Highlights
Public health relevance—How does this work relate to a public health issue?
- Chronic pain and opioid use in older adults are interlinked issues with major functional and mental health impacts.
- An integrated, system-level approach is needed beyond symptom control.
Public health significance—Why is this work of significance to public health?
- This review proposes a neuropsychiatric model to enhance safety, prevention, and care in later life.
- It highlights shared mechanisms linking pain, depression, and opioid misuse.
Public health implications—What are the key implications or messages for practitioners, policy makers and/or researchers in public health?
- Treatment should prioritise functional recovery, emotional stability, and cognitive preservation.
- Multimodal strategies combining pharmacological and psychosocial care are essential.
Abstract
Background: Chronic pain, opioid use, and mental health disorders frequently co-occur in older adults, forming a complex and mutually reinforcing triad. Neurobiological ageing processes—such as neuroinflammation, dopaminergic decline, and impaired top-down regulation—may increase vulnerability to maladaptive coping strategies, including opioid misuse. This review aims to integrate neurobiological, affective, and clinical evidence to propose a unified neuropsychiatric framework for understanding the intersection between chronic pain, emotional distress, and opioid vulnerability in later life, while highlighting implications for integrated treatment and opioid stewardship. Methods: This structured narrative review synthesised interdisciplinary evidence from neuroscience, geriatric psychiatry, and pain medicine. The literature was thematically organised to examine shared neurobiological and psychosocial mechanisms underlying chronic pain, affective disorders, and opioid use disorder (OUD) in older adults, with attention to treatment strategies and stewardship principles. Results: Converging evidence suggests a neuroprogressive continuum linking chronic pain, emotional distress, opioid misuse, and cognitive decline. Key mechanisms include frontolimbic dysfunction, impaired reward processing, and chronic allostatic load. Therapeutic approaches that integrate analgesia with emotional regulation—such as buprenorphine, serotonin–noradrenaline reuptake inhibitors (SNRIs), and multimodal tapering strategies—may offer neuroprotective benefits. Effective opioid stewardship appears to require integrated functional, cognitive, and affective monitoring. Conclusions: Pain management in older adults may benefit from moving beyond symptom-focused approaches toward a neuropsychiatric model of care aimed at preserving homeostatic balance across sensory, emotional, and motivational domains. Within this framework, opioid therapy can be conceptualised as a potential means of functional and neuroaffective restoration, rather than solely as a strategy for risk reduction.
1. Introduction
Chronic pain in older adults is a growing public health concern, not only because of its rising prevalence but also because of its profound entanglement with affective suffering, neurobiological vulnerability, and the increasing use of opioid medications [1,2]. Conventional approaches have often separated the somatic, psychological, and pharmacological domains of pain, resulting in fragmented care pathways and unintended iatrogenic outcomes—most notably, the emergence of opioid use disorder (OUD) in older adults [3,4].
In recent decades, mounting evidence has challenged the reductionist view of pain as a purely nociceptive event. Instead, chronic pain is increasingly understood as an affective-cognitive phenomenon, modulated by neuroplastic, emotional and social factors [5,6]. In older adults, this complexity is further amplified by age-related changes in brain circuits, cumulative medical comorbidities, and psychosocial stressors such as isolation, functional loss, and polypharmacy [7,8].
Despite this paradigm shift, the literature remains highly fragmented across disciplines—spanning neurology, psychiatry, pain medicine, geriatrics, and addiction science. Integrative overviews that capture the converging biological, affective, and contextual dynamics of chronic pain and opioid vulnerability in older adults are scarce. Furthermore, clinical practice often lacks a cohesive framework to guide multimodal and interdisciplinary interventions in this domain.
To address this gap, the present review synthesises current evidence across these interconnected domains. Rather than conducting a formal systematic review, we adopt a structured narrative approach, grounded in a concept-driven framework developed through extensive multidisciplinary literature mapping. An iterative thematic synthesis was performed, whereby recurrent neurobiological mechanisms, psychosocial determinants, and clinical trajectories were identified across disciplines and progressively clustered into higher-order conceptual domains. This method enables a panoramic yet analytically rigorous exploration of how chronic pain, ageing, affective instability, and opioid exposure interact to shape clinical trajectories and therapeutic needs in older adults.
Through iterative thematic synthesis of the multidisciplinary literature, convergent patterns were identified across neurobiological mechanisms, psychosocial determinants, and clinical trajectories linking chronic pain and opioid exposure in later life. These patterns were progressively clustered into four interrelated thematic domains, reflecting points of convergence rather than disciplinary boundaries: (1) Pain, Ageing and Psychosocial Vulnerability; (2) Neurobiological and Clinical Intersections; (3) Integrated Treatment Strategies; and (4) Special Populations, Coordination and Future Directions.
Together, these domains capture the convergence of biological vulnerability, affective dysregulation, and treatment complexity that characterises chronic pain and opioid exposure in older adults. By organising the evidence along these axes, the review offers a coherent conceptual framework that integrates mechanistic, clinical, and contextual dimensions and provides a structured basis for interpreting both preventive and interventional strategies within the context of opioid stewardship and affective pain care in late life.
2. Materials and Methods
This study adopts a structured narrative, concept-driven review design to synthesise heterogeneous yet conceptually related literature across the domains of pain, ageing, affective vulnerability, and opioid use in older adults. Although not a systematic review in the PRISMA sense, this approach follows an explicitly defined qualitative framework, including predefined selection criteria, qualitative evidence prioritisation, and iterative thematic synthesis, thereby ensuring transparency and conceptual reproducibility. As this review follows a structured narrative, concept-driven methodology rather than a systematic review protocol, it was not registered in PROSPERO or similar databases.
2.1. Objective and Scope
The review was designed to explore the shared neurobiological and psychosocial mechanisms underpinning chronic pain and vulnerability to opioid use in ageing populations, with a focus on affective dysregulation, multimorbidity, and integrated treatment strategies. The overarching goal was to develop a clinically useful, multidimensional framework that links chronic pain management with opioid stewardship in later life. Rather than aiming for exhaustive coverage of all available evidence, the review focuses on identifying convergent mechanisms, recurring clinical patterns, and treatment-relevant themes across disciplines.
2.2. Search Strategy and Data Source
The literature included in this review was identified through a two-phase process:
An initial concept-driven selection of peer-reviewed articles, guidelines, and academic reports published between 2000 and 2025, covering neurobiology, geriatric pain, affective disorders, opioid use, and addiction. The timeframe starting from the year 2000 was selected to capture literature reflecting contemporary neurobiological models of pain (including neuroplasticity-based and affective–cognitive frameworks), advances in the neuroscience of ageing, and the modern era of opioid prescribing and stewardship. Earlier literature, while historically relevant, was excluded to maintain conceptual coherence with current clinical and neuroscientific paradigms.
Sources were retrieved from PubMed, the Cochrane Library, WHO, CDC, EMCDDA, and NIHR/NICE, using combinations of the following keywords: “chronic pain”, “older adults”, “opioid use”, “addiction”, “neuroaffective regulation”, “multimorbidity”, “polypharmacy”, “opioid stewardship”, “integrated care”, “affective dysregulation”, and “psychosocial vulnerability”.
Grey literature, commercial websites, opinion pieces, and non–peer-reviewed materials were excluded.
Studies were included if they met the following criteria:
- Published between 2000 and 2025.
- Peer-reviewed primary studies, meta-analyses, systematic reviews, clinical guidelines, or policy papers.
- Focused on adults aged ≥65 with chronic non-cancer pain and/or opioid exposure.
- Addressed at least one of the following: neurobiological mechanisms, psychosocial factors, pharmacological management, non-pharmacological interventions, or healthcare coordination.
Studies focusing exclusively on cancer pain or palliative opioid use were excluded [9].
When systematic reviews or meta-analyses were identified, they were considered higher-level evidence and prioritised in the synthesis when methodologically robust and aligned with the objectives of the review. Primary studies were not automatically extracted from the reference lists of systematic reviews. Instead, individual studies were included only when they provided mechanistic, geriatric-specific, or clinical insights not sufficiently addressed by existing reviews, and when they met the same predefined thematic relevance and qualitative appraisal criteria.
2.3. Data Selection and Synthesis
Data selection, qualitative data extraction, and thematic synthesis were performed by the lead author through an iterative review process. Extraction focused on conceptual, neurobiological, psychosocial, and clinical features relevant to the objectives of the review, rather than on predefined quantitative variables. No dedicated systematic review software (e.g., Covidence) was used, as the extraction process was oriented toward conceptual and thematic integration rather than quantitative data abstraction.
229 sources were mapped and thematically classified into four conceptual domains that correspond to the review’s structure:
- Pain, ageing, and psychosocial vulnerability.
- Neurobiological and clinical intersections.
- Integrated treatment strategies.
- Special populations and coordinated and future direction.
Thematic synthesis was conducted iteratively, with selected papers coded for content relevance, methodological quality, and theoretical contribution. No statistical meta-analysis was performed because of heterogeneity in study types and outcomes. Priority was given to systematic reviews, large cohort studies, randomised controlled trials, and evidence-based guidelines.
Evidence weighting was qualitative rather than quantitative. Systematic reviews, meta-analyses, large cohort studies, randomised controlled trials, and evidence-based clinical guidelines were assigned greater interpretative weight. Observational, descriptive, and smaller studies were included selectively when they contributed unique mechanistic insights, addressed geriatric-specific vulnerabilities, or informed integrated models of care not otherwise captured by higher-level evidence.
When conflicting or heterogeneous evidence was identified, studies were not excluded or statistically pooled. Instead, discrepancies were examined in relation to study design, population characteristics, outcome definitions, and clinical or contextual factors. Greater interpretative weight was assigned to convergent findings supported by multiple high-quality sources across different disciplines, while divergent results were explicitly acknowledged and contextualised within the narrative synthesis.
Methodological rigour and relevance were assessed qualitatively. Where applicable, multiple sources were triangulated to ensure conceptual robustness and reduce bias. Sources were classified by their predominant thematic content, although several addressed overlapping domains. In such cases, the dominant focus was used for categorisation. Section 2 and Section 3 contain the highest concentration of sources, consistent with the extensive literature on the neurobiology of pain and integrated therapeutic strategies.
Interpretative statements that extend beyond direct empirical findings are explicitly framed as integrative interpretations grounded in the authors’ clinical and research expertise. Such interpretations were applied only after triangulation of consistent evidence across multiple sources and disciplines and are clearly distinguished from conclusions directly derived from the reviewed literature.
3. Results
3.1. Pain, Ageing and Psychosocial Vulnerability
3.1.1. Synthesis of Findings from the Literature
Pain, particularly when chronic, is increasingly conceptualised not merely as a symptom but as a disorder of neural plasticity, a maladaptive reorganisation of sensory, emotional, and cognitive circuits that may blur the boundary between nociception and affect [10,11,12,13]. Once adaptive as an alarm signal, the pain system may evolve into a self-sustaining network dysfunction, in which persistent activation of limbic and prefrontal regions—including the amygdala, anterior cingulate cortex (ACC), and nucleus accumbens (NAc)—appears to reshape how the brain encodes salience and emotion [14]. This reorganisation, often referred to as “pain-induced neuroplasticity”, has been shown to share mechanistic features with processes implicated in mood disorders and substance addiction, including altered dopaminergic tone, glutamatergic remodelling, neuroinflammation, and impaired top-down inhibitory control [15,16,17].
From a neurobiological perspective, converging evidence suggests that chronic pain and affective dysregulation may represent closely related facets of altered neural plasticity. Both engage the mesocorticolimbic system—the ventral tegmental area (VTA), nucleus accumbens (NAc), medial prefrontal cortex (mPFC), and extended amygdala—and are associated with decreased reward sensitivity, anhedonia, and negative affect [18,19,20]. Sustained nociceptive input and emotional distress have been reported to promote microglial activation, pro-inflammatory cytokine release (IL-6, TNF-α), and mitochondrial dysfunction, processes that may progressively erode synaptic integrity in limbic–prefrontal networks [21,22,23]. This putative “neuroprogressive” trajectory, analogous to that described in chronic stress and affective disorders, has been hypothesised to link pain chronification to cognitive decline and the neurodegenerative cascade of ageing [24,25]. In older adults, reductions in grey-matter volume within the insula, hippocampus, and dorsolateral PFC have been observed in association with both prolonged pain exposure and depressive symptom burden, suggesting a shared vulnerability pathway [26,27,28,29].
The affective dimension of pain (“pain affect”) appears to become particularly central in later life, when sensory thresholds, emotional regulation, and reward processing are altered by ageing [30,31]. Declines in dopaminergic and serotonergic signalling may reduce the brain’s capacity for endogenous analgesia and mood stabilisation, thereby amplifying suffering and catastrophising. This convergence of biological ageing, frailty, and emotional dysregulation has been proposed to generate a unique clinical phenotype in which chronic pain, depression, anxiety, and opioid responsiveness intertwine [32,33,34].
Opioid therapy appears to play an ambivalent role. Acting on μ-opioid receptors (MORs) in both nociceptive and reward circuits, opioids may transiently normalise dysregulated limbic activity and restore hedonic tone, but chronic exposure is associated with counter-adaptations, tolerance, dependence, and opioid-induced hyperalgesia [35,36,37].
Epidemiological data suggest that between 45% and 80% of adults aged ≥65 years experience chronic or persistent pain, with prevalence estimates reaching 70–85% in institutionalised populations [38,39]. Clinical variability in pain intensity and disability appears to be better explained by psychosocial determinants than by somatic pathology alone [33,40,41]. Comorbid depression and anxiety are common [42,43,44,45,46,47] and are consistently associated with poorer functional outcomes. Social isolation has been shown to further amplify pain via the anterior cingulate and insula [48,49,50] and may induce HPA axis dysregulation and inflammatory priming [51,52,53].
Cognitive factors, such as catastrophising, have been correlated with altered activity in prefrontal and brainstem circuits [54,55], promoting avoidance and physical deconditioning. These circuits overlap with those implicated in addiction and compulsive reward-seeking [15]. Frailty, reconceptualized by some authors as a neuropsychiatric syndrome, has been linked to dopaminergic and neurotrophic deficits and chronic inflammation [15,56,57,58,59].
