Simple Summary
Many men now live for many years after prostate cancer diagnosis. Hormone therapy is one of the main treatments available, especially when the cancer is advanced, but its long-term effects on daily life are not well understood. In this review, we looked at studies of men who survived at least 5 years after prostate cancer diagnosis and had received hormone therapy. The findings show that, compared with men treated only with local therapies, such as surgery or radiation, those who had hormone therapy often reported poorer overall health, more physical and emotional difficulties, lower energy, and greater problems with social life. They also experienced more bowel, urinary, and sexual problems. In addition, very little research has followed men beyond 15 years. These insights highlight the need for clearer discussions with patients about long-term effects and for future studies to better understand how different hormone treatments affect long-term quality of life.
Abstract
Prostate cancer accounts for the largest group of cancer survivors in men. Hormone therapy is essential, especially in advanced disease. While its short-term effects are well studied, research into the long-term effects on health-related quality of life (HRQOL) remains limited. Therefore, this review aims to synthesize and identify key gaps in the literature on HRQOL and symptom burden in long-term prostate cancer survivors who underwent hormone therapy. After searching four databases until 15 April 2025, we identified 14 observational studies that reported on general and prostate cancer-specific HRQOL in prostate cancer survivors ≥ 5 years post-diagnosis. Survivors who underwent hormone therapy reported worse global health status and physical, emotional, and social functioning compared to those treated with local therapies like prostatectomy or radiation. These survivors also experienced greater symptom burdens, alongside worse vitality and mental health. Prostate cancer-specific issues, such as bowel and urinary bother and sexual dysfunction, were also more pronounced in hormone therapy recipients. Nevertheless, outcomes beyond 15 years remain under-researched. The findings of this review highlight the importance of discussing long-term HRQOL compromises with patients who consider hormone therapy. However, they warrant cautious interpretation, particularly due to limited details on hormone therapy regimens and inadequate control for stage.
1. Introduction
Prostate cancer is the second most common cancer in men worldwide, with over 1.4 million new cases and more than 390,000 deaths each year [1,2]. The number of new cases of prostate cancer is predicted to double by 2040, due to population aging, increased life expectancy, and increasing rates of prostate-specific antigen (PSA) testing [3]. Advances in early diagnosis and treatment modalities have significantly improved prognosis, achieving a 5-year relative survival rate of 97.5% [4]. In addition, mortality from prostate cancer has decreased in most Western countries [5,6]. This has led to a growing population of long-term prostate cancer survivors, defined as those living 5 years or more after diagnosis [7].
Treatment options for prostate cancer include active surveillance, surgery, radiation therapy, hormone therapy, chemotherapy, immunotherapy, and targeted therapy, with the choice depending on the stage and aggressiveness of the disease [8]. Hormone therapy includes traditional androgen deprivation therapy (ADT), such as LHRH agonists and antagonists, as well as first- and second-generation anti-androgens, which bind to androgen receptors to prevent androgens from stimulating tumor growth [9]. Due to the significant androgen dependence of prostate cancer cells, ADT has been considered the cornerstone of systemic treatment for men with prostate cancer since 1941 [10]. One of the benefits of ADT is its ability to effectively reduce levels of circulating testosterones, which fuel the growth of prostate cancer cells [11]. Prostate cancer patients with advanced and recurrent disease often require systemic treatment, including ADT [12]. ADT is also increasingly used either as monotherapy or in combination with other treatment modalities for the management of clinically localized prostate cancer, and is often prescribed as a lifelong treatment [13,14]. Despite its efficacy in controlling disease progression, ADT has been associated with several adverse effects. These include cardiovascular complications, osteoporosis, impaired cognitive function, sexual dysfunction, fatigue, and reduced sleep quality. Certain side effects are specifically associated with the use of second-generation antiandrogens, such as hypertension and hypothyroidism [11]. These side effects can persist long after treatment, affecting multiple aspects of survivors’ health and health-related quality of life (HRQOL) [15]. Moreover, most of these symptoms are chronic in nature, which raises concerns about the long-term impact on prostate cancer survivors who undergo hormone therapy [15].
As hormone therapy becomes more broadly indicated, and an increasing number of prostate cancer survivors are maintained on long-term prescriptions, it is imperative to investigate the long-term effects of hormone therapy on the HRQOL, including physical and psychological wellbeing [16,17,18]. Consistent with established oncological definitions, long-term survivorship is defined as 5 or more years after diagnosis [19]. Several studies have explored the HRQOL of prostate cancer survivors treated with hormone therapy, particularly physical health, mental health, and sexual functioning [15,17,18,20]. However, most of these studies have been conducted on prostate cancer patients during or shortly after their treatment, with only few studies having examined the long-term HRQOL of prostate cancer survivors [15]. In addition, previous systematic reviews evaluating HRQOL in prostate cancer patients receiving hormone therapy have primarily focused on comparisons between different hormone therapy regimens rather than assessing the independent long-term impact of hormone therapy on overall quality of life [21,22]. These reviews have also concentrated on individual symptoms (e.g., erectile dysfunction, gynecomastia, etc.) rather than domain-specific HRQOL outcomes, often without applying a predefined follow-up threshold, with most included studies assessing patients within 5 years of diagnosis [21,22]. Hence, there is a need to explore the long-term HRQOL across its different domains. The purpose of this scoping review is therefore to synthesize and identify key gaps in the available literature on HRQOL in long-term prostate cancer survivors who have been treated with hormone therapy. The clinical relevance of HRQOL in long-term prostate cancer survivors is heightened by the expanding indications for hormone therapy and the increasing life expectancy of affected men, resulting in prolonged exposure to hormone treatment-related adverse effects. Consequently, understanding long-term, domain-specific HRQOL outcomes is essential for balancing hormone treatment benefits against impact on quality of life.
