Abstract
Background: Chemotherapy is a cornerstone of cancer treatment; however, resistance to first-line chemotherapeutic agents remains a major challenge. ROS1, one of fifty-eight receptor tyrosine kinases, has been implicated in various cancer subtypes, including glioblastoma, non-small-cell lung cancer, and cholangiocarcinoma. Notably, the Gly2032Arg mutation in the ROS1 protein has been linked to resistance against the kinase inhibitor crizotinib. Objectives: Given the challenge, we conducted a comprehensive in silico study to identify new drug candidates. Methods: The study starts with modeling the Gly2032Arg-mutated ROS1 protein, followed by structure-based screening of the PubChem database. Results: Out of 1760 molecules screened, we selected the top 4 molecules (PubChem CID: 67463531, 72544946, 139431449, and 139431487) with structural features similar to crizotinib, a high docking score, and drug likeness. To further validate the effectiveness of the identified compounds, we assessed their binding affinity using the Molecular Mechanics with Generalized Born Surface Area (MM-GBSA) scoring method. To underpin the behavior and stability of protein–ligand complexes, 500 ns molecular dynamics (MD) simulations were conducted, and parameters including RMSD, RMSF, and H-bond dynamics were studied and compared. Density functional theory (DFT) at the B3LYP/6-31G* level was performed to elucidate molecular features of the identified compounds. Conclusions: Overall, this study sheds light on a new series of compounds effective against mutated targets, thereby offering a new horizon in this area.
1. Introduction
Cancer remains the second leading cause of global mortality and morbidity, resulting in nearly 8 million deaths and 14 million new cases each year. A recent study indicates that both the incidence and mortality rates of cancer are expected to increase in the coming decades. Among various forms of cancers, non-small-cell lung cancer (NSCLC) is a prevalent form of lung cancer, constituting about 85% of all lung cancer cases [1,2]. Despite ongoing reports of potential treatments, fundamental challenges persist in this area, including late detection, metastasis, the absence of selective drugs, diverse treatment responses influenced by genetic, epigenetic, or environmental factors, and the almost inevitable emergence of tumour resistance [3,4,5]. Addressing these complexities remains a crucial focus in cancer research. The proto-oncogene receptor tyrosine kinase ROS1 is implicated in various cancers, including NSCLC [6,7,8,9,10]. Aberrations in the ROS1 gene, such as gene fusions, have been identified in a subset of NSCLC cases, leading to constitutive activation of the ROS1 kinase domain [11,12]. In NSCLC, ROS1 rearrangements are considered driver mutations that contribute to uncontrolled cell growth and proliferation [13,14]. Understanding the molecular mechanisms and signalling pathways regulated by ROS1 is essential for developing targeted therapies and advancing precision medicine to manage NSCLC [15,16]. Targeting ROS1 with kinase inhibitors, such as crizotinib, has shown therapeutic efficacy in treating ROS1-positive NSCLC [15,17,18]. However, most patients treated with crizotinib develop resistance within the first year.
Numerous theoretical and experimental studies have been undertaken to identify new drug candidates against ROS1 [19]. For example, Vanajothi et al. [20] studied ROS1 as a crucial biomarker in cancer progression and identified potential inhibitors using structure-based, atom-based, and pharmacophore-based virtual screening. Molecular docking and MM-GBSA calculations results suggested strong binding affinity and favorable ADME properties. However, the effectiveness of the inhibitors against mutated ROS1 remains uncertain. Additionally, 100 ns molecular dynamics (MD) simulation is insufficient to assess protein–ligand behavior. Wu et al. [21] explored the inhibitory mechanism of lorlatinib against WT and G2032R-mutated ROS1 using MD simulations and free energy calculations. They noted that the G2032R mutation reduces lorlatinib’s binding affinity by increasing conformational entropy and decreasing solvation energy. However, like the previous study, the 50 ns MD simulation duration is insufficient for a comprehensive trajectory analysis. Pathak et al. [22] identified potential inhibitors for WT and G2032R-mutated ROS1 using pharmacophore modelling, virtual screening, docking, and MD simulations. Although five compounds showed strong binding and stability, the study lacks longer MD simulations. Similarly, Hasan and co-workers [23] investigated the effects of the G2032R and D2033N mutations in ROS1 using MD simulations. They demonstrated that such mutation induces structural instability and drug resistance. In addition, conformational changes were apparent, especially at the ATP-binding site.
Considering the above-mentioned challenges and in pursuit of identifying new drugs effective in combating cancer resistance, we conducted an extensive in silico study of the Gly2032Arg-mutated ROS1 protein. We used the PDB ID 7Z5X protein as a template (residues 1937–2225) to model the complete structure [24]. The PubChem database was used to identify new molecules with characteristics similar to those of crizotinib, yielding 1760 molecules. Based on their binding free energy, MMGBSA scores, and drug-likeness, the top four candidates, namely PubChem CID: 67463531, 72544946, 139431449, and 139431487, were then selected. To assess their stability, MD simulations were performed, and root mean square deviation (RMSD), root mean square fluctuation (RMSF), secondary structure elements (SSEs), H-bonding, and protein–ligand interaction results were analyzed. Finally, density functional theory (DFT) calculations were conducted to evaluate molecular properties and descriptors. The results were compared against crizotinib as the reference.
2. Results
2.1. Homology Modeling and Validation
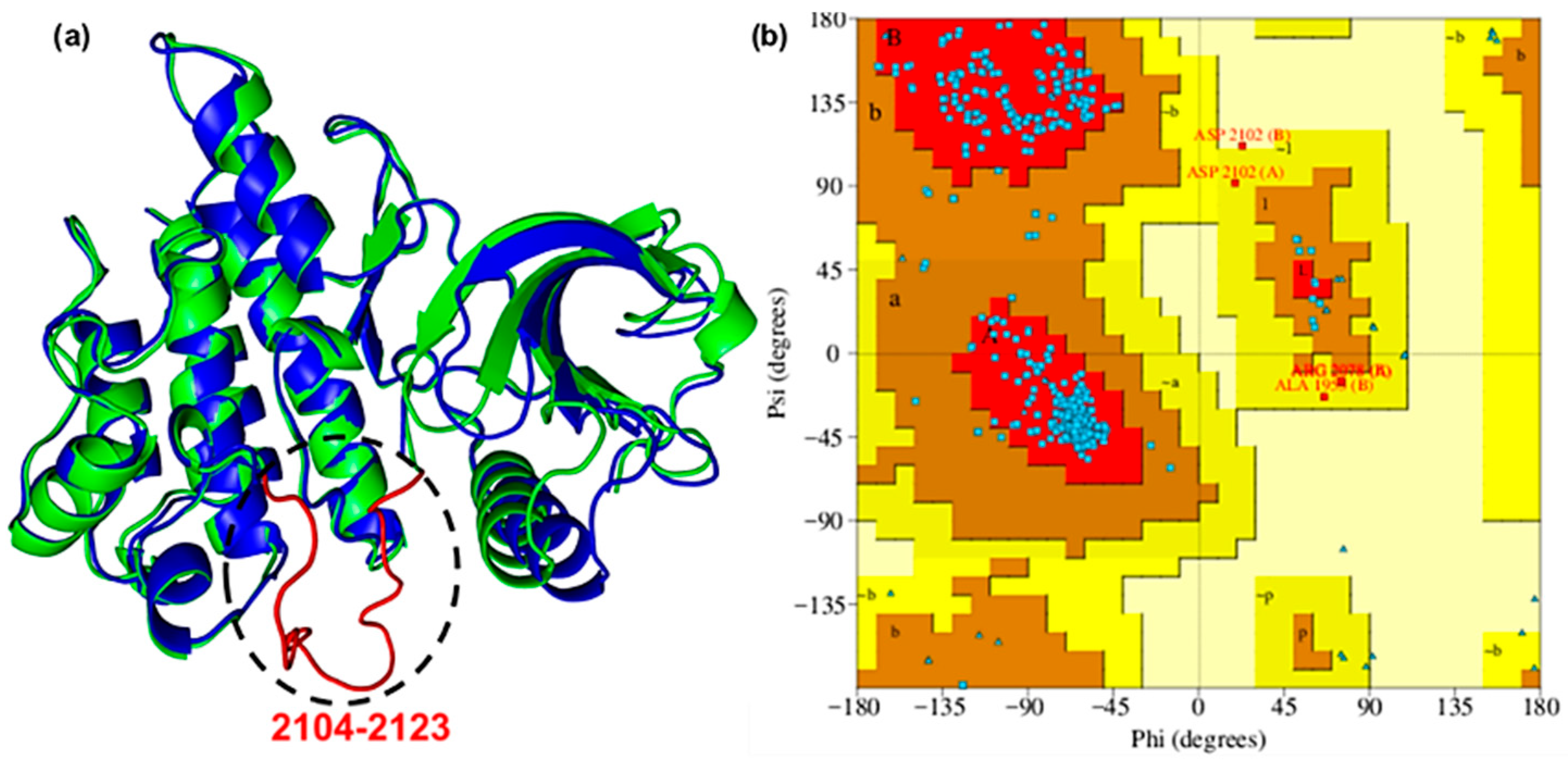
To obtain a detailed three-dimensional structure of the ROS1 protein incorporating the missing residues, homology modelling was performed using the Swiss-Model server. The missing segment (2104–2123: GLARDIYKNDYYRKRGEGLL) was modelled using PDB ID 7Z5X (residues 1937–2225) as a template [24,25]. The reconstructed loop was optimized and refined, which was confirmed by a low RMSD (0.71 Å) when aligned with the reference protein (Figure 1a) [26]. The Ramachandran plot revealed that 93.1% of the analyzed regions corresponded to the most favored conformations (Figure 1b). Additionally, 5.8% were allowed, and 0.9% were generously allowed, indicating less optimal but still permissible conformations. Only 0.2% of the plot was found in disallowed regions. Collectively, these findings substantiated the model’s adequacy and reliability [27].
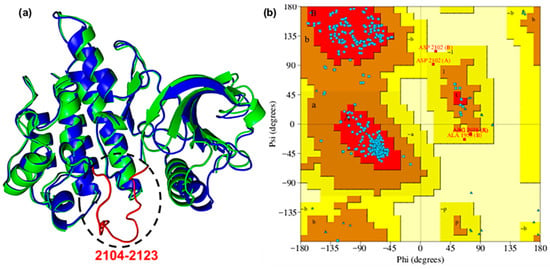
Figure 1.
(a) Superimposed ROS1 protein (PDB: 7Z5X, blue color) onto the model structure (green color), and the missing residues 21042123 (red loop). (b) Ramachandran plot.
2.2. Molecular Docking
The modelled protein, as obtained above, was then used as a receptor to identify suitable ligands that bind the protein’s active pocket. Molecular docking was performed using Glide XP, and the results are presented in Table 1 and Figure 2 and Figure 3. A comprehensive search of the PubChem database was conducted to identify potential ROS1 inhibitors, applying key drug-likeness filters such as molecular weight (MW < 500 Da), H-bond donors/acceptors, and logP values. This initial screening yielded 1760 compounds, which were subjected to molecular-docking-based virtual screening. Based on binding affinity, MM-GBSA scores, and drug-likeness properties, the top four promising candidates were shortlisted for further investigation.

