CMOS-Based Redox-Type Label-Free ATP Image Sensor for In Vitro Sensitive Imaging of Extracellular ATP
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials and Chemicals
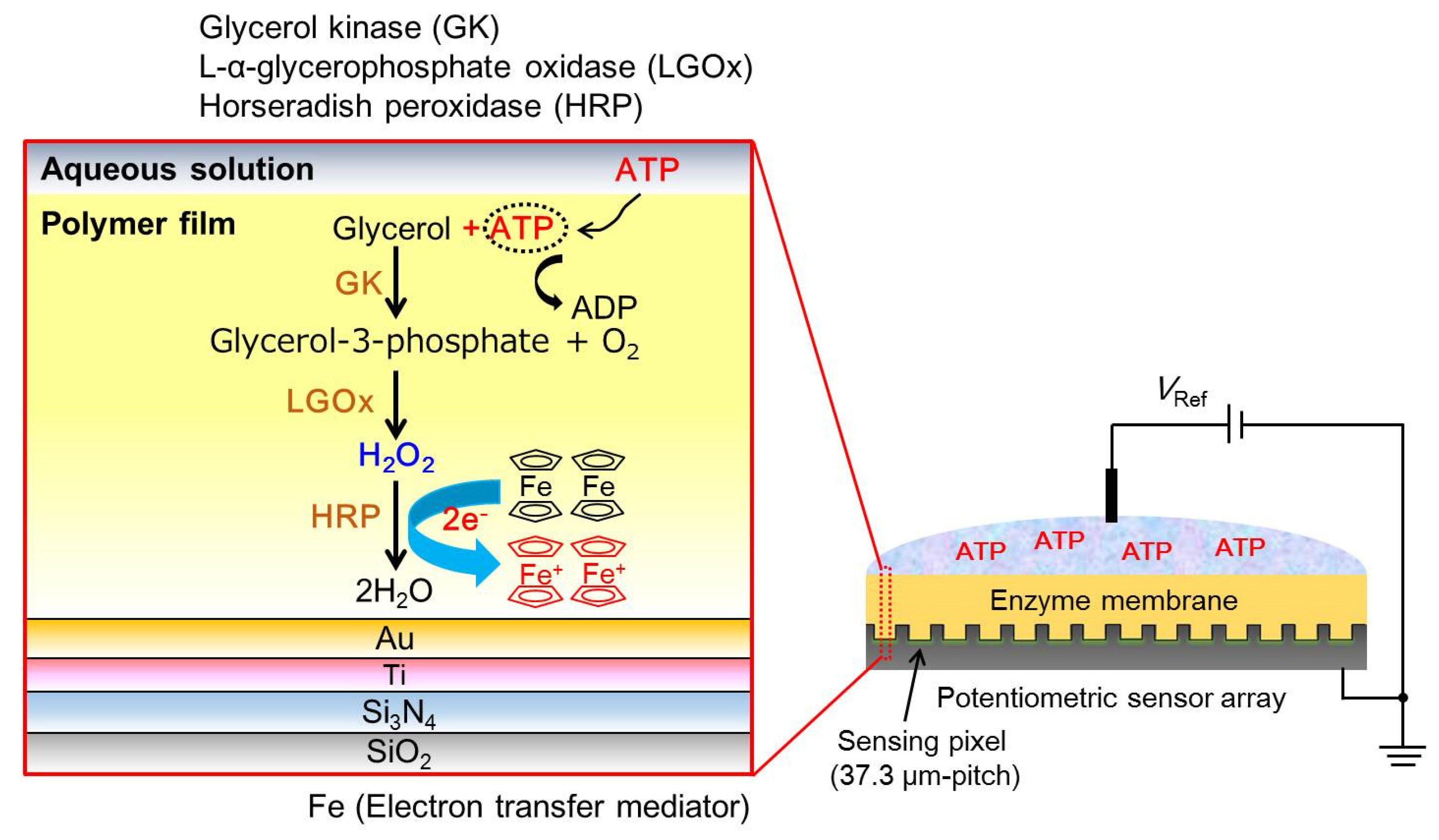
2.2. The Measurement Principle of the Redox-Type Label-Free ATP Image Sensor
2.3. Sensor Fabrication
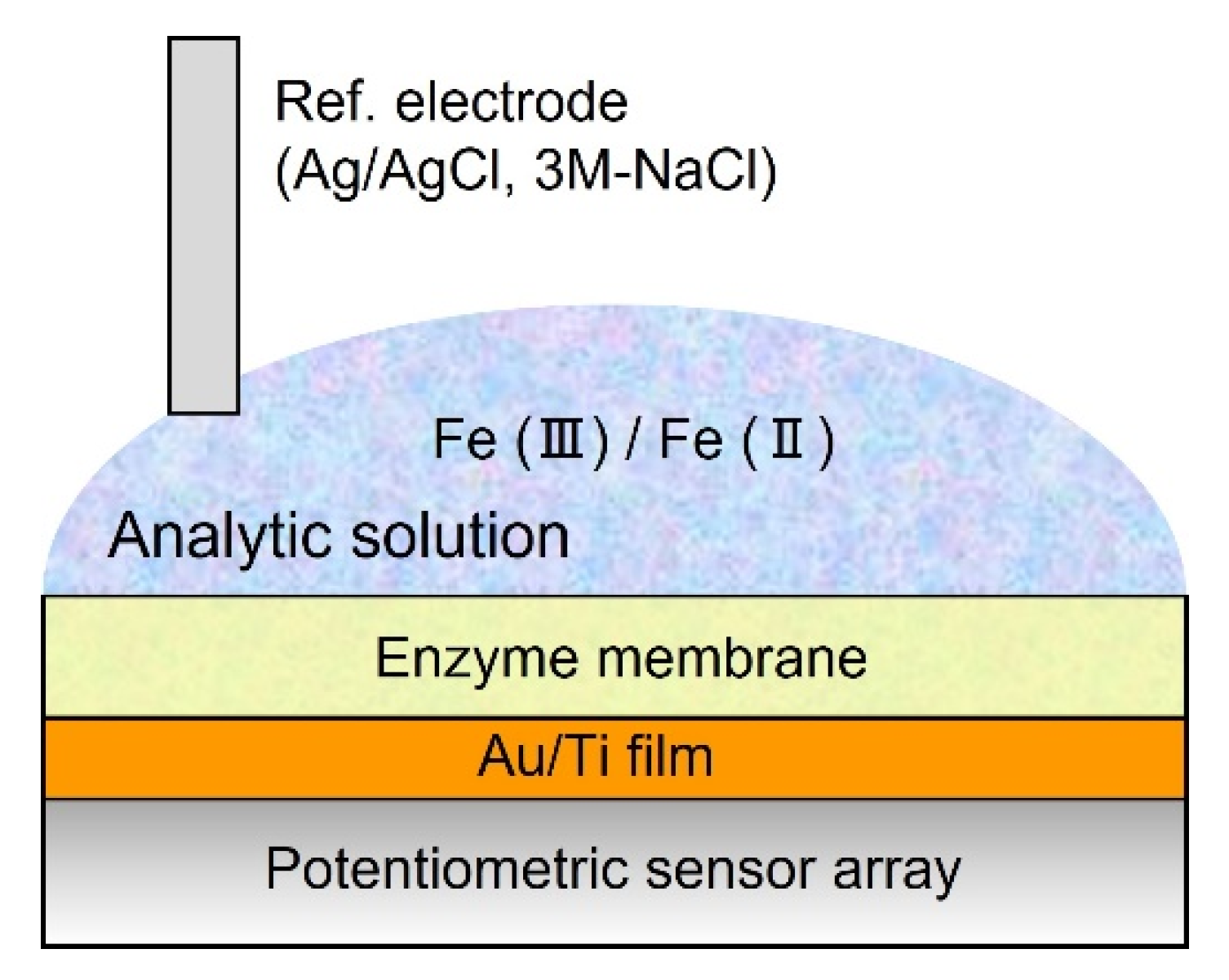
2.4. Measurement of Redox Response
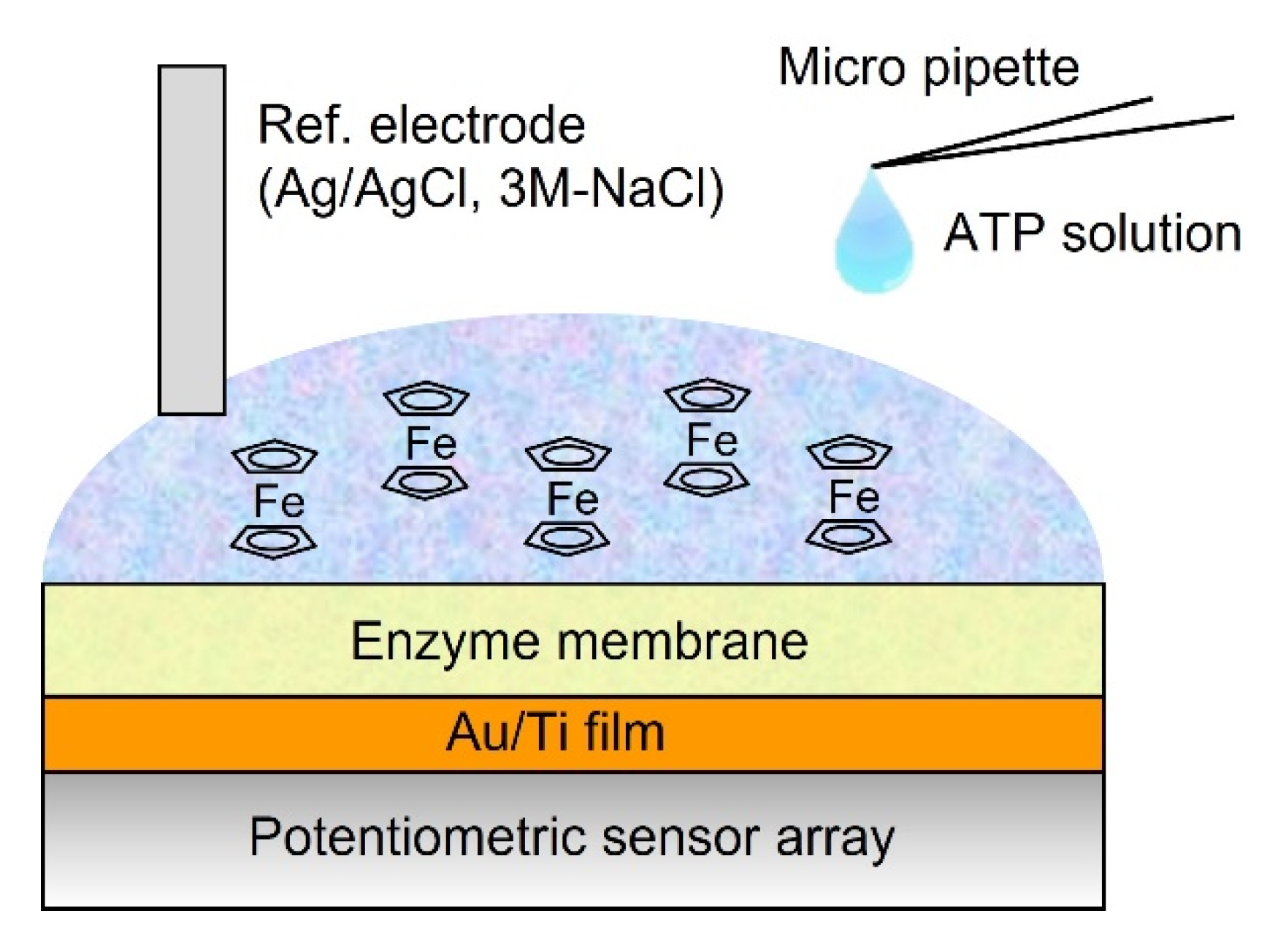
2.5. ATP Measurement
3. Results and Discussion
3.1. Fabricated Sensor
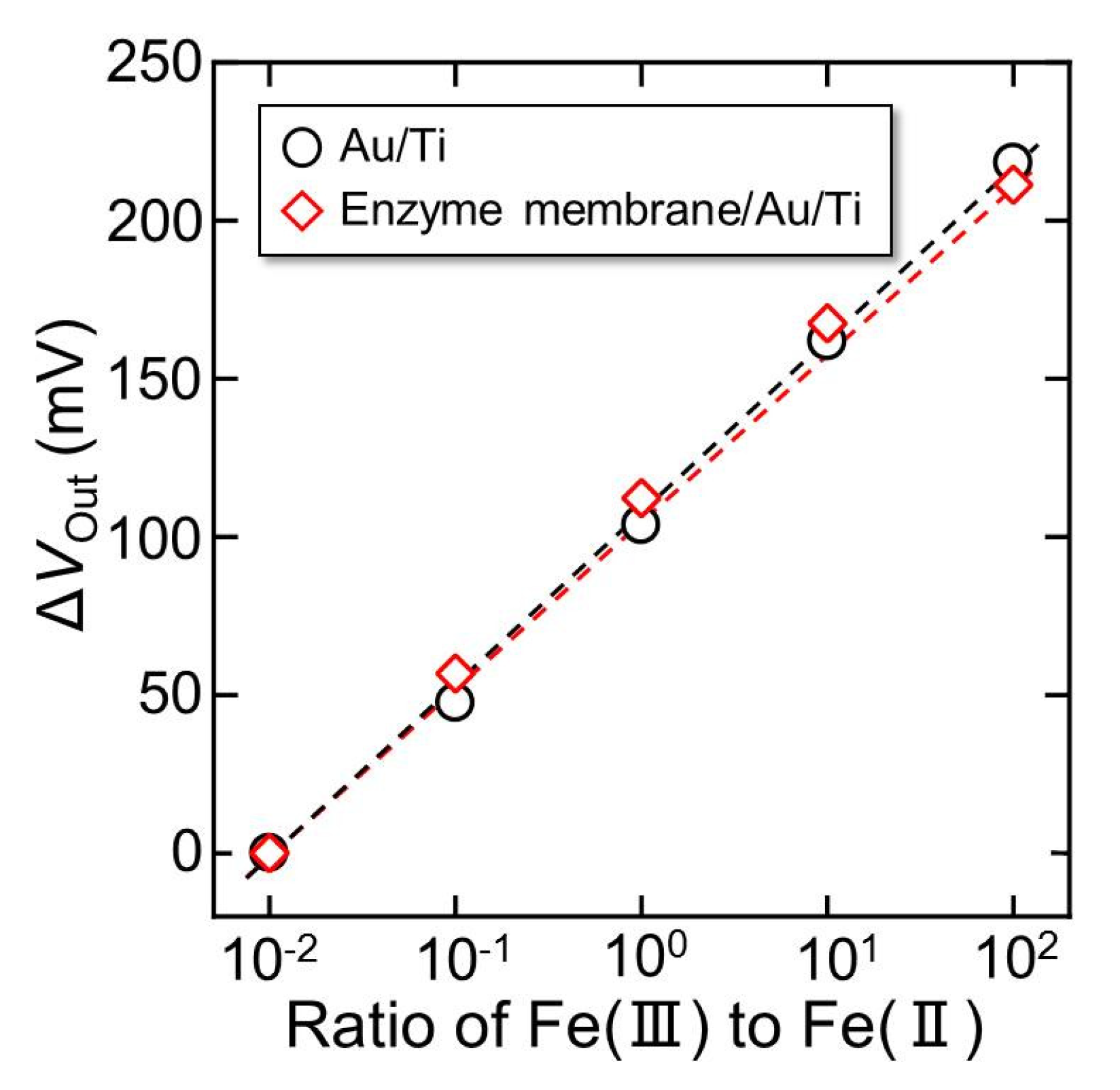
3.2. Redox Response
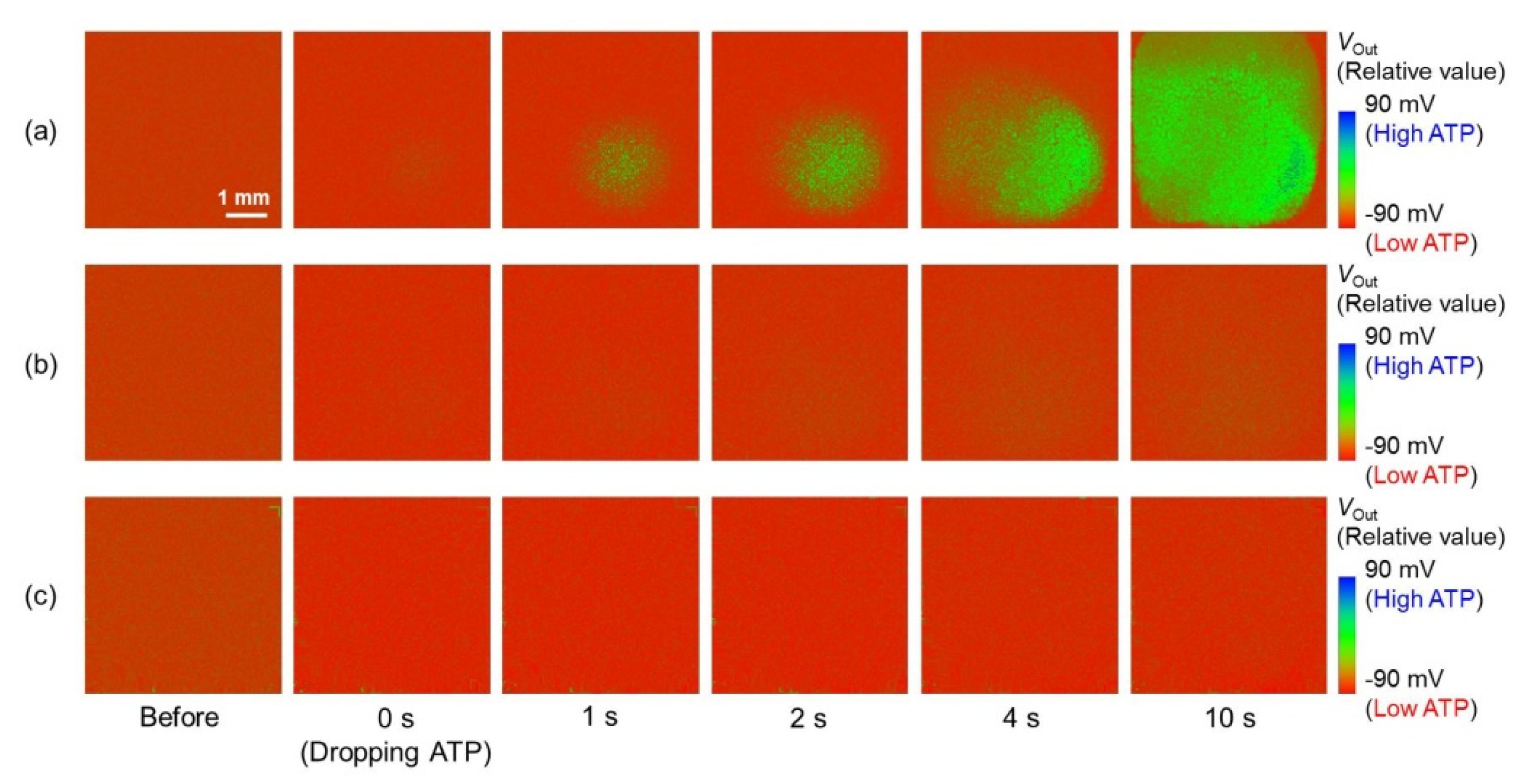
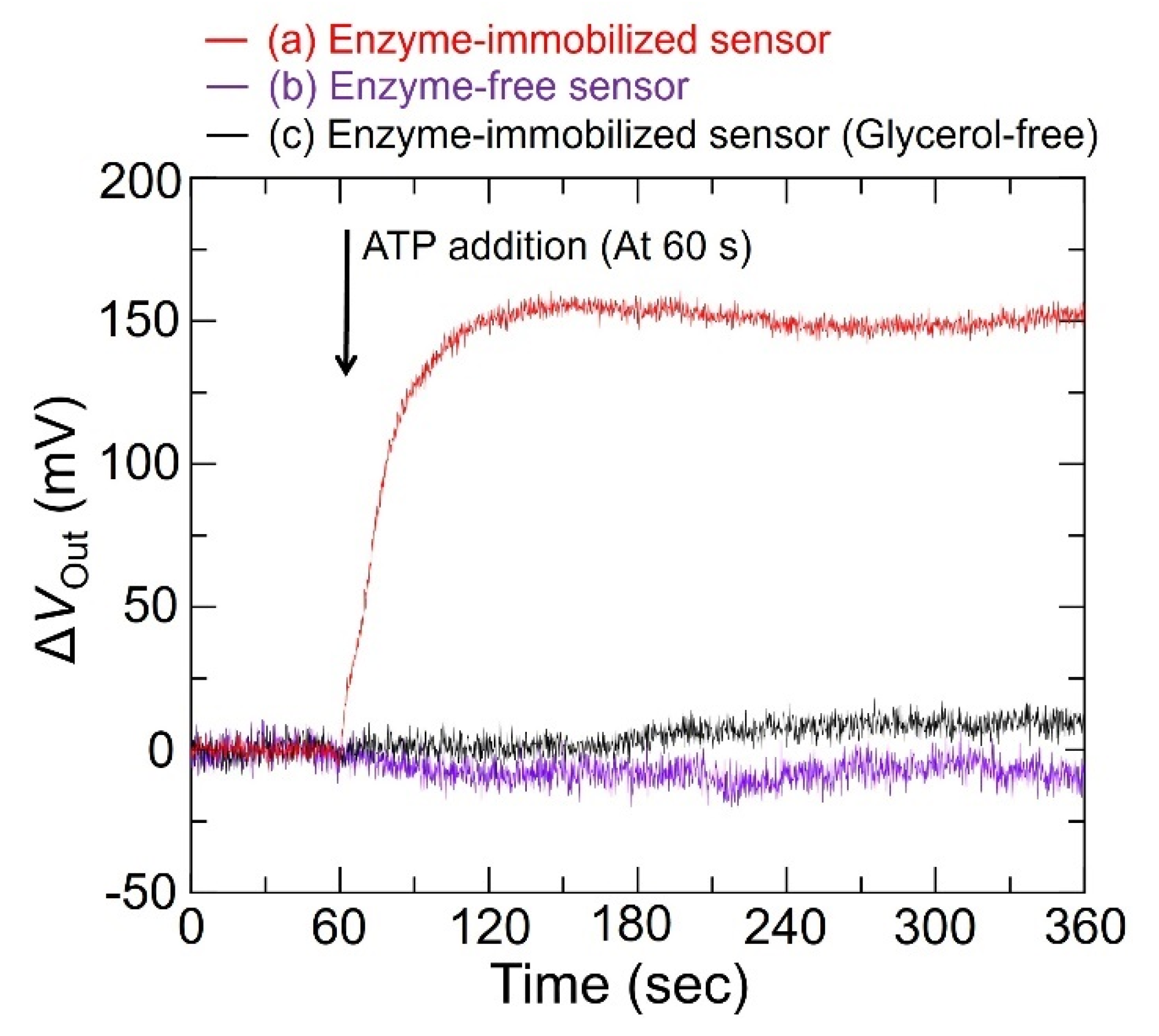
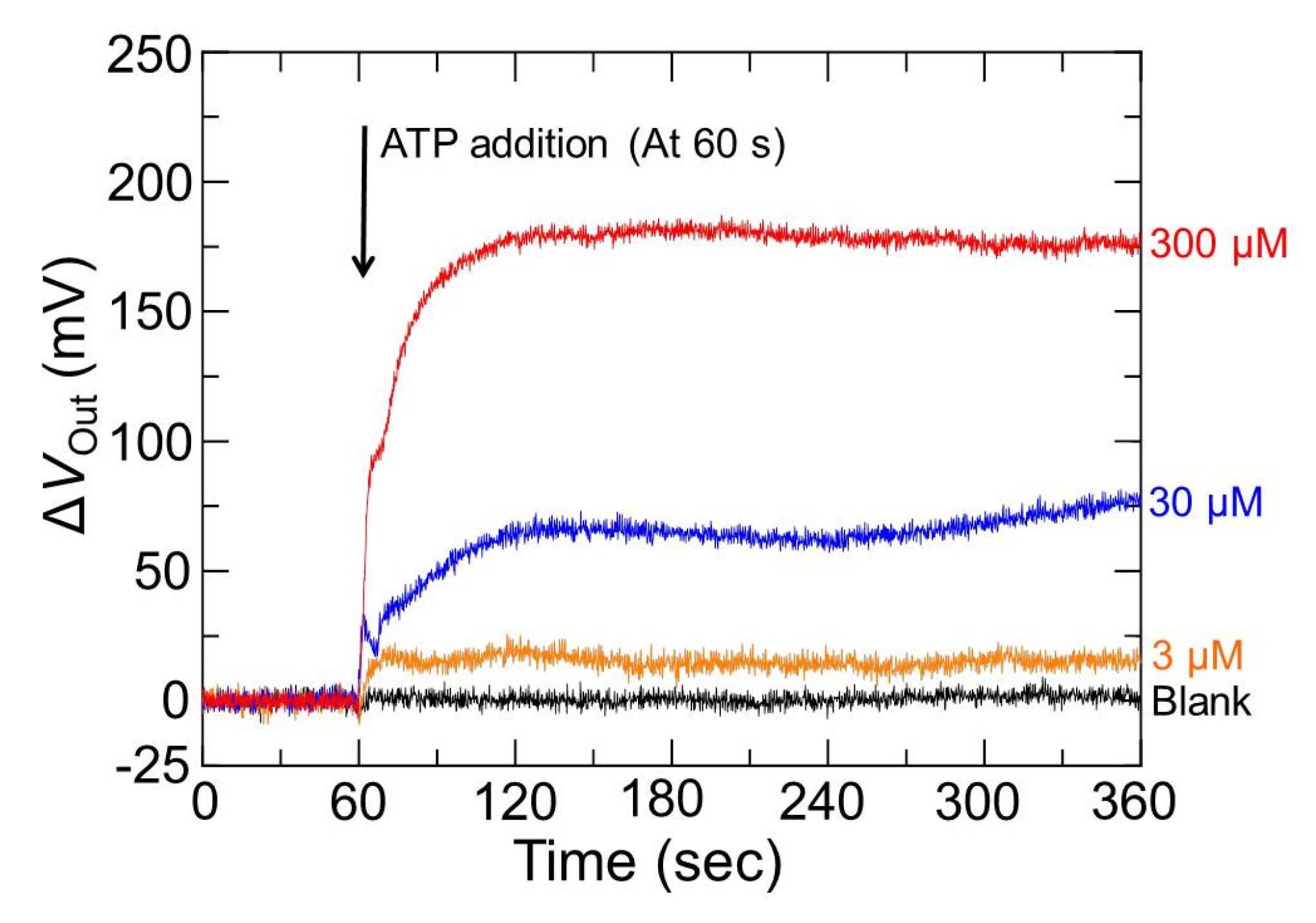
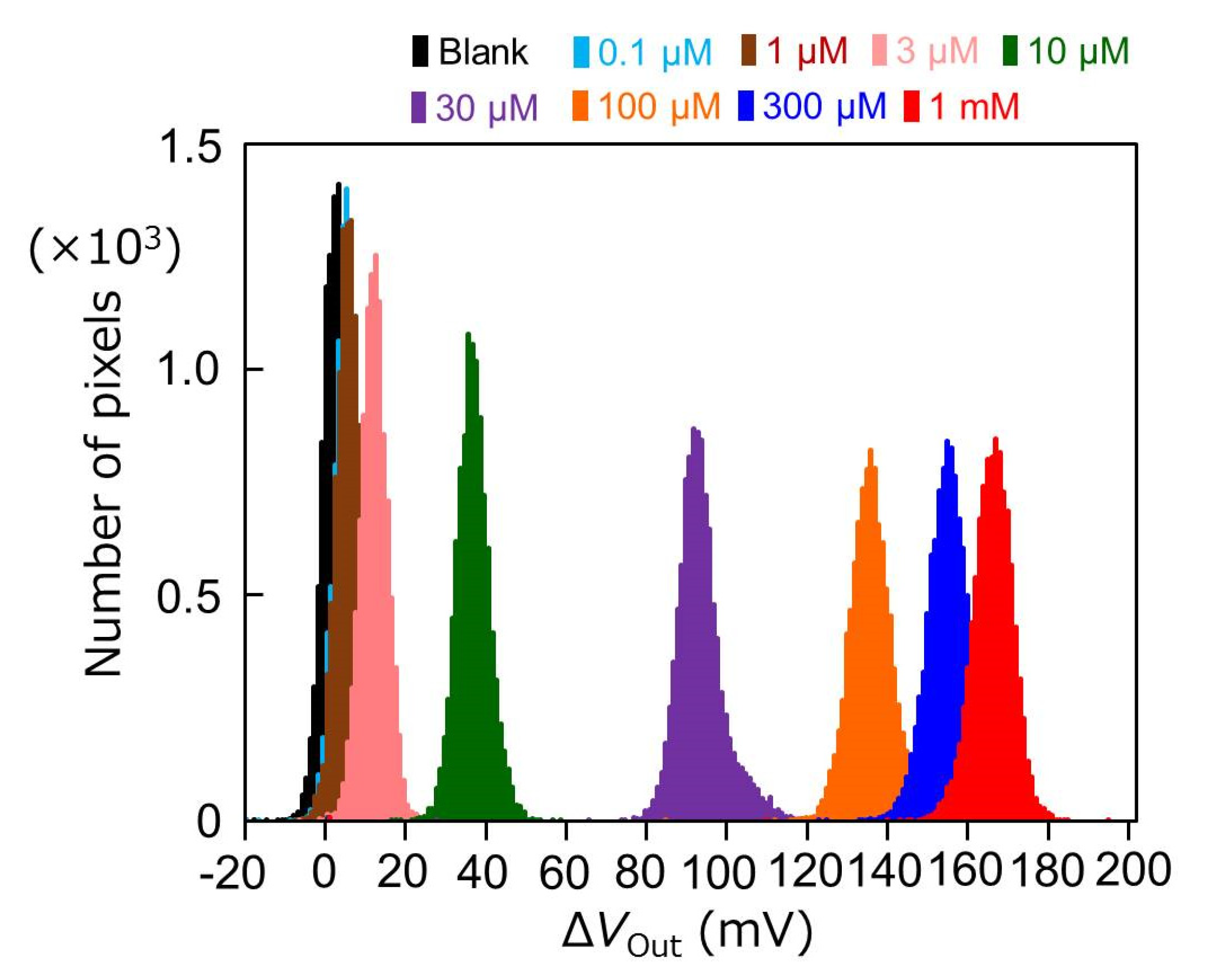
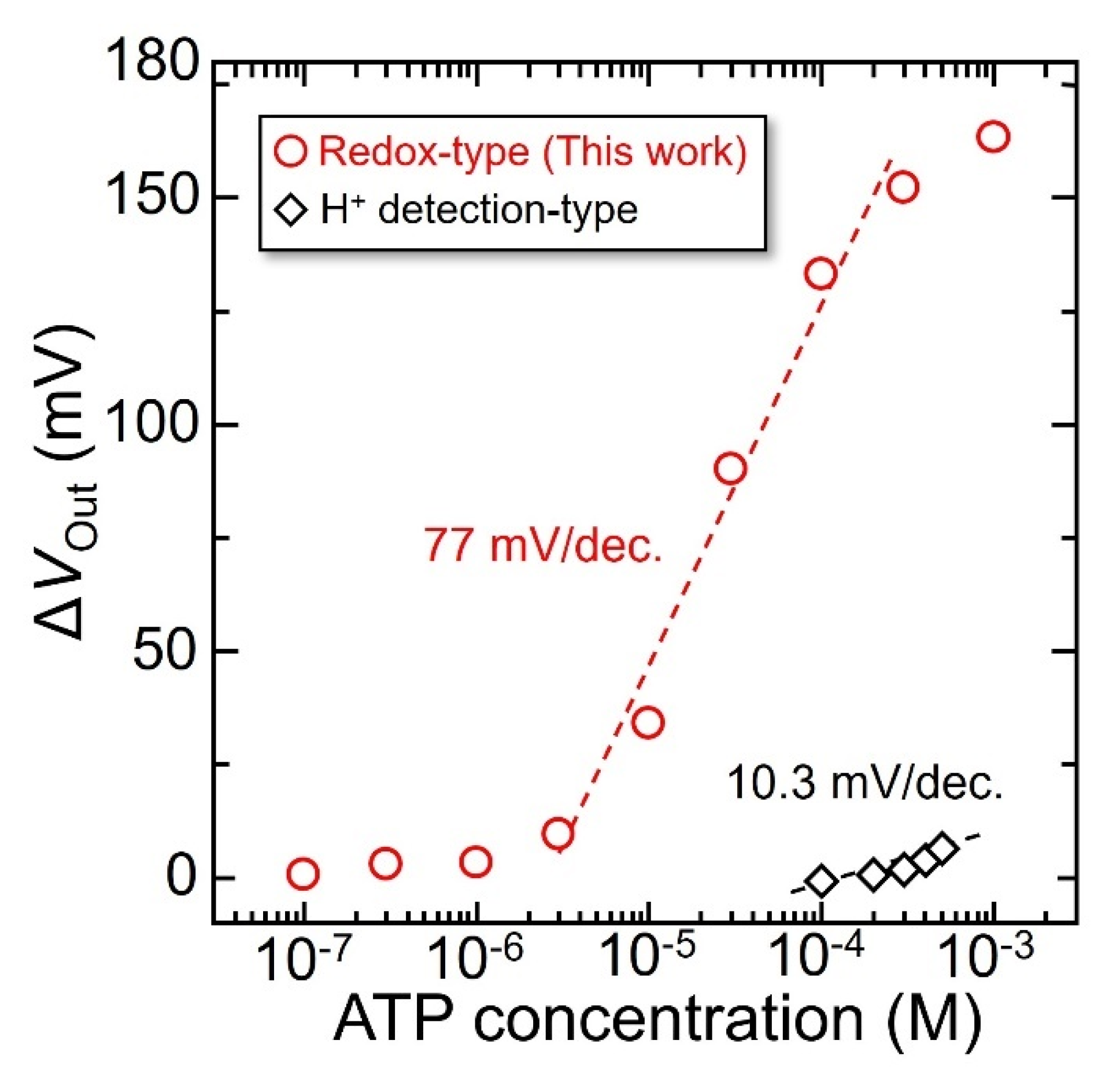
3.3. Output Characteristics of the Redox-Type ATP Image Sensor
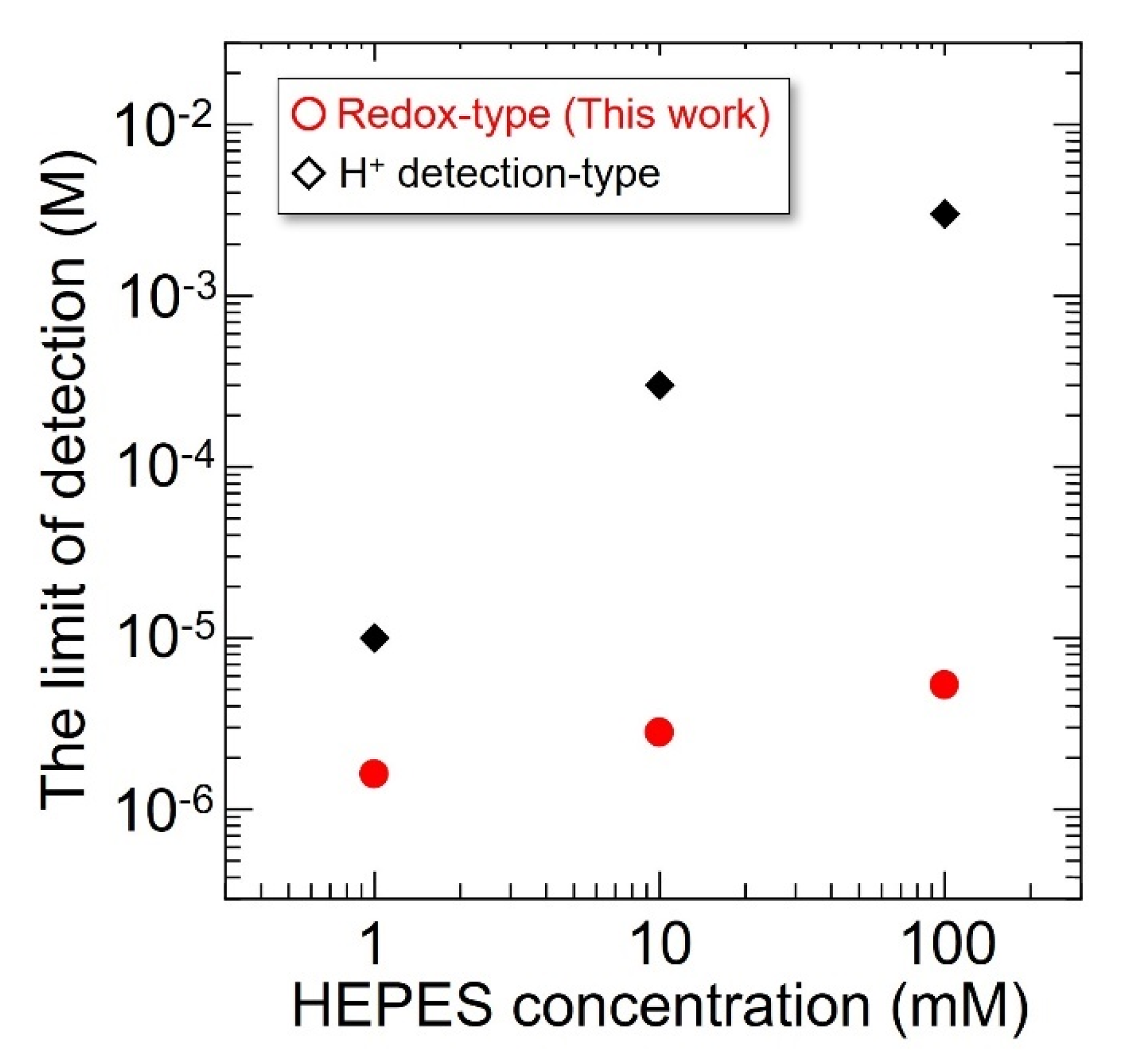
3.4. The Influence of pH Buffering Action on the Limit of Detection
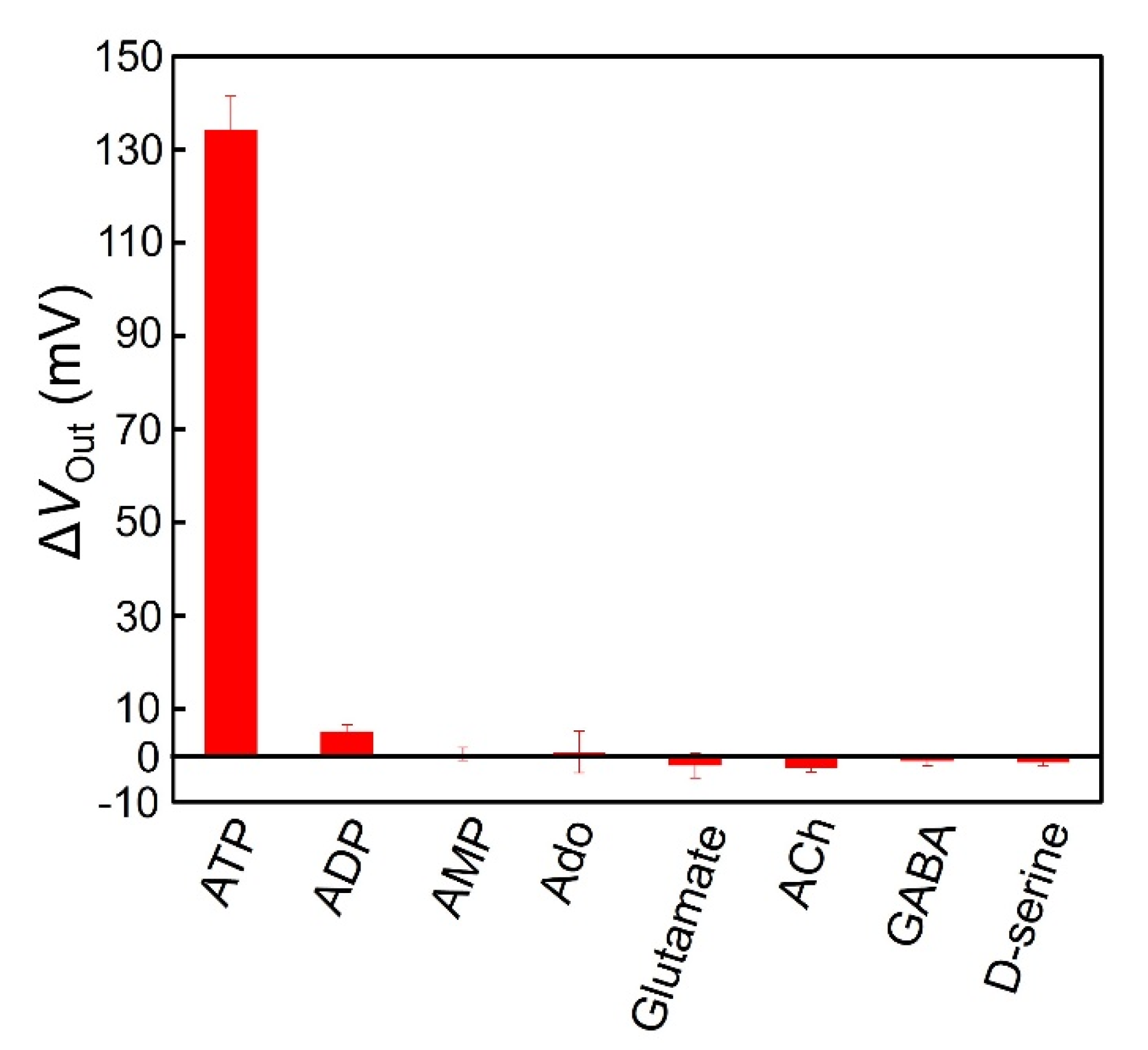
3.5. Selectivity for ATP
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Koizumi, S.; Fujishita, K.; Tsuda, M.; Shigemoto-Mogami, Y.; Inoue, K. Dynamic inhibition of excitatory synaptic transmission by astrocyte-derived ATP in hippocampal cultures. Proc. Natl. Acad. Sci. USA 2003, 100, 11023–11028. [Google Scholar] [CrossRef] [Green Version]
- Gourine, A.V.; Llaudet, E.; Dale, N.; Spyer, K.M. ATP is a mediator of chemosensory transduction in the central nervous system. Nature 2005, 436, 108–111. [Google Scholar] [CrossRef]
