Abstract
Background and Objectives: Scant data regarding early post-COVID-19 effects are available, especially in younger people. Therefore, the objective of this study was to explore the early clinical impacts of post-COVID-19 pneumonia, comparing severe and non-severe patients. Materials and Methods: A cross-sectional study was conducted in adult patients admitted with COVID-19 pneumonia from April to May 2021. Demographic data, symptoms and signs, quality of life, Hospital Anxiety and Depression Scale (HADS), chest radiograph (CXR), pulmonary function tests (spirometry, impulse oscillometry), fractional exhaled nitric oxide (FeNO), and exercise capacity were assessed one month after hospital discharge. Twenty-five healthy control subjects that were age- and gender-matched were recruited for comparisons. Results: One hundred and five patients, with a mean age of 35.6 ± 15.8 years and 54 (51.4%) males, participated and were categorized into the non-severe pneumonia (N = 68) and severe pneumonia groups (N = 37). At a one-month follow-up visit (the time from the onset of the disease symptoms = 45.4 ± 5.9 days), the severe group had more cough, fatigue, and skin rash with higher dyspnea scale, more residual CXR lesions, and lower quality of life scores. Forced vital capacity (FVC) was lower in the severe group (88.3% of predicted value) and non-severe group (94.6% of predicted value) than in the healthy controls (p = 0.001). The six-minute walk distance was significantly lower in the non-severe group, at 79.2 m, and in the severe group, at 103.8 m, than in the healthy control subjects (p < 0.001). Conclusions: Adult patients with COVID-19, especially those with clinically severe pneumonia, still had residual symptoms and chest radiographic abnormalities, together with poorer quality of life and lower exercise capacity, one month after hospital discharge.
1. Introduction
On 31 December 2019, cases of pneumonia caused by novel coronavirus SARS-CoV-2 (COVID-19 pneumonia) were reported from Wuhan city Hubei Province of China [1]. This virus outbreak spread to other countries, affecting nearly 200 million people, and was responsible for over 4 million deaths worldwide as of July 2021 [2]. In Thailand, 597,287 people were infected, and 4857 people died by the end of July 2021 [3]. In April 2021, phase 3 of the COVID-19 outbreak began in Chiang Mai, Thailand, especially among young adults, and infected more than one hundred people per day. At the end of May 2021, there were 4068 cases of COVID-19 in Chiang Mai [3].
The respiratory tract is the most common site of COVID-19 infection, with common symptoms including fever, cough, sputum production, fatigue, shortness of breath, myalgia, sore throat, rhinorrhea, and headache [4,5]. The World Health Organization (WHO) categorizes the clinical severity of COVID-19 as asymptomatic, mild, moderate (non-severe pneumonia), severe (severe pneumonia), and critical disease [6]. In cases of COVID-19 pneumonia, most of the chest radiographs (CXR) have shown ground-glass opacification (GGO), consolidation, or combination with bilateral lower lobe involvement [7].
Cohort studies in patients who have recovered from COVID-19 pneumonia have shown reduced health-related quality of life (HR-QoL) in all domains at two months after hospital discharge [8], with reduced physical activity and exercise performance and mild depression and anxiety at six weeks after hospital discharge [9]. These abnormalities might be related to abnormal lung function and might affect their performance [10]. Studies of CXR findings showed that many patients had imaging abnormalities after hospital discharge, such as residual GGO and pulmonary fibrosis [8,11]. The post-COVID-19 sequelae can affect various organs and can be explained by ongoing chronic inflammation and tissue damage [12]. However, patients with COVID-19 pneumonia in our setting were younger than in previous studies [8,9,11]. Therefore, we sought to explore the early impacts of post-COVID-19 pneumonia, including clinical manifestations, quality of life, mood disorders, pulmonary function tests, exercise capacity, FeNO, and CXR findings, among severe and non-severe COVID-19 pneumonia patients.
2. Materials and Methods
2.1. Study Design
This cross-sectional study was approved by the Research Ethics Committee, Faculty of Medicine, Chiang Mai University (Study code: MED-2564-08109, date of approval: 3 May 2021) and filed under the Clinical Trials Registry (Study ID: TCTR20210827005, date of approval: 27 August 2021) in compliance with the Declaration of Helsinki. Written informed consent was obtained from all participants. Data collection was conducted in subjects aged over 18 years old, with COVID-19 pneumonia, who were admitted during April–May 2021 and followed up one month after discharge from Maharaj Nakorn Chiang Mai Hospital, Chiang Mai, Thailand. Subjects who were unable to understand the Thai language were excluded. COVID-19 pneumonia was diagnosed by clinical symptoms and signs, pulmonary infiltration on CXR, and confirmed by positive reverse transcription polymerase chain reaction (RT-PCR). These patients were usually treated in our hospital for two weeks or until clinical improvement according to the current clinical practice guidelines of the Ministry of Public Health, Thailand [13]. Baseline demographics, patients’ medical records, and CXR during hospitalization were reviewed by radiologists. Symptoms, HR-QoL questionnaires, including the 36-item Short-Form Health Survey (SF-36) and Euro Quality of Life—5 Dimensions—5 Levels (EQ-5D-5L), Hospital Anxiety and Depression Scale (HADS), pulmonary function tests, exercise capacity, FeNO, and CXR were assessed at the follow-up visit.
