Nanomedicine as an Emerging Technology to Foster Application of Essential Oils to Fight Cancer
Abstract
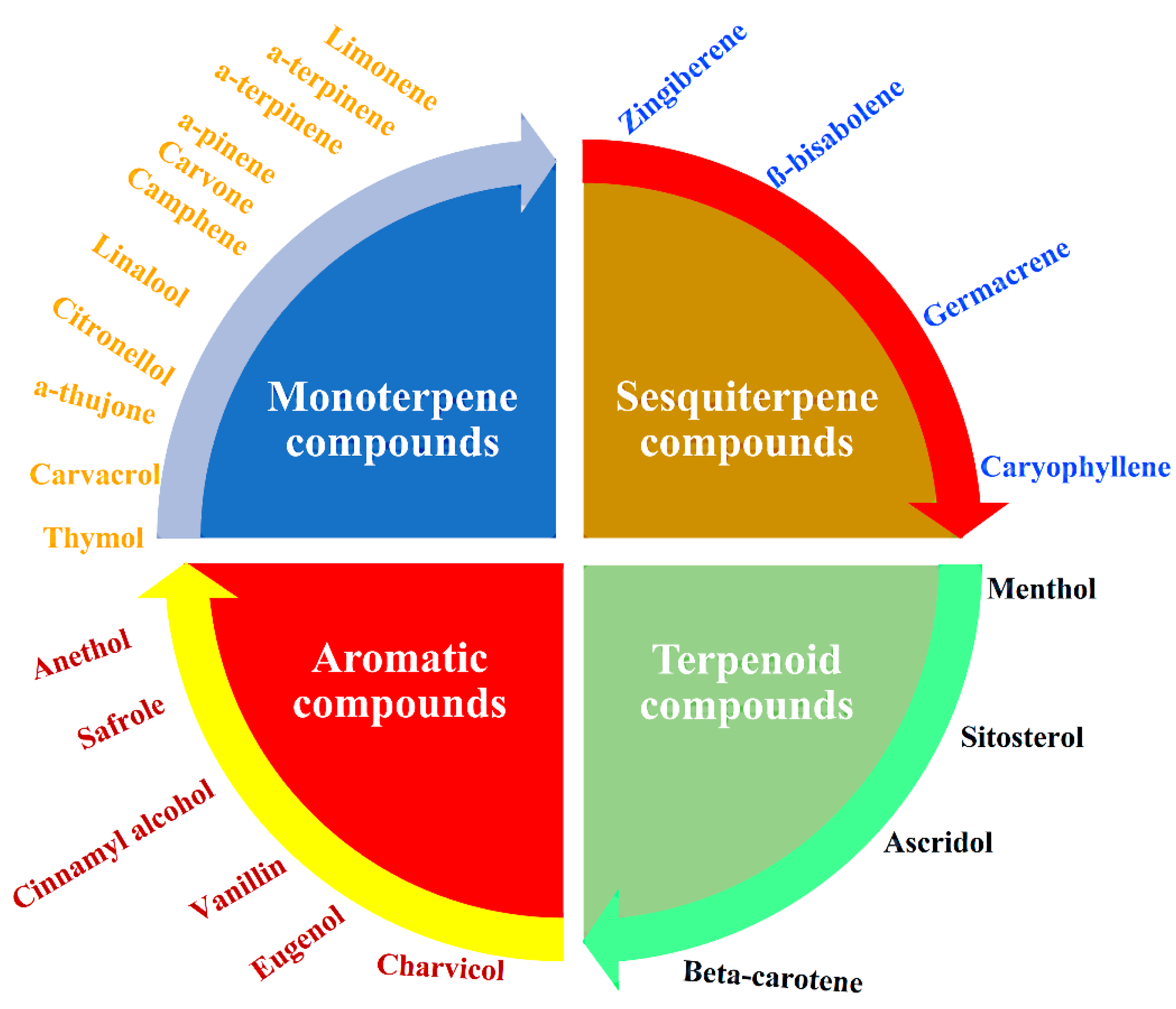
1. Introduction to Essential Oils
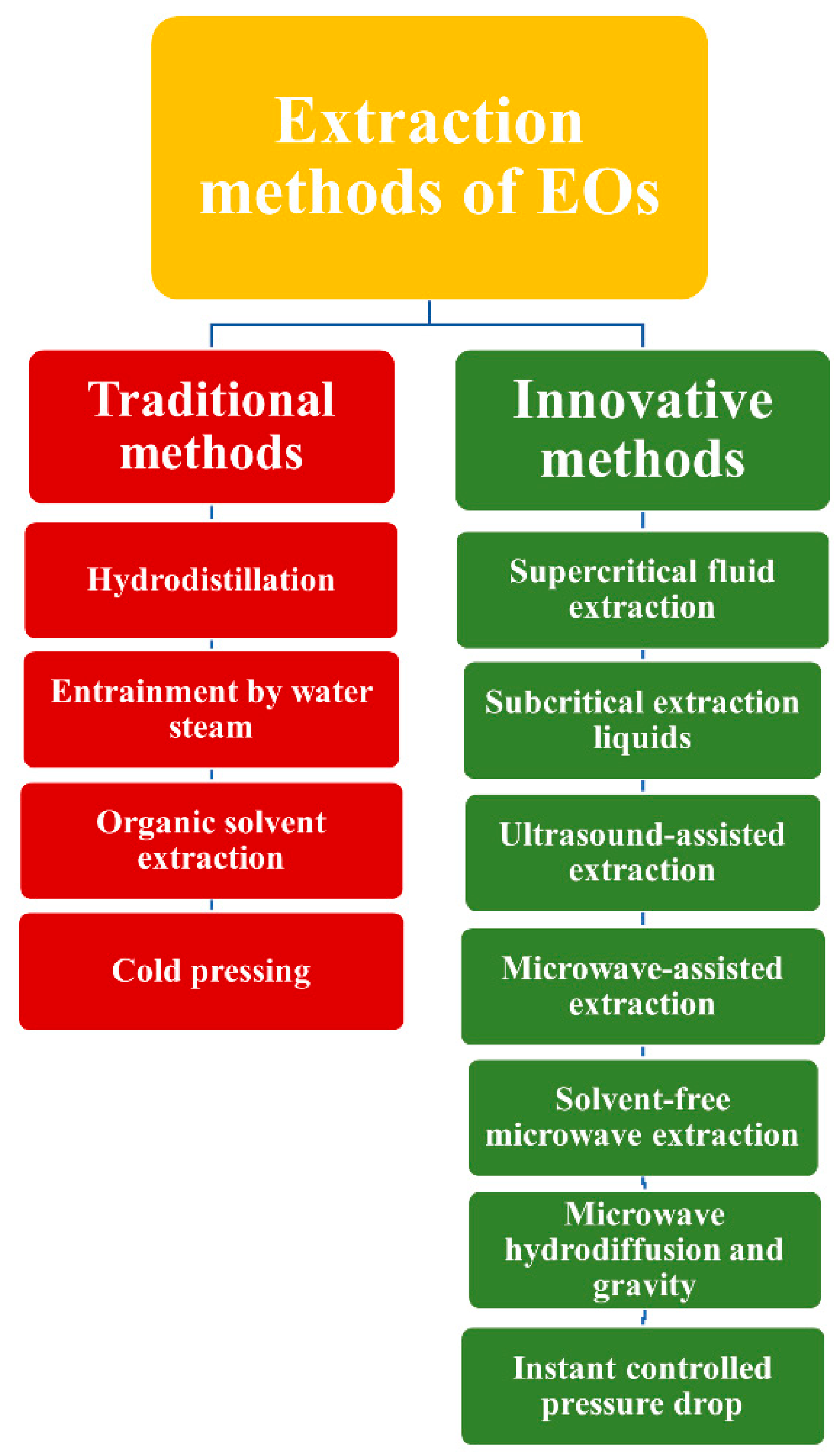
1.1. Methods of EOs Obtaining
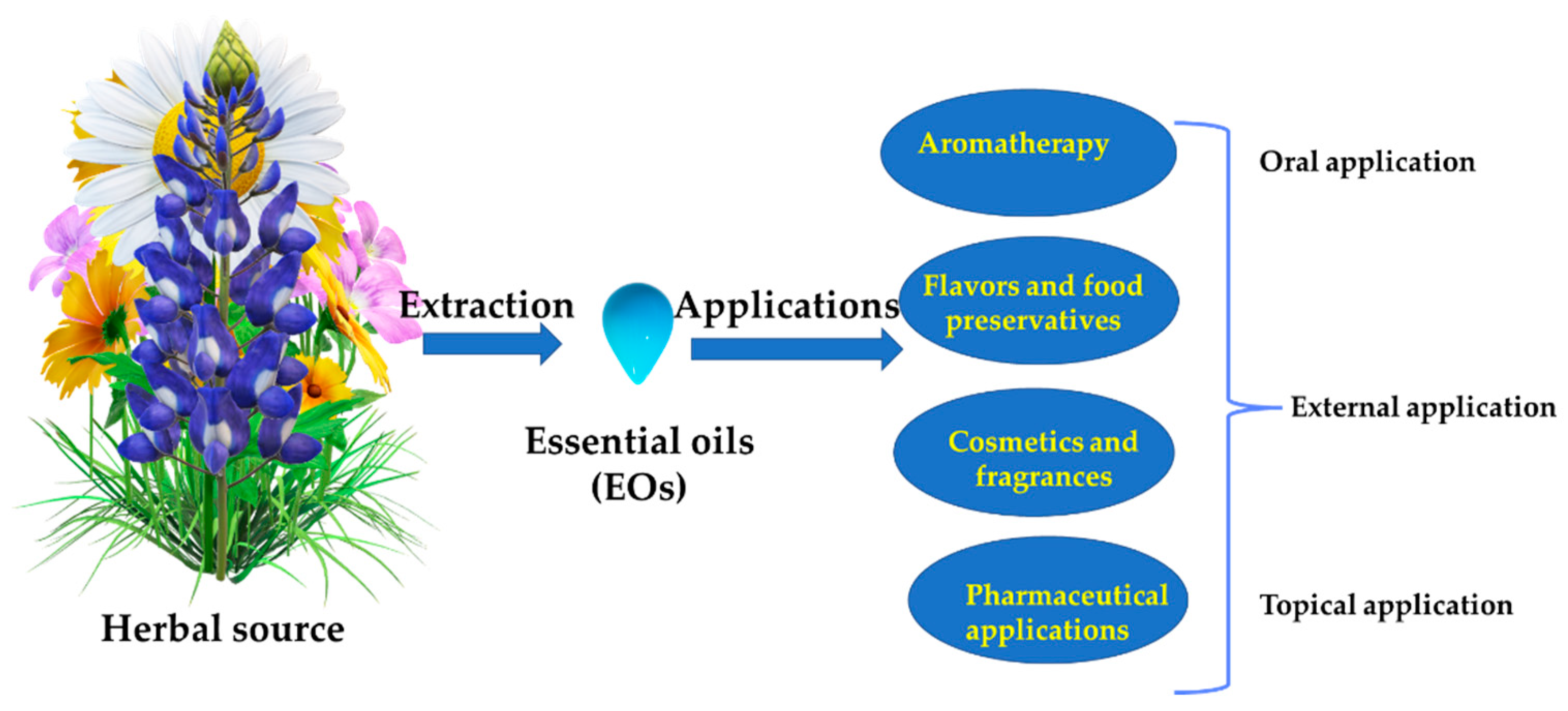
1.2. Applications of EOs in Health Care
1.3. Methods of EOs Administration
1.4. EO Side Effects
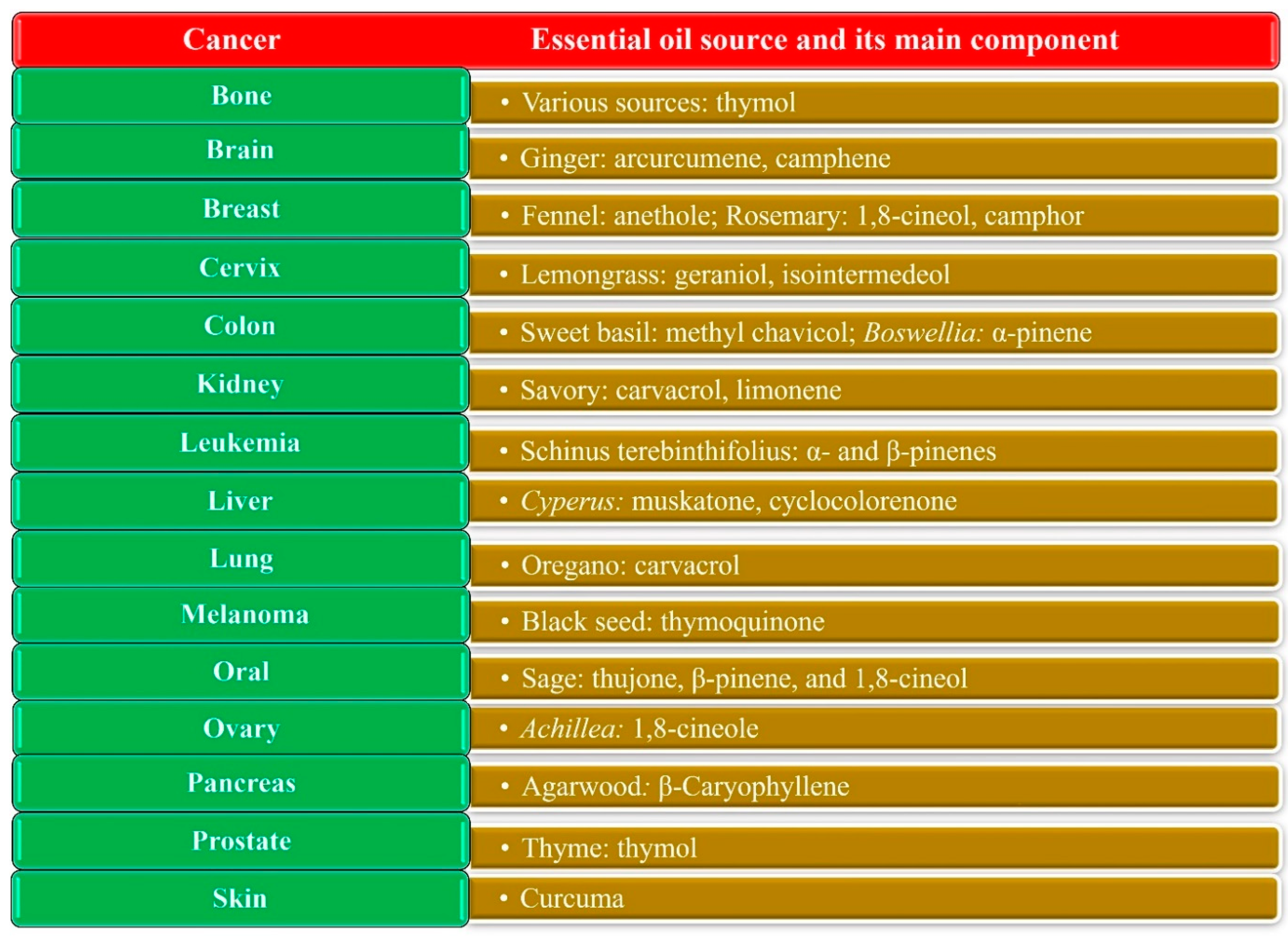
1.5. Anticancer Activity of EOs
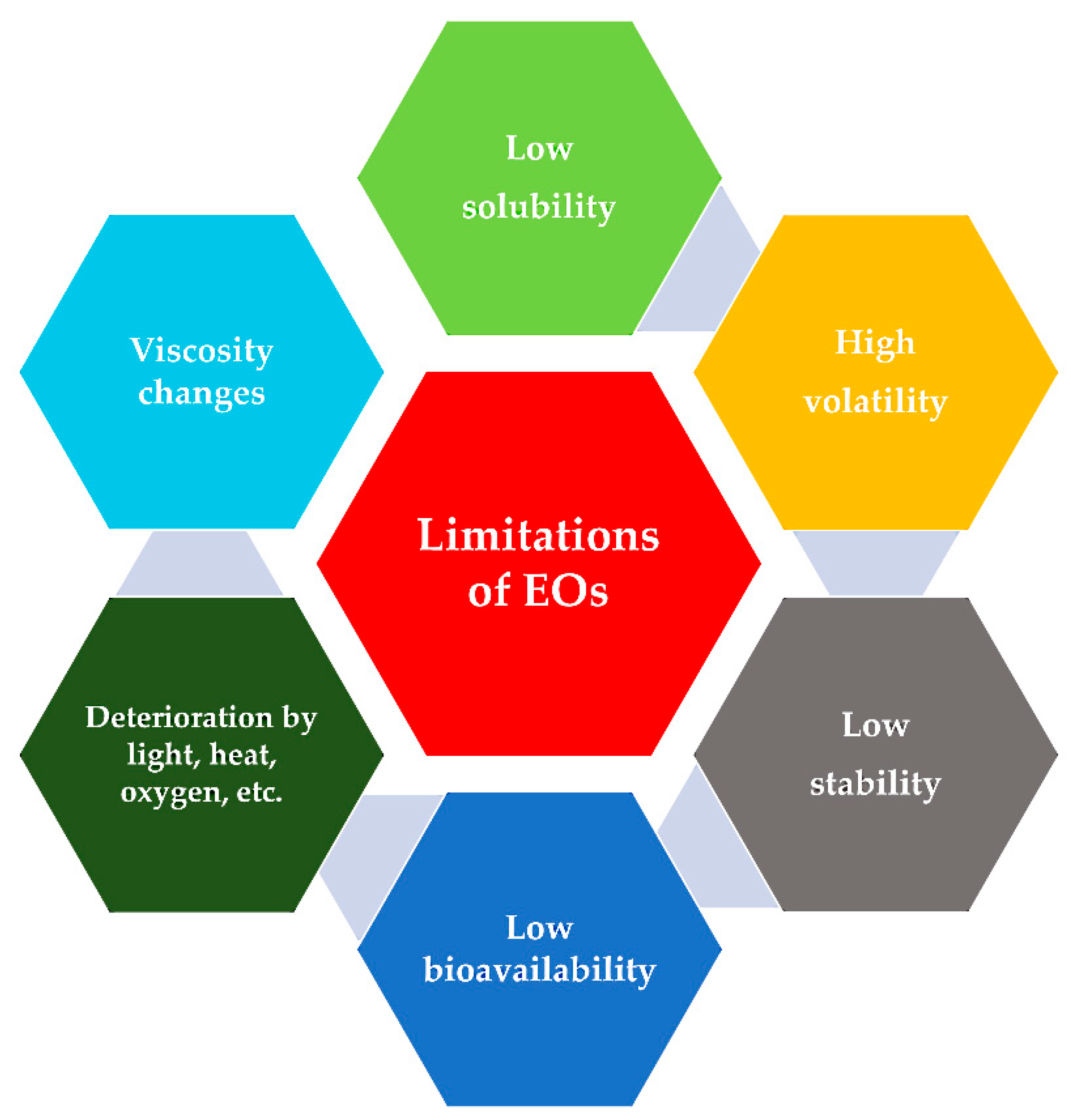
1.6. Limitations of Present Technology
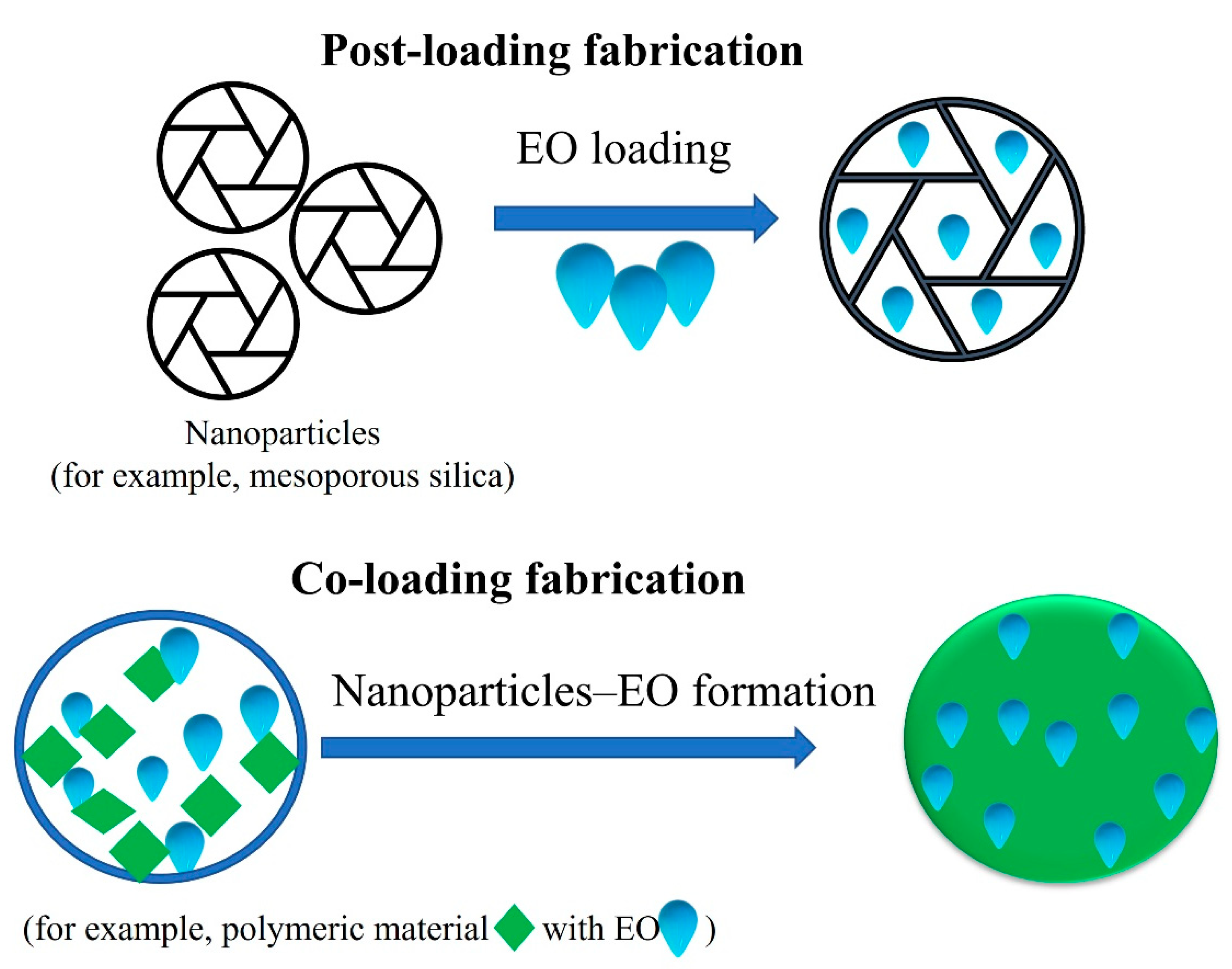
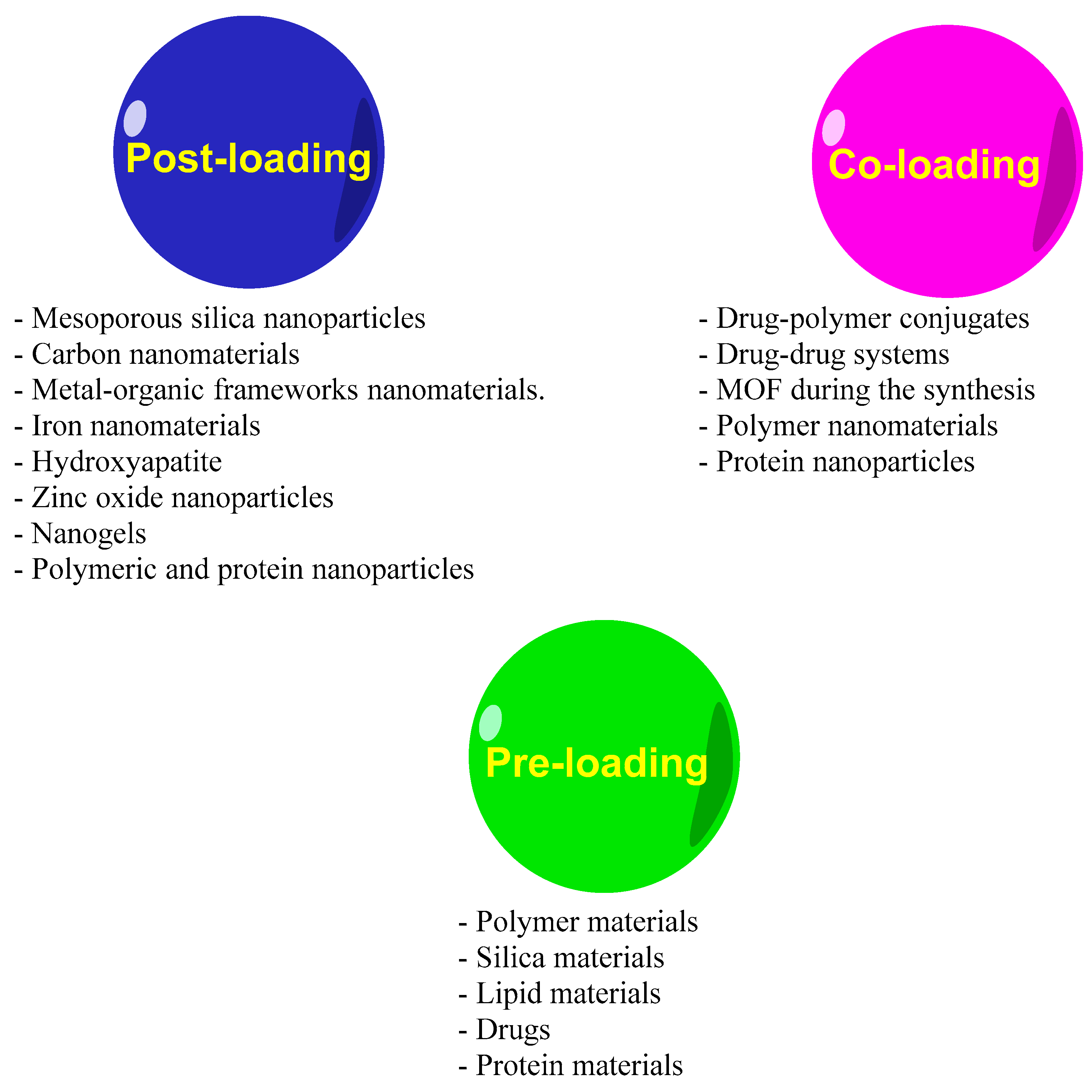
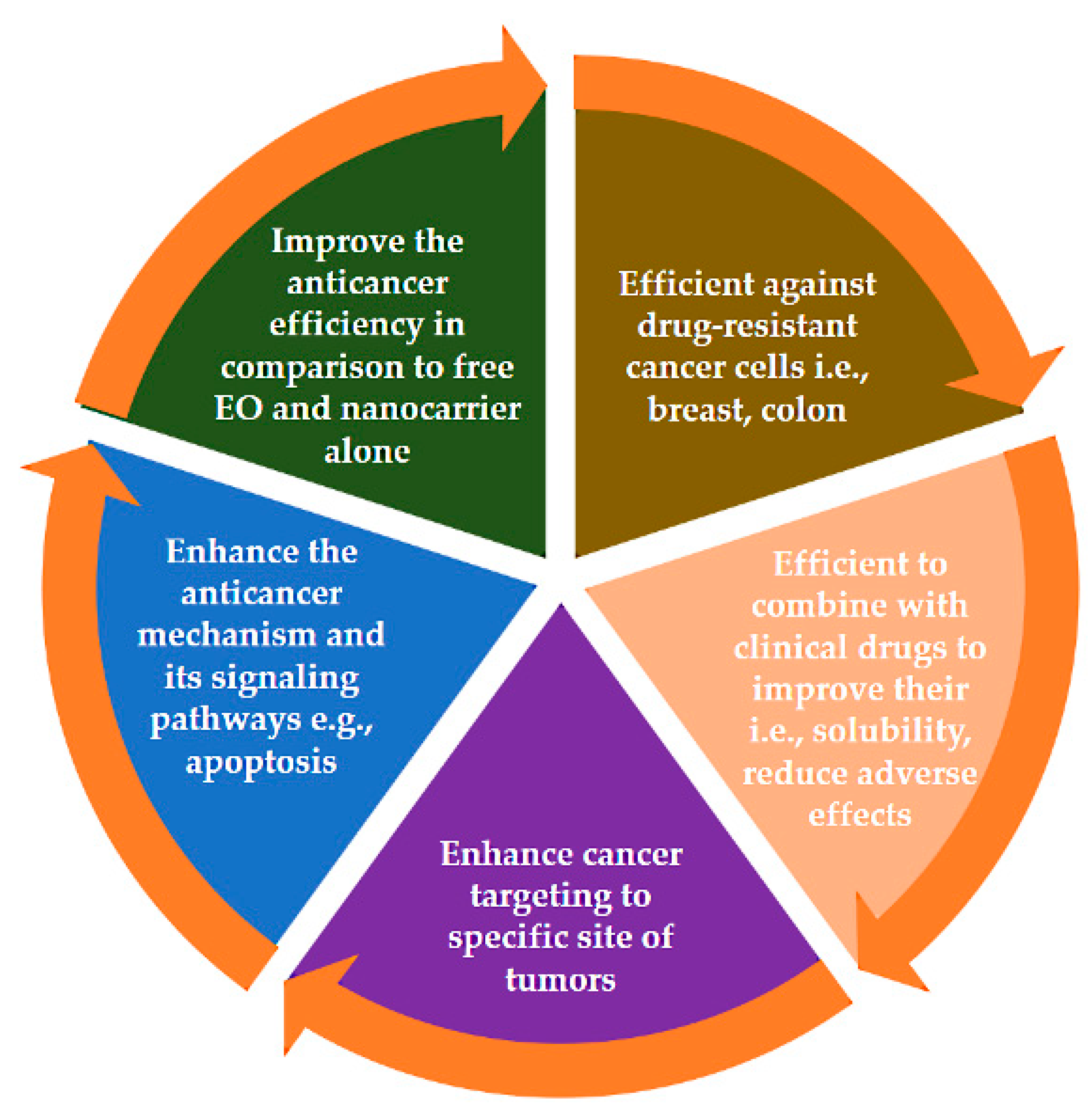
2. Nanomedicine Application to Enhance EOs Anticancer Efficiency
Nanodelivery Systems for EOs
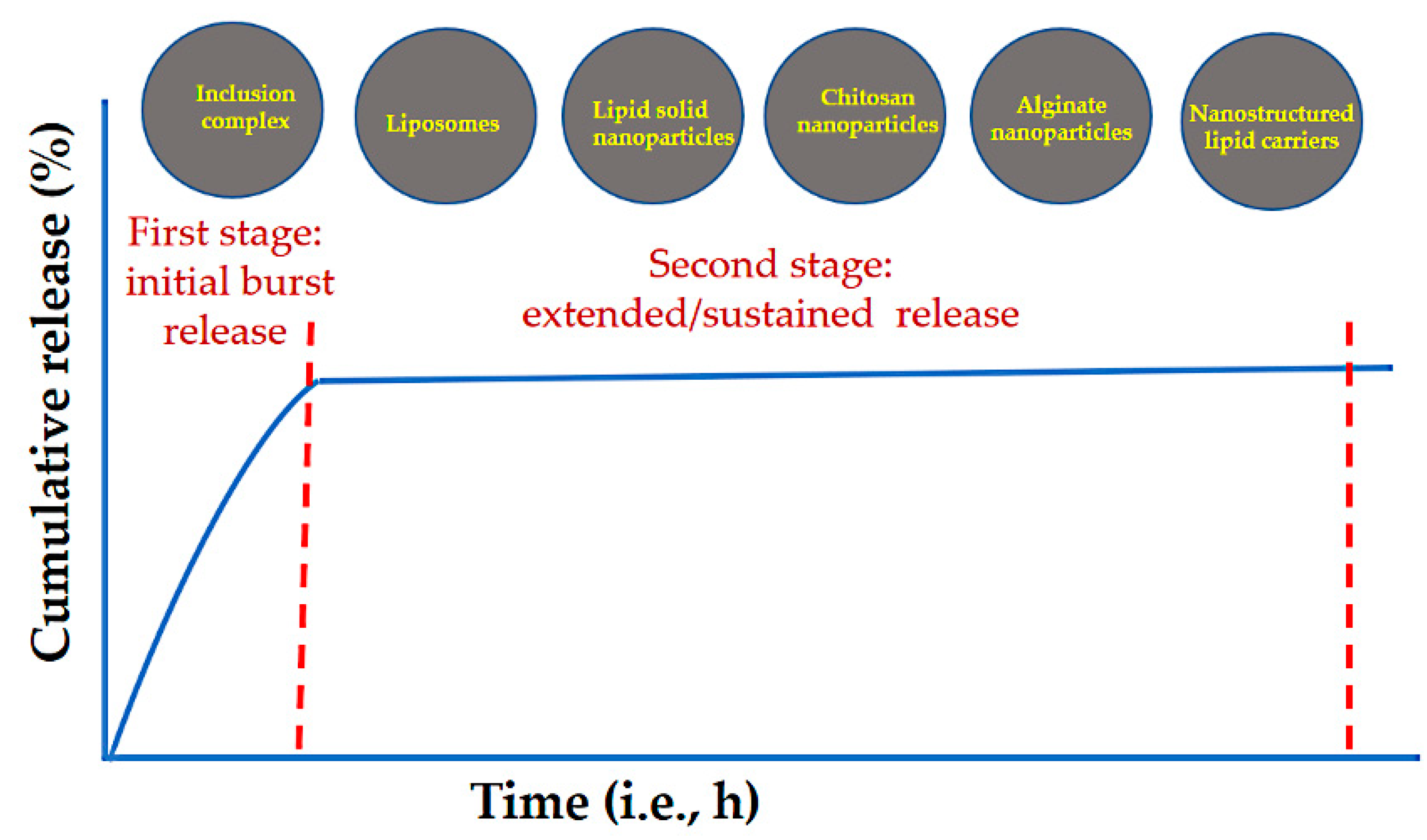
3. Controlled Release of EOs from Loaded Nanostructures
3.1. Controlled Release of EOs from Polymer Nanocarriers
3.2. Controlled Release of EOs from Lipids
3.3. Controlled Release of EOs from Inclusion Complexes
3.4. Controlled Release of EOs from Inorganic Nanocarriers
4. Targeted Nanodelivery Systems for Anticancer Applications
4.1. Nanodelivery Systems for Breast Cancer
4.2. Nanodelivery Systems for Lung Cancer
4.3. Nanodelivery Systems for Liver Cancer
4.4. Nanodelivery Systems for Colon Cancer
4.5. Nanodelivery Systems Fabricated for Brain Cancer
4.6. Nanodelivery Systems for Other Cancers
5. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Butler, M.S. Natural products to drugs: Natural product-derived compounds in clinical trials. Nat. Prod. Rep. 2008, 25, 475–516. [Google Scholar] [CrossRef]
- El Asbahani, A.; Miladi, K.; Badri, W.; Sala, M.; Aït Addi, E.H.; Casabianca, H.; El Mousadik, A.; Hartmann, D.; Jilale, A.; Renaud, F.N.; et al. Essential oils: From extraction to encapsulation. Int. J. Pharm. 2015, 483, 220–243. [Google Scholar] [CrossRef] [PubMed]
- Solórzano-Santos, F.; Miranda-Novales, M.G. Essential oils from aromatic herbs as antimicrobial agents. Curr. Opin. Biotechnol. 2012, 23, 136–141. [Google Scholar] [CrossRef] [PubMed]
- Aldred, E.M.; Buck, C.; Vall, K. Chapter 22—Terpenes. In Pharmacology; Aldred, E.M., Buck, C., Vall, K., Eds.; Churchill Livingstone: Edinburgh, UK, 2009; pp. 167–174. [Google Scholar]
- Kashyap, D.; Tuli, H.S.; Yerer, M.B.; Sharma, A.; Sak, K.; Srivastava, S.; Pandey, A.; Garg, V.K.; Sethi, G.; Bishayee, A. Natural product-based nanoformulations for cancer therapy: Opportunities and challenges. Semin. Cancer Biol. 2019, 69, 5–23. [Google Scholar] [CrossRef]
- Baser, K.H.C.; Buchbauer, G. Handbook of Essential Oils: Science, Technology, and Applications; CRC Press: Boca Raton, FL, USA, 2015. [Google Scholar]
- Masango, P. Cleaner production of essential oils by steam distillation. J. Clean. Prod. 2005, 13, 833–839. [Google Scholar] [CrossRef]
- Farhat, A.; Fabiano-Tixier, A.S.; Visinoni, F.; Romdhane, M.; Chemat, F. A surprising method for green extraction of essential oil from dry spices: Microwave dry-diffusion and gravity. J. Chromatogr. A 2010, 1217, 7345–7350. [Google Scholar] [CrossRef] [PubMed]
- Farhat, A.; Ginies, C.; Romdhane, M.; Chemat, F. Eco-friendly and cleaner process for isolation of essential oil using microwave energy: Experimental and theoretical study. J. Chromatogr. A 2009, 1216, 5077–5085. [Google Scholar] [CrossRef] [PubMed]
- Ferhat, M.A.; Meklati, B.Y.; Chemat, F. Comparison of different isolation methods of essential oil from Citrus fruits: Cold pressing, hydrodistillation and microwave ‘dry’ distillation. Flavour Fragr. J. 2007, 22, 494–504. [Google Scholar] [CrossRef]
- Vinatoru, M. An overview of the ultrasonically assisted extraction of bioactive principles from herbs. Ultrason. Sonochemistry 2001, 8, 303–313. [Google Scholar] [CrossRef]
- Svoboda, K.P.; Greenaway, R.I. Investigation of volatile oil glands of Satureja hortensis L. (summer savory) and phytochemical comparison of different varieties. Int. J. Aromather. 2003, 13, 196–202. [Google Scholar] [CrossRef]
- Bravo Cadena, M.; Preston, G.M.; Van der Hoorn, R.A.L.; Townley, H.E.; Thompson, I.P. Species-specific antimicrobial activity of essential oils and enhancement by encapsulation in mesoporous silica nanoparticles. Ind. Crops Prod. 2018, 122, 582–590. [Google Scholar] [CrossRef]
- Dhifi, W.; Bellili, S.; Jazi, S.; Bahloul, N.; Mnif, W. Essential Oils’ Chemical Characterization and Investigation of Some Biological Activities: A Critical Review. Medicines 2016, 3, 25. [Google Scholar] [CrossRef] [PubMed]
- Andrade, M.A.; Braga, M.A.; Cesar, P.H.S.; Trento, M.V.C.; Espósito, M.A.; Silva, L.F.; Marcussi, S. Anticancer Properties of Essential Oils: An Overview. Curr. Cancer Drug Targets 2018, 18, 957–966. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Weller, C.L. Recent advances in extraction of nutraceuticals from plants. Trends Food Sci. Technol. 2006, 17, 300–312. [Google Scholar] [CrossRef]
- Meyer-Warnod, B. Natural essential oils: Extraction processes and application to some major oils. Perfum. Flavorist 1984, 9, 93–104. [Google Scholar]
- Faborode, M.O.; Favier, J.F. Identification and significance of the oil-point in seed-oil expression. J. Agric. Eng. Res. 1996, 65, 335–345. [Google Scholar] [CrossRef]
- Herrero, M.; Cifuentes, A.; Ibañez, E. Sub- and supercritical fluid extraction of functional ingredients from different natural sources: Plants, food-by-products, algae and microalgae: A review. Food Chem. 2006, 98, 136–148. [Google Scholar] [CrossRef]
- Özel, M.Z.; Göğüş, F.; Lewis, A.C. Comparison of direct thermal desorption with water distillation and superheated water extraction for the analysis of volatile components of Rosa damascena Mill. using GCxGC-TOF/MS. Anal. Chim. Acta 2006, 566, 172–177. [Google Scholar] [CrossRef]
- Zizovic, I.; Stamenić, M.; Orlović, A.; Skala, D. Supercritical carbon dioxide extraction of essential oils from plants with secretory ducts: Mathematical modelling on the micro-scale. J. Supercrit. Fluids 2007, 39, 338–346. [Google Scholar] [CrossRef]
- Assami, K.; Pingret, D.; Chemat, S.; Meklati, B.Y.; Chemat, F. Ultrasound induced intensification and selective extraction of essential oil from Carum carvi L. seeds. Chem. Eng. Process. Process Intensif. 2012, 62, 99–105. [Google Scholar] [CrossRef]
- Golmakani, M.T.; Rezaei, K. Comparison of microwave-assisted hydrodistillation withthe traditional hydrodistillation method in the extractionof essential oils from Thymus vulgaris L. Food Chem. 2008, 109, 925–930. [Google Scholar] [CrossRef]
- Lucchesi, M.E.; Chemat, F.; Smadja, J. An original solvent free microwave extraction of essential oils from spices. Flavour. Fragr. J. 2004, 19, 134–138. [Google Scholar] [CrossRef]
- Vian, M.A.; Fernandez, X.; Visinoni, F.; Chemat, F. Microwave hydrodiffusion and gravity, a new technique for extraction of essential oils. J. Chromatogr. A 2008, 1190, 14–17. [Google Scholar] [CrossRef]
