Updated Guidelines for the Diagnosis and Treatment of Endometrial Carcinoma: The Polish Society of Gynecological Oncology (2025v)
Abstract
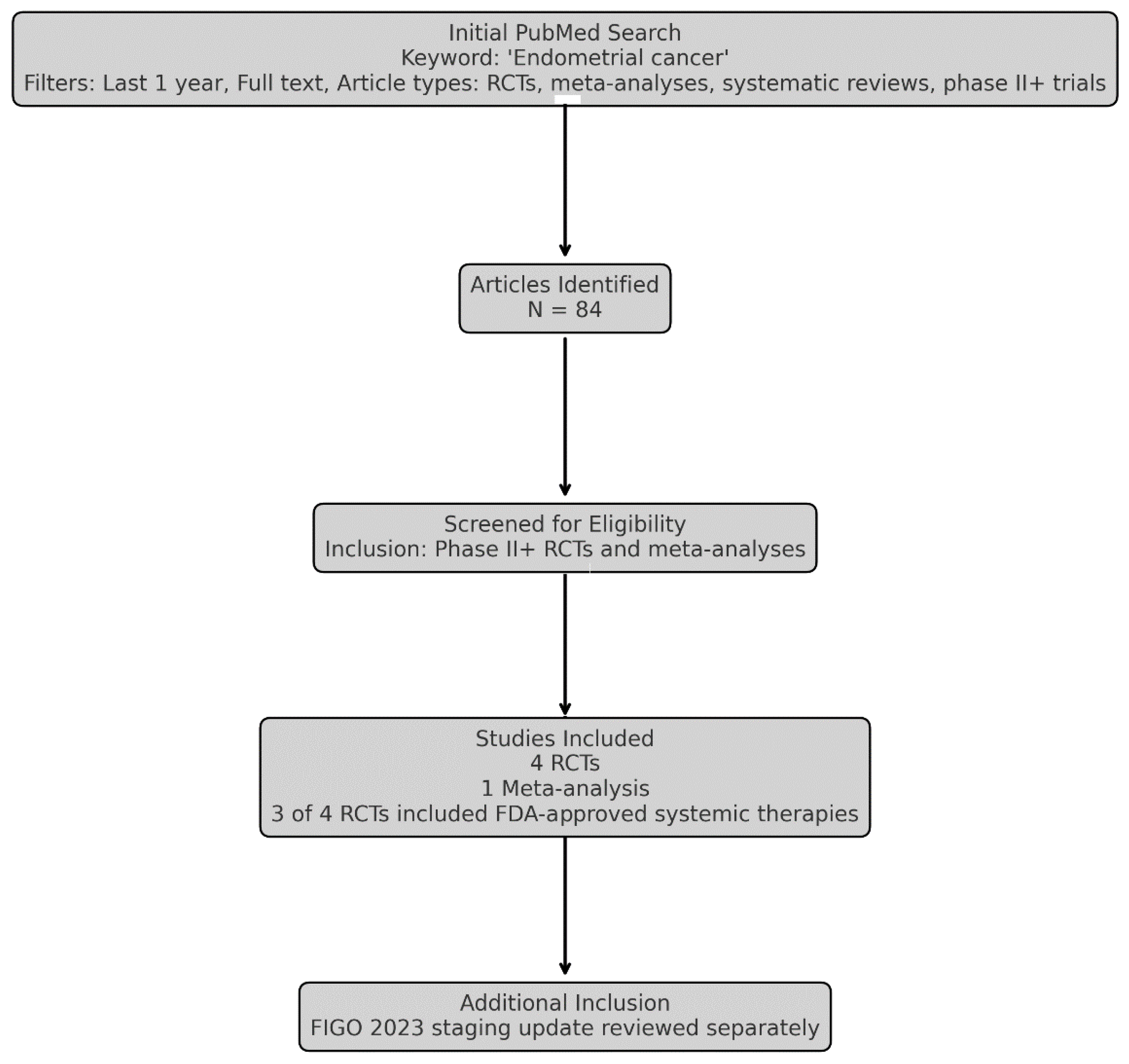
1. Background and Methodology
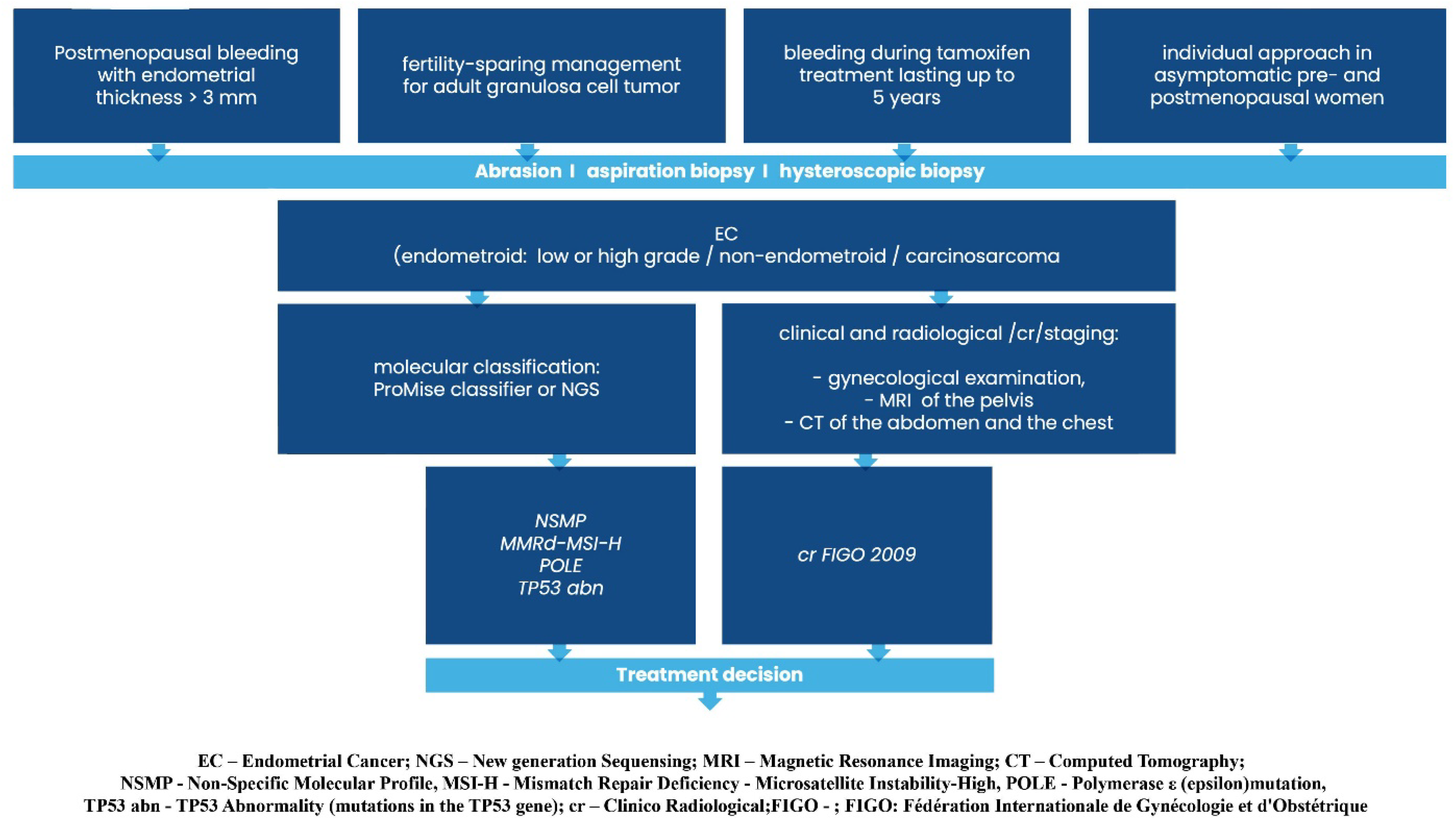
2. Key Findings
2.1. Comparison of FIGO 2023 and 2009
Expert Opinion
2.2. Newly Published RCTs & Meta-Analysis
2.2.1. RUBI (antyPD1)
European Medicines Agency (EMA) Registration
2.2.2. GY018/KEYNOTE-868 Trial (antyPD1)
European Medicines Agency (EMA) Registration:
