19β,28-Epoxy-18α-olean-3β-ol-2-furoate from Allobetulin (19β,28-Epoxy-18α-olean-3β-ol)
Abstract
1. Introduction
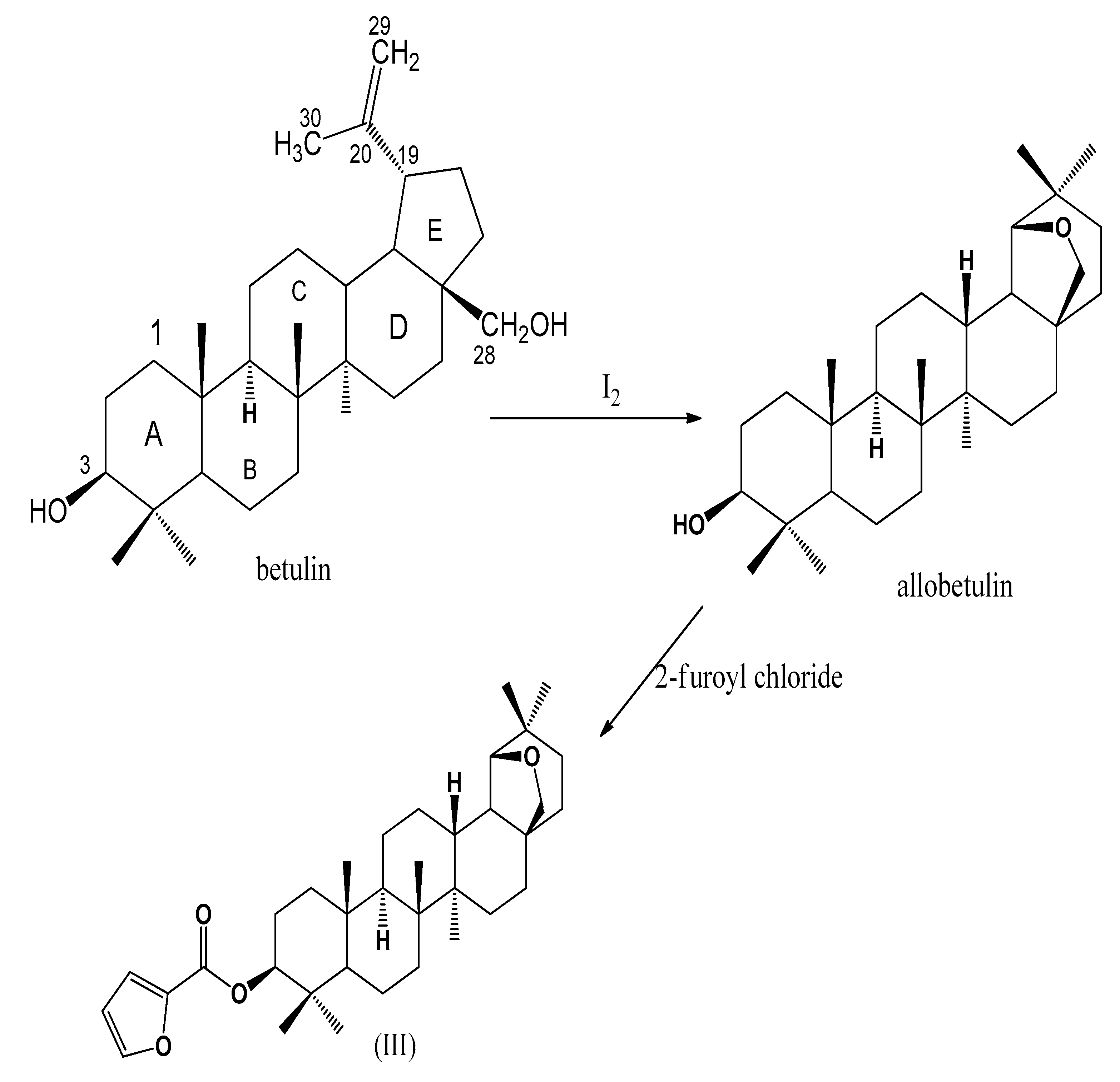
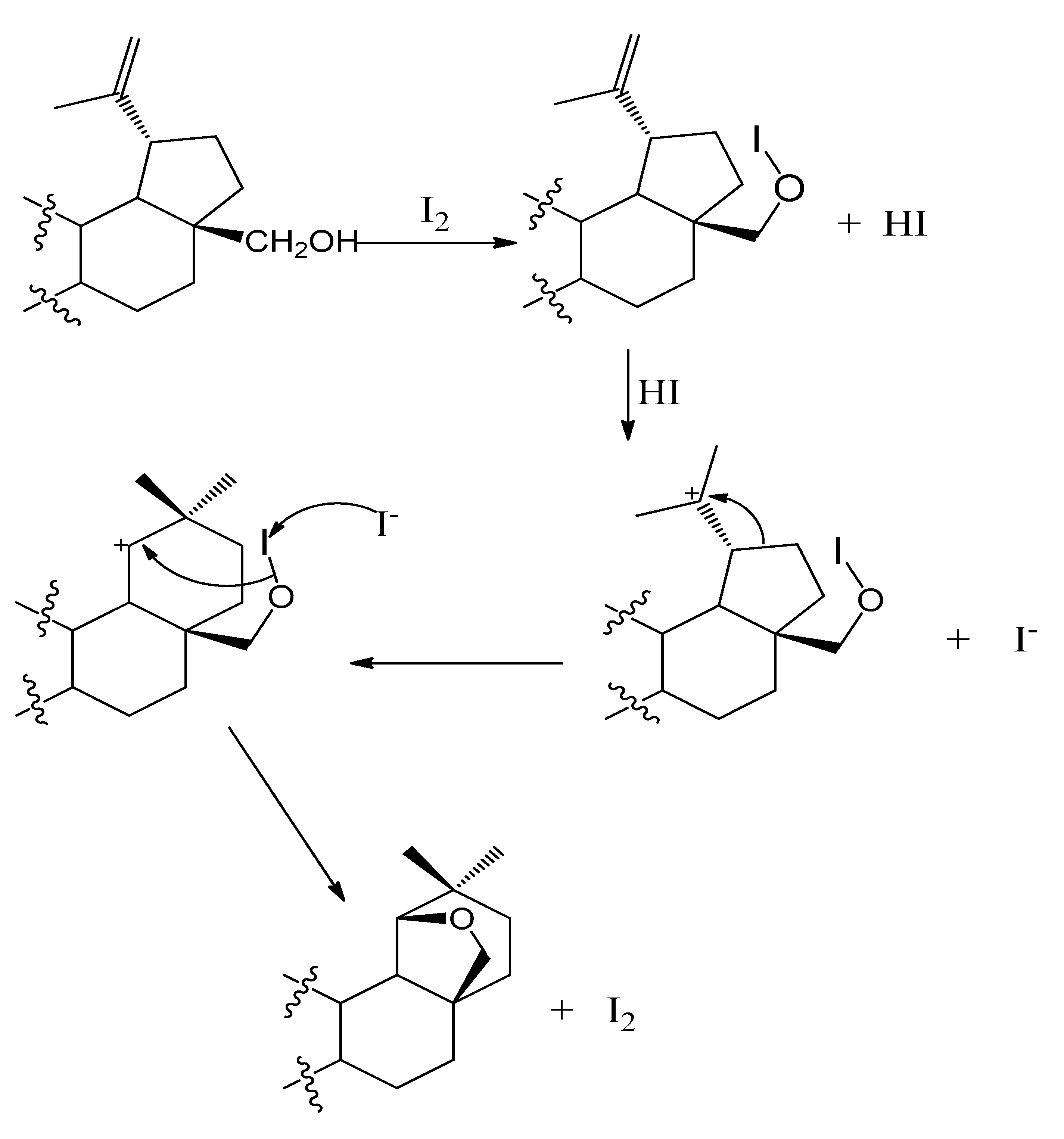
2. Results and Discussion
3. Experimental
3.1. 19β,28-Epoxy-18α-olean-3β-ol (II) (Allobetulin)
3.2. 19β,28-Epoxy-18α-olean-3β-ol-2-furoate (III)
Supplementary Materials
Funding
Data Availability Statement
Conflicts of Interest
References
- Krol, S.K.; Kiełbus, M.; Rivero-Müller, A.; Stepulak, A. Comprehensive Review on Betulin as a Potent Anticancer Agent. BioMed Res. Int. 2015, 2015, 584189. [Google Scholar] [CrossRef] [PubMed]
- Sami, A.; Taru, M.; Salme, K.; Jari, Y.-K. Pharmacological properties of the ubiquitous natural product betulin. Eur. J. Pharm. Sci. 2006, 29, 1–13. [Google Scholar]
- Soler, F.; Poujade, C.; Evers, M.; Carry, J.-C.; Hénin, Y.; Bousseau, A.; Huet, T.; Pauwels, R.; De Clercq, E.; Mayaux, J.-F.; et al. Betulinic Acid Derivatives: A New Class of Specific Inhibitors of Human Immunodeficiency Virus Type 1 Entry. J. Med. Chem. 1996, 39, 1069–1083. [Google Scholar] [CrossRef] [PubMed]
- Kashiwada, Y.; Hashimoto, F.; Cosentino, L.M.; Chen, C.-H.; Garrett, P.E.; Lee, K.-H. Betulinic Acid and Dihydrobetulinic Acid Derivatives as Potent Anti-HIV Agents. J. Med. Chem. 1996, 39, 1016–1017. [Google Scholar] [CrossRef] [PubMed]
- Tolstikov, G.A.; Flekhter, O.B.; Shultz, E.E.; Baltina, L.A.; Tolstikov, A.G. Betulin and Its Derivatives. Chemistry and Biological Activity. Chem. Sustain. Dev. 2005, 13, 1–29. [Google Scholar]
- Zhang, D.-M.; Xu, H.-G.; Wang, L.; Li, Y.-J.; Sun, P.-H.; Wu, X.-M.; Wang, G.-J.; Chen, W.-M.; Ye, W.-C. Betulinic Acid and its Derivatives as Potential Antitumor Agents. Med. Res. Rev. 2015, 35, 1127–1155. [Google Scholar] [CrossRef]