2.2. Data Collection
Clinical manifestations were assessed on various aspects including clinical symptoms and physiological signs, quality of life (SF-36 and EQ-5D-5L), and major mood disorders (anxiety and depression). The SF-36 questionnaire measures general health status and comprises 36 questions that cover eight domains: physical functioning, role limitations due to physical health problems, role limitations due to emotional problems, vitality (energy and fatigue), emotional well-being, social functioning, bodily pain, and general health perceptions. Each domain has a score ranging from 0 to 100, where a higher score indicates better health [14].
The EQ-5D-5L questionnaire measures health status and consists of the EQ-5D descriptive system and EQ visual analog scale (VAS). The EQ-5D descriptive system comprises five dimensions: mobility, self-care, usual activities, pain/discomfort, and anxiety/depression, where each dimension has five levels, from no problems to slight, moderate, severe, and extreme problems. The score ranges from 0 to 1, in which a score close to 1 shows better quality of life. EQ-VAS records the subject’s self-rated health on a vertical scale from 0 to 100, with the endpoints labeled “The best health you can imagine” and “The worst health you can imagine” [15].
The Hospital Anxiety and Depression Scale (HADS) questionnaire measures psychological distress resulting from a disease and contains 14 items with two subscales: anxiety and depression. Each item has a score of 0–3, giving a maximum score of 21 for anxiety and depression. A score of 11 or higher on either subscale is considered to indicate the probable presence of anxiety or depression, respectively [16].
Chest radiograph was evaluated and scored by radiologists. Several studies have demonstrated the association of scores and clinical manifestations, rate of ICU admission, and death [17,18]. The scoring method in this study was modified and adjusted by dividing the bilateral lung into three zones: the upper lung zone (area above aortic arch), the middle lung zone (area between aortic arch and the inferior margin of the left pulmonary hilum), and the lower lung zone (area below left hilum). The total score of six areas was a summation of each lung zone that was calculated by the multiplication point of the involved area with a density of opacity (0 point—lung involvement of 0%; 1, 2, 3, and 4 points—lung involvement in the range of 1–25%, 26–50%, 51–75%, and 76–100%, respectively) and density of opacity (0 points—no opacity, 1 point—ground-glass opacity, and 2 points—consolidation). The total scores were summarized and ranged from 0 to 48 [19,20].
All subjects were assessed for pre-bronchodilator pulmonary function tests including impulse oscillometry (IOS) and spirometry. IOS was performed before spirometry. IOS and spirometry were performed using combined IOS and spirometry equipment (MostGraph-02; Chest M.I., Co Ltd., Tokyo, Japan).
For IOS measurement, the subjects were asked to perform tidal breathing for 30–40 s via a mouthpiece that was connected to a loudspeaker that generated pressure oscillations composed of multiple frequencies. A minimum of three tests was performed, following the European Respiratory Society (ERS) standard [21]. The average values from three IOS measurements were recorded. We collected the following IOS parameters: airway resistance including resistance at 5 Hz (R5), resistance at 20 Hz (R20), heterogeneity of resistance (R5–R20), and airway reactance including reactance at 5 Hz (X5); resonant frequency (Fres); and area under reactance curve between 5 Hz and Fres (AX) [16]. The cut-off point of R5–R20 0.1 kPa/L/s or higher was defined as the presence of small airway disorder [22].
For spirometry assessment, a minimum of three acceptable tests was performed, following the American Thoracic Society/European Respiratory Society (ATS/ERS) guidelines [23]. The spirometry parameters, including forced vital capacity (FVC), forced expiratory volume in the first second (FEV1), the ratio of FEV1/FVC, and forced expiratory flow at 25–75% of FVC (FEF25–75%), were measured. Predicted values were calculated using the Global Lung Function Initiative (GLI) reference equation (Southeast Asian population) [24].
The FeNO was performed before IOS and spirometry using FeNO equipment (NIOX VERO® Circassia Inc., Morrisville, NC, USA). The FeNO was performed to evaluate airway inflammation according to the ATS/ERS guidelines [25]. A FeNO value higher than 25 ppb indicated a high possibility of eosinophilic airway inflammation [26].