2.2.3. ATtEnd Trial
European Medicines Agency (EMA) Registration
2.2.4. DUO-E (antyPD-L1)
European Medicines Agency (EMA) Registration
2.2.5. Review of Phase III Trials on Immunotherapy
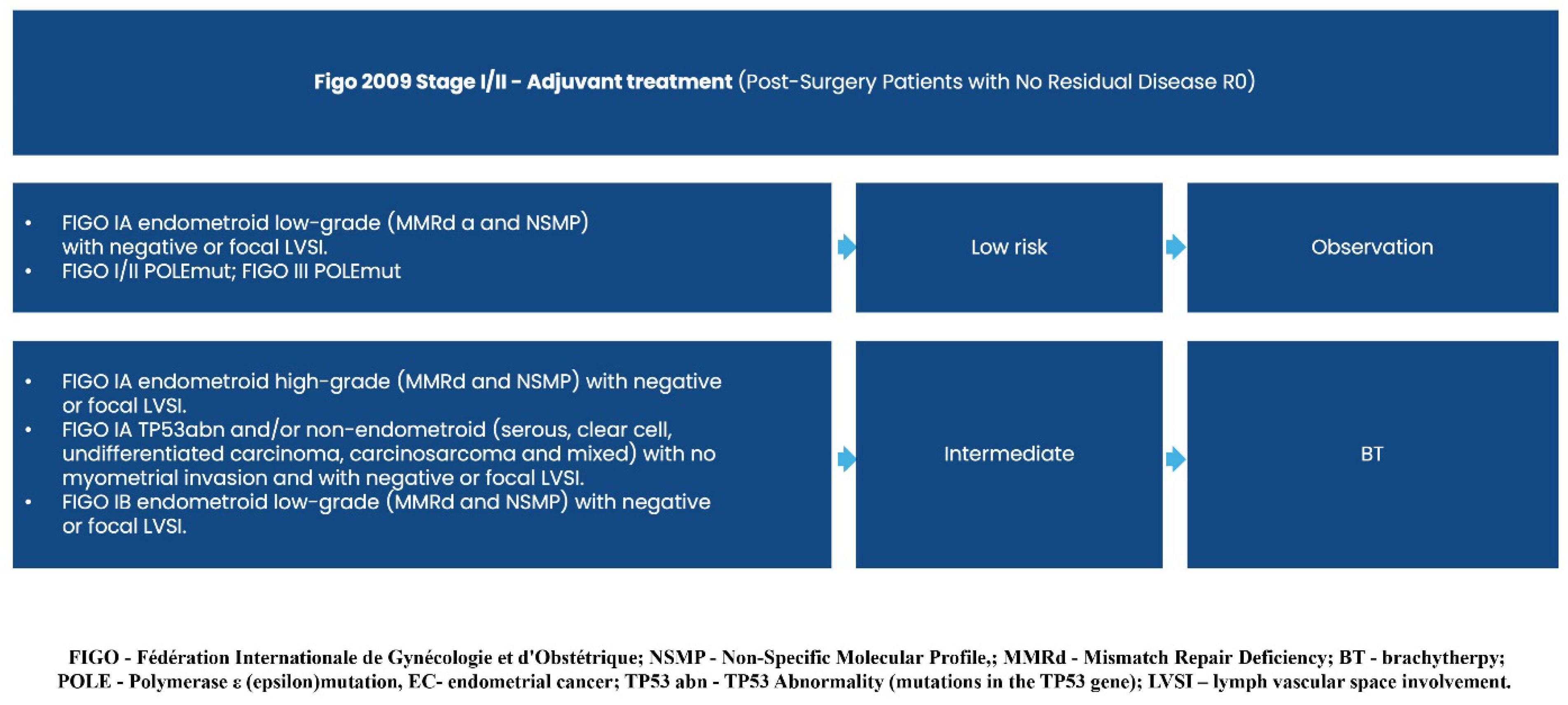
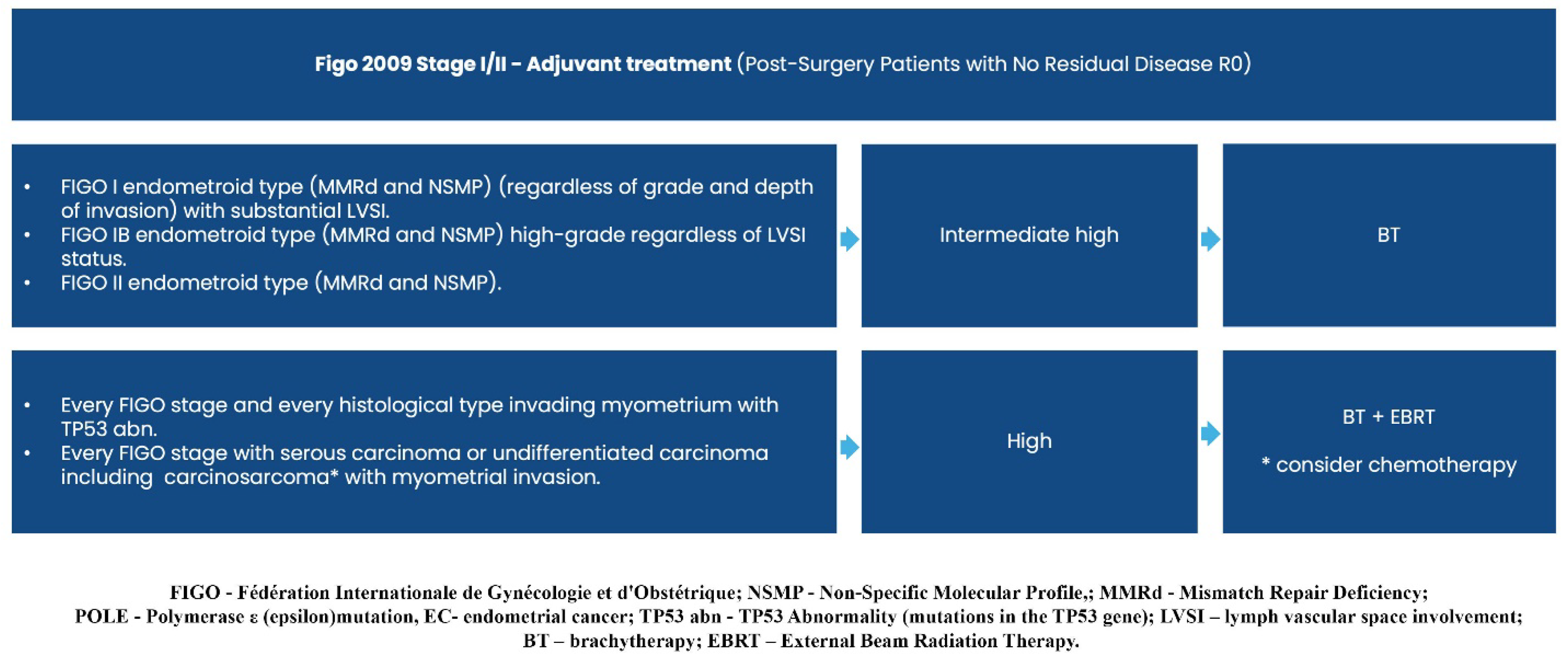
3. How Does New Evidence Contribute to Updates in Treatment Guidelines?
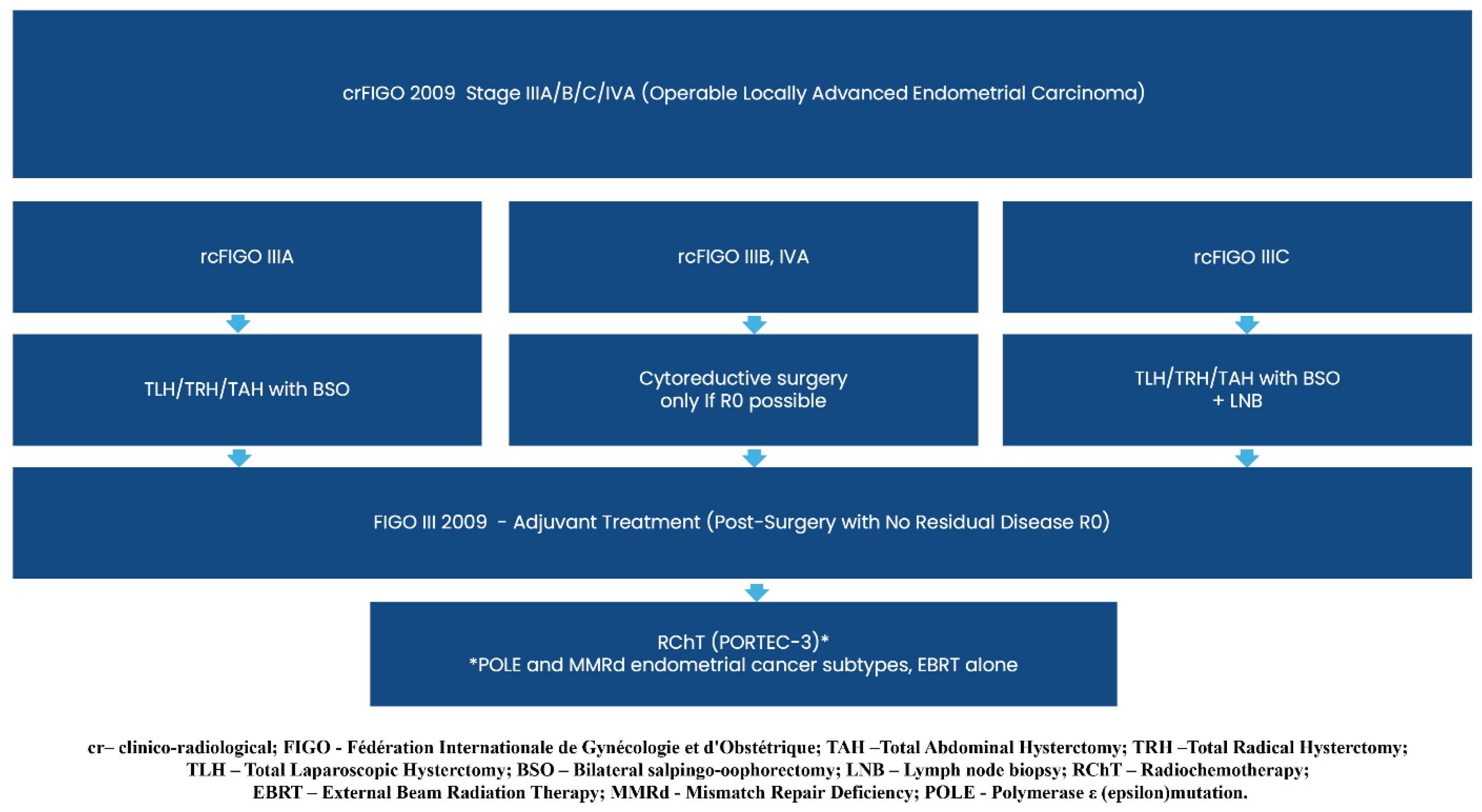
3.1. First Line Systemic Treatment Changes
3.2. Second-Line Systemic Treatment Changes
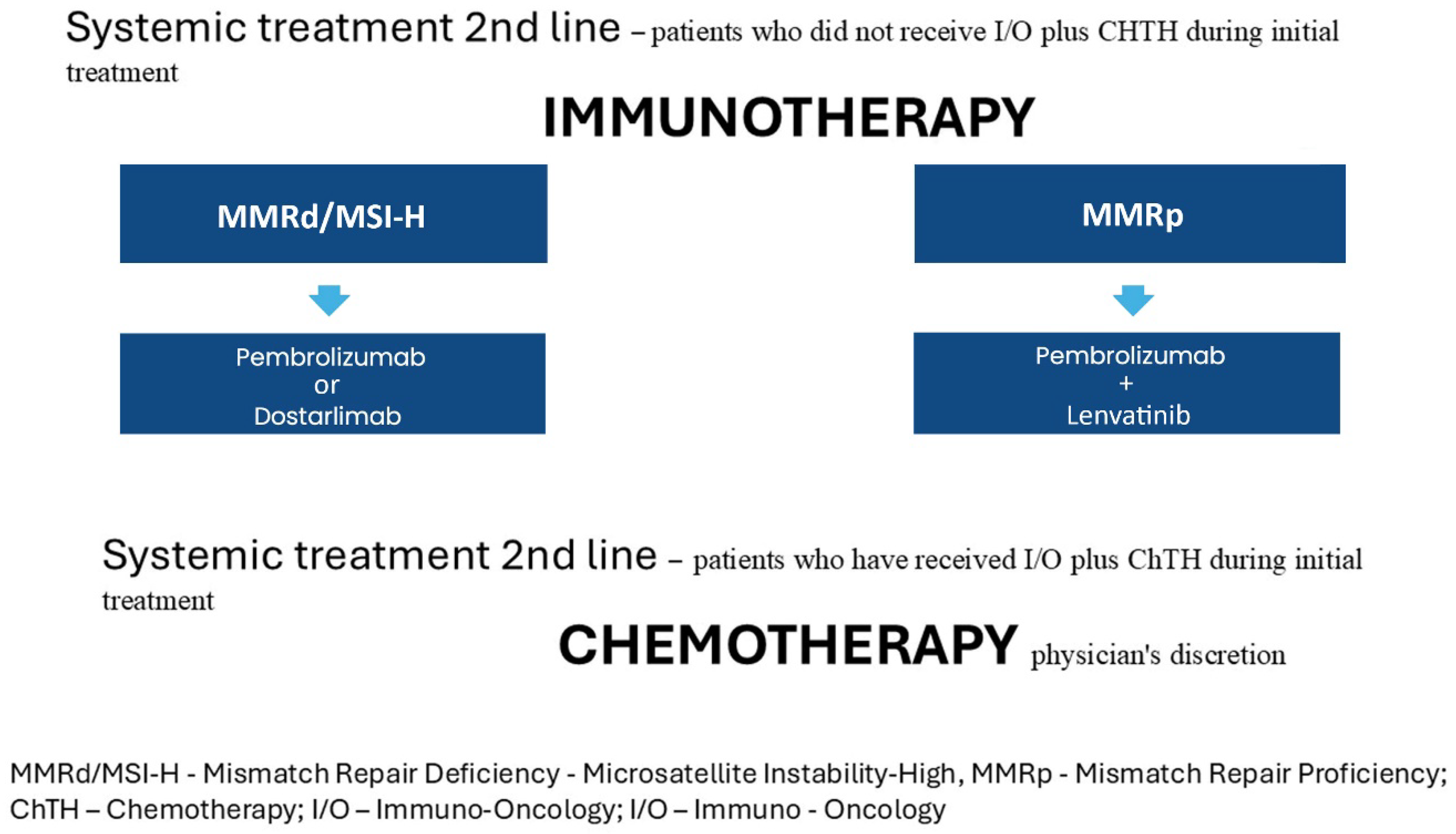
3.2.1. For Patients Who Progressed After at Least One Cycle of Platinum-Based Chemotherapy and Did Not Receive I/O Plus CHTH During Initial Treatment, the Recommendations Remain Unchanged from 2023v1 as Outlined Below
3.2.2. For Patients Who Received I/O Plus Chemotherapy as Initial Treatment, the Options for Second-Line Systemic Therapy Remain Unclear and Are Currently Determined by the Treating Physician’s Discretion
4. Conclusions: Key Changes from 2023v1 to 2025v1
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sznurkowski, J.J.; Rys, J.; Kowalik, A.; Zolciak-Siwinska, A.; Bodnar, L.; Chudecka-Glaz, A.; Blecharz, P.; Zielinska, A.; Marszalek, A.; Bidzinski, M.; et al. The Polish Society of Gynecological Oncology Guidelines for the Diagnosis and Treatment of Endometrial Carcinoma. J. Clin. Med. 2023, 12, 1480. [Google Scholar] [CrossRef] [PubMed]
- Berek, J.S.; Matias-Guiu, X.; Creutzberg, C.; Fotopoulou, C.; Gaffney, D.; Kehoe, S.; Lindemann, K.; Mutch, D.; Concin, N.; Endometrial Cancer Staging Subcommittee, FIGO Women’s Cancer Committee. FIGO staging of endometrial cancer: 2023. Int. J. Gynaecol. Obstet. 2023, 162, 383–394. [Google Scholar] [CrossRef] [PubMed]
- Pecorelli, S. Revised FIGO staging for carcinoma of the vulva, cervix, and endometrium. Int. J. Gynaecol. Obstet. 2009, 105, 103–104. [Google Scholar] [CrossRef] [PubMed]
- McCluggage, W.G.; Bosse, T.; Gilks, C.B.; Howitt, B.E.; McAlpine, J.N.; Nucci, M.R.; Rabban, J.T.; Singh, N.; Talia, K.L.; Parra-Herran, C. FIGO 2023 endometrial cancer staging: Too much, too soon? Int. J. Gynecol. Cancer. 2024, 34, 138–143. [Google Scholar] [CrossRef] [PubMed]
- Zannoni, G.F.; Santoro, A.; Arciuolo, D.; Travaglino, A.; Angelico, G.; Bragantini, E.; Rocco, E.G.; Inzani, F.; Pesci, A.; Troncone, G.; et al. Endometrial cancer and 2023 FIGO staging system: Not too soon, but maybe too much? Gynecol. Oncol. 2024, 189, 98–100. [Google Scholar] [CrossRef] [PubMed]