- Fellin, T.; Pascual, O.; Haydon, P.G. Astrocytes coordinate synaptic networks: Balanced excitation and inhibition. Physiology 2006, 21, 208–215. [Google Scholar] [CrossRef]
- Cao, X.; Li, L.P.; Wang, Q.; Wu, Q.; Hu, H.H.; Zhang, M.; Fang, Y.Y.; Zhang, J.; Li, S.J.; Xiong, W.C.; et al. Astrocyte-derived ATP modulates depressive-like behaviors. Nat. Med. 2013, 19, 773–777. [Google Scholar] [CrossRef]
- Abbasian, B.; Shair, A.; O’Gorman, D.B.; Pena-Diaz, A.M.; Brennan, L.; Engelbrecht, K.; Koenig, D.W.; Reid, G.; Burton, J.P. Potential Role of Extracellular ATP Released by Bacteria in Bladder Infection and Contractility. mSphere 2019, 4, e00439-19. [Google Scholar] [CrossRef] [Green Version]
- Amaral, S.S.; Oliveira, A.G.; Marques, P.E.; Quintão, J.L.D.; Pires, D.A.; Resende, R.R.; Sousa, B.R.; Melgaço, J.G.; Pinto, M.A.; Russo, R.C.; et al. Altered responsiveness to extracellular ATP enhances acetaminophen hepatotoxicity. Cell Commun. Signal. 2013, 11, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Ohyama, K.; Ikeshita, Y.; Fuchigami, Y.; Kawakami, S.; Nakashima, M.N.; Nakashima, M. Proteomic analysis and ATP assay reveal a positive effect of artificial cerebral spinal fluid perfusion following microdialysis sampling on repair of probe-induced brain damage. J. Neurosci. Methods 2019, 315, 1–5. [Google Scholar] [CrossRef] [Green Version]
- Kucherenko, I.S.; Didukh, D.Y.; Soldatkin, O.O.; Soldatkin, A.P. Amperometric biosensor system for simultaneous determination of adenosine-5′-triphosphate and glucose. Anal. Chem. 2014, 86, 5455–5462. [Google Scholar] [CrossRef]
