[99mTc]Tc-iFAP/SPECT Tumor Stroma Imaging: Acquisition and Analysis of Clinical Images in Six Different Cancer Entities
Abstract
:1. Introduction
2. Results
3. Discussion
4. Materials and Methods
4.1. Reagents
4.2. [99mTc]Tc-iFAP Preparation
4.3. Patients
4.4. Image Acquisition
4.5. Image Analysis
4.6. Tumor Tissue Samples
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| No. | Age (years) | Gender | Clinical Setting | Type of Cancer | Extent of Cancer |
|---|---|---|---|---|---|
| 1 | 69 | Male | Initial staging | Lung cancer, NSCLC (adenocarcinoma). | Primary, lymph node, bone |
| 2 | 51 | Female | Initial staging | Lung cancer, NSCLC (adenocarcinoma). | Primary, lymph node |
| 3 | 66 | Female | Restaging | Cervical cancer (squamous cell carcinoma). | Lymph node |
| 4 | 60 | Female | Initial staging | Cervical cancer (squamous cell carcinoma). | Primary |
| 5 | 91 | Female | Initial staging | Cervical cancer (squamous cell carcinoma). Breast cancer (ductal carcinoma; SBR 7, G2, moderate DR, Ki67 50%). Luminal B HER2+ | Primary Primary, lymph node |
| 6 | 70 | Female | Initial staging | Breast cancer (ductal carcinoma; SBR 8, G3, moderate DR, Ki67 70%). Triple-negative. | Primary, lymph node |
| 7 | 44 | Female | Initial staging | Breast cancer (ductal carcinoma; SBR 9, G3, mild DR, Ki67 70%). Triple-negative. | Primary, lymph node |
| 8 | 54 | Female | Initial staging | Breast cancer (ductal carcinoma; SBR 6, G2, mild DR). Luminal A | Primary, lymph node |
| 9 | 49 | Female | Initial staging | Breast cancer (ductal carcinoma; SBR 7, G2, mild DR, Ki67 40%). HER2+ | Primary, lymph node |
| 10 | 40 | Female | Initial staging | Breast cancer (ductal carcinoma; SBR 8, G3, moderate DR, Ki67 70%). Triple-negative. | Primary, lymph node |
| 11 | 28 | Female | Initial staging | Breast cancer (ductal carcinoma; SBR 8, G3, Ki67 60%). Triple-negative. | Primary, lymph node |
| 12 | 29 | Female | Initial staging | Breast cancer (ductal carcinoma; SBR 8, G3, moderate DR, Ki67 60%). Luminal B | Primary, lymph node |
| 13 | 60 | Female | Initial staging | Breast cancer (ductal carcinoma; SBR 9, G3, moderate DR, Ki67 30%). Luminal B HER2+ | Primary, lymph node |
| 14 | 55 | Female | Initial staging | Breast cancer (ductal carcinoma; SBR 6, G2, mild DR, Ki67 15%). Luminal B | Primary, lymph node |
| 15 | 55 | Female | Initial staging | Breast cancer (ductal carcinoma; SBR 5, G1, moderate DR). Luminal A. | Lymph node |
| 16 | 36 | Female | Initial staging | Breast cancer (ductal carcinoma; SBR 8, G3, mild DR, Ki67 80%). Triple-negative. | Primary, lymph node |
| 17 | 41 | Female | Initial staging | Breast cancer (ductal carcinoma; SBR 6, G2, moderate DR, Ki67 40%). Luminal B HER2+ | Primary, lymph node |