Table 1.
The results encompass drug-likeness properties, molecular docking XP Gscore, and MMGBSA calculations for the selected compounds.
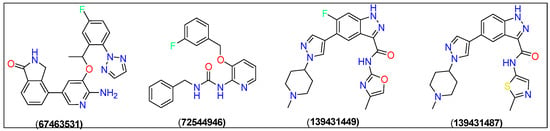
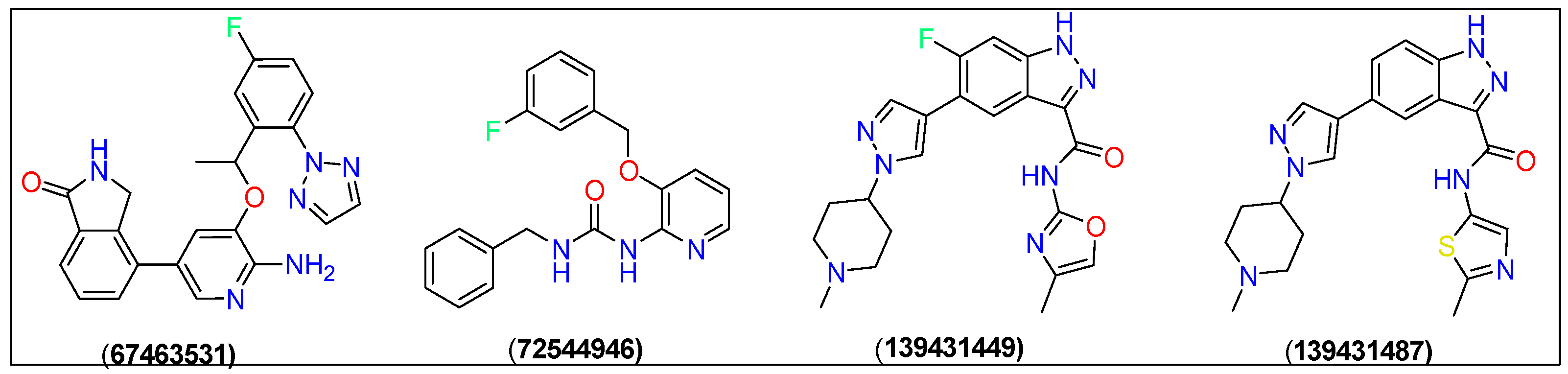
Figure 2.
Chemical structures of the top four candidates, namely PubChem CID: 67463531, 72544946, 139431449, and 139431487.
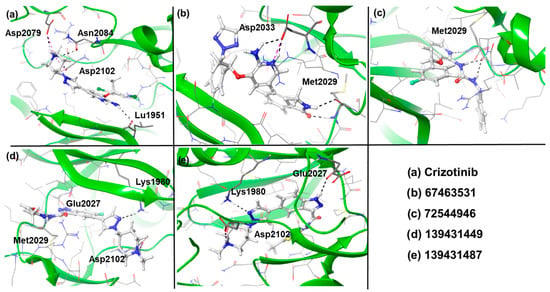
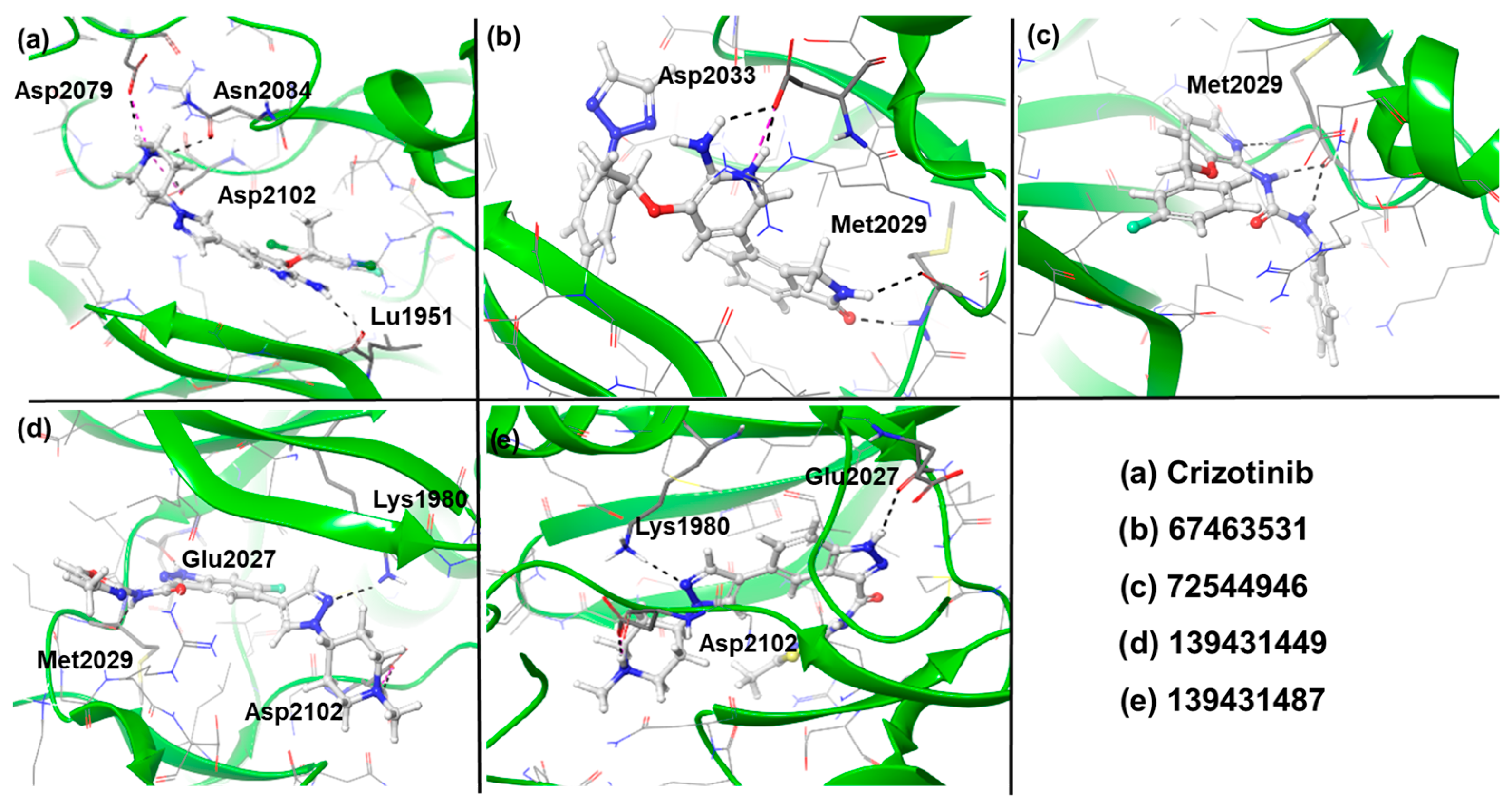
Figure 3.
Three-dimensional images of ROS1 with the reference and the identified compounds (67463531, 72544946, 139431449, and 139431487). H-bond interactions are shown as black dotted lines.
Docking studies yielded four new compounds 4-(6-amino-5-(1-(5-fluoro-2-(2H-1,2,3-triazol-2-yl)phenyl)ethoxy)pyridin-3-yl)isoindolin-1-one (67463531), 1-benzyl-3-(3-((3-fluorobenzyl)oxy) pyridin-2-yl)urea (72544946), 6-fluoro-N-(4-methyloxazol-2-yl)-5-(1-(1-methyl piperidin-4-yl)-1H-pyrazol-4-yl)-1H-indazole-3-carboxamide (139431449), and 5-(1-(1-methylpiperidin-4-yl)-1H-pyrazol-4-yl)-N-(2-methylthiazol-5-yl)-1H-indazole-3-carboxamide (139431487) as the ROS1 inhibitors (Figure 2). Crizotinib was used the reference compound in this study.
Docking results suggest that all the identified compounds displayed higher affinity (XP Gscore = −9.68 to −11.24 kcal/mol) than crizotinib (XP Gscore = −7.50 kcal/mol). The reference compound formed four H-bonds with amino acids Leu1951, Asp2079, Asn2084, and Asp2102 and interacted with residues such as Gly1952, Ser1953, Gly1954, Ala1955, Phe1956, Val1959, Lys1980, Ala1978, Leu2010, Leu2026, Glu2027, Leu2028, Met2029, Arg2032, Asp2033, Arg2083, Leu2086, and Leu2105 through Van der Waals interactions (Figure 3a and Figure 4a). Among the screened compounds, 67463531 showed the lowest XP Gscore (–11.24 kcal/mol) and formed three H-bonds involving Met2029 and Asp2033. Comparing the structure of this compound with the reference molecule, the 2,6-dichloro-3-fluorophenyl and piperidine groups in crizotinib are replaced by 5-fluoro-2-(2H-1,2,3-triazol) and isoindolin-1-one moieties, respectively, while the 2-aminopyridine group remains intact. The latter group (isoindoline-1-one) served as the HBD/HBA fragments, while the triazole ring was found to be exposed to the solvent. Such replacement also modified the way the 2-aminopyridine group interacted with the receptor, which is important when considering the ROS mutation. The ligand was found to be further stabilized by other non-covalent interactions (Figure 3b and Figure 4b). The compound 72544946 exhibited a slightly lower affinity (XP Gscore = −10.63 kcal/mol) and one H-bond (with residue Met2029). However, the two benzyl rings interacted with neighbouring residues, including Leu1949, Leu1951, Gly1952, Ser1953, Gly1954, Val1959, Glu1961, Lys2090, Lys1976, Ala1978, Lys1980, Leu2010, Leu2086, Leu2026, Glu2027, Leu2028, Glu2030, Gly2031, Arg2032, Asp2033, and Thr2036 (Figure 3c and Figure 4c).