The six-minute walk test (6-MWT) is a simple practical test measuring the distance that subjects can quickly walk in six minutes. The 6-MWT was performed following the instructions of the ATS [27]. The exercise desaturation was classified as a decrease of 3% or higher in oxygen saturation via pulse oximetry (SpO2) between resting and post 6-MWT [28].
Twenty-five age- and gender-matched healthy control subjects with no history of COVID-19 or other respiratory diseases were recruited to this study. Demographic data, symptoms, and signs, CXR, SF-36, ED-5Q-5L, HADS, IOS, spirometry, FeNO, and six-minute walking distance (6-MWD) were collected for comparison with COVID-19 pneumonia subjects.
After obtaining the consent of the ethics committee to record the data from all subjects, a database using Microsoft Excel was organized. In this database, the personal data of the patients were adequately encoded to guarantee data protection.
2.3. Study Size Estimation
Sample size calculation was based on the mean and SD of 6-MWD at one month after discharge between severe COVID-19 and non-severe COVID-19 in the previous study [11]. The means and SD of 6-MWD in severe COVID-19 and non-severe COVID-19 was 517.43 ± 44.55 m and 573.52 ± 38.38 m, respectively. We needed to study at least 20 subjects, 10 non-severe COVID-19 and 10 severe COVID-19, to be able to reject the null hypothesis that the population means of the severe and non-severe groups were equal with probability (power) of 0.8. The type I error probability associated with this test of the null hypothesis was 0.05.
2.4. Statistical Analysis
Continuous data were expressed as mean and standard deviation (SD), or median and interquartile range (IQR). Analysis of variance (ANOVA) with Bonferroni adjustment was used for analyzing clinical characteristics across the three groups for continuous data. The Kruskal–Wallis test was used for analyzing clinical characteristics across the three groups for non-parametric data. The Student t-test or Mann–Whitney U test was used for analyzing clinical characteristics between severe and non-severe groups for parametric and non-parametric data, respectively. Categorical data were expressed as frequencies and percentages. Comparison of categorical data between groups was performed using Fisher’s exact test. Gaussian regression adjusted for confounding factors including age, body mass index (BMI), and gender was used for analyzing differences in mean values of IOS parameters and 6-MWD across the three groups. A generalized linear model adjusted for confounding factors including age, BMI, and gender was used for analyzing differences in the proportion of exercise desaturation in 6-MWT between severe and non-severe groups. A p-value less than 0.05 was considered statistically significant. All statistical analyses were performed using STATA version 16 (StataCorp, College Station, TX, USA).
3. Results
During the phase 3 (April–May 2021) outbreak of COVID-19 in Chiang Mai, 193 patients with COVID-19 pneumonia were admitted to our hospital and ten patients (5.2%) died during hospitalization. One hundred and five discharged patients came for follow-up and participated in this study, with a mean age of 35.6 ± 15.7 years and with 54 males (51.4%). These patients were categorized into two groups: the non-severe pneumonia group, N = 68 (pneumonia with no oxygen therapy, N = 59, or treated with low-flow oxygen cannula, N = 9), and severe pneumonia group, N = 37 (pneumonia treated with high-flow nasal cannula oxygen (HFNC), N = 35, or mechanical ventilator, N = 2). Twenty-five age- and gender-matched healthy control subjects were also enrolled (Figure 1). The demographic data of the study population are shown in Table 1. The non-severe pneumonia patients were younger than the severe pneumonia patients, while the severe pneumonia patients had a higher BMI and had more cardiovascular co-morbidities, where hypertension was the most common (N = 16, 43.2%), compared to the non-severe pneumonia patients. More data are shown in Table 1.
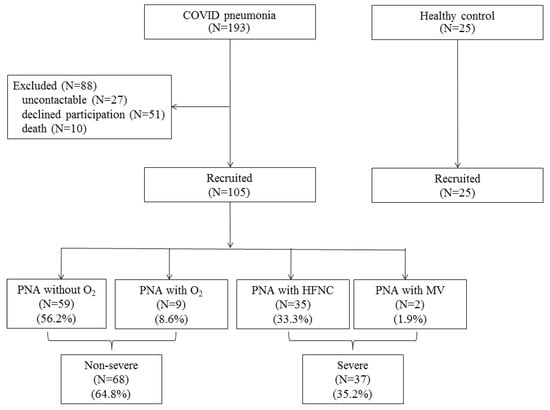
Figure 1.
Flow chart of the study population. Abbreviations: PNA, pneumonia; HFNC, high-flow nasal cannula oxygen; MV, mechanical ventilation.

Table 1.
Demographic data of study population (n = 130).
