A Systematic Review on Mobile Health Applications’ Education Program for Patients Taking Oral Anticoagulants
Abstract
1. Introduction
2. Methods
2.1. Literature Search Strategies
2.2. Quality Assessment
2.3. Data Extraction
3. Results
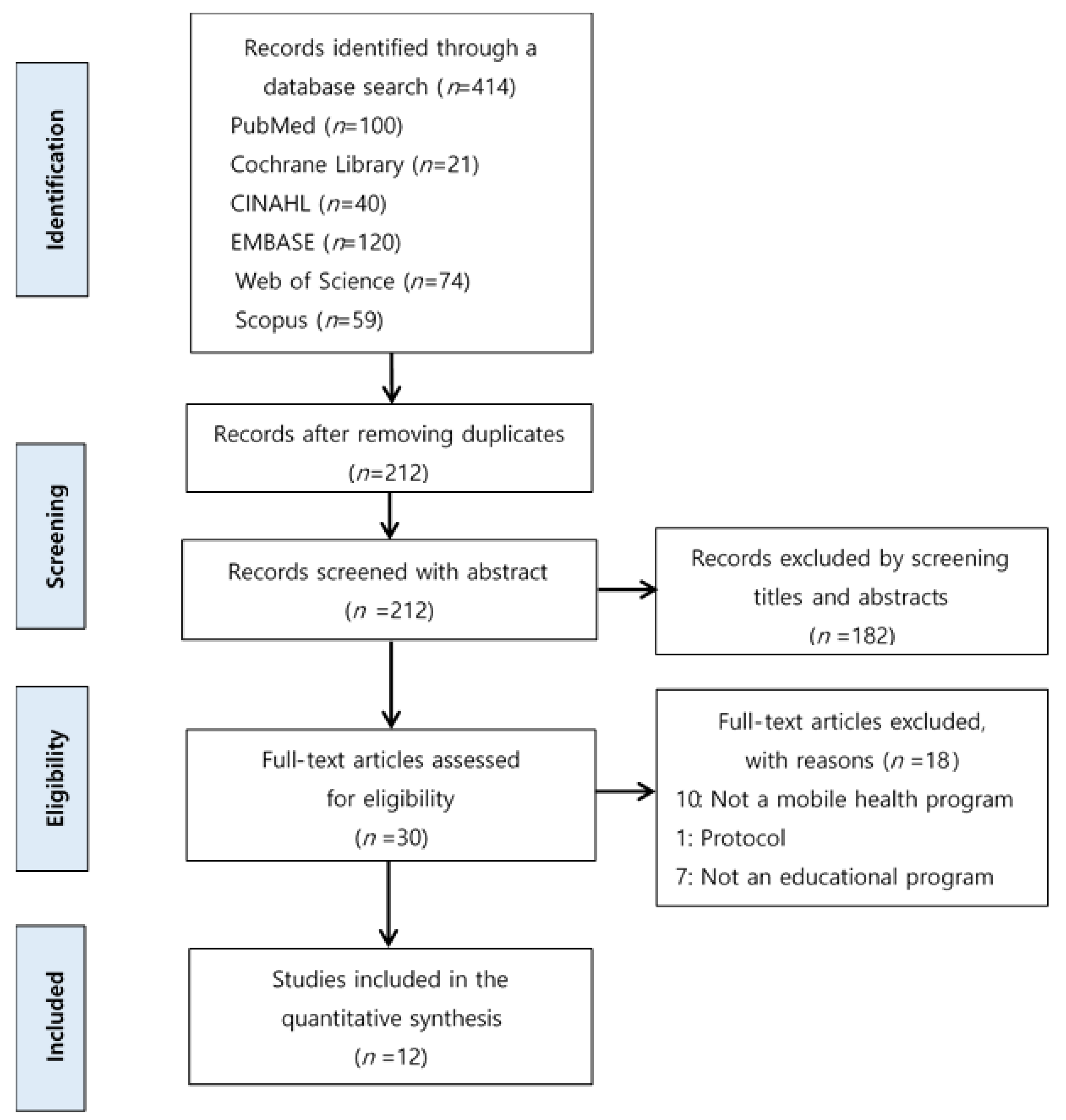
3.1. Search Outcome
3.2. Characteristics of the Included Studies
3.3. Overview of the Intervention
3.3.1. Clinical Outcomes
3.3.2. Self-Management Outcomes
3.4. Assessment of Study Quality
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Da Silva Praxedes Rn, M.F.; de Melo Mambrini Bstat, J.V.; Rph, A.M.M.R.; de Abreu Dds, M.H.N.G.; Rph, M.A.P.M. Assessment of patient knowledge on warfarin: An item response theory approach. J. Clin. Pharm. Ther. 2020, 45, 698–706. [Google Scholar] [CrossRef]
- Sjögren, V.; Grzymala-Lubanski, B.; Renlund, H.; Friberg, L.; Lip, G.Y.; Svensson, P.J.; Själander, A. Safety and efficacy of well managed warfarin. Thromb. Haemost. 2015, 113, 1370–1377. [Google Scholar] [CrossRef]
- Feng, X.; Sambamoorthi, U.; Innes, K.; Castelli, G.; Lemasters, T.; Xiong, L.; Williams, M.U.; Tan, X. Predictors of Major Bleeding Among Working-Age Adults with Atrial Fibrillation: Evaluating the Effects of Potential Drug-drug Interactions and Switching from Warfarin to Non-vitamin K Oral Anticoagulants. Cardiovasc. Drugs Ther. 2018, 32, 591–600. [Google Scholar] [CrossRef]
- Prochaska, J.H.; Göbel, S.; Keller, K.; Coldewey, M.; Ullmann, A.; Lamparter, H.; Jünger, C.; Al-Bayati, Z.; Baer, C.; Walter, U. Quality of oral anticoagulation with phenprocoumon in regular medical care and its potential for improvement in a telemedicine-based coagulation service–results from the prospective, multi-center, observational cohort study thrombEVAL. BMC Med. 2015, 13, 14. [Google Scholar] [CrossRef] [PubMed]
- Jang, I.; Choo, S.; Kim, K. Warfarin Therapy after Mechanical Valve Replacement: A Tool for Predicting Bleeding Events. J. Hear. Valve Dis. 2017, 26, 215–233. [Google Scholar]
- Nasser, S.; Mullan, J.; Bajorek, B. Challenges of older patients’ knowledge about warfarin therapy. J. Prim. Care Community Health 2012, 3, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Shilbayeh, S.A.R.; Almutairi, W.A.; Alyahya, S.A.; Alshammari, N.H.; Shaheen, E.; Adam, A. Validation of knowledge and adherence assessment tools among patients on warfarin therapy in a Saudi hospital anticoagulant clinic. Int. J. Clin. Pharm. 2018, 40, 56–66. [Google Scholar] [CrossRef]
- Sabate, E. Adherence to Long-Term Therapies: Evidence for Action; World Health Organization: Geneva, Switherland, 2003; Available online: http://www.who.int/chronic_conditions/en/adherence_report.pdf (accessed on 25 May 2021).
- Rakhshan, M.; Najafi, H.; Valizadeh, G.A. Lifestyle of patients with atrial fibrillation following self-management interventions: A randomized clinical trial. J. Caring Sci. 2019, 8, 83. [Google Scholar] [CrossRef] [PubMed]