- Liu, S.; Sun, Y. Co-immobilization of glucose oxidase and hexokinase on silicate hybrid sol-gel membrane for glucose and ATP detections. Biosens. Bioelectron. 2007, 22, 905–911. [Google Scholar] [CrossRef]
- Cui, Y.; Barford, J.P.; Renneberg, R. Amperometric trienzyme ATP biosensors based on the coimmobilization of salicylate hydroxylase, glucose-6-phosphate dehydrogenase, and hexokinase. Sens. Actuators B Chem. 2008, 132, 1–4. [Google Scholar] [CrossRef]
- Kucherenko, I.S.; Kucherenko, D.Y.; Soldatkin, O.O.; Lagarde, F.; Dzyadevych, S.V.; Soldatkin, A.P. A novel conductometric biosensor based on hexokinase for determination of adenosine triphosphate. Talanta 2016, 150, 469–475. [Google Scholar] [CrossRef]
- Patel, B.A.; Rogers, M.; Wieder, T.; O’Hare, D.; Boutelle, M.G. ATP microelectrode biosensor for stable long-term in vitro monitoring from gastrointestinal tissue. Biosens. Bioelectron. 2011, 26, 2890–2896. [Google Scholar] [CrossRef] [PubMed]
- Llaudet, E.; Hatz, S.; Droniou, M.; Dale, N. Microelectrode biosensor for real-time measurement of ATP in biological tissue. Anal. Chem. 2005, 77, 3267–3273. [Google Scholar] [CrossRef]
- Huckstepp, R.T.R.; Llaudet, E.; Gourine, A.V. CO2-Induced ATP-Dependent release of acetylcholine on the ventral surface of the medulla oblongata. PLoS ONE 2016, 11, e0167861. [Google Scholar] [CrossRef]
