1. Introduction
Chronic inflammatory dermatoses represent a heterogeneous group of skin disorders that are often characterized by persistent and relapsing inflammation, with complex underlying pathomechanisms. These disorders, which include, e.g., hidradenitis suppurativa (HS), psoriasis, atopic dermatitis (AD), and alopecia areata (AA), are driven by dysregulated immune responses, including Th1 and Th2 pathways [1]. Despite the diversity of these conditions, they share common features such as chronicity, substantial morbidity, and a significant impact on patients’ quality of life. As we launch this Special Issue of Current Issues in Molecular Biology focusing on chronic dermatoses, it is important to examine the current landscape of treatment strategies, from traditional systemic therapies to novel biologics, and to explore future directions in managing these complex conditions.
2. Traditional Systemic Therapies: Limitations and Challenges
For decades, the management of chronic inflammatory dermatoses has relied on the use of traditional systemic immunosuppressive and immunomodulatory drugs. Agents such as methotrexate (MTX), cyclosporine A, dapsone, and glucocorticosteroids (GCs) have been the mainstay of therapy, providing symptomatic relief for many patients. These medications have broad immunosuppressive effects, which can be beneficial in controlling inflammation. For instance, MTX, a folate antagonist, inhibits dihydrofolate reductase and reduces the proliferation of immune cells and keratinocytes, making it a successful therapy for psoriasis [2]. Among patients receiving MTX for 12–16 weeks, 60–70% showed a 75% improvement in the Psoriasis Area and Severity Index (PASI 75) [2]. Similarly, cyclosporine A, a calcineurin inhibitor, suppresses T-cell activation and cytokine production, leading to rapid disease control. It has effectively treated moderate-to-severe cases of AD and psoriasis [3,4]. GCs remain a mainstay in managing various chronic inflammatory dermatoses, including psoriasis, atopic dermatitis, and atopic dermatitis. Their broad-spectrum anti-inflammatory effects result in rapid symptom relief, making them indispensable for acute flares.
Nevertheless, it should be emphasized that despite their widespread use, these medications are not free of side effects. Due to their non-specific nature, they can suppress the immune system, leading to an increased risk of infections and malignancies. Moreover, their efficacy can be variable, with many patients experiencing partial responses or treatment failure. In addition, the chronic nature of these diseases often requires long-term therapy, which can lead to cumulative toxicity. For instance, the prolonged use of cyclosporine A is associated with nephrotoxicity and hypertension, while MTX can lead to hepatotoxicity and bone marrow suppression [5,6]. While highly effective in the short term, GCs are known for their systemic side effects, including osteoporosis, hyperglycemia, and adrenal suppression, when used chronically [7]. As a result, there has been a growing need for more targeted therapies that can offer better efficacy with a more favorable safety profile.
3. The Advent of Biologics: A Paradigm Shift in Treatment
The introduction of biological therapies has revolutionized the management of chronic inflammatory dermatoses, offering a more targeted approach to treatment. Biologics are humanized or human monoclonal antibodies or fusion proteins designed to specifically target key molecules involved in the pathogenesis of chronic inflammatory dermatoses, allowing for more effective disease control with a reduced risk of side effects.
The first biologic approved by the U.S. Food and Drug Administration (FDA) in 1998 for dermatological treatment was etanercept, a TNF-α inhibitor monoclonal antibody for psoriasis [8]. This initiated a new phase in dermatology, where treatment could be customized to target specific immune pathways. Since then, the repertoire of biologics has expanded significantly, offering targeted treatment options that modulate particular components of the immune response [9].
In psoriasis, the efficacy of TNF-α inhibitors (etanercept, infliximab, and adalimumab) is well established, providing rapid and sustained clearance of psoriatic lesions [10]. Ustekinumab, an IL-12/23 inhibitor, further advanced the treatment options by offering an alternative mechanism of action with a favorable safety profile. More recently, IL-17 inhibitors (secukinumab, ixekizumab, and bimekizumab) and IL-23 inhibitors (guselkumab and tildrakizumab) have demonstrated even greater efficacy, with some patients achieving complete or near-complete skin clearance [11].
