Improved Motion Artifact Correction in fNIRS Data by Combining Wavelet and Correlation-Based Signal Improvement
Abstract
1. Introduction
2. Materials and Methods
2.1. Motion Artifact Correction Methods
2.2. Experimental Procedure and fNIRS Data Recording
2.2.1. Participants
2.2.2. Experimental Setup and Procedure
2.2.3. fNIRS and IMU Data Recording and Processing
2.2.4. Data Processing and Movement Correction
3. Quality Metrics for Comparison among MA Correction Algorithms
- (1)
- Pearson’s correlation coefficient R was calculated between the averaged HRF of the reference signal (NHM) and of the movement-contaminated signals (SHM and LHM). Pearson correlation measures the similarity of the shapes of two signals and is scaled between −1 and 1.
- (2)
- Rooted Mean Square Error (RMSE) measures the unscaled average deviation between two signal time series. It was calculated with the following equation:Here, N is the number of samples in an epoch and i is the sample count.
- (3)
- Mean Absolute Percentage Error (MAPE) measures the deviation in relation to the momentary strength of the reference signal. It was obtained with the formula:
- (4)
- The area under the curve difference (ΔAUC) is a global measure that compares the overall deviation from the baseline of two curves. It was obtained with the formula:
4. Movement Analysis
5. Results
5.1. Movement Analysis
5.2. fNIRS Movement Artifacts
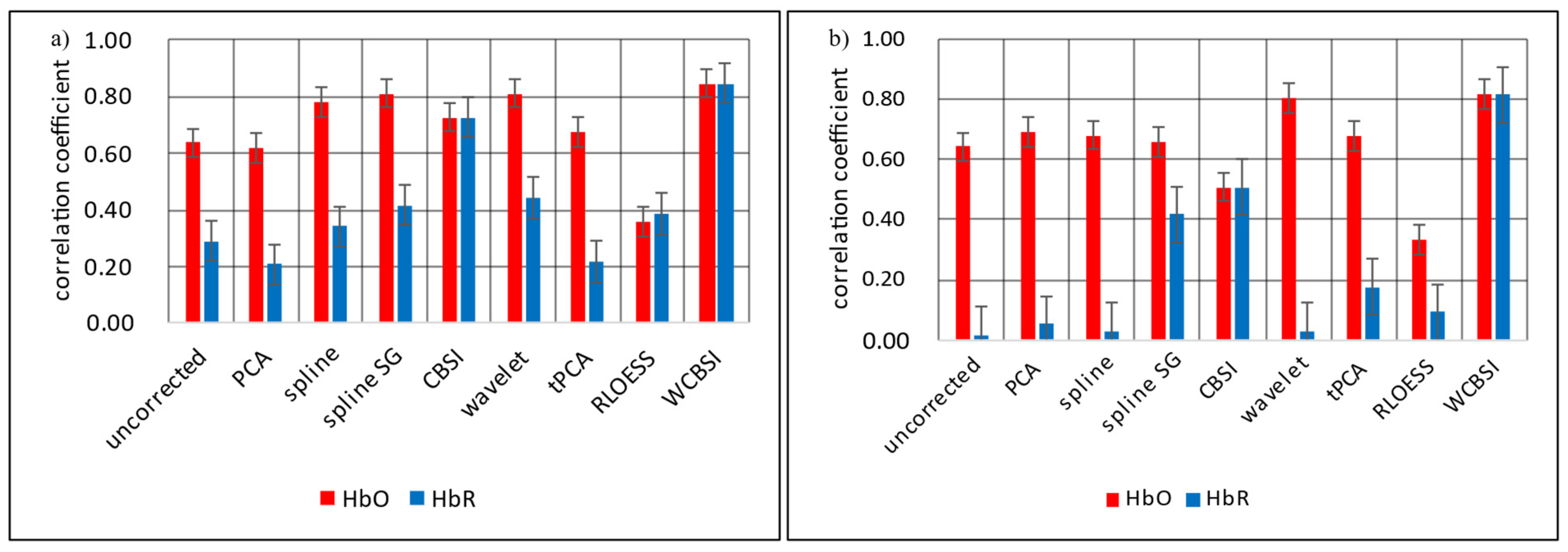
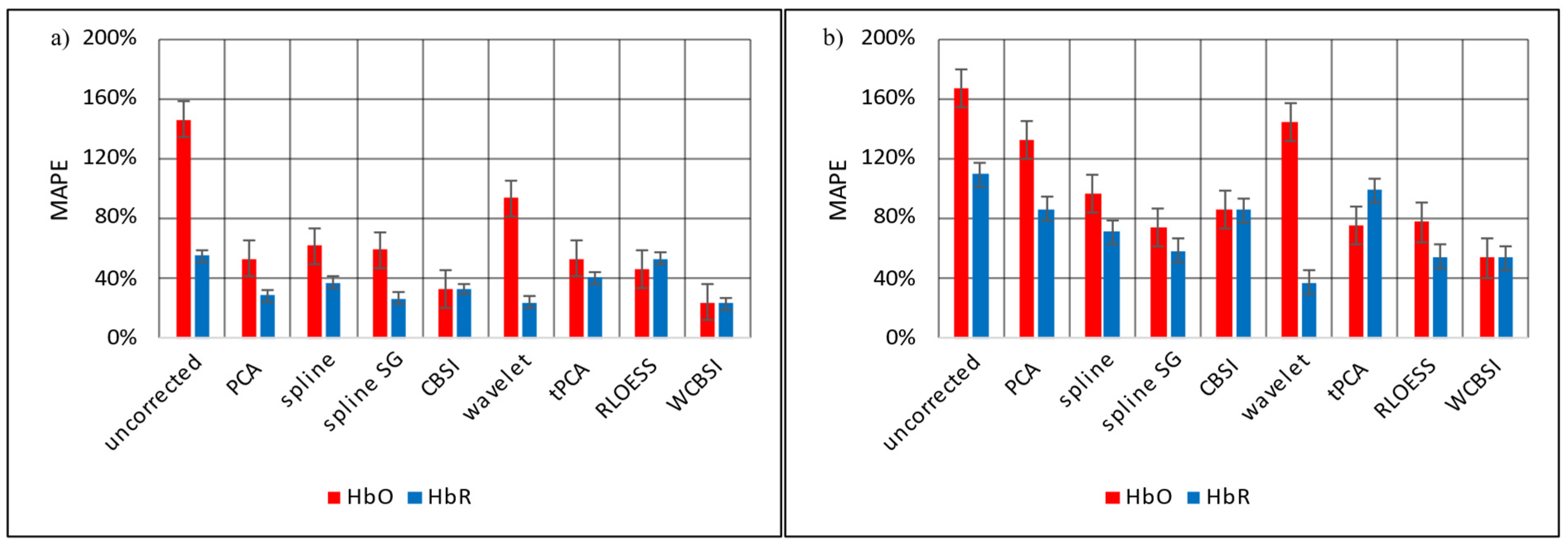
5.3. Comparison of Movement Correction Methods
6. Discussion
7. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Villringer, A.; Chance, B. Non-Invasive Optical Spectroscopy and Imaging of Human Brain Function. Trends Neurosci. 1997, 20, 435–442. [Google Scholar] [CrossRef] [PubMed]
- Jöbsis, F.F. Noninvasive, Infrared Monitoring of Cerebral and Myocardial Oxygen Sufficiency and Circulatory Parameters. Science 1977, 198, 1264–1266. [Google Scholar] [CrossRef] [PubMed]
- Huppert, T.; Angela Franceschini, M.; Boas, D. Noninvasive Imaging of Cerebral Activation with Diffuse Optical Tomography. Vivo Opt. Imaging Brain Funct. 2009, 393–433. [Google Scholar] [CrossRef]