| 18 | 48 | Female | Initial staging | Breast cancer (ductal carcinoma; SBR 8, G3, mild DR, Ki67 30%). Luminal B HER2+ | Primary, lymph node, lung |
| 19 | 46 | Female | Initial staging | Breast cancer (ductal carcinoma; SBR 7, G2). Luminal B HER2+ | Primary, lymph node, liver |
| 20 | 58 | Female | Initial staging | Breast cancer (ductal carcinoma; SBR 6, G2, Ki67 30%). Luminal B. | Primary, lymph node |
| 21 | 63 | Female | Initial staging | Breast cancer (ductal carcinoma; SBR 7, G2, mild DR, Ki67 50%). Her2+ | Primary, lymph node |
| 22 | 44 | Female | Initial staging | Breast cancer (ductal carcinoma; SBR 9, G3, mild DR, Ki67 70%). Triple-negative. | Primary, lymph node |
| 23 | 42 | Female | Initial staging | Breast cancer (ductal carcinoma; SBR 8, G3, mild DR, Ki67 80%). Triple-negative. | Primary, lymph node |
| 24 | 68 | Female | Initial staging | Breast cancer (ductal carcinoma; SBR 9, G3, Ki67 50%). Triple-negative. | Primary, lymph node, lung |
| 25 | 55 | Female | Initial staging | Breast cancer (ductal carcinoma; SBR 9, G3, moderate DR, Ki67 60%). Triple-negative. | Primary |
| 26 | 37 | Female | Restaging | Glioblastoma NOS (WHO IV) | Primary |
| 27 | 76 | Male | Initial staging | Anaplastic astrocytoma NOS (WHO III) | Primary |
| 28 | 40 | Female | Initial staging | Astrocytoma NOS (WHO II) | Primary |
| 29 | 32 | Female | Restaging | Anaplastic astrocytoma NOS (WHO III) | Primary |
| 30 | 27 | Male | Restaging | Anaplastic astrocytoma NOS (WHO III) | Primary |
| 31 | 47 | Male | Restaging | Colorectal cancer (adenocarcinoma). | Peritoneal carcinomatosis |
| 32 | 23 | Female | Initial staging | Adrenal cortical neuroendocrine tumor (poorly differentiated, Ki67 30%) | Primary, lung |
References
- Hamson, E.J.; Keane, F.M.; Tholen, S.; Schilling, O.; Gorrell, M.D. Understanding fibroblast activation protein (FAP): Substrates, activities, expression and targeting for cancer therapy. Proteom. Clin. Appl. 2014, 8, 454–463. [Google Scholar] [CrossRef] [PubMed]
- Altmann, A.; Haberkorn, U.; Siveke, J. The Latest Developments in Imaging of Fibroblast Activation Protein. J. Nucl. Med. 2021, 62, 160–167. [Google Scholar] [CrossRef] [PubMed]
- Lindner, T.; Altmann, A.; Krämer, S.; Kleist, C.; Loktev, A.; Kratochwil, C.; Giesel, F.; Mier, W.; Marme, F.; Debus, J.; et al. Design and Development of 99mTc-Labeled FAPI Tracers for SPECT Imaging and 188Re Therapy. J. Nucl. Med. 2020, 61, 1507–1513. [Google Scholar] [CrossRef] [PubMed]
- Loktev, A.; Lindner, T.; Mier, W.; Debus, J.; Altmann, A.; Jäger, D.; Giesel, F.; Kratochwil, C.; Barthe, P.; Roumestand, C.; et al. A tumor-imaging method targeting cancer-associated fibroblasts. J. Nucl. Med. 2018, 59, 1423–1429. [Google Scholar] [CrossRef]