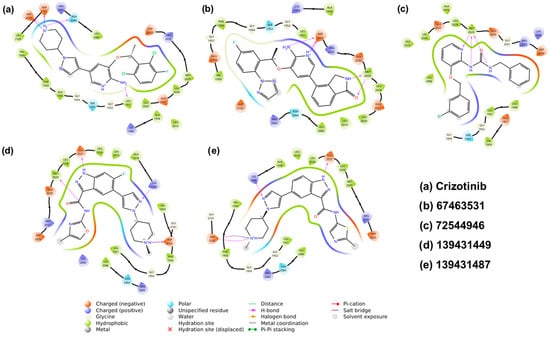
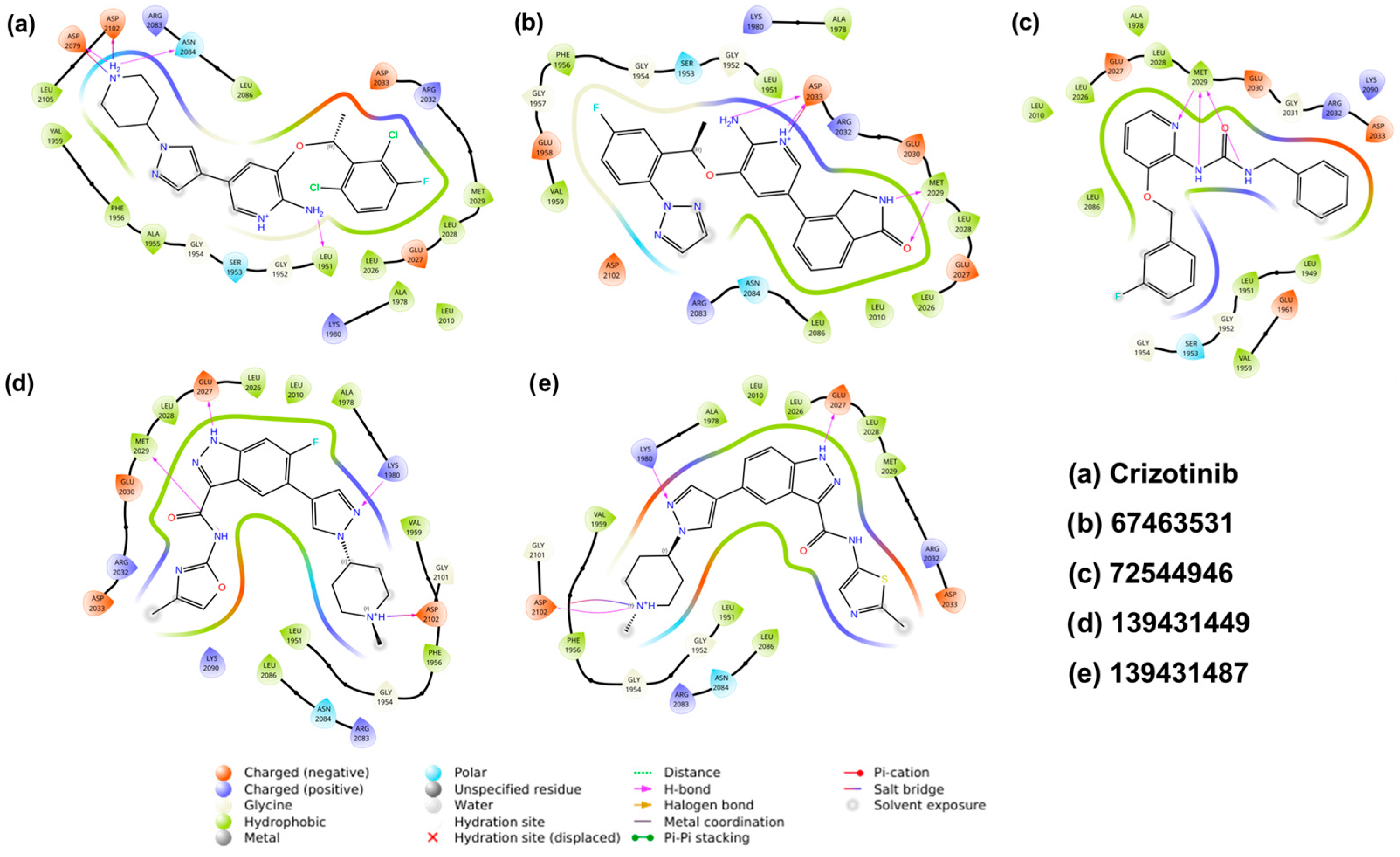
Figure 4.
Two-dimensional interactions of ROS1 with the reference and the screened compounds (67463531, 72544946, 139431449, and 139431487). H-bond interactions are shown as pink arrows.
The last two compounds, 139431449 and 139431487, are indazole-based and share a high degree of structural similarity. This also explains why they exhibited very close affinity (−9.77 and −9.68 kcal/mol, respectively). Both compounds formed three H-bonds with Lys1980, Glu2027, and Asp2102, while 139431449 formed an additional interaction with Met2029 (Figure 3d,e and Figure 4d,e). Following the docking studies, we also estimated the binding free energies (ΔGBind) from MM-GBSA calculations. Except for 139431449 (−55.31 kcal/mol), all other compounds displayed the highest negative binding energy (−56.10 to −58.31 kcal/mol) and were better than the reference compound (−55.59 kcal/mol).
2.3. Physicochemical and Pharmacokinetic Profiling
ADME/T and pharmacological profiling play a vital role in the drug discovery process. QikProp analysis confirmed that crizotinib and four shortlisted compounds (67463531, 72544946, 139431449, and 139431487) met all drug-like features and obeyed Lipinski’s Rule of Five (RO5) (Table 1) [28]. All selected compounds exhibited MW < 500 Da, a polar surface area (PSA) within the acceptable range, and favorable H-bonding fragments (donors: 1–3, acceptors: 3.75–8). Human oral absorption (%HOA) ranged from 77% to 92%, indicating good bioavailability. Additionally, properties such as blood–brain barrier permeability and solubility were within acceptable limits, making these compounds promising candidates for further studies.
The selected compounds were also evaluated for physicochemical and pharmacokinetic properties (Table 1) using the modules available in Qikprop [29]. All compounds exhibited promising permeability across cell membranes (QPPCaco > 88) and moderate brain penetration (QPlogBB between −0.14 and −1.27). Solubility values (QPlogS) ranged from −5.806 to −3.992, indicating good drug-likeness. Notably, all candidates showed acceptable human serum albumin binding (QPlogKhsa) and metabolic stability (metab: 3–6). The overall results suggest that these compounds possess favorable ADME profiles.
2.4. MD Simulation
2.4.1. RMSD Analysis
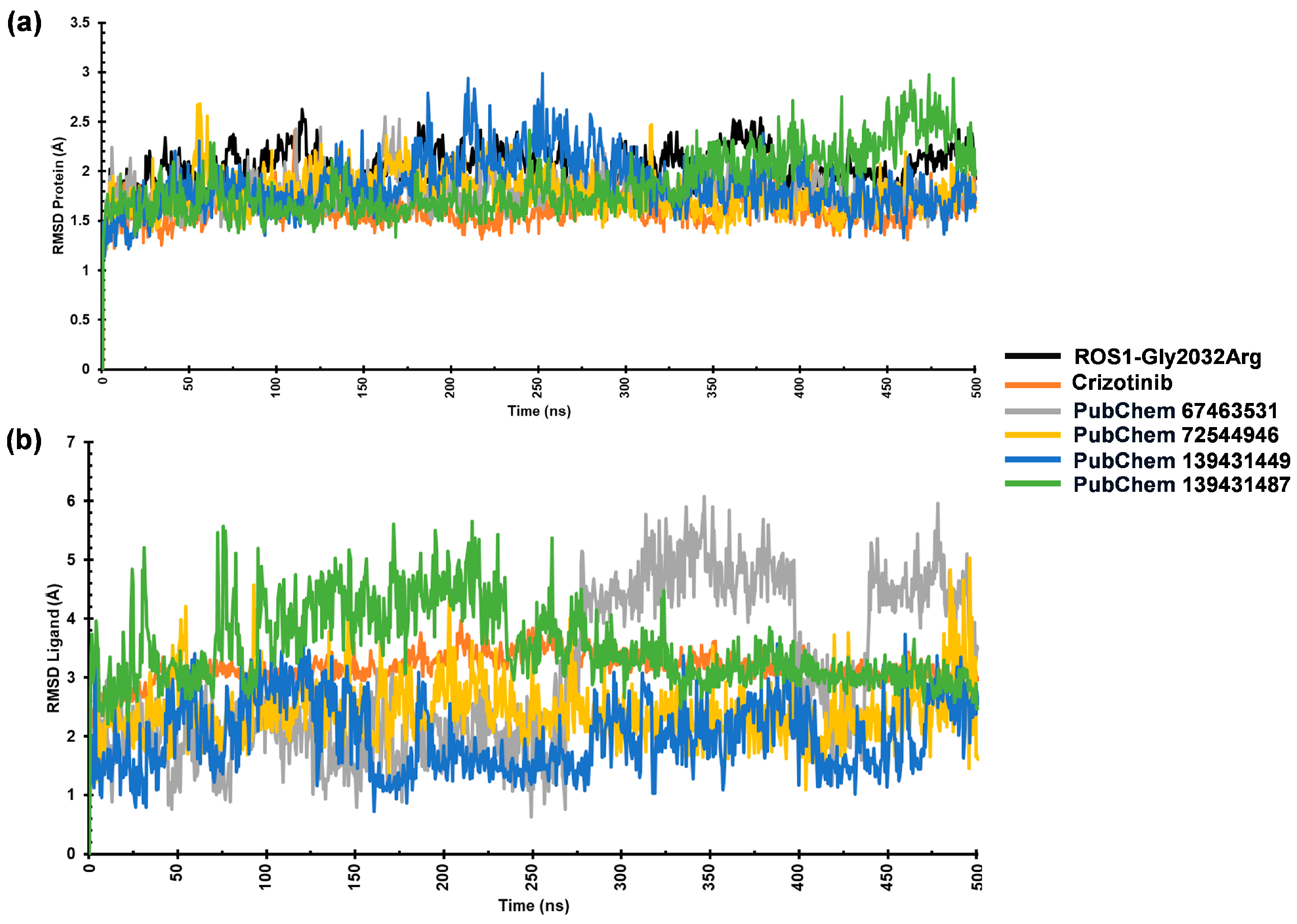
The stability of ROS1–ligand complexes was assessed through RMSD analysis during a 500 ns MD simulation. We monitored the RMSD values for both protein and ligand to evaluate structural stability and binding behavior. The average Cα-RMSD values for ROS1-Gly2032Arg and its complexes with the reference, 67463531, 72544946, 139431449, and 139431487 ranged from 1.61 Å to 2.04 Å. The complexes reached equilibrium within the first 50 ns, indicating stable interactions. However, the mutant ROS1 structure exhibited noticeable fluctuations between 60 and 120 ns, which persisted up to 375 ns. Among the studied compounds, the reference crizotinib, 67463531, and 72544946 demonstrated the lowest RMSD values (~1.61–1.62 Å) (Table 2). On the other hand, the ROS1-139431449 complex showed slight fluctuations between 175 and 300 ns, peaking around 2.5 Å before stabilizing. These findings suggest that the references 67463531 and 72544946 maintained a stable interaction with ROS1 (Figure 5a). Additionally, we analyzed the ligand RMSD to evaluate binding behavior during the simulation period. The ligand RMSD values for crizotinib, 67463531, 72544946, 139431449, and 139431487 were 3.16 Å, 2.91 Å, 2.41 Å, 2.21 Å, and 3.42 Å, respectively, indicating that ligands maintained structural integrity throughout the simulation. This also confirms their stable interactions within the ROS1 binding site (Figure 5b).

Table 2.
The average value of the RMSD of protein and ligand and the RMSF values.