- Hu, X.-S.; Arredondo, M.M.; Gomba, M.; Confer, N.; DaSilva, A.F.; Johnson, T.D.; Shalinsky, M.; Kovelman, I. Comparison of Motion Correction Techniques Applied to Functional Near-Infrared Spectroscopy Data from Children. J. Biomed. Opt. 2015, 20, 126003. [Google Scholar] [CrossRef]
- Roche-Labarbe, N.; Fenoglio, A.; Radhakrishnan, H.; Kocienski-Filip, M.; Carp, S.A.; Dubb, J.; Boas, D.A.; Grant, P.E.; Franceschini, M.A. Somatosensory Evoked Changes in Cerebral Oxygen Consumption Measured Non-Invasively in Premature Neonates. Neuroimage 2014, 85, 279–286. [Google Scholar] [CrossRef]
- Xu, S.Y.; Lu, F.M.; Wang, M.Y.; Hu, Z.S.; Zhang, J.; Chen, Z.Y.; Armada-da-Silva, P.A.S.; Yuan, Z. Altered Functional Connectivity in the Motor and Prefrontal Cortex for Children with Down’s Syndrome: An FNIRS Study. Front. Hum. Neurosci. 2020, 14, 6. [Google Scholar] [CrossRef] [PubMed]
- Medvedev, A.V. Does the Resting State Connectivity Have Hemispheric Asymmetry? A near-Infrared Spectroscopy Study. Neuroimage 2014, 85, 400–407. [Google Scholar] [CrossRef]
- Lin, X.A.; Wang, C.; Zhou, J.; Sai, L.; Fu, G. Neural Correlates of Spontaneous Deception in a Non-Competitive Interpersonal Scenario: A Functional near-Infrared Spectroscopy (FNIRS) Study. Brain Cogn. 2021, 150, 105704. [Google Scholar] [CrossRef]
- Wei, Y.Y.; Chen, Q.; Curtin, A.; Tu, L.; Tang, X.; Tang, Y.Y.; Xu, L.H.; Qian, Z.Y.; Zhou, J.; Zhu, C.Z.; et al. Functional Near-Infrared Spectroscopy (FNIRS) as a Tool to Assist the Diagnosis of Major Psychiatric Disorders in a Chinese Population. Eur. Arch. Psychiatry Clin. Neurosci. 2021, 271, 745–757. [Google Scholar] [CrossRef]
- Mihara, M.; Fujimoto, H.; Hattori, N.; Otomune, H.; Kajiyama, Y.; Konaka, K.; Watanabe, Y.; Hiramatsu, Y.; Sunada, Y.; Miyai, I.; et al. Effect of Neurofeedback Facilitation on Poststroke Gait and Balance Recovery: A Randomized Controlled Trial. Neurology 2021, 96, e2587–e2598. [Google Scholar] [CrossRef]
- Rahman, M.A.; Siddik, A.B.; Ghosh, T.K.; Khanam, F.; Ahmad, M. A Narrative Review on Clinical Applications of FNIRS. J. Digit. Imaging 2020, 33, 1167–1184. [Google Scholar] [CrossRef] [PubMed]