The data collected during hospitalization for COVID-19 pneumonia are shown in Table 2. The severe group had greater clinical severity, with a higher neutrophil–lymphocyte ratio (NLR), platelet–lymphocyte ratio (PLR), and c-reactive protein (CRP) level than the non-severe group. Most of the CXR pattern was GGO (N = 71, 67.6%) with multi-lobar involvement (N = 89, 84.8%) and lower lung zone predominance (N = 102, 97.1%). The CXR score in the severe group was higher, with more consolidation and mixed with GGO than the non-severe group. Remdesivir, empirical antibiotics, systemic corticosteroids, and more aggressive treatments, such as tocilizumab and hemoperfusion, were more frequently prescribed in the severe group. More data are shown in Table 2.

Table 2.
Data during hospitalization (n = 105).
At the one-month follow-up visit, the mean time of the onset of the disease symptoms to follow-up visit was 45.4 ± 5.9 days. Fifty subjects (47.6%) had at least one symptom, with a higher frequency in the severe group (N = 24, 64.9%). In the severe group, cough, fatigue, and skin rash were the most common symptoms, with a higher dyspnea scale and lower oxygen saturation (Table 3).

Table 3.
Symptoms and vital signs at one-month follow-up visit.
Health-related quality of life, measured by EQ-5D-5L and SF-36, was also significantly lower in the severe group compared to the non-severe and healthy control groups. In the severe group, quality of life was significantly worse in all domains. Only a few patients (N = 7, 6.7%) had anxiety and/or depression but there was no significant difference between groups. More data are shown in Table 4. In the severe group, CXR revealed more residual lesions with a higher score than the in non-severe group, while NLR and PLR decreased to values closer to the non-severe and healthy control groups (Table 5). Pulmonary embolism was diagnosed in two patients in the severe group during follow-up.

Table 4.
Dyspnea, quality of life, and mood disorders at one-month follow-up visit.

Table 5.
Laboratory results and chest radiograph at one-month follow-up visit.
Pulmonary function tests including spirometry and IOS, FeNO, and 6-MWT were assessed in 85 patients (31 patients in the severe group, 29 patients in the non-severe group, and 25 healthy control subjects). The %predicted of FVC was significantly lower in the severe and non-severe groups, and the %predicted of FEV1 was also significantly lower in the severe group compared to the healthy controls. Only one patient in the severe group had an obstructive defect defined by FEV1/FVC < LLN. The FeNO level was not significantly different across the three groups. The proportion of FeNO > 25 ppb was more frequent in the non-severe group compared to the severe and healthy control groups, but not statistically significant. More data are shown in Table 6.

Table 6.
Spirometry and FeNO at one-month follow-up visit (n = 85).
IOS parameters were also not significantly different across the three groups, except for R20, which was significantly lower in the non-severe group than healthy controls. Ten patients in the severe group (32.3%), eight patients in the non-severe group (27.6%), and nine healthy control subjects (36.0%) had evidence of small airway disorder defined by R5-R20 > 0.1 kPa/L/s. More data are shown in Table 7.

Table 7.
Impulse oscillometry at one-month follow-up visit (n = 85).
Subjects in the severe and non-severe groups had significantly lower 6-MWD compared to healthy controls. Subjects in the severe group had more oxygen desaturation during 6-MWT than the non-severe group, but this was not statistically significant (adjusted risk ratio = 3.31 (95%CI: 0.38, 28.96)). More data are shown in Table 8.

Table 8.
Exercise capacity and exercise desaturation test at one-month follow-up visit (N = 85).
4. Discussion
We studied the impacts of COVID-19 pneumonia on clinical manifestations including HR-QoL and psychological problems, pulmonary function, exercise capacity, FeNO, and chest radiograph at a one-month follow-up visit after hospitalization for COVID-19 pneumonia and found that patients with COVID-19 pneumonia, especially clinically severe patients, still had more symptoms, including dyspnea, worse quality of life, lower exercise capacity, and more residual CXR lesions. Although the spirometric parameters of these post-COVID-19 patients were within normal limits, there was a trend of lower lung volume with increased severity of pneumonia, similar to previous studies [8,9,11].
This study found that cough, fatigue, and skin rash were significantly more frequent in the severe COVID-19 pneumonia group than the non-severe group. Cough and fatigue may be affected by the residual inflammatory process. During the acute phase of COVID-19, our patients with severe pneumonia had more lung inflammation, demonstrated by higher CXR scores, as well as more systemic inflammation, demonstrated by increased inflammatory mediators and cytokines (cytokine storm), which presented with lymphopenia, high NLR, PLR, and serum CRP, similar to previous studies [29,30,31]. These findings are associated with disease severity and poor outcomes [32,33]. The greater lung and systemic inflammation might need more time to resolve than that in non-severe patients [29]. Skin manifestations such as maculopapular rash and urticaria were reported during active COVID-19 and after hospital discharge [34]. However, the symptoms described in this study were assessed using a patient-reported questionnaire, so we did not explore the details of the skin rash and its relationship with clinical severity during admission.