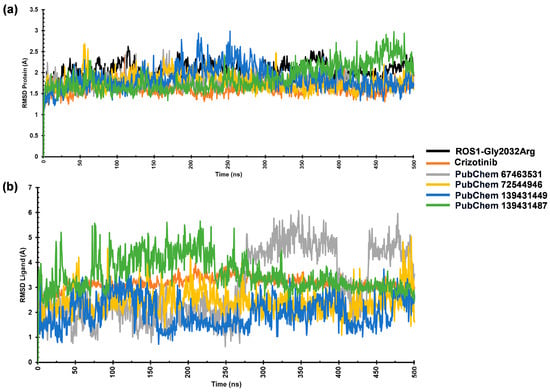
Figure 5.
The MD simulation results of ROS1-Gly2032Arg with crizotinib and compounds (67463531, 72544946, 139431449, and 139431487). RMSD of (a) protein and (b) ligands.
2.4.2. RMSF Analysis
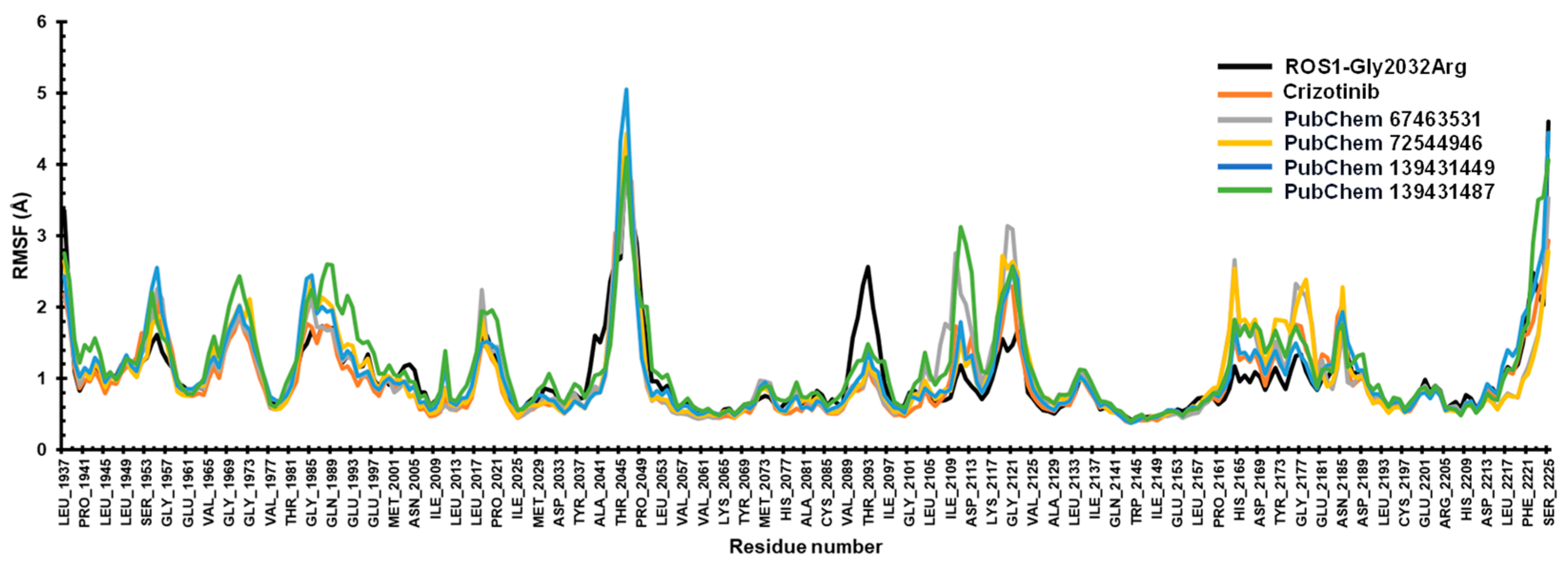
RMSF analysis helps in identifying the key residues of a protein engaged in interactions with a ligand. The mean RMSF values for the ROS1-mutated protein and the compounds crizotinib, 67463531, 72544946, 139431449, and 139431487 were 1.03 Å, 1.01 Å, 1.01 Å, 1.02 Å, 1.03 Å, and 1.11 Å, respectively. Significant fluctuations were observed in the loop regions of the ROS1-mutant protein, particularly in the Ala2041-Leu2037 (>3 Å), and Val2089-Ile2097 (~3 Å) residues. This can be attributed to the greater susceptibility of the N- and C-termini compared to other regions of the protein. We also noted a consistent trajectory for the compounds, indicating enhanced protein stability (Figure 6).
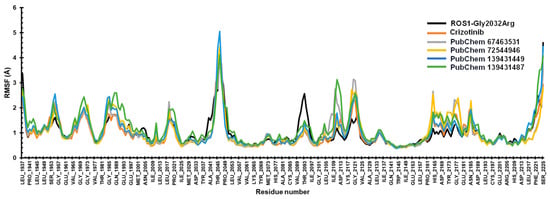
Figure 6.
The RMSF of the protein with crizotinib and the screened compounds (67463531, 72544946, 139431449, and 139431487).
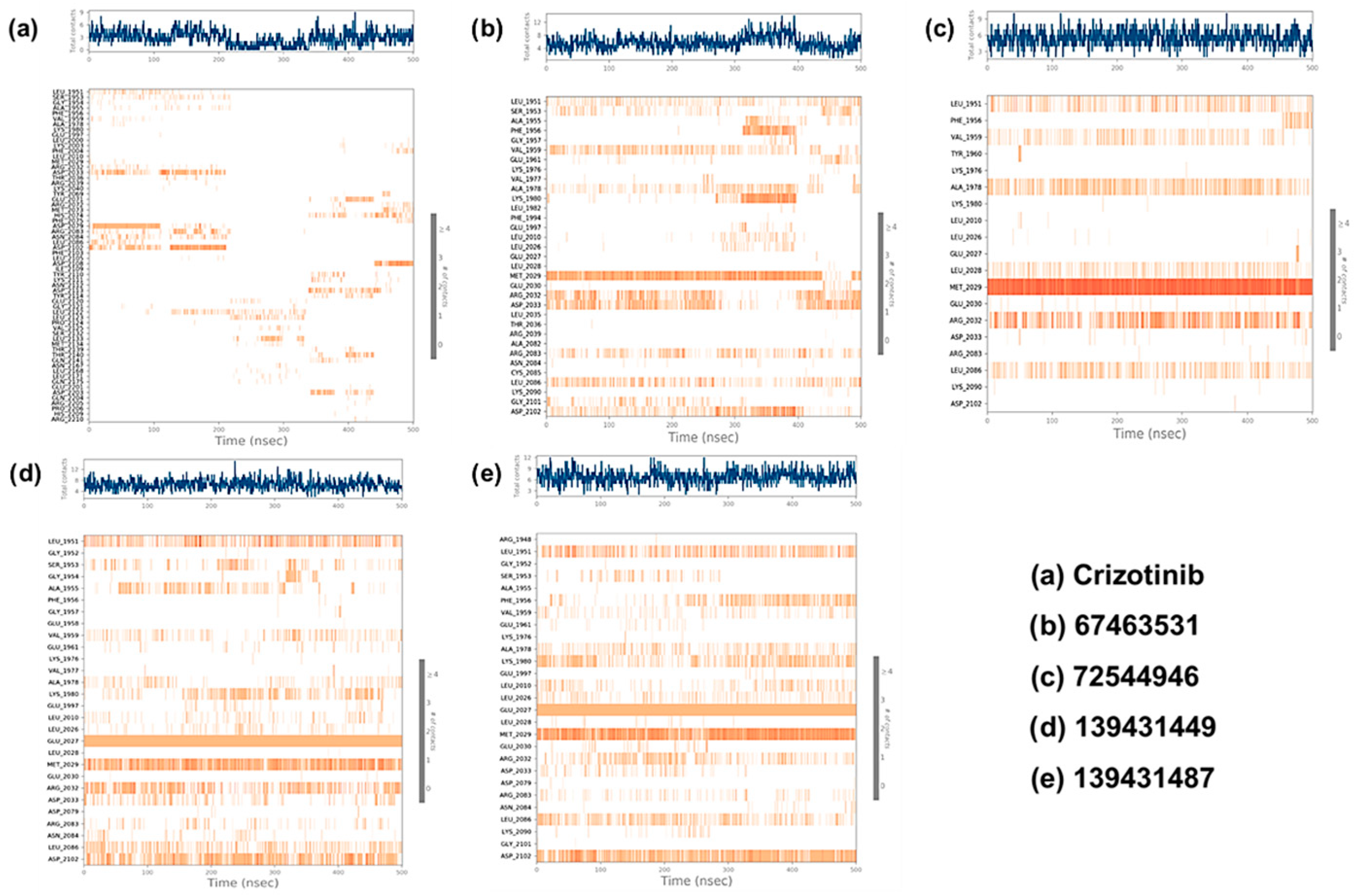
2.4.3. Contact Map Analysis
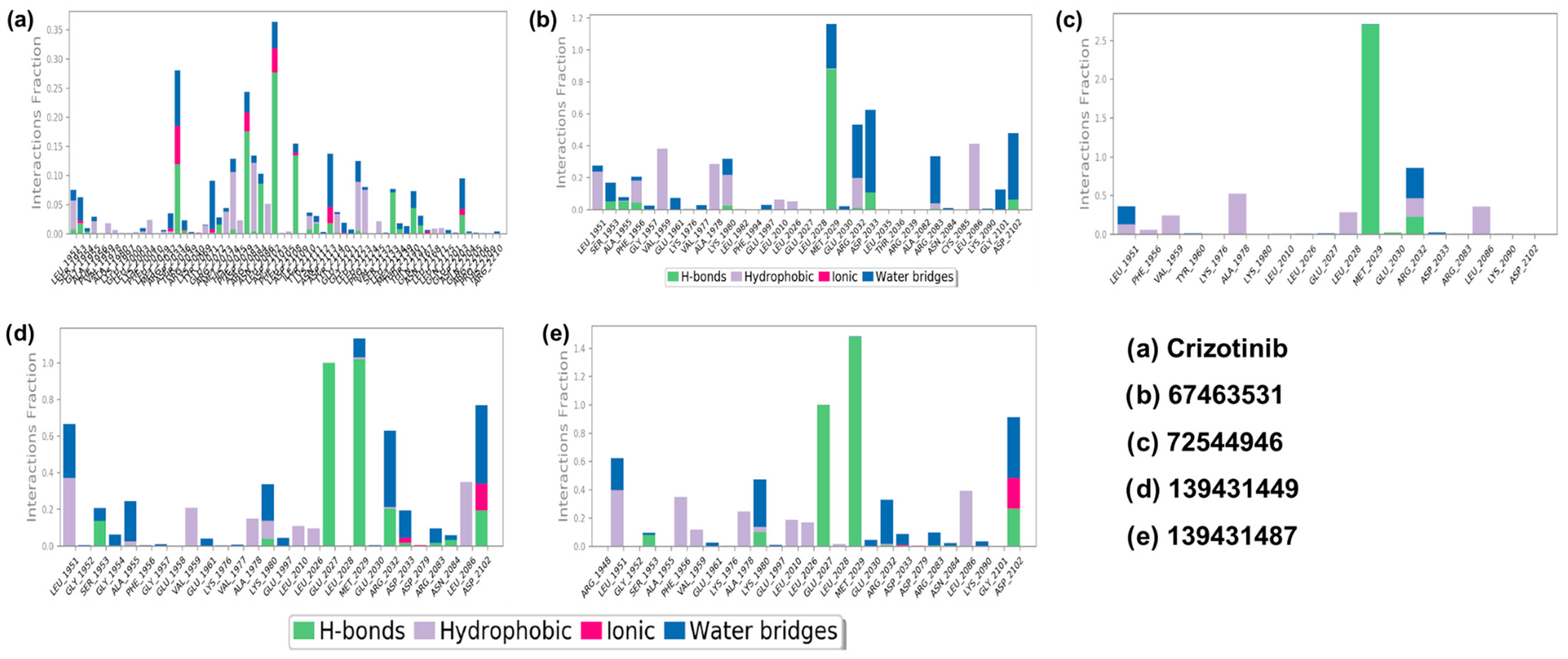
The contact maps for all the complexes were extracted from the simulated trajectory (Figure 7). White or light colors indicate weaker interactions, while darker colors indicate stronger interactions. As can be seen, the protein residues engaged with the ligands via both polar and non-polar non-covalent interactions (Figure 8). Crizotinib exhibits a notable binding profile, forming H-bonding with Ser1953, Arg2032, Asp2033, Glu2071, Met2073, Phe2075, Asp2079, Arg2083, Leu2086, Asp2108, Ile2109, Tyr2110, Asn2112, Gly2121, Ser2132, Leu2133, Met2134, Thr2139, Thr2140, and Gln2201 residues of the binding pocket of ROS1. On the other hand, Leu1951, Ser1953, Gly1954, Phe1959, Glu1997, Lys2003, Leu2010, Lys2040, Arg2072, Met2073, Asp2079, Asn2084, Phe2103, Ile2109, Asp2113, Gly2121, Leu2122, Leu2123, Pro2124, and Glu2201 assisted in forming hydrophobic interactions. Finally, we also noted water bridge interactions involving Leu1951, Ser1953, Leu2000, Leu2010, Met2029, Glu2071, Arg2072, Met2073, Phe2075, Asp2079, Arg2083, Asp2108, Ile2109, Tyr2110, Asn2112, Asp2113, Glu2120, Gly2121, Leu2122, Leu2133, Met2134, Thr2139, Thr2140, Leu2171, Gln2175, Asp2203, and Gln2204. Ionic bonds formed with Ser1953, Met2029, Arg2032, Tyr2069, Phe2075, Leu2105, Asn2112, Tyr2114, Gln2141, and Glu2201 (Figure 8a).