- Unni, A.; Ihme, K.; Jipp, M.; Rieger, J.W. Assessing the Driver’s Current Level of Working Memory Load with High Density Functional near-Infrared Spectroscopy: A Realistic Driving Simulator Study. Front. Hum. Neurosci. 2017, 11, 167. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Sobczak, G.; Colette, M.M.; Litovsky, R.Y. Comparing FNIRS Signal Qualities between Approaches with and without Short Channels. PLoS ONE 2021, 15, e0244186. [Google Scholar] [CrossRef] [PubMed]
- Scholkmann, F.; Spichtig, S.; Muehlemann, T.; Wolf, M. How to Detect and Reduce Movement Artifacts in Near-Infrared Imaging Using Moving Standard Deviation and Spline Interpolation. Physiol. Meas. 2010, 31, 649–662. [Google Scholar] [CrossRef]
- Nozawa, T.; Kondo, T. A Comparison of Artifact Reduction Methods for Real-Time Analysis of FNIRS Data. In Lecture Notes in Computer Science (Including Subseries Lecture Notes in Artificial Intelligence and Lecture Notes in Bioinformatics); Salvendy, G., Smith, M.J., Eds.; Springer: Berlin/Heidelberg, Germany, 2009; Volume 5618 LNCS, pp. 413–422. ISBN 3642025587. [Google Scholar]
- Vanegas, M.; Dementyev, A.; Mireles, M.; Carp, S.; Fang, Q. A Modular, Fiberless, 3-d Aware, Flexible-Circuit-Based Wearable FNIRS System. In Proceedings of the Optics InfoBase Conference Papers, Beijing, China, 24–27 October 2020; Volume Part F176-, p. BM3C.3. [Google Scholar]
- Brigadoi, S.; Ceccherini, L.; Cutini, S.; Scarpa, F.; Scatturin, P.; Selb, J.; Gagnon, L.; Boas, D.A.; Cooper, R.J. Motion Artifacts in Functional Near-Infrared Spectroscopy: A Comparison of Motion Correction Techniques Applied to Real Cognitive Data. Neuroimage 2014, 85, 181–191. [Google Scholar] [CrossRef]
- Zhang, Y.; Brooks, D.H.; Franceschini, M.A.; Boas, D.A. Eigenvector-Based Spatial Filtering for Reduction of Physiological Interference in Diffuse Optical Imaging. J. Biomed. Opt. 2005, 10, 011014. [Google Scholar] [CrossRef]
- Yücel, M.A.; Selb, J.; Cooper, R.J.; Boas, D.A. Targeted Principle Component Analysis: A New Motion Artifact Correction Approach for near-Infrared Spectroscopy. J. Innov. Opt. Health Sci. 2014, 7, 1350066. [Google Scholar] [CrossRef]
- Cui, X.; Bray, S.; Reiss, A.L. Functional near Infrared Spectroscopy (NIRS) Signal Improvement Based on Negative Correlation between Oxygenated and Deoxygenated Hemoglobin Dynamics. Neuroimage 2010, 49, 3039–3046. [Google Scholar] [CrossRef]