Two thirds of patients with community-acquired pneumonia had complete CXR resolution at four weeks and the rate of the resolution was inversely correlated with age and number of lobes involved [35], similar to our findings that two thirds of patients with COVID-19 pneumonia had complete CXR resolution at one month. However, CXR in the severe group showed more frequent residual lesions (59.5% vs. 10.3% in the severe and non-severe groups, respectively), among which most of them (19 of 22) were GGO, while others were mixed GGO and consolidation. Only a few patients (3 of 37, 8.1%) were stable or experienced progression, which might be explained by their age and the greater extent of lung inflammation (multi-lobar involvement). These residual lesions might affect respiratory symptoms such as cough and dyspnea, lower lung volume measured by spirometry, lower oxygen saturation, lower physical activity, and poorer HR-QoL.
Sixty patients (57.1%) were evaluated for pulmonary function by spirometry, small airway function by IOS, airway inflammation by FeNO, and exercise capacity by 6-MWT. We found that the value of each spirometric parameter was within the normal limits and not significantly different between the severe and non-severe groups; however, there was a trend of lower lung volume and lower exercise capacity by 6-MWD and exercise desaturation with increased severity of pneumonia, as found in other studies [8,11]. These abnormalities of pulmonary function after COVID-19 pneumonia, including low lung volume, impaired diffusion capacity, and reduced exercise capacity, might be caused by the residual process of inflammation and were correlated with disease severity. These affected quality of life and could improve thereafter [8,10,36].
According to the COVID-19 pathology of the main bronchi and bronchiolar branches, which showed mild, non-specific focal squamous metaplasia and mild transmural lymphocytic and monocytic infiltrates [37], we found evidence of small airway disorder by IOS in thirty percent of our patients (18 of 60), similar to a study in China that showed increased airway resistance (R5 and R20) at one-month follow-up [11]. However, the evidence of small airway disorder between patients and healthy controls was not different, which might be explained by many factors, such as occult airway diseases or the effect of smoking or air pollutants. Eosinophilic airway inflammation also might not be the cause of small airway disorder, as low FeNO levels were described in a study from Finland [38].
The HR-QoL scores, both physical and mental components, were poorer especially in the severe pneumonia group, which may be attributed to the severity of disease and management, which required more aggressive treatment [39] together with the patient’s isolation from their family and society. Although many factors are associated with worsened psychological status during the COVID-19 outbreak, such as lockdown conditions, social restrictions, fear of contamination, and uncertainty about the pandemic [40], only a few patients in our study had anxiety and/or depression, which might be explained by the lower incidence of COVID-19 in Chiang Mai during the time of assessment (late May–June 2021).
The strength of our study is its value as the first study that evaluates the residual symptoms, pulmonary function including small airway function, exercise capacity, and radiograph, together with quality of life and mood disorders, at one month after hospital discharge for COVID-19 pneumonia in Chiang Mai, Thailand. Healthy controls were also enrolled for comparison with severe and non-severe COVID-19 pneumonia. However, this study has some limitations. Firstly, our study included a single-center cohort. The results may differ at other clinical sites. Secondly, COVID-19 pneumonia during the study period was mostly caused by alpha variants (B.1.1.7), which have different clinical manifestations to other variants of more recent concern in Thailand (delta variant, B.1.617.2) [2,41]. Thirdly, some clinical features, pulmonary function, and abnormal chest radiographs require long-term follow-up to clarify the effect of COVID-19 pneumonia in Thailand.
5. Conclusions
Adult patients with COVID-19, especially those with clinically severe pneumonia, still have residual symptoms and chest radiographic abnormalities, together with worse quality of life and exercise capacity, at one month after hospital discharge.
Author Contributions
Conceptualization, N.N., A.D., W.C., C.B., T.T., A.L., P.T., K.T. and C.P.; Methodology, N.N., A.D., W.C., P.D. and C.P.; Software, W.C.; Validation, A.D., W.C. and C.P.; Formal Analysis, A.D. and W.C.; Investigation, N.N., A.D., W.C., P.D., C.L., J.E., Y.W., T.S. and C.P.; Resources, W.C., C.L. and C.P.; Data Curation, A.D., W.C., J.E., and C.P.; Writing—Original Draft Preparation, N.N., A.D. and W.C.; Writing—Review and Editing, N.N., A.D., W.C., P.D., J.E. and C.P.; Visualization, A.D., W.C., C.L. and C.P.; Supervision, C.L. and C.P.; Project Administration, A.D., W.C., T.T. and C.P.; Funding Acquisition, W.C. and C.P. All authors have read and agreed to the published version of the manuscript.
Funding
This study was funded by the Faculty of Medicine, Chiang Mai University Research Fund under grant No. 157/2564.