Finally, multimorbidity and polypharmacy may further undermine neuroplastic homeostasis. Most elderly patients present with ≥3 chronic diseases and are frequently exposed to high-risk CNS-active drug combinations [60,61,62,63,64,65].
3.1.2. Clinical and Conceptual Interpretation
The findings summarised above converge on a neuropsychosocial model in which chronic pain in older adults is closely intertwined with affective vulnerability, age-related neurodegeneration, and pharmacological fragility. Rather than a dichotomy between “analgesia” and “dependence”, the clinical scenario reflects a continuum of dysfunctions affecting motivation, stress regulation, and cognitive control. This is supported by evidence of overlapping circuitries and neurochemical processes between chronic pain and disorders such as depression and addiction [15,16,17].
The co-occurrence of affective symptoms and pain-related disability suggests that analgesic response may depend as much on restoring neuroplastic balance as on nociceptive control. However, opioids—while able to temporarily normalise hedonic tone—act on the same circuitry implicated in craving and emotional dysregulation. This dual effect, already recognised in younger adults, appears to be amplified in older patients by frailty-related neurochemical imbalances and structural vulnerabilities [33,34,37].
The concepts of “psychoneurobiological frailty” and “neuroprogression” [24,58,59] help frame the interplay among ageing, chronic pain, and emotional suffering. Interventions for this population are therefore likely to require addressing not only pain relief but also the preservation of reward sensitivity, cognitive function, and affective regulation [66]. In this perspective, pharmacological strategies (e.g., opioid stewardship, deprescribing) should be integrated with psychosocial and behavioural interventions targeting isolation, maladaptive cognitions, and functional decline [41,54,55].
Rather than focusing exclusively on symptom suppression, this integrated paradigm conceptualises chronic pain as a dynamic output of an ageing brain under stress, reflecting impaired equilibrium across interdependent systems of emotion, cognition, and somatic regulation.
3.2. Neurobiological and Clinical Intersection
3.2.1. Synthesis of Findings from the Literature
The convergence of chronic pain, mood disorders, and addiction appears to reflect a shared disruption of brain networks that mediate reward, motivation, and affect regulation. These conditions are not merely comorbid but have been proposed as parallel expressions of maladaptive plasticity within the same neural architecture, shaped by chronic stress, neuroinflammation, and impaired inhibitory control [67,68,69,70].
At the core of this convergence lies a set of interacting alterations within the mesocorticolimbic system, comprising the ventral tegmental area (VTA), nucleus accumbens (NAc), amygdala, and prefrontal cortex (PFC). In chronic pain, this system has been shown to undergo functional reorganisation: dopamine release has been reported to be blunted, NAc connectivity to the PFC appears to weaken, and the network shifts toward encoding aversive salience rather than reward [14,20,70,71]. Hypodopaminergic tone, also observed in opioid addiction and depression, has been associated with anhedonia and motivational impairment [72,73,74]. Limbic structures, including the amygdala and ACC, often exhibit hyperactivity and impaired top-down regulation, reinforcing emotional memories of suffering and avoidance behaviours [19,75,76,77]. These changes are mirrored in the PFC, particularly in ageing, where cortical atrophy and dopaminergic degeneration may further compromise inhibitory control and motivational balance [67,78,79,80,81,82,83].
Pain chronification and mood disorders share stress-related disruptions of plasticity and neuroimmune activation. Sustained nociceptive input has been shown to activate the HPA axis and to promote glial activation with pro-inflammatory cytokine release (IL-1β, IL-6, TNF-α), thereby contributing to central sensitisation and neurotoxicity [51,84,85,86]. This neuroinflammatory cascade has been shown to promote maladaptive long-term potentiation (LTP) in limbic circuits and long-term depression (LTD) in the PFC, creating a convergence of sensory and affective dysregulation. Reduced BDNF expression may limit synaptic resilience, leading to heightened cognitive and emotional vulnerability [87,88,89,90]. These alterations mirror those described in chronic opioid exposure, where μ-opioid receptor downregulation and NMDA-mediated sensitisation have been shown to remodel the same pathways [86,91].
In this context, chronic pain acts as a persistent stressor. The extended amygdala, bed nucleus of the stria terminalis (BNST), and HPA axis may maintain elevated corticotropin-releasing factor (CRF) and noradrenergic tone, thereby heightening vulnerability to craving and compulsive relief-seeking [15,92]. In older adults, these dynamics are further exacerbated by reduced dopaminergic reserve and diminished cognitive flexibility [37,93,94].
Analgesic efficacy increasingly appears to depend on affective and regulatory stability. Limbic hyperreactivity, impaired descending modulation, and negative affect have been associated with reduced responsiveness to both pharmacological and non-pharmacological treatments [14,93,95,96,97]. In older adults, these effects may be amplified by inflammatory priming and cognitive inflexibility [98,99,100].
Neuroimaging studies have revealed shared disruptions across chronic pain, depression, and opioid use disorder, including PFC–NAc dysconnectivity, amygdala hyperactivity, and abnormal salience processing [67,101,102,103]. Chronic opioid use may transiently restore affective balance but is associated with tolerance and emotional withdrawal over time [70,104]. With ageing, structural degeneration and neuroinflammatory mechanisms may further disrupt salience attribution and regulatory circuits [105,106].
The opioid–endocannabinoid interface has also been proposed to play a critical modulatory role. CB1 receptors in the amygdala, periaqueductal grey (PAG), and PFC are known to modulate stress and reward processing [107,108,109]. Chronic opioid exposure has been shown to impair CB1 receptor function, thereby contributing to affective instability. Reduced endocannabinoid tone, commonly observed in ageing, may increase pain sensitivity and vulnerability to dependence [110,111,112,113].
Taken together, chronic pain, mood disorders, and opioid use disorder may represent neuroprogressive outcomes of a shared disrupted system, sustained by chronic stress, neuroinflammation, and age-related degeneration (Figure 1). This integrated perspective helps reframe opioid vulnerability in older adults not as a moral failure but as a potential neurobiological endpoint of an overwhelmed affective regulatory system.
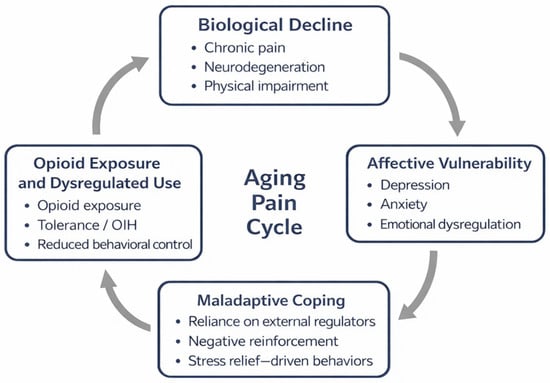
Figure 1.
Conceptual model of the “Aging Pain Cycle”, illustrating how biological decline, affective vulnerability, maladaptive coping strategies, and dysregulated opioid exposure may interact over time. The model does not equate chronic pain management with addiction, but rather highlights negative reinforcement processes that may contribute to the persistence of pain, emotional distress, and functional decline in vulnerable older adults.
3.2.2. Clinical and Conceptual Interpretation
Clinically, these convergences highlight the close interdependence of pain and affective regulation in ageing. Pain may amplify mood symptoms, and mood symptoms may in turn exacerbate pain. The reward and salience networks represent common substrates of both processes, and in older individuals with pre-existing neurodegenerative vulnerability, these networks appear particularly susceptible to destabilisation by stress, loss, or medication. Affective dysregulation should therefore not be viewed merely as a comorbidity but has been proposed as a central determinant of the pain trajectory itself [33,44,114].
The co-activation of stress and reward systems may help explain the clinical progression from chronic pain to craving and maladaptive opioid use. Neuroimaging studies have demonstrated shared disruptions in salience and executive networks [67,101,102]. Older adults appear more susceptible due to orbitofrontal and insular degeneration, which may amplify compulsive tendencies and reduce interoceptive accuracy [105,106].
Importantly, affective suffering often appears to underpin analgesic demand. When endogenous modulatory systems (dopamine, endorphins, endocannabinoids) are insufficient, opioids may fill this regulatory gap, creating patterns of use driven by emotional relief rather than by euphoria [15,115]. This learned behaviour has been described as mirroring the neurocognitive profile of late-onset addiction, characterised by prefrontal disinhibition and limbic sensitization [116,117,118,119].
Clinical classification should therefore distinguish physical dependence from pseudoaddiction and from full opioid use disorder (OUD). While physical dependence represents an expected pharmacological outcome [36,91], pseudoaddiction has been conceptualised as reflecting unmet therapeutic needs [120,121], and established OUD is associated with structural reorganisation of reward and control networks [104,122].
Ultimately, opioid stewardship in older adults requires recognition of emotional drivers—such as fear, loneliness, and unresolved grief—as integral components of the clinical picture. Affective stability, rather than analgesia alone, may represent a more appropriate therapeutic target. When mood, cognition, and interpersonal context are incorporated into the therapeutic framework, opioid therapy can be reframed not merely as a pharmacological intervention but as a potential means of restoring neuropsychological homeostasis [123,124].
3.3. Integrated Treatment Strategies
3.3.1. Synthesis of Findings from the Literature
The management of chronic pain in older adults increasingly appears to require a paradigm that integrates analgesic efficacy, emotional stabilisation, and neurobiological safety. Pharmacotherapy may therefore be most effective when conceptualised within a triadic framework—pain control, mood regulation, and dependence prevention—grounded in the recognition that these three dimensions share overlapping neural substrates.
Effective treatment typically begins with a comprehensive assessment that integrates somatic, affective, cognitive, and social dimensions (Table 1) [34,125,126]. Pain intensity alone provides limited prognostic information; accumulating evidence suggests that functional and emotional burden—such as interference with sleep, mood, and daily activities—more reliably predicts clinical outcomes [33,127]. Clinicians are therefore encouraged to routinely screen for depression, anxiety, sleep disorders, and cognitive impairment, as these comorbidities have been shown to influence pain perception and may modify pharmacodynamic responses through neuroinflammatory and monoaminergic pathways [68,128]. In older patients, loss of dopaminergic tone, increased oxidative stress, and reduced BDNF expression may lower thresholds for both analgesic failure and neurotoxic side effects.

Table 1.
Multidimensional Assessment Domains in Clinical Practice.
Pharmacological therapy is generally recommended to follow a multimodal, stepwise approach, combining agents that target distinct mechanisms—nociceptive, neuropathic, and affective—while aiming to minimise opioid exposure and preserve cognitive integrity (Table 2) [38,126,130]. Behavioural and psychotherapeutic interventions (CBT, ACT, mindfulness) are increasingly recognised as beneficial when embedded early, reinforcing adaptive neural plasticity and emotional regulation [115,132,133,134,135,136].

Table 2.
Pharmacological Options for Chronic Pain in Older Adults: Analgesic Effects, Mood/Affective Implications, and Geriatric Clinical Considerations.
Pharmacological management in older adults with chronic pain should be framed as a stepwise, multimodal strategy that balances analgesic efficacy with neuropsychiatric safety and functional preservation (Table 2) [38,126,130]. Rather than relying on pain intensity alone, treatment selection is guided by pain phenotype (nociceptive, inflammatory, neuropathic or mixed), comorbid affective and cognitive vulnerability, renal and hepatic reserve, and fall risk (Table 1) [34,125,126]. Current evidence supports the preferential use of non-opioid agents when possible, with careful titration and monitoring to minimise sedation, delirium, and cumulative neurotoxic burden in frail patients [34,38,61]. When opioid therapy is considered, it should be implemented within an opioid stewardship framework, characterised by low initial dosing, slow titration, frequent reassessment, and systematic monitoring of affective, cognitive, and functional trajectories rather than numerical pain scores alone [126,130,131]. Across pharmacological classes, particular attention is required for drug–drug interactions, polypharmacy, and cumulative sedative load, especially in the presence of multimorbidity and neurodegenerative vulnerability. In higher-risk clinical scenarios, international guidelines emphasise individualisation of therapy, integration with psychosocial and behavioural interventions, and regular deprescribing reviews to maintain neurobiological and functional homeostasis in later life [34,61,126,130,131].
3.3.2. Clinical and Conceptual Interpretation
Chronic pain in ageing rarely occurs in isolation from emotional, cognitive, and neurobiological decline. An integrated pharmacological management approach therefore appears to require alignment between neurobiological safety and functional and affective recovery. This perspective relies on understanding pain not solely as a sensory phenomenon but as a complex disturbance of brain homeostasis.
The emphasis on early affective screening reflects increasing awareness that neuropsychiatric fragility may modulate both pain perception and drug-related neurotoxic thresholds. Pharmacological choices are therefore best tailored not only to pain phenotype but also to emotional resilience, cognitive integrity, and pre-existing neuroinflammatory burden. Non-opioid therapies have been shown to modulate both nociceptive and limbic circuits and may delay or reduce the need for opioid escalation. The inclusion of agents such as SNRIs or gabapentinoids extends the therapeutic scope into the mood–pain interface.
When opioids are required, stewardship should extend beyond dosage control to include monitoring of affective and motivational trajectories. Agents such as buprenorphine and tapentadol are not pharmacologically neutral choices and have been proposed to confer neurobiological advantages that may be particularly relevant in aged, stress-sensitised brains. An affective–motivational stewardship approach frames opioid use within a neuropsychiatric model that prioritises preservation of cortical–limbic equilibrium. Clinical decisions are therefore increasingly informed by affective reactivity, reward sensitivity, and self-regulatory capacity—assessed through a combination of patient-reported outcomes and caregiver-informed measures (e.g., mood scales such as the Geriatric Depression Scale or PHQ-9, executive and cognitive screening tools such as the MoCA, and functional or behavioural monitoring including ADL/IADL changes)—rather than by pain intensity scores alone.
Ultimately, opioid prescribing may be conceptualised as an act of neuroaffective care, involving a dynamic balance between analgesia, emotional regulation, cognitive preservation, and neuroinflammatory restraint. This framework acknowledges the vulnerability of the ageing brain and reframes analgesic strategy as a component of integrated psychiatric–neurosomatic rehabilitation.
