Recent Advances in Doxorubicin Formulation to Enhance Pharmacokinetics and Tumor Targeting
Abstract
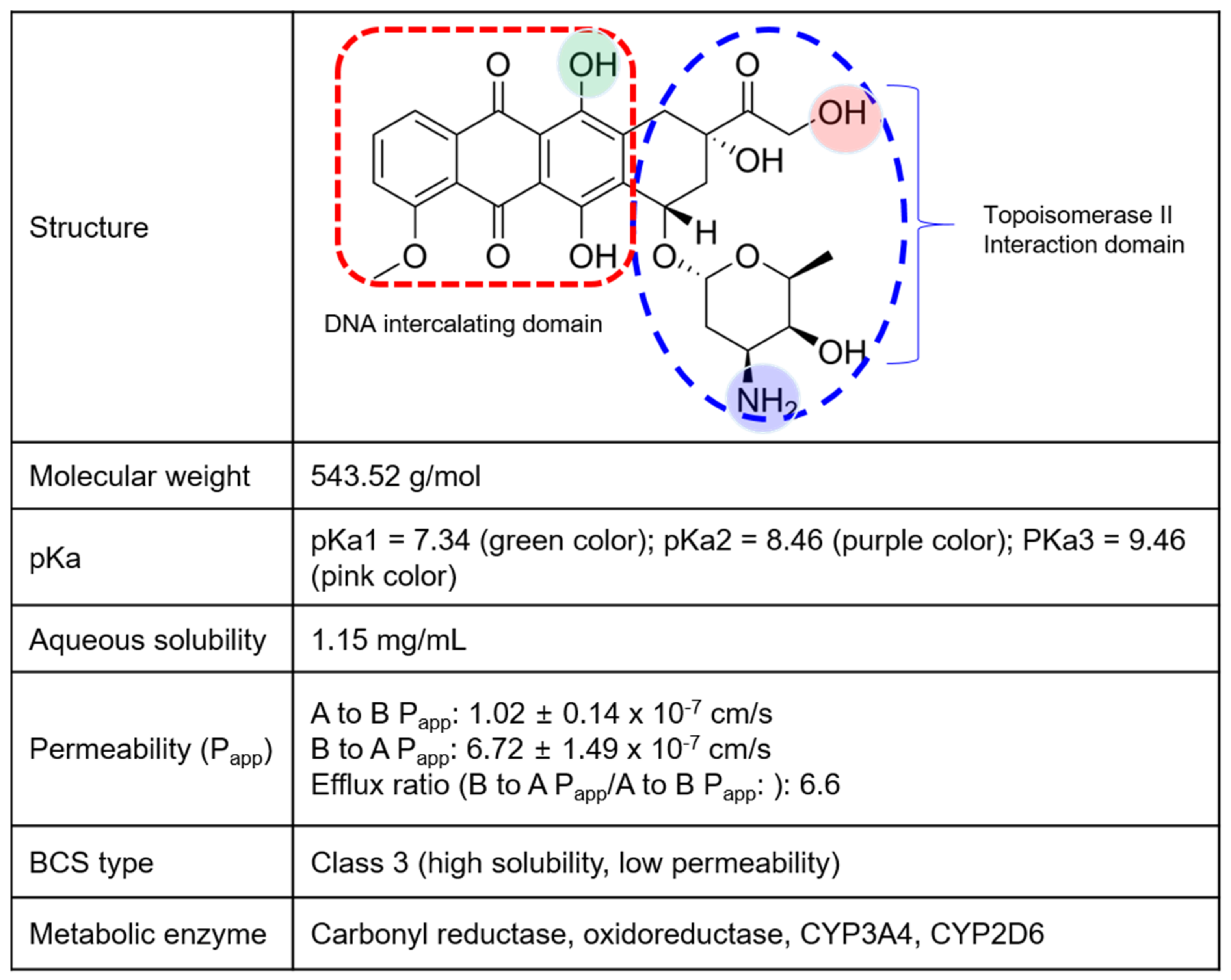
1. Introduction
2. DOX Formulations under Clinical Use or Trials
2.1. Liposomes
2.2. PMs
2.3. Polymeric Nanoparticles (PNPs)
2.4. Polymer-Drug Conjugates
| Carrier Type | Formulation and Route of Administration | Name | Clinical Results | References |
|---|---|---|---|---|
| Liposomes | PEGylated liposome (PL), IV | Doxil, approved by the FDA | EPR Treatment of ovarian cancer, Kaposi’s sarcoma, metastatic breast cancer, and multiple myeloma | [34] |
| PL, IV | Lipo-dox, approved by the FDA | EPR Therapeutic efficacy was not equivalent to that of Doxil in patients with ovarian cancer | [34] | |
| PL, IV | Caelyx, approved by the EMA | EPR Treatment of ovarian cancer, Kaposi’s sarcoma, metastatic breast cancer, and multiple myeloma | [26] | |
| PL, IV | Zolsketil, approved by the EMA | EPR Treatment of ovarian cancer, Kaposi’s sarcoma, metastatic breast cancer, and multiple myeloma | [26] | |
| PL, IV | JNS002 Phase III | EPR Treatment of ovarian cancer, primary fallopian tube cancer, and peritoneal cancer | [35] | |
| Non-PEGylated liposome, IV | Myocet, approved by the EMA | EPR A first-line treatment for metastatic breast cancer in adult women, in combination with cyclophosphamide | [36] | |
| Glutathione-conjugated PL, IV | GSH–PL–DOX Phase I/IIa | Brain targeting through a GSH transporter across the blood–brain barrier Safe and well tolerated with intracranial and extracranial antitumor activity | [39] | |
| HER2-targeted antibody anchoring PL, IV | HER2–PL–DOX Phase I | HER2-targeting Failed to provide beneficial effect superior to Doxil in patients with breast cancer | [34] | |
| EGFR-targeted antibody anchoring PL, IV | EGFR–PL–DOX Phase I | EGFR-targeting At 50 mg/m2, hand–foot syndrome, cardiotoxicity, or cumulative toxicity did not occur in any patient with glioblastoma and breast cancer | [40,41] | |
| PMs | PM with two nonionic pluronic block copolymers, IV | SP1049C Phase III | Inhibition of P-gp-mediated DOX efflux In a phase II trial involving 21 patients, 9 patients had a partial response and 8 patients had a minor response. The overall response rate was 47% | [46,47] |
| PEG–polyaspartic acid nanomicelle, IV | NK911 Phase II | EPR Well tolerated at 50 mg/m2 in 23 metastatic pancreatic cancer patients, and a partial response was achieved | [49] | |
| PNPs | Polyalkyl cyanoacrylate (PACA) nanoparticle, IV | Livatag Phase III | The phase III clinical trial did not achieve the desired effects in patients with advanced hepatocellular carcinoma | [51,52] |
| Polymer–drug conjugates | HPMA copolymer–GFLG–DOX, IV | FCE28069/PK1 Phase II | EPR and pinocytosis The maximum tolerable dose was 320 mg/m2, and no polymer-related toxicities were observed A considerable response occurred in some patients with breast and non-small cell lung cancer, but no response was noted in colorectal cancer patients Lack of biodegradability of the polymer main chain | [54,55,56] |
| HPMA copolymer–GFLG–DOX–galactosamine, IV | FCE28069/PK2 Phase II | Galactosamine-mediated uptake Liver-specific delivery using galactosamine-modified polymers, and a partial response was achieved in patients with liver cancer Grade 4 neutropenia and grade 3 mucositis | [57] |
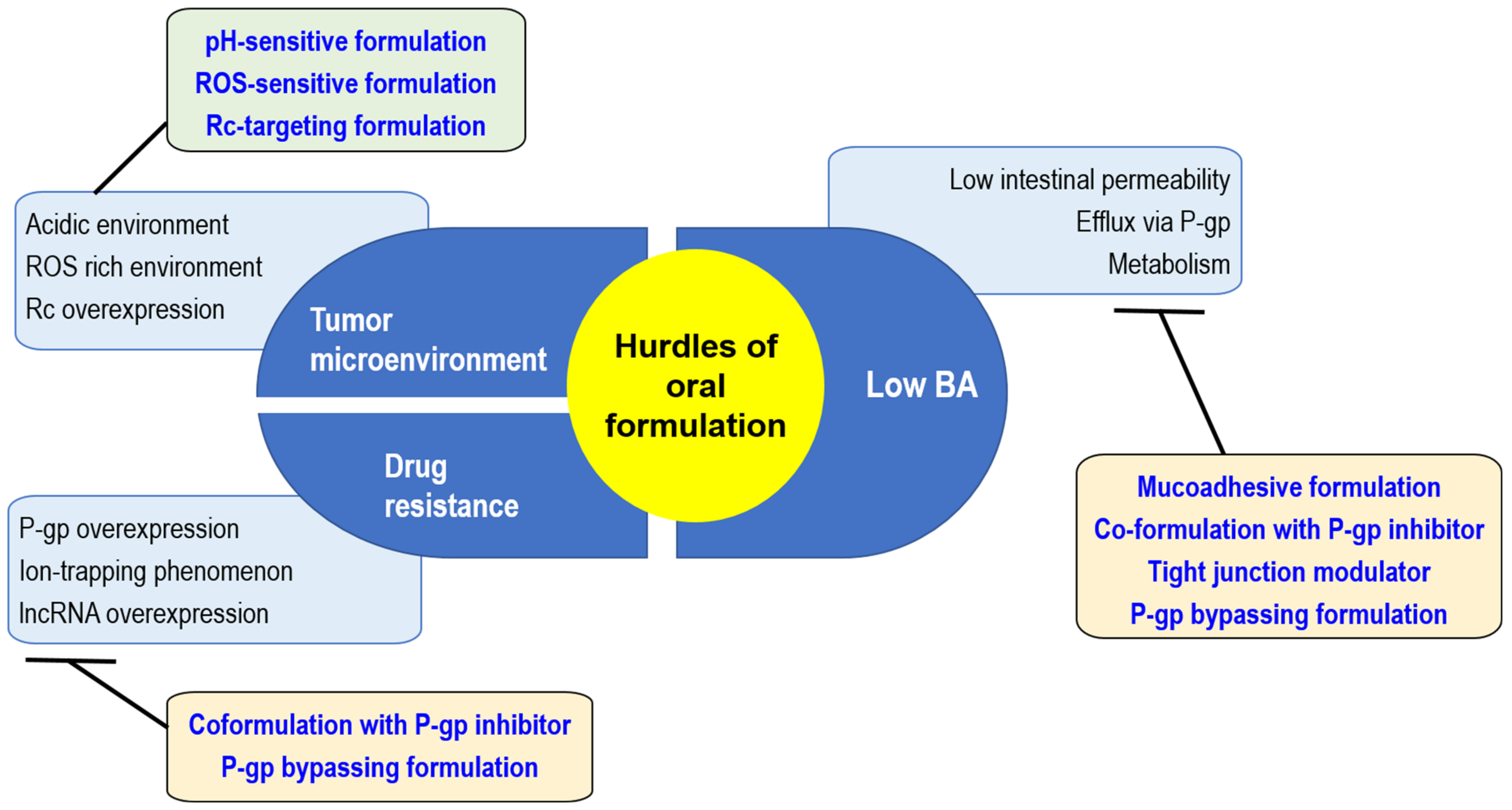
3. Obstacles in and Strategies for Formulating DOX to Enhance Oral BA and Tumor Targeting
3.1. Formulation Strategy Based on Tumor Microenvironments for the Targetability of DOX in the Preclinical Stage
3.1.1. pH-Sensitive Formulation
3.1.2. Reactive Oxygen Species (ROS)-Sensitive Formulation
3.1.3. Receptor (Rc)-Targeted Formulation
| Carrier–Type | Formulation & Route of Administration | Experimental Research | Findings | References |
|---|---|---|---|---|
| pH-sensitive PLs | DOX-loaded PL (DOPE: CHEMS: DOPE-PEG2000 = 5.8:3.7:0.5), IV | 4T1 tumor-bearing mice | Long circulating pH-sensitive liposome. Higher tumor uptake in 4T1 tumor-bearing mice | [63] |
| Healthy mice | Less QT interval prolongation on an electrocardiogram (reduced cardiotoxicity) | [64] | ||
| pH-sensitive PNPs | DOX and pHPMA conjugates via hydrazone bond (HPMA-NH-DOX), IV | 4T1, MCF-7 cell | A 5-fold faster DOX release in acidic intratumor (pH 6.5) and intratumor cellular (pH 5.5) environments than at pH 7.4 | [65] |
| pH-sensitive PMs | DOX-loaded micelle (DSPE-PEG2000/OA = 10:6), IV | 4T1 tumor-bearing mouse | pH-sensitive DOX release, 7-fold tumor shrinkage | [66] |
| ROS-sensitive liposomes | DOPE/Egg PC/DDA = 37.5/60/2.5%, IV | Walker 256 carcinosarcoma-bearing rat | A 3-fold faster DOX release at pH 5.0 than at pH 7.4 A 3-fold higher apoptosis rate | [68] |
| pH- and ROS-sensitive MSNs | Chitosan-folate conjugated MSN (DOX-MSN-SS-CH-FA), IV | C26-tumor-bearing mice | A 30% burst release in 0.1% H2O2 at pH 6.5 through the diselenide bond cleavage induced by the ROS signal The DOX-loaded liposome showed a 40-fold higher AUC than free DOX, efficient suppression of C26 tumor growth, and improved DOX distribution in tumors | [69] |
| Rc-targeted PMs | Folate targeted PM co-delivery of DOX and SIS3 (FA/DOX/SIS3), IV | SD rat | EPR and folate Rc-mediated endocytosis P-gp and BCRP inhibition by SIS3 6.1-fold increased AUC and 3.9-fold decreased clearance of DOX compared with free DOX | [77] |
| FA/DOX/SIS3, unilateral axillary injection | MCF-7/ADR bearing nude mice | EPR and folate Rc-mediated endocytosis P-gp and BCRP inhibition by SIS3 Increased DOX accumulation in tumor tissue Inhibited tumor growth and prolonged the lifetime in DOX resistant tumor mice | ||
| Rc-targeted and pH-sensitive PMs | HOD PM enclosed DOX-NN-VES, IV | MCF-7/ADR bearing nude mice | EPR and CD44-mediated endocytosis pH-sensitive DOX release at acidic intratumor organelles by hydrazone bond cleavage Increased DOX accumulation in tumor tissue Increased apoptosis and 2.28-fold decreased tumor weight compared with free DOX | [78] |
| Rc-targeted and pH-sensitive PNPs | Transferrin (Tf)- and poloxamer-integrated pH-sensitive PLGA NPs (Tf–DOX–PLGA), IV | NCI/ADR ovarian tumor cells | P-gp inhibition in tumor cells Significant decrease in cell viability from 80% to 20% compared with free DOX Arrested cell cycle in the G1 phase and increased apoptotic cell death by 2-fold | [79] |
3.2. Formulation Strategy for Overcoming DOX Resistance in the Preclinical Stage
3.2.1. Overexpression of P-gp in Tumor Cells
3.2.2. P-gp Inhibition in a Cellular Environment to Overcome Drug Resistance
3.2.3. Ion-Trapping Phenomenon
3.2.4. Long Noncoding RNA (lncRNA) Overexpression
3.2.5. Hypoglycemic Environment
| Carrier–Type | Formulation and Route of Administration | Experimental Model | Findings | References |
|---|---|---|---|---|
| PLs | PL incorporating DOX, ICG, and P-gp inhibitor quinine (ICG + PLDQ), IV | HT-29 MDR1 positive xenograft mice | P-gp inhibition in tumor cells Increased cellular uptake of DOX and reduction in tumor volume by 25% Increase in survival rate by 2-fold | [88] |
| Liposomes | DOX-loaded apolipoprotein A1-modified cationic liposome (ApoA1-LipDOX), IV | 4T1 tumor-bearing mice | P-gp inhibition in tumor cells with 3-fold higher DOX concentration in tumor tissue Decrease in tumor volume by 3-fold | [92] |
| DOX and palmitoyl ascorbate (PA)-loaded liposome (DOX-PA-liposome), IV | MCF-7 cells | P-gp inhibition in tumor cells Increased DOX uptake in MCF-7 cells by 2.5-fold | [102] | |
| SD rats | Elevation in DOX AUC by 10-fold compared with DOX–liposome AUC | |||
