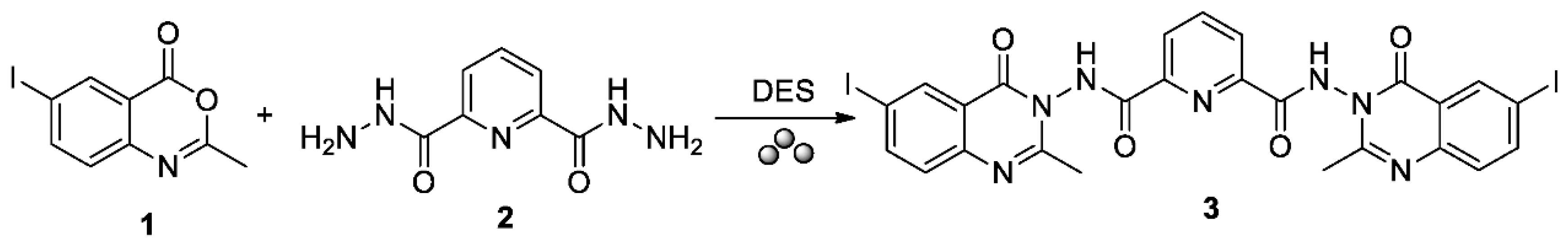
N2,N6-Bis(6-iodo-2-methyl-4-oxoquinazolin-3(4H)-yl)pyridine-2,6-dicarboxamide
Abstract
1. Introduction
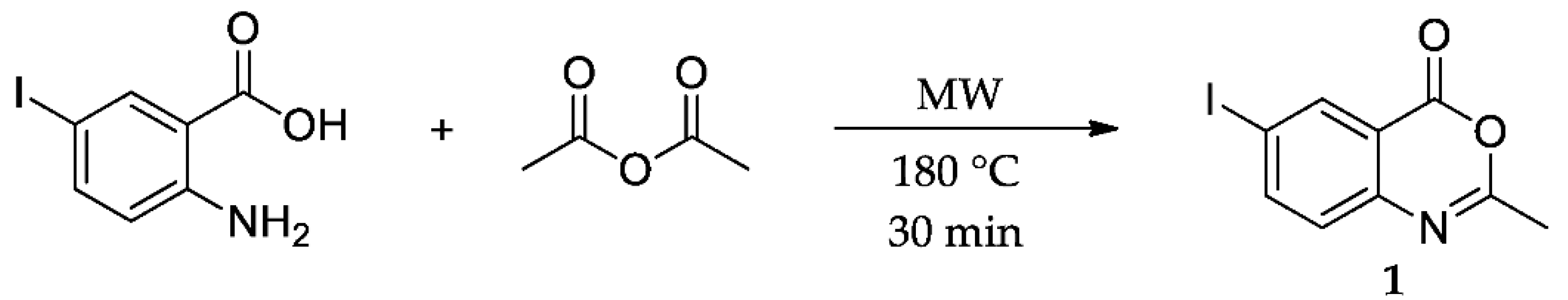
2. Results and Discussion
3. Materials and Methods
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sayyed, M.A.; Mokle, S.S.; Vibhute, Y.B. Synthesis of 6-Iodo/Bromo-3-Amino-2-Methylquinazolin-4(3H)-Ones by Direct Halogenation and Their Schiff Base Derivatives. ARKIVOC 2006, 2006, 221–226. [Google Scholar] [CrossRef]
- Atia, A.J.K.; Al-Mufrgeiy, S.S. Synthesis and Antibacterial Activities of New 3-Amino-2-Methyl-Quinazolin-4 (3H)-One Derivatives. Am. J. Chem. 2012, 2, 150–156. [Google Scholar] [CrossRef]
- Gatadi, S.; Gour, J.; Shukla, M.; Kaul, G.; Das, S.; Dasgupta, A.; Malasala, S.; Borra, R.S.; Madhavi, Y.V.; Chopra, S.; et al. Synthesis of 1,2,3-Triazole Linked 4(3H)-Quinazolinones as Potent Antibacterial Agents against Multidrug-Resistant Staphylococcus Aureus. Eur. J. Med. Chem. 2018, 157, 1056–1067. [Google Scholar] [CrossRef]
- Masri, A.; Anwar, A.; Khan, N.A.; Shahbaz, M.S.; Khan, K.M.; Shahabuddin, S.; Siddiqui, R. Antibacterial Effects of Quinazolin-4(3H)-One Functionalized-Conjugated Silver Nanoparticles. Antibiotics 2019, 8, 179. [Google Scholar] [CrossRef] [PubMed]
- Long, S.; Resende, D.I.S.P.; Kijjoa, A.; Silva, A.M.S.; Pina, A.; Fernández-Marcelo, T.; Vasconcelos, M.H.; Sousa, E.; Pinto, M.M.M. Antitumor Activity of Quinazolinone Alkaloids Inspired by Marine Natural Products. Mar. Drugs 2018, 16, 261. [Google Scholar] [CrossRef] [PubMed]
- Lai, K.-C.; Chia, Y.-T.; Yih, L.-H.; Lu, Y.-L.; Chang, S.-T.; Hong, Z.-X.; Chen, T.-L.; Hour, M.-J. Antitumor Effects of the Novel Quinazolinone Holu-12: Induction of Mitotic Arrest and Apoptosis in Human Oral Squamous Cell Carcinoma CAL27 Cells. Anticancer Res. 2021, 41, 259–268. [Google Scholar] [CrossRef]
- Wdowiak, P.; Matysiak, J.; Kuszta, P.; Czarnek, K.; Niezabitowska, E.; Baj, T. Quinazoline Derivatives as Potential Therapeutic Agents in Urinary Bladder Cancer Therapy. Front. Chem. 2021, 9, 765552. [Google Scholar] [CrossRef]
- Niu, Z.; Ma, S.; Zhang, L.; Liu, Q.; Zhang, S. Discovery of Novel Quinazoline Derivatives as Potent Antitumor Agents. Molecules 2022, 27, 3906. [Google Scholar] [CrossRef]