2. Materials and Methods
We used the Preferred Reporting Items for Systematic Reviews and Meta-Analyses extension for Scoping Reviews (PRISMA-ScR) checklist to report the review findings (Supplementary Materials, Table S1) [23]. The protocol for this scoping review is publicly accessible online on the Open Science Framework: https://osf.io/jn5xg [24].
2.1. Information Sources and Search Strategy
The first author (MB) developed a comprehensive search strategy and searched four databases (MEDLINE (OVID), Embase.com, CINAHL (EBSCO), and Cochrane). Tables S2–S5 in the Supplementary Materials provide the search strategy used in the aforementioned databases. The search was limited to articles published in English language, with no restrictions on date or study type. We used MeSH terms and relevant keywords related to prostate cancer, hormone therapy and HRQOL. We reviewed articles published up to 15 April 2025. An expert librarian (AH) at DKFZ reviewed and verified the search strategy. We also scrutinized the reference lists of eligible studies to retrieve potentially relevant publications. All included studies were imported and stored in EndNote, with duplicates removed, where applicable.
2.2. Eligibility Criteria
Study selection criteria for this scoping review are as follows:
Inclusion criteria:
- Peer-reviewed, published original research articles (observational and randomized controlled trials);
- Studies reporting on survivors of prostate cancer who are either at least 5 years post-diagnosis, or have been followed-up for 5 or more years post-diagnosis;
- Studies reporting on the HRQOL of prostate cancer survivors who received hormone therapy, either alone or combined with other treatment modalities.
Exclusion criteria:
- Commentaries, editorials, case reports, case series, conference abstracts, and reviews;
- Non-English articles;
- Studies in which both comparison groups receive some form of hormone therapy, either alone or in combination with other treatment modalities.
2.3. Outcomes
Our primary outcome was general HRQOL. Our secondary outcome was prostate cancer-specific HRQOL and symptom burden.
2.4. Study Selection and Evidence Abstraction
Two reviewers (MB, KY) screened titles and abstracts of all identified records. Subsequently, both reviewers screened full texts of potentially eligible articles, utilizing an a priori developed full-text screening sheet (Supplementary File S1). This process was carried out independently and in duplicate to ensure a thorough and unbiased assessment. Any exclusions made were documented, including the reasons for exclusion. We used the software Rayyan [25] for recording the full texts of screening decisions.
Using standardized extraction forms, both reviewers extracted data from all included records independently and in duplicate. Extracted data included the author’s name, country and year of publication, study design, population, patients’ demographics, sample size, type and duration of hormone treatment, other treatment modalities, HRQOL instrument, covariates and results, if available.
Any disagreements during these stages were resolved through discussion or the help of a third reviewer, who is an expert in the field (MT or VA).
2.5. Risk of Bias Assessment
While not required for scoping reviews, two reviewers (MB, KY) independently assessed the methodological quality and risk of bias of each included observational study, using the Joanna Briggs Institute critical appraisal checklists for observational studies to guide cautious interpretation of the findings [26]. Each question was rated as ‘yes’, ‘no’, ‘unclear’, or ‘not applicable’. We calculated the percentage of ‘yes’ responses for each study, and categorized the risk of bias as follows: low (>80%), moderate (51–80%), and high (≤50%).
2.6. Data Synthesis
Given the variability in outcome measures and years after diagnosis of prostate cancer, we summarized the findings of this review using a descriptive approach, emphasizing common patterns across studies. The results from the included studies were summarized in a table, and grouped by outcomes and corresponding HRQOL instruments.
3. Results
The search strategy identified 15,044 citations. Following duplicate removal, we screened 10,649 citations, of which 128 articles were potentially eligible. From the 128 articles, we retained a total of 14 articles (Figure 1), and excluded 114 articles for the reasons outlined in Table S6 (Supplementary Materials). All of the included articles were observational studies that reported on the HRQOL of prostate cancer survivors. We did not identify any eligible clinical trials.
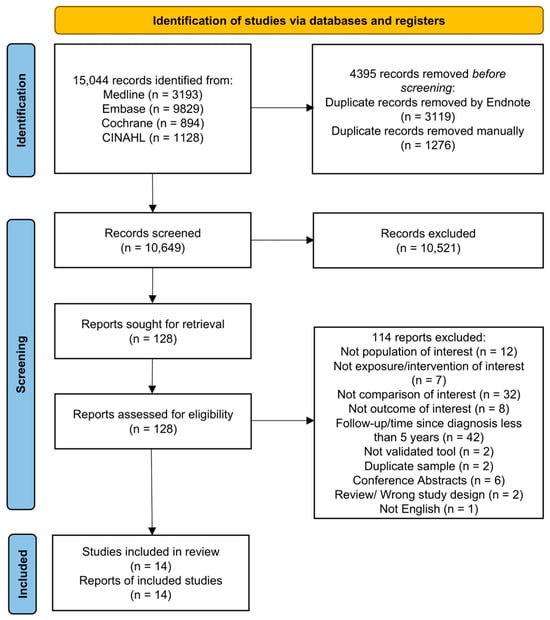
Figure 1.
