Central and Peripheral Fatigue in Recreational Trail Runners: A Pilot Study
Abstract
1. Introduction
2. Materials and Methods
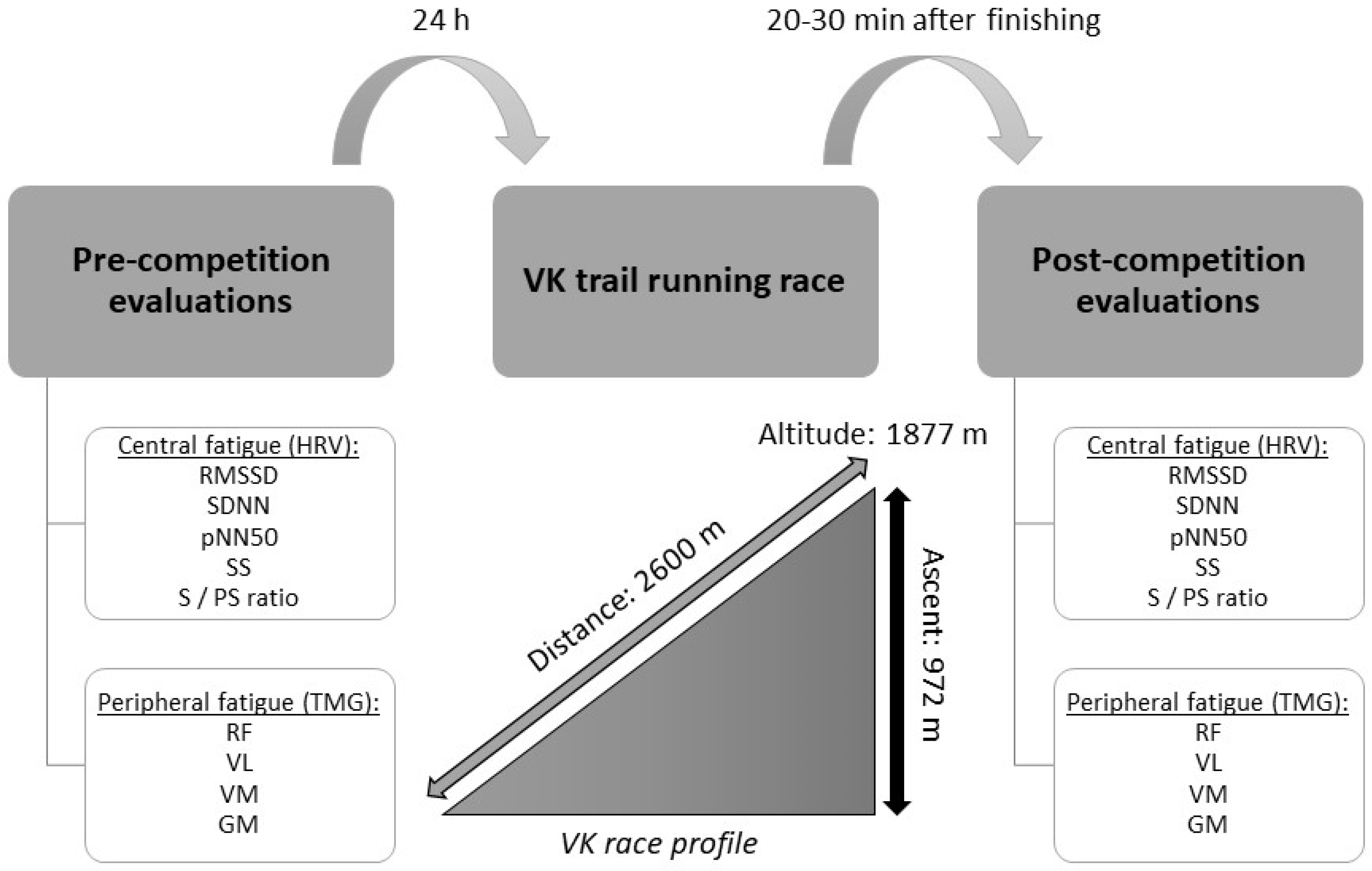
2.1. Study Design
2.2. Participants
2.3. Measurements
2.3.1. Central Fatigue Assessment: HRV
2.3.2. Peripheral Fatigue Assessment: Contractile Muscle Properties
2.4. Procedures
2.5. Statistical Analysis
3. Results
3.1. Central Fatigue Assessment: HRV
3.2. Peripheral Fatigue Assessment: TMG
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bassett, D.R.J.; Howley, E.T. Limiting factors for maximum oxygen uptake and determinants of endurance performance. Med. Sci. Sports Exerc. 2000, 32, 70–84. [Google Scholar] [CrossRef] [PubMed]
- Casado, A.; González-Mohíno, F.; González-Ravé, J.M.; Foster, C. Training Periodization, Methods, Intensity Distribution, and Volume in Highly Trained and Elite Distance Runners: A Systematic Review. Int. J. Sports Physiol. Perform. 2022, 17, 820–833. [Google Scholar] [CrossRef] [PubMed]
- Lazzer, S.; Salvadego, D.; Taboga, P.; Rejc, E.; Giovanelli, N.; di Prampero, P.E. Effects of the Etna Uphill Ultramarathon on Energy Cost and Mechanics of Running. Int. J. Sports Physiol. Perform. 2015, 10, 238–247. [Google Scholar] [CrossRef]
- Ehrström, S.; Tartaruga, M.P.; Easthope, C.S.; Brisswalter, J.; Morin, J.-B.; Vercruyssen, F. Short Trail Running Race: Beyond the Classic Model for Endurance Running Performance. Med. Sci. Sports Exerc. 2018, 50, 580–588. [Google Scholar] [CrossRef]
- Björklund, G.; Swarén, M.; Born, D.-P.; Stöggl, T. Biomechanical Adaptations and Performance Indicators in Short Trail Running. Front. Physiol. 2019, 10, 506. [Google Scholar] [CrossRef]
- Bascuas, P.J.; Bataller-Cervero, A.V.; Gutierrez, H.; Berzosa, C. Modifications of viscoelastic properties and physiological parameters after performing uphill and downhill running trials. J. Sports Med. Phys. Fit. 2020, 61, 625–635. [Google Scholar] [CrossRef]
- Lemire, M.; Hureau, T.J.; Favret, F.; Geny, B.; Kouassi, B.Y.L.; Boukhari, M.; Lonsdorfer, E.; Remetter, R.; Dufour, S.P. Physiological factors determining downhill vs uphill running endurance performance. J. Sci. Med. Sport 2021, 24, 85–91. [Google Scholar] [CrossRef]
- Nicot, F.; Sabater-Pastor, F.; Samozino, P.; Millet, G.Y.; Rupp, T. Effect of ground technicity on cardio-respiratory and biomechanical parameters in uphill trail running. Eur. J. Sport Sci. 2021, 22, 1836–1846. [Google Scholar] [CrossRef]