- Lindner, T.; Loktev, A.; Altmann, A.; Giesel, F.; Kratochwil, C.; Debus, J.; Jäger, D.; Mier, W.; Haberkorn, U. Development of quinoline-based theranostic ligands for the targeting of fibroblast activation protein. J. Nucl. Med. 2018, 59, 1415–1422. [Google Scholar] [CrossRef] [Green Version]
- Giesel, F.L.; Adeberg, S.; Syed, M.; Lindner, T.; Jiménez-Franco, L.D.; Mavriopoulou, E.; Staudinger, F.; Tonndorf-Martini, E.; Regnery, S.; Rieken, S.; et al. FAPI-74 PET/CT Using Either 18F-AlF or Cold-Kit 68Ga Labeling: Biodistribution, Radiation Dosimetry, and Tumor Delineation in Lung Cancer Patients. J. Nucl. Med. 2021, 62, 201–207. [Google Scholar] [CrossRef]
- Baum, R.P.; Schuchardt, C.; Singh, A.; Chantadisai, M.; Robiller, F.C.; Zhang, J.; Mueller, D.; Eismant, A.; Almaguel, F.; Zboralski, D.; et al. Biodistribution, and Preliminary Dosimetry in Peptide-Targeted Radionuclide Therapy of Diverse Adenocarcinomas Using 177Lu-FAP-2286: First-in-Humans Results. J. Nucl. Med. 2022, 63, 415–423. [Google Scholar] [CrossRef]
- Trujillo-Benítez, D.; Luna-Gutiérrez, M.; Ferro-Flores, G.; Ocampo-García, B.; Santos-Cuevas, C.; Bravo-Villegas, G.; Morales-Ávila, E.; Cruz-Nova, P.; Díaz-Nieto, L.; García-Quiroz, J.; et al. Design, Synthesis and Preclinical Assessment of 99mTc-iFAP for In Vivo Fibroblast Activation Protein (FAP) Imaging. Molecules 2022, 27, 264. [Google Scholar] [CrossRef]
- Coria-Domínguez, L.; Vallejo-Armenta, P.; Luna-Gutiérrez, M.; Ocampo-García, B.; Gibbens-Bandala, B.; García-Pérez, F.; Ramírez-Nava, G.; Santos-Cuevas, C.; Ferro-Flores, G. [99mTc]Tc-iFAP Radioligand for SPECT/CT Imaging of the Tumor Microenvironment: Kinetics, Radiation Dosimetry, and Imaging in Patients. Pharmaceuticals 2022, 15, 590. [Google Scholar] [CrossRef]
- Röhrich, M.; Loktev, A.; Wefers, A.K.; Altmann, A.; Paech, D.; Adeberg, S.; Windisch, P.; Hielscher, T.; Flechsig, P.; Floca, R.; et al. IDH-wildtype glioblastomas and grade III/IV IDH-mutant gliomas show elevated tracer uptake in fibroblast activation protein-specific PET/CT. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 2569–2580. [Google Scholar] [CrossRef]
- Feng, T.; Fang, F.; Zhang, C.; Li, T.; He, J.; Shen, Y.; Yu, H.; Liu, X. Fluid Shear Stress-Induced Exosomes from Liver Cancer Cells Promote Activation of Cancer-Associated Fibroblasts via IGF2-PI3K Axis. Front. Biosci. Landmark 2022, 27, 104. [Google Scholar] [CrossRef] [PubMed]
- The Human Protein Atlas. Available online: https://www.proteinatlas.org/ENSG00000078098-FAP/pathology (accessed on 6 May 2022).