3.4. Special Populations, Coordination and Future Direction
3.4.1. Findings from the Literature
Chronic pain and OUD have been increasingly described as intertwined conditions that share not only clinical overlap but also overlapping neurobiological substrates. Both appear to arise from maladaptive remodelling of reward–stress–pain circuitry, in which the mesolimbic dopamine system, the hypothalamic–pituitary–adrenal (HPA) axis, and the limbic forebrain operate under conditions of chronic allostatic load [15,137,149]. In ageing, this shared substrate may be further compromised by dopaminergic depletion, frontostriatal disconnection, and neuroinflammatory amplification, leaving older adults with prior opioid exposure particularly vulnerable.
For these patients, pain management cannot be adequately conceptualised as a dichotomy between “analgesia” and “addiction treatment”; instead, it has been proposed to follow an integrated neuropsychiatric logic, in which modulation of nociceptive, affective, and reward-related circuits occurs in parallel.
In patients receiving medications for opioid use disorder (MOUD), such as buprenorphine or methadone, analgesic regimens are generally recommended to build upon the existing opioid-maintenance substrate [150,151]. Abrupt discontinuation has been shown to destabilise reward homeostasis and pain modulation, thereby increasing relapse risk and pain sensitivity through mechanisms involving NMDA receptor upregulation and glial priming [37,152]. Divided dosing schedules or carefully monitored supplementation have been reported to optimise pain control without compromising MOUD efficacy [153,154,155].
A key concept in this context is the so-called “opioid debt”, referring to the mismatch between opioid receptor occupancy sufficient for withdrawal suppression and that required for effective analgesia. When this physiological gap is not addressed, patients may remain vulnerable to hyperalgesia and stress-induced craving [156,157]. Targeted correction, through scheduled dosing within a multimodal treatment plan, has been proposed as necessary to restore neurobiological stability [158,159].
Adjuvant agents such as SNRIs and gabapentinoids, together with non-pharmacological interventions, are considered important components of comprehensive care, whereas benzodiazepines and sedative agents are generally discouraged because of increased toxicity and overdose risk in older adults [130,131,141,160].
Buprenorphine has been shown to function as both an analgesic and an OUD treatment through partial μ-opioid agonism and κ-opioid receptor antagonism, potentially balancing nociceptive relief with affective stability [81,161,162]. It has been associated with modulation of amygdala–prefrontal circuits and reduced cue reactivity, suggesting potential utility in older adults in whom affective dysregulation coexists with chronic pain [163,164].
Methadone, through full μ-opioid agonism, NMDA receptor antagonism, and monoamine reuptake inhibition, engages peripheral, spinal, and supraspinal analgesic mechanisms. It has also been associated with stabilisation of affective tone and reduction in salience misattribution and has been proposed to exert antipsychotic-like effects in structured treatment settings [104,165,166]. Levomethadone, with reduced NMDA activity and lower cardiotoxic potential, may offer more targeted modulation of the opioid–glutamate–κ-opioid receptor axis, which could be particularly relevant in older adults with neurodegenerative vulnerability [167,168].
Finally, optimal outcomes appear to require coordinated care across pain medicine, psychiatry, and addiction services. Such triangular coordination has been shown to reduce iatrogenic instability and to support therapeutic continuity through integrated planning and shared clinical responsibility [169,170].
3.4.2. Clinical and Conceptual Interpretation
Older adults with chronic pain and a history of OUD often present with compounded neurobiological vulnerability. In this population, pain management may be more appropriately understood not merely as analgesic delivery but as a form of neuropsychiatric regulation. Strategies such as divided dosing of MOUD agents have been proposed to address neurochemical instability associated with inadequate analgesia, thereby potentially reducing stress-induced dysregulation and supporting functional opioid tone.
The concept of “opioid debt” reframes inadequate pain control as a physiological imbalance rather than a therapeutic failure. Failure to address this imbalance in MOUD-treated patients has been associated with heightened sympathetic activation and increased relapse risk [171]. Clinical correction through scheduled dosing and adjunctive therapies may help prevent both undertreatment of pain and escalation toward full μ-opioid agonists.
Buprenorphine has been described as a prototypical agent of “neuroaffective stewardship,” acting on pain, mood, and motivational circuits. Its suitability for older adults has been attributed to its balanced receptor activity and relatively limited impact on cognitive and endocrine functioning.
Methadone and levomethadone may offer distinct advantages for patients with dual disorder. Their capacity to modulate both nociceptive and affective pathways supports their use within structured, interdisciplinary treatment programmes. The additional mood-stabilising and antipsychotic-like properties of methadone have been highlighted as potentially relevant in complex affective states [172].
To move beyond fragmented care, integrated models have been increasingly advocated to coordinate analgesic modulation, affective regulation, and addiction containment. In the absence of such triangulated approaches, iatrogenic instability and neuroprogressive drift may occur. Therapeutic success is therefore increasingly framed not only in terms of pain reduction or substance use control, but also as the restoration of homeostatic balance across interdependent neural systems.
4. Integrative Neuroaffective Stewardship in Later-Life Opioid Therapy
Pain management for patients with OUD, particularly older adults, may benefit from moving beyond categorical thinking. Buprenorphine provides stability through MOR partial agonism and KOR antagonism, methadone offers broad-spectrum modulation of nociceptive and affective pathways, and levomethadone refines this approach by minimising excitotoxic and dysphoric liabilities.
Each of these agents appears to act not merely on receptors but on circuits of meaning and motivation, the same networks that, when dysregulated, produce craving, despair, and compulsive relief-seeking.
By conceptualising OUD and chronic pain as two stages of a shared neuroprogressive process, clinicians can deploy pharmacotherapy not as containment but as functional neurorehabilitation, a controlled restoration of dopaminergic, glutamatergic, and limbic balance.
This paradigm may reframe opioid therapy from a source of vulnerability into a neurobiologically grounded bridge to recovery, where analgesia and emotional regulation converge [173].
4.1. Neurobiological Rationale for Triadic Stewardship
The stewardship of opioid therapy in the elderly should not be reduced to dose titration or adverse-event surveillance. Instead, it must be understood as a neuropsychiatric process of preservation, maintaining cognition, emotional regulation, and adaptive plasticity in a brain that is both ageing and repeatedly challenged by pain, stress, and pharmacological exposure [174].
A truly geriatric approach has been proposed to rest on three interdependent axes, Pain, Mind, and Medication, each requiring systematic monitoring and dynamic rebalancing.
Pain. Chronic pain in older adults is both sensory and affective. Its evaluation must integrate objective severity with subjective burden, mobility, sleep continuity, participation, and motivation [34,126]. The goal of therapy is functional recovery, not zero pain [175]. Every follow-up should assess trajectory: Is pain improving, stable, or evolving into emotional amplification (catastrophising, fear-avoidance) [125,129,176]. Early recognition of the transition from pain signalling to pain memory (insula- and ACC-driven) may help prevent the consolidation of maladaptive circuits [177,178,179,180].
Mind. Depression, anxiety, and cognitive decline are not secondary phenomena; they are core components of the pain disorder [44,181,182]. Regular screening with tools such as the Geriatric Depression Scale (GDS), GAD-7, and MoCA enables early detection of affective and cognitive erosion [175]. Worsening mood or executive dysfunction during opioid therapy often signals neuroadaptive dysregulation, either excessive dopaminergic stimulation (euphoria → apathy) or frontal hypoactivity from prolonged μ-opioid engagement [122,183,184,185,186]. Emotional blunting and diminished initiative are not benign; they mark the onset of a neuroprogressive cascade in which motivational circuitry (VTA–NAc–PFC) loses flexibility and craving mechanisms emerge even at therapeutic doses [73,187].
Medication. The pharmacological plan must remain flexible, individualised, and regularly revised [126,188]. In geriatric practice, stewardship entails synchronising the pharmacodynamic rhythm with the patient’s neurobiological capacity for adaptation. This includes cautious dose titration, proactive management of constipation, hydration and nutritional support, and awareness that drug accumulation, sleep deprivation, and circadian disruption can trigger cognitive or affective decompensation [63].
4.2. Clinical Monitoring Across Pain, Mood and Cognition
To operationalise stewardship, clinicians should use a multidimensional follow-up matrix at every visit, integrating clinical observation, self-report, and caregiver feedback [126,135,175]. This checklist can be used to operationalise a neurocognitive surveillance model, transforming opioid follow-up into continuous observation of the brain’s adaptive state [34,152,189].
4.3. Deprescribing as Neuroadaptive Recalibration
In geriatric opioid therapy, deprescribing should not be viewed merely as drug discontinuation and may be better understood as a process of neural restoration. With each reduction in pharmacological load, the brain has an opportunity to re-engage its endogenous regulatory mechanisms. These include the resensitisation of μ-opioid receptors, the recalibration of dopaminergic tone, and the silencing of microglial overactivity, all essential for restoring affective and cognitive balance [190].
This delicate process requires careful planning and awareness of its neuropsychiatric implications. Before initiating tapering, clinicians must define clear functional and emotional targets. Reducing opioid dosage without such planning can trigger not only somatic withdrawal but also a rebound of limbic hyperactivity, manifesting as anxiety, dysphoria, or heightened pain perception [191,192].
Tapering should proceed gradually, with a typical reduction of 10–20% of the total daily dose every 2 to 4 weeks. If signs of withdrawal or neuroaffective destabilisation arise—such as insomnia, anxiety, or mood deterioration—the process must be slowed accordingly. Rapid tapering may provoke a neuroprogressive crisis, characterised by heightened suffering and cognitive disorganisation [152].
To cushion the descent, pharmacological bridging strategies are often necessary. Agents such as serotonin-norepinephrine reuptake inhibitors (SNRIs) or low-dose gabapentinoids can support descending inhibitory pain pathways and stabilise mood, thereby reducing the risk of affective rebound [193].
Non-pharmacological adjuncts are just as critical. Every step-down in dosing should be matched by increases in behavioural and psychosocial engagement. Psychotherapy, structured physical activity, and social reactivation provide alternative sources of dopaminergic stimulation and help re-establish motivational circuits [134,194,195].
Importantly, clinicians must avoid substitution cascades. Using benzodiazepines or sedative-hypnotics to manage withdrawal symptoms is contraindicated, particularly in older adults. These agents suppress prefrontal activity via GABAergic mechanisms, worsening cognitive decline and emotional blunting [63].
Ultimately, each deprescribing journey can be conceptualised as a cycle of neuroplastic recalibration. Success should not be measured solely by the final dose achieved, but by the recovery of emotional modulation, motivational resilience, and cognitive clarity.
4.4. Prevention of Delirium, Falls, and Affective Decompensation
Older adults receiving opioids are particularly susceptible to acute neuropsychiatric events, including delirium, falls, and sudden affective destabilisation [196]. These complications arise from the interplay of pharmacodynamic sensitivity, structural brain vulnerability, and environmental stressors.
Delirium often results from a transient collapse in cortical cholinergic and dopaminergic signalling, triggered by opioids, infections, dehydration, or polypharmacy [197,198]. Prevention typically requires proactive strategies, including regular cognitive screening, maintaining hydration, and ensuring stable environmental cues.
Falls, meanwhile, are typically associated with sedation, postural hypotension, and cerebellar-vestibular desynchronisation. Opioids with more favourable neurophysiological profiles—such as buprenorphine or tapentadol—may reduce these risks by preserving noradrenergic tone and reducing postural instability [146,148].
Affective decompensation—characterised by abrupt onset of apathy, irritability, or despair—may reflect underlying endocrine suppression or frontostriatal exhaustion induced by opioid exposure. Such events should prompt careful reassessment of treatment goals and consideration of opioid rotation or deprescribing [199].
Stabilising circadian rhythms, maintaining consistent light–dark cycles, promoting physical activity, and ensuring meaningful social contact are non-pharmacological interventions that support dopaminergic and serotonergic homeostasis, thereby reducing the risk of both neuropsychiatric crises and functional decline [73,200].
4.5. Toward Neuroprotective Opioid Stewardship
The overarching objective of opioid stewardship in older adults may be framed as neuroprotection. Each therapeutic decision—whether initiating, adjusting, or tapering opioids—alters the landscape of neural plasticity and network integrity. In this light, stewardship becomes an ongoing act of cortical care: balancing nociceptive control with the preservation of cognitive, emotional, and motivational circuits.
To prevent dopaminergic exhaustion, clinicians should consider rotating opioids or using partial agonists such as buprenorphine and tapentadol, which maintain analgesia while reducing neurochemical strain [201]. Adjunctive interventions, including physical activity, anti-inflammatory nutrition, and affective stimulation, counteract neuroinflammation and promote neural resilience [202,203].
Monitoring for early signs of emotional or cognitive dysregulation, promptly treating sleep or mood disturbances, and fostering rehabilitative engagement are critical to preserving the brain’s adaptive capacity. In this integrated model, stewardship extends beyond harm reduction and may be viewed as a form of neuropsychiatric support that sustains the ageing brain’s ability to feel, think, relate, and recover.
4.6. Limitations
This review has several limitations that should be acknowledged. First, although the literature synthesis followed a structured, concept-driven methodology with predefined thematic domains and qualitative evidence prioritisation, it was not conducted as a formal systematic review with quantitative meta-analysis. Consequently, the strength of the conclusions relies on the convergence of findings across heterogeneous study designs rather than on pooled effect estimates.
Second, the interdisciplinary scope of the review—spanning neuroscience, geriatric psychiatry, pain medicine, and addiction science—inevitably introduces variability in study populations, outcome measures, and methodological quality. While priority was given to systematic reviews, large cohort studies, randomised controlled trials, and evidence-based guidelines, some mechanistic and conceptual interpretations are derived from translational or observational studies and should therefore be interpreted with appropriate caution.
Third, given the narrative and integrative nature of the review, causal inferences cannot be established. Associations between chronic pain, affective dysregulation, opioid exposure, and neurobiological ageing processes should be understood as hypothesis-generating rather than definitive evidence of direct causality.
