Abstract
Many fungal quinazolinone metabolites, which contain the methyl-indole pyrazino [1,2-b]quinazoline-3,6-dione core, have been found to possess promising antitumor activity. The purpose of this work was to synthesize the enantiomeric pairs of two members of this quinazolinone family, to explore their potential as antitumor and their ability to revert multidrug resistance. The marine natural product fiscalin B (4c), and antienantiomers (4b, 5b, and 5c) were synthesized via a one-pot approach, while the syn enantiomers (4a, 4d, 5a, and 5d) were synthetized by a multi-step procedure. These strategies used anthranilic acid (i), chiral N-protected α-amino acids (ii), and tryptophan methyl esters (iii) to form the core ring of pyrazino[2,1-b]quinazoline-3,6-dione scaffold. Four enantiomeric pairs, with different enantiomeric purities, were obtained with overall yields ranging from 7 to 40%. Compounds 4a–d and 5a–d were evaluated for their growth inhibitory effect against two tumor cell lines. Differences between enantiomeric pairs were noted and 5a–d displayed GI50 values ranging from 31 to 52 μM, which are lower than those of 4a–d. Nevertheless, no effect on P-glycoprotein (P-gp) modulation was observed for all compounds. This study disclosed new data for fiscalin B (4c), as well as for its analogues for a future development of novel anticancer drug leads.
1. Introduction
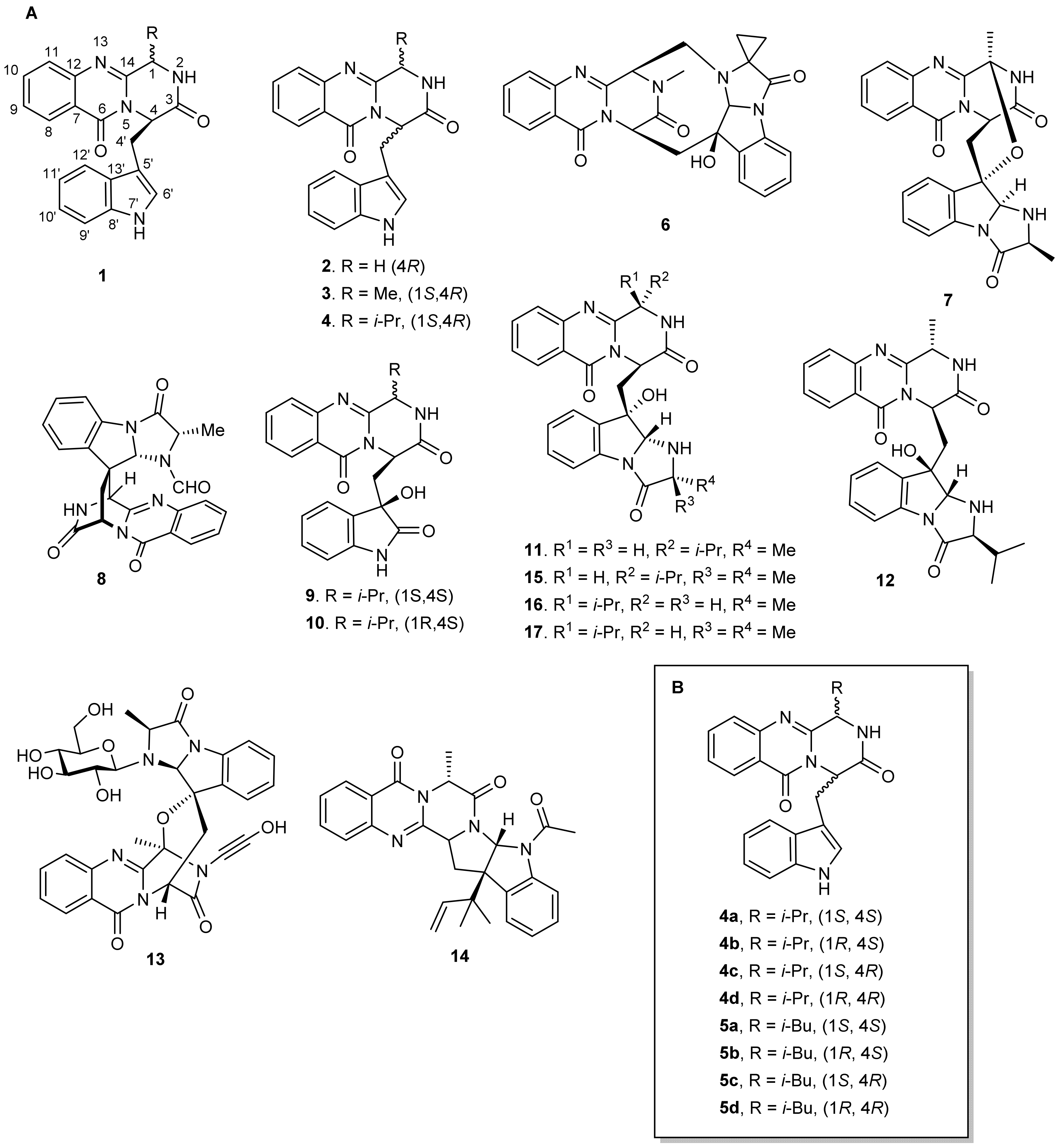
During the past ten years, great attention has been focused on drug development from Marine Natural Products (MNPs) as well as on their synthetic and semi-synthetic analogues. While terrestrial sources such as higher plants and microorganisms have reached the limelight, the marine environment increasingly becomes the newest and untapped resource of bioactive compounds [1]. A number of quinazolinone derivatives have played important roles in medicinal chemistry due to their broad spectrum of biological properties such as antibacterial, antifungal, anticonvulsant, anti-inflammatory, anti-HIV, anticancer, and analgesic activities [2,3]. One subclass of quinazolinone-derived Natural Products are the indolylmethyl pyrazinoquinazoline alkaloids (1) (Figure 1), characterized by a fused piperazine ring system linked to an indole moiety. So far, approximately 80 secondary metabolites of this subclass, covering structurally diverse compounds, have been isolated from fungi, mainly of marine origin. These include (i) compounds containing only a substituted piperazine ring such as glyantrypine (2), isolated from the culture broth of the mangrove-derived fungus Cladosporium sp. PJX-41 [4], fumiquinazoline F (3), isolated from the marine-derived fungus Aspergillus fumigatus strain H1-04 and Aspergillus sp. [5,6], and fiscalin B (4), isolated from the culture of the fugal strain Dichotomomyces cejpii which was recovered from sediments of the Brazilian coast [7]; (ii) compounds with a more complex structure containing several rings such as fumiquinazoline K (6), isolated from the Mediterranean sponge-derived fungus Aspergillus sp., the soft coral (Sinularia sp.)-associated fungus Aspergillus fumigatus KMM 4631 [8], and a gorgonian-associated fungus [9]; (iii) spiro compounds such as fumiquinazoline C (7), isolated from the marine-derived fungus Aspergillus fumigatus strain H1-04, and N-formyllapatin A (8), isolated from the marine-derived fungus Penicillium adametziodes (AS-53) [10]; (iv) compounds with complex 3-indolyl groups such as cladoquinazoline (9) and epi-cladoquinazoline (10), isolated from the mangrove-derived fungus Cladosporium sp. PJX-41 [4], neofiscalin A (11) from Neosartorya siamensis KUFC 6349, which was isolated from a forest soil [11], as well as from N. siamensis KUFC 6349, isolated from the sea fan Rumphella sp. [12], and fumiquinaziline S (12), isolated from a solid-substrate culture of Aspergillus sp., collected from a marine-submerged wood [13]; or (v) compounds with indole glucosides such as fumigatoside A (13), isolated from Aspergillus fumigatus which was derived from the jellyfish Nemopilema nomurai [14].
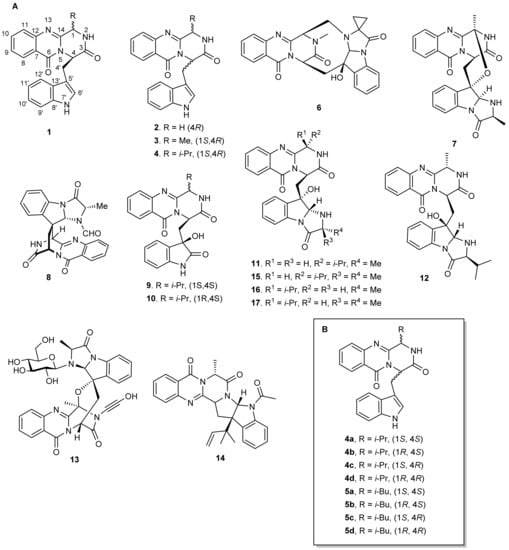
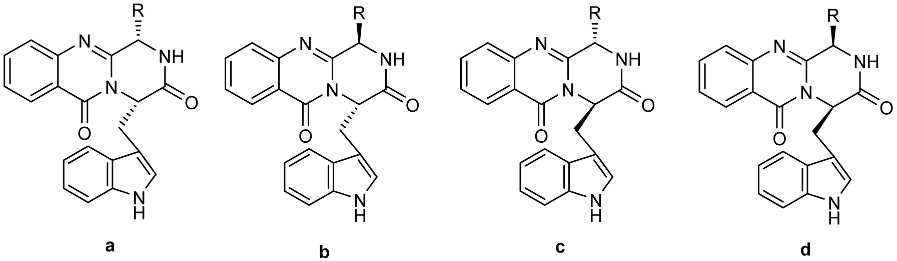
Figure 1.
(A) Pyrazino[2,1-b]quinazoline-3,6-dione ring system (1) and some examples of the marine-derived quinazolinone containing alkaloids (2–17) and (B) synthesized diastereomers of fiscalin B and their homologues (4–5).
Marine alkaloids containing indolymethyl pyrazinoquinazoline ring system can be considered conformationally constrained peptidomimetics exhibiting very interesting biological properties [15]. For instance, glyantrypine (2) is an antibacterial agent, active against Vibrio harveyi [16]; fumiquinazolines are antitumor compounds with moderate cytotoxicity [17]; fiscalins are substance P inhibitors and anticancer agents [7,18]; cladoquinazolines (9 and 10) are active against influenza A virus (H1N1); fumiquinazoline S (12) exhibits a weak inhibition against Na+/K+-ATPase, and N-acetylardeemin (14) is a potent inhibitor of multidrug resistant (MDR) tumor cells [4,13,19,20]. Moreover, the pyrazino[2,1-b]quinazoline ring system has been already ascribed as essential for the above-mentioned activities, and its enantioselective effects were observed for their antibacterial activity. For example, neofiscalin A (11) showed potent antibacterial activity against Gram-negative bacteria [12,21], whereas fiscalin C (15), epi-neofiscalin A (16), and epi-fiscalin C (17) were inactive in the same study. Interestingly, fiscalin C (15) displayed a synergistic effect against methicillin-resistant Staphylococcus aureus (MRSA) when combined with oxacillin [11,22]. Concerning antitumor activity, studies on quinazolinone compounds have mainly focused on natural or synthetic compounds with different substituents at the stereogenic C-1 and C-4; however, there is no report on analogues with different configurations at C-1 and C-4.
