Emergent Sources of Prebiotics: Seaweeds and Microalgae
Abstract
:1. Introduction
1.1. Definition of Dietary Fiber
1.2. Dietary Fibers in Seaweeds
| Type of Algae | Total Fiber (% DW) | Soluble Fiber (% DW) | Insoluble Fiber (% DW) | References |
|---|---|---|---|---|
| Brown seaweeds | 35–62 1 | 30–38 | 5–32 | [6] |
| Alaria esculenta | 42.86 2 | |||
| Cystoseira abies-marina | 56.34 | [7] | ||
| Eisenia bicyclis | 10–75 2 | |||
| Fucus spiralis | 63.88 | 27–40 1 | [7] | |
| F. vesiculosus a | 50.09 ± 1.77 | [8] | ||
| 45–59 2 | ||||
| Himanthalia elongata a | 32.7 | 25.7 | 7.0 | [9] |
| 33–37 2 | ||||
| Hizikia fusiforme b | 62.3 ± 0.7 | [10] | ||
| Laminaria sp. b | 36.0 ± 5.7 | 27–40 1 | [10] | |
| L. digitata | 37.3 | 32.6 | 4.7 | [9] |
| 36–37 2 | ||||
| Laminaria/Saccharina japonica | 10–41 2 | |||
| Saccahrina latissima | 30 2 | |||
| Sargassum fusiforme | 17–69 2 | |||
| Undaria pinnatifida a, b | 35.3–45.9 | 30.0–33 | 5.3–6 | [9,10,11] |
| 16–51 2 | ||||
| Red seaweeds | ||||
| Chondrus crispus | 10–34 2 | |||
| Gelidium microdon d | 57.37 | [7] | ||
| Gracilaria changii d | 28.0 c | [12] | ||
| Hypnea charoides | 50.3 ± 2.78 | [13] | ||
| H. japonica | 53.2 ± 0.56 | [13] | ||
| Osmundea pinnatifida | 33.82 | [7] | ||
| Palmaria palmate a, d | 29–46 2 | |||
| Porphyra sp. b, d | 48.6 ± 5.90 | 18 | [7,10] | |
| 40.98 | ||||
| 35–49 1 | ||||
| P. tenera d | 34.7 | 17.9 | 16.8 | [9] |
| 12–35 2 | ||||
| P. umbilicalis a, d | 43.0 c | 34 | 9 | [11] |
| 29–35 2 | ||||
| P. yezoensis | 30–59 2 | |||
| Porphyridium sp. | 35.5 (biomass) | 8.5 | 27 | [14] |
| 45 (EPS) | 37 | 8 | ||
| Pterocladia capillacea | 52.08 | [7] | ||
| Sphaerococcus coronopifolius | 41.25 | [7] | ||
| Green seaweeds | ||||
| Caulerpa lentillifera | 38–59 2 | |||
| C. racemosa | 33–41 2 | |||
| Codium reticulata | 39–67 2 | |||
| Enteromorpha spp. a | 33.4 | 17.2 | 16.2 | [7] |
| Ulva sp. | 381 | 21 | 17 | |
| U. compressa a | 41.16 | [7] | ||
| 29–45 2 | ||||
| U. lactuca a | 55.4 ± 2.00 | [9,11,13,15] | ||
| 38.1–43 c | 21.3–24 | 16.8–19 | ||
| 29–55 2 | ||||
| U. pertusa a | 52.1 | [16] | ||
| U. reticulata | 65.7 2 | |||
| U. rigida | 38–41 2 | |||
| Foods | ||||
| Apple | 2 g/100 g | 0.9 3 | [9,17] | |
| 14.2% DW | ||||
| Bean | 3.0 g/100 g 3, e | 0.2 | ||
| Brown rice | 3.8% DW | [18] | ||
| Cabbage | 2.3–2.9 g/100 g e | 0.3 3 | [9,17] | |
| 34.3% DW | ||||
| Lentils | 8.9 g/100 g | [17] | ||
| Rye | 20.5 g/100 g 3 | 4.8 | ||
| Wheat bran | 85% DW | [9] |
1.3. Dietary Fibers—Health Benefits
1.4. Techniques to Obtain Oligosaccharides
1.5. Health Benefits of Algal PS
| Polysaccharide/LMW-PS | Health Benefit | Main Glycosidic Linkages and Monomers along the Main Chain | Algal Genera |
|---|---|---|---|
| sPS | Antilipidaemic/hypocholesterolaemic | Porphyridium (R), Rhodella (R) | |
| s-laminaran | (1,3)- and (1,6)-β-glc | Ascophyllum (B), Fucus (B), Laminaria/Saccharina (B), Undaria (B) | |
| s-fucan | Sargassum (B) | ||
| s-galactofucan | (1,3)- and (1,4)-α-l-fuc (alternating) | Laminaria/Saccharina (B), | |
| s-galactan (porphyran) | (1,3)-β-d-gal or (1,4)-α-l-gal | Porphyra (R), Ulva (G) | |
| s-ulvan | (→4)-β-d-GlcAc-(1,4)-α-l-rham3S-(1→) | Ulva (G), Enteromorpha (G) | |
| (→4)-α-l-IduAc-(1,4)-α-l-rham3S-(1→) | |||
| sPS | Antiglycaemic | Porphyridium (R), Rhodella (R) | |
| (s)PS | Immunomodulatory | Chlorella (G), Gracilaria (R), Gyrodinium (Dino), Phaeodactlylum (Diat), Porphyridium (R), Ulva (G) | |
| s-fucan | (1,3)-α-l-fuc | Cladosiphon (aka Okinawa) (B) | |
| s-fucan | (1,3)- and (1,4)-α-l-fuc (alternating) | Ascophyllum (B), Fucus (B) | |
| s-laminaran | (1,3)- and (1,6)-β-glc | Ascophyllum (B), Fucus (B), Laminaria (B), Undaria (B) | |
| s-galactofucan | (1,3)- and (1,4)-α-l-fuc (alternating) | Laminaria (B), Undaria (B) | |
| s-ulvan | (→4)-β-d-GlcAc-(1,4)-α-l-rham3S-(1→) | Ulva (G), Enteromorpha (G) | |
| (→4)-α-l-IduAc-(1,4)-α-l-rham3S-(1→) | |||
| (s-) rhamnan | Enteromorpha (G), Monostroma (G) | ||
| LMW-sPS | Furcellaria (R), Soliera (R) | ||
| LMW-carrageenan | (1,3)-α-d-gal, and (1,4)-β-3,6-Agal or (1,4)-β-d-gal (alternating) | Kappaphycus (R) | |
| s-mannan | Capsosiphon (G) |
1.6. Algal PS as Dietary Fibers
2. Prebiotics
2.1. Definitions and Criteria
2.2. Benefits of Prebiotics to Human Health
| Effect | Mechanisms | References |
|---|---|---|
| IBD a, b | reduction of pro-inflammatory immune markers and also of calprotectin enhancement of cytokine production reduction of symptoms modulated by bifidobacteria (highest benefits usually when butyrate is used in the experiments) | [21,36,74,82,88,89,90,91,92,93] |
| Ulcerative colitis a, b | ||
| Pouchitis b | ||
| Crohn’s disease b | ||
| IBS a | ||