- Sahraoui, N.; Vian, M.A.; Bornard, I.; Boutekedjiret, C.; Chemat, F. Improved microwave steam distillation apparatus for isolation of essential oils: Comparison with conventional steam distillation. J. Chromatogr. A 2008, 1210, 229–233. [Google Scholar] [CrossRef]
- Berka-Zougali, B.; Hassani, A.; Besombes, C.; Allaf, K. Extraction of essential oils from Algerian myrtle leaves using instant controlled pressure drop technology. J. Chromatogr. A 2010, 1217, 6134–6142. [Google Scholar] [CrossRef]
- Elshafie, H.S.; Camele, I. An Overview of the Biological Effects of Some Mediterranean Essential Oils on Human Health. BioMed Res. Int. 2017, 2017, 9268468. [Google Scholar] [CrossRef]
- Wu, Y.; Luo, Y.; Wang, Q. Antioxidant and antimicrobial properties of essential oils encapsulated in zein nanoparticles prepared by liquid–liquid dispersion method. LWT Food Sci. Technol. 2012, 48, 283–290. [Google Scholar] [CrossRef]
- Chifiriuc, M.C.; Kamerzan, C.; Lazar, V. Chapter 12—Essential Oils and Nanoparticles: New Strategy to Prevent Microbial Biofilms. In Nanostructures for Antimicrobial Therapy; Ficai, A., Grumezescu, A.M., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 279–291. [Google Scholar]
- Bilia, A.R.; Guccione, C.; Isacchi, B.; Righeschi, C.; Firenzuoli, F.; Bergonzi, M.C. Essential Oils Loaded in Nanosystems: A Developing Strategy for a Successful Therapeutic Approach. Evid. Based Complement. Altern. Med. 2014, 2014, 651593. [Google Scholar] [CrossRef]
- Ali, B.; Al-Wabel, N.A.; Shams, S.; Ahamad, A.; Khan, S.A.; Anwar, F. Essential oils used in aromatherapy: A systemic review. Asian Pac. J. Trop. Biomed. 2015, 5, 601–611. [Google Scholar] [CrossRef]
- Edris, A.E. Pharmaceutical and therapeutic Potentials of essential oils and their individual volatile constituents: A review. Phytother. Res. 2007, 21, 308–323. [Google Scholar] [CrossRef]
- Cooke, B.; Ernst, E. Aromatherapy: A systematic review. Br. J. Gen. Pract. 2000, 50, 493–496. [Google Scholar]
- Vostinaru, O.; Heghes, S.C.; Filip, L. Safety profile of essential oils. In Essential Oils-Bioactive Compounds, New Perspectives and Applications; IntechOpen: London, UK, 2020; pp. 1–13. [Google Scholar]
- Farrar, A.J.; Farrar, F.C. Clinical Aromatherapy. Nurs. Clin. N. Am. 2020, 55, 489–504. [Google Scholar] [CrossRef]
- Zhang, N.; Yao, L. Anxiolytic Effect of Essential Oils and Their Constituents: A Review. J. Agric. Food Chem. 2019, 67, 13790–13808. [Google Scholar] [CrossRef]
- Steflitsch, W. Aromatherapy—From Traditional and Scientific Evidence into Clinical Practice. Dtsch. Med. Wochenschr. 2017, 142, 1936–1942. [Google Scholar] [CrossRef]
- Boukhatem, M.N.; Ferhat, M.A.; Kameli, A.; Saidi, F.; Kebir, H.T. Lemon grass (Cymbopogon citratus) essential oil as a potent anti-inflammatory and antifungal drugs. Libyan. J. Med. 2014, 9, 25431. [Google Scholar] [CrossRef]
- Mousavi, S.; Weschka, D.; Bereswill, S.; Heimesaat, M.M. Immune-Modulatory Effects upon Oral Application of Cumin-Essential-Oil to Mice Suffering from Acute Campylobacteriosis. Pathogens 2021, 10, 818. [Google Scholar] [CrossRef]
- Toniolo, J.; Delaide, V.; Beloni, P. Effectiveness of Inhaled Aromatherapy on Chemotherapy-Induced Nausea and Vomiting: A Systematic Review. J. Altern. Complement. Med. 2021, 27, 1058–1069. [Google Scholar] [CrossRef]
- Lua, P.L.; Salihah, N.; Mazlan, N. Effects of inhaled ginger aromatherapy on chemotherapy-induced nausea and vomiting and health-related quality of life in women with breast cancer. Complement. Med. 2015, 23, 396–404. [Google Scholar] [CrossRef]
- Kreye, G.; Wasl, M.; Dietz, A.; Klaffel, D.; Groselji-Strele, A.; Eberhard, K.; Glechner, A. Aromatherapy in Palliative Care: A Single-Institute Retrospective Analysis Evaluating the Effect of Lemon Oil Pads against Nausea and Vomiting in Advanced Cancer Patients. Cancers 2022, 14, 2131. [Google Scholar] [CrossRef]
- Imanishi, J.; Kuriyama, H.; Shigemori, I.; Watanabe, S.; Aihara, Y.; Kita, M.; Sawai, K.; Nakajima, H.; Yoshida, N.; Kunisawa, M.; et al. Anxiolytic effect of aromatherapy massage in patients with breast cancer. Evid. Based Complement. Altern. Med. 2009, 6, 123–128. [Google Scholar] [CrossRef]
- Izgu, N.; Ozdemir, L.; Bugdayci Basal, F. Effect of Aromatherapy Massage on Chemotherapy-Induced Peripheral Neuropathic Pain and Fatigue in Patients Receiving Oxaliplatin: An Open Label Quasi-Randomized Controlled Pilot Study. Cancer Nurs. 2019, 42, 139–147. [Google Scholar] [CrossRef]
- Bagetta, G.; Cosentino, M.; Sakurada, T. Aromatherapy: Basic Mechanisms and Evidence Based Clinical Use; CRC Press: Boca Raton, FL, USA, 2015; Volume 2. [Google Scholar]
- Schilcher, H. Effects and side-effects of essential oils. In Essential Oils and Aromatic Plants; Springer: Berlin/Heidelberg, Germany, 1985; pp. 217–231. [Google Scholar]
- Al-Otaibi, W.A.; Alkhatib, M.H.; Wali, A.N. Cytotoxicity and apoptosis enhancement in breast and cervical cancer cells upon coadministration of mitomycin C and essential oils in nanoemulsion formulations. Biomed. Pharmacother. 2018, 106, 946–955. [Google Scholar] [CrossRef]
- Attallah, O.A.; Shetta, A.; Elshishiny, F.; Mamdouh, W. Essential oil loaded pectin/chitosan nanoparticles preparation and optimization via Box–Behnken design against MCF-7 breast cancer cell lines. RSC Adv. 2020, 10, 8703–8708. [Google Scholar] [CrossRef]
- Sotelo-Boyás, M.; Correa-Pacheco, Z.; Bautista-Baños, S.; Gómez, Y.G.Y. Release study and inhibitory activity of thyme essential oil-loaded chitosan nanoparticles and nanocapsules against foodborne bacteria. Int. J. Biol. Macromol. 2017, 103, 409–414. [Google Scholar] [CrossRef]
- Khalili, S.T.; Mohsenifar, A.; Beyki, M.; Zhaveh, S.; Rahmani-Cherati, T.; Abdollahi, A.; Bayat, M.; Tabatabaei, M. Encapsulation of Thyme essential oils in chitosan-benzoic acid nanogel with enhanced antimicrobial activity against Aspergillus flavus. LWT Food Sci. Technol. 2015, 60, 502–508. [Google Scholar] [CrossRef]
- Ravi Kumar, M.N. Nano and microparticles as controlled drug delivery devices. J. Pharm. Pharm. Sci. 2000, 3, 234–258. [Google Scholar]
- Martins, I.M.; Barreiro, M.F.; Coelho, M.; Rodrigues, A.E. Microencapsulation of essential oils with biodegradable polymeric carriers for cosmetic applications. Chem. Eng. J. 2014, 245, 191–200. [Google Scholar] [CrossRef]
- van Soest, J.J.G. Encapsulation of Fragrances and Flavours: A Way to Control Odour and Aroma in Consumer Products. In Flavours and Fragrances: Chemistry, Bioprocessing and Sustainability; Berger, R.G., Ed.; Springer: Berlin/Heidelberg, Germany, 2007; pp. 439–455. [Google Scholar]
- Wang, N.; Cheng, X.; Li, N.; Wang, H.; Chen, H. Nanocarriers and Their Loading Strategies. Adv. Healthc. Mater. 2019, 8, 1801002. [Google Scholar] [CrossRef]
- Lammari, N.; Louaer, O.; Meniai, A.H.; Elaissari, A. Encapsulation of Essential Oils via Nanoprecipitation Process: Overview, Progress, Challenges and Prospects. Pharmaceutics 2020, 12, 431. [Google Scholar] [CrossRef]