The approval of dupilumab, an IL-4 receptor alpha antagonist, has marked a significant breakthrough in the treatment of AD [12]. Dupilumab blocks the signaling of both IL-4 and IL-13, which are cytokines with a crucial role in the Th2-mediated immune response characteristic of AD [13]. Additionally, clinical trials and real-world data have shown that dupilumab significantly improves skin clearance and reduces pruritus, with a safety profile superior to traditional systemic therapies [13,14]. Other biologics targeting IL-13 inhibitors (tralokinumab and lebrikizumab) have been recently approved for use in adult patients with moderate-to-severe AD. Clinical trials showed a significant decrease in AD and pruritus severity, as well as an important improvement in patients’ QoL [15].
Hidradenitis suppurativa, a condition notoriously resistant to treatment, saw the approval of adalimumab, a TNF-α inhibitor, as the first FDA-approved biologic for moderate-to-severe cases in 2015 [13,14]. While adalimumab provides relief for many patients, its efficacy is variable, and not all patients achieve significant improvement [16,17]. The FDA’s approval of bimekizumab, a dual IL-17A and IL-17F inhibitor, and secukinumab, an IL-17A inhibitor, has marked a significant milestone in the treatment of HS [18,19]. Both agents have demonstrated promising results in clinical trials, offering a new level of efficacy in reducing the inflammatory lesions and pain associated with HS. These approvals underscore the growing understanding of the pivotal role of IL-17 in the pathogenesis of this disease and represent a significant step forward in providing more effective and targeted treatment options for patients [18,19].
Alopecia areata, traditionally managed with corticosteroids, has seen limited success with biologics. Another significant development was the introduction of Janus kinase (JAK) inhibitors for the treatment of AA. Tofacitinib, the first JAK inhibitor approved by the FDA for dermatological use, has shown promise in treating moderate-to-severe AA [20]. Subsequently, other JAK inhibitors, including baricitinib, ruxolitinib, and ritlecitinib, have been approved for the treatment of moderate-to-severe AA [21].
JAK inhibitors have also shown efficacy in other chronic inflammatory dermatoses. Baricitinib, for example, is approved for the treatment of moderate-to-severe atopic dermatitis, offering another targeted option for patients. The potential of JAK inhibitors extends beyond current indications, with ongoing research exploring their use in psoriasis and HA [22]. The timeline of FDA and EMA acceptance of new drugs in dermatology is illustrated in Figure 1.
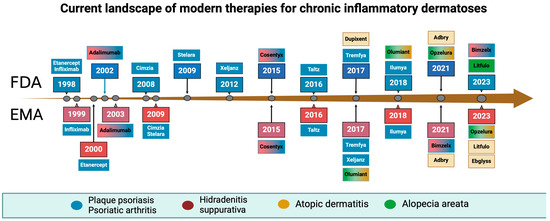
Figure 1.
Timeline of biological drugs and Janus kinase inhibitors acceptance by the Food and Drug Administration and the European Medical Agency. Created with BioRender.com.
The exact dates and indications for those drugs are mentioned in Table 1.

Table 1.
Biologics and Janus kinase inhibitors (JAKi) that are currently available for treating chronic inflammatory dermatoses.
4. Future Directions: Towards Precision Medicine in Dermatology
As we look to the future of chronic inflammatory dermatosis management, the emphasis is moving more towards targeted treatments. Identifying biomarkers that can predict disease severity, treatment effectiveness, and long-term outcomes will be crucial in guiding treatment strategies. The goal is to tailor treatment based on individual patient profiles, which could include genetic, immunological, and environmental factors.
Ongoing research on the molecular mechanisms involved in the pathophysiology of these diseases may yield new therapeutic targets. For instance, the exploration of the microbiome’s role in skin disease could lead to novel interventions aimed at modulating the skin’s microbial environment. Additionally, advances in gene therapy and the use of biologics targeting novel pathways, such as IL-17, IL-23, and IL-31, will continue to expand the therapeutic options.
Long-term studies should also refine the safety and efficacy of existing biologics and JAK inhibitors, providing insights into optimal dosing regimens and combination therapies. As these treatments become more widely used, it is also essential to monitor rare side effects and develop strategies to deal with treatment resistance that may occur with long-term use.
In conclusion, the treatment landscape for chronic inflammatory dermatoses has significantly evolved over the past two decades, with biologics and JAK inhibitors providing new hope for patients who previously had limited options. As we continue to unravel the molecular foundations of these complex diseases, the future of dermatological treatment promises to be increasingly personalized, with the potential to improve outcomes and quality of life for patients worldwide.