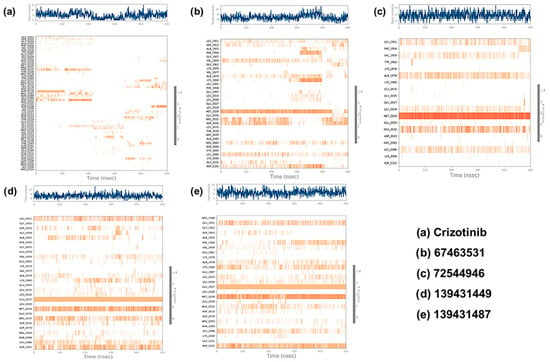
Figure 7.
The contact map of ROS1 with crizotinib and compounds (67463531, 72544946, 139431449, and 139431487).
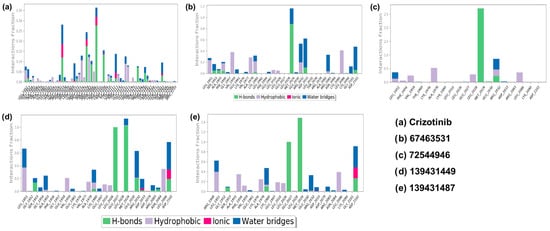
Figure 8.
The protein–ligand interaction diagrams for ROS1 with crizotinib and compounds (67463531, 72544946, 139431449, and 139431487 (a–e), respectively).
Similarly, in the ROS1-67463531 complex, H-bonds formed with Ser1953, Ala1955, Ala1978, Leu2028, Glu2030, Arg2032, Ala2082, and Gly2101 residues. Hydrophobic residues include Leu1951, Ala1955, Gly1957, Val1977, Ala1978, Glu1997, Leu2010, Glu2030, Ala2082, and Cys2085. Water bridge interactions involve Leu1951, Ser1953, Ala1955, Phe1956, Val1959, Lys1976, Ala1978, Phe1994, Met2029, Glu2030, Arg2032, Asp2033, Leu2036, Ala2082, Arg2083, Asn2084, Leu2086, Lys2090, Gly2101, and Asp2102 (Figure 8b). Moreover, in the ROS1-72544946 complex, the interaction pattern includes H-bonds with Leu2028, Met2029, Glu2030, and Arg2032. Hydrophobic residues involved are Leu1951, Phe1956, Val1959, Lys1976, Glu2027, Glu2030, and Arg2083. Water bridge interactions comprise Leu1951, Val1959, Glu2030, and Arg2032 (Figure 8c). In the ROS1-139431449 complex, the interaction pattern includes H-bonds with Ser1953, Ala1955, Ala1978, Leu2026, Leu2028, Glu2030, Arg2032, Asp2079, Arg2083, and Leu2086. Hydrophobic residues involved are Leu1951, Gly1954, Glu1958, Val1977, Glu1997, Leu2010, Glu2030, and Asn2084. Water bridge interactions comprise Leu1951, Gly1952, Gly1954, Phe1956, Val1959, Lys1976, Ala1978, Glu1997, Leu2010, Leu2028, Glu2030, Arg2032, Asp2033, Asp2079, and Asp2102. Ionic bonds formed with Arg2032, Asp2033, and Leu2086 (Figure 8d). The interaction pattern of the ROS1-139431487 complex includes H-bonds with Gly1952, Ala1978, Leu2026, Leu2028, Glu2030, Arg2032, Asp2079, and Asp2102. Hydrophobic residues involved are Arg1948, Ala1955, Phe1956, Lys1976, Ala1978, Glu1997, Leu2010, and Asn2084. Water bridge interactions comprise Arg1948, Gly1952, Val1959, Lys1980, Met2029, Glu2030, Arg2032, Asp2079, Arg2083, Leu2086, and Asp2102. Ionic bond formation occurs with Arg2032 and Asp2102 (Figure 8e).
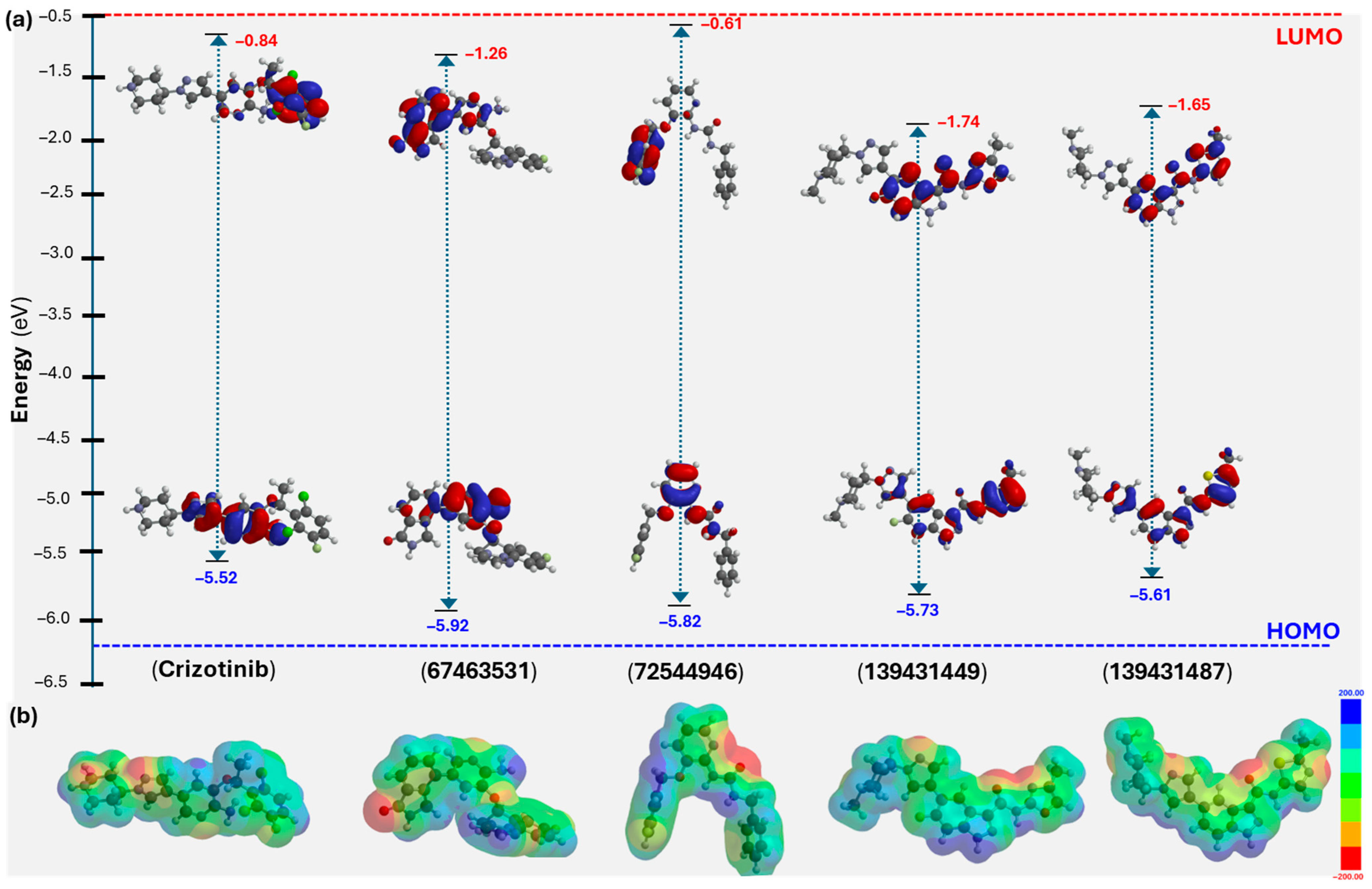
2.5. DFT Studies
Following the molecular docking, ADME/T analyses, and MD studies, DFT calculations at the B3LYP/6-31G* level were performed to explore the molecular features of the identified compounds [30]. While DFT is widely recognized for its accuracy and ability to predict the structure and properties of molecular systems, it has recently attracted interest among researchers for estimating molecular descriptors and correlating them with bioactivity [31,32]. The results of these findings are presented in Table 3 and Figure 9. As shown, the highest occupied molecular orbital (HOMO) is primarily distributed over a common motif, the pyridinyl ring, in both the reference (crizotinib) and the screened compounds (67463531 and 72544946, Figure 9a). As expected, the lowest unoccupied molecular orbital (LUMO) was found mainly over the 2,6-dichloro-3-fluorophenyl core in crizotinib. On the other hand, it was over isoindolin-1-one in 67463531 and 3-fluorobenzyl in 72544946 (Figure 9a). Compounds 139431449 and 139431487, having close structures (only differing in the oxazole/thiazole fragment), showed similar frontier molecular orbital distribution and energy levels. Therefore, in the case of 139431449 and 139431487, HOMO was delocalized over 6-fluoro-N-(4-methyloxazol-2-yl)-1H-indazole-3-carboxamide and N-(2-methylthiazol-5-yl)-1H-indazole-3-carboxamide, respectively. The HOMO levels for the screened compounds range from −5.52 to −5.92 eV, while the LUMO values vary between –1.74 and −0.84 eV, resulting in ΔE(H-L) values between 3.96 and 4.66 eV. These values were comparable with the reference compounds (ΔE(H-L) = 4.68 eV). Interestingly, we noted a relationship between the HOMO level and the XP GScore of the compounds. The electrostatic potential (ESP) map of the studied compounds is shown in Figure 9b. Different colors of the ESP indicate electron-poor or rich regions. Finally, using the ΔE(H-L) energies, we estimated the chemical reactivity descriptors such as chemical hardness (η = 1.98 to 2.34 eV), softness (σ = 0.19 to 0.25 eV−1), chemical potential (μ = −3.73 to −3.18 eV), electrophilicity index (ω = 1.97 to 3.48 eV), and electronegativity (χ = 3.18 to 3.73 eV) of the compounds.