- Bakry, A.M.; Abbas, S.; Ali, B.; Majeed, H.; Abouelwafa, M.Y.; Mousa, A.; Liang, L. Microencapsulation of Oils: A Comprehensive Review of Benefits, Techniques, and Applications. Compr. Rev. Food Sci. Food Saf. 2016, 15, 143–182. [Google Scholar] [CrossRef]
- Maes, C.; Bouquillon, S.; Fauconnier, M.-L. Encapsulation of Essential Oils for the Development of Biosourced Pesticides with Controlled Release: A Review. Molecules 2019, 24, 2539. [Google Scholar] [CrossRef]
- Majeed, H.; Bian, Y.-Y.; Ali, B.; Jamil, A.; Majeed, U.; Khan, Q.F.; Iqbal, K.J.; Shoemaker, C.F.; Fang, Z. Essential oil encapsulations: Uses, procedures, and trends. RSC Adv. 2015, 5, 58449–58463. [Google Scholar] [CrossRef]
- Ocak, B. Complex coacervation of collagen hydrolysate extracted from leather solid wastes and chitosan for controlled release of lavender oil. J. Environ. Manag. 2012, 100, 22–28. [Google Scholar] [CrossRef]
- Choi, M.-J.; Soottitantawat, A.; Nuchuchua, O.; Min, S.-G.; Ruktanonchai, U. Physical and light oxidative properties of eugenol encapsulated by molecular inclusion and emulsion–diffusion method. Food Res. Int. 2009, 42, 148–156. [Google Scholar] [CrossRef]
- Donsì, F.; Annunziata, M.; Sessa, M.; Ferrari, G. Nanoencapsulation of essential oils to enhance their antimicrobial activity in foods. LWT Food Sci. Technol. 2011, 44, 1908–1914. [Google Scholar] [CrossRef]
- Baranauskienė, R.; Bylaitė, E.; Žukauskaitė, J.; Venskutonis, R.P. Flavor Retention of Peppermint (Mentha piperita L.) Essential Oil Spray-Dried in Modified Starches during Encapsulation and Storage. J. Agric. Food Chem. 2007, 55, 3027–3036. [Google Scholar] [CrossRef]
- Abd-ElGawad, A.M.; Elshamy, A.I.; El-Nasser El Gendy, A.; Al-Rowaily, S.L.; Assaeed, A.M. Preponderance of oxygenated sesquiterpenes and diterpenes in the volatile oil constituents of Lactuca serriola L. revealed antioxidant and allelopathic activity. Chem. Biodivers. 2019, 16, e1900278. [Google Scholar] [CrossRef]
- Bayala, B.; Bassole, I.H.; Scifo, R.; Gnoula, C.; Morel, L.; Lobaccaro, J.-M.A.; Simpore, J. Anticancer activity of essential oils and their chemical components—A review. Am. J. Cancer Res. 2014, 4, 591–607. [Google Scholar]
- Blowman, K.; Magalhães, M.; Lemos, M.F.L.; Cabral, C.; Pires, I.M. Anticancer Properties of Essential Oils and Other Natural Products. Evid. Based Complement. Altern. Med. 2018, 2018, 3149362. [Google Scholar] [CrossRef]
- Russo, R.; Corasaniti, M.T.; Bagetta, G.; Morrone, L.A. Exploitation of cytotoxicity of some essential oils for translation in cancer therapy. Evid. Based Complement. Altern. Med. eCAM 2015, 2015, 397821. [Google Scholar] [CrossRef]
- Lesgards, J.-F.; Baldovini, N.; Vidal, N.; Pietri, S. Anticancer Activities of Essential Oils Constituents and Synergy with Conventional Therapies: A Review. Phytother. Res. 2014, 28, 1423–1446. [Google Scholar] [CrossRef]
- Aras, A.; Iqbal, M.J.; Naqvi, S.K.; Gercek, Y.C.; Boztas, K.; Gasparri, M.L.; Shatynska-Mytsyk, I.; Fayyaz, S.; Farooqi, A.A. Anticancer activity of essential oils: Targeting of protein networks in cancer cells. Asian Pac. J. Cancer Prev. 2014, 15, 8047–8050. [Google Scholar] [CrossRef]
- Gaonkar, R.; Shiralgi, Y.; Lakkappa, D.B.; Hegde, G. Essential oil from Cymbopogon flexuosus as the potential inhibitor for HSP90. Toxicol. Rep. 2018, 5, 489–496. [Google Scholar] [CrossRef]
- Gautam, N.; Mantha, A.K.; Mittal, S. Essential Oils and Their Constituents as Anticancer Agents: A Mechanistic View. BioMed Res. Int. 2014, 2014, 154106. [Google Scholar] [CrossRef]
- Jackson, A.L.; Loeb, L.A. The contribution of endogenous sources of DNA damage to the multiple mutations in cancer. Mutat. Res. 2001, 477, 7–21. [Google Scholar] [CrossRef]
- Storz, P. Reactive oxygen species in tumor progression. Front. Biosci. 2005, 10, 1881–1896. [Google Scholar] [CrossRef]
- Bayala, B.; Bassole, I.H.N.; Gnoula, C.; Nebie, R.; Yonli, A.; Morel, L.; Figueredo, G.; Nikiema, J.-B.; Lobaccaro, J.-M.A.; Simpore, J. Chemical composition, antioxidant, anti-inflammatory and anti-proliferative activities of essential oils of plants from Burkina Faso. PLoS ONE 2014, 9, e92122. [Google Scholar] [CrossRef]
- Fitsiou, E.; Mitropoulou, G.; Spyridopoulou, K.; Tiptiri-Kourpeti, A.; Vamvakias, M.; Bardouki, H.; Panayiotidis, M.Ι.; Galanis, A.; Kourkoutas, Y.; Chlichlia, K.; et al. Phytochemical Profile and Evaluation of the Biological Activities of Essential Oils Derived from the Greek Aromatic Plant Species Ocimum basilicum, Mentha spicata, Pimpinella anisum and Fortunella margarita. Molecules 2016, 21, 1069. [Google Scholar] [CrossRef]
- Becer, E.; Kabadayı, H.; Başer, K.H.C.; Vatansever, H.S. Boswellia sacra essential oil manages colon cancer stem cells proliferation and apoptosis: A new perspective for cure. J. Essent. Oil Res. 2021, 33, 53–62. [Google Scholar] [CrossRef]
- Islam, E.-G.; Sobhy Hassab El, N.; Ebtesam, N.; Samar, A. Foeniculum Vulgare and Pelargonium Graveolens Essential Oil Mixture Triggers the Cell Cycle Arrest and Apoptosis in MCF-7 Cells. Anti. Cancer Agents Med. Chem. 2019, 19, 1103–1113. [Google Scholar]
- Hussain, A.I.; Anwar, F.; Chatha, S.A.; Jabbar, A.; Mahboob, S.; Nigam, P.S. Rosmarinus officinalis essential oil: Antiproliferative, antioxidant and antibacterial activities. Braz. J. Microbiol. 2010, 41, 1070–1078. [Google Scholar] [CrossRef]
- Li, F.; Rajendran, P.; Sethi, G. Thymoquinone inhibits proliferation, induces apoptosis and chemosensitizes human multiple myeloma cells through suppression of signal transducer and activator of transcription 3 activation pathway. Br. J. Pharmacol. 2010, 161, 541–554. [Google Scholar] [CrossRef]
- Chen, W.; Lu, Y.; Gao, M.; Wu, J.; Wang, A.; Shi, R. Anti-angiogenesis effect of essential oil from Curcuma zedoaria in vitro and in vivo. J. Ethnopharmacol. 2011, 133, 220–226. [Google Scholar] [CrossRef]
- Santana, J.S.; Sartorelli, P.; Guadagnin, R.C.; Matsuo, A.L.; Figueiredo, C.R.; Soares, M.G.; da Silva, A.M.; Lago, J.H. Essential oils from Schinus terebinthifolius leaves—Chemical composition and in vitro cytotoxicity evaluation. Pharm. Biol. 2012, 50, 1248–1253. [Google Scholar] [CrossRef]
- Kumar, A.; Malik, F.; Bhushan, S.; Sethi, V.K.; Shahi, A.K.; kaur, J.; Taneja, S.C.; Qazi, G.N.; Singh, J. An essential oil and its major constituent isointermedeol induce apoptosis by increased expression of mitochondrial cytochrome c and apical death receptors in human leukaemia HL-60 cells. Chem. Biol. Interact. 2008, 171, 332–347. [Google Scholar] [CrossRef]
- Jo, J.-R.; Park, J.S.; Park, Y.-K.; Chae, Y.Z.; Lee, G.-H.; Park, G.-Y.; Jang, B.-C. Pinus densiflora leaf essential oil induces apoptosis via ROS generation and activation of caspases in YD-8 human oral cancer cells. Int. J. Oncol. 2012, 40, 1238–1245. [Google Scholar] [CrossRef]
- Sertel, S.; Eichhorn, T.; Plinkert, P.K.; Efferth, T. Anticancer activity of Salvia officinalis essential oil against HNSCC cell line (UMSCC1). Hno 2011, 59, 1203–1208. [Google Scholar] [CrossRef]