- Heinrich, A.; Andõ, R.D.; Túri, G.; Rõzsa, B.; Sperlágh, B. K+ depolarization evokes ATP, adenosine and glutamate release from glia in rat hippocampus: A microelectrode biosensor study. Br. J. Pharmacol. 2012, 167, 1003–1020. [Google Scholar] [CrossRef] [Green Version]
- Zhu, Q.; Liang, B.; Liang, Y.; Ji, L.; Cai, Y.; Wu, K.; Tu, T.; Ren, H.; Huang, B.; Wei, J.; et al. 3D bimetallic Au/Pt nanoflowers decorated needle-type microelectrode for direct in situ monitoring of ATP secreted from living cells. Biosens. Bioelectron. 2020, 153, 112019. [Google Scholar] [CrossRef]
- Pearson, R.A.; Dale, N.; Llaudet, E.; Mobbs, P. ATP released via gap junction hemichannels from the pigment epithelium regulates neural retinal progenitor proliferation. Neuron 2005, 46, 731–744. [Google Scholar] [CrossRef] [Green Version]
- Furuya, K.; Sokabe, M.; Grygorczyk, R. Real-time luminescence imaging of cellular ATP release. Methods 2014, 66, 330–344. [Google Scholar] [CrossRef]
- Tokunaga, T.; Namiki, S.; Yamada, K.; Imaishi, T.; Nonaka, H.; Hirose, K.; Sando, S. Cell surface-anchored fluorescent aptamer sensor enables imaging of chemical transmitter dynamics. J. Am. Chem. Soc. 2012, 134, 9561–9564. [Google Scholar] [CrossRef]
- Lobas, M.A.; Tao, R.; Nagai, J.; Kronschläger, M.T.; Borden, P.M.; Marvin, J.S.; Looger, L.L.; Khakh, B.S. A genetically encoded single-wavelength sensor for imaging cytosolic and cell surface ATP. Nat. Commun. 2019, 10, 711. [Google Scholar] [CrossRef] [Green Version]