Finally, although the proposed neuropsychiatric framework is grounded in convergent evidence and clinical plausibility, it has not been prospectively validated in longitudinal or interventional studies specifically designed to test its predictive or therapeutic utility. Future research should aim to empirically evaluate this model using longitudinal designs, multimodal biomarkers, and integrated clinical outcomes in older populations.
5. Concluding Integration and Clinical Implications
Chronic pain in late life is increasingly understood not merely as a failure of sensory transmission but as an expression of affective dysregulation within an ageing brain struggling to maintain homeostasis across sensation, emotion, and meaning. Across the lifespan, pain may progressively shift from an adaptive warning signal to a condition of affective embodiment, shaped by neuroplastic decline, chronic stress exposure, and neuroinflammatory processes [68,204]. In this perspective, pain in ageing appears less as a transient symptom and more as a persistent neuroaffective state, reflecting the brain’s reduced capacity to integrate threat, loss, and self-preservation signals.
At the neurobiological level, converging evidence points to a progressive disruption of integrative circuitry. Fronto-limbic and mesocorticolimbic networks—central to reward processing, motivation, and emotional salience—undergo cumulative deterioration due to age-related neurodegeneration and sustained allostatic load. Microglial activation, mitochondrial dysfunction, and glutamatergic dysregulation further erode the brain’s ability to distinguish nociceptive input from emotional distress, and pain from affect [205,206,207,208,209].
This continuum provides a framework for understanding why chronic pain, depression, and substance use disorders frequently co-occur in later life. Rather than reflecting moral weakness or isolated comorbidity, this convergence may represent a shared neuroprogressive phenotype characterised by exhausted regulatory capacity [210,211]. Within this context, dependence—whether pharmacological or behavioural—can be interpreted as a maladaptive attempt to restore affective equilibrium in neural systems no longer able to self-regulate effectively [104,122].
Ageing further amplifies this vulnerability. Declines in dopaminergic tone, reduced neurotrophic support (including BDNF), and impaired prefrontal inhibitory control weaken endogenous buffers against both pain amplification and craving. In this sense, the physiological neuroprogression of ageing may mirror the pathological neuroprogression observed in addiction, as both processes constrain adaptive flexibility and increase reliance on external regulators—such as opioids, repetitive behaviours, or relational scaffolding—to achieve emotional stability [212].
Recognising this shared continuum reframes the task of clinical care. The goal extends beyond symptom suppression toward the restoration of neuroaffective balance: supporting the ageing brain’s capacity to experience sensation without suffering, desire without compulsion, and memory without persistent distress. This perspective supports a neuropsychiatric model of pain care in which treatment aims to recalibrate the dialogue between cortical control and limbic experience, aligning neurochemistry with meaning and context.
Within this framework, pharmacological and psychological interventions should be coordinated rather than sequential [213,214,215]. Mechanism-guided agents—such as buprenorphine, tapentadol, or serotonin–norepinephrine reuptake inhibitors—may be combined with cognitive-behavioural, mindfulness-based, and rehabilitative interventions to co-modulate circuits involved in salience attribution, affect regulation, and motivational drive [134,135,216,217,218,219,220]. Effective opioid stewardship in older adults therefore extends beyond the binary opposition of analgesia versus addiction, emphasising the continuous integration of functional, affective, and cognitive monitoring, with the aim of preserving emotional, cognitive, and motivational homeostasis in a brain with declining adaptive plasticity [212].
Future research should prioritise integrative biomarkers capable of linking biological and experiential domains. Neuroinflammatory indices (e.g., IL-6, TNF-α, CRP, microglial PET imaging) combined with functional connectomic measures of affective integration (e.g., PFC–ACC–insula coupling) may help quantify how pharmacological and behavioural interventions converge on shared neural pathways [221,222,223].
Ultimately, chronic pain in ageing emerges as a disorder of affective integration rather than a purely sensory dysfunction. Its treatment is therefore not only therapeutic but reconstructive: supporting the brain’s effort to maintain coherence, motivation, and identity in the face of neurodegenerative vulnerability. In this light, to treat pain in older adults is to defend—and where possible restore—the emotional architecture of the self, reframing suffering from a marker of decline into a signal for adaptive, mechanism-guided care.
Author Contributions
Conceptualization, M.G.C., I.M. and A.G.I.M.; methodology, M.G.C., I.M. and A.G.I.M.; investigation, L.M., A.B., R.M., R.R., G.G., C.T. and F.D.R.; writing—original draft preparation, M.G.C.; writing—review and editing, All Authors; supervision, A.G.I.M. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
This study did not generate or analyse any original datasets. All data supporting the conclusions of this review are derived from previously published studies, which are cited in the reference list. Data sharing is therefore not applicable.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| 5-HT | 5-Hydroxytryptamine (Serotonin) |
| 6MWT | Six-Minute Walk Test |
| ACC | Anterior Cingulate Cortex |
| ADLs | Activities of Daily Living |
| BDNF | Brain-Derived Neurotrophic Factor |
| BNST | Bed Nucleus of the Stria Terminalis |
| BP | Blood Pressure |
| BPI | Brief Pain Inventory |
| CB1 | Cannabinoid Receptor Type 1 |
| CBT | Cognitive Behavioral Therapy |
| CDC | Centers for Disease Control and Prevention |
| CHF | Congestive Heart Failure |
| CKD | Chronic Kidney Disease |
| CNS | Central Nervous System |
| CRF | Corticotropin-Releasing Factor |
| CRP | C-Reactive Protein |
| ECG | Electrocardiogram |
| EMCDDA | European Monitoring Centre for Drugs and Drug Addiction |
| ER | Extended Release |
| ESS | Epworth Sleepiness Scale |
| GABA | Gamma-Aminobutyric Acid |
| GAD-7 | Generalized Anxiety Disorder 7-Item Scale |
| GDS | Geriatric Depression Scale |
| HADS | Hospital Anxiety fnd Depression Scale |
| HPA | Hypothalamic–Pituitary–Adrenal (Axis) |
| IL-1β | Interleukin-1 Beta |
| IL-6 | Interleukin-6 |
| IR | Immediate Release |
| KOR | Kappa Opioid Receptor |
| LTD | Long-Term Depression |
| LTP | Long-Term Potentiation |
| MAOIs | Monoamine Oxidase Inhibitors |
| MoCA | Montreal Cognitive Assessment |
| MORs | µ-Opioid Receptors |
| MOUD | Medications For Opioid Use Disorder |
| mPFC | Medial Prefrontal Cortex |
| NAc | Nucleus Accumbens |
| NE | Norepinephrine |
| NICE | National Institute for Health and Care Excellence |
| NIHR | National Institute for Health Research |
| NMDA | N-Methyl-D-Aspartate |
| OIH | Opioid-Induced Hyperalgesia |
| OUD | Opioid Use Disorder |
| PAG | Periaqueductal Gray |
| PET | Positron Emission Tomography |
| PFC | Prefrontal Cortex |
| PPI | Proton Pump Inhibitors |
| PRISMA | Preferred Reporting Items for Systematic Reviews and Meta-Analyses |
| QT | QT Interval |
| SL | Sublingual |
| SNRIs | Serotonin-Norepinephrine Reuptake Inhibitors |
| SSRIs | Selective Serotonin Reuptake Inhibitors |
| TD | Transdermal |
| TNF-α | Tumor Necrosis Factor Alpha |
| TUG | Timed Up and Go Test |
| VAS | Visual Analog Scale |
| VTA | Ventral Tegmental Area |
| WHO | World Health Organization |
References
- Potru, S.; Tang, Y.L. Chronic Pain, Opioid Use Disorder, and Clinical Management Among Older Adults. Focus 2021, 19, 294–302. [Google Scholar] [CrossRef] [PubMed]
- Doan, L.; Manders, T.; Wang, J. Neuroplasticity underlying the comorbidity of pain and depression. Neural Plast. 2015, 2015, 504691. [Google Scholar] [CrossRef] [PubMed]
- National Academies of Sciences, Engineering, and Medicine. Pain Management and the Opioid Epidemic: Balancing Societal and Individual Benefits and Risks of Prescription Opioid Use; The National Academies Press: Washington, DC, USA, 2017; p. 482. [Google Scholar]
- Schieber, L.Z.; Guy, G.P., Jr.; Seth, P.; Losby, J.L. Variation in Adult Outpatient Opioid Prescription Dispensing by Age and Sex—United States, 2008–2018. MMWR Morb. Mortal. Wkly. Rep. 2020, 69, 298–302. [Google Scholar] [CrossRef] [PubMed]
- Melzack, R. From the gate to the neuromatrix. Pain 1999, 82, S121–S126. [Google Scholar] [CrossRef]
- Apkarian, A.V.; Baliki, M.N.; Farmer, M.A. Predicting transition to chronic pain. Curr. Opin. Neurol. 2013, 26, 360–367. [Google Scholar] [CrossRef]
- Molton, I.R.; Terrill, A.L. Overview of persistent pain in older adults. Am. Psychol. 2014, 69, 197–207. [Google Scholar] [CrossRef]
- Cole, L.J.; Farrell, M.J.; Gibson, S.J.; Egan, G.F. Age-related differences in pain sensitivity and regional brain activity evoked by noxious pressure. Neurobiol. Aging 2010, 31, 494–503. [Google Scholar] [CrossRef]
- Chou, R.; Fanciullo, G.J.; Fine, P.G.; Adler, J.A.; Ballantyne, J.C.; Davies, P.; Donovan, M.I.; Fishbain, D.A.; Foley, K.M.; Fudin, J.; et al. Clinical guidelines for the use of chronic opioid therapy in chronic noncancer pain. J. Pain 2009, 10, 113–130.e22. [Google Scholar] [CrossRef]
- Apkarian, A.V.; Baliki, M.N.; Geha, P.Y. Towards a theory of chronic pain. Prog. Neurobiol. 2009, 87, 81–97. [Google Scholar] [CrossRef]
- Morton, D.L.; Sandhu, J.S.; Jones, A.K. Brain imaging of pain: State of the art. J. Pain Res. 2016, 9, 613–624. [Google Scholar] [CrossRef]
- Horta, M.; Wright, K.; Lighthall, N.R.; Fillingim, R.B. Choosing well: A narrative review on chronic pain and decision-making. J. Pain 2025, 35, 105525. [Google Scholar] [CrossRef] [PubMed]
- Mansour, A.R.; Farmer, M.A.; Baliki, M.N.; Apkarian, A.V. Chronic pain: The role of learning and brain plasticity. Restor. Neurol. Neurosci. 2014, 32, 129–139. [Google Scholar] [CrossRef] [PubMed]
- Baliki, M.N.; Apkarian, A.V. Nociception, Pain, Negative Moods, and Behavior Selection. Neuron 2015, 87, 474–491. [Google Scholar] [CrossRef] [PubMed]
- Koob, G.F.; Volkow, N.D. Neurobiology of addiction: A neurocircuitry analysis. Lancet Psychiatry 2016, 3, 760–773. [Google Scholar] [CrossRef]
- Koob, G.F. Drug Addiction: Hyperkatifeia/Negative Reinforcement as a Framework for Medications Development. Pharmacol. Rev. 2021, 73, 163–201. [Google Scholar] [CrossRef]
- Fields, H.L. How expectations influence pain. Pain 2018, 159, S3–S10. [Google Scholar] [CrossRef]
- Dresp-Langley, B. From Reward to Anhedonia-Dopamine Function in the Global Mental Health Context. Biomedicines 2023, 11, 2469. [Google Scholar] [CrossRef]
- Leknes, S.; Tracey, I. A common neurobiology for pain and pleasure. Nat. Rev. Neurosci. 2008, 9, 314–320. [Google Scholar] [CrossRef]
- Mitsi, V.; Zachariou, V. Modulation of pain, nociception, and analgesia by the brain reward center. Neuroscience 2016, 338, 81–92. [Google Scholar] [CrossRef]