- Chang, H.T.; Hsu, S.S.; Chou, C.T.; Cheng, J.S.; Wang, J.L.; Lin, K.L.; Fang, Y.C.; Chen, W.C.; Chien, J.M.; Lu, T.; et al. Effect of Thymol on Ca2+ Homeostasis and Viability in MG63 Human Osteosarcoma Cells. Pharmacology 2011, 88, 201–212. [Google Scholar] [CrossRef]
- Sharma, P.R.; Mondhe, D.M.; Muthiah, S.; Pal, H.C.; Shahi, A.K.; Saxena, A.K.; Qazi, G.N. Anticancer activity of an essential oil from Cymbopogon flexuosus. Chem. Biol. Interact. 2009, 179, 160–168. [Google Scholar] [CrossRef]
- Slamenová, D.; Horváthová, E.; Sramková, M.; Marsálková, L. DNA-protective effects of two components of essential plant oils carvacrol and thymol on mammalian cells cultured in vitro. Neoplasma 2007, 54, 108–112. [Google Scholar]
- Zu, Y.; Yu, H.; Liang, L.; Fu, Y.; Efferth, T.; Liu, X.; Wu, N. Activities of Ten Essential Oils towards Propionibacterium acnes and PC-3, A-549 and MCF-7 Cancer Cells. Molecules 2010, 15, 3200. [Google Scholar] [CrossRef] [PubMed]
- Yousefzadi, M.; Riahi-Madvar, A.; Hadian, J.; Rezaee, F.; Rafiee, R.; Biniaz, M. Toxicity of essential oil of Satureja khuzistanica: In vitro cytotoxicity and anti-microbial activity. J. Immunotoxicol. 2014, 11, 50–55. [Google Scholar] [CrossRef]
- Nogueira, M.L.; Lima, E.J.S.P.d.; Adrião, A.A.X.; Fontes, S.S.; Silva, V.R.; Santos, L.d.S.; Soares, M.B.P.; Dias, R.B.; Rocha, C.A.G.; Costa, E.V.; et al. Cyperus articulatus L. (Cyperaceae) Rhizome Essential Oil Causes Cell Cycle Arrest in the G2/M Phase and Cell Death in HepG2 Cells and Inhibits the Development of Tumors in a Xenograft Model. Molecules 2020, 25, 2687. [Google Scholar] [CrossRef]
- Abdalla, A.N.; Shaheen, U.; Abdallah, Q.M.A.; Flamini, G.; Bkhaitan, M.M.; Abdelhady, M.I.S.; Ascrizzi, R.; Bader, A. Proapoptotic Activity of Achillea membranacea Essential Oil and Its Major Constituent 1,8-Cineole against A2780 Ovarian Cancer Cells. Molecules 2020, 25, 1582. [Google Scholar] [CrossRef]
- Dahham, S.S.; Tabana, Y.M.; Ahmed Hassan, L.E.; Khadeer Ahamed, M.B.; Abdul Majid, A.S.; Abdul Majid, A.M.S. In vitro antimetastatic activity of Agarwood (Aquilaria crassna) essential oils against pancreatic cancer cells. Alex. J. Med. 2016, 52, 141–150. [Google Scholar] [CrossRef]
- Turek, C.; Stintzing, F.C. Stability of Essential Oils: A Review. Compr. Rev. Food Sci. Food Saf. 2013, 12, 40–53. [Google Scholar] [CrossRef]
- Watkins, R.; Wu, L.; Zhang, C.; Davis, R.M.; Xu, B. Natural product-based nanomedicine: Recent advances and issues. Int. J. Nanomed. 2015, 10, 6055–6074. [Google Scholar] [CrossRef]
- Aljuffali, I.A.; Fang, C.L.; Chen, C.H.; Fang, J.Y. Nanomedicine as a Strategy for Natural Compound Delivery to Prevent and Treat Cancers. Curr. Pharm. Des. 2016, 22, 4219–4231. [Google Scholar] [CrossRef]
- Fonseca-Santos, B.; Chorilli, M. The uses of resveratrol for neurological diseases treatment and insights for nanotechnology based-drug delivery systems. Int. J. Pharm. 2020, 589, 119832. [Google Scholar] [CrossRef]
- AbouAitah, K.; Swiderska-Sroda, A.; Farghali, A.A.; Wojnarowicz, J.; Stefanek, A.; Gierlotka, S.; Opalinska, A.; Allayeh, A.K.; Ciach, T.; Lojkowski, W. Folic acid-conjugated mesoporous silica particles as nanocarriers of natural prodrugs for cancer targeting and antioxidant action. Oncotarget 2018, 9, 26466–26490. [Google Scholar] [CrossRef]
- AbouAitah, K.; Hassan, H.A.; Swiderska-Sroda, A.; Gohar, L.; Shaker, O.G.; Wojnarowicz, J.; Opalinska, A.; Smalc-Koziorowska, J.; Gierlotka, S.; Lojkowski, W. Targeted Nano-Drug Delivery of Colchicine against Colon Cancer Cells by Means of Mesoporous Silica Nanoparticles. Cancers 2020, 12, 144. [Google Scholar] [CrossRef]
- Shahein, S.A.; Aboul-Enein, A.M.; Higazy, I.M.; Abou-Elella, F.; Lojkowski, W.; Ahmed, E.R.; Mousa, S.A.; AbouAitah, K. Targeted anticancer potential against glioma cells of thymoquinone delivered by mesoporous silica core-shell nanoformulations with pH-dependent release. Int. J. Nanomed. 2019, 14, 5503–5526. [Google Scholar] [CrossRef]
- Froiio, F.; Ginot, L.; Paolino, D.; Lebaz, N.; Bentaher, A.; Fessi, H.; Elaissari, A. Essential Oils-Loaded Polymer Particles: Preparation, Characterization and Antimicrobial Property. Polymers 2019, 11, 1071. [Google Scholar] [CrossRef]
- Ali, H.; Al-Khalifa, A.R.; Aouf, A.; Boukhebti, H.; Farouk, A. Effect of nanoencapsulation on volatile constituents, and antioxidant and anticancer activities of Algerian Origanum glandulosum Desf. essential oil. Sci. Rep. 2020, 10, 2812. [Google Scholar] [CrossRef]
- de Matos, S.P.; Teixeira, H.F.; de Lima, Á.A.N.; Veiga-Junior, V.F.; Koester, L.S. Essential Oils and Isolated Terpenes in Nanosystems Designed for Topical Administration: A Review. Biomolecules 2019, 9, 138. [Google Scholar] [CrossRef]
- Sharma, M.; Grewal, K.; Jandrotia, R.; Batish, D.R.; Singh, H.P.; Kohli, R.K. Essential oils as anticancer agents: Potential role in malignancies, drug delivery mechanisms, and immune system enhancement. Biomed. Pharmacother. 2022, 146, 112514. [Google Scholar] [CrossRef]
- Sousa, F.L.; Santos, M.; Rocha, S.M.; Trindade, T. Encapsulation of essential oils in SiO2 microcapsules and release behaviour of volatile compounds. J. Microencapsul. 2014, 31, 627–635. [Google Scholar] [CrossRef]
- Salehi, F.; Jamali, T.; Kavoosi, G.; Ardestani, S.K.; Vahdati, S.N. Stabilization of Zataria essential oil with pectin-based nanoemulsion for enhanced cytotoxicity in monolayer and spheroid drug-resistant breast cancer cell cultures and deciphering its binding mode with gDNA. Int. J. Biol. Macromol. 2020, 164, 3645–3655. [Google Scholar] [CrossRef]
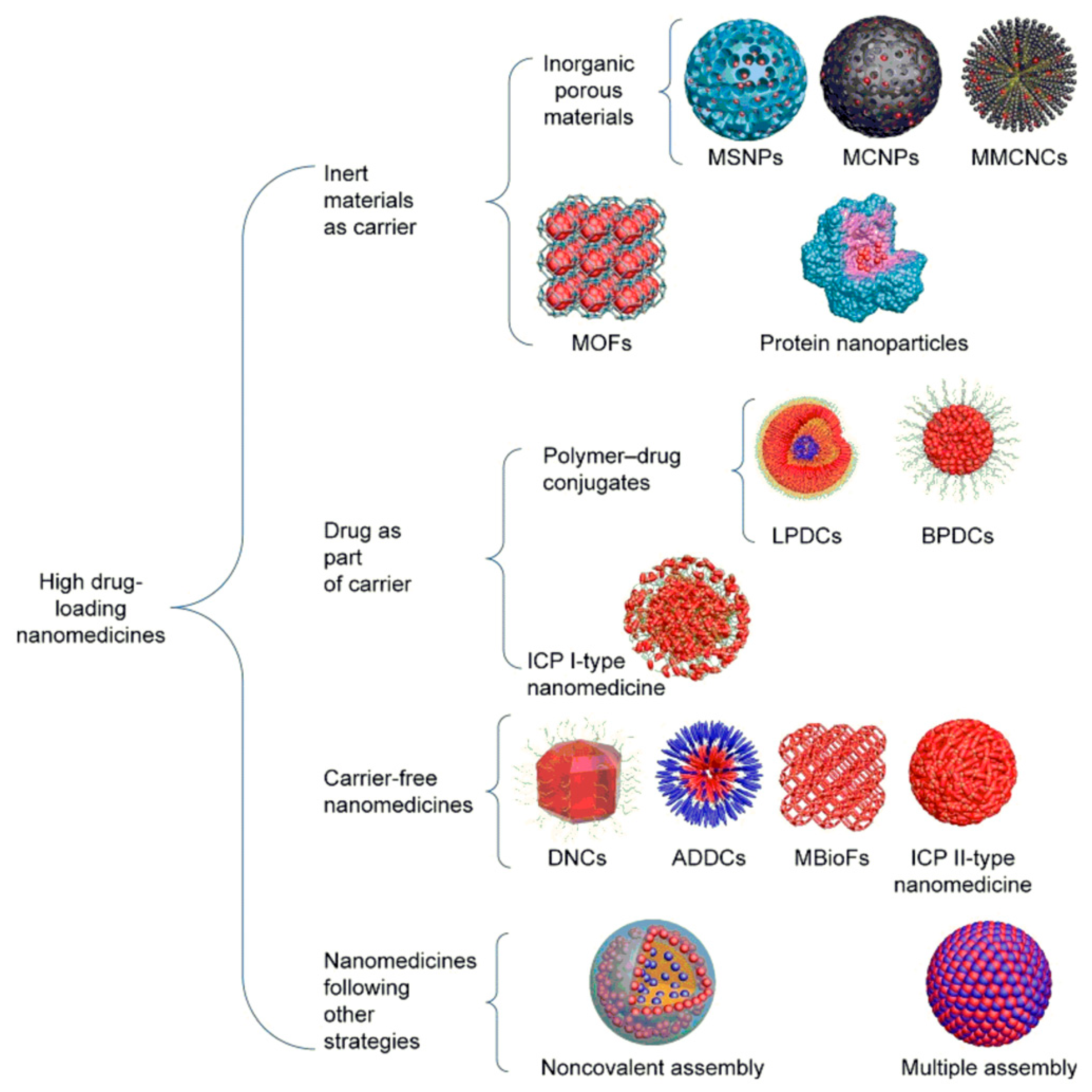
- Liu, Y.; Yang, G.; Jin, S.; Xu, L.; Zhao, C.-X. Development of High-Drug-Loading Nanoparticles. ChemPlusChem 2020, 85, 2143–2157. [Google Scholar] [CrossRef]
- Shen, S.; Wu, Y.; Liu, Y.; Wu, D. High drug-loading nanomedicines: Progress, current status, and prospects. Int. J. Nanomed. 2017, 12, 4085–4109. [Google Scholar] [CrossRef]
- de Matos, S.P.; Lucca, L.G.; Koester, L.S. Essential oils in nanostructured systems: Challenges in preparation and analytical methods. Talanta 2019, 195, 204–214. [Google Scholar] [CrossRef]
- Jia, J.; Liu, X.; Wu, K.; Zhou, X.; Ge, F. Loading zedoary oil into pH-sensitive chitosan grafted mesoporous silica nanoparticles via gate-penetration by supercritical CO2 (GPS). J. CO2 Util. 2019, 33, 12–20. [Google Scholar] [CrossRef]
- Gámez, E.; Elizondo-Castillo, H.; Tascon, J.; García-Salinas, S.; Navascues, N.; Mendoza, G.; Arruebo, M.; Irusta, S. Antibacterial Effect of Thymol Loaded SBA-15 Nanorods Incorporated in PCL Electrospun Fibers. Nanomaterials 2020, 10, 616. [Google Scholar] [CrossRef]
- Keawchaoon, L.; Yoksan, R. Preparation, characterization and in vitro release study of carvacrol-loaded chitosan nanoparticles. Colloids Surf. B Biointerfaces 2011, 84, 163–171. [Google Scholar] [CrossRef]