- Mirza, M.R.; Chase, D.M.; Slomovitz, B.M.; dePont Christensen, R.; Novák, Z.; Black, D.; Gilbert, L.; Sharma, S.; Valabrega, G.; Landrum, L.M.; et al. Dostarlimab for Primary Advanced or Recurrent Endometrial Cancer. N. Engl. J. Med. 2023, 388, 2145–2158. [Google Scholar] [CrossRef] [PubMed]
- Powell, M.A.; Bjørge, L.; Willmott, L.; Novák, Z.; Black, D.; Gilbert, L.; Sharma, S.; Valabrega, G.; Landrum, L.M.; Gropp-Meier, M.; et al. Overall survival in patients with endometrial cancer treated with dostarlimab plus carboplatin-paclitaxel in the randomized ENGOT-EN6/GOG-3031/RUBY trial. Ann. Oncol. 2024, 35, 728–738. [Google Scholar] [CrossRef] [PubMed]
- Eskander, R.N.; Sill, M.W.; Beffa, L.; Moore, R.G.; Hope, J.M.; Musa, F.B.; Mannel, R.; Shahin, M.S.; Cantuaria, G.H.; Girda, E.; et al. Pembrolizumab plus Chemotherapy in Advanced Endometrial Cancer. N. Engl. J. Med. 2023, 388, 2159–2170. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Eskander, R.; Sill, M.; Beffa, L.; Moore, R.; Hope, J.; Musa, F.; Mannelf, R.; Shahing, M.; Cantuariah, G.; Girda, E.; et al. Overall Survival and Progression Free Survival by PD-L1 status in Endometrial Cancer (EC) patients (pts) treated with pembrolizumab plus carboplatin/paclitaxel (CP) as compared to CP plus placebo (PBO) in the NRG GY018 trial. #SGO2024. Gynecol. Oncol. 2024, 190, S5. [Google Scholar]
- Colombo, N.; Biagioli, E.; Harano, K.; Galli, F.; Hudson, E.; Antill, Y.; Choi, C.H.; Rabaglio, M.; Marmé, F.; Marth, C.; et al. Atezolizumab and chemotherapy for advanced or recurrent endometrial cancer (AtTEnd): A randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2024, 25, 1135–1146. [Google Scholar] [CrossRef] [PubMed]
- Westin, S.N.; Moore, K.; Chon, H.S.; Lee, J.Y.; Thomes Pepin, J.; Sundborg, M.; Shai, A.; de la Garza, J.; Nishio, S.; Gold, M.A.; et al. Durvalumab Plus Carboplatin/Paclitaxel Followed by Maintenance Durvalumab With or Without Olaparib as First-Line Treatment for Advanced Endometrial Cancer: The Phase III DUO-E Trial. J. Clin. Oncol. 2024, 42, 283–299. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Bogani, G.; Monk, B.J.; Powell, M.A.; Westin, S.N.; Slomovitz, B.; Moore, K.N.; Eskander, R.N.; Raspagliesi, F.; Barretina-Ginesta, M.P.; Colombo, N.; et al. Adding immunotherapy to first-line treatment of advanced and metastatic endometrial cancer. Ann. Oncol. 2024, 35, 414–428. [Google Scholar] [CrossRef] [PubMed]