| Colon cancer, prevention | significant reduction of putrefactive compounds | [74,88,93,94,95] |
| production of butyrate to act as protective agent | ||
| mediation by colonic microbiota, as bifidobacteria may shift down carcinogenic promoters and genotoxins | ||
| reduction on biomarkers for cancer | ||
| reduction of cell proliferation | ||
| Bone mass/density | enhancement of calcium absorption a, b, due to the release of SCFAs | [21,74,88,96,97,98] |
| shift down of gut pH due to the production of SCFAs | ||
| Regulation of gut metabolism/transit | reduction of constipation of diarrhea and dysentery | [74] |
| Antibiotic-associated and traveller | reduction of the prevalence of/prevention from diarrhoea b reduction of the fever and vomiting in children b stimulation of the growth bifidobacteria | [93,99,100,101] |
| diarrhoea | ||
| Improvement of the immune system | production of pro-inflammatory cytokines (TNF-α) | [74,102] |
| expression of receptors on macrophages and lymphocytes T and B are stimulated |
Benefits of Prebiotics Reflected in the Morphology, Ecology and Microbiota of the Gut
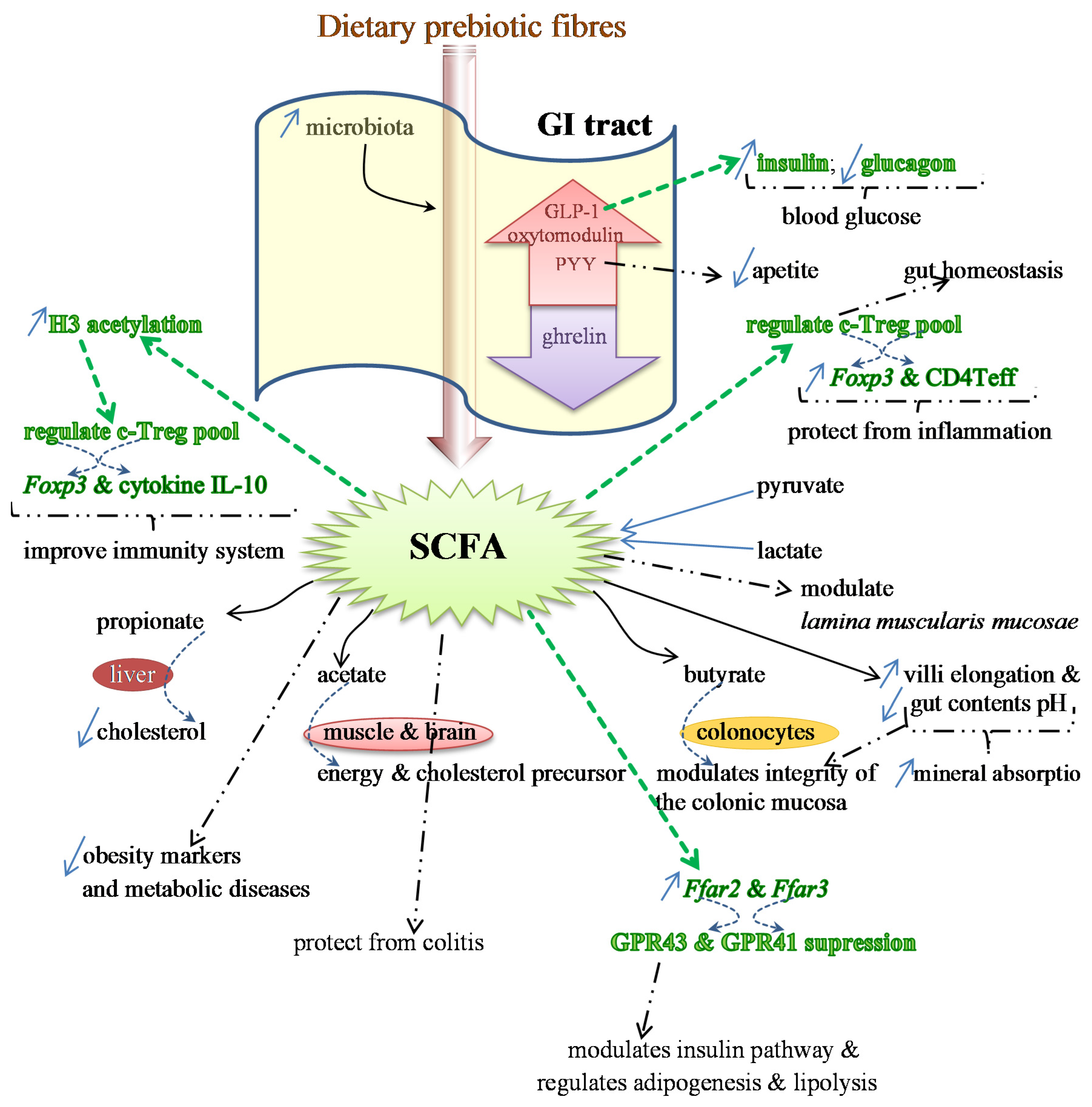
2.3. SCFAs—Molecular Mechanisms and Epigenetic Regulation
3. Seaweeds and Marine Microalgae and Their Active Compounds as Prebiotics
3.1. Brief Description of the PS Considered as Fibers
3.2. Prebiotic Benefits of Algal Biomass and Fibers, Oligo- and Polysaccharides
| Oligo-/PS | Algal Genus | Effects | In Vitro/in Vivo (Animal Model) | References |
|---|---|---|---|---|
| alginate | - | ↑ Bifidobacterium | rats | [61] |
| ↑ Lactobacillus | ||||
| NAOS (native and hydrolysates, DP 4–12) | - | ↑ Bifidobacterium | mice/rats (in vitro) | [158] |
| ↑ Lactobacillus | ||||
| ↓ Bacteroides and enterococci | ||||
| ↓ pH in medium | ||||
| ↓ putrefactive microorganisms | ||||
| laminaran | - | ↑ Bifidobacterium | rats (in vitro) | [165] |
| ↓ putrefactive compounds | ||||
| laminaran + fucoidan | - | ↑ lactobacilli | weanling pigs | [172] |
| ↓ diarrhoea | ||||
| extracts | Undaria/Porphyra | ↓ enzymes responsible for the transformation of pro- into carcinogens | rats | [173] |
| biomass | Ascophyllum | ↑ Lactobacillus/Escherichia coli | weanling pigs | [174] |
| biomass/extracts | Laminaria | ↑ SCFAs | weanling pigs | [175] |
| ↓ ammonia in the colon | ||||
| fucoidan | - | ↑ lactobacteria | pigs | [166,171,176] |
| ↑ fatty acids | ||||
| alginate | - | ↑ beneficial bacteria of microbiota | humans | [60] |
| ALGOS and native or LMW-PSs | Gelidium | ● positive effects on the microbiota and on the production of SCFAs ↓ putrefactive compounds | rats | [61,165,167,168] |
| ↓ putrefactive microorganisms | ||||
| FUCOS | - | ↑ beneficial bacteria | - | [158,168] |
| AGAROS | - | ↓ pro-inflammatory cytokines | - | [177,178] |
| ● act against glycosidase | ||||
| extracts | Gelidium | ↑ bifidobacteria; best with Gelidium-extract | in vitro | [179] |
| Gracilaria | ||||
| Ascophyllum | ||||
| ↑ total SCFAs, and acetic and propionic acids; best with Gelidium-extract | ||||
| biomass | Chondrus | ↑ beneficial bacteria | rats | [127] |
| ● improvement of microbiota | ||||
| ↑ SCFAs | ||||
| ● improvement in the histo-morphology of the colon | ||||
| ↑ holding-water capacity of stool | ||||
| ● enhancement of immune system: ↑ Ig-A and G | ||||
| biomass | Spirulina | ↑ L. casei, L. acidophilus, S. thermophillus and other beneficial bacteria, such as Bifidobacterium | in vitro | [87,180,181] |
| ↓ harmful bacteria: P. vulgaris, B. subtilis, B. pumulis | ||||
| biomass | Isochrysis | ↑ lactic acid bacteria | rats | [182] |
4. Conclusions and Final Remarks
Acknowledgments
Conflicts of Interest
References
- Clemente, J.C.; Ursell, L.K.; Parfrey, L.W.; Knight, R. The impact of the gut microbiota on human health: An integrative view. Cell 2012, 148, 1258–1270. [Google Scholar] [CrossRef] [PubMed]
- Institute of Medicine: Food and Nutrition Board. Dietary Reference Intakes: Energy, Carbohydrates, Fiber, Fat, Fatty Acids, Cholesterol, Protein and Amino Acids; National Academies Press: Washington, DC, USA, 2005. [Google Scholar]
- Turner, N.D.; Lupton, J.R. Dietary fiber. Adv. Nutr. 2011, 2, 151–152. [Google Scholar] [CrossRef] [PubMed]
- American Association of Cereal Chemists. The definition of dietary fiber. Cereal Foods World 2001, 46, 112–126. [Google Scholar]
- Codex Alimentarius Commission. Guidelines for the use of nutrition claims: draft table of conditions for nutrient contents (Part B) provisions on dietary fibre. In Proceedings of the Report of the 30th Session of the Codex Committee on Nutrition and Foods for Special Dietary Uses, Cape Town, South Africa, 3–7 November 2008. ALINORM 09/32/26.