- Shinde, P.; Agraval, H.; Srivastav, A.K.; Yadav, U.C.S.; Kumar, U. Physico-chemical characterization of carvacrol loaded zein nanoparticles for enhanced anticancer activity and investigation of molecular interactions between them by molecular docking. Int. J. Pharm. 2020, 588, 119795. [Google Scholar] [CrossRef]
- Shetta, A.; Kegere, J.; Mamdouh, W. Comparative study of encapsulated peppermint and green tea essential oils in chitosan nanoparticles: Encapsulation, thermal stability, in-vitro release, antioxidant and antibacterial activities. Int. J. Biol. Macromol. 2019, 126, 731–742. [Google Scholar] [CrossRef]
- Khosh manzar, M.; Pirouzifard, M.K.; Hamishehkar, H.; Pirsa, S. Cocoa butter and cocoa butter substitute as a lipid carrier of Cuminum cyminum L. essential oil; physicochemical properties, physical stability and controlled release study. J. Mol. Liq. 2020, 314, 113638. [Google Scholar] [CrossRef]
- Pires, F.Q.; da Silva, J.K.R.; Sa-Barreto, L.L.; Gratieri, T.; Gelfuso, G.M.; Cunha-Filho, M. Lipid nanoparticles as carriers of cyclodextrin inclusion complexes: A promising approach for cutaneous delivery of a volatile essential oil. Colloids Surf. B Biointerfaces 2019, 182, 110382. [Google Scholar] [CrossRef]
- Gupta, S.; Variyar, P.S. 15—Nanoencapsulation of essential oils for sustained release: Application as therapeutics and antimicrobials. In Encapsulations; Grumezescu, A.M., Ed.; Academic Press: Cambridge, MA, USA, 2016; pp. 641–672. [Google Scholar]
- Soppimath, K.S.; Aminabhavi, T.M.; Kulkarni, A.R.; Rudzinski, W.E. Biodegradable polymeric nanoparticles as drug delivery devices. J. Control. Release 2001, 70, 1–20. [Google Scholar] [CrossRef]
- Matshetshe, K.I.; Parani, S.; Manki, S.M.; Oluwafemi, O.S. Preparation, characterization and in vitro release study of β-cyclodextrin/chitosan nanoparticles loaded Cinnamomum zeylanicum essential oil. Int. J. Biol. Macromol. 2018, 118, 676–682. [Google Scholar] [CrossRef]
- Hariharan, S.; Bhardwaj, V.; Bala, I.; Sitterberg, J.; Bakowsky, U.; Ravi Kumar, M.N. Design of estradiol loaded PLGA nanoparticulate formulations: A potential oral delivery system for hormone therapy. Pharm. Res. 2006, 23, 184–195. [Google Scholar] [CrossRef]
- Hosseini, S.F.; Zandi, M.; Rezaei, M.; Farahmandghavi, F. Two-step method for encapsulation of oregano essential oil in chitosan nanoparticles: Preparation, characterization and in vitro release study. Carbohydr. Polym. 2013, 95, 50–56. [Google Scholar] [CrossRef]
- Karam, T.K.; Ortega, S.; Ueda Nakamura, T.; Auzély-Velty, R.; Nakamura, C.V. Development of chitosan nanocapsules containing essential oil of Matricaria chamomilla L. for the treatment of cutaneous leishmaniasis. Int. J. Biol. Macromol. 2020, 162, 199–208. [Google Scholar] [CrossRef]
- Natrajan, D.; Srinivasan, S.; Sundar, K.; Ravindran, A. Formulation of essential oil-loaded chitosan–alginate nanocapsules. J. Food Drug Anal. 2015, 23, 560–568. [Google Scholar] [CrossRef]
- Deng, X.; Chen, J.; Chen, W. Hydrogel particles as a controlled release delivery system for lavender essential oil using pH triggers. Colloids Surf. A Physicochem. Eng. Asp. 2020, 603, 125134. [Google Scholar] [CrossRef]
- Bautista, G.F.M.; Vidallon, M.L.P.; Salamanez, K.C.; Rodriguez, E.B. Nanodelivery system based on zein-alginate complexes enhances in vitro chemopreventive activity and bioavailability of pomelo [Citrus maxima (Burm.) Merr.] seed limonoids. J. Drug Deliv. Sci. Technol. 2019, 54, 101296. [Google Scholar] [CrossRef]
- Hasani, S.; Ojagh, S.M.; Ghorbani, M. Nanoencapsulation of lemon essential oil in Chitosan-Hicap system. Part 1: Study on its physical and structural characteristics. Int. J. Biol. Macromol. 2018, 115, 143–151. [Google Scholar] [CrossRef]
- Amiri, S.; Rahimi, A. Poly(ε-caprolactone) electrospun nanofibers containing cinnamon essential oil nanocapsules: A promising technique for controlled release and high solubility. J. Ind. Text. 2018, 48, 1527–1544. [Google Scholar] [CrossRef]
- Mallard, I.; Bourgeois, D.; Fourmentin, S. A friendly environmental approach for the controlled release of Eucalyptus essential oil. Colloids Surf. A Physicochem. Eng. Asp. 2018, 549, 130–137. [Google Scholar] [CrossRef]
- Esmaeili, A.; Asgari, A. In vitro release and biological activities of Carum copticum essential oil (CEO) loaded chitosan nanoparticles. Int. J. Biol. Macromol. 2015, 81, 283–290. [Google Scholar] [CrossRef]
- Detsi, A.; Kavetsou, E.; Kostopoulou, I.; Pitterou, I.; Pontillo, A.R.N.; Tzani, A.; Christodoulou, P.; Siliachli, A.; Zoumpoulakis, P. Nanosystems for the Encapsulation of Natural Products: The Case of Chitosan Biopolymer as a Matrix. Pharmaceutics 2020, 12, 669. [Google Scholar] [CrossRef]
- Abreu, F.O.M.S.; Oliveira, E.F.; Paula, H.C.B.; de Paula, R.C.M. Chitosan/cashew gum nanogels for essential oil encapsulation. Carbohydr. Polym. 2012, 89, 1277–1282. [Google Scholar] [CrossRef]
- Fang, Y.; Al-Assaf, S.; Phillips, G.O.; Nishinari, K.; Funami, T.; Williams, P.A. Binding behavior of calcium to polyuronates: Comparison of pectin with alginate. Carbohydr. Polym. 2008, 72, 334–341. [Google Scholar] [CrossRef]
- Spadari, C.d.C.; Lopes, L.B.; Ishida, K. Potential Use of Alginate-Based Carriers As Antifungal Delivery System. Front. Microbiol. 2017, 8, 97. [Google Scholar] [CrossRef]
- Hosseini, S.M.; Hosseini, H.; Mohammadifar, M.A.; Mortazavian, A.M.; Mohammadi, A.; Khosravi-Darani, K.; Shojaee-Aliabadi, S.; Dehghan, S.; Khaksar, R. Incorporation of essential oil in alginate microparticles by multiple emulsion/ionic gelation process. Int. J. Biol. Macromol. 2013, 62, 582–588. [Google Scholar] [CrossRef]
- de Oliveira, E.F.; Paula, H.C.B.; Paula, R.C.M.d. Alginate/cashew gum nanoparticles for essential oil encapsulation. Colloids Surf. B Biointerfaces 2014, 113, 146–151. [Google Scholar] [CrossRef]
- Makadia, H.K.; Siegel, S.J. Poly Lactic-co-Glycolic Acid (PLGA) as Biodegradable Controlled Drug Delivery Carrier. Polymers 2011, 3, 1377–1397. [Google Scholar] [CrossRef]
- Iannitelli, A.; Grande, R.; Di Stefano, A.; Di Giulio, M.; Sozio, P.; Bessa, L.J.; Laserra, S.; Paolini, C.; Protasi, F.; Cellini, L. Potential antibacterial activity of carvacrol-loaded poly(DL-lactide-co-glycolide) (PLGA) nanoparticles against microbial biofilm. Int. J. Mol. Sci. 2011, 12, 5039–5051. [Google Scholar] [CrossRef]
- Gomes, C.; Moreira, R.G.; Castell-Perez, E. Poly (DL-lactide-co-glycolide) (PLGA) nanoparticles with entrapped trans-cinnamaldehyde and eugenol for antimicrobial delivery applications. J. Food Sci. 2011, 76, N16–N24. [Google Scholar] [CrossRef]
- Zhang, Y.; Niu, Y.; Luo, Y.; Ge, M.; Yang, T.; Yu, L.; Wang, Q. Fabrication, characterization and antimicrobial activities of thymol-loaded zein nanoparticles stabilized by sodium caseinate–chitosan hydrochloride double layers. Food Chem. 2014, 142, 269–275. [Google Scholar] [CrossRef]
- Maryam, K.; Shakeri, S.; Kiani, K. Preparation and in vitro investigation of antigastric cancer activities of carvacrol-loaded human serum albumin nanoparticles. IET Nanobiotechnol. 2015, 9, 294–299. [Google Scholar] [CrossRef]