- Chen, L.; Zeng, L.; Li, W.; Li, J.-S. The Activation of Hippocampal Microglial Cells and Their Role in the Regulation of Pain. J. Integr. Neurosci. 2025, 24, 27730. [Google Scholar] [CrossRef]
- Campos, A.C.P.; Antunes, G.F.; Matsumoto, M.; Pagano, R.L.; Martinez, R.C.R. Neuroinflammation, Pain and Depression: An Overview of the Main Findings. Front. Psychol. 2020, 11, 1825. [Google Scholar] [CrossRef]
- Shraim, M.A.; Massé-Alarie, H.; Farrell, M.J.; Cavaleri, R.; Loggia, M.L.; Hodges, P.W. Neuroinflammatory activation in sensory and motor regions of the cortex is related to sensorimotor function in individuals with low back pain maintained by nociplastic mechanisms: A preliminary proof-of-concept study. Eur. J. Pain 2024, 28, 1607–1626. [Google Scholar] [CrossRef] [PubMed]
- Maletic, V.; Raison, C. Integrated neurobiology of bipolar disorder. Front. Psychiatry 2014, 5, 98. [Google Scholar] [CrossRef] [PubMed]
- Rouch, I.; Edjolo, A.; Laurent, B.; Pongan, E.; Dartigues, J.F.; Amieva, H. Association between chronic pain and long-term cognitive decline in a population-based cohort of elderly participants. Pain 2021, 162, 552–560. [Google Scholar] [CrossRef] [PubMed]
- Ezzati, A.; Zammit, A.R.; Lipton, M.L.; Lipton, R.B. The relationship between hippocampal volume, chronic pain, and depressive symptoms in older adults. Psychiatry Res. Neuroimaging 2019, 289, 10–12. [Google Scholar] [CrossRef]
- Seidel, M.F.; Hügle, T.; Morlion, B.; Koltzenburg, M.; Chapman, V.; MaassenVanDenBrink, A.; Lane, N.E.; Perrot, S.; Zieglgänsberger, W. Neurogenic inflammation as a novel treatment target for chronic pain syndromes. Exp. Neurol. 2022, 356, 114108. [Google Scholar] [CrossRef]
- Tu, Y.; Jung, M.; Gollub, R.L.; Napadow, V.; Gerber, J.; Ortiz, A.; Lang, C.; Mawla, I.; Shen, W.; Chan, S.T.; et al. Abnormal medial prefrontal cortex functional connectivity and its association with clinical symptoms in chronic low back pain. Pain 2019, 160, 1308–1318. [Google Scholar] [CrossRef]
- Shen, W.; Tu, Y.; Gollub, R.L.; Ortiz, A.; Napadow, V.; Yu, S.; Wilson, G.; Park, J.; Lang, C.; Jung, M.; et al. Visual network alterations in brain functional connectivity in chronic low back pain: A resting state functional connectivity and machine learning study. Neuroimage Clin. 2019, 22, 101775. [Google Scholar] [CrossRef]
- Borsook, D. A future without chronic pain: Neuroscience and clinical research. Cerebrum 2012, 2012, 7. [Google Scholar]
- Pinto, C.B.; Bielefeld, J.; Barroso, J.; Yip, B.; Huang, L.; Schnitzer, T.; Apkarian, A.V. Chronic pain domains and their relationship to personality, abilities, and brain networks. Pain 2023, 164, 59–71. [Google Scholar] [CrossRef]
- Li, A.; Peng, Y.B. Comorbidity of Depression and Pain: A review of shared contributing mechanisms. J. Neurol. Neuromed. 2017, 2, 4–11. [Google Scholar] [CrossRef]
- Cohen, S.P.; Vase, L.; Hooten, W.M. Chronic pain: An update on burden, best practices, and new advances. Lancet 2021, 397, 2082–2097. [Google Scholar] [CrossRef] [PubMed]
- Makris, U.E.; Abrams, R.C.; Gurland, B.; Reid, M.C. Management of persistent pain in the older patient: A clinical review. JAMA 2014, 312, 825–836. [Google Scholar] [CrossRef] [PubMed]
- Chu, L.F.; Angst, M.S.; Clark, D. Opioid-induced Hyperalgesia in Humans: Molecular Mechanisms and Clinical Considerations. Clin. J. Pain 2008, 24, 479–496. [Google Scholar] [CrossRef] [PubMed]
- Mercadante, S.; Arcuri, E.; Santoni, A. Opioid-Induced Tolerance and Hyperalgesia. CNS Drugs 2019, 33, 943–955. [Google Scholar] [CrossRef]
- Jamison, R.N.; Mao, J. Opioid Analgesics. Mayo Clin. Proc. 2015, 90, 957–968. [Google Scholar] [CrossRef]
- Schwan, J.; Sclafani, J.; Tawfik, V.L. Chronic Pain Management in the Elderly. Anesthesiol. Clin. 2019, 37, 547–560. [Google Scholar] [CrossRef]
- Tinnirello, A.; Mazzoleni, S.; Santi, C. Chronic Pain in the Elderly: Mechanisms and Distinctive Features. Biomolecules 2021, 11, 1256. [Google Scholar] [CrossRef]
- Dagnino, A.P.A.; Campos, M.M. Chronic Pain in the Elderly: Mechanisms and Perspectives. Front. Hum. Neurosci. 2022, 16, 736688. [Google Scholar] [CrossRef]
- Farrell, M.J. Age-related changes in the structure and function of brain regions involved in pain processing. Pain Med. 2012, 13, S37–S43. [Google Scholar] [CrossRef]
- Muscatell, K.A.; Brosso, S.N.; Humphreys, K.L. Socioeconomic status and inflammation: A meta-analysis. Mol. Psychiatry 2020, 25, 2189–2199. [Google Scholar] [CrossRef]
- Aaron, R.V.; Ravyts, S.G.; Carnahan, N.D.; Bhattiprolu, K.; Harte, N.; McCaulley, C.C.; Vitalicia, L.; Rogers, A.B.; Wegener, S.T.; Dudeney, J. Prevalence of Depression and Anxiety Among Adults With Chronic Pain: A Systematic Review and Meta-Analysis. JAMA Netw. Open 2025, 8, e250268. [Google Scholar] [CrossRef] [PubMed]
- Bair, M.J.; Robinson, R.L.; Katon, W.; Kroenke, K. Depression and Pain Comorbidity: A Literature Review. Arch. Intern. Med. 2003, 163, 2433–2445. [Google Scholar] [CrossRef] [PubMed]
- Zis, P.; Daskalaki, A.; Bountouni, I.; Sykioti, P.; Varrassi, G.; Paladini, A. Depression and chronic pain in the elderly: Links and management challenges. Clin. Interv. Aging 2017, 12, 709–720. [Google Scholar] [CrossRef] [PubMed]
- Karp, J.F.; Rollman, B.L.; Reynolds, C.F., III; Morse, J.Q.; Lotrich, F.; Mazumdar, S.; Morone, N.; Weiner, D.K. Addressing Both Depression and Pain in Late Life: The Methodology of the ADAPT Study. Pain Med. 2012, 13, 405–418. [Google Scholar] [CrossRef]
- Saint-Martin, H.; Rouch, I.; Laurent, B.; Edjolo, A.; Pongan, E.; Herrmann, M.; Creac’h, C.; Pérès, K.; Amieva, H.; Dorey, J.M. Chronic pain in older adults with psychiatric disorders: The DoCPPA study protocol. BMC Geriatr. 2025, 25, 661. [Google Scholar] [CrossRef]
- Muscatell, K.A.; Dedovic, K.; Slavich, G.M.; Jarcho, M.R.; Breen, E.C.; Bower, J.E.; Irwin, M.R.; Eisenberger, N.I. Neural mechanisms linking social status and inflammatory responses to social stress. Soc. Cogn. Affect. Neurosci. 2016, 11, 915–922. [Google Scholar] [CrossRef]
- Eisenberger, N.I. The neural bases of social pain: Evidence for shared representations with physical pain. Psychosom. Med. 2012, 74, 126–135. [Google Scholar] [CrossRef]
- Eisenberger, N.I. The pain of social disconnection: Examining the shared neural underpinnings of physical and social pain. Nat. Rev. Neurosci. 2012, 13, 421–434. [Google Scholar] [CrossRef]
- Dantzer, R.; O’Connor, J.; Freund, G.; Johnson, R.; Kelley, K. From inflammation to sickness and depression: When the immune system subjugates the brain. Nat. Rev. Neurosci. 2008, 9, 46–56. [Google Scholar] [CrossRef]
- Turkheimer, F.E.; Veronese, M.; Mondelli, V.; Cash, D.; Pariante, C.M. Sickness behaviour and depression: An updated model of peripheral-central immunity interactions. Brain Behav. Immun. 2023, 111, 202–210. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Wang, M.; Chen, D.; Jiang, X.; Xiong, Z. Inflammatory biomarkers in older adults with frailty: A systematic review and meta-analysis of cross-sectional studies. Aging Clin. Exp. Res. 2022, 34, 971–987. [Google Scholar] [CrossRef] [PubMed]
- Gracely, R.H.; Geisser, M.E.; Giesecke, T.; Grant, M.A.; Petzke, F.; Williams, D.A.; Clauw, D.J. Pain catastrophizing and neural responses to pain among persons with fibromyalgia. Brain 2004, 127, 835–843. [Google Scholar] [CrossRef] [PubMed]
- Loggia, M.L.; Berna, C.; Kim, J.; Cahalan, C.M.; Martel, M.O.; Gollub, R.L.; Wasan, A.D.; Napadow, V.; Edwards, R.R. The lateral prefrontal cortex mediates the hyperalgesic effects of negative cognitions in chronic pain patients. J. Pain 2015, 16, 692–699. [Google Scholar] [CrossRef]
- Fried, L.P.; Tangen, C.M.; Walston, J.; Newman, A.B.; Hirsch, C.; Gottdiener, J.; Seeman, T.; Tracy, R.; Kop, W.J.; Burke, G.; et al. Frailty in older adults: Evidence for a phenotype. J. Gerontol. A Biol. Sci. Med. Sci. 2001, 56, M146–M156. [Google Scholar] [CrossRef]
- Ma, L.; Chan, P. Understanding the Physiological Links Between Physical Frailty and Cognitive Decline. Aging Dis. 2020, 11, 405–418. [Google Scholar] [CrossRef]
- Soysal, P.; Stubbs, B.; Lucato, P.; Luchini, C.; Solmi, M.; Peluso, R.; Sergi, G.; Isik, A.T.; Manzato, E.; Maggi, S.; et al. Inflammation and frailty in the elderly: A systematic review and meta-analysis. Ageing Res. Rev. 2016, 31, 1–8. [Google Scholar] [CrossRef]
- Elman, I.; Pustilnik, A.; Borsook, D. Beating pain with psychedelics: Matter over mind? Neurosci. Biobehav. Rev. 2022, 134, 104482. [Google Scholar] [CrossRef]
- Rastogi, R.; Meek, B. Management of chronic pain in elderly, frail patients: Finding a suitable, personalized method of control. Clin. Interv. Aging 2013, 8, 37–46. [Google Scholar] [CrossRef]
- By the American Geriatrics Society 2015 Beers Criteria Update Expert Panel. American Geriatrics Society 2015 Updated Beers Criteria for Potentially Inappropriate Medication Use in Older Adults. J. Am. Geriatr. Soc. 2015, 63, 2227–2246.
- Beyth, R.J.; Shorr, R.I. Epidemiology of adverse drug reactions in the elderly by drug class. Drugs Aging 1999, 14, 231–239. [Google Scholar] [CrossRef]
- Clegg, A.; Young, J.B. Which medications to avoid in people at risk of delirium: A systematic review. Age Ageing 2011, 40, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Swart, L.M.; van der Zanden, V.; Spies, P.E.; de Rooij, S.E.; van Munster, B.C. The Comparative Risk of Delirium with Different Opioids: A Systematic Review. Drugs Aging 2017, 34, 437–443. [Google Scholar] [CrossRef] [PubMed]
- Virnes, R.E.; Tiihonen, M.; Karttunen, N.; van Poelgeest, E.P.; van der Velde, N.; Hartikainen, S. Opioids and Falls Risk in Older Adults: A Narrative Review. Drugs Aging 2022, 39, 199–207. [Google Scholar] [CrossRef] [PubMed]
- Navratilova, E.; Porreca, F. Reward and motivation in pain and pain relief. Nat. Neurosci. 2014, 17, 1304–1312. [Google Scholar] [CrossRef]
- Yang, S.; Chang, M.C. Chronic Pain: Structural and Functional Changes in Brain Structures and Associated Negative Affective States. Int. J. Mol. Sci. 2019, 20, 3130. [Google Scholar] [CrossRef]
- Apkarian, A.V.; Hashmi, J.A.; Baliki, M.N. Pain and the brain: Specificity and plasticity of the brain in clinical chronic pain. Pain 2011, 152, S49–S64. [Google Scholar] [CrossRef]
- Zhang, Z.; Gewandter, J.S.; Geha, P. Brain Imaging Biomarkers for Chronic Pain. Front. Neurol. 2022, 12, 734821. [Google Scholar] [CrossRef]
- Volkow, N.D.; Morales, M. The Brain on Drugs: From Reward to Addiction. Cell 2015, 162, 712–725. [Google Scholar] [CrossRef]
- Nees, F.; Becker, S. Psychological Processes in Chronic Pain: Influences of Reward and Fear Learning as Key Mechanisms—Behavioral Evidence, Neural Circuits, and Maladaptive Changes. Neuroscience 2018, 387, 72–84. [Google Scholar] [CrossRef]
- Sheng, J.; Liu, S.; Wang, Y.; Cui, R.; Zhang, X. The Link between Depression and Chronic Pain: Neural Mechanisms in the Brain. Neural Plast. 2017, 2017, 9724371. [Google Scholar] [CrossRef]
- Taylor, A.M.W.; Becker, S.; Schweinhardt, P.; Cahill, C. Mesolimbic dopamine signaling in acute and chronic pain: Implications for motivation, analgesia, and addiction. Pain 2016, 157, 1194–1198. [Google Scholar] [CrossRef]
- Gee, T.A.; Weintraub, N.C.; Lu, D.; Phelps, C.E.; Navratilova, E.; Heien, M.L.; Porreca, F. A pain-induced tonic hypodopaminergic state augments phasic dopamine release in the nucleus accumbens. Pain 2020, 161, 2376–2384. [Google Scholar] [CrossRef] [PubMed]