- Ethier, J.L.; Desautels, D.N.; Amir, E.; MacKay, H. Is hormonal therapy effective in advanced endometrial cancer? A systematic review and meta-analysis. Gynecol. Oncol. 2017, 147, 158–166. [Google Scholar] [CrossRef] [PubMed]
- Decruze, S.B.; Green, J.A. Hormone therapy in advanced and recurrent cancer: A systematic review. Int. J. Gynecol. Cancer 2007, 17, 964–978. [Google Scholar] [CrossRef] [PubMed]
- Miller, D.S.; Filiaci, V.L.; Mannel, R.S.; Cohn, D.E.; Matsumoto, T.; Tewari, K.S.; DiSilvestro, P.; Pearl, M.L.; Argenta, P.A.; Powell, M.A.; et al. Carboplatin and paclitaxel for advanced endometrial cancer: Final overall survival and adverse event analysis of a phase III trial (NRG oncology/GOG0209). J. Clin. Oncol. 2020, 38, 3841–3850. [Google Scholar] [CrossRef] [PubMed]
- Fader, A.N.; Roque, D.M.; Siegel, E.; Buza, N.; Hui, P.; Abdelghany, O.; Chambers, S.K.; Secord, A.A.; Havrilesky, L.; O’Malley, D.M.; et al. Randomized phase II trial of carboplatin paclitaxel versus carboplatin-paclitaxel-trastuzumab in uterine serous carcinomas that overexpress human epidermal growth factor receptor 2/neu. J. Clin. Oncol. 2018, 36, 2044–2051. [Google Scholar] [CrossRef] [PubMed]
- Fader, A.N.; Roque, D.M.; Siegel, E.; Buza, N.; Hui, P.; Abdelghany, O.; Chambers, S.; Secord, A.A.; Havrilesky, L.; O’Malley, D.M.; et al. Randomized Phase II Trial of Carboplatin-Paclitaxel Compared with Carboplatin-Paclitaxel-Trastuzumab in Advanced (Stage III-IV) or Recurrent Uterine Serous Carcinomas that Overexpress Her2/Neu (NCT01367002): Updated Overall Survival Analysis. Clin. Cancer Res. 2020, 26, 3928–3935. [Google Scholar] [CrossRef] [PubMed]
- Oaknin, A.; Tinker, A.V.; Gilbert, L.; Samouëlian, V.; Mathews, C.; Brown, J.; Barretina-Ginesta, M.P.; Moreno, V.; Gravina, A.; Abdeddaim, C.; et al. Clinical activity and safety of the anti-programmed death 1 monoclonal antibody Dostarlimab for patients with recurrent or advanced mismatch repair-deficient endometrial cancer: A nonrandomized phase 1 clinical trial. JAMA Oncol. 2020, 6, 1766–1772. [Google Scholar] [CrossRef] [PubMed]
- Makker, V.; Colombo, N.; Casado Herráez, A.; Santin, A.D.; Colomba, E.; Miller, D.S.; Fujiwara, K.; Pignata, S.; Baron-Hay, S.; Ray-Coquard, I.; et al. Study 309–KEYNOTE-775 investigators. Lenvatinib plus pembrolizumab for advanced endometrial cancer. N. Engl. J. Med. 2022, 386, 437–448. [Google Scholar] [CrossRef] [PubMed]