| MCF-7 breast cancer bearing mice | Tumor size decreased by 2-fold compared with DOX–liposome | |||
| DOX and NaHCO3-loaded HSPC-m2000PEG DSPE–liposome, IV | 4T1 breast cancer-bearing mouse | Increase in DOX concentration by 2–3-fold The average extracellular pH in tumor tissue increased to 7.38 | [109] | |
| NaHCO3-loaded liposome and Doxil combination, IV | colon26 tumor-bearing mouse | Decrease in tumor size by 9- and 2-fold compared with free DOX and Doxil, respectively | [111] | |
| PMs | DOX-loaded Pluronic F127 micelles with pH-sensitive poly(acrylic acid) at two terminals (PAA-PF127-PAA-PM), IV | Walker 256 carcinosarcoma-bearing mice | A 3-fold faster DOX release at pH 5.0 than at pH7.4 A 3-fold higher apoptosis rate compared with free DOX | [104] |
| PNPs | PEG-iPUTDN + NIR exposure, IP | H69AR lung cancer-bearing mice | Bypassed the P-gp-mediated efflux TPP-conjugated DOX was efficiently accumulated in mitochondria Tumor volume and weight decreased by 10-fold | [95] |
| MSNs | GOD@SiO2-Arg and DOX-MSN hydrogels, SC | MCF-7/ADR cells | Arg generated NO in the presence of H2O2 and decreased P-gp expression. Low pH facilitated DOX release from DOX–MSN and increased its therapeutic efficacy | [103] |
| MCF-7/ADR xenograft mice | Tumor volume reduced significantly by 8-fold without causing significant histological abnormalities Survival rate increased by 2-fold |
3.3. Challenges in the Development of Oral Formulations
3.3.1. Low Intestinal Permeability
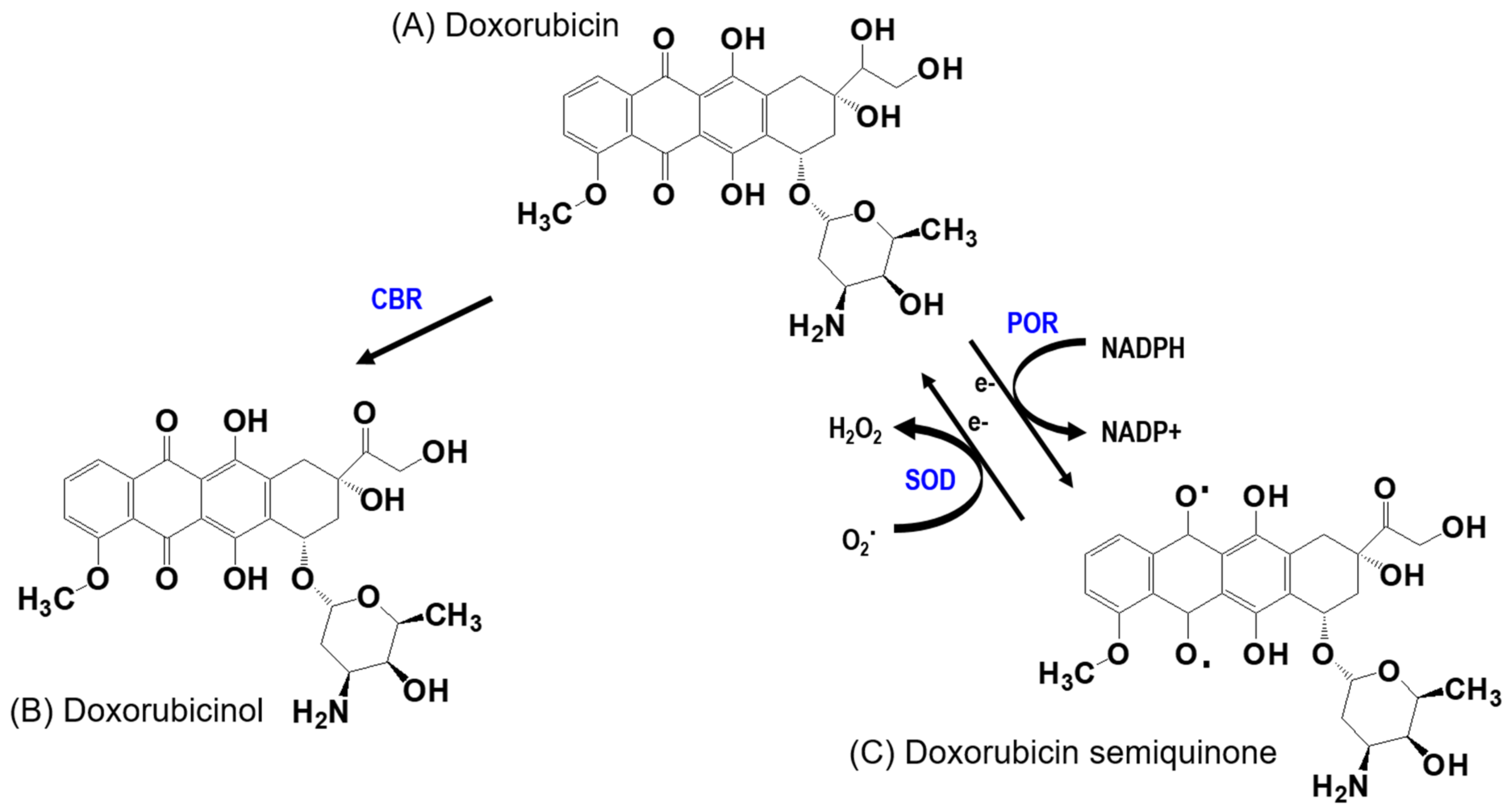
3.3.2. High First-Pass Metabolism of DOX
3.4. Formulation Strategy for Overcoming Low Oral BA in the Preclinical Stage
3.4.1. Coadministration with a P-gp Inhibitor to Increase Oral Absorption
3.4.2. Mucoadhesive Formulation
3.4.3. Formulations to Increase the Oral BA of DOX
- (1)
- PMs
- (2)
- PNPs
- (3)
- Multilayer micro-dispersing system (MMS)
- (4)
- MSNs
- (5)
- Clay mineral formulation
| Carrier–Type | Formulation & Route of Administration | Experimental Model | Findings | References |
|---|---|---|---|---|
| PMs | Linolenic acid–chitosan-based PMs (DOX-CS-LA), PO | SD rat | Mucoadhesive formulation Targeting the intestinal fatty acid transporter Increase in relative BA by 166% compared with that of free DOX | [152] |
| Lysine-linked ditocopherol polyethylene glycol 2000 succinate (PLV2K-DOX), PO | SD rat | Intestinal permeability of PLV2K–DOX was 3.19-, 1.61-, and 1.80-fold higher than that of free DOX in the duodenum, jejunum, and ileum Orally administered PLV2K–DOX showed 5.6-fold higher AUC than free DOX in rats | [153,154] | |
| Oleanolic acid conjugated methoxy-poly (ethylene glycol)-poly (D, L-lactide) (mPEG-PLA-OA), PO | Wistar rats | A 30-fold increased DOX circulation time and 30-fold reduced clearance time | [155] | |
| PNPs | DOX-loaded poly (lactic-co-glycolic acid) (PLGA) NPs, PO | SD rats | BA enhancement by 363% and reduced cardiotoxicity | [165] |
| Breast cancer bearing rats | Reduced tumor size, increased survival rate, and reduced cardiotoxicity | |||
| Chitosan coated–daunorubicin PLGA–NPs, PO | Wistar rats | Compared with free daunorubicin, a 11.3-fold higher AUC and 2.8-fold delay in the elimination of daunorubicin from the plasma | [168] | |
| PEGylated-DOX-loaded-PLGA–NPs, PO | Wistar rats | Compared with free DOX, a 11.8-fold higher AUC and 2.1-fold delay in the elimination of DOX from the plasma | [167] | |
| Chitosan modified chitosan diacetate (CDA) and chitosan triacetate (CTA)-NPs, PO | MCF-7 cells | Approximately 2-fold increased permeability of DOX in MCF-7 cells | [169] | |
| SD rats | Compared with free DOX, sustained release for 24 h, and 3-fold increase in the AUC of DOX–CTA NPs | |||
| Intestine-penetrating, pH-sensitive and double layered NPs, PO | H22-tumor bearing mice | Relative BA of 75.4% with effective inhibition of tumor growth DOX concentrations in major tissues did not exceed the maximum tolerated concentration Approximately 40% of the absorbed DOX accumulated in the tumor tissue | [170] | |
| Sodium caseinate (NaCN) NPs, PO | 4T1-breast cancer bearing mice | A 8-fold tumor shrinkage compared with that of free DOX Following the oral administration of DOX–NaCN NPs, DOX in tumor tissues showed 8.34-fold higher accumulation than IV DOX and 1.27-fold higher accumulation than IV DOX–NaCN NPs | [172] | |
| MMS | Mutilayer alginate beads with codelivery of chitosan-DOX nanogel and quercetin (DOX:CS/CMCS-NGs /Qu-M-ALG-Beads), PO | SD rats | pH-sensitive release at pH > 7.0. Chitosan increased DOX absorption via mucoadhesion and tight-junction opening Quercetin increased DOX absorption by inhibiting P-gp. BA of DOX:CS /CMCS-NGs/Qu-M-ALG-beads was 55.8% | [173] |
| MSNs | DOX loaded MSN (DOX-MSN), PO | SD rats | DOX–MSN with a rod shape and size of 200 nm showed 5.9-fold enhancement in relative BA compared with free DOX | [175] |
4. Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Gotwals, P.; Cameron, S.; Cipolletta, D.; Cremasco, V.; Crystal, A.; Hewes, B.; Mueller, B.; Quaratino, S.; Sabatos-Peyton, C.; Petruzzelli, L.; et al. Prospects for combining targeted and conventional cancer therapy with immunotherapy. Nat. Rev. Cancer 2017, 17, 286–301. [Google Scholar] [CrossRef] [PubMed]
- Le Lay, K.; Myon, E.; Hill, S.; Riou-Franca, L.; Scott, D.; Sidhu, M.; Dunlop, D.; Launois, R. Comparative cost-minimisation of oral and intravenous chemotherapy for first-line treatment of non-small cell lung cancer in the UK NHS system. Eur. J. Health Econ. 2007, 8, 145–151. [Google Scholar] [CrossRef] [PubMed]
- Cassidy, J.; Douillard, J.Y.; Twelves, C.; McKendrick, J.J.; Scheithauer, W.; Bustová, I.; Johnston, P.G.; Lesniewski-Kmak, K.; Jelic, S.; Fountzilas, G.; et al. Pharmacoeconomic analysis of adjuvant oral capecitabine vs intravenous 5-FU/LV in Dukes’ C colon cancer: The X-ACT trial. Br. J. Cancer 2006, 94, 1122–1129. [Google Scholar] [CrossRef] [PubMed]
- Terwogt, J.M.; Schellens, J.H.; Huinink, W.W.; Beijnen, J.H. Clinical pharmacology of anticancer agents in relation to formulations and administration routes. Cancer Treat. Rev. 1999, 25, 83–101. [Google Scholar] [CrossRef]
- Tu, L.; Cheng, M.; Sun, Y.; Fang, Y.; Liu, J.; Liu, W.; Feng, J.; Jin, Y. Fabrication of ultra-small nanocrystals by formation of hydrogen bonds: In vitro and in vivo evaluation. Int. J. Pharm. 2020, 573, 118730. [Google Scholar] [CrossRef]
- Mohammad, I.S.; Hu, H.; Yin, L.; He, W. Drug nanocrystals: Fabrication methods and promising therapeutic applications. Int. J. Pharm. 2019, 562, 187–202. [Google Scholar] [CrossRef]
- Lozoya-Agullo, I.; González-Álvarez, I.; González-Álvarez, M.; Merino-Sanjuán, M.; Bermejo, M. Development of an ion-pair to improve the colon permeability of a low permeability drug: Atenolol. Eur. J. Pharm. Sci. 2016, 93, 334–340. [Google Scholar] [CrossRef]
- Zhao, J.; Yang, J.; Xie, Y. Improvement strategies for the oral bioavailability of poorly water-soluble flavonoids: An overview. Int. J. Pharm. 2019, 570, 118642. [Google Scholar] [CrossRef]
- Yang, S.H.; Lee, M.G. Dose-independent pharmacokinetics of ondansetron in rats: Contribution of hepatic and intestinal first-pass effects to low bioavailability. Biopharm. Drug Dispos. 2008, 29, 414–426. [Google Scholar] [CrossRef]
- Rivankar, S. An overview of doxorubicin formulations in cancer therapy. J. Cancer Res. Ther. 2014, 10, 853–858. [Google Scholar] [CrossRef]
- Tewey, K.M.; Rowe, T.C.; Yang, L.; Halligan, B.D.; Liu, L.F. Adriamycin-induced DNA damage mediated by mammalian DNA topoisomerase II. Science 1984, 226, 466–468. [Google Scholar] [CrossRef] [PubMed]