- Kratochwil, C.; Flechsig, P.; Lindner, T.; Abderrahim, L.; Altmann, A.; Mier, W.; Adeberg, S.; Rathke, H.; Röhrich, M.; Winter, H.; et al. 68Ga-FAPI PET/CT: Tracer Uptake in 28 Different Kinds of Cancer. J. Nucl. Med. 2019, 60, 801–805. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jo, H.; Lee, J.; Jeon, J.; Kim, S.Y.; Chung, J.I.; Ko, H.Y.; Lee, M.; Yun, M. The critical role of glucose deprivation in epithelial-mesenchymal transition in hepatocellular carcinoma under hypoxia. Sci. Rep. 2020, 10, 1538. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ye, X.; Brabletz, T.; Kang, Y.; Longmore, G.D.; Nieto, M.A.; Stanger, B.Z.; Yang, J.; Weinberg, R.A. Upholding a role for EMT in breast cancer metastasis. Nature 2017, 547, E1–E3. [Google Scholar] [CrossRef]
- Arora, L.; Pal, D. Remodeling of Stromal Cells and Immune Landscape in Microenvironment During Tumor Progression. Front. Oncol. 2021, 11, 596798. [Google Scholar] [CrossRef]
- Costa, A.; Kieffer, Y.; Scholer-Dahirel, A.; Pelon, F.; Bourachot, B.; Cardon, M.; Sirven, P.; Magagna, I.; Fuhrmann, L.; Bernard, C.; et al. Fibroblast Heterogeneity and Immunosuppressive Environment in Human Breast Cancer. Cancer Cell 2018, 33, 463–479.e10. [Google Scholar] [CrossRef] [Green Version]
- Banerjee, S.; Lo, W.C.; Majumder, P.; Roy, D.; Ghorai, M.; Shaikh, N.K.; Kant, N.; Shekhawat, M.S.; Gadekar, V.S.; Ghosh, S.; et al. Multiple roles for basement membrane proteins in cancer progression and EMT. Eur. J. Cell Biol. 2022, 101, 151220. [Google Scholar] [CrossRef]
- Javadi, S.; Zhiani, M.; Mousavi, M.A.; Fathi, M. Crosstalk between Epidermal Growth Factor Receptors (EGFR) and integrins in resistance to EGFR tyrosine kinase inhibitors (TKIs) in solid tumors. Eur. J. Cell Biol. 2020, 99, 151083. [Google Scholar] [CrossRef]
- Wang, K.; Ma, Q.; Ren, Y.; He, J.; Zhang, Y.; Zhang, Y.; Chen, W. Geldanamycin destabilizes HER2 tyrosine kinase and suppresses Wnt/beta-catenin signaling in HER2 overexpressing human breast cancer cells. Oncol. Rep. 2007, 17, 89–96. [Google Scholar]
- Weinberg, F.; Peckys, D.B.; de Jonge, N. EGFR Expression in HER2-Driven Breast Cancer Cells. Int. J. Mol. Sci. 2020, 21, 9008. [Google Scholar] [CrossRef]
- Luque, M.; Sanz-Álvarez, M.; Santamaría, A.; Zazo, S.; Cristóbal, I.; de la Fuente, L.; Mínguez, P.; Eroles, P.; Rovira, A.; Albanell, J.; et al. Targeted Therapy Modulates the Secretome of Cancer-Associated Fibroblasts to Induce Resistance in HER2-Positive Breast Cancer. Int. J. Mol. Sci. 2021, 22, 13297. [Google Scholar] [CrossRef] [PubMed]