- Miles, C.; Arden-Close, E.; Thomas, M.; Bruton, A.; Yardley, L.; Hankins, M.; Kirby, S.E. Barriers and facilitators of effective self-management in asthma: Systematic review and thematic synthesis of patient and healthcare professional views. NPJ Prim. Care Respir. Med. 2017, 27, 1–21. [Google Scholar] [CrossRef]
- Hibbard, J.H.; Greene, J.; Tusler, M. Improving the outcomes of disease management by tailoring care to the patient’s level of activation. Am. J. Manag. Care 2009, 15, 353–360. [Google Scholar]
- Stephan, L.S.; Almeida, E.D.; Guimarães, R.B.; Ley, A.G.; Mathias, R.G.; Assis, M.V.; Leiria, T.L.L. Oral anticoagulation in atrial fibrillation: Development and evaluation of a mobile health application to support shared decision-making. Arq. Bras. Cardiol. 2018, 110, 7–15. [Google Scholar] [CrossRef]
- Seaburg, L.; Hess, E.P.; Coylewright, M.; Ting, H.H.; McLeod, C.J.; Montori, V.M. Shared decision making in atrial fibrillation: Where we are and where we should be going. Circulation 2014, 129, 704–710. [Google Scholar] [CrossRef] [PubMed]
- Kotecha, D.; Chua, W.W.; Fabritz, L.; Hendriks, J.; Casadei, B.; Schotten, U.; Vardas, P.; Heidbuchel, H.; Dean, V.; Kirchhof, P. European Society of Cardiology smartphone and tablet applications for patients with atrial fibrillation and their health care providers. EP Eur. 2018, 20, 225–233. [Google Scholar] [CrossRef] [PubMed]
- Whitehead, L.; Seaton, P. The effectiveness of self-management mobile phone and tablet apps in long-term condition management: A systematic review. J. Med. Internet Res. 2016, 18, e4883. [Google Scholar] [CrossRef]
- Hui, C.Y.; Creamer, E.; Pinnock, H.; McKinstry, B. Apps to support self-management for people with hypertension: Content analysis. JMIR Mhealth Uhealth 2019, 7, e13257. [Google Scholar] [CrossRef]
- Alessa, T.; Hawley, M.S.; Hock, E.S.; de Witte, L. Smartphone apps to support self-management of hypertension: Review and content analysis. JMIR Mhealth Uhealth 2019, 7, e13645. [Google Scholar] [CrossRef]
- Pearsons, A.; Hanson, C.L.; Gallagher, R.; O’Carroll, R.E.; Khonsari, S.; Hanley, J.; Strachan, F.E.; Mills, N.L.; Quinn, T.J.; McKinstry, B. Atrial fibrillation self-management: A mobile telephone app scoping review and content analysis. Eur. J. Cardiovasc. Nurs. 2021, 20, 305–314. [Google Scholar] [CrossRef] [PubMed]
- Lalloo, C.; Shah, U.; Birnie, K.A.; Davies-Chalmers, C.; Rivera, J.; Stinson, J.; Campbell, F. Commercially available smartphone apps to support postoperative pain self-management: Scoping review. JMIR Mhealth Uhealth 2017, 5, e162. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; Group, P. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef]
- Downs, S.H.; Black, N. The feasibility of creating a checklist for the assessment of the methodological quality both of randomised and non-randomised studies of health care interventions. J. Epidemiol. Community Health 1998, 52, 377–384. [Google Scholar] [CrossRef]
- O’Connor, S.R.; Tully, M.A.; Ryan, B.; Bradley, J.M.; Baxter, G.D.; McDonough, S.M. Failure of a numerical quality assessment scale to identify potential risk of bias in a systematic review: A comparison study. BMC Res. Notes 2015, 8, 224. [Google Scholar] [CrossRef] [PubMed]