Therefore, the aim of this study was to synthesize the diastereomers of fiscalin B (4c), i.e., 4a–d, and their homologues 5a–d, to further explore their potential as growth inhibitors of tumor cells, their ability to revert MDR by inhibiting P-gp activity, as well as to perform the SAR study.
2. Results and Discussion
2.1. Synthesis of Pyrazinoquinazoline Alkaloids
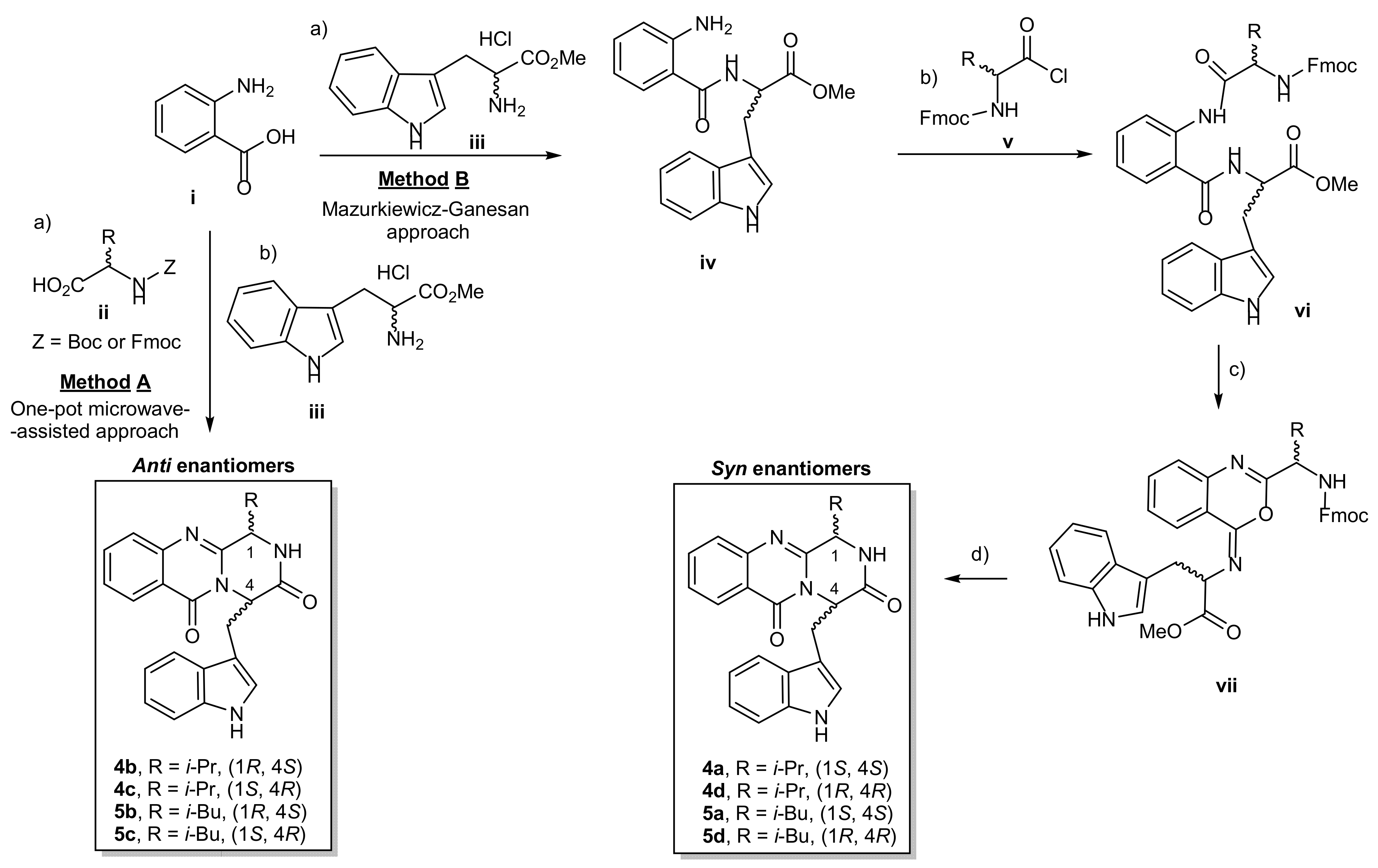
The pyrazino[2,1-b]quinazoline-3,6-dione ring system (1) is the core structure of fumiquinazoline-derived group of alkaloids. There are two main methods to synthesize compounds containing this scaffold (i) the Eguchi-aza Wittig approach that consists of a selective acylation of diketopiperazines with o-azidobenzoyl chloride, followed by dehydrative cyclization [23]; and (ii) the Mazurkiewicz–Ganesan approach, consisting of coupling of linear tripeptides followed by the isomerization of 4-imino-4H-3,1-benzoxozines to obtain the corresponding quinazolin-4-ones [24,25]. In 2005, Liu et al. [26] reported a highly effective and environmentally friendly approach using a microwave-assisted multicomponent one-pot one step polycondensation of amino acids for the total syntheses of glyantrypine (2), fumiquinazoline F (3), and fiscalin B (4). With this procedure, the authors reported that the addition of a N-protected α-amino acid (ii) to anthranilic acid (i), under a conventional heating condition at 55 °C with triphenylphosphite, (PhO)3P, generated the intermediate benzoxazin-4-one, followed by the addition of tryptophan (Trp) ester (iii), and then submitted to microwave irradiation at 220 °C for 1.5 min, to furnish the desired final products. Inspired by this simple and highly efficient methodology, we were able to prepare 4 and 5 (Scheme 1, method A). Attempts to obtain the syn enantiomers by this one-pot approach failed since only vestigial amounts could be detected due to the isomerization to the antienantiomers. The antienantiomers 4b/4c and 5b/5c were then obtained, starting from enantiomeric pure amino acids, from which no syn enantiomer were isolated (Entry 1–8, Table 1). Therefore, the syn enantiomers 4a/d and 5a/d were synthesized by the Mazurkiewicz–Ganesan method [25] (Scheme 1, method B) with some modifications. First, coupling of i with iii, using 1,1,3,3-tetramethylaminium tetrafluoroborate (TBTU) in basic conditions, afforded the dipeptide (iv). Coupling of iv with N-protected α-amino acids chloride (v) [27] in a two-phase Schotten–Baumann condition yielded the tripeptides vi. The intermediates vii were obtained by adding the dehydrating agent, triphenylphosphine (Ph3P), to convert β-keto amides (vi) to the oxazoles (vii), followed by Fmoc-deprotection by 20% piperidine to afford 4a/d and 5a/d with a moderate yields of 21–40%. After purification by chromatographic techniques, the purity of 4a–d and 5a–d, as determined by a reversed-phase HPLC (C18, MeOH:H2O; 60:40 or CH3CN:H2O; 50:50) was found to be higher than 90% (Supplementary Materials, Figures S1–S8). The enantiomers ratio was determined by the chiral HPLC equipped with amylose tris-3,5-dimethylphenylcarbamate column, using hexane: ethanol (80:20) as a mobile phase (Table 1, Supplementary Materials, Figures S9–S16).
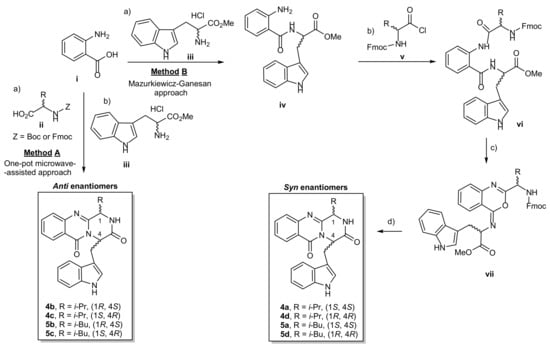
Scheme 1.
Synthesis of the pyrazinoquinazolinone alkaloids 4 and 5. (Method A) One-pot microwave-assisted approach. Reagents and conditions: (a) dried-pyridine, (PhO)3P, 55 °C, 16–24 h; (b) dried-pyridine, (PhO)3P, 220 °C, 1.5 min; (Method B) Mazurkiewicz–Ganesan approach. Reagents and conditions (a) CH3CN, TBTU, Et3N, rt, 5 h; (b) CH2Cl2/aq.Na2CO3, rt, 3 h; (c) dried CH2Cl2, Ph3P, I2, EtN(i-Pr)2, rt, overnight; (d) piperidine in CH2CH2, rt, 12 min, then CH3CN, DMAP, reflux 19 h. i-Pr = isopropyl; i-Bu = isobutyl, Boc = tert-butyloxycarbonyl; Fmoc = fluorenylmethyloxycarbonyl; DMAP = 4-(dimethylamino) pyridine, TBTU = 1,1,3,3-tetramethylaminium tetrafluoroborate.

Table 1.
Enantiomers and diastereomers of the pyrazinoquinazolinone alkaloids 4 and 5.
The overall yield of this one-step method ranged from 7 to 14% with different enantiomeric ratios (Table 1). The low yields of this one-pot reaction were attributed to a high temperature applied in microwave irradiation to convert the intermediate Boc-protected-benzoxazin-4-one to the final products. Moreover, the steric hindrance at C-1 could also be a reason, as previously noted by Liu et al. [26] and Wang et al. [24] in the synthesis of similar compounds. Although a partial epimerization was observed under these conditions, this one-pot procedure was proved successful in providing the pyrazinoquinazolinone scaffold and a series of compounds for further biological investigations. On the other hand, the moderate yields of compounds obtained by Mazurkiewicz–Ganesan method were related to the mild conditions and a multistep approach (Table 1 entry 9–12). In this study, the coupling agent 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide (ECD) was replaced by TBTU, which gave a similar yield for the dipeptide iv (81–94%) when compared to the previous reports. The tripeptides vi were also obtained in a very high yield (84–94%). The bottleneck of this multistep approach was the conversion of the intermediates vii to the final products since their decomposition to form the precursors vi, similar to what has been previously report by Ganesan et al. [25], was observed. Compounds 4a/d and 5a/d were purified by preparative TLC (EtOAc:CH2Cl2:MeOH: 50:47.5:2.5) after refluxing in CH3CN in the presence of 4-(dimethylamino)pyridine. The degrees of epimerization of 4a/d and 5a/d by Mazurkiewicz–Ganesan method were found to be lower than those obtained by the microwave method (Table 1). These results could be also associated with the mild conditions used in the multistep approach.