- Pelon, F.; Bourachot, B.; Kieffer, Y.; Magagna, I.; Mermet-Meillon, F.; Bonnet, I.; Costa, A.; Givel, A.M.; Attieh, Y.; Barbazan, J.; et al. Cancer-associated fibroblast heterogeneity in axillary lymph nodes drives metastases in breast cancer through complementary mechanisms. Nat. Commun. 2020, 11, 404. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kömek, H.; Can, C.; Güzel, Y.; Oruç, Z.; Gündoğan, C.; Yildirim, Ö.A.; Kaplan, İ.; Erdur, E.; Yıldırım, M.S.; Çakabay, B. 68Ga-FAPI-04 PET/CT, a new step in breast cancer imaging: A comparative pilot study with the 18F-FDG PET/CT. Ann. Nucl. Med. 2021, 35, 744–752. [Google Scholar] [CrossRef]
- Backhaus, P.; Burg, M.C.; Roll, W.; Büther, F.; Breyholz, H.J.; Weigel, S.; Heindel, W.; Pixberg, M.; Barth, P.; Tio, J.; et al. Simultaneous FAPI PET/MRI Targeting the Fibroblast-Activation Protein for Breast Cancer. Radiology 2022, 302, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.Y.; Wei, W.F.; Wu, H.Z.; Fan, L.S.; Wang, W. Cancer-Associated Fibroblast Heterogeneity: A Factor That Cannot Be Ignored in Immune Microenvironment Remodeling. Front. Immunol. 2021, 12, 671595. [Google Scholar] [CrossRef]
- Mhaidly, R.; Mechta-Grigoriou, F. Fibroblast heterogeneity in tumor micro-environment: Role in immunosuppression and new therapies. Semin. Immunol. 2020, 48, 101417. [Google Scholar] [CrossRef]
- Kieffer, Y.; Hocine, H.R.; Gentric, G.; Pelon, F.; Bernard, C.; Bourachot, B.; Lameiras, S.; Albergante, L.; Bonneau, C.; Guyard, A.; et al. Single-Cell Analysis Reveals Fibroblast Clusters Linked to Immunotherapy Resistance in Cancer. Cancer Discov. 2020, 10, 1330–1351. [Google Scholar] [CrossRef]
- Zhao, L.; Pang, Y.; Luo, Z.; Fu, K.; Yang, T.; Zhao, L.; Sun, L.; Wu, H.; Lin, Q.; Chen, H. Role of [68Ga]Ga-DOTA-FAPI-04 PET/CT in the evaluation of peritoneal carcinomatosis and comparison with [18F]-FDG PET/CT. Eur. J. Nucl. Med. Mol. Imaging. 2021, 48, 1944–1955. [Google Scholar] [CrossRef]








| Characteristics | Number |
|---|---|
| No. patient | 32 |
| Age (years) | 50.8 ± 16.7 |
| Gender (%) | |
| Female | 28 (88%) |
| Male | 4 (12%) |
| Diagnosis | Cases (%) |
| Breast cancer | 21 (66%) |
| Ductal carcinoma, Luminal A | 2 |
| Ductal carcinoma, Luminal B | 3 |
| Ductal carcinoma, Luminal B HER2+ | 5 |
| Ductal carcinoma, HER2+ | 2 |
| Ductal carcinoma, Triple negative | 9 |
| Lung cancer | 2 (6%) |
| NSCLC adenocarcinoma | |
| Cervical cancer | 3 (9%) |
| Squamous cell carcinoma | |
| Glioma | 5 (16%) |
| Astrocytoma NOS (WHO II) | 1 |
| Anaplastic astrocytoma NOS (WHO III) | 3 |
| Glioblastoma NOS (WHO IV) | 1 |
| Colorectal cancer | 1 (3%) |
| Adenocarcinoma | 1 |
| Adrenal cortical neuroendocrine tumor | 1 (3%) |
| Poorly differentiated, Ki67 30% | 1 |
| Clinical setting (%) | |
| Initial staging | 27 (84%) |
| Restaging | 5 (15%) |
| Diagnosis | Status Brain SPECT | T/Bc |
|---|---|---|
| Astrocytoma NOS (WHO II) | Negative | NA |
| Anaplastic astrocytoma NOS (WHO III) (n = 2) | Positive | 6.3 and 7.8 |