- Obrig, H.; Steinbrink, J. Non-Invasive Optical Imaging of Stroke. Philos. Trans. R. Soc. Math. Phys. Eng. Sci. 2011, 369, 4470–4494. [Google Scholar] [CrossRef]
- Savitzky, A.; Golay, M.J.E. Smoothing and Differentiation of Data by Simplified Least Squares Procedures. Anal. Chem. 1964, 36, 1627–1639. [Google Scholar] [CrossRef]
- Cleveland, W.S. Robust Locally Weighted Regression and Smoothing Scatterplots. J. Am. Stat. Assoc. 1979, 74, 829–836. [Google Scholar] [CrossRef]
- Jahani, S.; Setarehdan, S.K.; Boas, D.A.; Yücel, M.A. Motion Artifact Detection and Correction in Functional Near-Infrared Spectroscopy: A New Hybrid Method Based on Spline Interpolation Method and Savitzky–Golay Filtering. Neurophotonics 2018, 5, 1. [Google Scholar] [CrossRef] [PubMed]
- Molavi, B.; Dumont, G.A. Wavelet-Based Motion Artifact Removal for Functional near-Infrared Spectroscopy. Physiol. Meas. 2012, 33, 259–270. [Google Scholar] [CrossRef] [PubMed]
- Yücel, M.A.; Lühmann, A.v.; Scholkmann, F.; Gervain, J.; Dan, I.; Ayaz, H.; Boas, D.; Cooper, R.J.; Culver, J.; Elwell, C.E.; et al. Best Practices for FNIRS Publications. Neurophotonics 2021, 8, 12101. [Google Scholar] [CrossRef] [PubMed]
- Jurcak, V.; Tsuzuki, D.; Dan, I. 10/20, 10/10, and 10/5 Systems Revisited: Their Validity as Relative Head-Surface-Based Positioning Systems. Neuroimage 2007, 34, 1600–1611. [Google Scholar] [CrossRef]
- Brainard, D.H. The Psychophysics Toolbox. Spat. Vis. 1997, 10, 433–436. [Google Scholar] [CrossRef] [PubMed]
- Huppert, T.J.; Diamond, S.G.; Franceschini, M.A.; Boas, D.A. HomER: A Review of Time-Series Analysis Methods for near-Infrared Spectroscopy of the Brain. Appl. Opt. 2009, 48, D280–D298. [Google Scholar] [CrossRef] [PubMed]
- Scholkmann, F.; Wolf, M. General Equation for the Differential Pathlength Factor of the Frontal Human Head Depending on Wavelength and Age. J. Biomed. Opt. 2013, 18, 105004. [Google Scholar] [CrossRef]
- Bonilauri, A.; Sangiuliano Intra, F.; Baselli, G.; Baglio, F. Assessment of Fnirs Signal Processing Pipelines: Towards Clinical Applications. Appl. Sci. 2021, 12, 316. [Google Scholar] [CrossRef]
- Turner, H.L.; van Etten, J.; Firth, D.; Kosmidis, I. Modelling Rankings in R: The PlackettLuce Package. Comput. Stat. 2020, 35, 1027–1057. [Google Scholar] [CrossRef]