- Bori, I.D.; Hung, H.-Y.; Qian, K.; Chen, C.-H.; Morris-Natschke, S.L.; Lee, K.-H. Anti-AIDS agents 88. Anti-HIV conjugates of betulin and betulinic acid with AZT prepared via click chemistry. Tetrahedron Lett. 2012, 53, 1987–1989. [Google Scholar] [CrossRef]
- Hordyjewska, A.; Ostapiuk, A.; Horecka, A.; Kurzepa, J. Betulin and betulinic acid: Triterpenoids derivatives with a powerful biological potential. Phytochem. Rev. 2019, 18, 929–951. [Google Scholar] [CrossRef]
- Kommera, H.; Kaluđerović, G.N.; Kalbitz, J.; Paschke, R. Synthesis and Anticancer Activity of Novel Betulinic acid and Betulin Derivatives. Arch. Pharm. 2010, 343, 449–457. [Google Scholar] [CrossRef]
- Soica, C.M.; Dehelean, C.A.; Peev, C.; Aluas, M.; Zupkó, I.; Kása, P., Jr.; Alexa, E. Physico-chemical comparison of betulinic acid, betulin and birch bark extract and in vitro investigation of their cytotoxic effects towards skin epidermoid carcinoma (A431), breast carcinoma (MCF7) and cervix adenocarcinoma (HeLa) cell lines. Nat. Prod. Res. 2012, 26, 968–974. [Google Scholar] [CrossRef]
- Zhao, J.; Li, R.; Pawlak, A.; Henklewska, M.; Sysak, A.; Wen, L.; Yi, J.E.; Obmińska-Mrukowicz, B. Antitumor Activity of Bet-ulinic Acid and Betulin in Canine Cancer Cell Lines. In Vivo 2018, 32, 1081–1088. [Google Scholar] [CrossRef] [PubMed]
- Tubek, B.; Mituła, P.; Niezgoda, N.; Kempińska, K.; Wietrzyk, J.; Wawrzeńczyk, C. Synthesis and Cytotoxic Activity of New Betulin and Betulinic Acid Esters with Conjugated Linoleic Acid (CLA). Nat. Prod. Commun. 2013, 8, 435–438. [Google Scholar] [CrossRef] [PubMed]
- Shi, W.; Tang, N.; Yan, W.-D. Synthesis and cytotoxicity of triterpenoids derived from betulin and betulinic acid via click chemistry. J. Asian Nat. Prod. Res. 2014, 17, 159–169. [Google Scholar] [CrossRef] [PubMed]