- Conley, J.M.; Radhakrishnan, S.; Valentino, S.A.; Tantama, M. Imaging extracellular ATP with a genetically-encoded, ratiometric fluorescent sensor. PLoS ONE 2017, 12, e0187481. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kitajima, N.; Takikawa, K.; Sekiya, H.; Satoh, K.; Asanuma, D.; Sakamoto, H.; Takahashi, S.; Hanaoka, K.; Urano, Y.; Namiki, S.; et al. Real-time in vivo imaging of extracellular atp in the brain with a hybrid-type fluorescent sensor. eLife 2020, 9, e57544. [Google Scholar] [CrossRef]
- Futagawa, M.; Suzuki, D.; Otake, R.; Dasai, F.; Ishida, M.; Sawada, K. Fabrication of a 128 × 128 Pixels Charge Transfer Type Hydrogen Ion Image Sensor. IEEE Trans. Electron. Devices 2013, 60, 2634–2639. [Google Scholar] [CrossRef]
- Horiuchi, H.; Agetsuma, M.; Ishida, J.; Nakamura, Y.; Lawrence Cheung, D.; Nanasaki, S.; Kimura, Y.; Iwata, T.; Takahashi, K.; Sawada, K.; et al. CMOS-based bio-image sensor spatially resolves neural activity-dependent proton dynamics in the living brain. Nat. Commun. 2020, 11, 712. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Iwata, T.; Doi, H.; Okumura, K.; Horio, T.; Hattori, T.; Takahashi, K.; Sawada, K. Comparative study on the deposition of enzyme-entrapped membranes with spatial homogeneity for bioimaging. Sens. Actuators B Chem. 2017, 239, 800–806. [Google Scholar] [CrossRef] [Green Version]
- Takenaga, S.; Tamai, Y.; Okumura, K.; Ishida, M.; Sawada, K. Label-free acetylcholine image sensor based on charge transfer technology for biological phenomenon tracking. Jpn. J. Appl. Phys. 2012, 51, 2–7. [Google Scholar] [CrossRef]