- Berzosa, C.; Gutierrez, H.; Bascuas, P.J.; Arbones, I.; Bataller-Cervero, A.V. Muscle Tone and Body Weight Predict Uphill Race Time in Amateur Trail Runners. Int. J. Environ. Res. Public Health 2021, 18, 2040. [Google Scholar] [CrossRef]
- Millet, G.Y.; Tomazin, K.; Verges, S.; Vincent, C.; Bonnefoy, R.; Boisson, R.-C.; Gergelé, L.; Féasson, L.; Martin, V. Neuromuscular Consequences of an Extreme Mountain Ultra-Marathon. PLoS ONE 2011, 6, e17059. [Google Scholar] [CrossRef]
- Giovanelli, N.; Taboga, P.; Rejc, E.; Simunic, B.; Antonutto, G.; Lazzer, S. Effects of an Uphill Marathon on Running Mechanics and Lower-Limb Muscle Fatigue. Int. J. Sports Physiol. Perform. 2016, 11, 522–529. [Google Scholar] [CrossRef] [PubMed]
- Millet, G.Y.; Martin, V.; Temesi, J. The role of the nervous system in neuromuscular fatigue induced by ultra-endurance exercise. Appl. Physiol. Nutr. Metab. 2018, 43, 1151–1157. [Google Scholar] [CrossRef] [PubMed]
- Hewing, B.; Schattke, S.; Spethmann, S.; Sanad, W.; Schroeckh, S.; Schimke, I.; Halleck, F.; Peters, H.; Brechtel, L.; Lock, J.; et al. Cardiac and renal function in a large cohort of amateur marathon runners. Cardiovasc. Ultrasound 2015, 13, 13. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Marklund, P.; Mattsson, C.M.; Wåhlin-Larsson, B.; Ponsot, E.; Lindvall, B.; Lindvall, L.; Ekblom, B.; Kadi, F. Extensive inflammatory cell infiltration in human skeletal muscle in response to an ultraendurance exercise bout in experienced athletes. J. Appl. Physiol. 2012, 114, 66–72. [Google Scholar] [CrossRef]
- Kiviniemi, A.M.; Hautala, A.J.; Kinnunen, H.; Tulppo, M.P. Endurance training guided individually by daily heart rate variability measurements. Eur. J. Appl. Physiol. 2007, 101, 743–751. [Google Scholar] [CrossRef]
- Pichot, V.; Roche, F.; Celle, S.; Barthélémy, J.-C.; Chouchou, F. HRVanalysis: A Free Software for Analyzing Cardiac Autonomic Activity. Front. Physiol. 2016, 7, 557. [Google Scholar] [CrossRef]
- Varela-Sanz, A.; Tuimil, J.L.; Abreu, L.; Boullosa, D.A. Does Concurrent Training Intensity Distribution Matter? J. Strength Cond. Res. 2017, 31, 181–195. [Google Scholar] [CrossRef]
- Vesterinen, V.; Nummela, A.R.I.; Heikura, I.D.A.; Laine, T.; Hynynen, E.S.A.; Botella, J.; Häkkinen, K. Individual Endurance Training Prescription with Heart Rate Variability. Med. Sci. Sports Exerc. 2016, 48, 1347–1354. [Google Scholar] [CrossRef]
- Task Force of the European Society of Cardiology the North American Society of Pacing and Electrophysiology. Heart Rate Variability. Standards of measurement, physiological interpretation and clinical use. Circulation 1996, 93, 1043–1065. [Google Scholar] [CrossRef]