Author Contributions
Conceptualization, P.K.K. and A.S.; methodology, P.K.K. and A.S.; investigation, P.K.K. and A.S.; data curation, P.K.K. and A.S.; writing—original draft preparation, P.K.K. and A.S.; writing—review and editing, P.K.K. and A.S.; visualization, P.K.K. and A.S.; supervision, P.K.K.; project administration, P.K.K. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Biedermann, T.; Röcken, M.; Carballido, J.M. TH1 and TH2 lymphocyte development and regulation of TH cell-mediated immune responses of the skin. J. Investig. Dermatol. Symp. Proc. 2004, 9, 5–14. [Google Scholar] [CrossRef]
- Boffa, M.J.; Chalmers, R.J. Methotrexate for psoriasis. Clin. Exp. Dermatol. 1996, 21, 399–408. [Google Scholar] [CrossRef]
- Mozzanica, N.; Pigatto, P.D.; Finzi, A.F. Cyclosporin in psoriasis: Pathophysiology and experimental data. Dermatology 1993, 187 (Suppl. S1), 3–7. [Google Scholar] [CrossRef]
- Wang, C.; Lin, A. Efficacy of topical calcineurin inhibitors in psoriasis. J. Cutan. Med. Surg. 2014, 18, 8–14. [Google Scholar] [CrossRef]
- Di Martino, V.; Verhoeven, D.W.; Verhoeven, F.; Aubin, F.; Avouac, J.; Vuitton, L.; Lioté, F.; Thévenot, T.; Wendling, D. Busting the myth of methotrexate chronic hepatotoxicity. Nat. Rev. Rheumatol. 2022, 19, 96–110. [Google Scholar] [CrossRef]
- Brouhard, B.H.; Graham, R.M. Cyclosporine: Mechanisms of action and toxicity. Clevel. Clin. J. Med. 1994, 61, 308–313. [Google Scholar] [CrossRef]
- Oray, M.; Samra, K.A.; Ebrahimiadib, N.; Meese, H.; Foster, C.S. Long-term side effects of glucocorticoids. Expert Opin. Drug Saf. 2016, 15, 457–465. [Google Scholar] [CrossRef]
- Chamian, F.; Lowes, M.A.; Lin, S.-L.; Lee, E.; Kikuchi, T.; Gilleaudeau, P.; Sullivan-Whalen, M.; Cardinale, I.; Khatcherian, A.; Novitskaya, I.; et al. Alefacept reduces infiltrating T cells, activated dendritic cells, and inflammatory genes in psoriasis vulgaris. Proc. Natl. Acad. Sci. USA 2005, 102, 2075–2080. [Google Scholar] [CrossRef]
- Rodgers, M.; Epstein, D.; Bojke, L.; Yang, H.; Craig, D.; Fonseca, T.; Myers, L.; Bruce, I.; Chalmers, R.; Bujkiewicz, S.; et al. Etanercept, infliximab and adalimumab for the treatment of psoriatic arthritis: A systematic review and economic evaluation. Health Technol. Assess. 2011, 15, 1–329. [Google Scholar] [CrossRef]
- Gisondi, P.; Geat, D.; Conti, A.; Dapavo, P.; Piaserico, S.; De Simone, C.; Bianchi, L.; Costanzo, A.; Malagoli, P.; Malara, G.; et al. TNF-α inhibitors biosimilars as first line systemic treatment for moderate-to-severe chronic plaque psoriasis. Expert Rev. Clin. Immunol. 2020, 16, 591–598. [Google Scholar] [CrossRef]
- Wride, A.M.; Chen, G.F.; Spaulding, S.L.; Tkachenko, E.; Cohen, J.M. Biologics for Psoriasis. Dermatol. Clin. 2024, 42, 339–355. [Google Scholar] [CrossRef]
- Koskeridis, F.; Evangelou, E.; Ntzani, E.E.; Kostikas, K.; Tsabouri, S. Treatment with Dupilumab in Patients with Atopic Dermatitis: Systematic Review and Meta-Analysis. J. Cutan. Med. Surg. 2022, 26, 613–621. [Google Scholar] [CrossRef]