2.2. Structure Elucidation
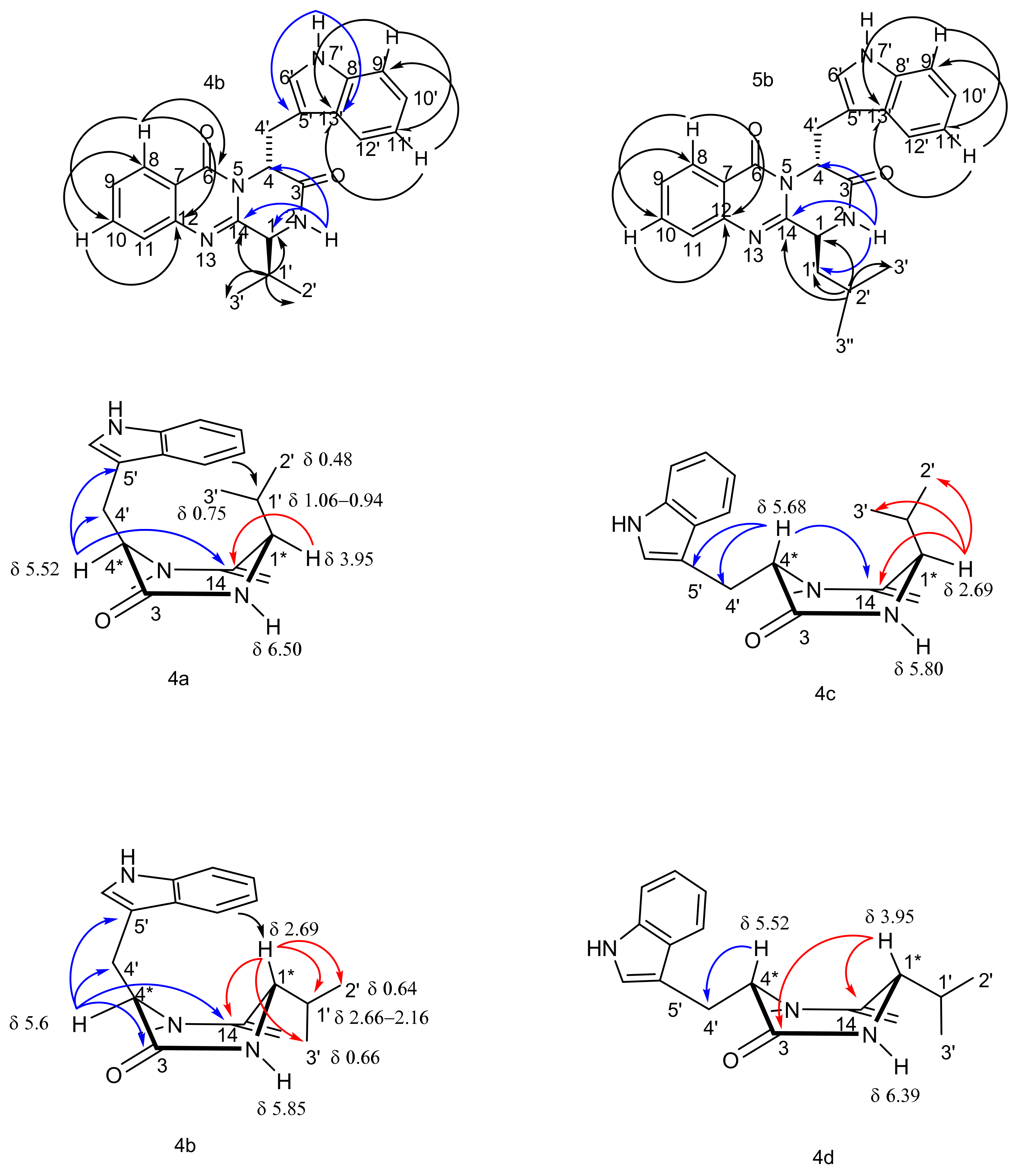
The structures of the new compounds, 4a, 4b, and 4d, and of the synthetic fiscalin B (4c) and their homologues 5a, 5b, 5d, and 5c, were established by extensive analyses of 1D and 2D NMR spectra and high-resolution mass spectrometry. The 1H (Table 2, Supplementary Materials, Figures S17–S30) and 13C NMR data (Table 3, Supplementary Materials, Figures S31–S44) of 4c were in agreement with those reported in the literature for fiscalin B, which was obtained from the marine sources [24,25]. The chemical shift values of some key protons allowed the determination of the relative configurations of both stereogenic centres as well as the conformation of the piperazine ring in 4 and 5, as shown in 4a, 4b and 4c, 4d (Figure 2). The substituent on C-4 is always in a pseudoaxial position while H-4 is shifted to δH 5.52–5.68 in all compounds, showing the characteristic anisotropic effect of the coplanar carbonyl group at C-14 on the quasi-equatorial proton, as explained by Hernández et al. [28]. The difference between anti and syn enantiomers were observed on H-1. In the antienantiomers, the chemical shift value of the axial H-1 of 4b/c and 5b/c at δH 2.69–2.73 indicates the folding of the C-4-indolyl substituent over the piperazine ring and the isopropyl group. This phenomenon was also reported by Hernández and coworkers for H-1 signals of quinazolinones whose chemical shift values were ca. 3 ppm for the boat conformation and the antienantiomers, due to the absence of the shielding effect by the aromatic ring [28]. Meanwhile, for the syn enantiomers, 4a/d and 5a/d, H-1 chemical shift values were at ca. δH 3.95–4.32, indicating the shielding effect from the aromatic ring over H-1. The chemical shifts of H-1′ of the isopropyl group were also different between the anti- and the syn enantiomers, being ca. δH 0.94–1.06 for 4a/d and δH 2.61–2.66 for 4c/d, indicating the different shielding effect of the aromatic ring on the group on C-1.

Table 2.
1H NMR data (300 MHz, CDCl3) for 4 and 5.

Table 3.
13C NMR data (75 MHz, CDCl3) for 4 and 5.
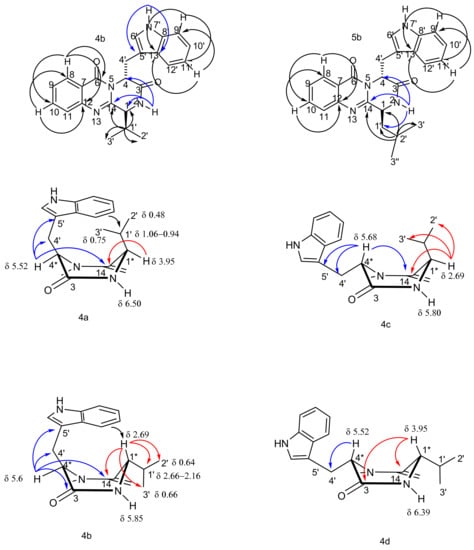
Figure 2.
Most relevant chemical shifts and key HMBC correlations of the protons to the stereogenic centers on the piperazine ring of 4a–d.
HMBC correlations were also used to distinguish the anti-isomers from the syn counterparts. For the anti isomer 4b (whose indole moiety derived from l-Trp), H-4 exhibited correlations to C-14, C-5′, C-4′, and C-3 whereas H-1 showed correlations to C-14, C-2′, C-3′, and C-1′. On the contrary, in 4c (whose indole moiety is derived from d-Trp), the HMBC correlations from H-4 to C-14, C-5′, C-4′ and from H-1 to C-14, C-3′, and C-2′, were observed. For the syn isomer 4a (whose indole moiety derived from l-Trp), the HMBC correlations from H-1 to C-14, and from H-4 to C-14, C-4′, and C-5′ were observed while the HMBC correlations from H-1 to C-3 and C-14 and from H-4 to only C-4′ were observed in the syn isomer 4d. Moreover, the NOESY spectrum revealed the cross peak between the C-1′ methyl groups and H-4 for the anti isomer 4c, while for the syn isomer 4a that correlation was absent (Supplementary Materials, Figures S45–S46). These observations support the identity/identification of the syn and anti conformational isomers.
2.3. Tumor Cell Growth Inhibitory Activity
Compounds 4a–d and 5a–d were tested for their tumor cell growth inhibitory activity against two human tumor cell lines: NCI-H460 (non-small cell lung cancer) and HCT-15 (colorectal adenocarcinoma), using the sulforhodamine B (SRB) colorimetric assay [29]. Five serial dilutions of each compound (at a maximum concentration of 150 μM) were tested for 48 h. Doxorubicin was used as a positive control, and the antitumor activity was reported as GI50 (drug concentration that inhibits the growth of cancer cells by 50%).
Compounds 4a–d, 5a, 5b, and 5d were also investigated for their possible modulatory activity of P-gp, a drug efflux pump associated with drug resistance. P-gp activity was determined by an assay which measures the mean fluorescence intensity of cells treated concomitantly with rhodamine 123 (Rh123, a substrate of P-gp), and the tested compounds [20]. The P-gp inhibitory activity of the compounds was tested on a drug resistant cell line which overexpresses P-gp (K562Dox), by measuring the intracellular accumulation of Rh123. After an incubation with the compounds and Rh123, cells were washed, and the fluorescence of Rh123 was detected by flow cytometry in the FL1 channel. The drug sensitive counterpart cells (K562) were used as control. The Rh123 accumulation ratio was calculated as: (Mean FL1K562Dox+Compound − Mean FL1K562Dox)/Mean FL1K562Dox [30].