- Carrasco-Poyatos, M.; González-Quílez, A.; Martínez-González-Moro, I.; Granero-Gallegos, A. HRV-Guided Training for Professional Endurance Athletes: A Protocol for a Cluster-Randomized Controlled Trial. Int. J. Environ. Res. Public Health 2020, 17, 5465. [Google Scholar] [CrossRef]
- Altini, M.; Plews, D. What Is behind Changes in Resting Heart Rate and Heart Rate Variability? A Large-Scale Analysis of Longitudinal Measurements Acquired in Free-Living. Sensors 2021, 21, 7932. [Google Scholar] [CrossRef] [PubMed]
- Pichot, V.; Busso, T.; Roche, F.; Garet, M.; Costes, F.; Duverney, D.; Lacour, J.-R.; Barthélémy, J.-C. Autonomic adaptations to intensive and overload training periods: A laboratory study. Med. Sci. Sports Exerc. 2002, 34, 1660–1666. [Google Scholar] [CrossRef] [PubMed]
- Gratze, G.; Rudnicki, R.; Urban, W.; Mayer, H.; Schlögl, A.; Skrabal, F. Hemodynamic and autonomic changes induced by Ironman: Prediction of competition time by blood pressure variability. J. Appl. Physiol. 2005, 99, 1728–1735. [Google Scholar] [CrossRef] [PubMed]
- Hautala, A.; Tulppo, M.P.; Mäkikallio, T.H.; Laukkanen, R.; Nissilä, S.; Huikuri, H.V. Changes in cardiac autonomic regulation after prolonged maximal exercise. Clin. Physiol. 2001, 21, 238–245. [Google Scholar] [CrossRef] [PubMed]
- Nicolas, M.; Banizette, M.; Millet, G.Y. Stress and recovery states after a 24 h ultra-marathon race: A one-month follow-up study. Psychol. Sport Exerc. 2011, 12, 368–374. [Google Scholar] [CrossRef]
- Easthope, C.S.; Hausswirth, C.; Louis, J.; Lepers, R.; Vercruyssen, F.; Brisswalter, J. Effects of a trail running competition on muscular performance and efficiency in well-trained young and master athletes. Eur. J. Appl. Physiol. 2010, 110, 1107–1116. [Google Scholar] [CrossRef]
- Carrasco, L.; Sañudo, B.; de Hoyo, M.; Pradas, F.; Da Silva, M.E. Effectiveness of low-frequency vibration recovery method on blood lactate removal, muscle contractile properties and on time to exhaustion during cycling at VO2max power output. Eur. J. Appl. Physiol. 2011, 111, 2271–2279. [Google Scholar] [CrossRef]
- García-García, O.; Cuba-Dorado, A.; Riveiro-Bozada, A.; Carballo-López, J.; Álvarez-Yates, T.; López-Chicharro, J. A Maximal Incremental Test in Cyclists Causes Greater Peripheral Fatigue in Biceps Femoris. Res. Q. Exerc. Sport 2020, 91, 460–468. [Google Scholar] [CrossRef]
- García-Manso, J.M.; Rodríguez-Ruiz, D.; Rodríguez-Matoso, D.; de Saa, Y.; Sarmiento, S.; Quiroga, M. Assessment of muscle fatigue after an ultra-endurance triathlon using tensiomyography (TMG). J. Sports Sci. 2011, 29, 619–625. [Google Scholar] [CrossRef]