- Dong, K.C.; Berger, J.M. Structural basis for gate-DNA recognition and bending by type IIA topoisomerases. Nature 2007, 450, 1201–1205. [Google Scholar] [CrossRef] [PubMed]
- Nitiss, J.L. DNA topoisomerase II and its growing repertoire of biological functions. Nat. Rev. Cancer 2009, 9, 327–337. [Google Scholar] [CrossRef] [PubMed]
- Nitiss, J.L. Targeting DNA topoisomerase II in cancer chemotherapy. Nat. Rev. Cancer 2009, 9, 338–350. [Google Scholar] [CrossRef]
- Perego, P.; Corna, E.; De Cesare, M.; Gatti, L.; Polizzi, D.; Pratesi, G.; Supino, R.; Zunino, F. Role of apoptosis and apoptosis-related genes in cellular response and antitumor efficacy of anthracyclines. Curr. Med. Chem. 2001, 8, 31–37. [Google Scholar] [CrossRef]
- Pang, B.; Qiao, X.; Janssen, L.; Velds, A.; Groothuis, T.; Kerkhoven, R.; Nieuwland, M.; Ovaa, H.; Rottenberg, S.; van Tellingen, O.; et al. Drug-induced histone eviction from open chromatin contributes to the chemotherapeutic effects of doxorubicin. Nat. Commun. 2013, 4, 1908. [Google Scholar] [CrossRef]
- Yang, F.; Kemp, C.J.; Henikoff, S. Doxorubicin enhances nucleosome turnover around promoters. Curr. Biol. 2013, 23, 782–787. [Google Scholar] [CrossRef]
- Gorovsky, M.A.; Keevert, J.B. Absence of histone F1 in a mitotically dividing, genetically inactive nucleus. Proc. Natl. Acad. Sci. USA 1975, 72, 2672–2676. [Google Scholar] [CrossRef]
- Kim, J.-E.; Cho, H.-J.; Kim, J.S.; Shim, C.-K.; Chung, S.-J.; Oak, M.-H.; Yoon, I.-S.; Kim, D.-D. The limited intestinal absorption via paracellular pathway is responsible for the low oral bioavailability of doxorubicin. Xenobiotica 2013, 43, 579–591. [Google Scholar] [CrossRef]
- Bukowski, K.; Kciuk, M.; Kontek, R. Mechanisms of Multidrug Resistance in Cancer Chemotherapy. Int. J. Mol. Sci. 2020, 21, 3233. [Google Scholar] [CrossRef]
- Lage, H. ABC-transporters: Implications on drug resistance from microorganisms to human cancers. Int. J. Antimicrob. Agents 2003, 22, 188–199. [Google Scholar] [CrossRef] [PubMed]
- Hershman, D.L.; McBride, R.B.; Eisenberger, A.; Tsai, W.Y.; Grann, V.R.; Jacobson, J.S. Doxorubicin, cardiac risk factors, and cardiac toxicity in elderly patients with diffuse B-cell non-Hodgkin’s lymphoma. J. Clin. Oncol. 2008, 26, 3159–3165. [Google Scholar] [CrossRef] [PubMed]
- Gou, M.; Shi, H.; Guo, G.; Men, K.; Zhang, J.; Zheng, L.; Li, Z.; Luo, F.; Qian, Z.; Zhao, X.; et al. Improving anticancer activity and reducing systemic toxicity of doxorubicin by self-assembled polymeric micelles. Nanotechnology 2011, 22, 095102. [Google Scholar] [CrossRef] [PubMed]
- Bangham, A.D.; Standish, M.M.; Watkins, J.C. Diffusion of univalent ions across the lamellae of swollen phospholipids. J. Mol. Biol. 1965, 13, 238–252. [Google Scholar] [CrossRef]
- Choi, M.K.; Nam, S.J.; Ji, H.Y.; Park, M.J.; Choi, J.S.; Song, I.S. Comparative pharmacokinetics and pharmacodynamics of a novel sodium-glucose cotransporter 2 Inhibitor, DWP16001, with dapagliflozin and ipragliflozin. Pharmaceutics 2020, 12, 268. [Google Scholar] [CrossRef]
- Taléns-Visconti, R.; Díez-Sales, O.; de Julián-Ortiz, J.V.; Nácher, A. Nanoliposomes in Cancer Therapy: Marketed Products and Current Clinical Trials. Int. J. Mol. Sci. 2022, 23, 4249. [Google Scholar] [CrossRef] [PubMed]
- Green, A.E.; Rose, P.G. Pegylated liposomal doxorubicin in ovarian cancer. Int. J. Nanomedicine 2006, 1, 229–239. [Google Scholar]
- Gheibi Hayat, S.M.; Jaafari, M.R.; Hatamipour, M.; Jamialahmadi, T.; Sahebkar, A. Harnessing CD47 mimicry to inhibit phagocytic clearance and enhance anti-tumor efficacy of nanoliposomal doxorubicin. Expert. Opin. Drug Deliv. 2020, 17, 1049–1058. [Google Scholar] [CrossRef]
- Waterhouse, D.N.; Tardi, P.G.; Mayer, L.D.; Bally, M.B. A comparison of liposomal formulations of doxorubicin with drug administered in free form: Changing toxicity profiles. Drug Saf. 2001, 24, 903–920. [Google Scholar] [CrossRef]
- Jain, R.K. Normalization of tumor vasculature: An emerging concept in antiangiogenic therapy. Science 2005, 307, 58–62. [Google Scholar] [CrossRef]
- Fang, C.; Shi, B.; Pei, Y.Y.; Hong, M.H.; Wu, J.; Chen, H.Z. In vivo tumor targeting of tumor necrosis factor-alpha-loaded stealth nanoparticles: Effect of MePEG molecular weight and particle size. Eur. J. Pharm. Sci. 2006, 27, 27–36. [Google Scholar] [CrossRef] [PubMed]
- Ambegia, E.; Ansell, S.; Cullis, P.; Heyes, J.; Palmer, L.; MacLachlan, I. Stabilized plasmid-lipid particles containing PEG-diacylglycerols exhibit extended circulation lifetimes and tumor selective gene expression. Biochim. Biophys. Acta 2005, 1669, 155–163. [Google Scholar] [CrossRef]
- Hatakeyama, H.; Akita, H.; Kogure, K.; Oishi, M.; Nagasaki, Y.; Kihira, Y.; Ueno, M.; Kobayashi, H.; Kikuchi, H.; Harashima, H. Development of a novel systemic gene delivery system for cancer therapy with a tumor-specific cleavable PEG-lipid. Gene Ther. 2007, 14, 68–77. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Lin, M. The Synthesis of Nano-Doxorubicin and its Anticancer Effect. Anticancer Agents Med. Chem. 2021, 21, 2466–2477. [Google Scholar] [CrossRef] [PubMed]
- Motohashi, T.; Yabuno, A.; Michimae, H.; Ohishi, T.; Nonaka, M.; Takano, M.; Nishio, S.; Fujiwara, H.; Fujiwara, K.; Kondo, E.; et al. Randomized phase III trial comparing pegylated liposomal doxorubicin (PLD) at 50 mg/m² versus 40 mg/m² in patients with platinum-refractory and -resistant ovarian carcinoma: The JGOG 3018 Trial. J. Gynecol. Oncol. 2021, 32, e9. [Google Scholar] [CrossRef]
- Swenson, C.E.; Perkins, W.R.; Roberts, P.; Janoff, A.S. Liposome technology and the development of Myocet™ (liposomal doxorubicin citrate). Breast 2001, 10, 1–7. [Google Scholar] [CrossRef]
- Rafiyath, S.M.; Rasul, M.; Lee, B.; Wei, G.; Lamba, G.; Liu, D. Comparison of safety and toxicity of liposomal doxorubicin vs. conventional anthracyclines: A meta-analysis. Exp. Hematol. Oncol. 2012, 1, 10. [Google Scholar] [CrossRef]
- Hudes, G. Boosting bioavailability of topotecan: What do we gain? J. Clin. Oncol. 2002, 20, 2918–2919. [Google Scholar] [CrossRef]
- Kerklaan, B.M.; Jager, A.; Aftimos, P.; Dieras, V.; Altintas, S.; Anders, C.; Arnedos, M.; Gelderblom, H.; Soetekouw, P.; Gladdines, W.; et al. NT-23 phase 1/2a study of gluthathione PEGylated liposomal doxorubicin (2B3-101) in breast cancer patients with brain metastasis (BCMB) or recurrent high grade gliomas (HGC). Neuro-Oncol. 2014, 16, v163. [Google Scholar] [CrossRef]
- Wicki, A.; Ritschard, R.; Loesch, U.; Deuster, S.; Rochlitz, C.; Mamot, C. Large-scale manufacturing of GMP-compliant anti-EGFR targeted nanocarriers: Production of doxorubicin-loaded anti-EGFR-immunoliposomes for a first-in-man clinical trial. Int. J. Pharm. 2015, 484, 8–15. [Google Scholar] [CrossRef]
- Mamot, C.; Ritschard, R.; Wicki, A.; Stehle, G.; Dieterle, T.; Bubendorf, L.; Hilker, C.; Deuster, S.; Herrmann, R.; Rochlitz, C. Tolerability, safety, pharmacokinetics, and efficacy of doxorubicin-loaded anti-EGFR immunoliposomes in advanced solid tumours: A phase 1 dose-escalation study. Lancet Oncol. 2012, 13, 1234–1241. [Google Scholar] [CrossRef] [PubMed]
- Kumari, P.; Ghosh, B.; Biswas, S. Nanocarriers for cancer-targeted drug delivery. J. Drug Target. 2016, 24, 179–191. [Google Scholar] [CrossRef]
- Oerlemans, C.; Bult, W.; Bos, M.; Storm, G.; Nijsen, J.F.; Hennink, W.E. Polymeric micelles in anticancer therapy: Targeting, imaging and triggered release. Pharm. Res. 2010, 27, 2569–2589. [Google Scholar] [CrossRef]