- Hooper, P.; Jutai, J.W.; Strong, G.; Russell-Minda, E. Age-related macular degeneration and low-vision rehabilitation: A systematic review. Can. J. Ophthalmol. 2008, 43, 180–187. [Google Scholar] [CrossRef]
- Silverman, S.; Schertz, L.; Yuen, H.; Lowman, J.; Bickel, C. Systematic review of the methodological quality and outcome measures utilized in exercise interventions for adults with spinal cord injury. Spinal Cord. 2012, 50, 718–727. [Google Scholar] [CrossRef] [PubMed]
- Garrard, J. Health Sciences Literature Review Made Easy: The Matrix Method; Jones & Bartlett Publishers: Burlington, MA, USA, 2011. [Google Scholar]
- Prochaska, J.H.; Göbel, S.; Keller, K.; Coldewey, M.; Ullmann, A.; Lamparter, H.; Schulz, A.; Schinzel, H.; Bickel, C.; Lauterbach, M.; et al. e-Health-based management of patients receiving oral anticoagulation therapy: Results from the observational thrombEVAL study. J. Thromb. Haemost. 2017, 15, 1375–1385. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.W.; Kang, W.Y.; Lin, D.T.; Lee, J.C.S.; Wu, F.L.L.; Chen, C.L.; Tseng, Y.J. Comparison of warfarin therapy clinical outcomes following implementation of an automated mobile phone-based critical laboratory value text alert system. BMC Med. Genom. 2014, 7, S13. [Google Scholar] [CrossRef]
- Guo, Y.; Lane, D.A.; Wang, L.; Zhang, H.; Wang, H.; Zhang, W.; Wen, J.; Xing, Y.; Wu, F.; Xia, Y. Mobile health technology to improve care for patients with atrial fibrillation. J. Am. Coll. Cardiol. 2020, 75, 1523–1534. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Guo, J.; Shi, X.; Yao, Y.; Sun, Y.; Xia, Y.; Yu, B.; Liu, T.; Chen, Y.; Lip, G.Y. Mobile health technology-supported atrial fibrillation screening and integrated care: A report from the mAFA-II trial Long-term Extension Cohort. Eur. J. Internet Med. 2020, 82, 105–111. [Google Scholar] [CrossRef]
- Shilbayeh, S.A.R.; Abutaily, S.A.; Al Ghwairi, L.S.; Al Madani, W.O.; Almoussa, A.F.; Alzahrani, S.A. Development and testing of an educational mobile application for improving knowledge among saudi patients receiving warfarin. Int. Res. J. Pharm. 2019, 10, 213–221. [Google Scholar] [CrossRef]
- Labovitz, D.L.; Shafner, L.; Reyes Gil, M.; Virmani, D.; Hanina, A. Using Artificial Intelligence to Reduce the Risk of Nonadherence in Patients on Anticoagulation Therapy. Stroke 2017, 48, 1416–1419. [Google Scholar] [CrossRef]
- Lee, J.A.; Evangelista, L.S.; Moore, A.A.; Juth, V.; Guo, Y.; Gago-Masague, S.; Lem, C.G.; Nguyen, M.; Khatibi, P.; Baje, M.; et al. Feasibility Study of a Mobile Health Intervention for Older Adults on Oral Anticoagulation Therapy. Gerontol. Geriatr. Med. 2016, 2, 2333721416672970. [Google Scholar] [CrossRef]
- Guo, Y.; Chen, Y.; Lane, D.A.; Liu, L.; Wang, Y.; Lip, G.Y. Mobile health technology for atrial fibrillation management integrating decision support, education, and patient involvement: mAF app trial. Am. J. Med. 2017, 130, 1388–1396. [Google Scholar] [CrossRef] [PubMed]