Flow diagram of articles related to use of hormone therapy and quality of life of prostate cancer survivors [27].
3.1. Description of Included Studies
Table S9 (Supplementary Materials) describes the characteristics of the 14 included observational studies, listed in descending chronological order from 2024 to 2002 [28,29,30,31,32,33,34,35,36,37,38,39,40,41]. These studies reported on general HRQOL (n = 13), prostate cancer-specific HRQOL (n = 12), and other psychological wellbeing issues (n = 2). The exposure was hormone therapy, either alone or in combination with other treatments, compared to one or more other treatment modalities. Nine studies included only ADT in their comparisons [28,30,32,33,35,36,37,38,41], while the remaining studies included any type of hormone therapy, including both classical ADT and anti-androgens [29,31,34,39,40]. The included studies were conducted in Europe (n = 8), North America (n = 5), and Australia (n = 1). Prostate cancer patients were recruited from population-based cancer registries (n = 7), national databases (n = 2), hospitals (n = 1), urology clinics (n = 3), and using a convenience sample (n = 1). The sample size ranged from 143 to 6944. Five studies were cohort, six were cross-sectional, and three were a combination of cross-sectional and case–control designs. Participants were followed from 6 months post-diagnosis up to 19.8 years after end of treatment in cohort studies. In cross-sectional studies, prostate cancer patients/survivors were 5 to 18 years post-diagnosis. In addition to survivors more than 5 years post-diagnosis, three cross-sectional studies also included survivors who were less than 5 years post-diagnosis [29,38,40].
Thirteen studies on general HRQOL used various questionnaires [28,29,30,31,32,33,34,35,37,38,39,40,41] (Table 1). Six of them measured general HRQOL using the European Organization for Research and Treatment of Cancer Core Quality of Life Questionnaire (EORTC QLQ-C30) [31,33,34,35,38,40], while the remaining studies used the 12-Item Short Form Health Survey (SF-12) [28,30,32], the 36-Item Short Form Health Survey (SF-36) [28,37,39], Functional Assessment of Cancer Therapy-General (FACT-G) [29], Quality of Life Cancer Survivors Questionnaire (QoL-CS) [39], or a questionnaire based on SF-12 and published literature [41].

Table 1.
Summary of general HRQOL and symptom burden in the included studies.
Eleven of the 13 studies that reported general HRQOL also reported prostate cancer-specific HRQOL and symptoms, in addition to one other study [28,29,30,32,33,34,35,36,37,38,40,41] (Table 2). Four studies used the European Organization for Research and Treatment of Cancer Prostate Cancer 25 Questionnaire (EORTC QLQ-PR25) [33,34,38,40]. The remaining studies used the Expanded Prostate Cancer Index Composite (EPIC) questionnaire [28,32,35], the University of California Los Angeles Prostate Cancer Index (UCLA PCI) [30,37], the Functional Assessment of Cancer Therapy-General (FACT-P) [29], and the Prostate Cancer Symptom Scale (PCSS)-based questionnaire [36].

Table 2.
Summary of prostate-cancer specific HRQOL in the included studies.
Tables S7 and S8 (Supplementary Materials) provide the detailed assessment of the risk of bias and the overall appraisal for each of the included studies. In the assessment of the cross-sectional studies, six were classified as having a low risk of bias, demonstrating adherence to rigorous methodological standards [31,33,35,36,38,39]. Two studies were assigned a moderate risk of bias, primarily due to their reliance on self-reported treatment modalities and adjustment for age as the only confounder, without accounting for other factors such as cancer stage [40,41]. Another study exhibited a high risk of bias, with significant concerns related to self-reporting of treatment and insufficient control for confounding variables [29]. Regarding the cohort studies, three were classified as having a low risk of bias, meeting robust methodological criteria [28,32,37]. One study was deemed to have a moderate risk of bias, primarily due to loss to follow-up and inadequate strategies to address incomplete follow-up [30]. The final cohort study exhibited a high risk of bias, with notable issues concerning incomplete follow-up and failure to adequately identify and control for confounding variables, significantly compromising the integrity of its findings [34].
3.2. Association of Hormone Therapy with HRQOL
3.2.1. General HRQOL–Functioning
Prostate cancer survivors with various treatment combinations, involving hormone therapy, reported worse HRQOL outcomes, including global health status/QOL, physical functioning, emotional functioning, social functioning, and physical and mental wellbeing, compared to survivors who received local treatment alone (e.g., RP and/or RT) or those who were in active surveillance/watchful waiting. One study compared the HRQOL of prostate cancer survivors who survived 2 to 18 years after diagnosis and received ADT exclusively to those who underwent radical prostatectomy [38]. Similarly, the analysis showed that worse global health status/QOL, physical functioning, role functioning, and social functioning were observed in the ADT group.
Two studies included primary hormone therapy in their comparisons. Punnen et al. reported a decline in physical health 5 years after diagnosis in prostate cancer survivors who received primary ADT compared to nerve-sparing radical prostatectomy (adjusted OR = 1.9, 95% CI [1.1–2.2]; p-value = 0.03) [37]. The second study, by Mols et al., also showed that primary hormone therapy was associated with worse physical functioning (adjusted mean score = 57.4 (31.8)) compared to radical prostatectomy (adjusted mean score = 75.7 (24.4); p-value < 0.05) and other treatment combinations that could also involve hormone therapy (RP ± RT ± HT), in prostate cancer patients who survived 5 to 10 years after diagnosis [39]. Vitality was also worse in the primary hormone therapy group (adjusted mean score = 62.1 (22.4)) compared to radical prostatectomy (adjusted mean score = 70.9 (20.8); p < 0.05) and other treatment combinations involving hormone therapy. Moreover, the authors of that study assessed generic HRQOL survival issues using QOL-CS, and also observed worse physical and psychological well-being in prostate cancer survivors who received primary hormone therapy.