- Marabelle, A.; Fakih, M.; Lopez, J.; Shah, M.; Shapira-Frommer, R.; Nakagawa, K.; Chung, H.C.; Kindler, H.L.; Lopez-Martin, J.A.; Miller, W.H., Jr.; et al. Association of tumour mutational burden with outcomes in patients with advanced solid tumours treated with pembrolizumab: Prospective biomarker analysis of the multicohort, open-label, phase 2 KEYNOTE-158 study. Lancet Oncol. 2020, 21, 1353–1365. [Google Scholar] [CrossRef] [PubMed]




| FIGO 2009 Stage I | FIGO 2023 Stage I |
|---|---|
| Tumor is limited to the uterine corpus. | A combination of the following features: histological typea, presence and depth of myometrial invasion (inner versus outer half), and absent or focal LVSI b |
| Subclassified as stage IA, indicating no myometrial invasion or invasion of less than 50% of the myometrial thickness, and stage IB, indicating myometrial invasion equal to or greater than 50%. | Classification into stage IA or IB now applies exclusively to non-aggressive histological subtypes a with absent or focal LVSI b |
| For non-aggressive histological types a with absent or focal LVSI b, the updated classification reintroduces a distinction between tumors confined to the endometrium (now classified as IA1), those with less than 50% myometrial invasion (IA2), and those with invasion equal to or exceeding 50% (IB). | |
| For aggressive histological types a, the updated system introduces stage IC for tumors without myometrial invasion and classifies any degree of myometrial invasion as stage IIC. | |
| Ovarian involvement is now permitted within staging criteria if all of the following conditions are met: low-grade endometrioid histology; absent or superficial myometrial invasion (<50%); absent or focal LVSI b; no evidence of additional metastatic disease; unilateral ovarian involvement confined to the ovary, without capsular invasion or rupture. | |
| FIGO 2009 Stage II | FIGO 2023 Stage II |
| Tumor confined to the uterus with extension into the cervical stroma | A combination of tumor features—including cervical stromal invasion, substantial LVSI b, and myometrial infiltration by an aggressive histological subtype a. |
| IIA—a non-aggressive histological type a with extension to | |
| IIB—a non-aggressive histological type a with substantial LVSI b | |
| IIC—an aggressive histological type with any degree of myometrial invasion. | |
| FIGO 2009 Stage III | FIGO 2023 Stage III |
| Local and/or regional spread outside of the uterus excluding bladder/intestinal lining, and distant sites. | Local and/or regional tumor spread. |
| Tubo-ovarian and serosal involvement are grouped under stage IIIA. | Stage IIIA is now subdivided into: |