- Kwon, M.; Lim, D.Y.; Lee, C.H.; Jeon, J.H.; Choi, M.K.; Song, I.S. Enhanced intestinal absorption and pharmacokinetic modulation of berberine and its metabolites through the inhibition of P-glycoprotein and intestinal metabolism in rats using a berberine mixed micelle formulation. Pharmaceutics 2020, 12, 882. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.A.; Yoon, Y.H.; Choi, K.; Kwon, M.; Goo, S.H.; Cha, J.S.; Choi, M.K.; Lee, H.S.; Song, I.S. Enhanced oral bioavailability of morin administered in mixed micelle formulation with PluronicF127 and Tween80 in rats. Biol. Pharm. Bull. 2015, 38, 208–217. [Google Scholar] [CrossRef] [PubMed]
- Danson, S.; Ferry, D.; Alakhov, V.; Margison, J.; Kerr, D.; Jowle, D.; Brampton, M.; Halbert, G.; Ranson, M. Phase I dose escalation and pharmacokinetic study of pluronic polymer-bound doxorubicin (SP1049C) in patients with advanced cancer. Br. J. Cancer 2004, 90, 2085–2091. [Google Scholar] [CrossRef]
- Valle, J.W.; Armstrong, A.; Newman, C.; Alakhov, V.; Pietrzynski, G.; Brewer, J.; Campbell, S.; Corrie, P.; Rowinsky, E.K.; Ranson, M. A phase 2 study of SP1049C, doxorubicin in P-glycoprotein-targeting pluronics, in patients with advanced adenocarcinoma of the esophagus and gastroesophageal junction. Investig. New Drugs 2011, 29, 1029–1037. [Google Scholar] [CrossRef]
- Varela-Moreira, A.; Shi, Y.; Fens, M.H.A.M.; Lammers, T.; Hennink, W.E.; Schiffelers, R.M. Clinical application of polymeric micelles for the treatment of cancer. Mater. Chem. Front. 2017, 1, 1485–1501. [Google Scholar] [CrossRef]
- Matsumura, Y.; Hamaguchi, T.; Ura, T.; Muro, K.; Yamada, Y.; Shimada, Y.; Shirao, K.; Okusaka, T.; Ueno, H.; Ikeda, M.; et al. Phase I clinical trial and pharmacokinetic evaluation of NK911, a micelle-encapsulated doxorubicin. Br. J. Cancer 2004, 91, 1775–1781. [Google Scholar] [CrossRef]
- Razak, S.A.; Mohd Gazzali, A.; Fisol, F.A.; Abdulbaqi, I.M.; Parumasivam, T.; Mohtar, N.; Wahab, H.A. Advances in Nanocarriers for Effective Delivery of Docetaxel in the Treatment of Lung Cancer: An Overview. Cancers 2021, 13, 400. [Google Scholar] [CrossRef]
- Mura, S.; Fattal, E.; Nicolas, J. From poly(alkyl cyanoacrylate) to squalene as core material for the design of nanomedicines. J. Drug Target. 2019, 27, 470–501. [Google Scholar] [CrossRef] [PubMed]
- Graur, F.; Puia, A.; Mois, E.I.; Moldovan, S.; Pusta, A.; Cristea, C.; Cavalu, S.; Puia, C.; Al Hajjar, N. Nanotechnology in the Diagnostic and Therapy of Hepatocellular Carcinoma. Materials 2022, 15, 3893. [Google Scholar] [CrossRef] [PubMed]
- Ekladious, I.; Colson, Y.L.; Grinstaff, M.W. Polymer-drug conjugate therapeutics: Advances, insights and prospects. Nat. Rev. Drug Discov. 2019, 18, 273–294. [Google Scholar] [CrossRef] [PubMed]
- Duncan, R. Development of HPMA copolymer-anticancer conjugates: Clinical experience and lessons learnt. Adv. Drug Deliv. Rev. 2009, 61, 1131–1148. [Google Scholar] [CrossRef] [PubMed]
- Vasey, P.A.; Kaye, S.B.; Morrison, R.; Twelves, C.; Wilson, P.; Duncan, R.; Thomson, A.H.; Murray, L.S.; Hilditch, T.E.; Murray, T.; et al. Phase I clinical and pharmacokinetic study of PK1 [N-(2-hydroxypropyl)methacrylamide copolymer doxorubicin]: First member of a new class of chemotherapeutic agents-drug-polymer conjugates. Cancer Research Campaign Phase I/II Committee. Clin. Cancer Res. 1999, 5, 83–94. [Google Scholar] [PubMed]
- Seymour, L.W.; Ferry, D.R.; Kerr, D.J.; Rea, D.; Whitlock, M.; Poyner, R.; Boivin, C.; Hesslewood, S.; Twelves, C.; Blackie, R.; et al. Phase II studies of polymer-doxorubicin (PK1, FCE28068) in the treatment of breast, lung and colorectal cancer. Int. J. Oncol. 2009, 34, 1629–1636. [Google Scholar] [CrossRef]
- Seymour, L.W.; Ferry, D.R.; Anderson, D.; Hesslewood, S.; Julyan, P.J.; Poyner, R.; Doran, J.; Young, A.M.; Burtles, S.; Kerr, D.J. Hepatic drug targeting: Phase I evaluation of polymer-bound doxorubicin. J. Clin. Oncol. 2002, 20, 1668–1676. [Google Scholar] [CrossRef]
- Eisenmann, E.D.; Talebi, Z.; Sparreboom, A.; Baker, S.D. Boosting the oral bioavailability of anticancer drugs through intentional drug-drug interactions. Basic Clin. Pharmacol. Toxicol. 2022, 130 (Suppl. 1), 23–35. [Google Scholar] [CrossRef]
- Boedtkjer, E.; Pedersen, S.F. The Acidic Tumor Microenvironment as a Driver of Cancer. Annu. Rev. Physiol. 2020, 82, 103–126. [Google Scholar] [CrossRef] [PubMed]
- Simões, S.; Moreira, J.N.; Fonseca, C.; Düzgüneş, N.; de Lima, M.C. On the formulation of pH-sensitive liposomes with long circulation times. Adv. Drug Deliv. Rev. 2004, 56, 947–965. [Google Scholar] [CrossRef]
- Karanth, H.; Murthy, R.S. pH-sensitive liposomes--principle and application in cancer therapy. J. Pharm. Pharmacol. 2007, 59, 469–483. [Google Scholar] [CrossRef]
- Koudelka, S.; Turanek Knotigova, P.; Masek, J.; Prochazka, L.; Lukac, R.; Miller, A.D.; Neuzil, J.; Turanek, J. Liposomal delivery systems for anti-cancer analogues of vitamin E. J. Control. Release 2015, 207, 59–69. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira Silva, J.; Miranda, S.E.M.; Leite, E.A.; de Paula Sabino, A.; Borges, K.B.G.; Cardoso, V.N.; Cassali, G.D.; Guimarães, A.G.; Oliveira, M.C.; de Barros, A.L.B. Toxicological study of a new doxorubicin-loaded pH-sensitive liposome: A preclinical approach. Toxicol. Appl. Pharmacol. 2018, 352, 162–169. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira Silva, J.; Fernandes, R.S.; Ramos Oda, C.M.; Ferreira, T.H.; Machado Botelho, A.F.; Martins Melo, M.; de Miranda, M.C.; Assis Gomes, D.; Dantas Cassali, G.; Townsend, D.M.; et al. Folate-coated, long-circulating and pH-sensitive liposomes enhance doxorubicin antitumor effect in a breast cancer animal model. Biomed. Pharmacother. 2019, 118, 109323. [Google Scholar] [CrossRef] [PubMed]
- Bobde, Y.; Biswas, S.; Ghosh, B. PEGylated N-(2 hydroxypropyl) methacrylamide-doxorubicin conjugate as pH-responsive polymeric nanoparticles for cancer therapy. React. Funct. Polym. 2020, 151, 104561. [Google Scholar] [CrossRef]
- Cavalcante, C.H.; Fernandes, R.S.; de Oliveira Silva, J.; Oda, C.M.R.; Leite, E.A.; Cassali, G.D.; Charlie-Silva, I.; Fernandes, B.H.V.; Ferreira, L.A.M.; de Barros, A.L.B. Doxorubicin-loaded pH-sensitive micelles: A promising alternative to enhance antitumor activity and reduce toxicity. Biomed. Pharmacother. 2021, 134, 111076. [Google Scholar] [CrossRef]
- Perillo, B.; Di Donato, M.; Pezone, A.; Di Zazzo, E.; Giovannelli, P.; Galasso, G.; Castoria, G.; Migliaccio, A. ROS in cancer therapy: The bright side of the moon. Exp. Mol. Med. 2020, 52, 192–203. [Google Scholar] [CrossRef]
- Mirhadi, E.; Mashreghi, M.; Askarizadeh, A.; Mehrabian, A.; Alavizadeh, S.H.; Arabi, L.; Badiee, A.; Jaafari, M.R. Redox-sensitive doxorubicin liposome: A formulation approach for targeted tumor therapy. Sci. Rep. 2022, 12, 11310. [Google Scholar] [CrossRef]
- Bhavsar, D.B.; Patel, V.; Sawant, K.K. Design and characterization of dual responsive mesoporous silica nanoparticles for breast cancer targeted therapy. Eur. J. Pharm. Sci. 2020, 152, 105428. [Google Scholar] [CrossRef]
- Mirzaei, S.; Zarrabi, A.; Hashemi, F.; Zabolian, A.; Saleki, H.; Azami, N.; Hamzehlou, S.; Farahani, M.V.; Hushmandi, K.; Ashrafizadeh, M. Nrf2 signaling pathway in chemoprotection and doxorubicin resistance: Potential application in drug discovery. Antioxidants 2021, 10, 349. [Google Scholar] [CrossRef]