- Loggia, M.L.; Kim, J.; Gollub, R.L.; Vangel, M.G.; Kirsch, I.; Kong, J.; Wasan, A.D.; Napadow, V. Default mode network connectivity encodes clinical pain: An arterial spin labeling study. Pain 2013, 154, 24–33. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Mawla, I.; Kong, J.; Lee, J.; Gerber, J.; Ortiz, A.; Kim, H.; Chan, S.T.; Loggia, M.L.; Wasan, A.D.; et al. Somatotopically specific primary somatosensory connectivity to salience and default mode networks encodes clinical pain. Pain 2019, 160, 1594–1605. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.Y.; Lee, S.; Son, S.J.; Park, R.W.; Park, B. Identifying affective state via clustering of temporal variability in limbic activity and mediating role of negative emotion. Mol. Psychiatry 2025, 31, 1546–1555. [Google Scholar] [CrossRef]
- Goldstein, R.Z.; Volkow, N.D. Dysfunction of the prefrontal cortex in addiction: Neuroimaging findings and clinical implications. Nat. Rev. Neurosci. 2011, 12, 652–669. [Google Scholar] [CrossRef]
- Vachon-Presseau, E.; Centeno, M.V.; Ren, W.; Berger, S.E.; Tétreault, P.; Ghantous, M.; Baria, A.; Farmer, M.; Baliki, M.N.; Schnitzer, T.J.; et al. The Emotional Brain as a Predictor and Amplifier of Chronic Pain. J. Dent. Res. 2016, 95, 605–612. [Google Scholar] [CrossRef]
- Arif, S.; Madadi, S.; Haidary, M. Neurobiological convergence of pain and substance use disorders: A focus on opioidergic and mesolimbic circuitry. Addict. Neurosci. 2025, 16, 100220. [Google Scholar] [CrossRef]
- Pergolizzi, J.V., Jr.; Raffa, R.B.; Rosenblatt, M.H. Opioid withdrawal symptoms, a consequence of chronic opioid use and opioid use disorder: Current understanding and approaches to management. J. Clin. Pharm. Ther. 2020, 45, 892–903. [Google Scholar] [CrossRef]
- Land, B.B.; Bruchas, M.R.; Lemos, J.C.; Xu, M.; Melief, E.J.; Chavkin, C. The Dysphoric Component of Stress Is Encoded by Activation of the Dynorphin κ-Opioid System. J. Neurosci. 2008, 28, 407–414. [Google Scholar] [CrossRef]
- Marchette, R.C.N.; Vendruscolo, L.F.; Koob, G.F. The Dynorphin/-Opioid Receptor System at the Interface of Hyperalgesia/Hyperkatifeia and Addiction. Curr. Addict. Rep. 2025, 12, 11. [Google Scholar] [CrossRef]
- Sapolsky, R.M. Glucocorticoids and hippocampal atrophy in neuropsychiatric disorders. Arch. Gen. Psychiatry 2000, 57, 925–935. [Google Scholar] [CrossRef] [PubMed]
- Suri, D.; Vaidya, V.A. Glucocorticoid regulation of brain-derived neurotrophic factor: Relevance to hippocampal structural and functional plasticity. Neuroscience 2013, 239, 196–213. [Google Scholar] [CrossRef] [PubMed]
- Ji, R.R.; Nackley, A.; Huh, Y.; Terrando, N.; Maixner, W. Neuroinflammation and Central Sensitization in Chronic and Widespread Pain. Anesthesiology 2018, 129, 343–366. [Google Scholar] [CrossRef] [PubMed]
- Maletic, V.; Raison, C.L. Neurobiology of depression, fibromyalgia and neuropathic pain. Front. Biosci. 2009, 14, 5291–5338. [Google Scholar] [CrossRef]
- Correia, A.S.; Cardoso, A.; Vale, N. BDNF Unveiled: Exploring Its Role in Major Depression Disorder Serotonergic Imbalance and Associated Stress Conditions. Pharmaceutics 2023, 15, 2081. [Google Scholar] [CrossRef]
- Yang, T.; Nie, Z.; Shu, H.; Kuang, Y.; Chen, X.; Cheng, J.; Yu, S.; Liu, H. The Role of BDNF on Neural Plasticity in Depression. Front. Cell. Neurosci. 2020, 14, 82. [Google Scholar] [CrossRef]
- Lukkahatai, N.; Ong, I.L.; Benjasirisan, C.; Saligan, L.N. Brain-Derived Neurotrophic Factor (BDNF) as a Marker of Physical Exercise or Activity Effectiveness in Fatigue, Pain, Depression, and Sleep Disturbances: A Scoping Review. Biomedicines 2025, 13, 332. [Google Scholar] [CrossRef]
- Lee, M.; Silverman, S.M.; Hansen, H.; Patel, V.B.; Manchikanti, L. A comprehensive review of opioid-induced hyperalgesia. Pain Physician 2011, 14, 145–161. [Google Scholar] [CrossRef]
- Sinha, R. Chronic stress, drug use, and vulnerability to addiction. Ann. N. Y Acad. Sci. 2008, 1141, 105–130. [Google Scholar] [CrossRef] [PubMed]
- Bushnell, M.C.; Ceko, M.; Low, L.A. Cognitive and emotional control of pain and its disruption in chronic pain. Nat. Rev. Neurosci. 2013, 14, 502–511. [Google Scholar] [CrossRef] [PubMed]
- Wakaizumi, K.; Vigotsky, A.D.; Jabakhanji, R.; Abdallah, M.; Barroso, J.; Schnitzer, T.J.; Apkarian, A.V.; Baliki, M.N. Psychosocial, Functional, and Emotional Correlates of Long-Term Opioid Use in Patients with Chronic Back Pain: A Cross-Sectional Case-Control Study. Pain Ther. 2021, 10, 691–709. [Google Scholar] [CrossRef] [PubMed]
- Martucci, K.T.; Ng, P.; Mackey, S. Neuroimaging chronic pain: What have we learned and where are we going? Future Neurol. 2014, 9, 615–626. [Google Scholar] [CrossRef]
- Martucci, K.T.; Mackey, S.C. Neuroimaging of Pain: Human Evidence and Clinical Relevance of Central Nervous System Processes and Modulation. Anesthesiology 2018, 128, 1241–1254. [Google Scholar] [CrossRef]
- Kucyi, A.; Moayedi, M.; Weissman-Fogel, I.; Goldberg, M.B.; Freeman, B.V.; Tenenbaum, H.C.; Davis, K.D. Enhanced Medial Prefrontal-Default Mode Network Functional Connectivity in Chronic Pain and Its Association with Pain Rumination. J. Neurosci. 2014, 34, 3969–3975. [Google Scholar] [CrossRef]
- Schmaal, L.; Pozzi, E.; Ho, T.C.; van Velzen, L.S.; Veer, I.M.; Opel, N.; Van Someren, E.J.W.; Han, L.K.M.; Aftanas, L.; Aleman, A.; et al. ENIGMA MDD: Seven years of global neuroimaging studies of major depression through worldwide data sharing. Transl. Psychiatry 2020, 10, 172. [Google Scholar] [CrossRef]
- Flor, H. Psychological pain interventions and neurophysiology: Implications for a mechanism-based approach. Am. Psychol. 2014, 69, 188–196. [Google Scholar] [CrossRef]
- Kim, D.J.; Jassar, H.; Lim, M.; Nascimento, T.D.; DaSilva, A.F. Dopaminergic Regulation of Reward System Connectivity Underpins Pain and Emotional Suffering in Migraine. J. Pain Res. 2021, 14, 631–643. [Google Scholar] [CrossRef]
- Martikainen, I.K.; Nuechterlein, E.B.; Peciña, M.; Love, T.M.; Cummiford, C.M.; Green, C.R.; Stohler, C.S.; Zubieta, J.K. Chronic Back Pain Is Associated with Alterations in Dopamine Neurotransmission in the Ventral Striatum. J. Neurosci. 2015, 35, 9957–9965. [Google Scholar] [CrossRef]
- Baliki, M.N.; Mansour, A.R.; Baria, A.T.; Apkarian, A.V. Functional Reorganization of the Default Mode Network across Chronic Pain Conditions. PLoS ONE 2014, 9, e106133. [Google Scholar] [CrossRef]
- Hu, Y.; Zhao, C.; Zhao, H.; Qiao, J. Abnormal functional connectivity of the nucleus accumbens subregions mediates the association between anhedonia and major depressive disorder. BMC Psychiatry 2023, 23, 282. [Google Scholar] [CrossRef] [PubMed]
- Koob, G.F.; Schulkin, J. Addiction and stress: An allostatic view. Neurosci. Biobehav. Rev. 2019, 106, 245–262. [Google Scholar] [CrossRef] [PubMed]
- Voicu, V.; Toader, C.; Șerban, M.; Covache-Busuioc, R.A.; Ciurea, A.V. Systemic Neurodegeneration and Brain Aging: Multi-Omics Disintegration, Proteostatic Collapse, and Network Failure Across the CNS. Biomedicines 2025, 13, 2025. [Google Scholar] [CrossRef] [PubMed]
- El Hayek, S. Addiction and Neurodegenerative Disorders: Unraveling the Nexus; CRC Press: Boca Raton, FL, USA, 2025. [Google Scholar]
- Wenzel, J.M.; Cheer, J.F. Endocannabinoid Regulation of Reward and Reinforcement through Interaction with Dopamine and Endogenous Opioid Signaling. Neuropsychopharmacology 2018, 43, 103–115. [Google Scholar] [CrossRef]
- Sun, Y.; Hasbi, A.; George, S.R. G Protein-Coupled Receptor Heteromers in Brain: Functional and Therapeutic Importance in Neuropsychiatric Disorders. Annu. Rev. Pharmacol. Toxicol. 2025, 65, 215–236. [Google Scholar] [CrossRef]
- Secondulfo, C.; Mazzeo, F.; Pastorino, G.M.G.; Vicidomini, A.; Meccariello, R.; Operto, F.F. Opioid and Cannabinoid Systems in Pain: Emerging Molecular Mechanisms and Use in Clinical Practice, Health, and Fitness. Int. J. Mol. Sci. 2024, 25, 9407. [Google Scholar] [CrossRef]
- Meccariello, R.; Aseer, K.R.; Kabir, M.; Santoro, A. Editorial: Multifaceted cannabinoids: Regulators of normal and pathological function in metabolic and endocrine organs, volume II. Front. Endocrinol. 2024, 15, 1503017. [Google Scholar] [CrossRef]
- Mato, S.; Pilar-Cuéllar, F.; Valdizán, E.M.; González-Maeso, J.; Rodríguez-Puertas, R.; Meana, J.; Sallés, J.; Crespo-Facorro, B.; Pazos, Á. Selective up-regulation of cannabinoid CB1 receptor coupling to Go-proteins in suicide victims with mood disorders. Biochem. Pharmacol. 2018, 157, 258–265. [Google Scholar] [CrossRef]
- Llorente-Ovejero, A.; Bengoetxea de Tena, I.; Martínez-Gardeazabal, J.; Moreno-Rodríguez, M.; Lombardero, L.; Manuel, I.; Rodríguez-Puertas, R. Cannabinoid Receptors and Glial Response Following a Basal Forebrain Cholinergic Lesion. ACS Pharmacol. Transl. Sci. 2022, 5, 791–802. [Google Scholar] [CrossRef]
- Alton, H.; Linz, E.; Bi, G.-H.; Soler-Cedeno, O.; Maras, M.; Xi, Z.-X. Cannabinoid CB1 receptor and mu-opioid receptor interaction: New insights from conditional knockout mice. Neuropsychopharmacology 2025, 51, 506–518. [Google Scholar] [CrossRef]
- Meda, R.T.; Nuguru, S.P.; Rachakonda, S.; Sripathi, S.; Khan, M.I.; Patel, N. Chronic Pain-Induced Depression: A Review of Prevalence and Management. Cureus 2022, 14, e28416. [Google Scholar] [CrossRef] [PubMed]
- Garland, E.L.; Trøstheim, M.; Eikemo, M.; Ernst, G.; Leknes, S. Anhedonia in chronic pain and prescription opioid misuse. Psychol. Med. 2020, 50, 1977–1988. [Google Scholar] [CrossRef] [PubMed]
- Naqvi, N.H.; Bechara, A. The insula and drug addiction: An interoceptive view of pleasure, urges, and decision-making. Brain Struct. Funct. 2010, 214, 435–450. [Google Scholar] [CrossRef] [PubMed]
- Höijer, I.; Ilonen, T.; Löyttyniemi, E.; Salokangas, R.K.R. Onset Age of Substance Use and Neuropsychological Performance in Hospital Patients. Clin. Neuropsychiatry 2020, 17, 271–280. [Google Scholar]
- Uhl, G.R.; Koob, G.F.; Cable, J. The neurobiology of addiction. Ann. N. Y Acad. Sci. 2019, 1451, 5–28. [Google Scholar] [CrossRef]
- Kuerbis, A.; Sacco, P.; Blazer, D.G.; Moore, A.A. Substance abuse among older adults. Clin. Geriatr. Med. 2014, 30, 629–654. [Google Scholar] [CrossRef]
- Passik, S.D.; Kirsh, K.L.; Webster, L. Pseudoaddiction revisited: A commentary on clinical and historical considerations. Pain Manag. 2011, 1, 239–248. [Google Scholar] [CrossRef]
- McCarberg, B.H.; Barkin, R.L. Long-acting opioids for chronic pain: Pharmacotherapeutic opportunities to enhance compliance, quality of life, and analgesia. Am. J. Ther. 2001, 8, 181–186. [Google Scholar] [CrossRef]
- Volkow, N.D.; McLellan, A.T. Opioid Abuse in Chronic Pain—Misconceptions and Mitigation Strategies. N. Engl. J. Med. 2016, 374, 1253–1263. [Google Scholar] [CrossRef]
- Manchikanti, L.; Kaye, A.M.; Knezevic, N.N.; McAnally, H.; Slavin, K.; Trescot, A.M.; Blank, S.; Pampati, V.; Abdi, S.; Grider, J.S.; et al. Responsible, Safe, and Effective Prescription of Opioids for Chronic Non-Cancer Pain: American Society of Interventional Pain Physicians (ASIPP) Guidelines. Pain Physician 2017, 20, S3–S92. [Google Scholar] [CrossRef]
- Papaleontiou, M.; Henderson, C.R., Jr.; Turner, B.J.; Moore, A.A.; Olkhovskaya, Y.; Amanfo, L.; Reid, M.C. Outcomes associated with opioid use in the treatment of chronic noncancer pain in older adults: A systematic review and meta-analysis. J. Am. Geriatr. Soc. 2010, 58, 1353–1369. [Google Scholar] [CrossRef] [PubMed]