- García-Manso, J.M.; Rodríguez-Matoso, D.; Sarmiento, S.; de Saa, Y.; Vaamonde, D.; Rodríguez-Ruiz, D.; Da Silva-Grigoletto, M.E. Effect of high-load and high-volume resistance exercise on the tensiomyographic twitch response of biceps brachii. J. Electromyogr. Kinesiol. 2012, 22, 612–619. [Google Scholar] [CrossRef]
- Macgregor, L.J.; Ditroilo, M.; Smith, I.J.; Fairweather, M.M.; Hunter, A.M. Reduced Radial Displacement of the Gastrocnemius Medialis Muscle After Electrically Elicited Fatigue. J. Sport Rehabil. 2016, 25, 241–247. [Google Scholar] [CrossRef] [PubMed]
- Raeder, C.; Wiewelhove, T.; Simola, R.Á.D.P.; Kellmann, M.; Meyer, T.; Pfeiffer, M.; Ferrauti, A. Assessment of Fatigue and Recovery in Male and Female Athletes After 6 Days of Intensified Strength Training. J. Strength Cond. Res. 2016, 30, 3412–3427. [Google Scholar] [CrossRef] [PubMed]
- Wiewelhove, T.; Raeder, C.; Meyer, T.; Kellmann, M.; Pfeiffer, M.; Ferrauti, A. Markers for Routine Assessment of Fatigue and Recovery in Male and Female Team Sport Athletes during High-Intensity Interval Training. PLoS ONE 2015, 10, e0139801. [Google Scholar] [CrossRef] [PubMed]
- González-Fimbres, R.A.; Hernández-Cruz, G.; Flatt, A.A. Ultrashort Versus Criterion Heart Rate Variability Among International-Level Girls’ Field Hockey Players. Int. J. Sports Physiol. Perform. 2021, 16, 985–992. [Google Scholar] [CrossRef] [PubMed]
- Bloomfield, D.M.; Magnano, A.; Bigger, J.T.; Rivadeneira, H.; Parides, M.; Steinman, R.C. Comparison of spontaneous vs. metronome-guided breathing on assessment of vagal modulation using RR variability. Am. J. Physiol. Circ. Physiol. 2001, 280, H1145–H1150. [Google Scholar] [CrossRef] [PubMed]
- Tarvainen, M.P.; Niskanen, J.-P.; Lipponen, J.A.; Ranta-aho, P.O.; Karjalainen, P.A. Kubios HRV—Heart rate variability analysis software. Comput. Methods Programs Biomed. 2014, 113, 210–220. [Google Scholar] [CrossRef] [PubMed]
- Naranjo Orellana, J.; de la Cruz Torres, B.; Sarabia Cachadiña, E.; de Hoyo, M.; Domínguez Cobo, S. Two New Indexes for the Assessment of Autonomic Balance in Elite Soccer Players. Int. J. Sports Physiol. Perform. 2015, 10, 452–457. [Google Scholar] [CrossRef]
- Gutiérrez-Vargas, R.; Martín-Rodríguez, S.; Sánchez-Ureña, B.; Rodríguez-Montero, A.; Salas-Cabrera, J.; Gutiérrez-Vargas, J.C.; Simunic, B.; Rojas-Valverde, D. Biochemical and Muscle Mechanical Postmarathon Changes in Hot and Humid Conditions. J. Strength Cond. Res. 2020, 34, 847–856. [Google Scholar] [CrossRef]