| IIIA1, Tubo-ovarian involvement | |
| IIIA2, Subserosal and serosal involvement The concept of the uterine subserosa as a distinct anatomical site has been introduced. | |
| Vaginal and parametrial tumor involvement are grouped under stage IIIB. | Stage IIIB is subdivided into IIIB1 indicating vaginal and/or parametrial tumor involvement IIIB2 indicating pelvic peritoneal involvement). |
| Nodal micro- and macrometastasis are grouped under stage IIIC1 (pelvic) and IIIC2 (para-aortic). | Stage IIIC1, indicating pelvic lymph node involvement, is now subdivided into IIIC1 for micrometastases and IIIC1 for macrometastases. Similarly, Stage IIIC2, indicating para-aortic lymph node involvement, is subdivided into IIIC2 for micrometastases and IIIC2 for macrometastases |
| FIGO 2009 Stage IV | FIGO 2023 Stage IV |
| Stage IVB includes abdominal peritoneal spread as well as distant metastases to the lungs, liver, brain, bone, and non-regional lymph nodes (the inguinal region or above the renal vessels). | Abdominal peritoneal spread is classified as stage IVB. Distant metastases to the lungs, liver, brain, bone, and non-regional lymph nodes (the inguinal region or above the renal vessels) are grouped as stage IVC |
| In FIGO 2023 staging for stages I and II, POLE-mutated tumors are designated as IAm POLEmut and p53-abnormal tumors as IICm p53abn, irrespective of anatomical extent, degree of LVSI, or histological subtype. Tumors with no specific molecular profile (NSMP) or mismatch repair deficiency (MMRd) do not influence staging | |
a Histological Classification:
| |
b Lymphovascular space invasion (LVSI):
| |
| RCT | Antibody | Cohort | Randomization Strategy | Patient Population | Duration | PFS Benefit | Grade3 Adverse Events IO vs. Control | |||
|---|---|---|---|---|---|---|---|---|---|---|
| Recurrent Disease | Newly Diagnosed Stage III/Stage IV | Overall Population | MMRd | MMRp | ||||||
| RUBY/ENGOT-EN6/GOG3031 | antyPD-1 | 494 | 1:1 A: CT+ Dostarlimab B: CT | 239 (47.7%) | 258 (52.3%) | up to progression max. 36 months | HR 0.64 (95% CI 0.51–0.80) p < 0.001 | HR 0.28 (95% CI 0.16–0.50) p < 0.0001 | HR 0.76 (95% CI 0.59–0.98) | 174 (72.2%) vs. 148 (60.2%) |
| NRG-GY018 | antyPD1 | 813 | 1:1 A: CT+ Pembrolizumab B: CT | not recorded | not recorded | up to progression max. 25.5 months | HR 0.30 (95% CI, 0.19–0.48), p < 0.001 | HR 0.30 (95% CI 0.19–0.48) p < 0.0001 | HR 0.54 (95% CI 0.41–0.71) | MMRd 69 (63.3%) vs. MMRp 50 (47.2%) vs. 124 (45.3%) |