- Doi, H.; Parajuli, B.; Horio, T.; Shigetomi, E.; Shinozaki, Y.; Noda, T.; Takahashi, K.; Hattori, T.; Koizumi, S.; Sawada, K. Development of a label-free ATP image sensor for analyzing spatiotemporal patterns of ATP release from biological tissues. Sens. Actuators B Chem. 2021, 335, 129686. [Google Scholar] [CrossRef]
- Iwabuchi, S.; Kawahara, K. Extracellular ATP-prinoceptor signaling and AMP-activated protein kinase regulate astrocytic glucose transporter 3 in an in vitro ischemia. Neurochem. Int. 2013, 63, 259–268. [Google Scholar] [CrossRef]
- Tchernookova, B.K.; Heer, C.; Young, M.; Swygart, D.; Kaufman, R.; Gongwer, M.; Shepherd, L.; Caringal, H.; Jacoby, J.; Kreitzer, M.A.; et al. Activation of retinal glial (Müller) cells by extracellular ATP induces pronounced increases in extracellular H+ flux. PLoS ONE 2018, 13, e0190893. [Google Scholar] [CrossRef] [Green Version]
- Okumura, Y.; Iwata, T.; Okumura, K.; Shiguma, H.; Horio, T.; Doi, H.; Takahashi, K.; Sawada, K. BS3.4—H2O2 and Glutamate Imaging with Improved Sensitivity Based on Charge-Transfer-Type Potentiometric Redox Sensor Arrays. Proc. IMCS 2020, 141–142. [Google Scholar] [CrossRef]
- Sensor, C.P. Redox Sensor Array with 23.5-µm Resolution for Real-Time Imaging of Hydrogen Peroxide and Glutamate Based on Charge-Transfer-Type Potentiometric Sensor. Sensors 2021, 21, 7682. [Google Scholar]
- Okumura, Y.; Mizutani, S.; Okumura, K.; Iwata, T.; Takahashi, K.; Sawada, K. Development of H2O2 image sensor for GABA and glutamate imaging. In Proceedings of the TRANSDUCERS 2017—19th International Conference on Solid-State Sensors, Actuators and Microsystems (TRANSDUCERS), Kaohsiung, Taiwan, 18–22 June 2017; pp. 1551–1554. [Google Scholar]