- Kraan, S. Algal polysaccharides, novel applications and outlook. In Carbohydrates—Comprehensive Studies on Glycobiology and Glycotechnology; InTech: Rijeka, Croatia, 2012; pp. 489–524. [Google Scholar]
- Patarra, R.F.; Paiva, L.; Neto, A.I.; Lima, E.; Baptista, J. Nutritional value of selected macroalgae. J. Appl. Phycol. 2011, 23, 205–208. [Google Scholar] [CrossRef]
- Rupérez, P.; Saura-Calixto, F. Dietary fiber and physicochemical properties of edible Spanish seaweeds. Eur. Food Res. Technol. 2001, 212, 349–354. [Google Scholar]
- Burtin, P. Nutritional value of seaweeds. EJEAFChe 2003, 2, 498–503. [Google Scholar]
- Dawczynski, C.; Schubert, R.; Jahrein, G. Amino acids, fatty acids, and dietary fibre in edible seaweed products. Food Chem. 2007, 103, 891–899. [Google Scholar] [CrossRef]
- Institut de Phytonutrition. Functional, Health and Therapeutic Effects of Algae and Seaweed, version 1.5; Institut de Phytonutrition Electronic Database: Beausoleil, France, 2004. [Google Scholar]
- Norziah, M.H.; Ching, C.Y. Nutritional composition of edible seaweed Gracilaria changgi. Food Chem. 2000, 68, 69–76. [Google Scholar] [CrossRef]
- Wong, K.H.; Cheung, P.C.K. Nutritional evaluation of some red and green seaweeds, Part I—Proximate composition, amino acid profiles and some physico-chemical properties. Food Chem. 2000, 71, 475–482. [Google Scholar] [CrossRef]
- Dvir, I.; Chayoth, R.; Sod-Moriah, U.; Shany, S.; Nyska, A.; Stark, A.H.; Madar, Z.; Arad, S.M. Soluble polysaccharide and biomass of red microalga Porphyridium sp. alter intestinal morphology and reduce cholesterol in rats. Br. J. Nutr. 2000, 84, 469–476. [Google Scholar] [PubMed]
- Lahaye, M.; Jegou, D. Chemical and physico-chemical characteristics of dietary fibers of Ulva lactuca (L.) Thuret and Enteromorpha compressa (L.) Grev. J. Appl. Phycol. 1993, 5, 195–200. [Google Scholar] [CrossRef]
- Yoshie, Y.; Suzuki, T.; Shirai, T.; Hirano, T. Analytical procedure and distribution of soluble and insoluble dietary fibers in seaweed foods. J. Tokyo Univ. Fish. 1997, 83, 1–12. [Google Scholar]
- McCance, R.A.; Widdowson, E.M.; Holland, B. McCance and Widdowson’s Composition of Foods, 6th ed.; Royal Society Chemistry: Cambridge, UK, 1993. [Google Scholar]
- MacArtain, P.; Gill, C.I.R.; Brooks, M.; Campbell, R.; Rowland, I.R. Nutritional value of edible seaweeds. Nutr. Rev. 2007, 65, 535–543. [Google Scholar] [CrossRef] [PubMed]
- McLachlan, A.J.; Morgan, P.R.; Howard-Williams, C.; McLachlan, S.M.; Bourn, D. Aspects of the recovery of a saline African lake following a dry period. Arch. Hydrobiol. 1972, 70, 325–340. [Google Scholar]
- Pereira, L. A review of the nutrient composition of selected edible seaweeds. In Seaweed: Ecology, Nutrient Composition and Medicinal Uses; Pomin, V.H., Ed.; Nova Science Publishers, Inc.: New York, NY, USA, 2011; pp. 15–47. [Google Scholar]
- Praznik, W.; Loeppert, R.; Viernstein, H.; Haslberger, A.G.; Unger, F.M. Dietary fiber and prebiotics. In Polysaccharides: Bioactivity and Biotechnology; Ramawat, K.G., Mérillon, J.M., Eds.; Springer International Publishing: Cham, Switzerland, 2015; pp. 891–925. [Google Scholar]
- Meyer, K.A.; Kushi, L.H.; Jacobs, D.R.; Slavin, J.; Sellers, T.A.; Folsom, A.R. Carbohydrates, dietary fiber, and incident type 2 diabetes in older women. Am. J. Clin. Nutr. 2002, 71, 921–931. [Google Scholar]
- Kasper, H. Ernährungsmedizin und Diätetic, 9th ed.; Elsevier: Amsterdam, The Netherlands; Urban & Fischer: München, Germany, 2000. [Google Scholar]
- Baghurst, P.A.; Baghurst, K.I.; Record, S.J. Dietary fiber, non-starch polysaccharides and resistant starch—A review. Food Aust. 1996, 48, S3–S35. [Google Scholar]
- Potty, V.H. Physico-chemical aspects, physiological functions, nutritional importance and technological significance of dietary fibers—A critical appraisal. J. Food Sci. Technol. 1996, 33, 1–18. [Google Scholar]
- Courtois, J. Oligosaccharides from land plants and algae: Production and applications in therapeutics and biotechnology. Curr. Opin. Microbiol. 2009, 12, 261–273. [Google Scholar] [CrossRef] [PubMed]
- Klarzynski, V.; Descamps, V.; Plesse, B.; Yvin, J.C.; Kloareg, B.; Fritig, B. Sulfated fucan oligosaccharides elicit defense responses in tobacco and local systemic resistance against tobacco mosaic virus. Mol. Plant Microb. Interact. 2003, 16, 115–122. [Google Scholar] [CrossRef] [PubMed]
- Chow, J.T.N.; Williamson, D.A.; Yates, K.M.; Goux, W.J. Chemical characterization of the immunomodulating polysaccharide of Aloe vera L. Carbohydr. Res. 2005, 340, 1131–1142. [Google Scholar] [CrossRef] [PubMed]
- Zaporozhets, T.S.; Besednova, N.N.; Kusnetsova, T.A.; Zvyagintseva, T.N.; Makarenkova, I.D.; Kryzhanovsky, S.P.; Melnikov, V.G. The prebiotic potential of polysaccharides and extracts of seaweeds. Russ. J. Mar. Bot. 2014, 40, 1–9. [Google Scholar] [CrossRef]
- Kardos, N.; Luche, J.L. Sonochemistry of carbohydrate compounds. Carbohydr. Res. 2001, 332, 115–131. [Google Scholar] [CrossRef]
- Lii, C.Y.; Chen, C.H.; Yeh, A.I.; Lai, V.M.F. Preliminary study on the degradation kinetics of agarose and carrageenans by ultrasound. Food Hydrocoll. 1999, 13, 477–481. [Google Scholar] [CrossRef]
- Sun, L.; Wang, C.; Shi, Q.; Ma, C. Preparation of different molecular weight polysaccharides from Porphyridium cruentum and their antioxidant activities. Int. J. Biol. Macromol. 2009, 45, 42–47. [Google Scholar] [CrossRef] [PubMed]
- Zúñiga, E.A.; Matsuhiro, B.; Mejías, E. Preparation of low-molecular weight fraction by free radical depolymerisation of the sulfated galactan from Schizymenia binderi (Gigartinales, Rhodophyta) and its anticoagulant activity. Carbohydr. Polym. 2006, 66, 208–215. [Google Scholar] [CrossRef]
- Leal, B.E.S. Obtenção de Oligossacarídeos Prebióticos a Partir da Hidrólise Fosfórica da Biomassa de Microalgas Utilizadas na Biomitigação de CO2 de Efluente Gasoso de Churrascaria. Master’s Thesis, Universidade Tecnológica Federal do Paraná, Curitiba, Brazil, 2015. [Google Scholar]
- Nardella, A.; Chaubet, F.; Boisson-Vidal, C.; Blondin, C.; Durand, P.; Jozefonvicz, J. Anticoagulant low molecular weight fucans produced by radical process and ion exchange chromatography of high molecular weight fucans extracted from the brown seaweed Ascophyllum nodosum. Carbohydr. Res. 1996, 289, 201–208. [Google Scholar] [CrossRef]
- Patel, S.; Goyal, A. The current trends and future perspectives of prebiotics research: A review. 3 Biotech 2012, 2, 115–125. [Google Scholar] [CrossRef]
- Enoki, T.; Okuda, S.; Kudo, Y.; Takashima, F.; Sagawa, H.; Kato, I. Oligosaccharides from agar inhibit pro-inflammatory mediator release by inducing heme oxygenase 1. Biosci. Biotechnol. Biochem. 2010, 74, 766–770. [Google Scholar] [CrossRef] [PubMed]
- Claudia, N.P.; Matsuhiro, B. Characterization of a fucoidan from Lessonia vadose (Phaeophyta) and its anticoagulant and elicitor properties. Int. J. Biol. Macromol. 2008, 42, 235–240. [Google Scholar]
- Zhou, G.; Sun, Y.P.; Xin, H.; Zhang, Y.; Li, Z.; Xu, Z. In vivo antitumor and immunomodulation activities of different molecular weight lambda-carrageenans from Chondrus ocellatus. Pharmacol. Res. 2004, 50, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Wang, L.; Zhou, Y. Immunomodulation and antitumor activities of different molecular-weight polysaccharides from Porphyridium cruentum. Carbohydr. Res. 2012, 87, 1206–1210. [Google Scholar] [CrossRef]