Table 3.
DFT (B3LYP/6-31G*)-calculated frontier molecular orbital energies, energy gap (ΔE(H-L)), and chemical reactivity descriptors computed from the HOMO-LUMO energies.
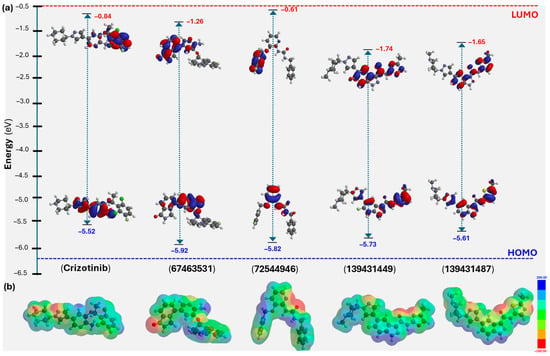
Figure 9.
(a) Frontier molecular orbitals and (b) electrostatic potential map of crizotinib and shortlisted compounds.
3. Discussion
The ROS1 gene encodes a proto-oncogene tyrosine-protein kinase that plays a role in cellular signalling and oncogenesis [6]. The protein consists of an extracellular domain with nine fibronectin III-like domains and three β-propeller structures, a 23-residue transmembrane segment, and an intracellular tyrosine kinase domain [6,34]. ROS1 has emerged as a significant oncogenic driver in multiple cancers [18]. Targeted therapies, such as crizotinib and lorlatinib, have been developed to inhibit ROS1 fusion proteins, offering effective treatment options for ROS1-positive lung cancer [17,35]. However, resistance mutations, particularly G2032R, present challenges, necessitating the discovery of next-generation inhibitors [36]. G2032R is the most prevalent resistance mechanism in ROS1-rearranged non-small-cell lung cancer (NSCLC), emerging in approximately one-third of patients after crizotinib therapy [37]. Current second-generation inhibitors, such as ceritinib and entrectinib, show limited efficacy against this mutation.
Despite significant progress in computational drug design and the synthesis of ROS1 inhibitors, bypassing resistance remains a challenge. Researchers continue to explore novel scaffolds and adopt optimization strategies to enhance potency, selectivity, and resistance profiles. While promising compounds have been identified [37,38,39], new drug candidates are needed to develop clinically effective ROS1 inhibitors. Recently, Park et al. [40] developed trisubstituted pyrazole-based inhibitors, with some compounds showing low IC50 values and strong interactions within ROS1. The inhibitors were tested primarily against wild-type ROS1, leaving their effectiveness against resistant mutations like G2032R unclear. Similarly, Guo et al. [41] focused on developing dual ALK/ROS1 inhibitors to overcome ALK G1202R mutation. They adopted a structure-guided approach, leading to the identification of diarylaminopyrimidine analogues with superior cytotoxicity and nanomolar inhibitory activity against ALK and ROS1. It was suggested that the introduction of heteroatoms, such as sulfur, is beneficial for activity. Drilon et al. [42] studied and identified the ROS1 D2033N mutation as a novel mechanism of acquired resistance to crizotinib in ROS1-rearranged lung cancer. Structural modeling and biochemical assays revealed that this mutation reduces crizotinib binding by altering electrostatic interactions. It was also reported that a heterocyclic small molecule N-(4-((6,7-dimethoxyquinolin-4-yl)oxy)phenyl)-N-(4-fluorophenyl)cyclopropane-1,1-dicarboxamide (cabozantinib) inhibits the mutant form. While the above-mentioned candidates are promising, further clinical studies will be needed to determine their broader applicability and potential off-target effects, which is indeed a time- and money-intensive step.
Considering this, we adopted a comprehensive computational approach to identify new candidates that could bind and inhibit the mutant form of ROS1. Fragment-based molecular docking identified four promising inhibitors (67463531, 72544946, 139431449, and 139431487) from 1760 molecules. These top four compounds showed strong binding affinities and favorable pharmacokinetic properties. MD simulations confirmed the stability of ROS1–ligand complexes over 500 ns, especially complexes containing 67463531 and 72544946. RMSF and contact map analyses highlighted several key residues involved in stable interactions. DFT results also supplement the docking results, as the values of frontier molecular orbital energies and other descriptors for the top two candidates (67463531 and 72544946) are close to those of the reference compound, crizotinib.
4. Materials and Methods
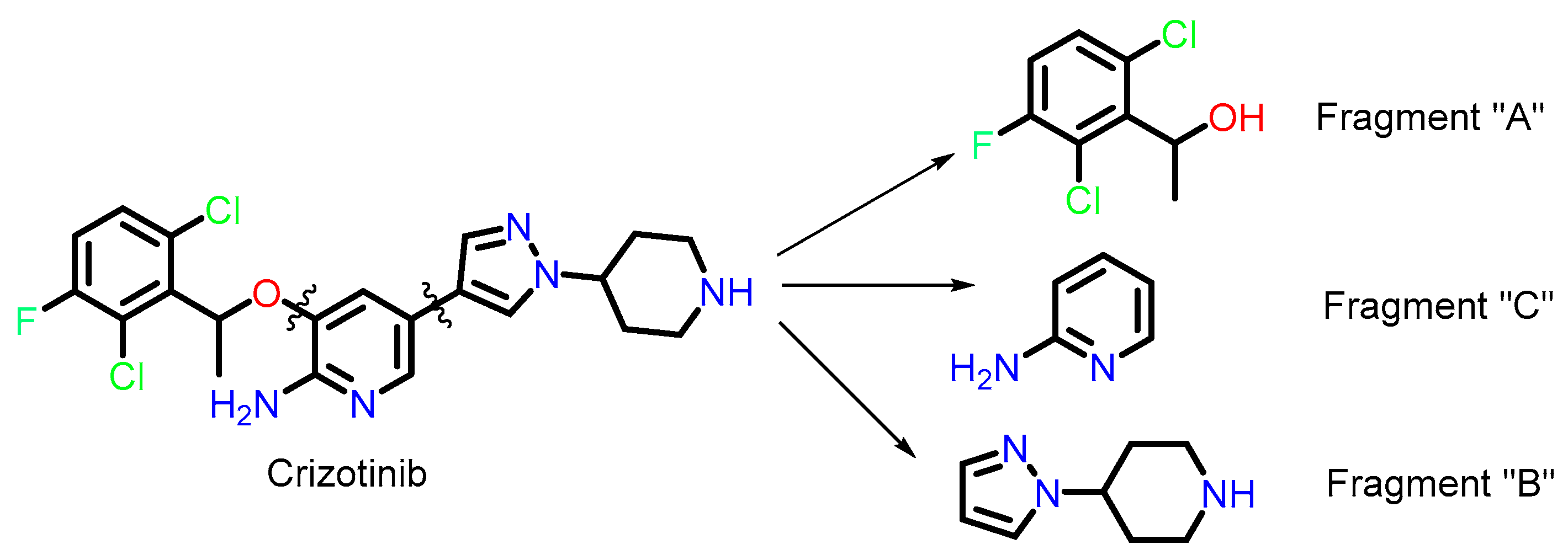
To collect the maximum diversity of molecules such as crizotinib, we adopted a fragment-based search strategy (Scheme 1). We divided crizotinib into three parts, namely fragments “A” (SMILES: (O[C@@H](C1=C(Cl)C=CC(F)=C1Cl)C), “B” (SMILES: N1(C2CC NCC2)C=CC=N1), and “C” (SMILES: NC1=C(O)C=CC=N1). We then queried these fragment-containing compounds available in the PubChem database [43], leading to the identification of 1760 molecules.
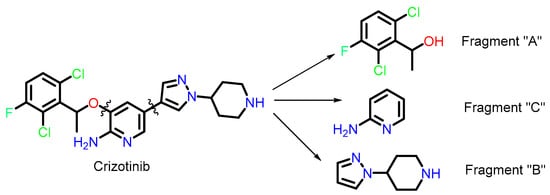
Scheme 1.
Chemical structures of the fragments used to search the PUBCHEM database.
4.1. Molecular Docking
4.1.1. Hardware and Software
All computational calculations were conducted on a Dell workstation with 64 GB of RAM with an NVIDIA GeForce RTX 3070 graphics card and 8 GB of GDDR6 memory.
4.1.2. Ligands and Receptor Preparation
A set of 1760 compounds from the PubChem database was obtained in sdf format. The structures were cleaned, energy-minimized, and converted to mae format using Schrödinger Maestro. LigPrep (Schrödinger) was then employed for further optimization and preparation for molecular docking studies [44]. The ROS1 protein consists of 2347 amino acids [6]. Its available structure (PDB ID: 7Z5X) spans residues 1937–2225 but lacks a segment (2104–2123) [24]. To complete the structure, the missing residues were modelled using SWISS-MODEL [45], incorporating the co-crystal ligand from the original structure. Active site residues were identified from UniProt (P08922), crystallographic data, and literature [46]. Further validation was performed using PDBsum and sources in the literature [47].
4.1.3. Pharmacological Properties Predictions
The pharmacological properties (absorption, distribution, metabolism, and toxicity, ADMET) of the compounds under investigation were assessed using the QikProp software V-2024.4 (Schrödinger) [48,49].