- Gregory, G. Liposome Technology, 3rd ed.; CRC Press: Boca Raton, FL, USA, 2006. [Google Scholar]
- Gulati, M.; Grover, M.; Singh, S.; Singh, M. Lipophilic drug derivatives in liposomes. Int. J. Pharm. 1998, 165, 129–168. [Google Scholar] [CrossRef]
- Mishra, G.P.; Bagui, M.; Tamboli, V.; Mitra, A.K. Recent Applications of Liposomes in Ophthalmic Drug Delivery. J. Drug Deliv. 2011, 2011, 863734. [Google Scholar] [CrossRef]
- Rongen, H.A.H.; Bult, A.; van Bennekom, W.P. Liposomes and immunoassays. J. Immunol. Methods 1997, 204, 105–133. [Google Scholar] [CrossRef]
- Voinea, M.; Simionescu, M. Designing of ‘intelligent’ liposomes for efficient delivery of drugs. J. Cell Mol. Med. 2002, 6, 465–474. [Google Scholar] [CrossRef]
- Risaliti, L.; Kehagia, A.; Daoultzi, E.; Lazari, D.; Bergonzi, M.C.; Vergkizi-Nikolakaki, S.; Hadjipavlou-Litina, D.; Bilia, A.R. Liposomes loaded with Salvia triloba and Rosmarinus officinalis essential oils: In vitro assessment of antioxidant, antiinflammatory and antibacterial activities. J. Drug Deliv. Sci. Technol. 2019, 51, 493–498. [Google Scholar] [CrossRef]
- Risaliti, L.; Pini, G.; Ascrizzi, R.; Donato, R.; Sacco, C.; Bergonzi, M.C.; Salvatici, M.C.; Bilia, A.R. Artemisia annua essential oil extraction, characterization, and incorporation in nanoliposomes, smart drug delivery systems against Candida species. J. Drug Deliv. Sci. Technol. 2020, 59, 101849. [Google Scholar] [CrossRef]
- Hammoud, Z.; Gharib, R.; Fourmentin, S.; Elaissari, A.; Greige-Gerges, H. Drug-in-hydroxypropyl-β-cyclodextrin-in-lipoid S100/cholesterol liposomes: Effect of the characteristics of essential oil components on their encapsulation and release. Int. J. Pharm. 2020, 579, 119151. [Google Scholar] [CrossRef]
- Hammoud, Z.; Gharib, R.; Fourmentin, S.; Elaissari, A.; Greige-Gerges, H. New findings on the incorporation of essential oil components into liposomes composed of lipoid S100 and cholesterol. Int. J. Pharm. 2019, 561, 161–170. [Google Scholar] [CrossRef]
- Thonggoom, O.; Punrattanasin, N.; Srisawang, N.; Promawan, N.; Thonggoom, R. In vitro controlled release of clove essential oil in self-assembly of amphiphilic polyethylene glycol-block-polycaprolactone. J. Microencapsul. 2016, 33, 239–248. [Google Scholar] [CrossRef]
- Zhao, Y.; Chang, Y.X.; Hu, X.; Liu, C.Y.; Quan, L.H.; Liao, Y.H. Solid lipid nanoparticles for sustained pulmonary delivery of Yuxingcao essential oil: Preparation, characterization and in vivo evaluation. Int. J. Pharm. 2017, 516, 364–371. [Google Scholar] [CrossRef]
- Rodenak-Kladniew, B.; Islan, G.A.; de Bravo, M.G.; Durán, N.; Castro, G.R. Design, characterization and in vitro evaluation of linalool-loaded solid lipid nanoparticles as potent tool in cancer therapy. Colloids Surf. B Biointerfaces 2017, 154, 123–132. [Google Scholar] [CrossRef]
- Moghimipour, E.; Ramezani, Z.; Handali, S. Solid lipid nanoparticles as a delivery system for Zataria multiflora essential oil: Formulation and characterization. Curr. Drug Deliv. 2013, 10, 151–157. [Google Scholar] [CrossRef]
- Li, Y.; Wu, Z.F.; Kuang, W.W.; Li, Y.H.; Chen, B.Y.; Yang, M. Effect of nanostructured lipid carriers (NLCs) in improving stability of essential oils and its application. Zhongguo Zhong Yao Za Zhi 2020, 45, 523–530. [Google Scholar] [CrossRef]
- Vieira, R.; Severino, P.; Nalone, L.A.; Souto, S.B.; Silva, A.M.; Lucarini, M.; Durazzo, A.; Santini, A.; Souto, E.B. Sucupira Oil-Loaded Nanostructured Lipid Carriers (NLC): Lipid Screening, Factorial Design, Release Profile, and Cytotoxicity. Molecules 2020, 25, 685. [Google Scholar] [CrossRef]
- Kfoury, M.; Balan, R.; Landy, D.; Nistor, D.; Fourmentin, S. Investigation of the complexation of essential oil components with cyclodextrins. Supramol. Chem. 2015, 27, 620–628. [Google Scholar] [CrossRef]
- Ciobanu, A.; Mallard, I.; Landy, D.; Brabie, G.; Nistor, D.; Fourmentin, S. Retention of aroma compounds from Mentha piperita essential oil by cyclodextrins and crosslinked cyclodextrin polymers. Food Chem. 2013, 138, 291–297. [Google Scholar] [CrossRef]
- Wadhwa, G.; Kumar, S.; Chhabra, L.; Mahant, S.; Rao, R. Essential oil–cyclodextrin complexes: An updated review. J. Incl. Phenom. Macrocycl. Chem. 2017, 89, 39–58. [Google Scholar] [CrossRef]
- Marques, H.M.C. A review on cyclodextrin encapsulation of essential oils and volatiles. Flavour. Fragr. J. 2010, 25, 313–326. [Google Scholar] [CrossRef]
- Cetin Babaoglu, H.; Bayrak, A.; Ozdemir, N.; Ozgun, N. Encapsulation of clove essential oil in hydroxypropyl beta-cyclodextrin for characterization, controlled release, and antioxidant activity. J. Food Process. Preserv. 2017, 41, e13202. [Google Scholar] [CrossRef]
- Li, Z.; Wen, W.; Chen, X.; Zhu, L.; Cheng, G.; Liao, Z.; Huang, H.; Ming, L. Release characteristics of an essential oil component encapsulated with cyclodextrin shell matrices. Curr. Drug Deliv. 2020, 18, 487–499. [Google Scholar] [CrossRef] [PubMed]
- Pednekar, P.P.; Godiyal, S.C.; Jadhav, K.R.; Kadam, V.J. Chapter 23—Mesoporous silica nanoparticles: A promising multifunctional drug delivery system. In Nanostructures for Cancer Therapy; Ficai, A., Grumezescu, A.M., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 593–621. [Google Scholar]
- Le Hoang Doan, T.; Mai, N.X.D.; Matsumoto, K.; Tamanoi, F. Chapter Four—Tumor Targeting and Tumor Growth Inhibition Capability of Mesoporous Silica Nanoparticles in Mouse Models. In The Enzymes; Tamanoi, F., Ed.; Academic Press: Cambridge, MA, USA, 2018; Volume 44, pp. 61–82. [Google Scholar]
- Mitran, R.-A.; Deaconu, M.; Matei, C.; Berger, D. Chapter 11—Mesoporous Silica as Carrier for Drug-Delivery Systems. In Nanocarriers for Drug Delivery; Mohapatra, S.S., Ranjan, S., Dasgupta, N., Mishra, R.K., Thomas, S., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 351–374. [Google Scholar]
- Ambrogio, M.W.; Thomas, C.R.; Zhao, Y.-L.; Zink, J.I.; Stoddart, J.F. Mechanized Silica Nanoparticles: A New Frontier in Theranostic Nanomedicine. Acc. Chem. Res. 2011, 44, 903–913. [Google Scholar] [CrossRef] [PubMed]
- Mekaru, H.; Lu, J.; Tamanoi, F. Development of mesoporous silica-based nanoparticles with controlled release capability for cancer therapy. Adv. Drug Deliv. Rev. 2015, 95, 40–49. [Google Scholar] [CrossRef]
- Zhang, Q.; Wang, X.; Li, P.-Z.; Nguyen, K.T.; Wang, X.-J.; Luo, Z.; Zhang, H.; Tan, N.S.; Zhao, Y. Biocompatible, Uniform, and Redispersible Mesoporous Silica Nanoparticles for Cancer-Targeted Drug Delivery In Vivo. Adv. Funct. Mater. 2014, 24, 2450–2461. [Google Scholar] [CrossRef]
- Slowing, I.I.; Vivero-Escoto, J.L.; Wu, C.-W.; Lin, V.S.Y. Mesoporous silica nanoparticles as controlled release drug delivery and gene transfection carriers. Adv. Drug Deliv. Rev. 2008, 60, 1278–1288. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Liu, T.; Fu, C.; Tan, L.; Meng, X.; Liu, H. Biodistribution, excretion, and toxicity of mesoporous silica nanoparticles after oral administration depend on their shape. Nanomed. Nanotechnol. Biol. Med. 2015, 11, 1915–1924. [Google Scholar] [CrossRef] [PubMed]