| Anaplastic astrocytoma NOS restaging (WHO III) | Positive | 15.4 |
| Glioblastoma NOS (WHO IV) | Positive | 13.9 |
| Primary Tumor | Lymph Node Metastases | Distant Metastases | Total | |
|---|---|---|---|---|
| All lesions (N) | 26 | 61 | 24 | 111 |
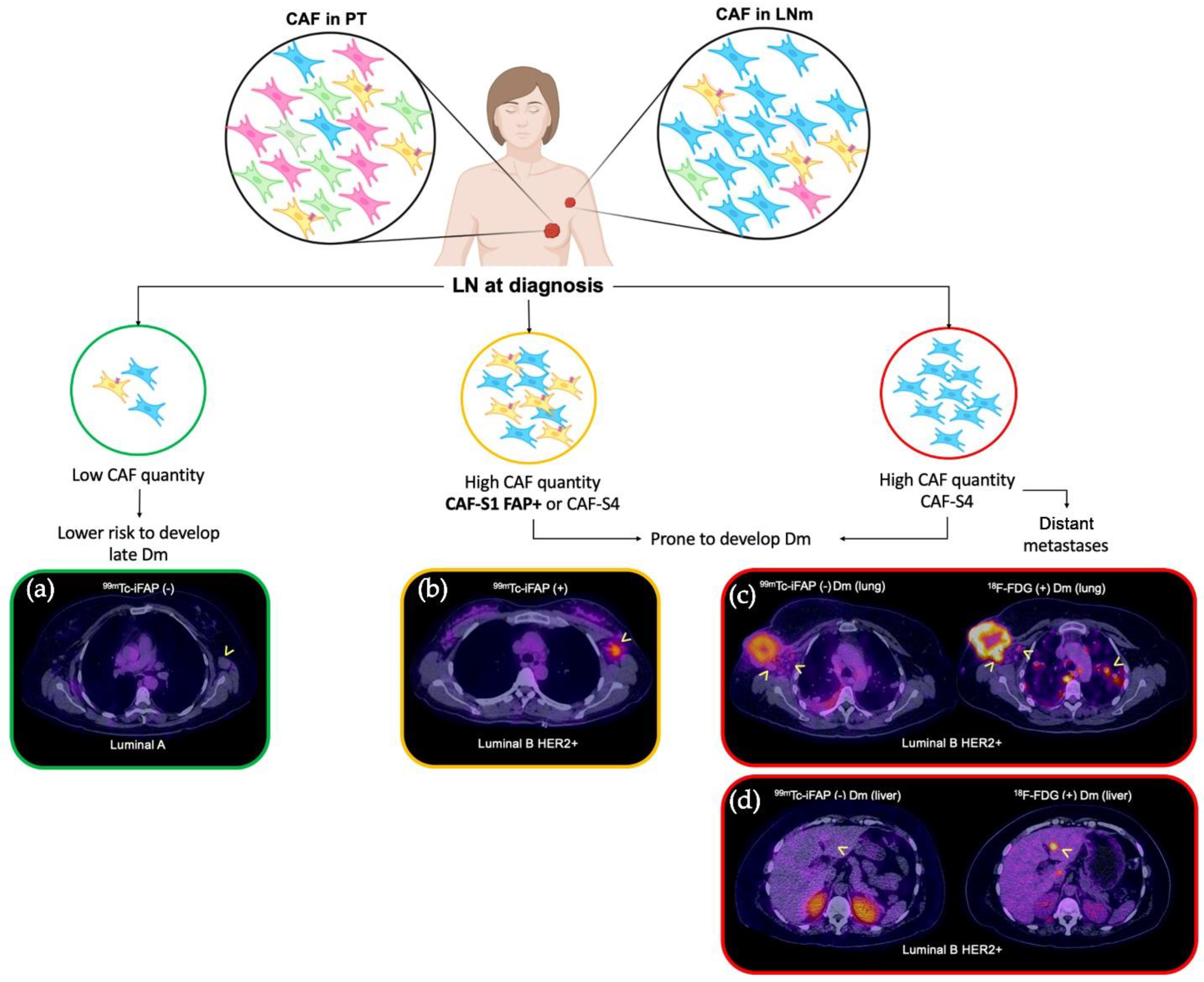
| [99mTc]Tc-iFAP | 26 (100%) | 31 (51%) | 4 (17%) | 61 (55%) |
| [18F]FDG | 26 (100%) | 61 (100%) | 21 (88%) | 108 (97%) |
| Diagnosis | Lymph node metastases | Distant metastases | ||
| [99mTc]Tc-iFAP | n = 31 (51%) | n = 4 (17%) | ||
| Lung cancer NSCLC | 3 | 0 | ||
| Triple-negative BC | 10 | 0 | ||
| Luminal A | 0 | 0 | ||
| Luminal B HER2+ BC | 7 | 2 | ||
| Luminal B BC | 4 | 0 | ||
| HER2+ BC | 5 | 0 | ||
| Cervical cancer | 2 | 0 | ||
| Colorectal cancer | 0 | 3 | ||
| Adrenal cortical NT | 0 | 1 | ||
| [18F]FDG | n = 61 (100%) | n = 21 (88%) | ||
| Lung cancer NSCLC | 3 | 1 | ||
| Triple-negative BC | 25 | 1 | ||
| Luminal A BC | 5 | 0 | ||
| Luminal B HER2+ BC | 8 | 16 | ||
| Luminal B BC | 10 | 1 | ||
| HER2+ BC | 7 | 0 | ||
| Cervical cancer | 2 | 0 | ||
| Colorectal cancer | 0 | 0 | ||
| Adrenal cortical NT | 1 | 2 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vallejo-Armenta, P.; Ferro-Flores, G.; Santos-Cuevas, C.; García-Pérez, F.O.; Casanova-Triviño, P.; Sandoval-Bonilla, B.; Ocampo-García, B.; Azorín-Vega, E.; Luna-Gutiérrez, M. [99mTc]Tc-iFAP/SPECT Tumor Stroma Imaging: Acquisition and Analysis of Clinical Images in Six Different Cancer Entities. Pharmaceuticals 2022, 15, 729. https://doi.org/10.3390/ph15060729
Vallejo-Armenta P, Ferro-Flores G, Santos-Cuevas C, García-Pérez FO, Casanova-Triviño P, Sandoval-Bonilla B, Ocampo-García B, Azorín-Vega E, Luna-Gutiérrez M. [99mTc]Tc-iFAP/SPECT Tumor Stroma Imaging: Acquisition and Analysis of Clinical Images in Six Different Cancer Entities. Pharmaceuticals. 2022; 15(6):729. https://doi.org/10.3390/ph15060729
Chicago/Turabian StyleVallejo-Armenta, Paola, Guillermina Ferro-Flores, Clara Santos-Cuevas, Francisco Osvaldo García-Pérez, Pamela Casanova-Triviño, Bayron Sandoval-Bonilla, Blanca Ocampo-García, Erika Azorín-Vega, and Myrna Luna-Gutiérrez. 2022. "[99mTc]Tc-iFAP/SPECT Tumor Stroma Imaging: Acquisition and Analysis of Clinical Images in Six Different Cancer Entities" Pharmaceuticals 15, no. 6: 729. https://doi.org/10.3390/ph15060729
APA StyleVallejo-Armenta, P., Ferro-Flores, G., Santos-Cuevas, C., García-Pérez, F. O., Casanova-Triviño, P., Sandoval-Bonilla, B., Ocampo-García, B., Azorín-Vega, E., & Luna-Gutiérrez, M. (2022). [99mTc]Tc-iFAP/SPECT Tumor Stroma Imaging: Acquisition and Analysis of Clinical Images in Six Different Cancer Entities. Pharmaceuticals, 15(6), 729. https://doi.org/10.3390/ph15060729