4.1.4. Molecular-Docking-Based Virtual Screening
The Glide module is an advanced, three-tiered molecular docking protocol designed to streamline the drug discovery process [50]. A systematic multi-stage virtual screening workflow was employed to identify potential ROS1 inhibitors. Initially, 1760 crizotinib-like compounds were retrieved from the PubChem database and screened using High-Throughput Virtual Screening (HTVS), followed by Standard Precision (SP) and Extra Precision (XP) docking, each retaining the top 50% of ranked molecules. From the XP stage, 25 leading compounds were shortlisted and evaluated [51]. Based on docking performance and key hydrogen bond interactions, four top candidates (PubChem CIDs 67463531, 72544946, 139431449, and 139431487) were selected. Their binding modes were visualized using PyMOL 3.1, and owing to their favorable binding affinities, interaction stability, and pharmacokinetic profiles, these compounds were advanced for MD simulations [52].
4.1.5. MD Simulations
To predict the stability of the protein–ligand docking complex, MD calculations were conducted using Desmond V4.1 following our previous protocol [53]. The complexes were immersed in a simple point charge (SPC) water model, neutralized with counterions, and adjusted to a physiological salt concentration of 0.15 M [54]. The Particle Mesh Ewald (PME) method was used for electrostatics, with a threshold of 10 for Lennard–Jones interactions. The SHAKE algorithm was employed to constrain the movements of covalent bonds involving hydrogen atoms [54,55]. Initial energy minimization (2000 steps) with solute restraints utilized a hybrid method of steepest descent (10 steps) and the LBFGS algorithm followed by energy minimization (2000 steps) without solute restraints. Simulations at 300 K involved 24 ps in the NPT ensemble with restrained solute non-hydrogen atoms, followed by an additional 24 ps with no constraints. Initial simulations utilized Berendsen thermostats and barostats to monitor temperatures and pressures [56]. Subsequently, a Nose–Hoover thermostat at 300 K and Martyna–Tobias–Klein barostats at 1.01 bar pressure were employed in the NPT ensemble for a relaxed method spanning 500 ns with a time steps of 2 fs [57]. Trajectories (around 20 K) were recorded at a time interval of 4.5 ps.
4.1.6. Density Functional Theory (DFT) Studies
DFT calculations were performed using the Becke three-parameter hybrid functional combined with Lee-Yang-Parr correlation functional (B3LYP) and 6-31G* basis set with the Spartan 24 software. Calculations were performed using the PCM solvation model, and water was selected as the solvent [58,59]. Chemical reactivity descriptors (ionization energy, electron affinity, electronegativity, chemical potential, chemical hardness, softness, and electrophilicity index) were estimated following a previous publication [33].
5. Conclusions
The proto-oncogene receptor tyrosine kinase ROS1 plays a pivotal role in regulating cancers, particularly non-small-cell lung cancer (NSCLC). Aberrations such as ROS1 gene fusions have been detected in a subset of NSCLC cases, leading to persistent activation of the ROS1 kinase domain. These mutations are recognized as driver mutations that contribute to unregulated cell growth. Targeting ROS1 with inhibitors like crizotinib has proven effective in treating ROS1-positive NSCLC. In this context, targeting ROS1 presents a promising strategy for developing effective anticancer drugs. A total of 1760 compounds, bearing crizotinib fragments, were retrieved from the PubChem database. This study identified four small-molecule inhibitors, namely 67463531, 72544946, 139431449, and 139431487, targeting mutated ROS1 with a favorable pharmacokinetic profile. The results of the molecular docking and MD simulations suggest high affinity of these compounds against mutated ROS1. While further in vitro and in vivo validation is required to substantiate these claims, we firmly believe that the compounds are promising and may serve as potential leads for next-generation ROS1 inhibitors.
Author Contributions
Conceptualization, M.J.A., A.H. and S.A.; methodology, M.J.A. and A.H.; software, M.J.A. and S.A.; writing—original draft preparation, M.J.A. and A.H.; resources, M.J.A., A.J. and S.D.H.; formal analysis, M.J.A. and D.G.; review and editing, A.H., A.J., S.A. and D.G.; supervision, M.J.A.; project administration, A.H.; funding acquisition, M.J.A. All authors have read and agreed to the published version of the manuscript.
Funding
This research has been funded by Scientific Research Deanship at University of Ha’il—Saudi Arabia through project number <<RG-24 095>>.
Informed Consent Statement
Not applicable.
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding authors.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| ADME | Adsorption, distribution, metabolism, excretion |
| DFT | Density functional theory |
| MD | Molecular dynamics |
| MMGBSA | Molecular Mechanics Generalized Born Surface |
| NSCLC | Non-small-cell lung cancer |
| PDB | Protein data bank |
| RMSD | Root mean square deviation |
| RMSF | Root mean square fluctuation |
References
- Yatabe, Y. Molecular pathology of non-small cell carcinoma. Histopathology 2024, 84, 55–66. [Google Scholar] [CrossRef]
- Gridelli, C.; Rossi, A.; Carbone, D.P.; Guarize, J.; Karachaliou, N.; Mok, T.; Petrella, F.; Spaggiari, L.; Rosell, R. Non-small-cell lung cancer. Nat. Rev. Dis. Primers 2015, 1, 15009. [Google Scholar] [CrossRef] [PubMed]
- Damen, M.P.; van Rheenen, J.; Scheele, C.L. Targeting dormant tumor cells to prevent cancer recurrence. FEBS J. 2021, 288, 6286–6303. [Google Scholar] [CrossRef]
- Dagogo-Jack, I.; Shaw, A.T. Tumour heterogeneity and resistance to cancer therapies. Nat. Rev. Clin. Oncol. 2018, 15, 81–94. [Google Scholar] [CrossRef] [PubMed]
- Turner, N.C.; Reis-Filho, J.S. Genetic heterogeneity and cancer drug resistance. Lancet Oncol. 2012, 13, e178–e185. [Google Scholar] [CrossRef] [PubMed]
- Roskoski, R., Jr. ROS1 protein-tyrosine kinase inhibitors in the treatment of ROS1 fusion protein-driven non-small cell lung cancers. Pharmacol. Res. 2017, 121, 202–212. [Google Scholar] [CrossRef]
- Esteban-Villarrubia, J.; Soto-Castillo, J.J.; Pozas, J.; San Román-Gil, M.; Orejana-Martín, I.; Torres-Jiménez, J.; Carrato, A.; Alonso-Gordoa, T.; Molina-Cerrillo, J. Tyrosine kinase receptors in oncology. Int. J. Mol. Sci. 2020, 21, 8529. [Google Scholar] [CrossRef]
- Moes-Sosnowska, J.; Szpechcinski, A.; Chorostowska-Wynimko, J. Clinical significance of TP53 alterations in advanced NSCLC patients treated with EGFR, ALK and ROS1 tyrosine kinase inhibitors: An update. Tumor Biol. 2024, 46, S309–S325. [Google Scholar] [CrossRef]
- Davies, K.D.; Doebele, R.C. Molecular pathways: ROS1 fusion proteins in cancer. Clin. Cancer Res. 2013, 19, 4040–4045. [Google Scholar] [CrossRef]
- Rimkunas, V.M.; Crosby, K.E.; Li, D.; Hu, Y.; Kelly, M.E.; Gu, T.-L.; Mack, J.S.; Silver, M.R.; Zhou, X.; Haack, H. Analysis of receptor tyrosine kinase ROS1-positive tumors in non–small cell lung cancer: Identification of a FIG-ROS1 fusion. Clin. Cancer Res. 2012, 18, 4449–4457. [Google Scholar] [CrossRef]
- Guaitoli, G.; Bertolini, F.; Bettelli, S.; Manfredini, S.; Maur, M.; Trudu, L.; Aramini, B.; Masciale, V.; Grisendi, G.; Dominici, M. Deepening the knowledge of ROS1 rearrangements in non-small cell lung cancer: Diagnosis, treatment, resistance and concomitant alterations. Int. J. Mol. Sci. 2021, 22, 12867. [Google Scholar] [CrossRef]
- Drilon, A.; Jenkins, C.; Iyer, S.; Schoenfeld, A.; Keddy, C.; Davare, M.A. ROS1-dependent cancers—Biology, diagnostics and therapeutics. Nat. Rev. Clin. Oncol. 2021, 18, 35–55. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, X.; Zhang, R.; Xu, Q.; Yang, H.; Lizaso, A.; Xu, C.; Liu, J.; Wang, W.; Ou, S.-H.I. Clinical and molecular factors that impact the efficacy of first-line crizotinib in ROS1-rearranged non-small-cell lung cancer: A large multicenter retrospective study. BMC Med. 2021, 19, 206. [Google Scholar] [CrossRef]
- Black, R.C.; Khurshid, H. NSCLC: An update of driver mutations, their role in pathogenesis and clinical significance. Rhode Isl. Med. J. 2015, 98, 25. [Google Scholar]
- Roys, A.; Chang, X.; Liu, Y.; Xu, X.; Wu, Y.; Zuo, D. Resistance mechanisms and potent-targeted therapies of ROS1-positive lung cancer. Cancer Chemother. Pharmacol. 2019, 84, 679–688. [Google Scholar] [CrossRef]
- Michelotti, A.; de Scordilli, M.; Bertoli, E.; De Carlo, E.; Del Conte, A.; Bearz, A. NSCLC as the paradigm of precision medicine at its finest: The rise of new druggable molecular targets for advanced disease. Int. J. Mol. Sci. 2022, 23, 6748. [Google Scholar] [CrossRef] [PubMed]
- D’Angelo, A.; Sobhani, N.; Chapman, R.; Bagby, S.; Bortoletti, C.; Traversini, M.; Ferrari, K.; Voltolini, L.; Darlow, J.; Roviello, G. Focus on ROS1-positive non-small cell lung cancer (NSCLC): Crizotinib, resistance mechanisms and the newer generation of targeted therapies. Cancers 2020, 12, 3293. [Google Scholar] [CrossRef]