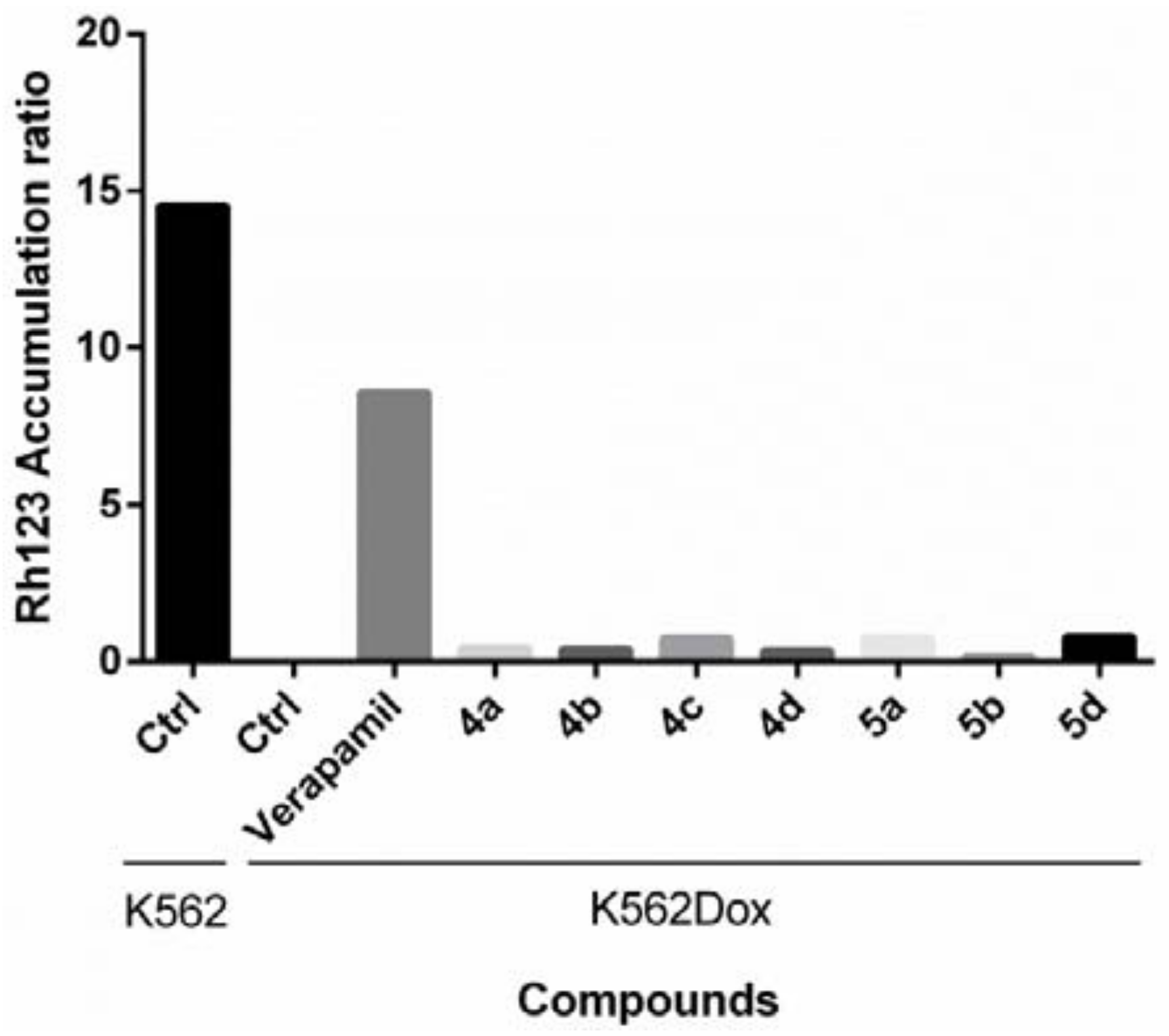
All the compounds tested showed weak to moderate activity, with the GI50 values ranging from 30 to 80 µM. Some differences were observed among the groups of 4 and 5. Compounds 4 were more potent in the HCT-15 cell line but exhibited higher GI50 values in the NCI-H460 cell lines. On the other hand, members of 5 were more potent than those of 4 in the NCI-H460 cell line. Compound 5c was the most promising in this panel of cell lines (Table 4). The substituent at C-1 was found to influence the inhibitory effects observed in the NCI-H460 cell. For example, 4a–d, whose C-1 bears the isopropyl group, exhibited GI50 values ranging from 57 to 81 µM, while 5a–d, whose C-1 bears the isobutyl group, displayed GI50 values ranging from 31 to 42 µM. These findings are in accordance with the SAR obtained with the natural compounds in vitro antitumor assays. For instance, glyantrypine (2), whose C-1 bears a hydrogen atom, showed no antitumor activity (GI50 > 100 µM) while the analogue, with the phenyl group on C-1, was more active (GI50 = 15 µM) [25]. Moreover, fumiquinazolines F (3) and G (4), whose C-1 bears a methyl group, showed moderate activity against P-388 cells (GI50 = 13.5 µM) [31]. Likewise, differences in the inhibitory effects against the two cell lines were observed between enantiomeric pairs; i.e., 4a (1S,4S)/4d (1R,4R) and 4b (1S,1R)/4c (1R,4S). Significant differences were detected for GI50 concentration values in the pair 4a/4d in NCI-H460 cells (p = 0.026). Among the fiscalin series such as epi-fiscalin A (16, 1S,4S), epi-fiscalin C (17, 1S,4S), fiscalin F (1S,4S), and fiscalin C (15, 1R,4S), the configurations of the stereogenic carbons of the isopropyl pyrazinone and imidazolone moieties have already been found to influence the antitumor activity [21,32]. Although this study brought insights into the antitumor activity of fiscalin B (4c) and the synthetic analogues, none of the compounds showed any effect on the intracellular accumulation of Rh123, when tested at 10 µM concentrations using verapamil as a positive control for P-gp inhibition (Figure 3).

Table 4.
The GI50 of 4a–d and 5a–d in the NCI-H460 and HCT-15 human tumor cell lines.
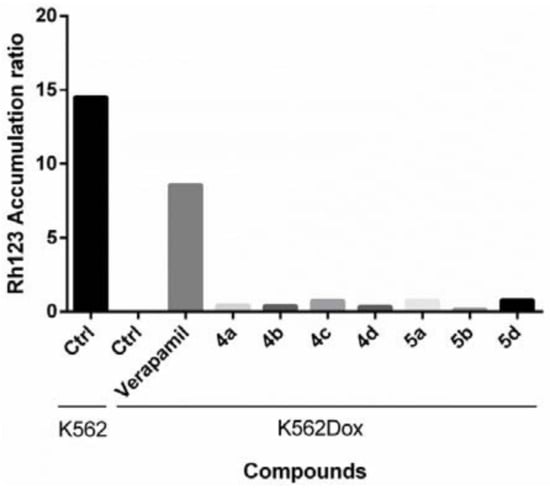
Figure 3.
Accumulation ratio of Rh123 in K562 and K562Dox cell lines. Cells were incubated for 1 h with 4a–d, 5a, 5b, and 5d at a final concentration of 10 µM. The activity of 5c was not analyzed due to its quantity we have obtained. Verapamil (10 µM) was used as a positive control (known P-gp inhibitor), and K562 cells were used as a negative control. The accumulation ratio in the untreated K562Dox cells was defined as zero; any value higher than that represents a potential inhibition of P-gp. Results are the mean of two independent experiments.
3. Materials and Methods
3.1. General Procedure
All reagents were from analytical grade. Dried pyridine and triphenylphosphite were purchased from Sigma (Sigma-Aldrich Co. Ltd., Gillinghan, UK). Protected amino acids (ii) and anthranilic acid (i) were purchased from TCI (Tokyo Chemical Industry Co. Ltd., Chuo-ku, Tokyo, Japan). Column chromatography purifications were performed using flash silica Merck 60, 230–400 mesh (EMD Millipore corporation, Billerica, MA, USA) and preparative TLC was carried out on precoated plates Merck Kieselgel 60 F254 (EMD Millipore corporation, Billerica, MA, USA), spots were visualized with UV light (Vilber Lourmat, Marne-la-Vallée, France). Melting points were measured in a Köfler microscope and are uncorrected. Infrared spectra were recorded in a KBr microplate in a FTIR spectrometer Nicolet iS10 from Thermo Scientific (Waltham, MA, USA) with Smart OMNI-Transmission accessory (Software 188 OMNIC 8.3). 1H and 13C NMR spectra were recorded in CDCl3 (Deutero GmbH, Kastellaun, Germany) at room temperature unless otherwise mentioned on Bruker AMC instrument (Bruker Biosciences Corporation, Billerica, MA, USA), operating at 300 MHz for 1H and 75 MHz for 13C). Carbons were assigned according to HSQC and or HMBC experiments. Optical rotation was measured at 25 °C using the ADP 410 polarimeter (Bellingham + Stanley Ltd., Tunbridge Wells, Kent, UK), using the emission wavelength of sodium lamp, concentrations are given in g/100 mL. Qualitative GC-MS analyses were performed on a Trace GC 2000 Series ThermoQuest gas chromatography (Thermo Fisher Scientific Inc., Austin, TX, USA) equipped with ion-trap GCQ Plus ThermoQuest Finnigan mass detector (Thermo Fisher Scientific Inc., Austin, TX, USA). Chromatographic separation was achieved using a capillary column (30 m × 0.25 mm × 0.25 μm, cross-linked 5% diphenyl and 95% dimethyl polysiloxane) from Thermo ScientificTM (Thermo Fisher Scientific Inc., Austin, TX, USA) and high-purity helium C-60 as carrier gas. High resolution mass spectra (HRMS) were measured on a Bruker FTMS APEX III mass spectrometer (Bruker Corporation, Billerica, MA, USA) recorded as ESI (Electrospray) made in Centro de Apoio Cientifico e Tecnolόxico á Investigation (CACTI, University of Vigo, Pontevendra, Spain). The purity of synthesized compounds was determined by reversed-phase HPLC with diode array detector (DAD) using C18 column (Kimetex®, 2.6 EV0 C18 100 Å, 150 × 4.6 mm), and the mobile phase was methanol: water (60:40) or acetonitrile:water (50:50). Enantiomeric ratio was determined by chiral HPLC (LCMS-2010EV, Shimadzu, Lisbon, Portugal), employing a system equipped with a chiral column (Lux® 5 µm Amylose-1, 250 × 4.6 mm) and UV-detection at 254 nm, mobile phase was hexane:ethanol (80:20) and the flow rate was 0.5 mL/min.
3.2. General Conditions for the Synthesis of 4-(1H-Indol-3-ylmethyl)-1-isopropyl-2H-pyrazino[2,1-b]quinazoline-3,6-(1H,4H)-diones (4b/4c)
In a closed vial, anthranilic acid (i) (28 mg, 200 µmol), N-Boc-l-valine (iia) for 4c or N-Boc-d-valine (iib) for 4b (44 mg, 200 µmol), and triphenylphosphite (63 µL, 220 µmol) were added along with 1 mL of dried pyridine. The vial was heated in heating block with stirring at 55 °C for 16–24 h. After cooling the mixture to room temperature, l-tryptophan methyl ester hydrochloride (iiia) for 4b or d-tryptophan methyl ester hydrochloride (iiib) for 4c (51 mg, 200 µmol) was added, and the mixture was irradiated in the microwave at a constant temperature at 220 °C for 1.5 min. Four reaction mixtures were prepared in the same conditions and treated in parallel. After removing the solvent with toluene, the crude product was purified by flash column chromatography using hexane: EtOAc (60:40) as a mobile phase. The preparative TLC was performed using CH2Cl2:Me2CO (95:5) as mobile phase. The major compound appeared as a black spot with no fluorescence under the UV light. The desired compounds 4b/c were collected as yellow solids. Before analysis, compounds were recrystallized from methanol.