- Calcabrini, C.; Maffei, F.; Turrini, E.; Fimognari, C. Sulforaphane potentiates anticancer effects of doxorubicin and cisplatin and mitigates their toxic effects. Front. Pharmacol. 2020, 11, 567. [Google Scholar] [CrossRef]
- Ryoo, I.-g.; Kim, G.; Choi, B.-h.; Lee, S.-h.; Kwak, M.-K. Involvement of NRF2 signaling in doxorubicin resistance of cancer stem cell-enriched colonospheres. Biomol. Ther. 2016, 24, 482. [Google Scholar] [CrossRef] [PubMed]
- Gu, T.-T.; Li, C.; Xu, Y.; Zhang, L.; Shan, X.; Huang, X.; Guo, L.; Chen, K.; Wang, X.; Ge, H. Stimuli-responsive combination therapy of cisplatin and Nrf2 siRNA for improving antitumor treatment of osteosarcoma. Nano Res. 2020, 13, 630–637. [Google Scholar] [CrossRef]
- Ojha, T.; Pathak, V.; Shi, Y.; Hennink, W.E.; Moonen, C.T.W.; Storm, G.; Kiessling, F.; Lammers, T. Pharmacological and physical vessel modulation strategies to improve EPR-mediated drug targeting to tumors. Adv. Drug Deliv. Rev. 2017, 119, 44–60. [Google Scholar] [CrossRef]
- Xu, W.; Cui, Y.; Ling, P.; Li, L.B. Preparation and evaluation of folate-modified cationic pluronic micelles for poorly soluble anticancer drug. Drug Deliv. 2012, 19, 208–219. [Google Scholar] [CrossRef] [PubMed]
- Jia, Y.; Chen, S.; Wang, C.; Sun, T.; Yang, L. Hyaluronic acid-based nano drug delivery systems for breast cancer treatment: Recent advances. Front. Bioeng. Biotechnol. 2022, 10, 990145. [Google Scholar] [CrossRef]
- Wang, S.; Tan, X.; Zhou, Q.; Geng, P.; Wang, J.; Zou, P.; Deng, A.; Hu, J. Co-delivery of doxorubicin and SIS3 by folate-targeted polymeric micelles for overcoming tumor multidrug resistance. Drug Deliv. Transl. Res. 2022, 12, 167–179. [Google Scholar] [CrossRef]
- Qiu, L.; Xu, J.; Ahmed, K.S.; Zhu, M.; Zhang, Y.; Long, M.; Chen, W.; Fang, W.; Zhang, H.; Chen, J. Stimuli-responsive, dual-function prodrug encapsulated in hyaluronic acid micelles to overcome doxorubicin resistance. Acta Biomater. 2022, 140, 686–699. [Google Scholar] [CrossRef]
- Scheeren, L.E.; Nogueira-Librelotto, D.R.; Macedo, L.B.; de Vargas, J.M.; Mitjans, M.; Vinardell, M.P.; Rolim, C.M. Transferrin-conjugated doxorubicin-loaded PLGA nanoparticles with pH-responsive behavior: A synergistic approach for cancer therapy. J. Nanopart. Res. 2020, 22, 1–18. [Google Scholar] [CrossRef]
- Swider, E.; Koshkina, O.; Tel, J.; Cruz, L.J.; de Vries, I.J.M.; Srinivas, M. Customizing poly (lactic-co-glycolic acid) particles for biomedical applications. Acta Biomater. 2018, 73, 38–51. [Google Scholar] [CrossRef]
- Waghray, D.; Zhang, Q. Inhibit or Evade Multidrug Resistance P-Glycoprotein in Cancer Treatment. J. Med. Chem. 2018, 61, 5108–5121. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.Y.; Shueng, P.W.; Chou, C.M.; Lin, B.X.; Lin, M.H.; Kuo, D.Y.; Tsai, I.L.; Wu, S.M.; Lin, C.W. Elevation of CD109 promotes metastasis and drug resistance in lung cancer via activation of EGFR-AKT-mTOR signaling. Cancer Sci. 2020, 111, 1652–1662. [Google Scholar] [CrossRef] [PubMed]
- Christowitz, C.; Davis, T.; Isaacs, A.; Van Niekerk, G.; Hattingh, S.; Engelbrecht, A.-M. Mechanisms of doxorubicin-induced drug resistance and drug resistant tumour growth in a murine breast tumour model. BMC Cancer 2019, 19, 757. [Google Scholar] [CrossRef]
- Ibrahim, I.M.; Abdelmalek, D.H.; Elfiky, A.A. GRP78: A cell’s response to stress. Life Sci. 2019, 226, 156–163. [Google Scholar] [CrossRef] [PubMed]
- Xie, J.; Tao, Z.-H.; Zhao, J.; Li, T.; Wu, Z.-H.; Zhang, J.-F.; Zhang, J.; Hu, X.-C. Glucose regulated protein 78 (GRP78) inhibits apoptosis and attentinutes chemosensitivity of gemcitabine in breast cancer cell via AKT/mitochondrial apoptotic pathway. Biochem. Biophys. Res. Commun. 2016, 474, 612–619. [Google Scholar] [CrossRef] [PubMed]
- Tian, S.; Chang, W.; Du, H.; Bai, J.; Sun, Z.; Zhang, Q.; Wang, H.; Zhu, G.; Tao, K.; Long, Y. The interplay between GRP78 expression and Akt activation in human colon cancer cells under celecoxib treatment. Anti-Cancer Drugs 2015, 26, 964–973. [Google Scholar] [CrossRef]
- Fu, Y.; Wey, S.; Wang, M.; Ye, R.; Liao, C.-P.; Roy-Burman, P.; Lee, A.S. Pten null prostate tumorigenesis and AKT activation are blocked by targeted knockout of ER chaperone GRP78/BiP in prostate epithelium. Proc. Natl. Acad. Sci. USA 2008, 105, 19444–19449. [Google Scholar] [CrossRef]
- Grabarnick, E.; Andriyanov, A.V.; Han, H.; Eyal, S.; Barenholz, Y. PEGylated liposomes remotely loaded with the combination of doxorubicin, quinine, and indocyanine green enable successful treatment of multidrug-resistant tumors. Pharmaceutics 2021, 13, 2181. [Google Scholar] [CrossRef]
- Treger, J.S.; Priest, M.F.; Iezzi, R.; Bezanilla, F. Real-time imaging of electrical signals with an infrared FDA-approved dye. Biophys. J. 2014, 107, L09–L12. [Google Scholar] [CrossRef]
- Tamai, K.; Mizushima, T.; Wu, X.; Inoue, A.; Ota, M.; Yokoyama, Y.; Miyoshi, N.; Haraguchi, N.; Takahashi, H.; Nishimura, J. Photodynamic therapy using indocyanine green loaded on super carbonate apatite as minimally invasive cancer treatment. Mol. Cancer Ther. 2018, 17, 1613–1622. [Google Scholar] [CrossRef]
- Master, A.; Livingston, M.; Gupta, A.S. Photodynamic nanomedicine in the treatment of solid tumors: Perspectives and challenges. J. Control. Release 2013, 168, 88–102. [Google Scholar] [CrossRef] [PubMed]
- An, D.; Yu, X.; Jiang, L.; Wang, R.; He, P.; Chen, N.; Guo, X.; Li, X.; Feng, M. Reversal of multidrug resistance by apolipoprotein A1-modified doxorubicin liposome for breast cancer treatment. Molecules 2021, 26, 1280. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Y.; Wang, W.; Wang, B.; Zhu, H.; Zhang, B.; Feng, M. Delivery of hydrophilic drug doxorubicin hydrochloride-targeted liver using apoAI as carrier. J. Drug Target. 2013, 21, 367–374. [Google Scholar] [CrossRef] [PubMed]
- Cui, H.; Huan, M.-l.; Ye, W.-l.; Liu, D.-z.; Teng, Z.-h.; Mei, Q.-B.; Zhou, S.-y. Mitochondria and nucleus dual delivery system to overcome DOX resistance. Mol. Pharm. 2017, 14, 746–756. [Google Scholar] [CrossRef]
- Zhou, M.-X.; Zhang, J.-Y.; Cai, X.-M.; Dou, R.; Ruan, L.-F.; Yang, W.-J.; Lin, W.-C.; Chen, J.; Hu, Y. Tumor-penetrating and mitochondria-targeted drug delivery overcomes doxorubicin resistance in lung cancer. Chin. J. Polym. Sci. 2023, 41, 525–537. [Google Scholar] [CrossRef]
- Zhou, X.-W.; Xia, Y.-Z.; Zhang, Y.-L.; Luo, J.-G.; Han, C.; Zhang, H.; Zhang, C.; Yang, L.; Kong, L.-Y. Tomentodione M sensitizes multidrug resistant cancer cells by decreasing P-glycoprotein via inhibition of p38 MAPK signaling. Oncotarget 2017, 8, 101965–101983. [Google Scholar] [CrossRef] [PubMed]
- Liao, X.; Gao, Y.; Liu, J.; Tao, L.; Xie, J.; Gu, Y.; Liu, T.; Wang, D.; Xie, D.; Mo, S. Combination of Tanshinone IIA and Cisplatin Inhibits Esophageal Cancer by Downregulating NF-κB/COX-2/VEGF Pathway. Front. Oncol. 2020, 10, 1756. [Google Scholar] [CrossRef]
- Wang, R.; Luo, Z.; Zhang, H.; Wang, T. Tanshinone IIA reverses gefitinib-resistance in human non-small-cell lung cancer via regulation of VEGFR/Akt pathway. Onco. Targets Ther. 2019, 9355–9365. [Google Scholar] [CrossRef]
- Ye, Y.T.; Zhong, W.; Sun, P.; Wang, D.; Wang, C.; Hu, L.M.; Qian, J.Q. Apoptosis induced by the methanol extract of Salvia miltiorrhiza Bunge in non-small cell lung cancer through PTEN-mediated inhibition of PI3K/Akt pathway. J. Ethnopharmacol. 2017, 200, 107–116. [Google Scholar] [CrossRef]