- Di Lorenzo, R.; Pirazzoli, L.; Blasi, A.; Bulgarelli, C.; Hakuno, Y.; Minagawa, Y.; Brigadoi, S. Recommendations for Motion Correction of Infant FNIRS Data Applicable to Multiple Data Sets and Acquisition Systems. Neuroimage 2019, 200, 511–527. [Google Scholar] [CrossRef] [PubMed]
- Hossain, M.S.; Chowdhury, M.E.H.; Reaz, M.B.I.; Ali, S.H.M.; Bakar, A.A.A.; Kiranyaz, S.; Khandakar, A.; Alhatou, M.; Habib, R.; Hossain, M.M. Motion Artifacts Correction from Single-Channel EEG and FNIRS Signals Using Novel Wavelet Packet Decomposition in Combination with Canonical Correlation Analysis. Sensors 2022, 22, 3169. [Google Scholar] [CrossRef] [PubMed]
- Cooper, R.J.; Selb, J.; Gagnon, L.; Phillip, D.; Schytz, H.W.; Iversen, H.K.; Ashina, M.; Boas, D.A. A Systematic Comparison of Motion Artifact Correction Techniques for Functional Near-Infrared Spectroscopy. Front. Neurosci. 2012, 6, 147. [Google Scholar] [CrossRef] [PubMed]
- Fishburn, F.A.; Ludlum, R.S.; Vaidya, C.J.; Medvedev, A.V. Temporal Derivative Distribution Repair (TDDR): A Motion Correction Method for FNIRS. Neuroimage 2019, 184, 171–179. [Google Scholar] [CrossRef]
- Croce, P.; Zappasodi, F.; Merla, A.; Chiarelli, A.M. Exploiting Neurovascular Coupling: A Bayesian Sequential Monte Carlo Approach Applied to Simulated EEG FNIRS Data. J. Neural Eng. 2017, 14, 46029. [Google Scholar] [CrossRef] [PubMed]
- Lawoyin, S.A.; Fei, D.-Y.; Bai, O. A Novel Application of Inertial Measurement Units (IMUs) as Vehicular Technologies for Drowsy Driving Detection via Steering Wheel Movement. Open J. Saf. Sci. Technol. 2014, 4, 166–177. [Google Scholar] [CrossRef]
- Munafò, M.R.; Nosek, B.A.; Bishop, D.V.M.; Button, K.S.; Chambers, C.D.; Percie du Sert, N.; Simonsohn, U.; Wagenmakers, E.-J.; Ware, J.J.; Ioannidis, J.P.A. A Manifesto for Reproducible Science. Nat. Hum. Behav. 2017, 1, 21. [Google Scholar] [CrossRef]








| Name | Function | Parameters and Values |
|---|---|---|
| Channel rejection | hmrR_PruneChannels | dRange (1 × 10−4–1 × 107), SNRthresh = 1, Sdrange = (0.0–45.0) |
| Motion detection | HmrMotionArtifactByChannel | tMotion = 0.5 Sec, tMask = 1.0 Sec, SDEVThresh = 20, AMPthresh = (0.05–0.5) |