3.2.1. (1R,4S)-4-(1H-Indol-3-ylmethyl)-1-isopropyl-2H-pyrazino[2,1-b]quinazoline-3,6-(1H,4H)-dione (4b)
Yield: 14%; m.p.: 168–169 °C; [α]D = +65.21 (c 0.46; CHCl3); IR vmax (KBr) 3411, 3066, 1684, 1596, 1471, 1389, 1293 cm−1; 1H NMR see Table 2; 13C NMR see Table 3; m/z (rel. intensity, %): 385.9 (M+, 2), 257.1 (29), 214.1 (3), 202.0 (7), 171.1 (10), 143.1 (4), 130.1 (100), 103.0 (18), 77.0 (16); (+)-HRESIMS m/z 387.1810 (M + H)+ (calculated for C23H22N4O2, 387.1776).
3.2.2. (1S,4R)-4-(1H-Indol-3-ylmethyl)-1-isopropyl-2H-pyrazino[2,1-b]quinazoline-3,6-(1H,4H)-dione (4c)
Yield: 8%; m.p.: 168–169 °C; [α]D = −248.1 (c 0.43; CHCl3); IR vmax (KBr) 3346, 3066, 1683, 1596, 1471, 1389, 1293 cm−1; 1H NMR see Table 2; 13C NMR see Table 3; m/z (rel. intensity, %): 385.9 (M+, 5), 257.1 (32), 298.9 (37), 284.2 (6), 254.0 (15), 238.8 (12), 201.8 (18), 189.0 (35), 171.1 (25), 149.1 (32), 130.1 (100), 103.0 (9), 77.0 (13); (+)-HRESIMS m/z 387.1809 (M + H)+ (calculated for C23H22N4O2, 387.1776).
3.3. General Condition for the Synthesis of 4-(1H-Indol-3-ylmethyl)-1-isobutyl-2H-pyrazino[2,1-b]quinazoline-3,6-(1H,4H)-diones (5b/5c)
In a closed vial, anthranilic acid (i) (28 mg, 200 µmol), N-Fmoc-l-leucine (iic) for 5c or N-Fmoc-d-leucine (iid) for 5b (70.68 mg, 200 µmol), and triphenylphosphite (63 µL, 220 µmol) were added along with 1 mL of dried pyridine. The vial was heated in heating block at 55 °C for 16–24 h. After cooling the mixture to room temperature, we added l-tryptophan methyl ester hydrochloride (iiia) for 5b (51 mg, 200 µmol), or d-tryptophan methyl ester hydrochloride (iiib) 5d (51 mg, 200 µmol), and the mixture was irradiated in the microwave at a constant temperature at 220 °C for 1.5 min. Four reaction mixtures were prepared in the same conditions and treated in parallel. After removing the solvent with toluene, the mixture was purified by flash column chromatography using hexane: EtOAc (60:40) as a mobile phase. The preparative TLC was performed using CH2Cl2:Me2CO (95:5) as a mobile phase. The major compound appeared as a black spot with no fluorescence under UV light. The desired compounds, 5b/5c, were collected as yellow solids. Before analysis, compounds were recrystallized from methanol.
3.3.1. (1R,4S)-4-(1H-Indol-3-ylmethyl)-1-isobutyl-2H-pyrazino[2,1-b]quinazoline-3,6-(1H,4H)-dione (5b)
3.3.2. (1S,4R)-4-(1H-Indol-3-ylmethyl)-1-isobutyl-2H-pyrazino[2,1-b]quinazoline-3,6-(1H,4H)-dione (5c)
3.4. General Condition for the Synthesis of Compounds 4a and 5a
3.4.1. Synthesis of N-(2-Aminobenzoyl)-l-tryptophan methyl ester (iv-a)
To a mixture of anthranilic acid (287 mg, 2.39 mmol) and TBTU (920 mg, 2.86 mmol, 1.2 equiv) in acetonitrile (20 mL) was added Et3N (833 µL, 4.78 mmol, 2 equiv) and l-tryptophan methyl ester (521 mg, 2.39 mmol) at room temperature. After stirring for 5 h, the reaction mixture was concentrated under reduced pressure. The residue was dissolved in CH2Cl2 and washed with 1 M HCl, extracted with CH2Cl2 (3 × 100 mL), dried with Na2SO4, filtered, and concentrated. The residue was purified by flash chromatography (eluent 1% MeOH in CH2Cl2) to yield iv-a as a white solid (675.5 mg, 96%), m.p. 134–135 °C, IR vmax (KBr): 3424, 1746, 1727, 1645, 1611, 1581; 1H NMR (300, MHz, CDCl3): 8.15 (s, 1H), 7.56 (d, 1H, J = 7.9 Hz), 7.35 (d, 1H, J = 8.1 Hz), 7.19–7.15 (m, 3H), 7.09 (t, 1H, J = 7.4 Hz), 7.01 (d, 1H, J = 2.3 Hz), 6.65 (dd, 1H, J = 8,7 and 1.1 Hz), 6.56 (ddd, 1H, J = 7.5 and 0.8 Hz), 5.08 (dt, 1H, J = 7.5 and 5.3 Hz), 3.71 (s, 3H), 3.43 (m, 2H); 13C NMR (75, MHz, CDCl3) 172.6 (CO), 168.8 (CO), 148.8 (C), 136.1 (C), 132.6 (CH), 127.6 (CH), 127.6 (C), 122.8 (CH), 122.3 (CH), 119.8 (CH), 118.7 (CH), 117.3 (CH), 116.7 (CH), 115.3 (C), 111.3 (CH), 110.1 (C), 53.1 (CH), 52.7 (CH3), 27.7 (CH2).
3.4.2. Synthesis of N-[9H-Fluoren-9-ylmethoxy)carbonyl]-l-valinyl-2-aminobenzoyl-l-tryptophan methyl ester (vi-a)
To a solution of iv-a (160 mg, 0.474 mmol) in dried CH2Cl2 (10 mL) was added N-Fmoc-l-valine-Cl [27] (v-a,182 mg, 0.5 mmol). The mixture was stirred for 30 min, followed by addition of aqueous Na2CO3 (1 M, 8 mL, 8 mmol). After continuous stirring for 3 h, the mixture was extracted with CH2Cl2 (4 × 100 mL), dried with Na2SO4, filtered, and concentrated. The residue was purified by flash chromatography (eluent: 5% MeOH in CH2Cl2 to give vi-a as a white solid (296.4 mg, 95%), m.p.: 194.4–196.3 °C, = +13.22 (c 0.126, CHCl3), IR vmax (KBr) 3318, 1727, 1698, 1588 cm−1; 1H NMR (300, MHz, CDCl3): 11.4 (s, 1H), 8.50 (d, 1H, J = 8.3 Hz), 8.24 (s, 1H), 7.67 (d, 2H, J = 7.4 Hz), 7.57 (d, 1H, J = 7.4 Hz), 7.51 (d, J = 7.3), 7.43–7.19 (m, 8H), 7.08 (t, 1H, J = 7.4 Hz), 6.97 (t, 1H, J = 7.4 Hz), 6.91 (d, 1H, J = 7.5 Hz) 6.87 (s, 1H), 6.68 (d, 1H, J = 7.6 Hz), 5.50 (d, 1H, J = 8.5 Hz), 4.97 (dd, 1H, J = 12.7 and 5.2 Hz), 4.32 (dt, 3H, J = 17.5 and 10.4 Hz), 4.20 (dt, 1H, J = 13.6 and 6.1 Hz), 3.72 (s, 3H), 3.38 (dd, 1H, J = 15.0 and 5.2 Hz), 3.31 (dd, 1H J = 14.9 and 5.3 Hz), 1.82 (m, 1H), 0.97 (d, 1H, J = 6.7 Hz), 0.90 (d, 6H, J = 6.8 Hz) 13C NMR (75, MHz, CDCl3) 172.1 (CO), 170.3 (CO), 168.3 (CO), 156.6 (C), 144.1 (2C), 141.3 (2C), 138.9 (C), 136.1 (C), 132.8 (CH), 127.7 (2CH), 127.5 (C), 127.1 (2CH), 127.1 (2CH), 127.0 (CH), 125.3 (CH), 125.2 (CH), 123.2 (CH), 122.8 (CH), 122.3 (CH), 121.4 (C), 120.0 (2CH), 119.8 (CH), 118.4 (C), 111.4 (CH), 109.6 (C), 67.3 (CH2), 61.4 (CH), 53.4 (CH), 52.6 (CH3), 47.3 (CH), 31.3 (CH), 27.3 (CH2), 19.4 (CH3), 17.6 (CH3).
3.4.3. Synthesis of (1S,4S)-4-(1H-Indol-3-ylmethyl)-1-isopropyl-2H-pyrazino[2,1-b]quinazolin-3,6-(1H, 4H)-dione (4a)
To a solution of vi-a (290 mg, 0.440 mmol) in dried CH2Cl2 (20 mL) was added Ph3P (576 mg, 2.2 mmol, 5 equiv), I2 (448 mg, 2.16 mmol. 4.9 equiv), and N,N-diisopropylethylamine (774 µL, 4.44 mmol, 10 equiv). The reaction mixture was stirred at room temperature for 5 h, quenched with aqueous Na2CO3, and extracted with CH2Cl2 (3 × 100 mL), dried with Na2SO4, filtered and concentrated. Hexane was added to remove an excess of Ph3P, the precipitate was filtered and was treated with CH2Cl2 (10 mL) and piperidine (2.5 mL, 20%) at room temperature for 20 min, followed by solvent evaporation to provide the solid which was triturated with hexane (1 × 200 mL), CH2Cl2/PhMe (1 × 200 mL), and hexane (1 × 200 mL). The vacuum-dried crude residue was dissolved in CH3CN (10 mL) in the presence of DMAP (64 mg, 0.53 mmol) and refluxed for 19 h. The reaction mixture was purified by preparative TLC (EtOAc: MeOH: CH2Cl2, 50:2.5:47.5) to afford 4a (45.5 g, 28%); m.p.: 112.8–114.1 °C, = +300.55 (c 0.061, CHCl3), IR vmax (KBr) 3417, 3068, 1683, 1594, 1471, 1387, 1333 cm−1; 1H NMR see Table 2; 13C NMR see Table 3; m/z (rel. intensity, %): 386.3 (M+, 8), 341.0 (5), 315 (10), 282.0 (8), 257.1 (55), 241.9 (7), 217.1 (17), 186.1 (10), 171.0 (18), 130.1 (100), 103.0 (14), 77.0 (16); (+)-HRESIMS m/z 387.1810 (M + H)+ (calculated for C23H22N4O2, 387.1776).