- Saeedi, M.; Mohammadi-Khanaposhtani, M.; Pourrabia, P.; Razzaghi, N.; Ghadimi, R.; Imanparast, S.; Faramarzi, M.A.; Bandarian, F.; Esfahani, E.N.; Safavi, M.; et al. Design and Synthesis of Novel Quinazolinone-1,2,3-Triazole Hybrids as New Anti-Diabetic Agents: In Vitro α-Glucosidase Inhibition, Kinetic, and Docking Study. Bioorg. Chem. 2019, 83, 161–169. [Google Scholar] [CrossRef]
- Khalifa, M.M.; Sakr, H.M.; Ibrahim, A.; Mansour, A.M.; Ayyad, R.R. Design and Synthesis of New Benzylidene-Quinazolinone Hybrids as Potential Anti-Diabetic Agents: In Vitro α-Glucosidase Inhibition, and Docking Studies. J. Mol. Struct. 2022, 1250, 131768. [Google Scholar] [CrossRef]
- Barmak, A.; Niknam, K.; Mohebbi, G. Synthesis, Structural Studies, and α-Glucosidase Inhibitory, Antidiabetic, and Antioxidant Activities of 2,3-Dihydroquinazolin-4(1H)-Ones Derived from Pyrazol-4-Carbaldehyde and Anilines. ACS Omega 2019, 4, 18087–18099. [Google Scholar] [CrossRef] [PubMed]
- Chaitanya, P.; Reddy, G.D.; Varun, G.; Srikanth, L.M.; Prasad, V.V.S.R.; Ravindernath, A. Design and Synthesis of Quinazolinone Derivatives as Anti-Inflammatory Agents: Pharmacophore Modeling and 3D QSAR Studies. Med. Chem. 2014, 10, 711–723. [Google Scholar] [CrossRef] [PubMed]
- Krishnarth, N.; Verma, S.K.; Chaudhary, A. Synthesis and Anti-Inflammatory Activity of Some Novel Quinazolinone Derivatives. FABAD J. Pharm. Sci. 2020, 45, 205–210. [Google Scholar]
- Poojari, S.; Krishnamurthy, G.; KS, J.K.; Kumar, S.; Naik, S. Anti-Inflammatory, Antibacterial and Molecular Docking Studies of Novel Spiro-Piperidine Quinazolinone Derivatives. J. Taibah Univ. Sci. 2017, 11, 497–511. [Google Scholar] [CrossRef]
- Altaf, A.A.; Shahzad, A.; Gul, Z.; Rasool, N.; Badshah, A.; Lal, B.; Khan, E. A Review on the Medicinal Importance of Pyridine Derivatives. J. Drug Des. Med. Chem. 2015, 1, 1. [Google Scholar] [CrossRef]
- Ling, Y.; Hao, Z.-Y.; Liang, D.; Zhang, C.-L.; Liu, Y.-F.; Wang, Y. The Expanding Role of Pyridine and Dihydropyridine Scaffolds in Drug Design. Drug Des. Dev. Ther. 2021, 15, 4289–4338. [Google Scholar] [CrossRef]
- Anastas, P.T.; Warner, J.C. Green Chemistry: Theory and Practice; Oxford University Press: Oxford, UK, 2000; ISBN 978-0-19-850698-0. [Google Scholar]
- David, E.; Niculescu, V.-C. Volatile Organic Compounds (VOCs) as Environmental Pollutants: Occurrence and Mitigation Using Nanomaterials. Int. J. Environ. Res. Public Health 2021, 18, 13147. [Google Scholar] [CrossRef]
- Bedlovičová, Z. Green Synthesis—An Overview. In Green Synthesis of Silver Nanomaterials; Elsevier Inc.: Amsterdam, The Netherlands, 2022; pp. 547–569. [Google Scholar]