- Jiang, Q.; Chen, X.; Tian, X.; Zhang, J.; Xue, S.; Jiang, Y.; Liu, T.; Wang, X.; Sun, Q.; Hong, Y.; et al. Tanshinone I inhibits doxorubicin-induced cardiotoxicity by regulating Nrf2 signaling pathway. Phytomedicine 2022, 106, 154439. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zhang, L.; Ma, Z.; Tian, L.; Liu, Y.; Liu, Y.; Chen, Q.; Li, Y.; Ma, E. Ascorbate promotes the cellular accumulation of doxorubicin and reverses the multidrug resistance in breast cancer cells by inducing ROS-dependent ATP depletion. Free Radic. Res. 2019, 53, 758–767. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Lu, X.; Liu, Q.; Dai, Y.; Zhu, X.; Wen, Y.; Xu, J.; Lu, Y.; Zhao, D.; Chen, X.; et al. Palmitoyl ascorbate and doxorubicin co-encapsulated liposome for synergistic anticancer therapy. Eur. J. Pharm. Sci. 2017, 105, 219–229. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zhu, M.; Meng, M.; Wang, Q.; Wang, Y.; Lei, Y.; Zhang, Y.; Weng, L.; Chen, X. A dual-responsive hyaluronic acid nanocomposite hydrogel drug delivery system for overcoming multiple drug resistance. Chin. Chem. Lett. 2023, 34, 107583. [Google Scholar] [CrossRef]
- Zhao, H.; Sun, S.; Wang, Z.; Hong, Y.; Shi, L.; Lan, M. pH-sensitive DOX-loaded PAA-PF127-PAA micelles combined with cryotherapy for treating walker 256 carcinosarcoma in a rat model. J. Nanosci. Nanotechnol. 2018, 18, 8070–8077. [Google Scholar] [CrossRef]
- Mahoney, B.P.; Raghunand, N.; Baggett, B.; Gillies, R.J. Tumor acidity, ion trapping and chemotherapeutics: I. Acid pH affects the distribution of chemotherapeutic agents in vitro. Biochem. Pharmacol. 2003, 66, 1207–1218. [Google Scholar] [CrossRef] [PubMed]
- Yoneda, T.; Hiasa, M.; Nagata, Y.; Okui, T.; White, F. Contribution of acidic extracellular microenvironment of cancer-colonized bone to bone pain. Biochim. Biophys. Acta 2015, 1848, 2677–2684. [Google Scholar] [CrossRef]
- Dubowchik, G.M.; Padilla, L.; Edinger, K.; Firestone, R.A. Reversal of doxorubicin resistance and catalytic neutralization of lysosomes by a lipophilic imidazole. Biochim. Biophys. Acta 1994, 1191, 103–108. [Google Scholar] [CrossRef]
- Altan, N.; Chen, Y.; Schindler, M.; Simon, S.M. Tamoxifen inhibits acidification in cells independent of the estrogen receptor. Proc. Natl. Acad. Sci. USA 1999, 96, 4432–4437. [Google Scholar] [CrossRef]
- Abumanhal-Masarweh, H.; Koren, L.; Zinger, A.; Yaari, Z.; Krinsky, N.; Kaneti, G.; Dahan, N.; Lupu-Haber, Y.; Suss-Toby, E.; Weiss-Messer, E. Sodium bicarbonate nanoparticles modulate the tumor pH and enhance the cellular uptake of doxorubicin. J. Control. Release 2019, 296, 1–13. [Google Scholar] [CrossRef]
- Kato, Y.; Ozawa, S.; Miyamoto, C.; Maehata, Y.; Suzuki, A.; Maeda, T.; Baba, Y. Acidic extracellular microenvironment and cancer. Cancer Cell Int. 2013, 13, 89. [Google Scholar] [CrossRef]
- Ando, H.; Ikeda, A.; Tagami, M.; Matsuo, N.C.A.; Shimizu, T.; Ishima, Y.; Eshima, K.; Ishida, T. Oral administration of sodium bicarbonate can enhance the therapeutic outcome of Doxil® via neutralizing the acidic tumor microenvironment. J. Control. Release 2022, 350, 414–420. [Google Scholar] [CrossRef] [PubMed]
- Robey, I.F.; Baggett, B.K.; Kirkpatrick, N.D.; Roe, D.J.; Dosescu, J.; Sloane, B.F.; Hashim, A.I.; Morse, D.L.; Raghunand, N.; Gatenby, R.A. Bicarbonate increases tumor pH and inhibits spontaneous metastases. Cancer Res. 2009, 69, 2260–2268. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Luo, Y.; Zheng, Y.; Zheng, L.; Lin, W.; Chen, Z.; Wu, S.; Chen, J.; Xie, Y. Long non-coding RNA LINC00426 contributes to doxorubicin resistance by sponging miR-4319 in osteosarcoma. Biol. Direct. 2020, 15, 1–11. [Google Scholar] [CrossRef]
- Huang, L.; Zeng, L.; Chu, J.; Xu, P.; Lv, M.; Xu, J.; Wen, J.; Li, W.; Wang, L.; Wu, X. Chemoresistance-related long non-coding RNA expression profiles in human breast cancer cells Erratum in/10.3892/mmr. 2019.10003. Mol. Med. Rep. 2018, 18, 243–253. [Google Scholar] [CrossRef]
- Gooding, A.J.; Zhang, B.; Gunawardane, L.; Beard, A.; Valadkhan, S.; Schiemann, W.P. The lncRNA BORG facilitates the survival and chemoresistance of triple-negative breast cancers. Oncogene 2019, 38, 2020–2041. [Google Scholar] [CrossRef]
- Wang, W.; Chen, D.; Zhu, K. SOX2OT variant 7 contributes to the synergistic interaction between EGCG and Doxorubicin to kill osteosarcoma via autophagy and stemness inhibition. J. Exp. Clin. Cancer Res. 2018, 37, 1–16. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, B.; Wang, Y.; Zhao, Q.; Wu, W.; Zhang, P.; Miao, L.; Sun, S. NOTCH3 Overexpression and Posttranscriptional Regulation by miR-150 Were Associated with EGFR-TKI Resistance in Lung Adenocarcinoma. Oncol. Res. 2019, 27, 751–761. [Google Scholar] [CrossRef] [PubMed]
- Pandey, V.; Chaube, B.; Bhat, M.K. Hyperglycemia regulates MDR-1, drug accumulation and ROS levels causing increased toxicity of carboplatin and 5-fluorouracil in MCF-7 cells. J. Cell Biochem. 2011, 112, 2942–2952. [Google Scholar] [CrossRef] [PubMed]
- Chouhan, S.; Singh, S.; Athavale, D.; Ramteke, P.; Vanuopadath, M.; Nair, B.G.; Nair, S.S.; Bhat, M.K. Sensitization of hepatocellular carcinoma cells towards doxorubicin and sorafenib is facilitated by glucose-dependent alterations in reactive oxygen species, P-glycoprotein and DKK4. J. Biosci. 2020, 45, 1–23. [Google Scholar] [CrossRef]
- Liao, C.H.; Yeh, C.T.; Huang, Y.H.; Wu, S.M.; Chi, H.C.; Tsai, M.M.; Tsai, C.Y.; Liao, C.J.; Tseng, Y.H.; Lin, Y.H. Dickkopf 4 positively regulated by the thyroid hormone receptor suppresses cell invasion in human hepatoma cells. Hepatology 2012, 55, 910–920. [Google Scholar] [CrossRef]
- Baehs, S.; Herbst, A.; Thieme, S.E.; Perschl, C.; Behrens, A.; Scheel, S.; Jung, A.; Brabletz, T.; Göke, B.; Blum, H. Dickkopf-4 is frequently down-regulated and inhibits growth of colorectal cancer cells. Cancer Lett. 2009, 276, 152–159. [Google Scholar] [CrossRef]
- Xi, Y.; Formentini, A.; Nakajima, G.; Kornmann, M.; Ju, J. Validation of biomarkers associated with 5-fluorouracil and thymidylate synthase in colorectal cancer. Oncol. Rep. 2008, 19, 257–262. [Google Scholar] [CrossRef] [PubMed]
- Ensign, L.M.; Cone, R.; Hanes, J. Oral drug delivery with polymeric nanoparticles: The gastrointestinal mucus barriers. Adv. Drug Deliv. Rev. 2012, 64, 557–570. [Google Scholar] [CrossRef] [PubMed]
- Cone, R.A. Barrier properties of mucus. Adv. Drug Deliv Rev. 2009, 61, 75–85. [Google Scholar] [CrossRef]
- Ma, T.Y.; Tran, D.; Hoa, N.; Nguyen, D.; Merryfield, M.; Tarnawski, A. Mechanism of extracellular calcium regulation of intestinal epithelial tight junction permeability: Role of cytoskeletal involvement. Microsc. Res. Tech. 2000, 51, 156–168. [Google Scholar] [CrossRef]
- Speth, P.; Van Hoesel, Q.; Haanen, C. Clinical pharmacokinetics of doxorubicin. Clin. Pharmacokine 1988, 15, 15–31. [Google Scholar] [CrossRef] [PubMed]
- Rawat, P.S.; Jaiswal, A.; Khurana, A.; Bhatti, J.S.; Navik, U. Doxorubicin-induced cardiotoxicity: An update on the molecular mechanism and novel therapeutic strategies for effective management. Biomed. Pharmacother. 2021, 139, 111708. [Google Scholar] [CrossRef]
- Reis-Mendes, A.; Carvalho, F.; Remião, F.; Sousa, E.; Bastos, M.d.L.; Costa, V.M. The main metabolites of fluorouracil+ adriamycin+ cyclophosphamide (FAC) are not major contributors to FAC toxicity in H9c2 cardiac differentiated cells. Biomolecules 2019, 9, 98. [Google Scholar] [CrossRef]
- Mordente, A.; Meucci, E.; Silvestrini, A.; Martorana, G.E.; Giardina, B. New developments in anthracycline-induced cardiotoxicity. Curr. Med. Chem. 2009, 16, 1656–1672. [Google Scholar] [CrossRef]
- Green, P.S.; Leeuwenburgh, C. Mitochondrial dysfunction is an early indicator of doxorubicin-induced apoptosis. Biochim. Biophys. Acta 2002, 1588, 94–101. [Google Scholar] [CrossRef]