- Talboom-Kamp, E.; Verdijk, N.A.; Kasteleyn, M.J.; Harmans, L.M.; Talboom, I.; Numans, M.E.; Chavannes, N.H. Effect of a combined education and eHealth programme on the control of oral anticoagulation patients (PORTALS study): A parallel cohort design in Dutch primary care. BMJ Open 2017, 7, e017909. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Guhl, E.; Althouse, A.D.; Pusateri, A.M.; Kimani, E.; Paasche-Orlow, M.K.; Bickmore, T.W.; Magnani, J.W. The Atrial Fibrillation Health Literacy Information Technology Trial: Pilot Trial of a Mobile Health App for Atrial Fibrillation. JMIR Cardio 2020, 4, e17162. [Google Scholar] [CrossRef] [PubMed]
- Hoppe, C.D.; Cade, J.E.; Carter, M. An evaluation of diabetes targeted apps for Android smartphone in relation to behaviour change techniques. J. Hum. Nutr. Diet. 2017, 30, 326–338. [Google Scholar] [CrossRef]
- Bonoto, B.C.; de Araújo, V.E.; Godói, I.P.; de Lemos, L.L.P.; Godman, B.; Bennie, M.; Diniz, L.M.; Junior, A.A.G. Efficacy of mobile apps to support the care of patients with diabetes mellitus: A systematic review and meta-analysis of randomized controlled trials. JMIR Mhealth Uhealth 2017, 5, e4. [Google Scholar] [CrossRef] [PubMed]
- Poli, D.; Antonucci, E.; Pengo, V.; Migliaccio, L.; Testa, S.; Lodigiani, C.; Coffetti, N.; Facchinetti, R.; Serricchio, G.; Falco, P.; et al. Mechanical prosthetic heart valves: Quality of anticoagulation and thromboembolic risk. The observational multicenter PLECTRUM study. Int. J. Cardiol. 2018, 267, 68–73. [Google Scholar] [CrossRef]
| PubMed | (“Anticoagulants”[Mesh] OR Anticoagulants[ti] OR Anticoagulation[ti] OR “Warfarin”[Mesh] OR Warfarin[ti]) AND (“Mobile health application”[ti] OR Telemedicine[ti] OR “Telemedicine”[mesh] OR “Mobile Health”[ti] OR mHealth[ti] OR “Mobile Applications”[Mesh] OR “Mobile App”[ti] OR “Mobile Applications”[ti] OR “Mobile Application”[ti] OR “home monitoring”[ti] OR Platform[ti] OR “Smartphone”[Mesh] OR “Smart phone”[ti] OR Smartphone[ti] OR “Cell Phone”[ti] OR “Cell Phones”[ti] OR “Mobile Phone”[ti] OR “Mobile Phones”[ti]) |
| Embase | (‘anticoagulant agent’/mj OR ‘anti coagulant’:ti OR ‘anti coagulant agent’:ti OR ‘anti coagulant drug’:ti OR ‘anti coagulating agent’:ti OR … OR ‘wafarin’:ti OR ‘waran’:ti OR ‘warf compound 42’:ti OR ‘warfar’:ti OR ‘warfarin’:ti OR ‘warfarin 2 (dimethylamino) ethanol’:ti OR ‘warfarin potassium’:ti OR ‘warfarin sodium’:ti OR ‘warfarine’:ti OR ‘warfarinum sodium’:ti OR ‘warfil 5’:ti OR ‘warfilone’:ti OR ‘warnerin’:ti ) AND (‘mobile health application’/mj OR ‘moovcare’:ti OR ‘neomate’:ti OR ‘ispo2 (mobile health application)’:ti OR ‘mobile health application’:ti OR … OR ‘smartphone’/mj OR ‘smart phone’:ti OR ‘smartphone’:ti OR ‘smartphones’:ti OR ‘mobile phone’/mj OR ‘cell phone’:ti OR ‘cell phones’:ti OR ‘cellphone’:ti OR ‘cellphones’:ti OR ‘cellular phone’:ti OR ‘cellular telephone’:ti OR ‘mobile phone’:ti OR ‘mobile telephone’:ti) |