- Gatchel, R.J.; Peng, Y.B.; Peters, M.L.; Fuchs, P.N.; Turk, D.C. The biopsychosocial approach to chronic pain: Scientific advances and future directions. Psychol. Bull. 2007, 133, 581–624. [Google Scholar] [CrossRef] [PubMed]
- Reid, M.C.; Eccleston, C.; Pillemer, K. Management of chronic pain in older adults. BMJ 2015, 350, h532. [Google Scholar] [CrossRef] [PubMed]
- Haack, M.; Simpson, N.; Sethna, N.; Kaur, S.; Mullington, J. Sleep deficiency and chronic pain: Potential underlying mechanisms and clinical implications. Neuropsychopharmacology 2020, 45, 205–216. [Google Scholar] [CrossRef]
- Saggu, S.; Pless, A.; Dew, E.; Ware, D.; Jiao, K.; Wang, Q. Monoamine signaling and neuroinflammation: Mechanistic connections and implications for neuropsychiatric disorders. Front. Immunol. 2025, 16, 1543730. [Google Scholar] [CrossRef]
- Turk, D.C.; Okifuji, A. Psychological factors in chronic pain: Evolution and revolution. J. Consult. Clin. Psychol. 2002, 70, 678–690. [Google Scholar] [CrossRef]
- Dowell, D.; Ragan, K.R.; Jones, C.M.; Baldwin, G.T.; Chou, R. CDC Clinical Practice Guideline for Prescribing Opioids for Pain—United States, 2022. MMWR Recomm. Rep. 2022, 71, 1–95. [Google Scholar] [CrossRef]
- Krčevski Škvarč, N.; Morlion, B.; Vowles, K.E.; Bannister, K.; Buchsner, E.; Casale, R.; Chenot, J.-F.; Chumbley, G.; Drewes, A.M.; Dom, G.; et al. European clinical practice recommendations on opioids for chronic noncancer pain—Part 2: Special situations. Eur. J. Pain 2021, 25, 969–985. [Google Scholar] [CrossRef]
- Parisi, A.; Roberts, R.L.; Hanley, A.W.; Garland, E.L. Mindfulness-Oriented Recovery Enhancement for Addictive Behavior, Psychiatric Distress, and Chronic Pain: A Multilevel Meta-Analysis of Randomized Controlled Trials. Mindfulness 2022, 13, 2396–2412. [Google Scholar] [CrossRef]
- Hughes, L.S.; Clark, J.; Colclough, J.A.; Dale, E.; McMillan, D. Acceptance and Commitment Therapy (ACT) for Chronic Pain: A Systematic Review and Meta-Analyses. Clin. J. Pain 2017, 33, 552–568. [Google Scholar] [CrossRef] [PubMed]
- Garland, E.L.; Hudak, J.; Hanley, A.W.; Nakamura, Y. Mindfulness-oriented recovery enhancement reduces opioid dose in primary care by strengthening autonomic regulation during meditation. Am. Psychol. 2020, 75, 840–852. [Google Scholar] [CrossRef] [PubMed]
- Hooten, W.M. Chronic Pain and Mental Health Disorders: Shared Neural Mechanisms, Epidemiology, and Treatment. Mayo Clin. Proc. 2016, 91, 955–970. [Google Scholar] [CrossRef] [PubMed]
- Garland, E.L.; Hanley, A.W.; Riquino, M.R.; Reese, S.E.; Baker, A.K.; Salas, K.; Yack, B.P.; Bedford, C.E.; Bryan, M.A.; Atchley, R.; et al. Mindfulness-oriented recovery enhancement reduces opioid misuse risk via analgesic and positive psychological mechanisms: A randomized controlled trial. J. Consult. Clin. Psychol. 2019, 87, 927–940. [Google Scholar] [CrossRef]
- Higgins, C.; Smith, B.H.; Matthews, K. Evidence of opioid-induced hyperalgesia in clinical populations after chronic opioid exposure: A systematic review and meta-analysis. Br. J. Anaesth. 2019, 122, e114–e126. [Google Scholar] [CrossRef]
- Chou, R.; Cruciani, R.A.; Fiellin, D.A.; Compton, P.; Farrar, J.T.; Haigney, M.C.; Inturrisi, C.; Knight, J.R.; Otis-Green, S.; Marcus, S.M.; et al. Methadone safety: A clinical practice guideline from the American Pain Society and College on Problems of Drug Dependence, in collaboration with the Heart Rhythm Society. J. Pain 2014, 15, 321–337. [Google Scholar] [CrossRef]
- Savvas, S.M.; Gibson, S.J. Overview of Pain Management in Older Adults. Clin. Geriatr. Med. 2016, 32, 635–650. [Google Scholar] [CrossRef]
- Evoy, K.E.; Morrison, M.D.; Saklad, S.R. Abuse and Misuse of Pregabalin and Gabapentin. Drugs 2017, 77, 403–426. [Google Scholar] [CrossRef]
- Bykov, K.; Bateman, B.T.; Franklin, J.M.; Vine, S.M.; Patorno, E. Association of Gabapentinoids With the Risk of Opioid-Related Adverse Events in Surgical Patients in the United States. JAMA Netw. Open 2020, 3, e2031647. [Google Scholar] [CrossRef]
- Lunn, M.P.; Hughes, R.A.; Wiffen, P.J. Duloxetine for treating painful neuropathy, chronic pain or fibromyalgia. Cochrane Database Syst. Rev. 2014, 2014, CD007115. [Google Scholar] [CrossRef]
- Likar, R. Transdermal buprenorphine in the management of persistent pain—Safety aspects. Ther. Clin. Risk Manag. 2006, 2, 115–125. [Google Scholar] [PubMed]
- Deeks, E.D. Tapentadol Prolonged Release: A Review in Pain Management. Drugs 2018, 78, 1805–1816. [Google Scholar] [CrossRef] [PubMed]
- Polati, E.; Canonico, P.L.; Schweiger, V.; Collino, M. Tapentadol: An overview of the safety profile. J. Pain Res. 2019, 12, 1569–1576. [Google Scholar] [CrossRef] [PubMed]
- Biondi, D.M.; Xiang, J.; Etropolski, M.; Moskovitz, B. Tolerability and efficacy of tapentadol extended release in elderly patients ≥ 75 years of age with chronic osteoarthritis knee or low back pain. J. Opioid Manag. 2015, 11, 393–403. [Google Scholar] [CrossRef]
- Miotto, K.; Cho, A.K.; Khalil, M.A.; Blanco, K.; Sasaki, J.D.; Rawson, R. Trends in Tramadol: Pharmacology, Metabolism, and Misuse. Anesth. Analg. 2017, 124, 44–51. [Google Scholar] [CrossRef]
- Pergolizzi, J.V.; Raffa, R.B.; Marcum, Z.; Colucci, S.; Ripa, S.R. Safety of buprenorphine transdermal system in the management of pain in older adults. Postgrad. Med. 2017, 129, 92–101. [Google Scholar] [CrossRef]
- Borsook, D.; Hargreaves, R.; Bountra, C.; Porreca, F. Lost but making progress—Where will new analgesic drugs come from? Sci. Transl. Med. 2014, 6, 249sr3. [Google Scholar] [CrossRef]
- Lembke, A.; Ottestad, E.; Schmiesing, C. Patients Maintained on Buprenorphine for Opioid Use Disorder Should Continue Buprenorphine Through the Perioperative Period. Pain Med. 2019, 20, 425–428. [Google Scholar] [CrossRef]
- Alford, D.P.; Compton, P.; Samet, J.H. Acute pain management for patients receiving maintenance methadone or buprenorphine therapy. Ann. Intern. Med. 2006, 144, 127–134. [Google Scholar] [CrossRef]
- Manhapra, A.; Sullivan, M.D.; Ballantyne, J.C.; MacLean, R.R.; Becker, W.C. Complex Persistent Opioid Dependence with Long-term Opioids: A Gray Area That Needs Definition, Better Understanding, Treatment Guidance, and Policy Changes. J. Gen. Intern. Med. 2020, 35, 964–971. [Google Scholar] [CrossRef]
- Kahan, M.; Srivastava, A.; Wilson, L.; Gourlay, D.; Midmer, D. Misuse of and dependence on opioids: Study of chronic pain patients. Can. Fam. Physician 2006, 52, 1081–1087. [Google Scholar] [PubMed]
- Sandhu, S.; Calcaterra, S.L. How Do I Manage Acute Pain for Patients Prescribed Buprenorphine for Opioid Use Disorder? NEJM Evid. 2024, 3, EVIDccon2300275. [Google Scholar] [CrossRef] [PubMed]
- Raub, J.N.; Vettese, T.E. Acute Pain Management in Hospitalized Adult Patients with Opioid Dependence: A Narrative Review and Guide for Clinicians. J. Hosp. Med. 2017, 12, 375–379. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.-P.; Compton, P. Management of chronic pain with chronic opioid therapy in patients with substance use disorders. Addict. Sci. Clin. Pract. 2013, 8, 21. [Google Scholar] [CrossRef]
- St Marie, B.; Broglio, K. Managing Pain in the Setting of Opioid Use Disorder. Pain Manag. Nurs. 2020, 21, 26–34. [Google Scholar] [CrossRef]
- Coluzzi, F.; Bifulco, F.; Cuomo, A.; Dauri, M.; Leonardi, C.; Melotti, R.M.; Natoli, S.; Romualdi, P.; Savoia, G.; Corcione, A. The challenge of perioperative pain management in opioid-tolerant patients. Ther. Clin. Risk Manag. 2017, 13, 1163–1173. [Google Scholar] [CrossRef]
- Kaye, A.D.; Kandregula, S.; Kosty, J.; Sin, A.; Guthikonda, B.; Ghali, G.E.; Craig, M.K.; Pham, A.D.; Reed, D.S.; Gennuso, S.A.; et al. Chronic pain and substance abuse disorders: Preoperative assessment and optimization strategies. Best. Pract. Res. Clin. Anaesthesiol. 2020, 34, 255–267. [Google Scholar] [CrossRef]
- Macfarlane, G.; Kronisch, C.; Atzeni, F.; Häuser, W.; Choy, E.; Amris, K.; Branco, J.; Dincer, F.; Leino-Arjas, P.; Longley, K.; et al. EULAR recommendations for management of fibromyalgia. Ann. Rheum. Dis. 2017, 76, e54. [Google Scholar] [CrossRef]
- Lin, C.; Karim, H.T.; Pecina, M.; Aizenstein, H.J.; Lenze, E.J.; Blumberger, D.M.; Mulsant, B.H.; Kharasch, E.D.; Reynolds, C.F., III; Karp, J.F. Low-dose augmentation with buprenorphine increases emotional reactivity but not reward activity in treatment resistant mid- and late-life depression. Neuroimage Clin. 2019, 21, 101679. [Google Scholar] [CrossRef]
- Bershad, A.K.; Ruiz, N.A.; de Wit, H. Effects of Buprenorphine on Responses to Emotional Stimuli in Individuals with a Range of Mood Symptomatology. Int. J. Neuropsychopharmacol. 2018, 21, 120–127. [Google Scholar] [CrossRef]
- Garfinkel, S.; Liberzon, I. Neurobiology of PTSD: A Review of Neuroimaging Findings. Psychiatr. Ann. 2009, 39, 370–381. [Google Scholar] [CrossRef]
- Gorfinkel, L.R.; Malte, C.A.; Fink, D.S.; Mannes, Z.L.; Wall, M.M.; Olfson, M.; Livne, O.; Keyhani, S.; Keyes, K.M.; Martins, S.S.; et al. Trends in Opioid Use Disorder in the Veterans Health Administration, 2005–2022. JAMA Netw. Open 2024, 7, e2451821. [Google Scholar] [CrossRef] [PubMed]
- Kreutzwiser, D.; Tawfic, Q.A. Methadone for Pain Management: A Pharmacotherapeutic Review. CNS Drugs 2020, 34, 827–839. [Google Scholar] [CrossRef] [PubMed]
- Maremmani, A.G.I.; Della Rocca, F.; Bacciardi, S.; Carbone, M.G.; Maremmani, I. Antipsychotic Potential of Opioids: Rethinking Substance-Induced Psychosis and Treatment Stratification. J. Clin. Med. 2025, 14, 5596. [Google Scholar] [CrossRef]
- Santin, L.; Verlato, G.; Tfaily, A.; Manera, R.; Zinfollino, G.; Fusina, F.; Lugoboni, F. Methadone Maintenance and QT-Interval: Prevalence and Risk Factors—Is It Effective to Switch Therapy to Levomethadone? Biomedicines 2023, 11, 2109. [Google Scholar] [CrossRef]
- Havig, S.M.; Berg-Pedersen, R.M.; Krabseth, H.M.; Müller, L.D.; Haugaa, K.; Zare, H.K.; Gjesdal, K.; Krajci, P.; Opdal, M.S. Effect on QTc interval by switching from methadone to equipotent R-methadone dose in methadone maintenance treatment patients. Basic Clin. Pharmacol. Toxicol. 2024, 134, 519–530. [Google Scholar] [CrossRef]
- Stewart, J.L.; May, A.C.; Paulus, M.P. Bouncing back: Brain rehabilitation amid opioid and stimulant epidemics. Neuroimage Clin. 2019, 24, 102068. [Google Scholar] [CrossRef]
- Sandbrink, F.; Murphy, J.L.; Johansson, M.; Olson, J.L.; Edens, E.; Clinton-Lont, J.; Sall, J.; Spevak, C. The Use of Opioids in the Management of Chronic Pain: Synopsis of the 2022 Updated U.S. Department of Veterans Affairs and U.S. Department of Defense Clinical Practice Guideline. Ann. Intern. Med. 2023, 176, 388–397. [Google Scholar] [CrossRef]
- Hyland, S.J.; Brockhaus, K.K.; Vincent, W.R.; Spence, N.Z.; Lucki, M.M.; Howkins, M.J.; Cleary, R.K. Perioperative Pain Management and Opioid Stewardship: A Practical Guide. Healthcare 2021, 9, 333. [Google Scholar] [CrossRef]
- Petersson, M.; Uvnäs-Moberg, K. Interactions of Oxytocin and Dopamine-Effects on Behavior in Health and Disease. Biomedicines 2024, 12, 2440. [Google Scholar] [CrossRef]
- Gazerani, P. The neuroplastic brain: Current breakthroughs and emerging frontiers. Brain Res. 2025, 1858, 149643. [Google Scholar] [CrossRef] [PubMed]
- Tschacher, W.; Ribeiro, E.; Gonçalves, A.; Sampaio, A.; Moreira, P.; Coutinho, J. Electrodermal synchrony of patient and therapist as a predictor of alliance and outcome in psychotherapy. Front. Psychol. 2025, 16, 1545719. [Google Scholar] [CrossRef] [PubMed]