- Holck, J.; Hjernø, K.; Lorentzen, A.; Vigsnæs, L.K.; Hemmingsen, L.; Licht, T.R.; Mikkelsen, J.D.; Meyer, A.S. Tailored enzymatic production of oligosaccharides from sugar beet pectin and evidence of differential effects of a single DP chain length difference on human faecal microbiota composition after in vitro fermentation. Proc. Biochem. 2011, 46, 1039–1049. [Google Scholar] [CrossRef]
- Wang, Z.; Roberts, A.B.; Buffa, A.J.; Levison, B.S.; Zhu, W.; Org, E.; Gu, X.; Huang, Y.; Zamanian-Daryoush, M.; Culley, M.K.; et al. Non-lethal inhibition of gut microbial trimethylamine production for the treatment of atherosclerosis. Cell 2015, 163, 1585–1595. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, D.; Rocha-Santos, T.A.P.; Gomes, A.M.; Goodfellow, B.J.; Freitas, A.C. Lipolysis in probiotic and symbiotic cheese: The influence of probiotic bacteria, prebiotic compounds and ripening time on free fatty acid profiles. Food Chem. 2012, 131, 1414–1421. [Google Scholar] [CrossRef]
- Raposo, M.F.J.; Morais, A.M.M.B. Microalgae for the prevention of cardiovascular disease and stroke. Life Sci. 2015, 125, 32–41. [Google Scholar] [CrossRef]
- Raposo, M.F.J.; Morais, A.M.M.B.; Morais, R.M.S.C. Marine polysaccharides from algae with potential biomedical applications. Mar. Drugs 2015, 13, 2967–3028. [Google Scholar] [CrossRef] [PubMed]
- Raposo, M.F.J.; Morais, A.M.M.B.; Morais, R.M.S.C. Polysaccharides from marine microalgae. In Polysaccharides: Bioactivity and Biotechnology; Ramawat, K.G., Mérillon, J.M., Eds.; Springer International Publishing: Cham, Switzerland, 2015; pp. 1683–1727. [Google Scholar]
- Werman, M.J.; Sukenik, A.; Mokady, S. Effects of the marine unicellular alga Nannochloropsis sp. to reduce the plasma and liver cholesterol levels in male rats fed on diets with cholesterol. Biosci. Biotechnol. Biochem. 2003, 67, 2266–2268. [Google Scholar] [CrossRef] [PubMed]
- De Morais, M.G.; Vaz, B.S.; de Morais, E.G.; Costa, J.A.V. Biologically active metabolites synthesized by microalgae. Biomed. Res. Int. 2015, 2015. [Google Scholar] [CrossRef] [PubMed]
- Jimenez-Escrig, A.; Sanchez-Muniz, F.J. Dietary fiber from edible seaweeds: Chemical, structure, physicochemical properties and effects on cholesterol metabolism. Nutr. Res. 2000, 20, 585–598. [Google Scholar] [CrossRef]
- Pal, A.; Kamthania, M.C.; Kumar, A. Bioactive compounds and properties of seaweeds—A review. OALib J. 2014, 1, e752. [Google Scholar] [CrossRef]
- Lahaye, M. Marine-algae as sources of fibers—Determination of soluble and insoluble dietary fiber contents in some sea vegetables. J. Sci. Food Agric. 1991, 54, 5987–594. [Google Scholar] [CrossRef]
- Lahaye, M.; Thibault, J.F. Chemical and physico-chemical properties of fibers from algae extraction by-products. In Dietary Fibre: Chemical and Biological Aspects; Southgate, D.A.T., Waldron, K., Johnson, I.T., Fenwick, G.R., Eds.; Royal Society Chemistry: Cambridge, UK, 1990; pp. 68–72. [Google Scholar]
- Wood, P.J. Cereal β-glucans: Structure, properties and health claims. In Advanced Dietary Fibre Technology; McCleary, B.V., Prosky, L., Eds.; Blackwell Science: Oxford, UK, 2001; pp. 315–327. [Google Scholar]
- Fleury, N.; Lahaye, M. Chemical and physicochemical characterization of fibers from Laminaria digitata (Kombu Breton)—A physiological approach. J. Sci. Food Agric. 1991, 55, 389–400. [Google Scholar] [CrossRef]
- Lahaye, M.; Roneau-Mouro, C.; Deniaud, E.; Buléon, A. Solid-state 13C NMR spectroscopy studies of xylans in the cell wall of Palmaria palmata (L. Kuntze, Rhodophyta). Carbohydr. Res. 2003, 338, 1559–1569. [Google Scholar] [CrossRef]
- Kimura, Y.; Watanabe, K.; Okuda, H. Effects of soluble sodium alginate on cholesterol excretion and glucose tolerance in rats. J. Ethnopharmacol. 1996, 54, 47–54. [Google Scholar] [CrossRef]
- Zee, S. Body weight loss with the aid of alginic acid. Med. Arh. 1991, 45, 113–114. [Google Scholar]
- Vaugelade, P.; Hoebler, C.; Bernard, F.; Guillon, F.; Lahaye, M.; Duee, P.H.; Darcy-Vrillon, B. Non-starch polysaccharides extracted from seaweed can modulate intestinal absorption of glucose and insulin response in the pig. Reprod. Nutr. Dev. 2000, 40, 33–47. [Google Scholar] [CrossRef] [PubMed]
- Torsdottir, I.; Alpsten, M.; Holm, G.; Sandberg, A.S.; Tolli, J. A small dose of soluble alginate-fiber affects postprandial glycemia and gastric-emptying in humans with diabetes. J. Nutr. 1991, 121, 795–799. [Google Scholar] [PubMed]
- Terada, A.; Hara, H.; Mitsuoka, T. Effect of dietary alginate on the faecal microbiota and faecal metabolic activity in humans. Microbial Ecol. Health Dis. 1995, 8, 259–266. [Google Scholar] [CrossRef]
- Wang, Y.; Han, F.; Hu, B.; Li, J.; Yu, W. In vivo prebiotic properties of alginate oligosaccharides prepared through enzymatic hydrolysis of alginate. Nutr. Res. 2006, 26, 597–603. [Google Scholar] [CrossRef]
- Morrissey, J.; Kraan, S.; Guiry, M.D. A Guide to Commercially Important Seaweeds on the Irish Coast; Bord Iascaigh Mhara/Irish Sea Fisheries Board: Dublin, Ireland, 2001. [Google Scholar]
- Dumelod, B.D.; Ramirez, R.P.B.; Tiangson, C.L.P.; Barrios, E.B.; Panlasigui, L.N. Carbohydrate availability of arroz caldo with lambda-carrageenan. Int. J. Food Sci. Nutr. 1999, 50, 283–289. [Google Scholar] [PubMed]
- Deville, C.; Damas, J.; Forget, J.; Dandrifosse, G.; Peulen, O. Laminarin in the dietary fiber concept. J. Sci. Food Agric. 2004, 84, 1030–1038. [Google Scholar] [CrossRef]
- Deville, C.; Gharbi, M.; Dandrifosse, G.; Peulen, O. Study of the effects of laminarin, a polysaccharide from seaweed, on gut characteristics. J. Sci. Food Agric. 2007, 87, 1717–1725. [Google Scholar] [CrossRef]
- Lahaye, M.; Michel, C.; Barry, J.L. Chemical, physicochemical and in vitro fermentation characteristics of dietary-fibers from Palmaria palmata (L.) Kuntze. Food Chem. 1993, 47, 29–36. [Google Scholar] [CrossRef]
- Li, N.; Zhang, Q.B.; Song, J.M. Toxicological evaluation of fucoidan extracted from Laminaria japonica in Wistar rats. Food Chem. Toxicol. 2005, 43, 421–426. [Google Scholar] [CrossRef] [PubMed]
- Burri, S.; Tato, I.; Nunes, M.L.; Morais, R. Functional vegetable-based sausages for consumption by children. Food Nutr. Sci. 2011, 2, 494–501. [Google Scholar] [CrossRef]
- Raposo, M.F.J.; Morais, R.M.S.C.; Morais, A.M.M.B. Bioactivity and applications of sulphated polysaccharides from marine microalgae. Mar. Drugs 2013, 11, 233–252. [Google Scholar] [CrossRef] [PubMed]
- Southgate, D.A.T. Duetary fiber and health. In Dietary Fiber: Chemical and Biological Aspects; Southgate, D.A.T., Waldron, K., Johnson, I.T., Fenwick, G.R., Eds.; Royal Society Chemistry: Cambridge, UK, 1990. [Google Scholar]
- Roberfroid, M.B. Dietary fiber, inulin, and oligofructose: A review comparing their physiological effects. Crit. Rev. Food Sci. Nutr. 1993, 33, 103–148. [Google Scholar] [CrossRef] [PubMed]
- Gibson, G.R.; Roberfroid, M.B. Dietary modulation of the human colonic microbiota: Introducing the concept of prebiotics. J. Nutr. 1995, 125, 1401–1412. [Google Scholar] [PubMed]
- Gibson, G.R.; Probert, H.M.; van Loo, J.A.E.; Roberfroid, M.B. Dietary modulation of human colonic microbiota: Updating the concept of prebiotics. Nutr. Res. Rev. 2004, 17, 257–259. [Google Scholar] [CrossRef]