Finally, one study compared medical to surgical castration and reported a lower mental score with time in the medical castration group [30]. Another study assessed the effect of castration, either medical or surgical, on the HRQOL of prostate cancer survivors 7 to 8 years after prostatectomy. Survivors in the castration group reported greater prostate cancer- and treatment-related concerns, including body image concerns, and worries about cancer and death, and had lower scores in general health, mental health and activity index [41].
Only one study showed no difference in the general HRQOL of survivors of localized prostate cancer who received ADT compared to those who did not, 10 years after diagnosis [35].
3.2.2. General HRQOL—Symptom Burden
Prostate cancer survivors who received hormone therapy also reported higher burden of fatigue, dyspnea, nausea and vomiting, insomnia, diarrhea, appetite loss, and financial difficulties (Table 1).
3.2.3. Prostate Cancer-Specific HRQOL
Prostate cancer survivors who received hormone therapy with various other combinations had worse bowel symptoms and function, hormone treatment-related symptoms and function, urinary bother, particularly burning on urination, and sexual function than survivors who received no hormone therapy or who were observed only (Table 2). Survivors 2 to 18 years after diagnosis who received ADT exclusively reported less sexual activity and more ADT-related symptoms, compared with those who had radical prostatectomy [38]. One study reported worse urinary incontinence, urinary and bowel bother, and sexual function in survivors who received ADT, with or without external beam radiation therapy, at both 10 and 15 years post-diagnosis, compared to age-matched controls [32]. Particularly, urinary incontinence and urinary bother worsened between year 10 and year 15, whereas sexual function showed improvement during the same period. Similarly, another study compared ADT as a single therapy and in combination with either radiotherapy or radical prostatectomy, or both, with age-matched controls [36]. Survivors who received ADT alone reported more erectile dysfunction compared with controls (adjusted OR = 2.32, 95% CI [1.33–4.05]). In addition, survivors who received ADT in combination with other forms of therapy had worse erectile and bowel dysfunction and urinary urgency and incontinence. The only study comparing primary hormone therapy to radical prostatectomy showed a decline in bowel function (adjusted OR = 2, 95% CI [1.1–3.6]; p-value = 0.03) and bother (adjusted OR = 2, 95% CI [1.1–3.8]; p-value = 0.03) 5 years after diagnosis [37].
In addition, one study showed that medical castration was associated with worse urinary and hormone function, and bowel bother, compared to surgical castration [30]. Finally, one study reported on self-treatment side effects of castration in prostate cancer survivors 7 to 8 years after prostatectomy. Survivors in the castration group reported worse sexual function and significantly fewer days with sexual drive [41].
3.2.4. Psychological Well-Being Issues
Evidence on psychological outcomes is limited, as only two studies explored psychological issues related to prostate cancer survivors on hormone treatment [29,35]. One used a cross-sectional design, and one was a combination of cross-sectional and case–control designs. There was substantial heterogeneity across these studies in terms of measurement tools and the psychological outcomes evaluated. The authors found no differences in depression, anxiety, or psychological flexibility between long-term prostate cancer survivors who underwent hormone therapy and those who did not. However, one study showed that prostate cancer patients who survived an average of 5 years and who received any form of hormone therapy reported lower masculine self-esteem compared with those who did not receive any (Table 3).

Table 3.
Summary of psychological wellbeing issues in the included studies.
4. Discussion
Hormone therapy in prostate cancer is associated with worse general and prostate cancer-specific HRQOL outcomes in long-term survivors, i.e., 5 and more years after diagnosis/treatment. Studies using validated questionnaires revealed that survivors who underwent hormone therapy reported worse global health status and physical, emotional, and social functioning compared to those treated with local therapies like radical prostatectomy or radiation. These survivors also experienced greater symptom burdens, alongside worse vitality and mental health. Prostate cancer-specific issues, such as bowel and urinary bother and sexual dysfunction, were also more pronounced in hormone therapy recipients. While psychological well-being outcomes like depression and anxiety showed no differences according to hormone therapy status, lower masculine self-esteem was noted in survivors on hormone therapy.
This review builds upon previous literature examining hormone therapy and HRQOL in prostate cancer patients by specifically focusing on long-term survivors. While earlier studies have reported that ADT is associated with reduced HRQOL, alongside increased emotional and cognitive impairment, depressive symptoms, and fatigue, they often did not distinguish between short- and long-term survivors [42,43]. Despite evidence that newer antiandrogens can delay HRQOL deterioration and reduced pain progression, they also reported increased fatigue and hot flushes [43,44]. However, emotional well-being was generally unaffected [44]. Our findings align with some of these results, but provide a more detailed perspective on HRQOL in long-term survivors.