| CINAHL | ((MM “Anticoagulants+”) OR (MM “Warfarin”) OR TI (Anticoagulants OR Anticoagulation OR Warfarin)) AND ((MM “Telemedicine+”) OR (MM “Telehealth+”) OR (MM “Mobile Applications”) OR (MM “Cellular Phone+”) OR (MM “Smartphone”) OR TI (“Mobile health application” OR Telemedicine OR “Mobile Health” OR Telehealth OR mHealth OR “Mobile App” OR “Mobile Applications” OR “Mobile Application” OR “home monitoring” OR Platform OR “Smart phone” OR Smartphone OR “Cellular Phone” OR “Cell Phone” OR “Cell Phones” OR “Mobile Phone” OR “Mobile Phones”)) |
| Cochrane | ((MeSH descriptor: [Anticoagulants] explode all trees) OR (MeSH descriptor: [Warfarin] explode all trees) OR ((Anticoagulants OR Anticoagulation OR Warfarin):ti)) AND ((MeSH descriptor: [Telemedicine] explode all trees) OR (MeSH descriptor: [Mobile Applications] explode all trees) OR (MeSH descriptor: [Smartphone] explode all trees) OR (MeSH descriptor: [Cell Phone] explode all trees) OR ((“Mobile health application” OR Telemedicine OR “Mobile Health” OR Telehealth OR mHealth OR “Mobile App” OR “Mobile Applications” OR “Mobile Application” OR “home monitoring” OR Platform OR “Smart phone” OR Smartphone OR “Cellular Phone” OR “Cell Phone” OR “Cell Phones” OR “Mobile Phone” OR “Mobile Phones”):ti)) |
| Author Year Country | Research Design | Sample Size | Age | Setting | Inclusion Criteria | Program | Contents | Study Outcomes | Duration of Intervention | Teaching Methods |
|---|---|---|---|---|---|---|---|---|---|---|
| Guhl et al. 2020 USA | RCT | 120 Exp. 61 Cont. 59 | 72.1 ± 9.1 | Hospital | ∙ age ≥ 18 years ∙ history of chronic AF ∙ prescribed oral anticoagulation ∙ English-speaking | Kardia | ∙ education ∙ symptoms ∙ adherence ∙ patient activation | ∙ HRQoL with the Atrial Fibrillation Effect on Quality of Life (AFEQT) ∙ self-reported adherence | 30 days | mHealth app (Kardia apps) |
| Guo et al. 2017 China | RCT | 209 Exp. 113 Cont. 96 | 69.0 | Hospital | ∙ age > 18 years ∙ patients with atrial fibrillation ∙ patients without valvular atrial fibrillation | Mobile AF (mAF) | ∙ clinical decision-support tools (CHA2DS2-VASc, HAS-BLED, SAMe-TT2R2) ∙ education program ∙ self-care items (Euro EQ-5D-Y) ∙ structured follow-up | ∙ knowledge ∙ QOL ∙ drug adherence ∙ anticoagulation satisfaction ∙ usability, feasibility, acceptability of the mAF App | 3 months | mHealth app |
| Guo et al. 2020 China | RCT | 3324 Exp. 1646 Cont. 1678 | 68.5 | Hospital | ∙ age ≥ 18 years ∙ patients with a new-onset of paroxysmal, persistent, or permanent atrial fibrillation with ECG ∙ CHA2DS2-VASc ≥ 2 | mAFA | ∙ clinical decision-support tools (CHA2DS2-VASc, HAS-BLED, SAMe-TT2R2) ∙ education program ∙ self-care items (Euro EQ-5D-Y) ∙ structured follow-up ∙ the ABC pathway for AF management | ∙ clinical outcomes (thromboembolism, bleeding events, cardiovascular outcomes, all-cause death, re-hospitalization, composite outcome) | 12 months | mHealth app |