- Gibson, G.R.; Scott, K.P.; Rastall, R.A.; Tuohy, K.M.; Hotchkiss, A.; Dubert-Ferrandon, A.; Gareau, M.; Murphy, E.F.; Saulnier, D.; Loh, G.; et al. Dietary prebiotics: Current status and new definition. Food Sci. Technol. Bull. Funct. Foods 2010, 7, 1–19. [Google Scholar] [CrossRef]
- Roberfroid, M. Prebiotics: The concept revisited. J. Nutr. 2007, 137, 830S–837S. [Google Scholar] [PubMed]
- Binns, N. Probiotics, Prebiotics and the Gut Microbiota; ILSI Europe: Brussels, Belgium, 2013; p. 32. [Google Scholar]
- Fernandes, J.; Su, W.; Rahat-Rozenbloom, S.; Wolever, T.M.S.; Comelli, E.M. Adiposity, gut microbiota, and faecal short chain fatty acids are linked in adult humans. Nutr. Diabetes 2014, 4, e121. [Google Scholar] [CrossRef] [PubMed]
- Vogt, J.A.; Wolever, T.M.S. Faecal acetate is inversely related to acetate absorption from the human rectum and distal colon. J. Nutr. 2003, 133, 3145–3148. [Google Scholar] [PubMed]
- Cummings, J.H. Short chain fatty acids. In Human Colonic Bacteria: Role in Nutrition, Physiology and Pathology; Gibson, G.R., Macfarlane, G.T., Eds.; CRC Press: Boca Raton, FL, USA, 1995; pp. 101–130. [Google Scholar]
- Macfarlane, G.T.; Gibson, G.R.; Cummings, J.H. Comparison of fermentation reactions in different regions of the colon. J. Appl. Bacteriol. 1992, 72, 57–64. [Google Scholar] [PubMed]
- Barcenilla, A.; Pride, S.E.; Martin, J.C.; Duncan, S.H.; Stewart, C.S.; Henderson, C.; Flint, J.Y. Phylogenetic relationships of butyrate-producing bacteria from the human gut. Appl. Environ. Microbiol. 2000, 66, 1654–1661. [Google Scholar] [CrossRef] [PubMed]
- Salminen, S.; Bouley, C.; Boutron-Ruault, M.C.; Cummings, J.H.; Franck, A.; Gibson, G.R.; Isolauri, I.; Moreau, M.C.; Roberfroid, M.; Rowland, I.R. Functional food science and gastrointestinal function. Br. J. Nutr. 1998, 80, S147–S171. [Google Scholar] [CrossRef] [PubMed]
- Mykkanen, H.; Laiho, K.; Salminen, S. Variations in faecal bacterial enzyme activities and associations with bowel function and diet in elderly subjects. J. Appl. Microbiol. 1998, 85, 37–41. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Gibson, G.R.; Roberfroid, M.B. Colonic Microbiota Nutrition and Health; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1999. [Google Scholar]
- Macfarlane, G.T.; McBain, A.J. The human colonic microbiota. In Colonic Microbiota Nutrition and Health; Gibson, G.R., Roberfroid, M.B., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1999; pp. 1–25. [Google Scholar]
- Van der Waaij, D. Microbial ecology of the intestinal microflora: Influence of interactions with the host organism. In Probiotics, Other Nutritional Factors, and Intestinal Microflora; Hanson, L.A., Yolken, R.H., Eds.; Lippincott-Raven Publishers: Philadelphia, PA, USA, 1999; pp. 1–16. [Google Scholar]
- Bhowmik, D.; Dubey, J.; Mehra, S. Probiotic efficiency of Spirulina platensis-stimulating growth of lactic acid bacteria. World J. Dairy Food Sci. 2009, 42, 160–163. [Google Scholar]
- Conway, P.L. Prebiotics and human health: The state-of-the-art and future perspectives. Food Nutr. Res. 2001, 45, 13–21. [Google Scholar] [CrossRef]
- Furrie, E.; Macfarlane, S.; Kennedy, A.; Cummings, J.H.; Walsh, S.V.; O’Neil, D.A.; Macfarlane, G.T. Synbiotic therapy (Bifidobacterium longum/Synergy 1) initiates resolution of inflammation in patients with active ulcerative colitis: A randomized controlled pilot trial. Gut 2005, 54, 242–249. [Google Scholar] [CrossRef] [PubMed]
- Lindsay, J.O.; Whelan, K.; Stagg, A.J.; Gobin, P.; Al-Hassi, H.O.; Rayment, N.; Kamm, M.A.; Knight, S.C.; Forbes, A. Clinical, microbiological, and immunological effects of fructo-oligosaccharide in patients with Crohn’s disease. Gut 2006, 55, 348–355. [Google Scholar] [CrossRef] [PubMed]
- Silk, D.B.; Davis, A.; Vulevic, J.; Tzortzis, G.; Gibson, G.R. Clinical trial: The effects of a trans-galactooligosaccharide prebiotic on faecal microbiota and symptoms in irritable bowel syndrome. Aliment. Pharmacol. Ther. 2009, 29, 508–518. [Google Scholar] [CrossRef] [PubMed]
- Uchida, M.; Mogami, O.; Matsueda, K. Characteristic of milk whey culture with Propioni-bacterium freudenreichii ET-3 and its application to the inflammatory bowel disease therapy. Inflammopharmacology 2007, 15, 105–108. [Google Scholar] [CrossRef] [PubMed]
- Kasubuchi, M.; Hasegawa, S.; Hiramatsu, T.; Ichimura, A.; Kimura, I. Dietary gut microbial metabolites, short-chain fatty acids, and host metabolic regulation. Nutrients 2015, 7, 2839–2849. [Google Scholar] [CrossRef] [PubMed]
- Rafter, J.; Bennett, M.; Caderni, G.; Clune, Y.; Hughes, R.; Karlson, P.C.; Klinder, A.; O’Riordan, M.; O’Sullivan, G.C.; Pool.Zobel, B.; et al. Dietary synbiotics reduce cancer risk factors in polypectomized and colon cancer patients. Am. J. Clin. Nutr. 2007, 85, 488–496. [Google Scholar] [PubMed]
- Rowland, I.R.; Rumney, C.J.; Coutts, J.T.; Lievense, L.C. Effect of Bifidobacterium longum and inulin on gut bacterial metabolism and carcinogen-induced aberrant crypt focci in rats. Carcinogenesis 1998, 19, 281–285. [Google Scholar] [CrossRef] [PubMed]
- Demigne, C.; Jacobs, H.; Moundras, C.; Davicco, M.J.; Horcajada, M.N.; Berlanier, A.; Coxam, V. Comparison of native or reformulated chicory fructans, or non-purified chicory, on rat cecal fermentation and mineral metabolism. Eur. L. Nutr. 2008, 47, 366–374. [Google Scholar] [CrossRef] [PubMed]
- Abrams, S.A.; Griffin, I.J.; Hawthorne, K.M.; Liang, L.; Gunn, S.K.; Darlington, G.; Ellis, K.J. A combination of prebiotic short- and long-chain inulin-type fructans enhance calcium absorption and bone mineralization in young adolescents. Am. J. Clin. Nutr. 2005, 82, 471–476. [Google Scholar] [PubMed]
- Cieślik, E.; Topolska, K.; Pisulewski, P.M. Effect of inulin-type fructans on body weight gain and selected biochemical parameters at calcium hypoalimentation in rats. Pol. J. Food Nutr. Sci. 2009, 59, 163–168. [Google Scholar]
- Cummings, J.H.; Christie, S.; Cole, T.J. A study of fructo-oligosaccharide in the prevention of travellers’ diarrhoea. Aliment. Pharmacol. Ther. 2001, 15, 1139–1145. [Google Scholar] [CrossRef] [PubMed]
- Lewis, S.; Burmeister, S.; Brazier, J. Effect of the prebiotic oligofructose on relapse of Clostridium difficile-associated diarrhea: A randomized, controlled study. Clin. Gastroenterol. Hepatol. 2005, 3, 442–448. [Google Scholar] [CrossRef]
- Waligora-Dupriet, A.J.; Campeotto, F.; Nicolis, I.; Bonet, A.; Soulaines, P.; Dupont, C.; Butel, M.J. Effect of oligofructose supplementation on gut microflora and well-being in young children attending a day care centre. Int. J. Food Microbiol. 2007, 113, 108–113. [Google Scholar] [CrossRef] [PubMed]
- Sotnikova, N.; Antsiferova, I.; Malyshkina, A. Cytokine network of eutopic and ectopic endometrium in women with adenomyosis. Am. J. Reprod. Immunol. 2002, 47, 251–255. [Google Scholar] [CrossRef] [PubMed]
- Macfarlane, G.T.; Gibson, S.R. The control and consequences of bacterial fermentation in the human colon. J. Appl. Bacteriol. 1991, 70, 443–459. [Google Scholar]
- Ley, R.E.; Bäckhed, F.; Turnbaugh, P.J.; Lozupone, C.A.; Knight, R.D.; Gordon, J.I. Obesity alters gut microbial ecology. Proc. Natl. Acad. Sci. USA 2005, 102, 11070–11075. [Google Scholar] [CrossRef] [PubMed]