To our knowledge, our scoping review is the first to systematically map domain-specific HRQOL outcomes in long-term prostate cancer survivors who received hormone therapy, using a predefined follow-up threshold (≥5 years after diagnosis). Unlike previous studies, which primarily focused on early survivorship [43], our approach enables a clearer understanding of which specific dimensions of HRQOL remain affected in long-term survivors who received hormone therapy, thus providing clinicians with a domain-specific overview of persistent HRQOL impairments.
Due to the scarcity of longitudinal studies that have tracked prostate cancer survivors for more than 5 years post-diagnosis, only a limited number of studies were eligible for inclusion in this review. Most of included studies focused on prostate cancer survivors up to 15 years post-diagnosis, with little evidence available on HRQOL beyond 15 years. Additionally, the majority of studies in the existing literature have explored primarily the effect of local treatment options, like radical prostatectomy and radiation therapy, with little focus on hormone therapy. Moreover, the variation in how prostate cancer stage was reported and adjusted for across studies highlights an important methodological gap. While some studies specified disease stage [28,30,32,33,34,35,36,37] or adjusted for it as a covariate [38,39], others included all stages without adjustment [29,40] or did not report stage at all [31,41]. When comparing studies that did not report or adjust for stage with those that did, studies adjusting for stage reported similar results but showed additional burden in social functioning, appetite loss, psychological wellbeing, vitality, and bowel and urinary symptoms. This inconsistency in accounting for a key clinical characteristic like stage limits the ability to draw firm conclusions regarding the independent impact of disease stage on long-term HRQOL and underscores the need for more standardized reporting in future research. Furthermore, there is a lack of detailed information regarding hormone therapy regimens and their duration, and whether they were administered intermittently or continuously, which further complicates the analysis. A systematic review and meta-analysis comparing intermittent to continuous ADT showed that most included trials reported an improvement in physical and sexual functioning with intermittent therapy [45]. Contrasting results were highlighted in a review by Luo and collaborators, which found different effects on HRQOL and symptoms burden among recipients of the new generation of antiandrogens [43]. Therefore, this review cannot determine whether particular hormone therapy regimens or treatment durations are associated with differential long-term HRQOL effects. In addition, the disparity in HRQOL measurement tools and differences in follow-up duration introduced further challenges to the analysis. On the other hand, using a range of HRQOL questionnaires captures the full spectrum of relevant domains, enhancing generalizability by incorporating aspects assessed in some questionnaires but absent in others. In our review, we included only two studies that specifically examined primary hormone therapy [37,39], whereas the others involved combined treatment modalities, which complicates further the comparison across studies. While hormone therapy is a crucial treatment for prostate cancer, its use varies based on stage, patient characteristics and treatment goals [46]. Primary hormone therapy is often used as the main treatment for patients who are not candidates for local therapy, typically in advanced diseases, and its goal is systemic control and symptom management [46]. However, hormone therapy in combination with surgery or radiation therapy is usually administered to target microscopic residual disease [46]. Patients receiving adjuvant or neoadjuvant hormone therapy often have earlier-stage disease but adverse pathological features [46]. These differences highlight the importance of tailoring comparisons to specific patients’ characteristics and treatment goals.
5. Conclusions
By applying a scoping methodology, we were able to provide a structured overview of methodological challenges, which provides a clear agenda for future research studies. To summarize, the findings of this review highlight the importance of discussing HRQOL compromises with patients who consider hormone therapy for prostate cancer. These discussions should consider cancer stage and clinical characteristics, as they can influence the decision to initiate hormone therapy and its impact on HRQOL. Future research studies should also prioritize longitudinal designs to evaluate the trajectory of HRQOL changes over time in long-term prostate cancer survivors and explore interventions to improve HRQOL for recipients of hormone therapy.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/curroncol33030137/s1, Table S1. Preferred Reporting Items for Systematic reviews and Meta-Analyses extension for Scoping Reviews (PRISMA-ScR) Checklist. Table S2. Medline Search Strategy. Table S3. Embase Search Strategy. Table S4. Cochrane Search Strategy. Table S5. CINAHL Search Strategy. Table S6. Reasons for exclusion. Table S7. Risk of bias and quality assessment of included cross-sectional studies using the Joanna Briggs Institute (JBI) critical appraisal tool. Table S8. Risk of bias and quality assessment of included cohort studies using the Joanna Briggs Institute (JBI) critical appraisal tool. Table S9. Characteristics of the included studies. Supplementary File S1. Full-text screening form.
Author Contributions
Conceptualization, M.B., V.A. and M.S.Y.T.; methodology, M.B., V.A. and M.S.Y.T.; formal analysis, M.B. and K.Y.; investigation, M.B. and K.Y.; data curation, M.B. and K.Y.; writing—original draft preparation, M.B.; writing—review and editing, M.B., K.Y., V.A. and M.S.Y.T.; visualization, M.B. All authors have read and agreed to the published version of the manuscript.
Funding
M.B. is supported by the DKFZ International PhD Program Scholarship.
Data Availability Statement
No new data were created in this study.
Acknowledgments
The authors would like to thank Andrea Heppert, medical librarian at DKFZ, for her professional assistance in conducting the literature search for this review.