| Guo et al. 2020 China | RCT | 2473 Exp. 1261 Cont. 1212 | 56.7 ± 13.7 | Hospital | ∙ age > 18 years ∙ patients with atrial fibrillation with ECG ∙ CHA2DS2-VASc ≥ 2 | mAFAII | ∙ the ABC pathway for AF management | ∙ clinical outcomes (thromboembolism, bleeding events, recurrent AF or AF symptom, heart failure, re-hospitalization. all-cause death) ∙ adherence and persistence | 1 year | mHealth app |
| Labovitz et al. 2017 USA | RCT | 28 Exp. 15 Cont. 13 | 57.0 ± 13.2 | Hospital | ∙ patients diagnosed with ischemic stroke ∙ oral anticoagulation therapy | ∙ visual confirmation of ingestion ∙ self-reported dose via the AI app ∙ self-reported dose by clinic staff ∙ missed dose ∙ dose taken in a clinic | ∙ adherence ∙ PT/INR, APTT ∙ usability and feasibility | 12 weeks | AI platform | |
| Lee et al. 2016 USA | One group pre-posttest design | 21 | 67.3 ± 8.7 | Hospital | ∙ age ≥ 55 years ∙ taking an oral anticoagulant ∙ English-speaking or Spanish-speaking ∙ not cognitively impaired ∙ not living in a long-term care facility | Mobile Applications for Seniors to enhance Safe anticoagulation therapy (MASS) | ∙ education about anticoagulation therapies and safety tips ∙ medication self-monitoring and reminders ∙ Vitamin K content of foods ∙ monitoring signs and symptoms of bleeding ∙ monitoring blood (INR) ∙ connecting with trusted other people ∙ message to tell doctors | ∙ OAK test ∙ Perception of Anticoagulant Treatment Questionnaire (PACTQ) ∙ Duke Anticoagulation Satisfaction Scale (DASS) ∙ Patient Health Questionnaire (PHQ-9) ∙ Brief Symptom Inventory (BSI) ∙ Morisky medication adherence scale (MMAS) ∙ Perceived Health Web Site Usability Questionnaire (PHWSUQ-12) | 3 months | mHealth app |
| Lin et al. 2014 Taiwan | Retrospective cohort design | 7278 Exp. 3781 Cont. 3497 | 59.3 | Hospital | ∙ patients engaged in warfarin therapy | Personal Handy-phone System (PHS) | ∙ PHS alert system | ∙ PT/INR ∙ complications (thromboembolic or hemorrhagic events) ∙ major hemorrhage, event ∙ treatment with vitamin K | 2 years | Mobile phone network system |
| Prochaska et al. 2017 Germany | Prospective cohort design | 2318 Exp. 760 Cont. 1558 | 73.0 | Hospital | ∙ patients with an oral anticoagulation therapy | Poltavita | ∙ INR values ∙ computer-assisted dosing algorithms | ∙ efficacy ∙ safety ∙ all-cause mortality ∙ hospitalization | 12–14 months | web-based electronic patient file |
| Prochaska et al. 2015 Germany | Prospective cohort design | 2771 Exp. 760 Cont. 2011 | 73.0 | Hospital | ∙ age ≥ 18 years ∙ taking an oral anticoagulant ≥ 4 months | ThrombEVAL study | ∙ self-management of OAC with INR values | ∙ TTR of INR | 3 months | Telemedicine-based coagulation service |
| Shilbayeh et al. 2019 Saudi Arabia | One group pre-posttest design | 45 | 45.8 ± 12.8 | Hospital | ∙ age > 18 years ∙ Arabic-speaking ∙ receiving OAT (mainly warfarin) and visiting the clinic regularly ∙ a naïve patient ∙ using smartphones or tablets with Android | Coagulation and Anticoagulant Therapy and Awareness (CATA) | ∙ Oral anticoagulation knowledge (OAK) scale ∙ Anti-clot Treatment Scale (ACTS) ∙ Treatment Satisfaction Questionnaire for Medication (TSQM 1.4) | ∙ Oral anticoagulation knowledge (OAK) scale ∙ Anti-clot Treatment Scale (ACTS) ∙ Treatment Satisfaction Questionnaire for Medication (TSQM 1.4) | 45 days | mHealth app |