Institutional Review Board Statement
This study was conducted in accordance with the Declaration of Helsinki and approved by the Research Ethics Committee, Faculty of Medicine, Chiang Mai University (Study code: MED-2564-08109, date of approval: 3 May 2021).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Acknowledgments
The authors would like to thank all subjects who kindly participated in this study. The authors acknowledge the physicians and nurses of the Division of Pulmonary, Critical Care and Allergy, Department of Internal Medicine, Faculty of Medicine, Chiang Mai University for their contribution to this trial. The authors would like to thank Ruth Leatherman, Research Administration Section, Faculty of Medicine, Chiang Mai University for the native English proofreading.
Conflicts of Interest
The authors have no potential conflict of interest.
References
- World Health Organization. Novel Coronavirus (2019-nCoV) Situation Report-1. 21 January 2020. Available online: https://apps.who.int/iris/handle/10665/330760 (accessed on 15 May 2021).
- World Health Organization. COVID-19 Weekly Epidemiological Update. Edition 50 Published 27 July 2021. 2021. Available online: https://www.who.int/publications/m/item/weekly-epidemiological-update-on-covid-19 (accessed on 5 August 2021).
- Corona Virus Disease (COVID-19 in Thailand). Available online: https://ddc.moph.go.th/viralpneumonia/index.php (accessed on 5 August 2021).
- Guan, W.J.; Ni, Z.Y.; Hu, Y.; Liang, W.H.; Ou, C.Q.; He, J.X.; Liu, L.; Shan, H.; Lei, C.L.; Hui, D.S.C.; et al. China Medical Treatment Expert Group for COVID-19. Clinical Characteristics of Coronavirus Disease 2019 in China. N. Engl. J. Med. 2020, 382, 1708–1720. [Google Scholar] [CrossRef] [PubMed]
- Pongpirul, W.A.; Wiboonchutikul, S.; Charoenpong, L.; Panitantum, N.; Vachiraphan, A.; Uttayamakul, S.; Pongpirul, K.; Manosuthi, W.; Prasithsirikul, W. Clinical course and potential predictive factors for pneumonia of adult patients with Coronavirus Disease 2019 (COVID-19): A retrospective observational analysis of 193 confirmed cases in Thailand. PLoS Negl. Trop. Dis. 2020, 14, e0008806. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. COVID-19 Clinical Management-Living Guidance 25 January 2021. Available online: https://apps.who.int/iris/handle/10665/338882 (accessed on 15 May 2021).
- Zhang, Q.; Wang, Z.; Lv, Y.; Zhao, J.; Dang, Q.; Xu, D.; Zhao, D.; Liu, H.; Wang, Z.; Zhao, X.; et al. Clinical features and prognostic factors of patients with COVID-19 in Henan Province, China. Hum. Cell. 2021, 34, 419–435. [Google Scholar] [CrossRef] [PubMed]
- Strumiliene, E.; Zeleckiene, I.; Bliudzius, R.; Samuilis, A.; Zvirblis, T.; Zablockiene, B.; Strumila, A.; Gruslys, V.; Malinauskiene, L.; Kasiulevicius, V.; et al. Follow-Up Analysis of Pulmonary Function, Exercise Capacity, Radiological Changes, and Quality of Life Two Months after Recovery from SARS-CoV-2 Pneumonia. Medicina 2021, 57, 568. [Google Scholar] [CrossRef] [PubMed]
- Daher, A.; Balfanz, P.; Cornelissen, C.; Müller, A.; Bergs, I.; Marx, N.; Müller-Wieland, D.; Hartmann, B.; Dreher, M.; Müller, T. Follow up of patients with severe coronavirus disease 2019 (COVID-19): Pulmonary and extrapulmonary disease sequelae. Respir. Med. 2020, 174, 106197. [Google Scholar] [CrossRef] [PubMed]
- Van der Sar-van der Brugge, S.; Talman, S.; Boonman-de Winter, L.; de Mol, M.; Hoefman, E.; van Etten, R.W.; De Backer, I.C. Pulmonary function and health-related quality of life after COVID-19 pneumonia. Respir. Med. 2021, 176, 106272. [Google Scholar] [CrossRef]
- Huang, Y.; Tan, C.; Wu, J.; Chen, M.; Wang, Z.; Luo, L.; Zhou, X.; Liu, X.; Huang, X.; Yuan, S.; et al. Impact of coronavirus disease 2019 on pulmonary function in early convalescence phase. Respir. Res. 2020, 21, 163. [Google Scholar] [CrossRef]
- Crook, H.; Raza, S.; Nowell, J.; Young, M.; Edison, P. Long covid-mechanisms, risk factors, and management. BMJ 2021, 374, n1648. [Google Scholar] [CrossRef]
- Department of Disease Control, Ministry of Public Health, Thailand. Guidelines on Clinical Practice, Diagnosis, Treatment, and Prevention of Healthcare-Associated Infection for COVID-19. Available online: https://ddc.moph.go.th/viralpneumonia/eng/guideline_hcw.php (accessed on 10 December 2021).