Conflicts of Interest
Dr. Arndt is the corresponding author and the principal investigator of one of the studies included in this review. The remaining authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| AA | Antiandrogens |
| ADT | Androgen Deprivation Therapy |
| AS | Active Surveillance |
| BT | Brachytherapy |
| CI | Confidence Interval |
| CompACT | Comprehensive Assessment of Acceptance and Commitment Therapy |
| DASS-21 | Depression, Anxiety, and Stress Scale-21 items |
| EBRT | External Beam Radiation Therapy |
| EORTC QLQ-C30 | European Organization for Research and Treatment of Cancer Quality of Life-Core 30 |
| EORTC QLQ-PR25 | European Organization for Research and Treatment of Cancer Quality of Life-Prostate 25 |
| EPIC | Expanded Prostate Cancer Index Composite |
| FACT-G | Functional Assessment of Cancer Therapy-General |
| FACT-P | Functional Assessment of Cancer Therapy-Prostate |
| HADS | Hospital Anxiety and Depression Scale |
| HRQOL | Health-Related Quality of Life |
| HT | Hormone Therapy |
| IQR | Interquartile Range |
| LHRH | Luteinizing Hormone Releasing Hormone |
| MeSH | Medical Subject Heading |
| MSE | Masculine Self Esteem |
| NSRP | Nerve-Sparing Radical Prostatectomy |
| NTX | No Treatment Reported |
| OR | Odds Ratio |
| PC | Prostate Cancer |
| PCSS | Prostate Cancer Symptom Scale |
| PRISMA-ScR | Preferred Reporting Items for Systematic Reviews and Meta-Analyses extension for Scoping Reviews |
| PSA | Prostate Specific Antigen |
| QOL | Quality of Life |
| QoL-CS | Quality of Life-Cancer Survivors |
| RP | Radical Prostatectomy |
| RT | Radiation Therapy |
| SD | Standard Deviation |
| SF-12 | The 12-Item Short Form Health Survey |
| SF-36 | The 36-Item Short Form Health Survey |
| TX | Treatment |
| UCLA PCI | University of California Los Angeles Prostate Cancer Index |
| WW | Watchful Waiting |
References
- Vieira Lima Aguiar Melão, B.; Pekala, K.R.; Matsoukas, K.; Bratt, O.; Carlsson, S.V. SIU-ICUD: Epidemiology of prostate cancer. Soc. Int. Urol. J. 2025, 6, 44. [Google Scholar] [CrossRef]
- Ferlay, J.; Ervik, M.; Lam, F.; Laversanne, M.; Colombet, M.; Mery, L.; Piñeros, M.; Znaor, A.; Soerjomataram, I.; Bray, F. Global Cancer Observatory: Cancer Today (Version 1.1); International Agency for Research on Cancer: Lyon, France, 2024; Available online: https://gco.iarc.who.int/today (accessed on 15 September 2025).
- James, N.D.; Tannock, I.; N’Dow, J.; Feng, F.; Gillessen, S.; Ali, S.A.; Trujillo, B.; Al-Lazikani, B.; Attard, G.; Bray, F.; et al. The Lancet Commission on prostate cancer: Planning for the surge in cases. Lancet 2024, 403, 1683–1722. [Google Scholar] [CrossRef]
- Cancer Trends Progress Report, National Cancer Institute, NIH, DHHS, Bethesda, MD. Available online: https://progressreport.cancer.gov (accessed on 15 September 2025).
- Schafer, E.J.; Laversanne, M.; Sung, H.; Soerjomataram, I.; Briganti, A.; Dahut, W.; Bray, F.; Jemal, A. Recent patterns and trends in global prostate cancer incidence and mortality: An update. Eur. Urol. 2025, 87, 302–313. [Google Scholar] [CrossRef]
- Culp, M.B.; Soerjomataram, I.; Efstathiou, J.A.; Bray, F.; Jemal, A. Recent Global Patterns in Prostate Cancer Incidence and Mortality Rates. Eur. Urol. 2020, 77, 38–52. [Google Scholar] [CrossRef]
- Miller, K.D.; Nogueira, L.; Devasia, T.; Mariotto, A.B.; Yabroff, K.R.; Jemal, A.; Kramer, J.; Siegel, R.L. Cancer treatment and survivorship statistics, 2022. CA Cancer J. Clin. 2022, 72, 409–436. [Google Scholar] [CrossRef]
- American Cancer Society Treating Prostate Cancer. Available online: https://www.cancer.org/cancer/types/prostate-cancer/treating.html (accessed on 25 September 2025).
- American Cancer Society Hormone Therapy for Prostate Cancer. Available online: https://www.cancer.org/cancer/types/prostate-cancer/treating/hormone-therapy.html (accessed on 25 September 2025).