| Stephan et al. 2018 Brazil | One group pre-posttest design | 30 Exp. 20 Pilot 10 | 67.7 ± 9.4 | Hospital | ∙ patients with AF and low socioeconomic and cultural status ∙ using OAC | ∙ knowing the disease (video) ∙ individualizing the risks ∙ understanding risks and benefits ∙ knowing the treatment option ∙ making a choice | ∙ AF knowledge questionnaire ∙ Decisional Conflict Scaled in Health (DSCH) | During medical visits | dactor’s tablet computer | |
| Talboom-Kamp et al. 2017 Netherlands | Parallel cohort design | 195 E-learning 52 Group course 58 Basic training 85 | 66.9 | Hospital | ∙ long-term indication for anticoagulants ∙ self-management | PORTALS study: Poltavita | ∙ disease-specific knowledge ∙ self-testing skills ∙ use of the web portal ∙ self-adjustment of medication | ∙ TTR of INR ∙ usage of an eHealth platform ∙ Generalized Self-Efficacy Scale (GSES), education level | 18 months | eHealth platform |
| Author (Year) | Reporting (11) * | External Validity (3) * | Internal Validity: Bias (7) * | Internal Validity: Confounding (6) * | Power (1) * | Total Score (28) |
|---|---|---|---|---|---|---|
| Guhl et al. (2020) | 9 | 3 | 7 | 4 | 1 | 24 |
| Guo et al. (2017) | 10 | 3 | 7 | 5 | 1 | 26 |
| Guo et al. (2020) | 10 | 3 | 6 | 4 | 1 | 24 |
| Guo et al. (2020) | 10 | 3 | 6 | 5 | 1 | 24 |
| Labovitz et al. (2017) | 6 | 0 | 3 | 2 | 0 | 11 |
| Lee et al. (2016) | 7 | 0 | 5 | 3 | 0 | 15 |
| Lin et al. (2014) | 7 | 1 | 4 | 2 | 1 | 15 |
| Prochaska et al. (2015) | 6 | 1 | 5 | 2 | 1 | 15 |
| Prochaska et al. (2017) | 6 | 1 | 4 | 2 | 1 | 14 |
| Shilbayeh et al. (2019) | 7 | 0 | 5 | 2 | 0 | 14 |
| Stephan et al. (2018) | 6 | 0 | 5 | 2 | 0 | 13 |
| Talboom-Kamp et al. (2017) | 8 | 1 | 5 | 2 | 0 | 16 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jang, I. A Systematic Review on Mobile Health Applications’ Education Program for Patients Taking Oral Anticoagulants. Int. J. Environ. Res. Public Health 2021, 18, 8902. https://doi.org/10.3390/ijerph18178902
Jang I. A Systematic Review on Mobile Health Applications’ Education Program for Patients Taking Oral Anticoagulants. International Journal of Environmental Research and Public Health. 2021; 18(17):8902. https://doi.org/10.3390/ijerph18178902
Chicago/Turabian StyleJang, Insil. 2021. "A Systematic Review on Mobile Health Applications’ Education Program for Patients Taking Oral Anticoagulants" International Journal of Environmental Research and Public Health 18, no. 17: 8902. https://doi.org/10.3390/ijerph18178902
APA StyleJang, I. (2021). A Systematic Review on Mobile Health Applications’ Education Program for Patients Taking Oral Anticoagulants. International Journal of Environmental Research and Public Health, 18(17), 8902. https://doi.org/10.3390/ijerph18178902