- Lim, L.L.; Seubsman, S.A.; Sleigh, A. Thai SF-36 health survey: Tests of data quality, scaling assumptions, reliability and validity in healthy men and women. Health Qual. Life Outcomes 2008, 6, 52. [Google Scholar] [CrossRef] [Green Version]
- Devlin, N.J.; Krabbe, P.F. The development of new research methods for the valuation of EQ-5D-5L. Eur. J. Health Econ. 2013, 14, S1–S3. [Google Scholar] [CrossRef] [Green Version]
- Snaith, R.P. The Hospital Anxiety And Depression Scale. Health Qual. Life Outcomes 2003, 1, 29. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Toussie, D.; Voutsinas, N.; Finkelstein, M.; Cedillo, M.A.; Manna, S.; Maron, S.Z.; Jacobi, A.; Chung, M.; Bernheim, A.; Eber, C.; et al. Clinical and Chest Radiography Features Determine Patient Outcomes in Young and Middle-aged Adults with COVID-19. Radiology 2020, 297, E197–E206. [Google Scholar] [CrossRef] [PubMed]
- Galloway, J.B.; Norton, S.; Barker, R.D.; Brookes, A.; Carey, I.; Clarke, B.D.; Jina, R.; Reid, C.; Russell, M.D.; Sneep, R.; et al. A clinical risk score to identify patients with COVID-19 at high risk of critical care admission or death: An observational cohort study. J. Infect. 2020, 81, 282–288. [Google Scholar] [CrossRef]
- Monaco, C.G.; Zaottini, F.; Schiaffino, S.; Villa, A.; Della Pepa, G.; Carbonaro, L.A.; Menicagli, L.; Cozzi, A.; Carriero, S.; Arpaia, F.; et al. Chest X-ray severity score in COVID-19 patients on emergency department admission: A two-centre study. Eur. Radiol. Exp. 2020, 4, 68. [Google Scholar] [CrossRef]
- Litmanovich, D.E.; Chung, M.; Kirkbride, R.R.; Kicska, G.; Kanne, J.P. Review of Chest Radiograph Findings of COVID-19 Pneumonia and Suggested Reporting Language. J. Thorac. Imaging 2020, 35, 354–360. [Google Scholar] [CrossRef]
- King, G.G.; Bates, J.; Berger, K.I.; Calverley, P.; de Melo, P.L.; Dellacà, R.L.; Farré, R.; Hall, G.L.; Ioan, I.; Irvin, C.G.; et al. Technical standards for respiratory oscillometry. Eur. Respir. J. 2020, 55, 1900753. [Google Scholar] [CrossRef]
- Manoharan, A.; Anderson, W.J.; Lipworth, J.; Lipworth, B.J. Assessment of spirometry and impulse oscillometry in relation to asthma control. Lung 2015, 193, 47–51. [Google Scholar] [CrossRef]
- Miller, M.R.; Hankinson, J.; Brusasco, V.; Burgos, F.; Casaburi, R.; Coates, A.; Crapo, R.; Enright, P.; van der Grinten, C.P.; Gustafsson, P.; et al. ATS/ERS Task Force. Standardisation of spirometry. Eur. Respir. J. 2005, 26, 319–338. [Google Scholar] [CrossRef] [Green Version]
- Quanjer, P.H.; Stanojevic, S.; Cole, T.J.; Baur, X.; Hall, G.L.; Culver, B.H.; Enright, P.L.; Hankinson, J.L.; Ip, M.S.; Zheng, J.; et al. ERS Global Lung Function Initiative. Multi-ethnic reference values for spirometry for the 3-95-yr age range: The global lung function 2012 equations. Eur. Respir. J. 2012, 40, 1324–1343. [Google Scholar] [CrossRef]
- American Thoracic Society; European Respiratory Society. ATS/ERS recommendations for standardized procedures for the online and offline measurement of exhaled lower respiratory nitric oxide and nasal nitric oxide, 2005. Am. J. Respir. Crit. Care. Med. 2005, 171, 912–930. [Google Scholar] [CrossRef]
- Bjermer, L.; Alving, K.; Diamant, Z.; Magnussen, H.; Pavord, I.; Piacentini, G.; Price, D.; Roche, N.; Sastre, J.; Thomas, M.; et al. Current evidence and future research needs for FeNO measurement in respiratory diseases. Respir. Med. 2014, 108, 830–841. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- ATS Statement. Guidelines for the six-minute walk test. Am. J. Respir. Crit. Care. Med. 2002, 166, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Kalin, A.; Javid, B.; Knight, M.; Inada-Kim, M.; Greenhalgh, T. Direct and indirect evidence of efficacy and safety of rapid exercise tests for exertional desaturation in COVID-19: A rapid systematic review. Syst. Rev. 2021, 10, 77. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Zheng, K.I.; Liu, S.; Yan, Z.; Xu, C.; Qiao, Z. Plasma CRP level is positively associated with the severity of COVID-19. Ann. Clin. Microbiol. Antimicrob. 2020, 19, 18. [Google Scholar] [CrossRef] [PubMed]