- Seegräber, M.; Srour, J.; Walter, A.; Knop, M.; Wollenberg, A. Dupilumab for treatment of atopic dermatitis. Expert Rev. Clin. Pharmacol. 2018, 11, 467–474. [Google Scholar] [CrossRef]
- Simpson, E.L.; Paller, A.S.; Siegfried, E.C.; Boguniewicz, M.; Sher, L.; Gooderham, M.J.; Beck, L.A.; Guttman-Yassky, E.; Pariser, D.; Blauvelt, A.; et al. Efficacy and Safety of Dupilumab in Adolescents with Uncontrolled Moderate to Severe Atopic Dermatitis: A Phase 3 Randomized Clinical Trial. JAMA Dermatol. 2020, 156, 44–56. [Google Scholar] [CrossRef]
- Kim, R.W.; Lam, M.; Abuabara, K.; Simpson, E.L.; Drucker, A.M. Targeted Systemic Therapies for Adults with Atopic Dermatitis: Selecting from Biologics and JAK Inhibitors. Am. J. Clin. Dermatol. 2024, 25, 179–193. [Google Scholar] [CrossRef]
- Kimball, A.B.; Okun, M.M.; Williams, D.A.; Gottlieb, A.B.; Papp, K.A.; Zouboulis, C.C.; Armstrong, A.W.; Kerdel, F.; Gold, M.H.; Forman, S.B.; et al. Two Phase 3 Trials of Adalimumab for Hidradenitis Suppurativa. N. Engl. J. Med. 2016, 375, 422–434. [Google Scholar] [CrossRef]
- Zouboulis, C.C.; Okun, M.M.; Prens, E.P.; Gniadecki, R.; Foley, P.A.; Lynde, C.; Weisman, J.; Gu, Y.; Williams, D.A.; Jemec, G.B. Long-term adalimumab efficacy in patients with moderate-to-severe hidradenitis suppurativa/acne inversa: 3-year results of a phase 3 open-label extension study. J. Am. Acad. Dermatol. 2019, 80, 60–69.e2. [Google Scholar] [CrossRef]
- Kimball, A.B.; Jemec, G.B.E.; Sayed, C.J.; Kirby, J.S.; Prens, E.; Ingram, J.R.; Garg, A.; Gottlieb, A.B.; Szepietowski, J.C.; Bechara, F.G.; et al. Efficacy and safety of bimekizumab in patients with moderate-to-severe hidradenitis suppurativa (BE HEARD I and BE HEARD II): Two 48-week, randomised, double-blind, placebo-controlled, multicentre phase 3 trials. Lancet 2024, 403, 2504–2519. [Google Scholar] [CrossRef]
- Kimball, A.B.; Jemec, G.B.E.; Alavi, A.; Reguiai, Z.; Gottlieb, A.B.; Bechara, F.G.; Paul, C.; Bourboulis, E.J.G.; Villani, A.P.; Schwinn, A.; et al. Secukinumab in moderate-to-severe hidradenitis suppurativa (SUNSHINE and SUNRISE): Week 16 and week 52 results of two identical, multicentre, randomised, placebo-controlled, double-blind phase 3 trials. Lancet 2023, 401, 747–761. [Google Scholar] [CrossRef]
- Jabbari, A.; Sansaricq, F.; Cerise, J.; Chen, J.; Bitterman, A.; Ulerio, G.; Borbon, J.; Clynes, R.; Christiano, A.; Mackay-Wiggan, J. An Open-Label Pilot Study to Evaluate the Efficacy of Tofacitinib in Moderate to Severe Patch-Type Alopecia Areata, Totalis, and Universalis. J. Investig. Dermatol. 2018, 138, 1539–1545. [Google Scholar] [CrossRef]
- Lee, H.; Park, S.H.; Lew, B.-L.; Park, H. The new era of JAK inhibitors: Impelling updates in Alopecia Areata Guideline. J. Eur. Acad. Dermatol. Venereol. 2023, 38, e602–e606. [Google Scholar] [CrossRef] [PubMed]
- Miot, H.A.; Criado, P.R.; de Castro, C.C.S.; Ianhez, M.; Talhari, C.; Ramos, P.M. JAK-STAT pathway inhibitors in dermatology. An. Bras. Dermatol. 2023, 98, 656–677. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).