- Turnbaugh, P.J.; Ley, R.E.; Mahowald, M.A.; Magrini, V.; Mardis, E.R.; Gordon, J.I. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature 2006, 444, 1027–1031. [Google Scholar] [CrossRef] [PubMed]
- Ley, R.E.; Turnbaugh, P.J.; Klein, S.; Gordon, J.I. Microbial ecology: Human gut microbes associated with obesity. Nature 2006, 444, 1022–1023. [Google Scholar] [CrossRef] [PubMed]
- Topping, D.L.; Clifton, P.M. Short-chain fatty acids and human colonic function: Roles of resistant starch and non-starch polysaccharides. Physiol. Rev. 2001, 81, 1031–1064. [Google Scholar]
- Bird, A.R.; Brown, I.L.; Topping, D.L. Starches, resistant starches, the gut microflora and human health. Curr. Issues Intest. Microbiol. 2000, 1, 25–37. [Google Scholar] [PubMed]
- Pluske, J.R.; Durmic, Z.; Pethick, D.W.; Mullan, B.P.; Hampson, D.J. Confirmation of the role of rapidly fermentable carbohydrates in the expression of swine dysentery in pigs after experimental infection. J. Nutr. 1998, 128, 1737–1744. [Google Scholar] [PubMed]
- Topping, D.L.; Mock, S.; Trimble, R.P.; Illman, R.J. Effects of varying the content and proportions of gum Arabic and cellulose on cecal volatile fatty acids in the rat. Nutr. Res. 1988, 8, 1013–1020. [Google Scholar] [CrossRef]
- Baghurst, K.I.; Hope, A.K.; Down, E.C. Dietary intake in a group of institutionalized elderly and the effects of a fibre supplementation program on nutrient intake and weight gain. Communit. Health Stud. 1985, 9, 99–108. [Google Scholar] [CrossRef]
- Cherrington, C.A.; Hinton, M.; Pearson, G.R.; Chopra, I. Short-chain organic acids at pH 5.0 kill Escherichia coli and Salmonella spp. without causing membrane perturbation. J. Appl. Bacteriol. 1991, 70, 161–165. [Google Scholar] [CrossRef] [PubMed]
- Slavin, J. Fiber and prebiotics: Mechanisms and health benefits. Nutrients 2013, 5, 1417–1435. [Google Scholar] [CrossRef] [PubMed]
- Brown, I.L.; Warhurst, M.; Arcot, J.; Playne, M.; Illman, R.J.; Topping, D.L. Fecal numbers of bifidobacteria are high in pigs fed Bifidobacterium longum with a high amylose (amylomayze) starch than with a low amylomayze starch. J. Nutr. 1997, 127, 1822–1827. [Google Scholar] [PubMed]
- Campbell, J.M.; Fahey, G.C., Jr.; Wolf, B.W. Selected indigestible oligosaccharides affect large bowel mass, cecal and fecal short-chain fatty acids, pH and microflora in rats. J. Nutr. 1997, 127, 130–136. [Google Scholar] [PubMed]
- Kleessen, B.; Sykura, B.; Zunft, H.J.; Blaut, M. Effects of inulin and lactose on fecal microflora, microbial activity, and bowel habit in elderly constipated persons. Am. J. Clin. Nutr. 1997, 65, 1397–1402. [Google Scholar] [PubMed]
- Ohkusa, T.; Ozaki, Y.; Sato, C.; Mikuni, K.; Ikeda, H. Long-term ingestion of lactosucrose increases Bifidobacterium sp. in human fecal flora. Digestion 1995, 56, 415–420. [Google Scholar] [CrossRef] [PubMed]
- Dimitroglou, A.; Davies, S.J.; Sweetman, J.; Divanach, P.; Chatzifotis, S. Dietary supplementation of mannan oligosaccharide on white sea bream (Diplodus sargus L.) larvae: Effects on development, gut morphology and salinity tolerance. Aquacult. Res. 2010, 41, e245–e251. [Google Scholar] [CrossRef]
- Marinho, M.C.; Pinho, M.A.; Mascarenhas, R.D.; Silva, F.C.; Lordelo, M.M.; Cunha, L.F.; Freire, J.P.B. Effect of prebiotic or probiotic supplementation and ileo rectal anastomosis on intestinal morphology of weaned piglets. Livest. Sci. 2007, 108, 240–243. [Google Scholar] [CrossRef]
- Pérez-Conesa, D.; López, G.; Ros, G. Effects of probiotic, prebiotic and symbiotic follow-up infant formulas on large intestine morphology and bone mineralisation in rats. J. Sci. Food Agric. 2007, 87, 1059–1068. [Google Scholar] [CrossRef]
- Mourão, J.L.; Pinheiro, V.; Alves, A.; Guedes, C.M.; Pinto, L.; Saavedra, M.J.; Spring, P.; Kocher, A. Effect of mannan oligosaccharides on the performance, intestinal morphology and cecal fermentation of fattening rabbits. Anim. Feed Sci. Technol. 2006, 126, 107–120. [Google Scholar] [CrossRef]
- Spencer, J.D.; Touchete, K.J.; Liu, H.; Alle, G.L.; Newcom, M.D.; Kerley, M.S.; Pace, L.W. Effect of spray-dried plasma and fructooligosaccharide on nursery performance and small intestinal morphology on weaned pigs. J. Anim. Sci. 1997, 75, 199. [Google Scholar]
- Ohta, A.; Osakabe, N.; Yamada, K.; Saito, Y.; Hidaka, H. Effects of fructooligosaccharides and other saccharides on Ca, Mg and P absorption in rats. J. Jpn. Soc. Nutr. Food Sci. 1993, 46, 123–129. [Google Scholar] [CrossRef]
- Chonan, O.; Watanuki, M. Effect of galactooligosaccharides on calcium absorption in rats. J. Nutr. Sci. Vitaminol. (Tokyo) 1995, 41, 95–104. [Google Scholar] [CrossRef] [PubMed]
- Salgado, P.; Martins, J.M.; Carvalho, F.; Abreu, M.; Freire, J.P.B.; Toullec, R.; Lalles, J.P.; Bento, O. Component digestibility of lupin (Lupinus angustifolius) and pea (Pisum sativum) seeds and effects on the small intestine and body organs in anastomosed and intact growing pigs. Anim. Feed Sci. Technol. 2002, 98, 187–201. [Google Scholar] [CrossRef]
- Fuller, M.F. Methodologies of the measurement of digestion. In Proceedings of the 5th International Symposium on Digestive Physiology in Pigs, Wageningen, The Netherlands, 24–26 April 1991; Verstegen, M.W.A., Huisman, J., den Hartog, L.A., Eds.; 1991; pp. 273–288. [Google Scholar]
- Liu, J.; Kandasamy, S.; Zhang, J.; Kirby, C.W.; Karakach, T.; Hafting, J.; Crichley, A.T.; Evans, F.; Prithiviraj, B. Prebiotic effects of diet supplemented with the cultivated red seaweed Chondrus crispus or with fructo-oligosaccharide on host immunity, colonic microbiota and gut microbial metabolites. BMC Complement. Altern. Med. 2015, 15, 279–290. [Google Scholar] [CrossRef] [PubMed]
- Resta-Lenert, S.; Barrett, K.E. Live probiotics protect intestinal epithelial cells from the effects of infection with enteroinvasive Escherichia coli (EIEC). Gut 2003, 52, 988–997. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.J.; Goldworthy, S.M.; Barnes, A.A.; Eilert, M.M.; Tcheang, I.; Daniels, D.; Muir, A.I.; Wigglesworth, M.J.; Kinghorn, I.; Fraser, N.J.; et al. The orphan G protein-coupled receptors GPR41 and GPR43 are activated by propionate and other short chain carboxylic acids. J. Biol. Chem. 2003, 278, 11312–11319. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, N.E.; Kotarsky, K.; Owman, C.; Olde, B. Identification of a free fatty acid receptor, FFAR2R, expressed on leukocytes and activated by short-chain fatty acids. Biochem. Biophys. Res. Commun. 2003, 303, 1047–1052. [Google Scholar] [CrossRef]
- Kimura, I.; Inoue, D.; Hirano, K.; Tsujimoto, G. The SCFA receptor GPR43 and energy metabolism. Front. Endocrinol. 2014, 5, 85. [Google Scholar] [CrossRef] [PubMed]
- Du, H.; van der A, D.L.; Boshuizen, H.C.; Forouhi, N.G.; Wareham, N.J.; Halkjaer, J.; TjØnnland, A.; Overvad, K.; Jakobsen, M.U.; Boeing, H.; et al. Dietary fiber and subsequent changes in body weight and waist circumference in European men and women. Am. J. Clin. Nutr. 2010, 91, 329–336. [Google Scholar] [CrossRef] [PubMed]
- Anderson, J.W.; Baird, P.; Davis, R.H., Jr.; Ferreri, S.; Knudtson, M.; Koraym, A.; Waters, V.; Williams, C.L. Health benefits of dietary fibre. Nutr. Rev. 2009, 67, 88–205. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Willet, W.C.; Manson, J.E.; Hu, F.B.; Rosner, B.; Colditz, G. Relation between changes in intakes of dietary fibre and grain products and changes in weight and development of obesity among middle-aged women. Am. J. Clin. Nutr. 2003, 78, 920–927. [Google Scholar] [PubMed]