- Pereira, L.A.; Ramirez-Campillo, R.; Martín-Rodríguez, S.; Kobal, R.; Abad, C.C.C.; Arruda, A.F.S.; Guerriero, A.; Loturco, I. Is tensiomyography-derived velocity of contraction a sensitive marker to detect acute performance changes in elite team-sport athletes? Int. J. Sports Physiol. Perform. 2020, 15, 31–37. [Google Scholar] [CrossRef]
- Rey, E.; Padrón-Cabo, A.; Barcala-Furelos, R.; Mecías-Calvo, M. Effect of High and Low Flexibility Levels on Physical Fitness and Neuromuscular Properties in Professional Soccer Players. Int. J. Sports Med. 2016, 37, 878–883. [Google Scholar] [CrossRef]
- Rey, E.; Lago-Peñas, C.; Lago-Ballesteros, J. Tensiomyography of selected lower-limb muscles in professional soccer players. J. Electromyogr. Kinesiol. 2012, 22, 866–872. [Google Scholar] [CrossRef]
- Loturco, I.; Pereira, L.A.; Kobal, R.; Kitamura, K.; Ramírez-Campillo, R.; Zanetti, V.; Abad, C.C.C.; Nakamura, F.Y. Muscle contraction velocity: A suitable approach to analyze the functional adaptations in elite soccer players. J. Sports Sci. Med. 2016, 15, 483. [Google Scholar] [PubMed]
- De Paula Simola, R.Á.; Harms, N.; Raeder, C.; Kellmann, M.; Meyer, T.; Pfeiffer, M.; Ferrauti, A. Assessment of Neuromuscular Function After Different Strength Training Protocols Using Tensiomyography. J. Strength Cond. Res. 2015, 29, 1339–1348. [Google Scholar] [CrossRef]
- De Paula Simola, R.Á.; Raeder, C.; Wiewelhove, T.; Kellmann, M.; Meyer, T.; Pfeiffer, M.; Ferrauti, A. Muscle mechanical properties of strength and endurance athletes and changes after one week of intensive training. J. Electromyogr. Kinesiol. 2016, 30, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Ellis, P.D. The Essential Guide to Effect Sizes; Cambridge University Press: Cambridge, UK, 2010; ISBN 9780511761676. [Google Scholar]
- Kerby, D.S. The Simple Difference Formula: An Approach to Teaching Nonparametric Correlation. Compr. Psychol. 2014, 3, 11. [Google Scholar] [CrossRef]
- Medina Corrales, M.; de la Cruz Torres, B.; Garrido Esquivel, A.; Garrido Salazar, M.A.; Naranjo Orellana, J. Normal values of heart rate variability at rest in a young, healthy and active Mexican population. Health 2012, 4, 377–385. [Google Scholar] [CrossRef]
- Sammito, S.; Böckelmann, I. Reference values for time- and frequency-domain heart rate variability measures. Heart Rhythm 2016, 13, 1309–1316. [Google Scholar] [CrossRef] [PubMed]
- Melanson, E.L. Resting heart rate variability in men varying in habitual physical activity. Med. Sci. Sports Exerc. 2000, 32, 1894–1901. [Google Scholar] [CrossRef]