| AtTEnd | antyPDL1 | 549 | 2:1 A: CT+ Atezolizumab B: CT | 243 (67.5%) vs. 126 (66.7%) | 117 (32,5%) vs. 62 (33,3%) | up to progression | HR 0.74 (95% CI 0.61–0.91) p< 0.003 | HR 0.38 (95%CI 0.23–0.57) p< 0.0005 | HR 0.92 (95% CI 0.73–1.16) | No data |
| DUO-E | antyPDL1 | 479 | 1:1:1 A: CT B: CT + Durvalumab C: CT + Durvalumab +olaparib | 125 vs. 126 | 116 vs.114 | up to progression | HR 0.71 (95% CI 0.57–0.89), p < 0.0219 | HR 0.42 (95%CI 0.22–0.80) | HR 0.77 (95% CI 0.60–0.97) p < 0.0001 | No data |
| DUO-E | antyPDL1 + Olaparib | 480 | 1:1:1 A: CT B: CT + Durvalumab C: CT + durvalumab +olaparib | 125 vs. 126 | 114 vs. 116 | up to progression | HR:0.55 (95% CI 0.43–0.69); p < 0.0001 | HR:0.28 (95%CI, 0.10–0.68) | No data | No data |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sznurkowski, J.J.; Bodnar, L.; Dańska-Bidzińska, A.; Marszałek, A.; Blecharz, P.; Chudecka-Głaz, A.; Klasa-Mazurkiewicz, D.; Kowalik, A.; Zołciak-Siwinska, A.; Zielinska, A.; et al. Updated Guidelines for the Diagnosis and Treatment of Endometrial Carcinoma: The Polish Society of Gynecological Oncology (2025v). Curr. Oncol. 2025, 32, 340. https://doi.org/10.3390/curroncol32060340
Sznurkowski JJ, Bodnar L, Dańska-Bidzińska A, Marszałek A, Blecharz P, Chudecka-Głaz A, Klasa-Mazurkiewicz D, Kowalik A, Zołciak-Siwinska A, Zielinska A, et al. Updated Guidelines for the Diagnosis and Treatment of Endometrial Carcinoma: The Polish Society of Gynecological Oncology (2025v). Current Oncology. 2025; 32(6):340. https://doi.org/10.3390/curroncol32060340
Chicago/Turabian StyleSznurkowski, Jacek J., Lubomir Bodnar, Anna Dańska-Bidzińska, Andrzej Marszałek, Pawel Blecharz, Anita Chudecka-Głaz, Dagmara Klasa-Mazurkiewicz, Artur Kowalik, Agnieszka Zołciak-Siwinska, Aleksandra Zielinska, and et al. 2025. "Updated Guidelines for the Diagnosis and Treatment of Endometrial Carcinoma: The Polish Society of Gynecological Oncology (2025v)" Current Oncology 32, no. 6: 340. https://doi.org/10.3390/curroncol32060340
APA StyleSznurkowski, J. J., Bodnar, L., Dańska-Bidzińska, A., Marszałek, A., Blecharz, P., Chudecka-Głaz, A., Klasa-Mazurkiewicz, D., Kowalik, A., Zołciak-Siwinska, A., Zielinska, A., Bidziński, M., & Sawicki, W. (2025). Updated Guidelines for the Diagnosis and Treatment of Endometrial Carcinoma: The Polish Society of Gynecological Oncology (2025v). Current Oncology, 32(6), 340. https://doi.org/10.3390/curroncol32060340