3.4.4. Synthesis of N-[9H-Fluoren-9-ylmethoxy)carbonyl]-l-methylpentanyl-2H-aminobenzoyl-l-tryptophan methyl ester (vi-b)
To a solution of compound iv-a (129 mg, 0.382 mmol) in dried CH2Cl2 (10 mL) was added N-Fmoc-l-leucine-Cl [27] (v-b, 171 mg, 0.458 mmol). The mixture was stirred for 30 min, followed by addition of aqueous Na2CO3 (1 M, 7.6 mL, 7.6 mmol). After being stirred for a total 3 h, the mixture was extracted with CH2Cl2 (4 × 100 mL), dried with Na2SO4, filtered, and concentrated. The residue was purified by flash chromatography (eluent 5% MeOH in CH2Cl2) to give vi-b as a white solid (231.7 mg, 92%), m.p.: 193.7–194.9 °C, = +18.42 (c 0.398, CHCl3), IR vmax (KBr) 3405, 1744, 1695, 1586 cm−1; 1H NMR (300, MHz, CDCl3): 11.4 (s, 1H), 8.59 (d, 1H, J 8.3 Hz), 8.19 (s, 1H), 7.76 (d, 2H, J 7.4 Hz), 7.65 (d, 1H, J 7.3 Hz), 7.59 (d, 1H, J 7.4 Hz), 7.52-7.28 (m, 8H), 7.17 (t, 1H, J 7.3 Hz), 7.06 (d, 1H, J 7.4 Hz), 7.00 (d, 1H, 7.7 Hz) 6.96 (s, 1H), 6.73 (d, 1H, J 7.6 Hz), 5.55 (d, 1H, J 8.5 Hz), 5.05 (dd, 1H, J 12.6 and 5.2 Hz), 4.39 (d, 2H, J 6.6 Hz), 4.28 (m, 2H), 3.72 (s, 3H), 3.38 (m, 2H), 2.33 (dt, 1H J 13.0 and 6.4 Hz), 1.06 (d, 3H, J 6.8 Hz), 0.98 (d, 3H, J 6.8 Hz); 13C NMR (75, MHz, CDCl3): 172.1 (CO), 170.2 (CO), 168.3 (CO), 156.5 (CO), 143.8 (2C), 141.3 (2C), 139.0 (C), 136.1 (C), 132.9 (CH), 127.7 (2CH), 127.5 (C), 127.1 (2CH), 127.1 (2CH), 126.9 (CH), 125.3 (CH), 125.2 (CH), 123.2 (CH), 122.8 (CH), 122.4 (CH), 121.4 (C), 120.2 (2CH), 119.8 (CH), 118.5 (C), 111.4 (CH), 109.7 (CH), 67.2 (CH2), 61.3 (CH), 53.3 (CH), 52.6 (CH3), 38.6 (CH), 47.3 (CH2), 31.4 (CH), 27.3 (CH2), 19.4 (CH3), 17.5 (CH3).
3.4.5. Synthesis of (1S,4S)-4-(1H-Indol-3-ylmethyl)-1-isobutyl-2H-pyrazino[2,1-b]quinazolin-3,6-(1H,4H)-dione (5a)
To a solution of vi-b (232 mg, 0.344 mmol) in dried CH2Cl2 (20 mL) was added Ph3P (451 mg, 1.72 mmol, 5 equiv), I2 (428 mg, 1.68 mmol. 4.9 equiv), and N,N-diisopropylethylamine (605 µL, 3.47 mmol, 10 equiv). The reaction mixture was stirred at room temperature for 5 h, quenched with aqueous Na2CO3, and extracted with CH2Cl2 (3 × 100 mL), dried with Na2SO4, filtered and concentrated. Hexane was added to remove an excess of Ph3P, the precipitate was filtered and was treated with CH2Cl2 (10 mL) and piperidine (2.5 mL, 20%) at room temperature for 20 min, followed by solvent evaporation to provide the solid which was triturated with hexane (1 × 200 mL), CH2Cl2/PhMe (1 × 200 mL), and hexane (1 × 200 mL). The vacuum-dried crude residue was dissolved in CH3CN (10 mL) in the presence of DMAP (80 mg, 0.66 mmol) and refluxed for 19 h. The reaction mixture was purified by preparative TLC (EtOAc:MeOH:CH2Cl2, 50:2.5:47.5) to afford 5a (39 mg, 28%); m.p.: 105.9–106.3 °C, = +81.76 (c 0.106, CHCl3), IR vmax (KBr) 3435, 3060, 1686, 1602, 1387, 1292 cm−1; 1H NMR see Table 2; 13C NMR see Table 3; (+)-HRESIMS m/z 401.1933 (M + H)+ (calculated for C24H24N4O2, 401.1933).
3.5. General Condition for the Synthesis Compound 4d and 5d
3.5.1. Synthesis of N-(2-Aminobenzoyl)-d-tryptophan methyl ester (iv-b)
To a mixture of anthranilic acid (287 mg, 2.39 mmol) and TBTU (920 mg, 2.86mmol, 1.2 equiv) in CH3CN (20 mL) was added Et3N (833 µL, 4.78 mmol, 2 equiv) and D-tryptophan methyl ester (521 mg, 2.39 mmol) at room temperature with stirring. After being stirred for 5 h, the reaction mixture was concentrated under reduced pressure. The residue was dissolved in CH2Cl2 and washed with 1M HCl, extracted with CH2Cl2 (3 × 100 mL), dried with Na2SO4, filtered and concentrated. The residue was purified by flash chromatography (eluent: 1% MeOH in CH2Cl2) to yield iv-b as a white solid (569.7 mg, 81%), m.p. 131.9–134.3 °C, IR vmax (KBr) 3423, 1746, 1644 cm−1, 1H NMR (300, MHz, CDCl3): 8.14 (s, 1H), 7.56 (d, 1H, J = 7.9 Hz), 7.35 (d, 1H, J = 8.1 Hz), 7.19–7.15 (m, 3H), 7.10 (ddd, 1H, J = 8.0, 7.1, and 1.1 Hz), 7.01 (d, 1H, J = 2.4 Hz), 6.65 (dd, 1H, J = 8,7 and 1.1), 6.60 (s, 1H), 6.59–6.52 (ddd, 2H, J = 7.5 and 0.8 Hz), 5.08 (dt, 1H, J = 7.5 and 5.3 Hz), 3.72 (s, 3H), 3.43 (m, 2H); 13C NMR (75, MHz, CDCl3): 172.6 (CO), 168.8 (CO), 148.8 (C), 136.1 (C), 132.6 (CH), 127.6 (CH), 127.5 (C), 122.8 (CH), 122.3 (CH), 119.8 (CH), 118.7 (CH), 117.3 (CH), 116.7 (CH), 115.3 (C), 111.3 (CH), 110.1 (C), 53.1 (CH), 52.5 (CH3), 27.7 (CH2).
3.5.2. Synthesis of N-[9H-Fluoren-9-ylmethoxy)carbonyl]-d-valinyl-2-aminobenzoyl-d-tryptophan methyl ester (vi-c)
To a solution of iv-b (140 mg, 0.416 mmol) in dried CH2Cl2 (10 mL) was added N-Fmoc-d-valine-Cl [27] (v-c,182 mg, 0.5 mmol). The mixture was stirred for 30 min, followed by addition of aqueous Na2CO3 (1 M, 8 mL, 8 mmol). After continuous stirring for 3 h, the mixture was extracted with CH2Cl2 (4 × 100 mL), dried with Na2SO4, filtered, and concentrated. The residue was purified by flash chromatography (eluent: 5% MeOH in CH2Cl2 to give vi-c as a white solid (220.4 mg, 84%), m.p.: 197.8–200.2 °C, = −22.72 (c 0.088, CHCl3), IR vmax (KBr) 3423, 1724, 1670, 1589 cm−1; 1H NMR (300, MHz, CDCl3): 11.42 (s, 1H), 8.59 (d, 1H, J = 8.3 Hz), 8.18 (s, 1H), 7.76 (d, 2H, J = 7.4 Hz), 7.66 (d, 1H, J = 7.3 Hz), 7.60 (d, 1H, J = 7.3 Hz); 7.53–7.28 (m, 9H), 7.17 (t, 1H, J = 7.3 Hz), 7.08 (d, 1H, J = 7.4 Hz), 7.02 (m, 1H), 6.96 (s, 1H), 6.72 (d, 1H, J = 7.6 Hz), 5.55 (d, 1H, J = 8.5 Hz), 5.06 (dd, 1H, J = 12.5 and 5.2 Hz), 4.40 (d, 1H, J = 6.6 Hz), 4.32–4.24 (m, 1H), 3.73 (s, 3H), 3.45–3.31 (m, 2H), 1.65 (s, 3H) 2.40–2.31 (m, 1H), 1.06 (d, 3H, J = 6.8 Hz), 0.98 (d, 3H, J = 6.9 Hz); 13C NMR (75, MHz, CDCl3) 172.0 (CO), 170.2 (CO), 168.3 (CO), 156.5 (C), 144.1 (2C), 141.3 (2C), 139.0 (C), 136.1 (C), 132.9 (CH), 127.7 (2CH), 127.5 (C), 127.1 (2CH), 127.1 (2CH), 126.9 (CH), 125.3 (CH), 125.2 (CH), 123.2 (CH), 122.8 (CH), 122.4 (CH), 121.4 (C), 120.2 (2CH), 119.8 (CH), 118.5 (C), 111.4 (CH), 109.7 (C), 67.2 (CH2), 61.3 (CH), 53.3 (CH), 52.6 (CH3), 47.3 (CH), 31.3 (CH), 27.3 (CH2), 19.4 (CH3), 17.6 (CH3).