- Horgas, A.L.; Wu, J.; Booker, S.Q. Pain Management for Older Adults Across the Cognitive Trajectory. Curr. Geriatr. Rep. 2023, 12, 220–227. [Google Scholar] [CrossRef] [PubMed]
- Finan, P.H.; Goodin, B.R.; Smith, M.T. The association of sleep and pain: An update and a path forward. J. Pain 2013, 14, 1539–1552. [Google Scholar] [CrossRef]
- Melzack, R.; Coderre, T.J.; Katz, J.; Vaccarino, A.L. Central neuroplasticity and pathological pain. Ann. N. Y. Acad. Sci. 2001, 933, 157–174. [Google Scholar] [CrossRef]
- Jensen, K.B.; Kosek, E.; Petzke, F.; Carville, S.; Fransson, P.; Marcus, H.; Williams, S.C.; Choy, E.; Giesecke, T.; Mainguy, Y.; et al. Evidence of dysfunctional pain inhibition in Fibromyalgia reflected in rACC during provoked pain. Pain 2009, 144, 95–100. [Google Scholar] [CrossRef]
- Baliki, M.N.; Geha, P.Y.; Apkarian, A.V.; Chialvo, D.R. Beyond feeling: Chronic pain hurts the brain, disrupting the default-mode network dynamics. J. Neurosci. 2008, 28, 1398–1403. [Google Scholar] [CrossRef]
- Hashmi, J.A.; Baliki, M.N.; Huang, L.; Baria, A.T.; Torbey, S.; Hermann, K.M.; Schnitzer, T.J.; Apkarian, A.V. Shape shifting pain: Chronification of back pain shifts brain representation from nociceptive to emotional circuits. Brain 2013, 136, 2751–2768. [Google Scholar] [CrossRef]
- Surah, A.; Baranidharan, G.; Morley, S. Chronic pain and depression. Contin. Educ. Anaesth. Crit. Care Pain 2014, 14, 85–89. [Google Scholar] [CrossRef]
- Kroenke, K.; Wu, J.; Bair, M.J.; Krebs, E.E.; Damush, T.M.; Tu, W. Reciprocal relationship between pain and depression: A 12-month longitudinal analysis in primary care. J. Pain 2011, 12, 964–973. [Google Scholar] [CrossRef]
- Sasaoka, T.; Harada, T.; Sato, D.; Michida, N.; Yonezawa, H.; Takayama, M.; Nouzawa, T.; Yamawaki, S. Neural basis for anxiety and anxiety-related physiological responses during a driving situation: An fMRI study. Cereb. Cortex Commun. 2022, 3, tgac025. [Google Scholar] [CrossRef] [PubMed]
- Rischer, K.M.; Anton, F.; González-Roldán, A.M.; Montoya, P.; van der Meulen, M. Better Executive Functions Are Associated With More Efficient Cognitive Pain Modulation in Older Adults: An fMRI Study. Front. Aging Neurosci. 2022, 14, 828742. [Google Scholar] [CrossRef] [PubMed]
- Volkow, N.D.; Koob, G.F.; McLellan, A.T. Neurobiologic Advances from the Brain Disease Model of Addiction. N. Engl. J. Med. 2016, 374, 363–371. [Google Scholar] [CrossRef] [PubMed]
- Savage, S.R.; Kirsh, K.L.; Passik, S.D. Challenges in using opioids to treat pain in persons with substance use disorders. Addict. Sci. Clin. Pract. 2008, 4, 4–25. [Google Scholar] [CrossRef]
- Koob, G.F.; Le Moal, M. Drug addiction, dysregulation of reward, and allostasis. Neuropsychopharmacology 2001, 24, 97–129. [Google Scholar] [CrossRef]
- Chau, D.L.; Walker, V.; Pai, L.; Cho, L.M. Opiates and elderly: Use and side effects. Clin. Interv. Aging 2008, 3, 273–278. [Google Scholar] [CrossRef]
- Lavin, R.; Park, J. Depressive symptoms in community-dwelling older adults receiving opioid therapy for chronic pain. J. Opioid Manag. 2011, 7, 309–319. [Google Scholar] [CrossRef]
- Cahill, C.M.; Lueptow, L.; Kim, H.; Shusharla, R.; Bishop, A.; Evans, C.J. Kappa Opioid Signaling at the Crossroads of Chronic Pain and Opioid Addiction. Handb. Exp. Pharmacol. 2022, 271, 315–350. [Google Scholar]
- Frank, J.W.; Lovejoy, T.I.; Becker, W.C.; Morasco, B.J.; Koenig, C.J.; Hoffecker, L.; Dischinger, H.R.; Dobscha, S.K.; Krebs, E.E. Patient Outcomes in Dose Reduction or Discontinuation of Long-Term Opioid Therapy: A Systematic Review. Ann. Intern. Med. 2017, 167, 181–191. [Google Scholar] [CrossRef]
- Fenton, J.J.; Magnan, E.; Tseregounis, I.E.; Xing, G.; Agnoli, A.L.; Tancredi, D.J. Long-term Risk of Overdose or Mental Health Crisis After Opioid Dose Tapering. JAMA Netw. Open 2022, 5, e2216726. [Google Scholar] [CrossRef]
- Davis, M.P.; Digwood, G.; Mehta, Z.; McPherson, M.L. Tapering opioids: A comprehensive qualitative review. Ann. Palliat. Med. 2020, 9, 586–610. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Hou, S.; Tian, C.; Fu, Z.; Jie, J. Effects of Intranasal Oxytocin Across Various Depressive Disorders. Psychiatry Investig. 2025, 22, 964–978. [Google Scholar] [CrossRef] [PubMed]
- Koole, S.L.; Tschacher, W. Synchrony in Psychotherapy: A Review and an Integrative Framework for the Therapeutic Alliance. Front. Psychol. 2016, 7, 862. [Google Scholar] [CrossRef] [PubMed]
- Wiech, K. Deconstructing the sensation of pain: The influence of cognitive processes on pain perception. Science 2016, 354, 584–587. [Google Scholar] [CrossRef]
- Hassamal, S.; Jolles, P.; Pandurangi, A. Reversal of cerebral glucose hypometabolism on positron emission tomography with electroconvulsive therapy in an elderly patient with a psychotic episode. Psychogeriatrics 2016, 16, 376–381. [Google Scholar] [CrossRef]
- El Sheikh, W.G.; Sleem, B.; Kobeissy, F.; Bizri, M. Biomarkers of delirium and relation to dementia among the elderly in the intensive care unit: A narrative review. Biomark. Neuropsychiatry 2023, 8, 100064. [Google Scholar] [CrossRef]
- Paul, A.K.; Smith, C.M.; Rahmatullah, M.; Nissapatorn, V.; Wilairatana, P.; Spetea, M.; Gueven, N.; Dietis, N. Opioid Analgesia and Opioid-Induced Adverse Effects: A Review. Pharmaceuticals 2021, 14, 1091. [Google Scholar] [CrossRef]
- Samson, L.D.; Buisman, A.M.; Ferreira, J.A.; Picavet, H.S.J.; Verschuren, W.M.M.; Boots, A.M.; Engelfriet, P. Inflammatory marker trajectories associated with frailty and ageing in a 20-year longitudinal study. Clin. Transl. Immunol. 2022, 11, e1374. [Google Scholar] [CrossRef]
- Maremmani, I.; Dematteis, M.; Gorzelanczyk, E.J.; Mugelli, A.; Walcher, S.; Torrens, M. Long-Acting Buprenorphine Formulations as a New Strategy for the Treatment of Opioid Use Disorder. J. Clin. Med. 2023, 12, 5575. [Google Scholar] [CrossRef]
- Zhou, B.; Wang, Z.; Zhu, L.; Huang, G.; Li, B.; Chen, C.; Huang, J.; Ma, F.; Liu, T.C. Effects of different physical activities on brain-derived neurotrophic factor: A systematic review and bayesian network meta-analysis. Front. Aging Neurosci. 2022, 14, 981002. [Google Scholar] [CrossRef]
- Peng, L.N.; Lee, P.L.; Chou, K.H.; Lee, W.J.; Lin, C.P.; Chang, C.W.; Liang, C.K.; Chung, C.P.; Hsiao, F.Y.; Chen, L.K. Enhancing Neurocognitive Health via Activity, Nutrition and Cognitive Exercise (ENHANCE): A Randomized Controlled Trial. J. Cachexia Sarcopenia Muscle 2025, 16, e13830. [Google Scholar] [CrossRef]
- Borsook, D.; Youssef, A.M.; Simons, L.; Elman, I.; Eccleston, C. When pain gets stuck: The evolution of pain chronification and treatment resistance. Pain 2018, 159, 2421–2436. [Google Scholar] [CrossRef] [PubMed]
- Pedersini, P.; Gobbo, M.; Bishop, M.D.; Arendt-Nielsen, L.; Villafañe, J.H. Functional and Structural Neuroplastic Changes Related to Sensitization Proxies in Patients with Osteoarthritis: A Systematic Review. Pain Med. 2022, 23, 488–498. [Google Scholar] [CrossRef] [PubMed]
- Inagaki, T.; Muscatell, K.; Cole, S.; Eisenberger, N. Inflammation Selectively Enhances Amygdala Activity to Socially Threatening Images. NeuroImage 2011, 59, 3222–3226. [Google Scholar] [CrossRef] [PubMed]
- Holton, K.F. Modulation of Glutamate for Chronic Pain Management. In Glutamate and Neuropsychiatric Disorders: Current and Emerging Treatments; Pavlovic, Z.M., Ed.; Springer International Publishing: Cham, Switzerland, 2022; pp. 381–402. [Google Scholar]
- Albrecht, D.S.; Forsberg, A.; Sandström, A.; Bergan, C.; Kadetoff, D.; Protsenko, E.; Lampa, J.; Lee, Y.C.; Höglund, C.O.; Catana, C.; et al. Brain glial activation in fibromyalgia—A multi-site positron emission tomography investigation. Brain Behav. Immun. 2019, 75, 72–83. [Google Scholar] [CrossRef]
- Grace, P.M.; Hutchinson, M.R.; Maier, S.F.; Watkins, L.R. Pathological pain and the neuroimmune interface. Nat. Rev. Immunol. 2014, 14, 217–231. [Google Scholar] [CrossRef]
- Elman, I.; Borsook, D. Common Brain Mechanisms of Chronic Pain and Addiction. Neuron 2016, 89, 11–36. [Google Scholar] [CrossRef]
- Fang, S.; Qin, Y.; Yang, S.; Zhang, H.; Zheng, J.; Wen, S.; Li, W.; Liang, Z.; Zhang, X.; Li, B.; et al. Differences in the neural basis and transcriptomic patterns in acute and persistent pain-related anxiety-like behaviors. Front. Mol. Neurosci. 2023, 16, 1185243. [Google Scholar] [CrossRef]
- Diniz, B.S.; Teixeira, A.L. Brain-derived neurotrophic factor and Alzheimer’s disease: Physiopathology and beyond. Neuromolecular Med. 2011, 13, 217–222. [Google Scholar] [CrossRef]
- Wager, T.D.; Rilling, J.K.; Smith, E.E.; Sokolik, A.; Casey, K.L.; Davidson, R.J.; Kosslyn, S.M.; Rose, R.M.; Cohen, J.D. Placebo-induced changes in FMRI in the anticipation and experience of pain. Science 2004, 303, 1162–1167. [Google Scholar] [CrossRef]
- Seminowicz, D.A.; Shpaner, M.; Keaser, M.L.; Krauthamer, G.M.; Mantegna, J.; Dumas, J.A.; Newhouse, P.A.; Filippi, C.G.; Keefe, F.J.; Naylor, M.R. Cognitive-behavioral therapy increases prefrontal cortex gray matter in patients with chronic pain. J. Pain 2013, 14, 1573–1584. [Google Scholar] [CrossRef] [PubMed]
- Jensen, K.B.; Berna, C.; Loggia, M.L.; Wasan, A.D.; Edwards, R.R.; Gollub, R.L. The use of functional neuroimaging to evaluate psychological and other non-pharmacological treatments for clinical pain. Neurosci. Lett. 2012, 520, 156–164. [Google Scholar] [CrossRef] [PubMed]
- Zeidan, F.; Emerson, N.M.; Farris, S.R.; Ray, J.N.; Jung, Y.; McHaffie, J.G.; Coghill, R.C. Mindfulness Meditation-Based Pain Relief Employs Different Neural Mechanisms Than Placebo and Sham Mindfulness Meditation-Induced Analgesia. J. Neurosci. 2015, 35, 15307–15325. [Google Scholar] [CrossRef] [PubMed]
- Garland, E.L.; Hanley, A.W.; Nakamura, Y.; Barrett, J.W.; Baker, A.K.; Reese, S.E.; Riquino, M.R.; Froeliger, B.; Donaldson, G.W. Mindfulness-Oriented Recovery Enhancement vs Supportive Group Therapy for Co-occurring Opioid Misuse and Chronic Pain in Primary Care: A Randomized Clinical Trial. JAMA Intern. Med. 2022, 182, 407–417. [Google Scholar] [CrossRef]
- Brewer, J.A.; Worhunsky, P.D.; Gray, J.R.; Tang, Y.Y.; Weber, J.; Kober, H. Meditation experience is associated with differences in default mode network activity and connectivity. Proc. Natl. Acad. Sci. USA 2011, 108, 20254–20259. [Google Scholar] [CrossRef]
- Horvath, A.O.; Del Re, A.C.; Flückiger, C.; Symonds, D. Alliance in individual psychotherapy. Psychotherapy 2011, 48, 9–16. [Google Scholar] [CrossRef]
- Hruschak, V.; Cochran, G.; Wasan, A.D. Psychosocial interventions for chronic pain and comorbid prescription opioid use disorders: A narrative review of the literature. J. Opioid Manag. 2018, 14, 345–358. [Google Scholar] [CrossRef]
- Butler, S.H. Neuroinflammation in chronic pain: Myth or reality? Scand. J. Pain 2025, 25, 20240065. [Google Scholar] [CrossRef]
- Coussens, N.P.; Sittampalam, G.S.; Jonson, S.G.; Hall, M.D.; Gorby, H.E.; Tamiz, A.P.; McManus, O.B.; Felder, C.C.; Rasmussen, K. The Opioid Crisis and the Future of Addiction and Pain Therapeutics. J. Pharmacol. Exp. Ther. 2019, 371, 396–408. [Google Scholar] [CrossRef]
- Holmes, S.A.; Mar’i, J.; Green, S.; Borsook, D. Towards a deeper understanding of pain: How machine learning and deep learning algorithms are needed to provide the next generation of pain medicine for use in the clinic. Neurobiol. Pain 2022, 12, 100108. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.