- Migita, S.; Ozasa, K.; Tanaka, T.; Haruyama, T. Enzyme-based field-effect transistor for adenosine triphosphate (ATP) sensing. Anal. Sci. 2007, 23, 45–48. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, Y.N.; Okumura, K.; Iwata, T.; Takahashi, K.; Hattori, T.; Ishida, M.; Sawada, K. Development of an ATP and hydrogen ion image sensor using a patterned apyrase-immobilized membrane. Talanta 2016, 161, 419–424. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Duarte-Guevara, C.; Swaminathan, V.; Reddy, B.; Wen, C.H.; Huang, Y.J.; Huang, J.C.; Liu, Y.S.; Bashir, R. Characterization of a 1024 × 1024 DG-BioFET platform. Sens. Actuators B Chem. 2017, 250, 100–110. [Google Scholar] [CrossRef]
- Lee, Y.N.; Araki, T.; Kimura, Y.; Dasai, F.; Iwata, T.; Takahashi, K.; Sawada, K. High-Density 2-μm-Pitch pH Image Sensor with High-Speed Operation up to 1933 fps. IEEE Trans. Biomed. Circuits Syst. 2019, 13, 352–363. [Google Scholar] [CrossRef] [PubMed]

| Bio-Recognition | Pixel-Pitch | Pixel Number | Sensitivity | LOD | HEPES | pH | Reference |
|---|---|---|---|---|---|---|---|
| H+-Apyrase | 37.3 µm | 16,384 | No data | 100 µM | 1 mM | 8.0 | [25] |
| H+-Apyrase | 37.3 µm | 16,384 | 15.5 mV/dec | 10 µM | 1 mM | 7.4 | [27] |
| H+-Apyrase | 37.3 µm | 16,384 | 37.8 mV/mM | 1.3 µM | 1 mM | 8.0 | [34] |