- Berthiaume, J.; Wallace, K.B. Adriamycin-induced oxidative mitochondrial cardiotoxicity. Cell Biol. Toxicol. 2007, 23, 15–25. [Google Scholar] [CrossRef]
- Barth, E.; Stämmler, G.; Speiser, B.; Schaper, J. Ultrastructural quantitation of mitochondria and myofilaments in cardiac muscle from 10 different animal species including man. J. Mol. Cell Cardiol. 1992, 24, 669–681. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Sun, H.; Hu, J.; Han, X.; Liu, H.; Hu, Y. Transferrin gated mesoporous silica nanoparticles for redox-responsive and targeted drug delivery. Colloids Surf. B Biointerfaces 2017, 152, 77–84. [Google Scholar] [CrossRef]
- Thomas, H.; Coley, H.M. Overcoming multidrug resistance in cancer: An update on the clinical strategy of inhibiting p-glycoprotein. Cancer Control. 2003, 10, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Fox, E.; Bates, S.E. Tariquidar (XR9576): A P-glycoprotein drug efflux pump inhibitor. Expert Rev. Anticancer Ther. 2007, 7, 447–459. [Google Scholar] [CrossRef] [PubMed]
- Meerum Terwogt, J.M.; Malingré, M.M.; Beijnen, J.H.; ten Bokkel Huinink, W.W.; Rosing, H.; Koopman, F.J.; van Tellingen, O.; Swart, M.; Schellens, J.H. Coadministration of oral cyclosporin A enables oral therapy with paclitaxel. Clin. Cancer Res. 1999, 5, 3379–3384. [Google Scholar]
- Malingre, M.; Beijnen, J.; Rosing, H.; Koopman, F.; Jewell, R.; Paul, E.; Huinink, W.; Schellens, J. Co-administration of GF120918 significantly increases the systemic exposure to oral paclitaxel in cancer patients. Br. J. Cancer 2001, 84, 42–47. [Google Scholar] [CrossRef]
- Herben, V.; Rosing, H.; Huinink, W.; Van Zomeren, D.; Batchelor, D.; Doyle, E.; Beusenberg, F.; Beijnen, J.; Schellens, J. Oral topotecan: Bioavailability and effect of food co-administration. Br. J. Cancer 1999, 80, 1380–1386. [Google Scholar] [CrossRef]
- Kruijtzer, C.; Beijnen, J.; Rosing, H.; ten Bokkel Huinink, W.; Schot, M.; Jewell, R.; Paul, E.; Schellens, J. Increased oral bioavailability of topotecan in combination with the breast cancer resistance protein and P-glycoprotein inhibitor GF120918. J. Clin. Oncol. 2002, 20, 2943–2950. [Google Scholar] [CrossRef]
- Kuppens, I.E.; Witteveen, E.O.; Jewell, R.C.; Radema, S.A.; Paul, E.M.; Mangum, S.G.; Beijnen, J.H.; Voest, E.E.; Schellens, J.H. A phase I, randomized, open-label, parallel-cohort, dose-finding study of elacridar (GF120918) and oral topotecan in cancer patients. Clin. Cancer Res. 2007, 13, 3276–3285. [Google Scholar] [CrossRef]
- Jackson, C.G.; Hung, T.; Segelov, E.; Barlow, P.; Prenen, H.; McLaren, B.; Hung, N.A.; Clarke, K.; Chao, T.Y.; Dai, M.S. Oral paclitaxel with encequidar compared to intravenous paclitaxel in patients with advanced cancer: A randomised crossover pharmacokinetic study. Br. J. Clin. Pharmacol. 2021, 87, 4670–4680. [Google Scholar] [CrossRef]
- Kuppens, I.; Bosch, T.; Van Maanen, M.; Rosing, H.; Fitzpatrick, A.; Beijnen, J.; Schellens, J. Oral bioavailability of docetaxel in combination with OC144-093 (ONT-093). Cancer Chemother. Pharmacol. 2005, 55, 72–78. [Google Scholar] [CrossRef] [PubMed]
- Rushing, D.A.; Raber, S.R.; Rodvold, K.A.; Piscitelli, S.C.; Plank, G.S.; Tewksbury, D.A. The effects of cyclosporine on the pharmacokinetics of doxorubicin in patients with small cell lung cancer. Cancer 1994, 74, 834–841. [Google Scholar] [CrossRef] [PubMed]
- Giaccone, G.; Linn, S.C.; Welink, J.; Catimel, G.; Stieltjes, H.; Van der Vijgh, W.; Eeltink, C.; Vermorken, J.B.; Pinedo, H.M. A dose-finding and pharmacokinetic study of reversal of multidrug resistance with SDZ PSC 833 in combination with doxorubicin in patients with solid tumors. Clin. Cancer Res. 1997, 3, 2005–2015. [Google Scholar] [PubMed]
- Onafuye, H.; Pieper, S.; Mulac, D.; Cinatl, J., Jr.; Wass, M.N.; Langer, K.; Michaelis, M. Doxorubicin-loaded human serum albumin nanoparticles overcome transporter-mediated drug resistance in drug-adapted cancer cells. Beilstein J. Nanotechnol. 2019, 10, 1707–1715. [Google Scholar] [CrossRef]
- Nielsen, R.B.; Holm, R.; Pijpers, I.; Snoeys, J.; Nielsen, U.G.; Nielsen, C.U. Oral etoposide and zosuquidar bioavailability in rats: Effect of co-administration and in vitro-in vivo correlation of P-glycoprotein inhibition. Int. J. Pharm. X 2021, 3, 100089. [Google Scholar] [CrossRef]
- Morschhauser, F.; Zinzani, P.L.; Burgess, M.; Sloots, L.; Bouafia, F.; Dumontet, C. Phase I/II trial of a P-glycoprotein inhibitor, Zosuquidar. 3HCl trihydrochloride (LY335979), given orally in combination with the CHOP regimen in patients with non-Hodgkin’s lymphoma. Leuk. Lymphoma 2007, 48, 708–715. [Google Scholar] [CrossRef]
- Toffoli, G.; Corona, G.; Basso, B.; Boiocchi, M. Pharmacokinetic optimisation of treatment with oral etoposide. Clin. Pharmacokinet 2004, 43, 441–466. [Google Scholar] [CrossRef]
- Chen, M.-C.; Mi, F.-L.; Liao, Z.-X.; Hsiao, C.-W.; Sonaje, K.; Chung, M.-F.; Hsu, L.-W.; Sung, H.-W. Recent advances in chitosan-based nanoparticles for oral delivery of macromolecules. Adv. Drug Deliv. Rev. 2013, 65, 865–879. [Google Scholar] [CrossRef]
- Sung, H.-W.; Sonaje, K.; Liao, Z.-X.; Hsu, L.-W.; Chuang, E.-Y. pH-responsive nanoparticles shelled with chitosan for oral delivery of insulin: From mechanism to therapeutic applications. Acc. Chem. Res. 2012, 45, 619–629. [Google Scholar] [CrossRef]
- Hu, F.-Q.; Liu, L.-N.; Du, Y.-Z.; Yuan, H. Synthesis and antitumor activity of doxorubicin conjugated stearic acid-g-chitosan oligosaccharide polymeric micelles. Biomaterials 2009, 30, 6955–6963. [Google Scholar] [CrossRef]
- Yang, Y.; Chen, Y.; Li, D.; Lin, S.; Chen, H.; Wu, W.; Zhang, W. Linolenic acid conjugated chitosan micelles for improving the oral absorption of doxorubicin via fatty acid transporter. Carbohydr. Polym. 2023, 300, 120233. [Google Scholar] [CrossRef]
- Wang, J.; Li, L.; Du, Y.; Sun, J.; Han, X.; Luo, C.; Ai, X.; Zhang, Q.; Wang, Y.; Fu, Q.; et al. Improved oral absorption of doxorubicin by amphiphilic copolymer of lysine-linked ditocopherol polyethylene glycol 2000 succinate: In vitro characterization and in vivo evaluation. Mol. Pharm. 2015, 12, 463–473. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Sun, J.; Chen, Q.; Gao, Y.; Li, L.; Li, H.; Leng, D.; Wang, Y.; Sun, Y.; Jing, Y.; et al. Star-shape copolymer of lysine-linked di-tocopherol polyethylene glycol 2000 succinate for doxorubicin delivery with reversal of multidrug resistance. Biomaterials 2012, 33, 6877–6888. [Google Scholar] [CrossRef] [PubMed]
- Kumbham, S.; Paul, M.; Itoo, A.; Ghosh, B.; Biswas, S. Oleanolic acid-conjugated human serum albumin nanoparticles encapsulating doxorubicin as synergistic combination chemotherapy in oropharyngeal carcinoma and melanoma. Int. J. Pharm. 2022, 614, 121479. [Google Scholar] [CrossRef]
- Sarfraz, M.; Afzal, A.; Raza, S.M.; Bashir, S.; Madni, A.; Khan, M.W.; Ma, X.; Xiang, G. Liposomal co-delivered oleanolic acid attenuates doxorubicin-induced multi-organ toxicity in hepatocellular carcinoma. Oncotarget 2017, 8, 47136–47153. [Google Scholar] [CrossRef]
- Kalaria, D.R.; Sharma, G.; Beniwal, V.; Ravi Kumar, M.N. Design of biodegradable nanoparticles for oral delivery of doxorubicin: In vivo pharmacokinetics and toxicity studies in rats. Pharm. Res. 2009, 26, 492–501. [Google Scholar] [CrossRef] [PubMed]
- Astete, C.E.; Sabliov, C.M. Synthesis and characterization of PLGA nanoparticles. J. Biomater. Sci. Polym. Ed. 2006, 17, 247–289. [Google Scholar] [CrossRef] [PubMed]