- Freeland, K.R.; Wolever, T.M. Acute effects of intravenous and rectal acetate on glucagon-like peptide-1, peptide YY, ghrelin, adiponectin and tumour necrosis factor-alpha. Br. J. Nutr. 2010, 103, 460–466. [Google Scholar] [CrossRef] [PubMed]
- Tarini, J.; Wolever, T.M. The fermentable fibre inulin increases postprandial serum short-chain fatty acids and reduces free-fatty acids and ghrelin in healthy subjects. Appl. Physiol. Nutr. Metab. 2010, 35, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Atarashi, K.; Tanoue, T.; Shima, T.; Imaoka, A.; Kuwahara, T.; Momose, Y.; Cheng, G.; Yamasaki, S.; Saito, T.; Ohba, Y.; et al. Induction of colonic regulatory T cells by indigenous Clostridium species. Science 2011, 331, 337–341. [Google Scholar] [CrossRef] [PubMed]
- Round, J.L.; Mazmanian, S.K. Inducible Foxp3+ regulatory T-cell development by a commensal bacterium of the intestinal microbiota. Proc. Natl. Acad. Sci. USA 2010, 107, 12204–12209. [Google Scholar] [CrossRef]
- HØverstad, T.; Midtvedt, T. Short-chain fatty acids in germfree mice and rats. J. Nutr. 1986, 116, 1772–1776. [Google Scholar] [PubMed]
- Smith, P.M.; Howitt, M.R.; Panikov, N.; Michaud, M.; Gallini, C.A.; Bohlooly-Y, M.; Glockman, J.N.; Garrett, W.S. The microbial metabolites, short chain fatty acids, regulate colonic Treg cell homeostasis. Science 2013, 341, 569–573. [Google Scholar] [CrossRef] [PubMed]
- Cummings, J.H.; Pomare, E.W.; Branch, W.J.; Naylor, C.P.; Macfarlane, G.T. Short chain fatty acids in human large intestine, portal, hepatic, and venous blood. Gut 1987, 28, 1221–1227. [Google Scholar] [CrossRef] [PubMed]
- Vinolo, M.A.R.; Rodrigues, H.G.; Nachbar, R.T.; Curi, R. Regulation of inflammation by short chain fatty acids. Nutrients 2011, 3, 858–876. [Google Scholar] [CrossRef] [PubMed]
- Waldecker, M.; Kautenburger, T.; Daumann, H.; Busch, C.; Schrenk, D. Inhibition of histone-deacetylase activity by short-chain fatty acids and some polyphenol metabolites formed in the colon. J. Nutr. Biochem. 2008, 19, 587–593. [Google Scholar] [CrossRef] [PubMed]
- Hinnebusch, B.F.; Meng, S.; Wu, J.T.; Archer, S.Y.; Hodin, R.A. The effects of short-chain fatty acids on human colon cancer cell-phenotype are associated with histone hyperacetylation. J. Nutr. 2002, 132, 1012–1017. [Google Scholar] [PubMed]
- Sealy, L.; Chalkley, R. The effect of sodium butyrate on histone modification. Cell 1978, 14, 115–121. [Google Scholar] [CrossRef]
- Morgan, D.O. Principles of CDK regulation. Nature 1995, 374, 131–134. [Google Scholar] [CrossRef] [PubMed]
- Newmark, H.L.; Young, C.W. Butyrate and phenylacetate as differentiating agents: Practical problems and opportunities. J. Cell Biochem. 1995, 22, 247–253. [Google Scholar] [CrossRef]
- Boffa, L.C.; Lupton, J.R.; Mariani, M.R. Modulation of colonic epithelial cell proliferation, histone acetylation, and luminal short chain fatty acids by variation of dietary fiber in rats. Cancer Res. 1992, 52, 5906–5912. [Google Scholar] [PubMed]
- Medina, V.; Young, G.P.; Edmonds, B.; James, R.; Appleton, S.; Zalewski, D.P. Induction of caspase-3 protease activity and apoptosis by butyrate and trichostatin a (inhibitors of histone deacetylase): Dependence on protein synthesis and synergy with a mitochondrial/cytochrome C-dependent pathway. Cancer Res. 1997, 57, 3697–3707. [Google Scholar] [PubMed]
- Grunstein, M. Histone acetylation in chromatin structure and transcription. Nature 1997, 389, 349–352. [Google Scholar] [CrossRef] [PubMed]
- Meijer, K.; de Vos, P.; Priebe, M.G. Butyrate and other short-chain fatty acids as modulators of immunity: What relevance for health? Curr. Opin. Nutr. Metab. Care 2010, 13, 715–721. [Google Scholar] [CrossRef]
- Cavaglieri, C.R.; Nishiyama, A.; Fernandes, L.C.; Curi, R.; Miles, E.A.; Calder, P.C. Differential effects of short-chain fatty acids on proliferation and production of pro- and anti-inflammatory cytokines by cultured lymphocytes. Life Sci. 2003, 73, 1683–1690. [Google Scholar] [CrossRef]
- Doty, M.S.; Caddy, J.F.; Santelices, B. (Eds.) Case Studies of Seven Commercial Seaweed Resources. In FAO Fisheries Technical Paper-281, Food and Agriculture Organization of the United Nations; FAO: Rome, Italy, 1987; ISBN 92-5-102540-1.
- Agrimer, Algues Marines. Available online: http://www.agrimer.com/en/algues/2-brown/7-ascophyllum-nodosum.html (accessed on 18 December 2015).
- Agrimer, Algues Marines. Available online: http://www.agrimer.com/en/algues/2-brown/11-fucus-vesiculosus.html (accessed on 18 December 2015).
- Ramsden, L. Plant and algal gums and mucilages. In Chemical and Functional Properties of Food Saccharides; Tomasik, P., Ed.; CRC Press: Boca Raton, FL, USA, 2004; pp. 231–254. [Google Scholar]
- Wu, J.H.; Xu, C.; Shan, C.Y.; Tan, R.X. Antioxidant properties and PC12 cell protective effects of APS-1, a polysaccharide from Aloe vera var. chinensis. Life Sci. 2006, 78, 622–630. [Google Scholar] [CrossRef]
- Hu, B.; Gong, Q.N.; Wang, Y.; Ma, Y.; Li, J.; Yu, W. Prebiotic effects of neoagaro-oligosaccharides prepared by enzymatic hydrolysis of agarose. Anaerobe 2006, 12, 260–266. [Google Scholar] [CrossRef]
- Muraoka, T.; Ishihara, K.; Oyamada, C.; Kunitake, H.; Hirayama, I.; Kimura, T. Fermentation properties of low-quality red alga Susabinori Porphyra yezoensis by intestinal bacteria. Biosci. Biotechnol. Biochem. 2008, 72, 1731–1739. [Google Scholar] [CrossRef] [PubMed]
- Ray, B.; Lahaye, M. Cell-wall polysaccharides from the marine green alga Ulva rigida (Ulvales, Chlorophyta); chemical structure of ulvan. Carbohydr. Res. 1995, 274, 313–318. [Google Scholar] [CrossRef]
- Akiyama, H.; Endo, T.; Nakakita, R.; Murata, K.; Yonemoto, Y.; Okayama, K. Effect of depolymerized alginates on the growth of bifidobacteria. Biosci. Biotechnol. Biochem. 1992, 56, 355–356. [Google Scholar] [CrossRef] [PubMed]
- Pokusaeva, K.; Fitzgerald, G.; Sinderen, D. Carbohydrate metabolism in bifidobacteria. Genes Nutr. 2011, 6, 285–306. [Google Scholar] [CrossRef] [PubMed]
- Osipov, G.A.; Parfenov, A.I.; Verkhovtseva, N.V.; Ruchkina, I.N.; Kurchavov, V.A.; Boiko, N.B.; Rogatina, E.L. The clinical significance of the study of microorganisms on the intestinal mucous membrane by cultural-biochemical and chromatography-mass spectrometry methods. Exp. Klin. Gastroenterol. 2003, 4, 59–67. (In Russian) [Google Scholar]
- Kusaikin, M.I. O-glycosyl hydrolases of marine invertebrates. Properties and specifics of fucoidanases, sulfatases, and 1-3-beta-d-glucanases. Ph.D. Thesis, Pacific Institute of Bioorganic Chemistry FEB RAS, Vladivostok, Russia, 2003. [Google Scholar]
- Kuda, T.; Yano, T.; Matsuda, N.; Nishizawa, M. Inhibitory effects of laminaran and low molecular alginate against the putrefactive compounds produced by intestinal microflora in vitro and in rats. Food Chem. 2005, 91, 745–749. [Google Scholar] [CrossRef]
- Kusnetsova, T.A.; Zaporozhets, T.S.; Makarenkova, I.D.; Besednova, N.N.; Timchenko, N.F.; Zvyagintseva, T.N.; Shevchenko, N.M.; Mandrakova, N.V.; Melnikov, V.G. The prebiotic potential of polysaccharides from the brown alga Fucus evanescens and significance for the clinical use. Pac. Med. J. 2012, 1, 37–40. [Google Scholar]
- Gibson, G.R.; Roberfroid, M.B. Handbook of Prebiotics; Taylor and Francis: Boca Raton, FL, USA, 2008. [Google Scholar]