- Sandercock, G.R.H.; Hardy-Shepherd, D.; Nunan, D.; Brodie, D. The relationships between self-assessed habitual physical activity and non-invasive measures of cardiac autonomic modulation in young healthy volunteers. J. Sports Sci. 2008, 26, 1171–1177. [Google Scholar] [CrossRef]
- Hoshi, R.A.; Pastre, C.M.; Vanderlei, L.C.M.; Godoy, M.F. Poincaré plot indexes of heart rate variability: Relationships with other nonlinear variables. Auton. Neurosci. 2013, 177, 271–274. [Google Scholar] [CrossRef]
- Plews, D.J.; Laursen, P.B.; Kilding, A.E.; Buchheit, M. Heart rate variability in elite triathletes, is variation in variability the key to effective training? A case comparison. Eur. J. Appl. Physiol. 2012, 112, 3729–3741. [Google Scholar] [CrossRef] [PubMed]
- Nicoll, J.X.; Hatfield, D.L.; Melanson, K.J.; Nasin, C.S. Thyroid hormones and commonly cited symptoms of overtraining in collegiate female endurance runners. Eur. J. Appl. Physiol. 2018, 118, 65–73. [Google Scholar] [CrossRef] [PubMed]
- Boullosa, D.; Esteve-Lanao, J.; Casado, A.; Peyré-Tartaruga, L.A.; da Rosa, R.; Del Coso, J. Factors Affecting Training and Physical Performance in Recreational Endurance Runners. Sports 2020, 8, 35. [Google Scholar] [CrossRef]
- Rodríguez-Matoso, D.; Mantecón, A.; Barbosa-Almeida, E.; Valverde, T.; García-Manso, J.M.; Rodríguez-Ruiz, D. Mechanical response of knee muscles in high level bodyboarders during performance. Rev. Bras. Med. Esporte 2015, 21, 144–147. [Google Scholar] [CrossRef]
- Westerblad, H.; Lännergren, J.; Allen, D.G. Slowed Relaxation in Fatigued Skeletal Muscle Fibers of Xenopus and Mouse: Contribution of [Ca2+]i and Cross-bridges. J. Gen. Physiol. 1997, 109, 385–399. [Google Scholar] [CrossRef] [PubMed]
- García-García, O.; Cuba-Dorado, A.; Álvarez-Yates, T.; Carballo-López, J.; Iglesias-Caamaño, M. Clinical utility of tensiomyography for muscle function analysis in athletes. Open Access J. Sports Med. 2019, 10, 49–69. [Google Scholar] [CrossRef]
- Carpes, F.P.; Mota, C.B.; Faria, I.E. On the bilateral asymmetry during running and cycling—A review considering leg preference. Phys. Ther. Sport 2010, 11, 136–142. [Google Scholar] [CrossRef]
- Zghal, F.; Cottin, F.; Kenoun, I.; Rebaï, H.; Moalla, W.; Dogui, M.; Tabka, Z.; Martin, V. Improved tolerance of peripheral fatigue by the central nervous system after endurance training. Eur. J. Appl. Physiol. 2015, 115, 1401–1415. [Google Scholar] [CrossRef]
| Pre-VK | Post-VK | p-Value | 95% CI | Cohen’s d ES | 95% CI | |||