- Mamaeva, V.; Sahlgren, C.; Lindén, M. Mesoporous silica nanoparticles in medicine—Recent advances. Adv. Drug Deliv. Rev. 2013, 65, 689–702. [Google Scholar] [CrossRef]
- Liong, M.; Lu, J.; Kovochich, M.; Xia, T.; Ruehm, S.G.; Nel, A.E.; Tamanoi, F.; Zink, J.I. Multifunctional inorganic nanoparticles for imaging, targeting, and drug delivery. ACS Nano 2008, 2, 889–896. [Google Scholar] [CrossRef]
- AbouAitah, K.; Lojkowski, W. Delivery of Natural Agents by Means of Mesoporous Silica Nanospheres as a Promising Anticancer Strategy. Pharmaceutics 2021, 13, 143. [Google Scholar] [CrossRef]
- Bisht, S.; Feldmann, G.; Soni, S.; Ravi, R.; Karikar, C.; Maitra, A.; Maitra, A. Polymeric nanoparticle-encapsulated curcumin (“nanocurcumin”): A novel strategy for human cancer therapy. J. Nanobiotechnol. 2007, 5, 3. [Google Scholar] [CrossRef]
- Pool, H.; Quintanar, D.; Figueroa, J.d.D.; Marinho Mano, C.; Bechara, J.E.H.; Godínez, L.A.; Mendoza, S. Antioxidant Effects of Quercetin and Catechin Encapsulated into PLGA Nanoparticles. J. Nanomater. 2012, 2012, 145380. [Google Scholar] [CrossRef]
- Rana, A.; Matiyani, M.; Tewari, C.; Negi, P.B.; Chandra Arya, M.; Das, V.; Pal, M.; Sahoo, N.G. Functionalized graphene oxide based nanocarrier for enhanced cytotoxicity of Juniperus squamata root essential oil against breast cancer cells. J. Drug Deliv. Sci. Technol. 2022, 72, 103370. [Google Scholar] [CrossRef]
- Bashlouei, S.G.; Karimi, E.; Zareian, M.; Oskoueian, E.; Shakeri, M. Heracleum persicum Essential Oil Nanoemulsion: A Nanocarrier System for the Delivery of Promising Anticancer and Antioxidant Bioactive Agents. Antioxidants 2022, 11, 831. [Google Scholar] [CrossRef] [PubMed]
- Lalami, Z.A.; Tafvizi, F.; Naseh, V.; Salehipour, M. Characterization and optimization of co-delivery Farnesol-Gingerol Niosomal formulation to enhance anticancer activities against breast cancer cells. J. Drug Deliv. Sci. Technol. 2022, 72, 103371. [Google Scholar] [CrossRef]
- Salehi, F.; Behboudi, H.; Kavoosi, G.; Ardestani, S.K. Incorporation of Zataria multiflora essential oil into chitosan biopolymer nanoparticles: A nanoemulsion based delivery system to improve the in-vitro efficacy, stability and anticancer activity of ZEO against breast cancer cells. Int. J. Biol. Macromol. 2020, 143, 382–392. [Google Scholar] [CrossRef]
- Periasamy, V.S.; Athinarayanan, J.; Alshatwi, A.A. Anticancer activity of an ultrasonic nanoemulsion formulation of Nigella sativa L. essential oil on human breast cancer cells. Ultrason. Sonochem. 2016, 31, 449–455. [Google Scholar] [CrossRef]
- Kavaz, D.; Idris, M.; Onyebuchi, C. Physiochemical characterization, antioxidative, anticancer cells proliferation and food pathogens antibacterial activity of chitosan nanoparticles loaded with Cyperus articulatus rhizome essential oils. Int. J. Biol. Macromol. 2019, 123, 837–845. [Google Scholar] [CrossRef]
- Alkhatib, M.H.; AlMotwaa, S.M.; Alkreathy, H.M. Incorporation of ifosfamide into various essential oils -based nanoemulsions ameliorates its apoptotic effect in the cancers cells. Sci. Rep. 2019, 9, 695. [Google Scholar] [CrossRef]
- Panyajai, P.; Chueahongthong, F.; Viriyaadhammaa, N.; Nirachonkul, W.; Tima, S.; Chiampanichayakul, S.; Anuchapreeda, S.; Okonogi, S. Anticancer activity of Zingiber ottensii essential oil and its nanoformulations. PLoS ONE 2022, 17, e0262335. [Google Scholar] [CrossRef]
- Khan, I.; Bahuguna, A.; Kumar, P.; Bajpai, V.K.; Kang, S.C. In vitro and in vivo antitumor potential of carvacrol nanoemulsion against human lung adenocarcinoma A549 cells via mitochondrial mediated apoptosis. Sci. Rep. 2018, 8, 144. [Google Scholar] [CrossRef]
- Rajivgandhi, G.; Saravanan, K.; Ramachandran, G.; Li, J.-L.; Yin, L.; Quero, F.; Alharbi, N.S.; Kadaikunnan, S.; Khaled, J.M.; Manoharan, N.; et al. Enhanced anti-cancer activity of chitosan loaded Morinda citrifolia essential oil against A549 human lung cancer cells. Int. J. Biol. Macromol. 2020, 164, 4010–4021. [Google Scholar] [CrossRef]
- Manju, S.; Malaikozhundan, B.; Vijayakumar, S.; Shanthi, S.; Jaishabanu, A.; Ekambaram, P.; Vaseeharan, B. Antibacterial, antibiofilm and cytotoxic effects of Nigella sativa essential oil coated gold nanoparticles. Microb. Pathog. 2016, 91, 129–135. [Google Scholar] [CrossRef]
- Trinh, L.H.; Takzare, A.; Ghafoor, D.D.; Siddiqi, A.F.; Ravali, S.; Shalbaf, M.; Bakhtiar, M. Trachyspermum copticum essential oil incorporated niosome for cancer treatment. J. Drug Deliv. Sci. Technol. 2019, 52, 818–824. [Google Scholar] [CrossRef]
- Parvez, S.; Karole, A.; Mudavath, S.L. Fabrication, physicochemical characterization and In vitro anticancer activity of nerolidol encapsulated solid lipid nanoparticles in human colorectal cell line. Colloids Surf. B Biointerfaces 2022, 215, 112520. [Google Scholar] [CrossRef]
- Khatamian, N.; Homayouni Tabrizi, M.; Ardalan, P.; Yadamani, S.; Darchini Maragheh, A. Synthesis of Carum Carvi essential oil nanoemulsion, the cytotoxic effect, and expression of caspase 3 gene. J. Food Biochem. 2019, 43, e12956. [Google Scholar] [CrossRef]
- Marchese, E.; D‘Onofrio, N.; Balestrieri, M.L.; Castaldo, D.; Ferrari, G.; Donsì, F. Bergamot essential oil nanoemulsions: Antimicrobial and cytotoxic activity. Z Nat. C J. Biosci 2020, 75, 279–290. [Google Scholar] [CrossRef]
- Detoni, C.B.; de Oliveira, D.M.; Santo, I.E.; Pedro, A.S.; El-Bacha, R.; da Silva Velozo, E.; Ferreira, D.; Sarmento, B.; de Magalhães Cabral-Albuquerque, E.C. Evaluation of thermal-oxidative stability and antiglioma activity of Zanthoxylum tingoassuiba essential oil entrapped into multi- and unilamellar liposomes. J. Liposome Res. 2012, 22, 1–7. [Google Scholar] [CrossRef]
- Celia, C.; Trapasso, E.; Locatelli, M.; Navarra, M.; Ventura, C.A.; Wolfram, J.; Carafa, M.; Morittu, V.M.; Britti, D.; Di Marzio, L.; et al. Anticancer activity of liposomal bergamot essential oil (BEO) on human neuroblastoma cells. Colloids Surf. B Biointerfaces 2013, 112, 548–553. [Google Scholar] [CrossRef]
- Gomes, M.R.F.; Schuh, R.S.; Jacques, A.L.B.; Augustin, O.A.; Bordignon, S.A.L.; Dias, D.O.; Kelmann, R.G.; Koester, L.S.; Gehring, M.P.; Morrone, F.B.; et al. Citotoxic activity evaluation of essential oils and nanoemulsions of Drimys angustifolia and D. brasiliensis on human glioblastoma (U-138 MG) and human bladder carcinoma (T24) cell lines in vitro. Rev. Bras. Farmacogn. 2013, 23, 259–267. [Google Scholar] [CrossRef][Green Version]
- Li, Z.; Veeraraghavan, V.P.; Mohan, S.K.; Bolla, S.R.; Lakshmanan, H.; Kumaran, S.; Aruni, W.; Aladresi, A.A.M.; Shair, O.H.M.; Alharbi, S.A.; et al. Apoptotic induction and anti-metastatic activity of eugenol encapsulated chitosan nanopolymer on rat glioma C6 cells via alleviating the MMP signaling pathway. J. Photochem. Photobiol. B 2020, 203, 111773. [Google Scholar] [CrossRef]