- Kvasnica, M.; Sarek, J.; Klinotova, E.; Dzubak, P.; Hajduch, M. Synthesis of phthalates of betulinic acid and betulin with cytotoxic activity. Bioorg. Med. Chem. 2005, 13, 3447–3454. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, K.; Lu, H.; Lu, J.; Chen, G.; Yokoyama, T.; Sagara, Y.; Manabe, M.; Kodama, H. Effect of three triterpenoids, lupeol, betulin, and betulinic acid on the stimulus-induced superoxide generation and tyrosyl phosphorylation of proteins in human neutrophils. Clin. Chim. Acta 2002, 325, 91–96. [Google Scholar] [CrossRef]
- Gauthier, C.; Legault, J.; Lavoie, S.; Rondeau, S.; Tremblay, S.; Pichette, A. Synthesis and Cytotoxicity of Bidesmosidic Betulin and Betulinic Acid Saponins. J. Nat. Prod. 2008, 72, 72–81. [Google Scholar] [CrossRef]
- Oloyede, H.; Ajiboye, H.; Salawu, M. Influence of oxidative stress on the antibacterial activity of betulin, betulinic acid and ursolic acid. Microb. Pathog. 2017, 111, 338–344. [Google Scholar] [CrossRef]
- Arrous, I.; Boudebouz, O.; Voronova, O.; Plotnikov, E.; Bakibaev, A. Synthesis and antioxidant evaluation of some new allobetulin esters. Rasayan J. Chem. 2019, 12, 1032–1037. [Google Scholar] [CrossRef]
- Dehaen, W.; Mashentseva, A.A.; Seitembetov, T.S. Allobetulin and Its Derivatives: Synthesis and Biological Activity. Molecules 2011, 16, 2443–2466. [Google Scholar] [CrossRef]
- Green, B.; Bentley, M.D.; Chung, B.Y.; Lynch, N.G.; Jensen, B.L. Isolation of Betulin and Rearrangement to Allobetulin. A Biomimetic Natural Product Synthesis. J. Chem. Educ. 2007, 84, 1985. [Google Scholar] [CrossRef]
- Grymel, M.; Adamek, J. Allobetulin. Molbank 2022, 2022, M1446. [Google Scholar] [CrossRef]
- Kasa, A.; Varala, R.; Swami, P.M.; Zubaidha, P. Selective and efficient etherification of secondary cinnamyl alcohols in the presence of iodine catalyst and evaluation of their anti-candida activity. Chem. J. 2013, 03, 66–74. [Google Scholar]