|---|---|---|---|---|---|---|---|---|
| (Mean ± SD) | (Mean ± SD) | Lower | Upper | Lower | Upper | |||
| HR (bpm) | 56.10 ± 6.96 | 71.90 ± 11.78 | 0.01 | −26.99 | −4.51 | −1.17 | −2.07 | −0.23 |
| Time domain | ||||||||
| SDNN (ms) | 58.10 ± 27.06 | 33.40 ± 18.99 | 0.02 | 4.79 | 44.52 | 1.04 | 0.14 | 1.89 |
| pNN50 (%) | 23.71 ± 12.56 | 8.26 ± 8.94 | 0.23 | 3.04 | 24.49 | 0.89 | ||
| RMSSD (ms) | 48.45 ± 19.29 | 27.21 ± 12.16 | 0.01 | 8.13 | 34.35 | 1.35 | 0.35 | 2.31 |
| Non-linear measurements | ||||||||
| SS (a.u.) | 14.97 ± 7.76 | 28.42 ± 13.66 | 0.01 | −22.31 | −4.59 | −1.27 | −2.20 | −0.30 |
| S/PS ratio (a.u.) | 0.58 ± 0.45 | 2.36 ± 2.32 | 0.02 | −3.89 | −0.21 | −0.94 | ||
| Muscle Group | TMG | Pre-VK (Mean ± SD) | Post-VK (Mean ± SD) | Difference | p-Value | 95% CI | Cohen’s d ES | 95% CI | ||
|---|---|---|---|---|---|---|---|---|---|---|
| (%) | Lower | Upper | Lower | Upper | ||||||
| Rectus Femoris (RF) | Td | 47.45 ± 2.93 | 45.34 ± 2.66 | −4.7 | 0.02 * | 0.41 | 3.47 | 0.85 | 0.14 | 1.54 |
| Tc | 57.19 ± 7.09 | 57.41 ± 8.40 | 0.4 | 0.88 | −5.0 | 4.35 | −0.05 | −0.64 | 0.55 | |
| Ts | 307.43 ± 599.85 | 143.03 ± 131.92 | −114.9 | 0.17 | −97.48 | 481.47 | 0.44 | −0.19 | 1.06 | |
| Tr | 121.65 ± 230.21 | 53.69 ± 55.11 | −126.6 | 0.15 | −33.34 | 188.73 | 0.47 | −0.17 | 1.09 | |
| Dm | 14.30 ± 3.43 | 15.60 ± 3.76 | 8.3 | 0.18 | −3.10 | 0.68 | −0.43 | −1.04 | 0.20 | |
| Vc | 0.27 ± 0.06 | 0.30 ± 0.06 | 9.2 | 0.12 | −0.06 | 0.008 | −0.50 | −1.13 | 0.14 | |
| Vastus Lateralis (VL) | Td | 43.93 ± 2.31 | 42.81 ± 2.49 | −2.6 | 0.09 | −0.19 | 2.20 | 0.56 | −0.09 | 1.19 |
| Tc | 45.85 ± 4.99 | 45.20 ± 5.88 | −1.4 | 0.52 | −1.22 | 2.24 | 0.19 | −0.40 | 0.79 | |
| Ts | 90.84 ± 47.23 | 89.07 ± 25.18 | −2.0 | 0.81 | −28.24 | 35.47 | 0.07 | −0.52 | 0.67 | |
| Tr | 38.82 ± 39.04 | 35.05 ± 17.45 | −10.8 | 0.66 | −21.23 | 32.03 | 0.13 | −0.46 | 0.73 | |
| Dm | 10.37 ± 1.82 | 11.16 ± 1.91 | 7.0 | 0.12 | −1.68 | 0.23 | −0.50 | −1.13 | 0.13 | |
| Vc | 0.23 ± 0.04 | 0.25 ± 0.04 | 9.2 | 0.06 | −0.04 | 0.001 | −0.63 | −1.28 | 0.03 | |
| Vastus Medialis (VM) | Td | 42.83 ± 1.52 | 42.51 ± 1.01 | −0.7 | 0.49 | −0.72 | 1.38 | 0.21 | −0.39 | 0.81 |
| Tc | 47.64 ± 4.52 | 48.15 ± 5.28 | 1.0 | 0.41 | −1.89 | 0.83 | −0.26 | −0.86 | 0.34 | |
| Ts | 411.90 ± 165.18 | 371.33 ± 59.77 | −10.9 | 0.23 | −34.64 | 127.56 | 0.38 | −0.24 | 0.99 | |
| Tr | 230.80 ± 65.42 | 151.23 ± 68.48 | −52.6 | 0.01 * | 18.01 | 126.10 | 0.89 | 0.17 | 1.59 | |
| Dm | 15.58 ± 3.46 | 16.09 ± 3.38 | 3.2 | 0.24 | −1.28 | 0.36 | −0.37 | −0.98 | 0.25 | |