- Xue, G.; Gan, X.; Wu, Z.; Xie, D.; Xiong, Y.; Hua, L.; Zhou, B.; Zhou, N.; Xiang, J.; Li, J. Novel serological biomarkers for inflammation in predicting disease severity in patients with COVID-19. Int. Immunopharmacol. 2020, 89, 107065. [Google Scholar] [CrossRef] [PubMed]
- Yılmaz Demirci, N.; Uğraş Dikmen, A.; Taşçı, C.; Doğan, D.; Arslan, Y.; Öcal, N.; Taşar, M.; Bozlar, U.; Artuk, C.; Yılmaz, G.; et al. Relationship between chest computed tomography findings and clinical conditions of coronavirus disease (COVID-19): A multicentre experience. Int. J. Clin. Pract. 2021, 75, e14459. [Google Scholar] [CrossRef]
- Terpos, E.; Ntanasis-Stathopoulos, I.; Elalamy, I.; Kastritis, E.; Sergentanis, T.N.; Politou, M.; Psaltopoulou, T.; Gerotziafas, G.; Dimopoulos, M.A. Hematological findings and complications of COVID-19. Am. J. Hematol. 2020, 95, 834–847. [Google Scholar] [CrossRef] [Green Version]
- Palladino, M. Complete blood count alterations in COVID-19 patients: A narrative review. Biochem. Med. 2021, 31, 030501. [Google Scholar] [CrossRef]
- Thuangtong, R.; Angkasekwinai, N.; Leeyaphan, C.; Triwongwaranat, D.; Thanomkitti, K.; Munprom, K.; Kulthanan, K. Patient Recovery from COVID-19 Infections: Follow-Up of Hair, Nail, and Cutaneous Manifestations. Biomed. Res. Int. 2021, 2021, 5595016. [Google Scholar] [CrossRef]
- Mittl, R.L., Jr.; Schwab, R.J.; Duchin, J.S.; Goin, J.E.; Albeida, S.M.; Miller, W.T. Radiographic resolution of community-acquired pneumonia. Am. J. Respir. Crit. Care Med. 1994, 149, 630–635. [Google Scholar] [CrossRef]
- Wu, X.; Liu, X.; Zhou, Y.; Yu, H.; Li, R.; Zhan, Q.; Ni, F.; Fang, S.; Lu, Y.; Ding, X.; et al. 3-month, 6-month, 9-month, and 12-month respiratory outcomes in patients following COVID-19-related hospitalisation: A prospective study. Lancet Respir. Med. 2021, 9, 747–754. [Google Scholar] [CrossRef]
- Carsana, L.; Sonzogni, A.; Nasr, A.; Rossi, R.S.; Pellegrinelli, A.; Zerbi, P.; Rech, R.; Colombo, R.; Antinori, S.; Corbellino, M.; et al. Pulmonary post-mortem findings in a series of COVID-19 cases from northern Italy: A two-centre descriptive study. Lancet Infect. Dis. 2020, 20, 1135–1140. [Google Scholar] [CrossRef]
- Lindahl, A.; Reijula, J.; Malmberg, L.P.; Aro, M.; Vasankari, T.; Mäkelä, M.J. Small airway function in Finnish COVID-19 survivors. Resp. Res. 2021, 22, 237. [Google Scholar] [CrossRef] [PubMed]
- Todt, B.C.; Szlejf, C.; Duim, E.; Linhares, A.O.M.; Kogiso, D.; Varela, G.; Campos, B.A.; Baghelli Fonseca, C.M.; Polesso, L.E.; Bordon, I.N.S.; et al. Clinical outcomes and quality of life of COVID-19 survivors: A follow-up of 3 months post hospital discharge. Respir. Med. 2021, 184, 106453. [Google Scholar] [CrossRef] [PubMed]
- Cielo, F.; Ulberg, R.; Giacomo, D.D. Psychological impact of the COVID-19 outbreak on mental health outcomes among youth: A rapid narrative review. Int. J. Environ. Res. Public Health 2021, 18, 6067. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. COVID-19 Weekly Epidemiological Update as of 11 April 2021. Available online: https://www.epid.gov.lk/web/images/pdf/corona_virus_report/sitrep-gl-en-19-04_10_21.pdf (accessed on 15 May 2021).
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).