| GK-LGOx-HRP | 37.3 µm | 16,384 | 77 ± 3.8 mV/dec | 2.8 µM | 10 mM | 7.4 | This work |
| Approach | Bio-Recognition | LOD | Linear Range | Spatiotemporal Analysis | Reference |
|---|---|---|---|---|---|
| Amperometric | GOD-HEX | 9.9 ± 3.2 nM | 0.25–4 µM | × | [12] |
| Amperometric | GK-G3PO | 200 nM | 200 nM–50 µM | × | [13] |
| Amperometric | HEX | 15 µM | 15–300 µM | × | [11] |
| Amperometric | SHL-G6PDH-HEX | 3 µM | 5 µM–4 mM | × | [10] |
| Potentiometric | GK-LGOx-HRP | 2.8 µM | 3–300 µM | ◯ | This work |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Doi, H.; Horio, T.; Choi, Y.-J.; Takahashi, K.; Noda, T.; Sawada, K. CMOS-Based Redox-Type Label-Free ATP Image Sensor for In Vitro Sensitive Imaging of Extracellular ATP. Sensors 2022, 22, 75. https://doi.org/10.3390/s22010075
Doi H, Horio T, Choi Y-J, Takahashi K, Noda T, Sawada K. CMOS-Based Redox-Type Label-Free ATP Image Sensor for In Vitro Sensitive Imaging of Extracellular ATP. Sensors. 2022; 22(1):75. https://doi.org/10.3390/s22010075
Chicago/Turabian StyleDoi, Hideo, Tomoko Horio, Yong-Joon Choi, Kazuhiro Takahashi, Toshihiko Noda, and Kazuaki Sawada. 2022. "CMOS-Based Redox-Type Label-Free ATP Image Sensor for In Vitro Sensitive Imaging of Extracellular ATP" Sensors 22, no. 1: 75. https://doi.org/10.3390/s22010075
APA StyleDoi, H., Horio, T., Choi, Y.-J., Takahashi, K., Noda, T., & Sawada, K. (2022). CMOS-Based Redox-Type Label-Free ATP Image Sensor for In Vitro Sensitive Imaging of Extracellular ATP. Sensors, 22(1), 75. https://doi.org/10.3390/s22010075