| Vc | 0.35 ± 0.09 | 0.36 ± 0.08 | 2.9 | 0.33 | −0.03 | 0.01 | −0.31 | −0.91 | 0.30 | |
| Gastrocnemius Medialis (GM) | Td | 39.94 ± 1.91 | 37.72 ± 3.82 | −5.9 | 0.10 | −0.43 | 4.12 | 0.54 | −0.10 | 1.17 |
| Tc | 43.22 ± 6.08 | 44.10 ± 13.28 | 2.0 | 0.66 | −8.64 | 5.73 | −0.13 | −0.73 | 0.46 | |
| Ts | 480.16 ± 242.51 | 622.28 ± 353.28 | 22.8 | 0.01 * | −239.42 | −39.12 | −0.93 | −1.63 | −0.20 | |
| Tr | 175.05 ± 129.45 | 143.02 ± 118.09 | −22.4 | 0.61 | −96.87 | 157.16 | 0.15 | −0.44 | 0.75 | |
| Dm | 5.07 ± 2.17 | 4.76 ± 2.14 | −6.6 | 0.65 | −1.07 | 1.65 | 0.14 | −0.46 | 0.73 | |
| Vc | 0.12 ± 0.05 | 0.12 ± 0.05 | −4.8 | 0.71 | −0.02 | 0.04 | 0.11 | −0.48 | 0.71 | |
| Side & Muscle Group | TMG | Pre-VK (Mean ± SD) | Post-VK (Mean ± SD) | p-Value | 95% CI | Cohen’s d ES | 95% CI | ||
|---|---|---|---|---|---|---|---|---|---|
| Lower | Upper | Lower | Upper | ||||||
| Right RF | Td | 23.74 ± 1.70 | 22.38 ± 1.95 | 0.02 | 0.02 | 2.46 | 0.94 | 0.13 | 1.71 |
| Vc | 0.14 ± 0.04 | 0.16 ± 0.036 | 0.03 | −0.04 | −0.002 | −0.86 | −1.62 | −0.07 | |
| Right VL | Td | 22.08 ± 1.48 | 21.13 ± 1.12 | 0.02 | 0.20 | 1.71 | 0.97 | 0.15 | 1.75 |
| Tc | 23.1 ± 1.83 | 21.85 ± 2.19 | 0.02 | 0.24 | 2.27 | 0.95 | 0.13 | 1.73 | |
| Left VM | Tr | 117.4 ± 48.07 | 67.18 ± 39.76 | 0.02 | 8.87 | 91.55 | 0.93 | 0.12 | 1.71 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Muñoz-Pérez, I.; Varela-Sanz, A.; Lago-Fuentes, C.; Navarro-Patón, R.; Mecías-Calvo, M. Central and Peripheral Fatigue in Recreational Trail Runners: A Pilot Study. Int. J. Environ. Res. Public Health 2023, 20, 402. https://doi.org/10.3390/ijerph20010402
Muñoz-Pérez I, Varela-Sanz A, Lago-Fuentes C, Navarro-Patón R, Mecías-Calvo M. Central and Peripheral Fatigue in Recreational Trail Runners: A Pilot Study. International Journal of Environmental Research and Public Health. 2023; 20(1):402. https://doi.org/10.3390/ijerph20010402
Chicago/Turabian StyleMuñoz-Pérez, Iker, Adrián Varela-Sanz, Carlos Lago-Fuentes, Rubén Navarro-Patón, and Marcos Mecías-Calvo. 2023. "Central and Peripheral Fatigue in Recreational Trail Runners: A Pilot Study" International Journal of Environmental Research and Public Health 20, no. 1: 402. https://doi.org/10.3390/ijerph20010402
APA StyleMuñoz-Pérez, I., Varela-Sanz, A., Lago-Fuentes, C., Navarro-Patón, R., & Mecías-Calvo, M. (2023). Central and Peripheral Fatigue in Recreational Trail Runners: A Pilot Study. International Journal of Environmental Research and Public Health, 20(1), 402. https://doi.org/10.3390/ijerph20010402