- Koneva, E.L. Substantiation and development of technologies for alginate-containing functional products. Ph.D. Thesis, Pacific Institute of Bioorganic Chemistry FEB RAS, Vladivostok, Russia, 2009. [Google Scholar]
- Yamada, Y.; Miyoshi, T.; Tanada, S.; Imaki, M. Digestibility and energy availability of wakame (Undaria pinnatifida) seaweed in Japanese. Nippon Eiseigaku Zasshi 1991, 46, 788–794. [Google Scholar] [CrossRef] [PubMed]
- Michel, C.; Lahaye, M.; Bonnet, C.; Mabeau, S.; Barry, J.L. In vitro fermentation by human faecal bacteria of total and purified dietary fibers from brown seaweeds. Br. J. Nutr. 1996, 75, 263–280. [Google Scholar] [CrossRef] [PubMed]
- O’Doherty, J.V.; Dillon, S.; Figat, S.; Callan, J.J.; Sweeney, T. The effects of lactose inclusion and seaweed extract derived from Laminaria spp. on performance, digestibility of diet components and microbial populations in newly weaned pigs. Anim. Feed Sci. Technol. 2010, 157, 173–180. [Google Scholar] [CrossRef]
- McDonnel, P.; Figat, S.; O’Doherty, J.V. The effect of dietary laminarin and fucoidan in the diet of the weanling piglet on performance, selected fecal microbial populations and volatile fatty acid concentrations. Animal 2010, 4, 579–585. [Google Scholar] [CrossRef] [PubMed]
- Gudiel-Urbano, M.; Goni, I. Effect of edible seaweeds (Undaria pinnatifida and Porphyra tenera) on the metabolic activities of intestinal microflora in rats. Nutr. Res. 2002, 22, 323–331. [Google Scholar] [CrossRef]
- Dierick, N.; Ovyn, A.; de Smet, S. Effect of feeding intact brown seaweed Ascophyllum nodosum on some digestive parameters and on iodine content in edible tissues in pigs. J. Sci. Food Agric. 2009, 89, 584–594. [Google Scholar] [CrossRef]
- Reilly, P.; O’Doherty, J.V.; Pierce, K.M. The effects of seaweed extract inclusion on gut morphology, selected intestinal microbiota, nutrient digestibility, volatile fatty acids concentration and the immune status of the weaned pig. Animal 2008, 2, 1465–1473. [Google Scholar] [CrossRef] [PubMed]
- Lynch, M.B.; Sweeney, T.; Callan, J.J.; O’Sullivan, J.T.; O’Doherty, J.V. The effects of dietary Laminaria-derived laminarin and fucoidan on nutrient digestibility, nitrogen utilization, intestinal microflora and volatile fatty acid concentration in pigs. J. Sci. Food Agric. 2010, 90, 430–437. [Google Scholar] [PubMed]
- Enoki, T.; Sagawa, H.; Tominaga, T.; Nishiyama, E.; Koyama, N.; Sakai, T.; Kato, I. Drugs, Foods or Drinks with the Use of Algae-Derived Physiologically Active Substances. U.S. Patent 20,030,105,029, 5 June 2003. [Google Scholar]
- Fernandez, L.I.; Valiente, O.G.; Mainardi, V.; Bello, J.L.; Velez, H.; Rosado, A. Isolation and characterization of an antitumor active agar-type polysaccharide of Gracilaria dominguensis. Carbohydr. Res. 1989, 190, 77–83. [Google Scholar] [CrossRef]
- Ramnani, P.; Chitarrari, R.; Tuohy, K.; Grant, J.; Hotchkiss, S.; Philp, K.; Campbell, R.; Gill, C.; Rowland, I. In vitro fermentation and prebiotic potential of novel low molecular weight polysaccharides derived from agar and alginate seaweeds. Anaerobe 2012, 18, 1–16. [Google Scholar] [CrossRef]
- Parada, J.L.; de Caire, G.Z.; de Mule, M.C.Z.; de Cano, M.M.S. Lactic acid bacteria growth promoters from Spirulina platensis. Int. J. Food Microbiol. 1998, 45, 222–228. [Google Scholar] [CrossRef]
- Beheshtipour, H.; Mortazavian, A.M.; Haratian, P.; Darani, K.K. Effects of Chlorella vulgaris and Arthrospira platensis addition on viability of probiotic bacteria in yogurt and its biochemical properties. Eur. J. Food Res. Technol. 2012, 235, 719–728. [Google Scholar] [CrossRef]
- Nuño, K.; Villaruel-López, A.; Puella-Pérez, A.M.; Romero-Velarde, E.; Puela-Mora, A.G.; Ascencio, F. Effects of the marine microalgae Isochrysis galbana and Nannochloropsis oculata in diabetic rats. J. Funct. Foods 2013, 5, 106–115. [Google Scholar] [CrossRef]
- Tokita, Y.; Nakajima, K.; Mochida, H.; Iha, M.; Nagamine, T. Development of a fucoidan-specific antibody and measurement of fucoidan in serum and urine by sandwich ELISA. Biosci. Biotechnol. Biochem. 2010, 74, 350–357. [Google Scholar] [CrossRef] [PubMed]
- Irhimeh, M.R.; Fitton, J.H.; Lowenthal, R.M.; Kongtawelert, P. A quantitative method to detect fucoidan in human plasma using a novel antibody. Methods Find. Exp. Clin. Pharmacol. 2005, 27, 705–711. [Google Scholar] [CrossRef] [PubMed]
- Kuda, T.; Goto, H.; Yokoyama, M.; Fujii, T. Fermentable dietary fiber in dried products of brown algae and their effects on caecal microflora and levels of plasma lipids in rats. Fish. Sci. 1998, 64, 582–588. [Google Scholar]
- Sugano, Y.; Terada, I.; Arita, M.; Noma, M.; Matsumoto, T. Purification and characterization of a new agarase from a marine bacterium, Vibrio sp. strain JT0107. Appl. Environ. Microbiol. 1993, 59, 1549–1554. [Google Scholar] [PubMed]
- Kuda, T.; Enomoto, T.; Yano, T. Effects of two storage β-1,3-glucans, laminaran from Eicenia bicyclis and paramylon from Euglena gracilis, on cecal environment and plasma lipid levels in rats. J. Funct. Foods 2009, 1, 399–404. [Google Scholar] [CrossRef]
- Janczyk, P.; Pieper, R.; Smidt, H.; Souffrant, W.B. Effect of alginate and inulin on intestinal microbial ecology of weanling pigs reared under different husbandry conditions. FEMS Microbiol. Ecol. 2010, 72, 132–142. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Li, D.; Wang, J.; Wu, H.; Xia, X.; Bi, W.; Guan, H.; Zhang, L. Effects of polymannuronate on performance, antioxidant capacity, immune status, cecal microflora, and volatile fatty acids in broiler chickens. Poult. Sci. 2015, 94, 345–352. [Google Scholar] [CrossRef] [PubMed]
- Gawronski, M.; Conrad, H.; Springer, T.; Stahmann, K.P. Conformational changes of the polysaccharide Cinerean in aqueous solution. Macromolecules 1996, 29, 7820–7825. [Google Scholar] [CrossRef]
- Yangilar, F. The application of dietary fibre in food industry: Structural features, effects on health and definition, obtaining and analysis of dietary fibre: A review. J. Food Nutr. Res. 2013, 1, 13–23. [Google Scholar]
- Raposo, M.F.J.; Morais, R.M.S.C.; Morais, A.M.M.B. Health applications of bioactive compounds from marine microalgae. Life Sci. 2013, 93, 479–486. [Google Scholar] [CrossRef] [PubMed]
- Shenderov, B.A. Medical microbial ecology and functional foods. In Prebiotics and Functional Foods; GRANT Publ.: Moscow, Russia, 2001; Volume 3. (In Russian) [Google Scholar]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
De Jesus Raposo, M.F.; De Morais, A.M.M.B.; De Morais, R.M.S.C. Emergent Sources of Prebiotics: Seaweeds and Microalgae. Mar. Drugs 2016, 14, 27. https://doi.org/10.3390/md14020027
De Jesus Raposo MF, De Morais AMMB, De Morais RMSC. Emergent Sources of Prebiotics: Seaweeds and Microalgae. Marine Drugs. 2016; 14(2):27. https://doi.org/10.3390/md14020027
Chicago/Turabian StyleDe Jesus Raposo, Maria Filomena, Alcina Maria Miranda Bernardo De Morais, and Rui Manuel Santos Costa De Morais. 2016. "Emergent Sources of Prebiotics: Seaweeds and Microalgae" Marine Drugs 14, no. 2: 27. https://doi.org/10.3390/md14020027
APA StyleDe Jesus Raposo, M. F., De Morais, A. M. M. B., & De Morais, R. M. S. C. (2016). Emergent Sources of Prebiotics: Seaweeds and Microalgae. Marine Drugs, 14(2), 27. https://doi.org/10.3390/md14020027









