (g/L) | 139 | Miscible | 42 | – | Negligible | – |
| Pure DMC in | DMC/Diesel in | DMC/Gasoline in | ||
|---|---|---|---|---|
| Diesel Engines | Diesel Engines | Gasoline Engines | ||
| No effect [4] | Improved [13] | Improved [8] | ||
| Engine | Negative effect [4] | Improved [13] | Improved [67] | |
| Emissions | Negative effect [8] | |||
| Improved [4] | Controversial [61,63,65] | Controversial [8,66,67] | ||
| Engine performance | Brake specific fuel consumption | Negative effect [4] | Negative effect [64] | Negative effect [8] |
| Exhaust gases temperature | Improved [4] | Improved [65] | Negative effect [8] | |
| Brake thermal efficiency | Improved [4] | Improved [61] | Improved [67] |
| Equivalence Ratio | at 298 K (cm/s) | at 313 K (cm/s) | at 338 K (cm/s) | at 358 K (cm/s) |
|---|---|---|---|---|
| 0.65 | 13.05 | – | – | – |
| 0.70 | 16.45 | 18 | 20.25 | 23.25 |
| 0.8 | 22.43 | 24.45 | 27.3 | 30.2 |
| 0.9 | 26.34 | 29.14 | 32.15 | 35.76 |
| 1 | 28.96 | 32.15 | 35.13 | 39.01 |
| 1.1 | – | 32.98 | 36.17 | 39.87 |
| 1.2 | – | 31.87 | 35.13 | 38.67 |
| 1.3 | – | 29.19 | 31.95 | 35.64 |
| 1.4 | – | 24.51 | 27.24 | 29.75 |
| 1.5 | – | 19.99 | 21.3 | 24.8 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abdalla, A.O.G.; Liu, D. Dimethyl Carbonate as a Promising Oxygenated Fuel for Combustion: A Review. Energies 2018, 11, 1552. https://doi.org/10.3390/en11061552
Abdalla AOG, Liu D. Dimethyl Carbonate as a Promising Oxygenated Fuel for Combustion: A Review. Energies. 2018; 11(6):1552. https://doi.org/10.3390/en11061552
Chicago/Turabian StyleAbdalla, Ayoub O. G., and Dong Liu. 2018. "Dimethyl Carbonate as a Promising Oxygenated Fuel for Combustion: A Review" Energies 11, no. 6: 1552. https://doi.org/10.3390/en11061552
APA StyleAbdalla, A. O. G., & Liu, D. (2018). Dimethyl Carbonate as a Promising Oxygenated Fuel for Combustion: A Review. Energies, 11(6), 1552. https://doi.org/10.3390/en11061552