| PCA | hmrR_PCAFilter | nSV = (0.96 ± 0.02) |
| tPCA | hmrR_MotionCorrectPCArecurse | tMotion = 0.5 Sec, tMask = 1.0 Sec, SDEVThresh = 20, AMPthresh = (0.1–0.5), nSV = 0.97, maxlter = 5 |
| Spline | hmrR_MotionCorrectSpline | p = 0.99 |
| SplineSG | hmrR_MotionCorrectionSplineSG | p = 0.99, FrameSize_Sec = 10 |
| RLOEES | hmrR_MotionCorrectRLOEES | span = 0.02 |
| Wavelet | hmrR_MotionCorrectWavelet | iqr = 1.5 |
| CBSI | hmrR_MotionCorrectCBSI | On |
| Band-pass filter | hmrR_BandpassFilt | hpf = 0.01 Hz, lpf = 0.1 Hz |
| OD change | hmrR_OD2Conc | 1.0 1.0 1.0 |
| Average | hmrR_BlockAvg | −2.0 Sec 20.0 Sec |
| Method | Grand Mean Rank | Rank std |
|---|---|---|
| WCBSI | 1.25 | 0.77 |
| RLOESS | 4.00 | 2.48 |
| CBSI | 4.13 | 2.13 |
| Wavelet | 4.25 | 2.27 |
| SplineSG | 4.94 | 2.24 |
| Spline | 5.44 | 1.46 |
| tPCA | 5.63 | 1.59 |
| PCA | 6.31 | 1.78 |
| Uncorrected | 8.63 | 0.81 |
| Method | Rank Grand Mean | Rank std |
|---|---|---|
| WCBSI | 1.25 | 0.77 |
| Uncorrected | 8.63 | 0.81 |
| Spline | 5.44 | 1.46 |
| tPCA | 5.63 | 1.59 |
| PCA | 6.31 | 1.78 |
| CBSI | 4.13 | 2.13 |
| SplineSG | 4.94 | 2.24 |
| Wavelet | 4.25 | 2.27 |
| RLOESS | 4.00 | 2.48 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al-Omairi, H.R.; Fudickar, S.; Hein, A.; Rieger, J.W. Improved Motion Artifact Correction in fNIRS Data by Combining Wavelet and Correlation-Based Signal Improvement. Sensors 2023, 23, 3979. https://doi.org/10.3390/s23083979
Al-Omairi HR, Fudickar S, Hein A, Rieger JW. Improved Motion Artifact Correction in fNIRS Data by Combining Wavelet and Correlation-Based Signal Improvement. Sensors. 2023; 23(8):3979. https://doi.org/10.3390/s23083979
Chicago/Turabian StyleAl-Omairi, Hayder R., Sebastian Fudickar, Andreas Hein, and Jochem W. Rieger. 2023. "Improved Motion Artifact Correction in fNIRS Data by Combining Wavelet and Correlation-Based Signal Improvement" Sensors 23, no. 8: 3979. https://doi.org/10.3390/s23083979
APA StyleAl-Omairi, H. R., Fudickar, S., Hein, A., & Rieger, J. W. (2023). Improved Motion Artifact Correction in fNIRS Data by Combining Wavelet and Correlation-Based Signal Improvement. Sensors, 23(8), 3979. https://doi.org/10.3390/s23083979