- Lin, J.J.; Shaw, A.T. Recent advances in targeting ROS1 in lung cancer. J. Thorac. Oncol. 2017, 12, 1611–1625. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Lv, Y.; Zhang, C.; Fu, B.; Liu, Y.; Hu, J. Recent advances in the development of dual ALK/ROS1 inhibitors for non-small cell lung cancer therapy. Eur. J. Med. Chem. 2023, 257, 115477. [Google Scholar] [CrossRef]
- Vanajothi, R.; Vedagiri, H.; Al-Ansari, M.M.; Al-Humaid, L.A.; Kumpati, P. Pharmacophore based virtual screening, molecular docking and molecular dynamic simulation studies for finding ROS1 kinase inhibitors as potential drug molecules. J. Biomol. Struct. Dyn. 2022, 40, 3385–3399. [Google Scholar] [CrossRef]
- Wu, X.; Wang, Y.; Wan, S.; Zhang, J. Investigation on the binding mechanism of loratinib with the c-ros oncogene 1 (ROS1) receptor tyrosine kinase via molecular dynamics simulation and binding free energy calculations. J. Biomol. Struct. Dyn. 2018, 36, 3106–3113. [Google Scholar] [CrossRef]
- Pathak, D.; Chadha, N.; Silakari, O. Identification of non-resistant ROS-1 inhibitors using structure based pharmacophore analysis. J. Mol. Graph. Model. 2016, 70, 85–93. [Google Scholar] [CrossRef]
- Hasan, S.I. Exploring the Profiles of ROS1 Tyrosine Kinase: A Structural Analysis of G2032R and D2033N Mutations. Int. J. Appl. Basic Med. Res. 2025, 15, 4–10. [Google Scholar] [CrossRef]
- Petrovic, D.; Scott, J.S.; Bodnarchuk, M.S.; Lorthioir, O.; Boyd, S.; Hughes, G.M.; Lane, J.; Wu, A.; Hargreaves, D.; Robinson, J. Virtual screening in the cloud identifies potent and selective ROS1 kinase inhibitors. J. Chem. Inf. Model. 2022, 62, 3832–3843. [Google Scholar] [CrossRef]
- Schwede, T.; Kopp, J.; Guex, N.; Peitsch, M.C. SWISS-MODEL: An automated protein homology-modeling server. Nucleic Acids Res. 2003, 31, 3381–3385. [Google Scholar] [CrossRef]
- Bodade, R.; Beedkar, S.; Manwar, A.; Khobragade, C. Homology modeling and docking study of xanthine oxidase of Arthrobacter sp. XL26. Int. J. Biol. Macromol. 2010, 47, 298–303. [Google Scholar] [CrossRef]
- Laskowski, R.A.; MacArthur, M.W.; Moss, D.S.; Thornton, J.M. PROCHECK: A program to check the stereochemical quality of protein structures. J. Appl. Crystallogr. 1993, 26, 283–291. [Google Scholar] [CrossRef]
- Ntie-Kang, F.; Nyongbela, K.D.; Ayimele, G.A.; Shekfeh, S. “Drug-likeness” properties of natural compounds. Phys. Sci. Rev. 2019, 4, 20180169. [Google Scholar] [CrossRef]
- Almasoudi, H.H.; Nahari, M.H. Targeting Plasmodium falciparum Schizont Egress Antigen-1 in Infected Red Blood Cells: Docking-Based Fingerprinting, Density Functional Theory, Molecular Dynamics Simulations, and Binding Free Energy Analysis. Pharmaceuticals 2025, 18, 237. [Google Scholar] [CrossRef] [PubMed]
- Sabe, V.T.; Ntombela, T.; Jhamba, L.A.; Maguire, G.E.; Govender, T.; Naicker, T.; Kruger, H.G. Current trends in computer aided drug design and a highlight of drugs discovered via computational techniques: A review. Eur. J. Med. Chem. 2021, 224, 113705. [Google Scholar] [CrossRef]
- Arias, F.; Franco-Montalban, F.; Romero, M.; Carrión, M.D.; Camacho, M.E. Synthesis, bioevaluation and docking studies of new imidamide derivatives as nitric oxide synthase inhibitors. Biorg. Med. Chem. 2021, 44, 116294. [Google Scholar] [CrossRef]
- Adekoya, O.C.; Adekoya, G.J.; Sadiku, E.R.; Hamam, Y.; Ray, S.S. Application of DFT calculations in designing polymer-based drug delivery systems: An overview. Pharmaceutics 2022, 14, 1972. [Google Scholar] [CrossRef]
- Haque, A.; Alenezi, K.M.; Khan, M.W.A.; Soury, R.; Khan, M.S.; Ahamad, S.; Ahmad, S.; Gupta, D. In silico evaluation of 4-thiazolidinone-based inhibitors against the receptor for advanced glycation end products (RAGE). J. Biomol. Struct. Dyn. 2025, 43, 985–996. [Google Scholar] [CrossRef] [PubMed]
- Charest, A. The ROS1 receptor family. In Receptor Tyrosine Kinases: Family and Subfamilies; Springer: Berlin/Heidelberg, Germany, 2015; pp. 641–684. [Google Scholar]
- Muminovic, M.; Uribe, C.R.C.; Alvarez-Pinzon, A.; Shan, K.; Raez, L.E. Importance of ROS1 gene fusions in non-small cell lung cancer. Cancer Drug Resist. 2023, 6, 332. [Google Scholar] [CrossRef]
- Jóri, B.; Falk, M.; Hövel, I.; Weist, P.; Tiemann, M.; Heukamp, L.C.; Griesinger, F. Acquired G2032R resistance mutation in ROS1 to lorlatinib therapy detected with liquid biopsy. Curr. Oncol. 2022, 29, 6628–6634. [Google Scholar] [CrossRef] [PubMed]
- Oesterich, L.G.; Riess, J.W. ROS1. In Targeted Therapies for Lung Cancer; Springer: Berlin/Heidelberg, Germany, 2019; pp. 55–78. [Google Scholar]
- Cui, J.J.; Rogers, E.; Zhai, D.; Deng, W.; Huang, Z.; Whitten, J.; Li, Y. Ending the endless acquired tyrosine kinase resistance mutations—Design of TPX-0005, a multi-target ALK/ROS1/TRK inhibitor with broad spectrum activity against wild-type and mutants including ALK G1202R, ROS1 G2032R and TRKA G595R. Cancer Res. 2016, 76, 2133. [Google Scholar] [CrossRef]
- Cui, J.J.; Zhai, D.; Deng, W.; Rogers, E.; Huang, Z.; Whitten, J.; Lim, J.; Li, Y. Abstract B185: TPX-0005, a supreme ROS1 inhibitor, overcomes crizotinib-resistant ROS1 mutations including solvent front mutation G2032R and gatekeeper mutation L2026M. Mol. Cancer Ther. 2018, 17, B185. [Google Scholar] [CrossRef]
- Park, B.S.; Al-Sanea, M.M.; Abdelazem, A.Z.; Park, H.M.; Roh, E.J.; Park, H.-M.; Yoo, K.H.; Sim, T.; Tae, J.S.; Lee, S.H. Structure-based optimization and biological evaluation of trisubstituted pyrazole as a core structure of potent ROS1 kinase inhibitors. Bioorganic Med. Chem. 2014, 22, 3871–3878. [Google Scholar] [CrossRef]
- Guo, M.; Zuo, D.; Zhao, T.; Li, X.; Cao, J.; Qiu, Y.; Wei, S.; Zhai, X. Structure-based optimization identified novel furyl-containing 2, 4-diarylaminopyrimidine analogues as ALK/ROS1 dual inhibitors with anti-mutation effects. Eur. J. Med. Chem. 2021, 214, 113259. [Google Scholar] [CrossRef] [PubMed]
- Drilon, A.; Somwar, R.; Wagner, J.P.; Vellore, N.A.; Eide, C.A.; Zabriskie, M.S.; Arcila, M.E.; Hechtman, J.F.; Wang, L.; Smith, R.S. A novel crizotinib-resistant solvent-front mutation responsive to cabozantinib therapy in a patient with ROS1-rearranged lung cancer. Clin. Cancer Res. 2016, 22, 2351–2358. [Google Scholar] [CrossRef]
- Kim, S.; Chen, J.; Cheng, T.; Gindulyte, A.; He, J.; He, S.; Li, Q.; Shoemaker, B.A.; Thiessen, P.A.; Yu, B. PubChem 2019 update: Improved access to chemical data. Nucleic Acids Res. 2019, 47, D1102–D1109. [Google Scholar] [CrossRef]
- Sterling, T.; Irwin, J.J. ZINC 15–ligand discovery for everyone. J. Chem. Inf. Model. 2015, 55, 2324–2337. [Google Scholar] [CrossRef]
- Waterhouse, A.; Bertoni, M.; Bienert, S.; Studer, G.; Tauriello, G.; Gumienny, R.; Heer, F.T.; de Beer, T.A.P.; Rempfer, C.; Bordoli, L. SWISS-MODEL: Homology modelling of protein structures and complexes. Nucleic Acids Res. 2018, 46, W296–W303. [Google Scholar] [CrossRef] [PubMed]
- Consortium, U. UniProt: A worldwide hub of protein knowledge. Nucleic Acids Res. 2019, 47, D506–D515. [Google Scholar] [CrossRef] [PubMed]
- Laskowski, R.A. PDBsum: Summaries and analyses of PDB structures. Nucleic Acids Res. 2001, 29, 221–222. [Google Scholar] [CrossRef]
- Ioakimidis, L.; Thoukydidis, L.; Mirza, A.; Naeem, S.; Reynisson, J. Benchmarking the reliability of QikProp. Correlation between experimental and predicted values. QSAR Comb. Sci. 2008, 27, 445–456. [Google Scholar] [CrossRef]
- Ahamad, S.; Junaid, I.T.; Gupta, D. Computational design of novel tau-tubulin kinase 1 inhibitors for neurodegenerative diseases. Pharmaceuticals 2024, 17, 952. [Google Scholar] [CrossRef]
- Sandor, M.; Kiss, R.; Keseruű, G.r.M. Virtual fragment docking by Glide: A validation study on 190 protein−fragment complexes. J. Chem. Inf. Model. 2010, 50, 1165–1172. [Google Scholar] [CrossRef]
- Pagadala, N.S.; Syed, K.; Tuszynski, J. Software for molecular docking: A review. Biophys. Rev. 2017, 9, 91–102. [Google Scholar] [CrossRef] [PubMed]
- DeLano, W.L. Pymol: An open-source molecular graphics tool. CCP4 Newsl. Protein Crystallogr. 2002, 40, 82–92. [Google Scholar]
- Desmond, S. Schrödinger. 2021. Available online: https://www.schrodinger.com/desmond (accessed on 5 April 2024).
- Petersen, H.G. Accuracy and efficiency of the particle mesh Ewald method. J. Chem. Phys. 1995, 103, 3668–3679. [Google Scholar] [CrossRef]
- Steinbrecher, T.; Mobley, D.L.; Case, D.A. Nonlinear scaling schemes for Lennard-Jones interactions in free energy calculations. J. Chem. Phys. 2007, 127, 214108. [Google Scholar] [CrossRef]
- Ke, Q.; Gong, X.; Liao, S.; Duan, C.; Li, L. Effects of thermostats/barostats on physical properties of liquids by molecular dynamics simulations. J. Mol. Liq. 2022, 365, 120116. [Google Scholar] [CrossRef]
- Martyna, G.J.; Tobias, D.J.; Klein, M.L. Constant pressure molecular dynamics algorithms. J. Chem. Phys. 1994, 101, 4177–4189. [Google Scholar] [CrossRef]
- Mennucci, B.; Tomasi, J.; Cammi, R.; Cheeseman, J.R.; Frisch, M.J.; Devlin, F.J.; Gabriel, S.; Stephens, P.J. Polarizable continuum model (PCM) calculations of solvent effects on optical rotations of chiral molecules. J. Phys. Chem. A 2002, 106, 6102–6113. [Google Scholar] [CrossRef]
- Kruse, S.J.; Forbes, T.Z.; MacGillivray, L.R. Effects of Ionizing Radiation on Halogen-Bonded Dipyridyl-Naphthalenediimide Cocrystals. Cryst. Growth Des. 2025, 25, 4968–4973. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.