3.5.3. Synthesis of (1R,4R)-4-(1H-Indol-3-ylmethyl)-1-isopropyl-2H-pyrazino[2,1-b]quinazolin-3,6-(1H,4H)-dione (4d)
To a solution of vi-c (183 mg, 0.278 mmol) in dried CH2Cl2 (20 mL) was added Ph3P (365 mg, 1.4 mmol, 5 equiv), I2 (345 mg, 1.36 mmol, 4.9 equiv), and N,N-diisopropylethylamine (489 µL, 2.81 mmol, 10 equiv). The reaction mixture was stirred at room temperature for 5 h, quenched with aqueous Na2CO3, and extracted with CH2Cl2 (3 × 100 mL), dried with Na2SO4, filtered, and concentrated. Hexane was added to remove an excess of Ph3P, the precipitate was filtered and treated with CH2Cl2 (10 mL) and piperidine (2.5 mL, 20%) at room temperature for 20 min, followed by solvent evaporation to provide the solid which was triturated with hexane (1 × 200mL), CH2Cl2/PhMe (1 × 200 mL), and hexane (1 × 200 mL). The vacuum-dried crude residue was dissolved in CH3CN (10 mL in the presence of DMAP (64 mg, 0.53 mmol) and refluxed for 19 h. The reaction mixture was purified by preparative TLC (EtOAc:MeOH:CH2Cl2, 50:2.5:47.5) to afford 4d (22.4 mg, 21%), m.p.: 111.9–113.0 °C, = −210.53 (c 0.114, MeOH), IR vmax (KBr) 3321, 3068, 1683, 1593, 1471, 1387, 1291 cm−1; 1H NMR see Table 2; 13C NMR see Table 3; m/z (rel. intensity, %): 385.9 (M+, 12), 257.1 (29), 214.1 (3), 202.0 (7), 171.1 (10), 143.1 (4), 130.1 (100), 103.0 (18), 77 (16); (+)-HRESIMS m/z 389.1776 (M + H)+ (calculated for C23H22N4O2, 387.1809).
3.5.4. Synthesis of N-[9H-Fluoren-9-ylmethoxy)carbonyl]-d-methylpentyl-2-aminobenzoyl-d-tryptophan methyl ester (vi-d)
To a solution of iv-b (130 mg, 0.386 mmol) in dried CH2Cl2 (10 mL) was added N-Fmoc-d-leucine-Cl [27] (v-d,172.5 mg, 0.464 mmol). The mixture was stirred for 30 min, followed by addition of aqueous Na2CO3 (1 M, 7.7 mL, 7.7 mmol). After continuous stirring for 3 h, the mixture was extracted with CH2Cl2 (4×), dried with Na2SO4, filtered, and concentrated. The residue was purified by flash chromatography (eluent: 5% MeOH in CH2Cl2) to give vi-d as a white solid (251 mg, 98%), m.p.: 194.9–196.3 °C, = −31.75 (c 0.105, CHCl3), IR vmax (KBr) 1740, 1645, 1584 cm−1; 1H NMR (300, MHz, CDCl3): 11.5 (s, 1H), 8.58 (d, 1H, J = 8.3 Hz), 8.23 (s, 1H), 7.76 (d, 2H, J = 7.4 Hz), 7.66 (d, 1H, J = 7.4 Hz), 7.57 (d, 1H, J = 7.4 Hz), 7.51–7.26 (m, 8H), 7.16 (t, 1H, J = 7.4 Hz), 7.07 (d, 1H, J = 7.4 Hz), 7.00 (t, 1H, J = 7.5 Hz) 6.93 (s, 1H), 6.74 (d, 1H, J = 7,6 Hz), 5.43 (d, 1H, J = 7.9 Hz), 5.04 (dd, 1H, J = 12.7 and 5.2 Hz), 4.41 (dt, 3H, J = 17.5 and 10.4 Hz), 4.25 (t, 1H, J = 7.0 Hz), 3.72 (s, 3H), 3.38 (dd, 1H, J = 15.0 and 5.2 Hz), 3.31 (dd, 1H J = 14.9 and 5.3 Hz), 1.82 (m, 1H), 1.00 (d, 6H, J = 6.2 Hz); 13C NMR (75 MHz, CDCl3): 172.1 (CO), 170.2 (CO), 168.3 (CO), 156.4 (CO), 143.8 (2C), 141.3 (2C), 139.0 (C), 136.1 (C), 132.9 (CH), 127.7 (2CH), 127.5 (C), 127.1 (2CH), 127.1 (2CH), 126.9 (CH), 125.3 (CH), 125.2 (CH), 123.2 (CH), 122.8 (CH), 122.4 (CH), 121.4 (C), 120.2 (2CH), 119.8 (CH), 118.5 (C), 111.4 (CH), 109.7 (CH), 67.2 (CH2), 61.3 (CH), 53.3 (CH), 52.6 (CH3), 47.3 (CH2), 38.6 (CH), 31.4 (CH), 27.3 (CH2), 19.4 (CH3), 17.5 (CH3).
3.5.5. Synthesis of (1R,4R)-4-(1H-Indol-3-ylmethyl)-1-isobutyl-2H-pyrazino[2,1-b]quinazolin-3,6-(1H,4H)-dione (5d)
To a solution of vi-d (251 mg, 0.373 mmol) in dried CH2Cl2 (20 mL) was added Ph3P (489 mg, 1.9 mmol, 5 equiv), I2 (464 mg, 1.83 mmol, 4.9 equiv), and N,N-diisopropylethylamine (656 µL, 3.77 mmol, 10 equiv). The reaction mixture was stirred at room temperature for 5 h, quenched with aqueous Na2CO3, and extracted with CH2Cl2 (3 × 100 mL), dried with Na2SO4, filtered, and concentrated. Hexane was added to remove an excess of Ph3P, the precipitate was filtered and was treated with CH2Cl2 (10 mL) and piperidine (2.5 mL, 20%) at room temperature for 20 min, followed by solvent evaporation to provide the solid which was triturated with hexane (1 × 200 mL), CH2Cl2/PhMe (1 × 200 mL), and hexane (1 × 200 mL). The vacuum-dried crude residue was dissolved in CH3CN (10 mL in the presence of DMAP (84 mg, 0.82 mmol) and refluxed for 19 h. The reaction mixture was purified by preparative TLC (EtOAc:MeOH:CH2Cl2, 50:2.5:47.5) to afford 5d (61.6 mg, 40%), m.p.: 103.2–105.6 °C, = −186.04 (c 0.086, CHCl3), IR vmax (KBr) 3333, 3061, 1687, 1603, 1296 cm−1; 1H NMR see Table 2; 13C NMR see Table 3; (+)-HRESIMS m/z 401.1966 (M + H)+ (calculated for C24H24N4O2, 401.1933).
3.6. Screening Test for Antitumor and Anti-P-Glycoprotein Activity
Compounds 4a–d and 5a–d were reconstituted in sterile DMSO to the final concentration of 60 mM, and several aliquots were made and stored at −20 °C to avoid repeated freeze-thaw cycles. For experiments, the compounds were freshly diluted in medium to the desired concentration. Screening for tumor cell growth inhibition was carried out in two human tumor cell lines (NCI-H460 and HCT-15), with the sulforhodamine B (SRB) assay, as previously described [30]. Briefly, tumor cells were plated in 96-well plates, incubated at 37 °C for 24 h, and then treated for 48 h with 5 serial dilutions (1:2) of each compound (ranging from 150 μM to 9.375 μM). The effect of the vehicle solvent (DMSO) was also analyzed as a control. Cells were fixed with 10% ice-cold trichloroacetic acid, washed with water and stained with SRB. Finally, the plates were washed with 1% acetic acid and the bound SRB was solubilized with 10 mM Tris Base. Absorbance was measured in a microplate reader (Synergy Mx, Biotek Instruments Inc., Winooski, VT, USA) at 510 nm. For each compound, the corresponding GI50 (concentration which inhibited 50% of net cell growth) was determined, as previously described [33]. For the screening of compounds for drug-efflux inhibitory activity, the flow cytometry determination of rhodamine-123 cellular accumulation was carried out as previously described [34]. Briefly, K562 and K562Dox cells were incubated for 1 h at 37 °C with 20 μM of the compounds, and 1 μM of rhodamine-123 (Rh123, from Sigma, USA). Verapamil was used as a positive control. Cells were then washed, resuspended in ice cold PBS, and analyzed in a BD Accuri™ C6 Flow Cytometer (BD Biosciences, San Jose, CA, USA). Data were analyzed using the FlowJo software (version 7.6.1, Tree Star, Inc.). The ratio of Rh123 accumulation in the cells was then calculated as MFIK562Dox+Compound -MFIK562Dox)/MFIK562Dox [30].
4. Conclusions
Inspired by the marine-derived fiscalin B (4c), quinazolinone alkaloid derivatives were synthesized using two different methodologies: a highly efficient and straightforward three-component one-pot microwave-assisted approach and also a multistep Mazurkiewicz–Ganesan approach. While the former proved to be efficient and practical for broad screening libraries of the compounds, the latter, although with a more intricate methodology, proved to be a good approach for the synthesis of the syn enantiomers. Moreover, we have found that partial epimerization under the reaction conditions could occur. In vitro growth inhibitory activity of two tumor cell lines revealed that among this series of synthesized compounds, six new analogues were found to exhibit tumor cell growth inhibitory activity. Consequently, this marine-inspired synthesis can bring new insights into discovery of new lead compounds in the oncology area.
Supplementary Materials
The following are available online at http://www.mdpi.com/1660-3397/16/8/261/s1.
Author Contributions
E.S. and A.K. conceived the study design. S.L. synthesized the compounds and elucidated their structure and, A.M.S.S., E.S., and M.M.M.P. analysed the data. D.I.S.P.R. performed the HPLC analysis. A.P. and T.F.-M. performed the cytotoxicity and anti-Pgp assays, and T.F.-M. and M.H.V. analyzed the data and discussed results. S.L. and E.S. wrote the manuscript, while all authors gave significant contributions in discussion and revision. All authors agreed to the final version of the manuscript.
Funding
This research was partially supported by the Strategic Funding UID/Multi/04423/2013 through national funds provided by FCT—Foundation for Science and Technology and European Regional Development Fund (ERDF), in the framework of the program PT2020. The authors thank to national funds provided by FCT—Foundation for Science and Technology and European Regional Development Fund (ERDF) and COMPETE under the Strategic Funding UID/Multi/04423/2013, the projects POCI-01-0145-FEDER-028736, PTDC/MAR-BIO/4694/2014 (POCI-01-0145-FEDER-016790; 3599-PPCDT), and INNOVMAR—Innovation and Sustainability in the Management and Exploitation of Marine Resources, reference NORTE-01-0145-FEDER-000035, Research Line NOVELMAR. The work was also funded by FEDER—Fundo Europeu de Desenvolvimento Regional através do COMPETE 2020—Programa Operacional para a Competitividade e Internacionalização (POCI), Portugal 2020, and by Portuguese fundings through FCT—Fundação para a Ciência e a Tecnologia/Ministério da Ciência, Tecnologia e Inovação, no âmbito do projeto “Instituto de Investigação e Inovação em Ciências da Saúde “(POCI—01-0145-FEDER—007274)”.