- Huggins, C.; Hodges, C.V. Studies on prostatic cancer. I. The effect of castration, of estrogen and androgen injection on serum phosphatases in metastatic carcinoma of the prostate. CA Cancer J. Clin. 1972, 22, 232–240. [Google Scholar] [CrossRef]
- Damber, J.E. Endocrine therapy for prostate cancer. Acta Oncol. 2005, 44, 605–609. [Google Scholar] [CrossRef]
- Parker, C.; Gillessen, S.; Heidenreich, A.; Horwich, A.; Committee, E.G. Cancer of the prostate: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2015, 26, v69–v77. [Google Scholar] [CrossRef] [PubMed]
- Cooperberg, M.R.; Grossfeld, G.D.; Lubeck, D.P.; Carroll, P.R. National practice patterns and time trends in androgen ablation for localized prostate cancer. J. Natl. Cancer Inst. 2003, 95, 981–989. [Google Scholar] [CrossRef] [PubMed]
- Pound, C.R.; Partin, A.W.; Eisenberger, M.A.; Chan, D.W.; Pearson, J.D.; Walsh, P.C. Natural history of progression after PSA elevation following radical prostatectomy. JAMA 1999, 281, 1591–1597. [Google Scholar] [CrossRef]
- Mohile, S.G.; Mustian, K.; Bylow, K.; Hall, W.; Dale, W. Management of complications of androgen deprivation therapy in the older man. Crit. Rev. Oncol. Hematol. 2009, 70, 235–255. [Google Scholar] [CrossRef][Green Version]
- Jacobsen, P.B.; Nipp, R.D.; Ganz, P.A. Addressing the Survivorship Care Needs of Patients Receiving Extended Cancer Treatment. Am. Soc. Clin. Oncol. Educ. Book 2017, 37, 674–683. [Google Scholar] [CrossRef] [PubMed]
- Alibhai, S.; Breunis, H.; Timilshina, N.; Naglie, G.; Tannock, I.; Krahn, M.; Warde, P.; Fleshner, N.E.; Canning, S.D.; Tomlinson, G. Long-term impact of androgen-deprivation therapy on physical function and quality of life. Cancer 2015, 121, 2350–2357. [Google Scholar] [CrossRef] [PubMed]
- Alibhai, S.M.; Breunis, H.; Timilshina, N.; Marzouk, S.; Stewart, D.; Tannock, I.; Naglie, G.; Tomlinson, G.; Fleshner, N.; Krahn, M.; et al. Impact of androgen-deprivation therapy on cognitive function in men with nonmetastatic prostate cancer. J. Clin. Oncol. 2010, 28, 5030–5037. [Google Scholar] [CrossRef]
- O’Sullivan, B.; Brierley, J.; D’Cruz, A.; Fey, M.; Pollock, R.E.; Vermorken, J.; Huang, S.H. UICC Manual of Clinical Oncology; Wiley Online Library: Hoboken, NJ, USA, 2015; Volume 267. [Google Scholar]
- Cappuccio, F.; Buonerba, C.; Scafuri, L.; Di Trolio, R.; Dolce, P.; Trabucco, S.O.; Erbetta, F.; Tulimieri, E.; Sciscio, A.; Ingenito, C. Study on the impact of hormone therapy for prostate cancer on the quality of life and the psycho-relational sphere of patients: ProQoL. Oncol. Ther. 2025, 13, 233–249. [Google Scholar] [CrossRef]
- Shahzad, M.; Basharat, A.; Mujtaba, G.; Khalid, M.F.; Dave, P.; Naeem, A.; Warraich, S.Z.; Jaglal, M.V. Comparing quality of life in prostate cancer patients: Continuous vs. intermittent androgen deprivation therapy—A systematic review and meta-analysis. J. Clin. Oncol. 2025, 43, 117. [Google Scholar] [CrossRef]
- Osanto, S.; van de Vliert-Bout, A.-L.; Gomez de Segura, C.A.; Efficace, F.; Sparano, F.; Willemse, P.P.; Schoones, J.; Cohen, A.; van Waalwijk van Doorn-Khosrovani, S.B. Health-related quality of life outcomes in randomized controlled trials in metastatic hormone-sensitive prostate cancer: A systematic review. EClinicalMedicine 2024, 78, 102914. [Google Scholar] [CrossRef]
- Tricco, A.C.; Lillie, E.; Zarin, W.; O’Brien, K.K.; Colquhoun, H.; Levac, D.; Moher, D.; Peters, M.D.J.; Horsley, T.; Weeks, L.; et al. PRISMA Extension for Scoping Reviews (PRISMA-ScR): Checklist and Explanation. Ann. Intern. Med. 2018, 169, 467–473. [Google Scholar] [CrossRef]
- Basbous, M. Health-related Quality of Life in Prostate Cancer Survivors Who Underwent Hormone Therapy: A Scoping Review. OSF. 2024. Available online: https://osf.io/jn5xg/ (accessed on 4 September 2024).