- Do, J.-L.; Friščić, T. Mechanochemistry: A Force of Synthesis. ACS Cent. Sci. 2017, 3, 13–19. [Google Scholar] [CrossRef]
- Komar, M.; Prašnikar, F.; Kraljević, T.G.; Aladić, K.; Molnar, M. 3-Amino-2-Methylquinazolin-4-(3H)-One Schiff Bases Synthesis—A Green Chemistry Approach—A Comparison of Microwave and Ultrasound Promoted Synthesis with Mechanosynthesis. Curr. Green Chem. 2021, 8, 62–69. [Google Scholar] [CrossRef]
- Molnar, M.; Pavić, V.; Šarkanj, B.; Čačić, M.; Vuković, D.; Klenkar, J. Mono- and Bis-Dipicolinic Acid Heterocyclic Derivatives—Thiosemicarbazides, Triazoles, Oxadiazoles and Thiazolidinones as Antifungal and Antioxidant Agents. Heterocycl Commun 2017, 23, 35–42. [Google Scholar] [CrossRef]
- Ramanathan, M.; Hsu, M.-T.; Liu, S.-T. Preparation of 4(3H)-Quinazolinones from Aryldiazonium Salt, Nitriles and 2-Aminobenzoate via a Cascade Annulation. Tetrahedron 2019, 75, 791–796. [Google Scholar] [CrossRef]
- Rajput, C.S.; Kumar, A.; Kumar Bhati, S.; Singh, J. Synthesis and Antiinflammatory Activity of 2-[5′-(4-Pyridinyl)-1′,2′,3′-Oxadiazol-2-Yl-Thiomethyl]-3-Substituted-Aryl-6-Substituted-Quinazolin-4-Ones. Asian J. Chem. 2008, 20, 6246–6252. [Google Scholar]
- Marinho, E.; Proença, M.F. The Reaction of 2-(Acylamino)Benzonitriles with Primary Aromatic Amines: A Convenient Synthesis of 2-Substituted 4-(Arylamino)Quinazolines. Synthesis 2015, 47, 1623–1632. [Google Scholar] [CrossRef]
- Ajani, O.O.; Audu, O.Y.; Germann, M.W.; Bello, B.L. Expeditious Synthesis and Spectroscopic Characterization of 2-Methyl-3-Substituted-Quinazolin-4(3H)-One Derivatives. Orient. J. Chem. 2017, 33, 562–574. [Google Scholar] [CrossRef]
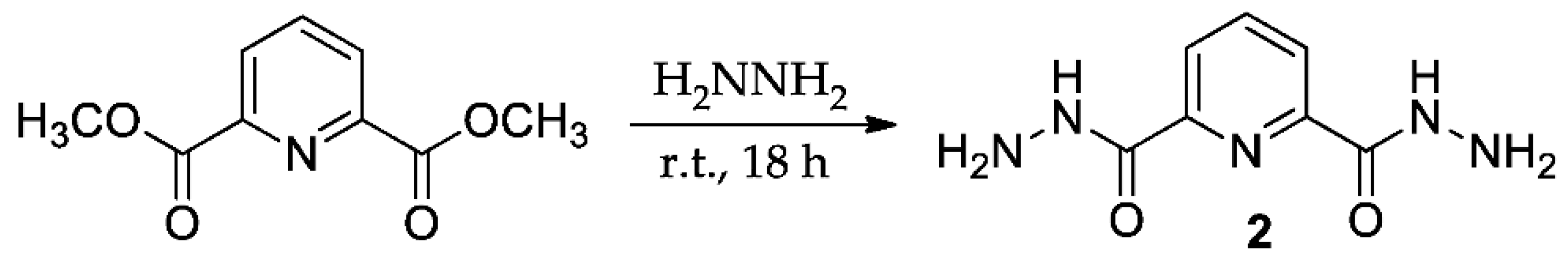
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Molnar, M.; Komar, M.; Jerković, I. N2,N6-Bis(6-iodo-2-methyl-4-oxoquinazolin-3(4H)-yl)pyridine-2,6-dicarboxamide. Molbank 2022, 2022, M1500. https://doi.org/10.3390/M1500
Molnar M, Komar M, Jerković I. N2,N6-Bis(6-iodo-2-methyl-4-oxoquinazolin-3(4H)-yl)pyridine-2,6-dicarboxamide. Molbank. 2022; 2022(4):M1500. https://doi.org/10.3390/M1500
Chicago/Turabian StyleMolnar, Maja, Mario Komar, and Igor Jerković. 2022. "N2,N6-Bis(6-iodo-2-methyl-4-oxoquinazolin-3(4H)-yl)pyridine-2,6-dicarboxamide" Molbank 2022, no. 4: M1500. https://doi.org/10.3390/M1500
APA StyleMolnar, M., Komar, M., & Jerković, I. (2022). N2,N6-Bis(6-iodo-2-methyl-4-oxoquinazolin-3(4H)-yl)pyridine-2,6-dicarboxamide. Molbank, 2022(4), M1500. https://doi.org/10.3390/M1500