- Kasashima, Y.; Fujimoto, H.; Mino, T.; Sakamoto, M.; Fujita, T. An Efficient Synthesis of Five-membered Cyclic Ethers from 1,3-Diols Using Molecular Iodine as a Catalyst. J. Oleo Sci. 2008, 57, 437–443. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kasashima, Y.; Uzawa, A.; Hashimoto, K.; Nishida, T.; Murakami, K.; Mino, T.; Sakamoto, M.; Fujita, T. Synthesis of Cinnamyl Ethers from .ALPHA.-Vinylbenzyl Alcohol Using Iodine as Catalyst. J. Oleo Sci. 2010, 59, 549–555. [Google Scholar] [CrossRef] [PubMed]
- Abraham, R.J.; Bernstein, H.J. The proton resonance spectra of furan and pyrrole. Can. J. Chem. 1959, 37, 1056–1065. [Google Scholar] [CrossRef]
- Bardsley, B.; Smith, M.S.; Gibbon, B.H. Structure elucidation and spectroscopic analysis of photodegradants of the anti-rhinitis drug fluticasone furoate. Org. Biomol. Chem. 2010, 8, 1876–1880. [Google Scholar] [CrossRef]
- Schulze, H.; Pierok, K. Zur Kenntnis des Betulins. Ber. Dtsch. Chem. Ges. 1922, 55, 2332. [Google Scholar] [CrossRef]
- Pettit, G.R.; Green, B.; Bowyer, W.J. Steroids and Related Natural Products. VI. The Structure of α-Apoallobetulin. J. Org. Chem. 1961, 8, 2879–2883. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lugemwa, F.N. 19β,28-Epoxy-18α-olean-3β-ol-2-furoate from Allobetulin (19β,28-Epoxy-18α-olean-3β-ol). Molbank 2022, 2022, M1499. https://doi.org/10.3390/M1499
Lugemwa FN. 19β,28-Epoxy-18α-olean-3β-ol-2-furoate from Allobetulin (19β,28-Epoxy-18α-olean-3β-ol). Molbank. 2022; 2022(4):M1499. https://doi.org/10.3390/M1499
Chicago/Turabian StyleLugemwa, Fulgentius Nelson. 2022. "19β,28-Epoxy-18α-olean-3β-ol-2-furoate from Allobetulin (19β,28-Epoxy-18α-olean-3β-ol)" Molbank 2022, no. 4: M1499. https://doi.org/10.3390/M1499
APA StyleLugemwa, F. N. (2022). 19β,28-Epoxy-18α-olean-3β-ol-2-furoate from Allobetulin (19β,28-Epoxy-18α-olean-3β-ol). Molbank, 2022(4), M1499. https://doi.org/10.3390/M1499