- Ouzzani, M.; Hammady, H.; Fedorowicz, Z.; Elmagarmid, A. Rayyan—A web and mobile app for systematic reviews. Syst. Rev. 2016, 5, 210. [Google Scholar] [CrossRef]
- Aromataris, E.; Lockwood, C.; Porritt, K.; Pilla, B.; Jordan, Z. (Eds.) Systematic reviews of etiology and risk. JBI Man. Evid. Synth. 2024, 1, 293–308. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Al Hussein Al Awamlh, B.; Wallis, C.J.D.; Penson, D.F.; Huang, L.C.; Zhao, Z.; Conwill, R.; Talwar, R.; Morgans, A.K.; Goodman, M.; Hamilton, A.S.; et al. Functional Outcomes after Localized Prostate Cancer Treatment. JAMA 2024, 331, 302–317. [Google Scholar] [CrossRef]
- Chowdhury, E.; Horrocks, T.; McAteer, G.; Gillanders, D. Examining the impact of androgen deprivation therapy, masculine self-esteem, and psychological flexibility on distress and quality of life in men with prostate cancer. Psycho-Oncology 2024, 33, e6277. [Google Scholar] [CrossRef]
- Gaither, T.W.; Kwan, L.; Villatoro, J.; Litwin, M.S. Quality of life in low-income men after surgical castration for metastatic prostate cancer. Urol. Oncol. 2022, 40, 343.e7–343.e14. [Google Scholar] [CrossRef] [PubMed]
- Jackson, M.D.; Walker, E.; Tulloch-Reid, M.K. Health-related quality of life in long-term Caribbean prostate cancer survivors: Comparisons with prostate cancer-free men. Qual. Life Res. 2022, 31, 3391–3401. [Google Scholar] [CrossRef]
- Mazariego, C.G.; Egger, S.; King, M.T.; Juraskova, I.; Woo, H.; Berry, M.; Armstrong, B.K.; Smith, D.P. Fifteen year quality of life outcomes in men with localised prostate cancer: Population based Australian prospective study. BMJ 2020, 371, m3503. [Google Scholar] [CrossRef] [PubMed]
- Adam, S.; Koch-Gallenkamp, L.; Bertram, H.; Eberle, A.; Holleczek, B.; Pritzkuleit, R.; Waldeyer-Sauerland, M.; Waldmann, A.; Zeissig, S.R.; Rohrmann, S.; et al. Health-related quality of life in long-term survivors with localised prostate cancer by therapy-Results from a population-based study. Eur. J. Cancer Care 2019, 28, e13076. [Google Scholar] [CrossRef]
- Hammerer, P.G.; Wirth, M.P. Health-related quality of life in 536 long-Term prostate cancer survivors after treatment with leuprorelin acetate: A combined retrospective and prospective analysis. Urol. Int. 2018, 100, 72–78. [Google Scholar] [CrossRef] [PubMed]
- Kerleau, C.; Guizard, A.V.; Daubisse-Marliac, L.; Heutte, N.; Mercier, M.; Grosclaude, P.; Joly, F. Long-term quality of life among localised prostate cancer survivors: QALIPRO population-based study. Eur. J. Cancer 2016, 63, 143–153. [Google Scholar] [CrossRef]
- Carlsson, S.; Drevin, L.; Loeb, S.; Widmark, A.; Lissbrant, I.F.; Robinson, D.; Johansson, E.; Stattin, P.; Fransson, P. Population-based study of long-term functional outcomes after prostate cancer treatment. BJU Int. 2016, 117, E36–E45. [Google Scholar] [CrossRef]
- Punnen, S.; Cowan, J.E.; Chan, J.M.; Carroll, P.R.; Cooperberg, M.R. Long-term health-related quality of life after primary treatment for localized prostate cancer: Results from the CaPSURE registry. Eur. Urol. 2015, 68, 600–608. [Google Scholar] [CrossRef] [PubMed]
- Drummond, F.J.; Kinnear, H.; O’Leary, E.; Donnelly; Gavin, A.; Sharp, L. Long-term health-related quality of life of prostate cancer survivors varies by primary treatment. Results from the PiCTure (Prostate Cancer Treatment, your experience) study. J. Cancer Surviv. 2015, 9, 361–372. [Google Scholar] [CrossRef]
- Mols, F.; van de Poll-Franse, L.V.; Vingerhoets, A.J.; Hendrikx, A.; Aaronson, N.K.; Houterman, S.; Coebergh, J.W.; Essink-Bot, M.L. Long-term quality of life among Dutch prostate cancer survivors: Results of a population-based study. Cancer 2006, 107, 2186–2196. [Google Scholar] [CrossRef]
- Voerman, B.; Fischer, M.; Visser, A.; Garssen, B.; Van Andel, G.; Bensing, J. Health-related quality of life in Dutch men with prostate cancer. J. Psychosoc. Oncol. 2006, 24, 49–64. [Google Scholar] [CrossRef]
- Fowler, F.J., Jr.; Collins, M.M.; Corkery, E.W.; Elliott, D.B.; Barry, M.J. The impact of androgen deprivation on quality of life after radical prostatectomy for prostate carcinoma. Cancer 2002, 95, 287–295. [Google Scholar] [CrossRef] [PubMed]
- Tucci, M.; Leone, G.; Buttigliero, C.; Zichi, C.; Di Stefano, R.F.; Pignataro, D.; Vignani, F.; Scagliotti, G.V.; Di Maio, M. Hormonal treatment and quality of life of prostate cancer patients: New evidence. Minerva Urol. Nefrol. 2018, 70, 144–151. [Google Scholar] [CrossRef] [PubMed]
- Luo, J.; Graff, J.N. Impact of enzalutamide on patient-related outcomes in metastatic castration-resistant prostate cancer: Current perspectives. Res. Rep. Urol. 2016, 8, 217–224. [Google Scholar] [CrossRef]
- Afferi, L.; Longoni, M.; Moschini, M.; Gandaglia, G.; Morgans, A.K.; Cathomas, R.; Mattei, A.; Breda, A.; Scarpa, R.M.; Papalia, R.; et al. Health-related quality of life in patients with metastatic hormone-sensitive prostate cancer treated with androgen receptor signaling inhibitors: The role of combination treatment therapy. Prostate Cancer Prostatic Dis. 2023, 27, 173–182. [Google Scholar] [CrossRef]
- Magnan, S.; Zarychanski, R.; Pilote, L.; Bernier, L.; Shemilt, M.; Vigneault, E.; Fradet, V.; Turgeon, A.F. Intermittent vs. Continuous Androgen Deprivation Therapy for Prostate Cancer: A Systematic Review and Meta-analysis. JAMA Oncol. 2015, 1, 1261–1269. [Google Scholar] [CrossRef]
- NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®): Prostate Cancer. Version 2. 2025. Available online: www.nccn.org (accessed on 15 September 2025).
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.