- Zhang, Y.; Xin, C.; Cheng, C.; Wang, Z. Antitumor activity of nanoemulsion based on essential oil of Pinus koraiensis pinecones in MGC-803 tumor-bearing nude mice. Arab. J. Chem. 2020, 13, 8226–8238. [Google Scholar] [CrossRef]
- Alkhatib, M.H. Apoptotic Effect of Bleomycin Formulated in Cinnamon Oil Nanoemulsion on HeLa Cervical Cancer Cells. Asian J. Pharm. 2020, 14, 361. [Google Scholar] [CrossRef]
- Nirmala, M.J.; Durai, L.; Gopakumar, V.; Nagarajan, R. Preparation of Celery Essential Oil-Based Nanoemulsion by Ultrasonication and Evaluation of Its Potential Anticancer and Antibacterial Activity. Int. J. Nanomed. 2020, 15, 7651–7666. [Google Scholar] [CrossRef]
- Tubtimsri, S.; Limmatvapirat, C.; Limsirichaikul, S.; Akkaramongkolporn, P.; Inoue, Y.; Limmatvapirat, S. Fabrication and characterization of spearmint oil loaded nanoemulsions as cytotoxic agents against oral cancer cell. Asian J. Pharm. Sci. 2018, 13, 425–437. [Google Scholar] [CrossRef]
- Nirmala, M.J.; Durai, L.; Rao, K.A.; Nagarajan, R. Ultrasonic Nanoemulsification of Cuminum cyminum Essential Oil and Its Applications in Medicine. Int. J. Nanomed. 2020, 15, 795–807. [Google Scholar] [CrossRef]
- Valizadeh, A.; Khaleghi, A.A.; Roozitalab, G.; Osanloo, M. High anticancer efficacy of solid lipid nanoparticles containing Zataria multiflora essential oil against breast cancer and melanoma cell lines. BMC Pharm. Toxicol. 2021, 22, 52. [Google Scholar] [CrossRef]
- Dawaba, A.M.; Dawaba, H.M. Application of Optimization Technique to Develop Nano-Based Carrier of Nigella Sativa Essential Oil: Characterization and Assessment. Recent Pat. Drug Deliv. 2019, 13, 228–240. [Google Scholar] [CrossRef]
- Han, H.D.; Cho, Y.-J.; Cho, S.K.; Byeon, Y.; Jeon, H.N.; Kim, H.-S.; Kim, B.-G.; Bae, D.-S.; Lopez-Berestein, G.; Sood, A.K.; et al. Linalool-Incorporated Nanoparticles as a Novel Anticancer Agent for Epithelial Ovarian Carcinoma. Mol. Cancer Ther. 2016, 15, 618–627. [Google Scholar] [CrossRef]

| EO | Nanocarrier | Loading Capacity | Loading Method | References |
|---|---|---|---|---|
| Jasmine | Pectin/chitosan nanoparticles | 0.43–6.06% | Direct preparation method | [49] |
| Zedoary | Chitosan-grafted mesoporous silica nanoparticles | 41% | Gate-penetration by supercritical CO2 | [109] |
| Thymol compound | SBA-15 mesoporous silica nanoparticles | ~71 | Post-fabrication method | [110] |
| Carvacrol compound | Chitosan | Up to 21 | Direct preparation method | [111] |
| Carvacrol compound | Zein | Up to 13% | Direct preparation method | [112] |
| Peppermint and green tea | Chitosan nanoparticles | Up to 23% | Direct preparation method | [113] |
| Cuminum cyminum | Nano lipid carrier | Over 70% | Direct preparation method | [114] |
| Lippia origanoides | Nanostructured lipid carriers/cyclodextrin inclusion complexes | 2.6% | Direct preparation method | [115] |
| EO | Nanocarrier | In Vitro Experiment | Analysis Technique | References |
|---|---|---|---|---|
| Peppermint and green tea | Chitosan nanoparticles | Lyophilized encapsulated nanoparticles were placed in a dialysis bag containing release media: PBS or acetate buffer. This bag was incubated in an additional volume of the release medium. Left under gentle shaking at room temperature. Sampling: a predetermined volume of release media was taken at specific intervals, then an equal volume of fresh release media was added during the entire experiment. | UV–Vis spectrophotometer | [113] |
| Chamomile | Chitosan nanocapsules | Freeze-dried nanocapsules placed in PBS buffer release medium containing 0.5% Tween 80 adjusted to pH 5.5 or 7.4. The solution was orbitally stirred (150 rpm) at 37 °C. Sampling: at predetermined times, the solution was washed twice with hexane (using a separatory funnel), and the supernatant was used for analysis. | Gas chromatography (GC) | [121] |
| Turmeric and lemongrass | Chitosan-alginate nanocapsules | The loaded nanocapsules were dispersed in a small volume of PBS solution and put in a dialysis bag, then surrounded by PBS containing 20% ethanol adjusted to pH 1.5 and 7.4. Next, the solution was incubated at 37 °C under gentle agitation. Sampling: a specific volume was withdrawn and replaced with fresh medium. | UV–Vis spectrophotometer | [122] |
| Lavender | Hydrogel microparticles | Headspace GC | GC analysis | [123] |
| Pomelo citrus | Zein-alginate solid nanoparticles | The lyophilized encapsulated nanoparticles were placed in simulated fluid into dialysis bags, sealed, and suspended in the same simulated fluid. The solution was stirred (100 rpm) at 37 °C. Sampling: a known volume was taken from the solution and replaced with the same volume of fresh simulated fluid. For analysis, and the withdrawn sample was partitioned with an equal volume of ethyl acetate. | UV–Vis spectrophotometer | [124] |
| Lemon | Chitosan-modified starch nanocapsules | Freeze-dried nanocapsules were placed in a microtube containing 40% ethanol. This mixture solution was incubated at room temperature under stirring. Sampling: at specified time intervals, the samples were centrifuged. The supernatant was removed for analysis. | UV–Vis spectrophotometer | [125] |
| Cinnamon | Poly(e-caprolactone) electrospun nanofibers | The nanofibers were placed in a simulated dissolution medium at 37 °C, and the paddle was set at 100 r/min. Sampling: at fixed time intervals, aliquots were taken and replaced with an equal volume of fresh release medium. The samples were filtered and diluted to be used for analysis. | UV–vis spectrophotometer | [126] |
| Eucalyptus | Β-cyclodextrins, functionalized zeolite materials | Headspace extraction with GC | GC analysis | [127] |
| Cinnamon | Β-cyclodextrin/ chitosan nanoparticles | The loaded nanoparticles were dispersed in acetate and phosphate buffers and vortexed at room temperature. Sampling: at predetermined specific time intervals, the samples were centrifuged (for 5 min at 14,000 rpm); then, aliquots of the supernatant were withdrawn (for analysis) and replaced with the same volume of buffer, and the experiment continued. | UV–Vis spectrophotometer | [118] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
AbouAitah, K.; Lojkowski, W. Nanomedicine as an Emerging Technology to Foster Application of Essential Oils to Fight Cancer. Pharmaceuticals 2022, 15, 793. https://doi.org/10.3390/ph15070793
AbouAitah K, Lojkowski W. Nanomedicine as an Emerging Technology to Foster Application of Essential Oils to Fight Cancer. Pharmaceuticals. 2022; 15(7):793. https://doi.org/10.3390/ph15070793
Chicago/Turabian StyleAbouAitah, Khaled, and Witold Lojkowski. 2022. "Nanomedicine as an Emerging Technology to Foster Application of Essential Oils to Fight Cancer" Pharmaceuticals 15, no. 7: 793. https://doi.org/10.3390/ph15070793
APA StyleAbouAitah, K., & Lojkowski, W. (2022). Nanomedicine as an Emerging Technology to Foster Application of Essential Oils to Fight Cancer. Pharmaceuticals, 15(7), 793. https://doi.org/10.3390/ph15070793