- Hussain, N.; Jaitley, V.; Florence, A.T. Recent advances in the understanding of uptake of microparticulates across the gastrointestinal lymphatics. Adv. Drug Deliv. Rev. 2001, 50, 107–142. [Google Scholar] [CrossRef]
- Nguyen, T.T.; Duong, V.A.; Maeng, H.J. Pharmaceutical formulations with P-glycoprotein Inhibitory effect as promising approaches for enhancing oral drug absorption and bioavailability. Pharmaceutics 2021, 13, 1103. [Google Scholar] [CrossRef]
- Choi, M.-K.; Lee, J.; Song, I.-S. Pharmacokinetic modulation of substrate drugs via the inhibition of drug-metabolizing enzymes and transporters using pharmaceutical excipients. J. Pharm. Investig. 2023, 53, 1–18. [Google Scholar] [CrossRef]
- Maksimenko, O.; Malinovskaya, J.; Shipulo, E.; Osipova, N.; Razzhivina, V.; Arantseva, D.; Yarovaya, O.; Mostovaya, U.; Khalansky, A.; Fedoseeva, V.; et al. Doxorubicin-loaded PLGA nanoparticles for the chemotherapy of glioblastoma: Towards the pharmaceutical development. Int. J. Pharm. 2019, 572, 118733. [Google Scholar] [CrossRef]
- Pereverzeva, E.; Treschalin, I.; Treschalin, M.; Arantseva, D.; Ermolenko, Y.; Kumskova, N.; Maksimenko, O.; Balabanyan, V.; Kreuter, J.; Gelperina, S. Toxicological study of doxorubicin-loaded PLGA nanoparticles for the treatment of glioblastoma. Int. J. Pharm. 2019, 554, 161–178. [Google Scholar] [CrossRef]
- Cao, D.; Zhang, X.; Akabar, M.D.; Luo, Y.; Wu, H.; Ke, X.; Ci, T. Liposomal doxorubicin loaded PLGA-PEG-PLGA based thermogel for sustained local drug delivery for the treatment of breast cancer. Artif. Cells Nanomed. Biotechnol. 2019, 47, 181–191. [Google Scholar] [CrossRef] [PubMed]
- Jain, A.K.; Swarnakar, N.K.; Das, M.; Godugu, C.; Singh, R.P.; Rao, P.R.; Jain, S. Augmented anticancer efficacy of doxorubicin-loaded polymeric nanoparticles after oral administration in a breast cancer induced animal model. Mol. Pharm. 2011, 8, 1140–1151. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.; Yang, Z.; Wang, S.; Wang, L.; He, Q.; Tang, H.; Ji, P.; Chen, T. Zwitterionic polydopamine modified nanoparticles as an efficient nanoplatform to overcome both the mucus and epithelial barriers. Chem. Eng. J. 2022, 428, 132107. [Google Scholar] [CrossRef]
- Ahmad, N.; Ahmad, R.; Alam, M.A.; Ahmad, F.J. Enhancement of oral bioavailability of doxorubicin through surface modified biodegradable polymeric nanoparticles. Chem. Cent. J. 2018, 12, 65. [Google Scholar] [CrossRef]
- Ahmad, N.; Ahmad, R.; Alam, M.A.; Ahmad, F.J.; Amir, M.; Pottoo, F.H.; Sarafroz, M.; Jafar, M.; Umar, K. Daunorubicin oral bioavailability enhancement by surface coated natural biodegradable macromolecule chitosan based polymeric nanoparticles. Int. J. Biol. Macromol. 2019, 128, 825–838. [Google Scholar] [CrossRef]
- Khdair, A.; Hamad, I.; Alkhatib, H.; Bustanji, Y.; Mohammad, M.; Tayem, R.; Aiedeh, K. Modified-chitosan nanoparticles: Novel drug delivery systems improve oral bioavailability of doxorubicin. Eur. J. Pharm. Sci. 2016, 93, 38–44. [Google Scholar] [CrossRef]
- Sun, M.; Li, D.; Wang, X.; He, L.; Lv, X.; Xu, Y.; Tang, R. Intestine-penetrating, pH-sensitive and double-layered nanoparticles for oral delivery of doxorubicin with reduced toxicity. J. Mater. Chem. B 2019, 7, 3692–3703. [Google Scholar] [CrossRef]
- Głąb, T.K.; Boratyński, J. Potential of Casein as a Carrier for Biologically Active Agents. Top. Curr. Chem. 2017, 375, 71. [Google Scholar] [CrossRef]
- Rehan, F.; Emranul Karim, M.; Ahemad, N.; Farooq Shaikh, M.; Gupta, M.; Gan, S.H.; Chowdhury, E.H. A comparative evaluation of anti-tumor activity following oral and intravenous delivery of doxorubicin in a xenograft model of breast tumor. J. Pharm. Investig. 2022, 52, 787–804. [Google Scholar] [CrossRef]
- Feng, C.; Li, J.; Mu, Y.; Kong, M.; Li, Y.; Raja, M.A.; Cheng, X.J.; Liu, Y.; Chen, X.G. Multilayer micro-dispersing system as oral carriers for co-delivery of doxorubicin hydrochloride and P-gp inhibitor. Int. J. Biol. Macromol. 2017, 94, 170–180. [Google Scholar] [CrossRef]
- Benezra, M.; Penate-Medina, O.; Zanzonico, P.B.; Schaer, D.; Ow, H.; Burns, A.; DeStanchina, E.; Longo, V.; Herz, E.; Iyer, S.; et al. Multimodal silica nanoparticles are effective cancer-targeted probes in a model of human melanoma. J. Clin. Investig. 2011, 121, 2768–2780. [Google Scholar] [CrossRef]
- Zheng, N.; Li, J.; Xu, C.; Xu, L.; Li, S.; Xu, L. Mesoporous silica nanorods for improved oral drug absorption. Artif. Cells Nanomed. Biotechnol. 2018, 46, 1132–1140. [Google Scholar] [CrossRef]
- Wang, Y.; Sun, Y.; Wang, J.; Yang, Y.; Li, Y.; Yuan, Y.; Liu, C. Charge-Reversal APTES-Modified Mesoporous Silica Nanoparticles with High Drug Loading and Release Controllability. ACS Appl. Mater. Interfaces 2016, 8, 17166–17175. [Google Scholar] [CrossRef]
- Han, N.; Wang, Y.; Bai, J.; Liu, J.; Wang, Y.; Gao, Y.; Jiang, T.; Kang, W.; Wang, S. Facile synthesis of the lipid bilayer coated mesoporous silica nanocomposites and their application in drug delivery. Microporous and Mesoporous Materials 2016, 219, 209–218. [Google Scholar] [CrossRef]
- Zhao, Q.; Liu, J.; Zhu, W.; Sun, C.; Di, D.; Zhang, Y.; Wang, P.; Wang, Z.; Wang, S. Dual-stimuli responsive hyaluronic acid-conjugated mesoporous silica for targeted delivery to CD44-overexpressing cancer cells. Acta Biomater. 2015, 23, 147–156. [Google Scholar] [CrossRef] [PubMed]
- Bekaroğlu, G.M.; İşçi, S. Raw and Purified Clay Minerals for Drug Delivery Applications. ACS Omega 2022, 7, 38825–38831. [Google Scholar] [CrossRef] [PubMed]
- Gerami, S.E.; Pourmadadi, M.; Fatoorehchi, H.; Yazdian, F.; Rashedi, H.; Nigjeh, M.N. Preparation of pH-sensitive chitosan/polyvinylpyrrolidone/α-Fe2O3 nanocomposite for drug delivery application: Emphasis on ameliorating restrictions. Int. J. Biol. Macromol. 2021, 173, 409–420. [Google Scholar] [CrossRef]
- Rahmani, E.; Pourmadadi, M.; Ghorbanian, S.A.; Yazdian, F.; Rashedi, H.; Navaee, M. Preparation of a pH-responsive chitosan-montmorillonite-nitrogen-doped carbon quantum dots nanocarrier for attenuating doxorubicin limitations in cancer therapy. Eng. Life Sci. 2022, 22, 634–649. [Google Scholar] [CrossRef] [PubMed]
- Liao, J.; Qian, Y.; Sun, Z.; Wang, J.; Zhang, Q.; Zheng, Q.; Wei, S.; Liu, N.; Yang, H. In Vitro Binding and Release Mechanisms of Doxorubicin from Nanoclays. J. Phys. Chem. Lett. 2022, 13, 8429–8435. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.-J.; Huang, S.-Y.; Wang, T.-H.; Lin, T.-Y.; Huang, N.-C.; Shih, O.; Jeng, U.S.; Chu, C.-Y.; Chiang, W.-H. Clay nanosheets simultaneously intercalated and stabilized by PEGylated chitosan as drug delivery vehicles for cancer chemotherapy. Carbohydr. Polym. 2023, 302, 120390. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, J.; Choi, M.-K.; Song, I.-S. Recent Advances in Doxorubicin Formulation to Enhance Pharmacokinetics and Tumor Targeting. Pharmaceuticals 2023, 16, 802. https://doi.org/10.3390/ph16060802
Lee J, Choi M-K, Song I-S. Recent Advances in Doxorubicin Formulation to Enhance Pharmacokinetics and Tumor Targeting. Pharmaceuticals. 2023; 16(6):802. https://doi.org/10.3390/ph16060802
Chicago/Turabian StyleLee, Jihoon, Min-Koo Choi, and Im-Sook Song. 2023. "Recent Advances in Doxorubicin Formulation to Enhance Pharmacokinetics and Tumor Targeting" Pharmaceuticals 16, no. 6: 802. https://doi.org/10.3390/ph16060802
APA StyleLee, J., Choi, M.-K., & Song, I.-S. (2023). Recent Advances in Doxorubicin Formulation to Enhance Pharmacokinetics and Tumor Targeting. Pharmaceuticals, 16(6), 802. https://doi.org/10.3390/ph16060802