Acknowledgments
S.L. thanks Erasmus Mundus Action 2 (LOTUS+, LP15DF0205) for full PhD scholarship. To Sara Cravo for technical support. To Centro de Apoio Cientifico e Tecnolόxico á Investigation (CACTI, University of Vigo, Pontevedra, Spain) for HRMS analysis.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Montaser, R.; Luesch, H. Marine natural products: A new wave of drugs? Future Med. Chem. 2011, 3, 1475–1489. [Google Scholar] [CrossRef] [PubMed]
- Panda, S.; Tripathy, U.P. Quinazolone: A molecule of significant pharmacological and biological activity. Res. J. Pharm. Technol. 2013, 6, 849–855. [Google Scholar]
- Eguchi, S. Quinazoline alkaloids and related chemistry. In Bioactive Heterocycles i; Eguchi, S., Ed.; Springer: Berlin/Heidelberg, Germany, 2006; pp. 113–156. [Google Scholar]
- Peng, J.; Lin, T.; Wang, W.; Xin, Z.; Zhu, T.; Gu, Q.; Li, D. Antiviral alkaloids produced by the mangrove-derived fungus cladosporium sp. Pjx-41. J. Nat. Prod. 2013, 76, 1133–1140. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Debbab, A.; Mándi, A.; Wray, V.; Schulz, B.; Müller, W.E.G.; Kassack, M.; Lin, W.; Kurtán, T.; Proksch, P.; et al. Alkaloids from the sponge-associated fungus Aspergillus sp. Eur. J. Org. Chem. 2013, 2013, 894–906. [Google Scholar] [CrossRef]
- He, F.; Han, Z.; Peng, J.; Qian, P.Y.; Qi, S.H. Antifouling indole alkaloids from two marine derived fungi. Nat. Prod. Commun. 2013, 8, 329–332. [Google Scholar] [PubMed]
- Rodrigues, B.S.F.; Sahm, B.D.B.; Jimenez, P.C.; Pinto, F.C.L.; Mafezoli, J.; Mattos, M.C.; Rodrigues-Filho, E.; Pfenning, L.H.; Abreu, L.M.; Costa-Lotufo, L.V.; et al. Bioprospection of cytotoxic compounds in fungal strains recovered from sediments of the brazilian coast. Chem. Biodivers. 2015, 12, 432–442. [Google Scholar] [CrossRef] [PubMed]
- Afiyatullov, S.S.; Zhuravleva, O.I.; Antonov, A.S.; Kalinovsky, A.I.; Pivkin, M.V.; Menchinskaya, E.S.; Aminin, D.L. New metabolites from the marine-derived fungus Aspergillus fumigatus. Nat. Prod. Commun. 2012, 7, 497–500. [Google Scholar] [PubMed]
- Shao, C.L.; Xu, R.F.; Wei, M.Y.; She, Z.G.; Wang, C.Y. Structure and absolute configuration of fumiquinazoline l, an alkaloid from a gorgonian-derived Scopulariopsis sp. Fungus. J. Nat. Prod. 2013, 76, 779–782. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Li, X.M.; Meng, L.H.; Wang, B.G. N-formyllapatin A, a new N-formylspiroquinazoline derivative from the marine-derived fungus Penicillium adametzioides As-53. Phytochem. Lett. 2014, 10, 145–148. [Google Scholar] [CrossRef]
- Zin, W.W.M.; Prompanya, C.; Buttachon, S.; Kijjoa, A. Bioactive secondary metabolites from a Thai collection of soil and marine-derived fungi of the genera Neosartorya and Aspergillus. Curr. Drug Deliv. 2016, 13, 378–388. [Google Scholar] [PubMed]
- Prata-Sena, M.; Ramos, A.A.; Buttachon, S.; Castro-Carvalho, B.; Marques, P.; Dethoup, T.; Kijjoa, A.; Rocha, E. Cytotoxic activity of secondary metabolites from marine-derived fungus Neosartorya siamensis in human cancer cells. Phytother. Res. 2016, 30, 1862–1871. [Google Scholar] [CrossRef] [PubMed]
- Liao, L.; You, M.; Chung, B.K.; Oh, D.-C.; Oh, K.-B.; Shin, J. Alkaloidal metabolites from a marine-derived Aspergillus sp. Fungus. J. Nat. Prod. 2015, 78, 349–354. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Wei, X.; Kim, E.L.; Lin, X.; Yang, X.W.; Zhou, X.; Yang, B.; Jung, J.H.; Liu, Y. Erratum: Fumigatosides A–D, four new glucosidated pyrazinoquinazoline indole alkaloids from a jellyfish-derived fungus Aspergillus fumigatus (org. Lett. (2014)). Org. Lett. 2014, 16, 2574. [Google Scholar] [CrossRef] [PubMed]
- Martine, D.; Isabelle, B. Survey of recent literature related to the biologically active 4(3H)-quinazolinones containing fused heterocycles. Curr. Med. Chem. 2013, 20, 794–814. [Google Scholar] [CrossRef]
- Penn, J.; Purcell, M.; Mantle, P.G. Biosynthesis of glyantrypine by aspergillus clavatus. FEMS Microbiol. Lett. 1992, 92, 229–233. [Google Scholar] [CrossRef]
- Takahashi, C.; Matsushita, T.; Doi, M.; Minoura, K.; Shingu, T.; Kumeda, Y.; Numata, A. Fumiquinazolines A–G, novel metabolites of a fungus separated from a pseudolabrus marine fish. J. Chem. Soc. Perkin Trans. 1 1995, 2345–2353, 2345–2353. [Google Scholar] [CrossRef]
- Wong, S.M.; Musza, L.L.; Kydd, H.G.C.; Kullnig, R.; Gillum, A.M.; Cooper, R. Fiscalins: New substance P inhibitors produced by the fungus neosartorya fischeri taxonomy, fermentation, structures, and biological properties. J. Antibiot. 1993, 46, 545–553. [Google Scholar] [CrossRef] [PubMed]
- Avendaño, C.; Caballero, E.; Méndez-Vidal, C.; De Quesada, A.R.; Menéndez, J.C. MDR reversal by deprenylated tetracyclic and hexacyclic analogues of N-acetylardeemin: Confirmation of the ardeemin pharmacophore. Lett. Drug Des. Discov. 2006, 3, 369–377. [Google Scholar] [CrossRef]
- Hayashi, D.; Tsukioka, N.; Inoue, Y.; Matsubayashi, Y.; Iizuka, T.; Higuchi, K.; Ikegami, Y.; Kawasaki, T. Synthesis and ABCG2 inhibitory evaluation of 5-N-acetylardeemin derivatives. Bioorg. Med. Chem. 2015, 23, 2010–2023. [Google Scholar] [CrossRef] [PubMed]
- Buttachon, S.; Chandrapatya, A.; Manoch, L.; Silva, A.; Gales, L.; Bruyère, C.; Kiss, R.; Kijjoa, A. Sartorymensin, a new indole alkaloid, and new analogues of tryptoquivaline and fiscalins produced by Neosartorya siamensis (KUFC 6349). Tetrahedron 2012, 68, 3253–3262. [Google Scholar] [CrossRef]
- Bessa, L.J.; Buttachon, S.; Dethoup, T.; Martins, R.; Vasconcelos, V.; Kijjoa, A.; da Costa, P.M. Neofiscalin A and fiscalin C are potential novel indole alkaloid alternatives for the treatment of multidrugresistant gram-positive bacterial infections. FEMS Microbiol. Lett. 2016, 363. [Google Scholar] [CrossRef] [PubMed]
- Cledera, P.; Avendaño, C.; Menéndez, J.C. A new route toward 4-substituted pyrazino[2,1-b]quinazoline-3,6-dione systems. Total synthesis of glyantrypine. J. Org. Chem. 2000, 65, 1743–1749. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Ganesan, A. Total Synthesis of the quinazoline alkaloids (−)-Fumiquinazoline G and (−)-Fiscalin B. J. Org. Chem. 1998, 63, 2432–2433. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Ganesan, A. Total synthesis of the fumiquinazoline alkaloids: Solution-phase studies. J. Org. Chem. 2000, 65, 1022–1030. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.F.; Ye, P.; Zhang, B.; Bi, G.; Sargent, K.; Yu, L.; Yohannes, D.; Baldino, C.M. Three-component one-pot total syntheses of glyantrypine, Fumiquinazoline F, and Fiscalin B promoted by microwave irradiation. J. Org. Chem. 2005, 70, 6339–6345. [Google Scholar] [CrossRef] [PubMed]
- Kantharaju; Patil, B.S.; Suresh Babu, V.V. Synthesis of fmoc-amino acid chlorides assisted by ultrasonication, a rapid approach. Int. J. Pept. Res. Ther. 2002, 9, 227–229. [Google Scholar]
- Hernández, F.; Buenadicha, F.L.; Avendao, C.; Söllhuber, M. 1-alkyl-2,4-dihydro-1h-pyrazino[2,1-b]quinazoline-3,6-diones as glycine templates. Synthesis of Fiscalin B. Tetrahedron Asymmetry 2002, 12, 3387–3398. [Google Scholar] [CrossRef]
- Vichai, V.; Kirtikara, K. Sulforhodamine B colorimetric assay for cytotoxicity screening. Nat. Protoc. 2006, 1, 1112–1116. [Google Scholar] [CrossRef] [PubMed]
- Lopes-Rodrigues, V.; Oliveira, A.; Correia-da-Silva, M.; Pinto, M.; Lima, R.T.; Sousa, E.; Vasconcelos, M.H. A novel curcumin derivative which inhibits P-glycoprotein, arrests cell cycle and induces apoptosis in multidrug resistance cells. Bioorg. Med. Chem. 2017, 25, 581–596. [Google Scholar] [CrossRef] [PubMed]
- Han, X.X.; Xu, X.Y.; Cui, C.B.; Gu, Q.Q. Alkaloidal compounds produced by a marine-derived fungus, aspergillus fumigatus H1-04, and their antitumor activities. J. Chin. Med. Chem. 2007, 4, 232. [Google Scholar]
- Yu, G.; Zhou, G.; Zhu, M.; Wang, W.; Zhu, T.; Gu, Q.; Li, D. Neosartoryadins A and B, fumiquinazoline alkaloids from a mangrove-derived fungus neosartorya udagawae HDN13-313. Org. Lett. 2016, 18, 244–247. [Google Scholar] [CrossRef] [PubMed]
- Kijjoa, A.; Santos, S.; Dethoup, T.; Manoch, L.; Almeida, A.P.; Vasconcelos, M.H.; Silva, A.; Gales, L.; Herz, W. Sartoryglabrins, analogs of ardeemins, from neosartorya glabra. Nat. Prod. Commun. 2011, 6, 807–812. [Google Scholar] [PubMed]
- Palmeira, A.; Vasconcelos, M.H.; Paiva, A.; Fernandes, M.X.; Pinto, M.; Sousa, E. Dual inhibitors of p-glycoprotein and tumor cell growth: (Re)discovering thioxanthones. Biochem. Pharmacol. 2012, 83, 57–68. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).