Abstract
The title compound was synthesized by multicomponent condensation of 3-amino-1,2,4-triazole, acetone and 5-bromo-2-hydroxy-3-methoxybenzaldehyde. The structure was established by 1H-NMR, 13C-NMR, IR spectroscopy and LC-MS.
1. Introduction
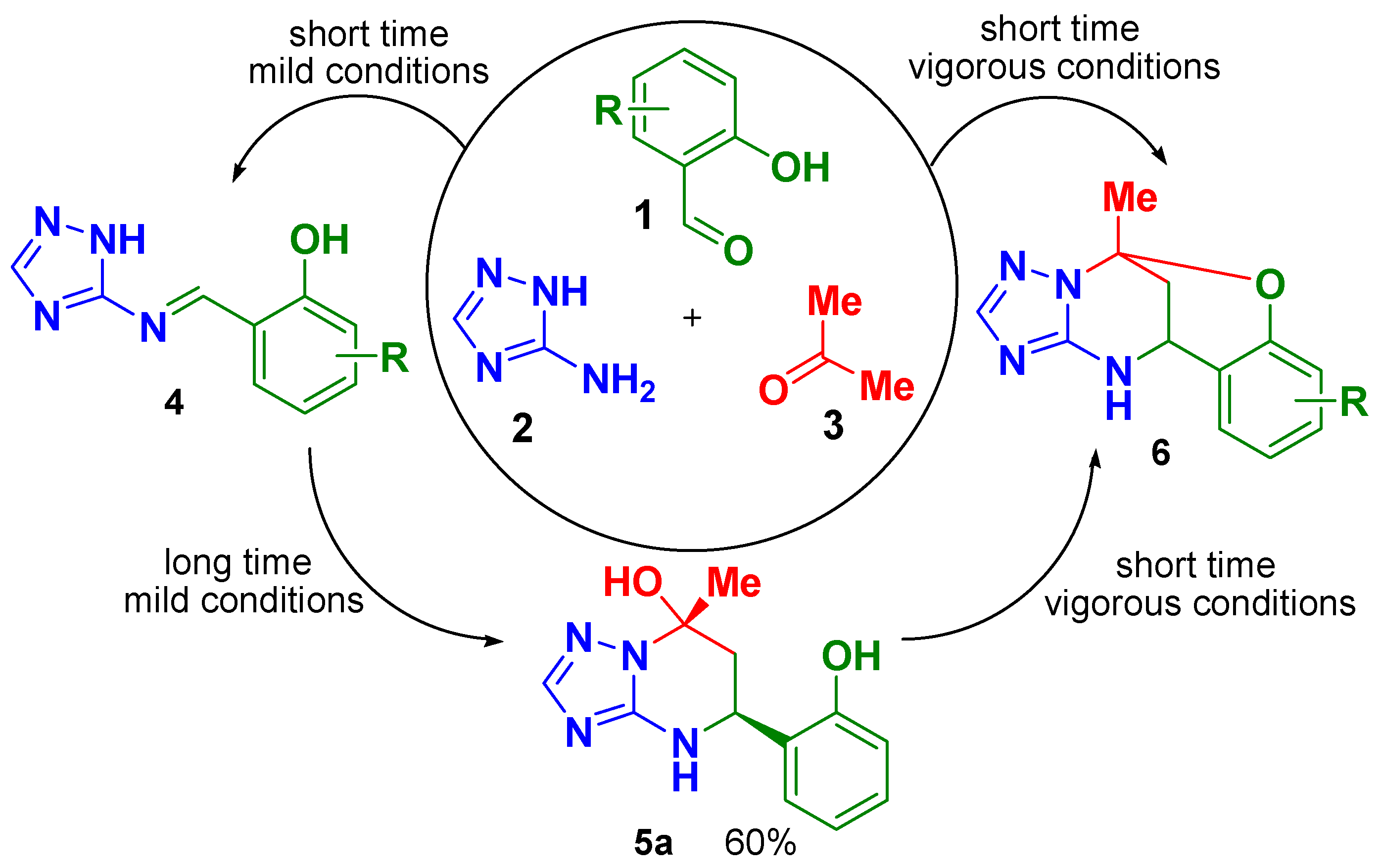
Application of multicomponent reactions (MCRs) in organic synthesis is a powerful method to create a chemical complexity from simple starting building blocks [1,2,3]. The use of aminoazoles in MCRs as building blocks having two or more nonequivalent reaction centers is often associated with ambiguous selectivity of the process and different reaction outcomes depending on the reaction conditions applied [4,5,6]. In particular, Biginelli-like MCRs of 3-amino-1,2,4-triazole with aldehydes and α-carbonyl CH-acids may result in several types of heterocyclic products (for a review see [7]). In our previous works we have shown that MCR of 3-amino-1,2,4-triazole (2) with acetone (3) and 2-hydroxybenzaldehydes (1) under acidic conditions leads to formation of different products, see Scheme 1. Under vigorous conditions, a series of oxygen-bridged compounds 6 was obtained with different substituents R, and only one example of open-bridge structure 5a (R = H) was isolated in high yield under mild conditions [5].
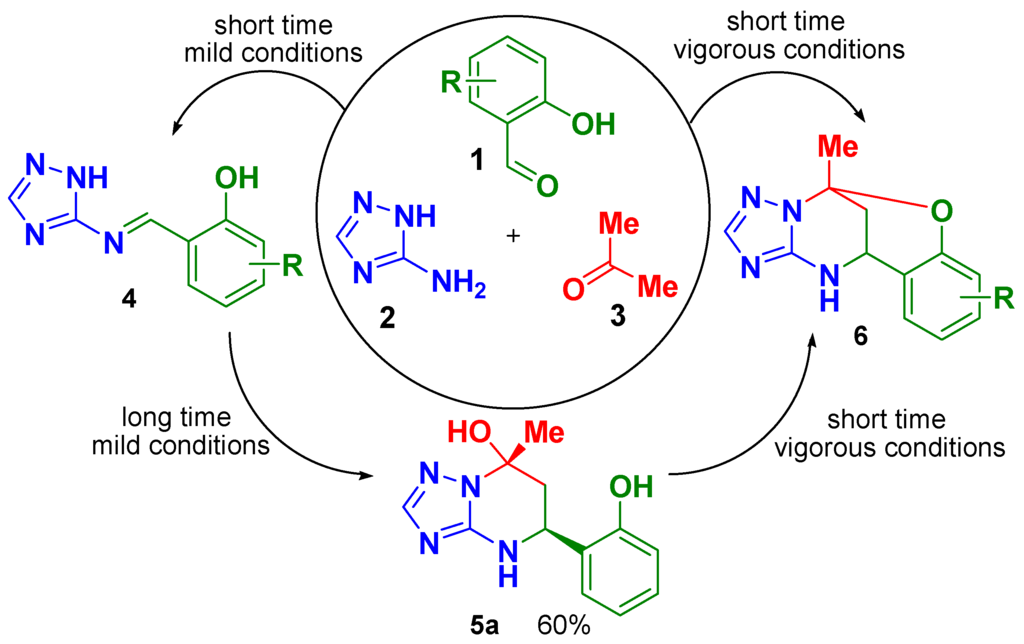
Scheme 1.
Different products from the studied multicomponent reaction (MCR).
Here we aimed to examine the possibility of using different substituted 2-hydroxybenzaldehydes to obtain corresponding products 5b–h with variable substituent R.
2. Results and Discussion
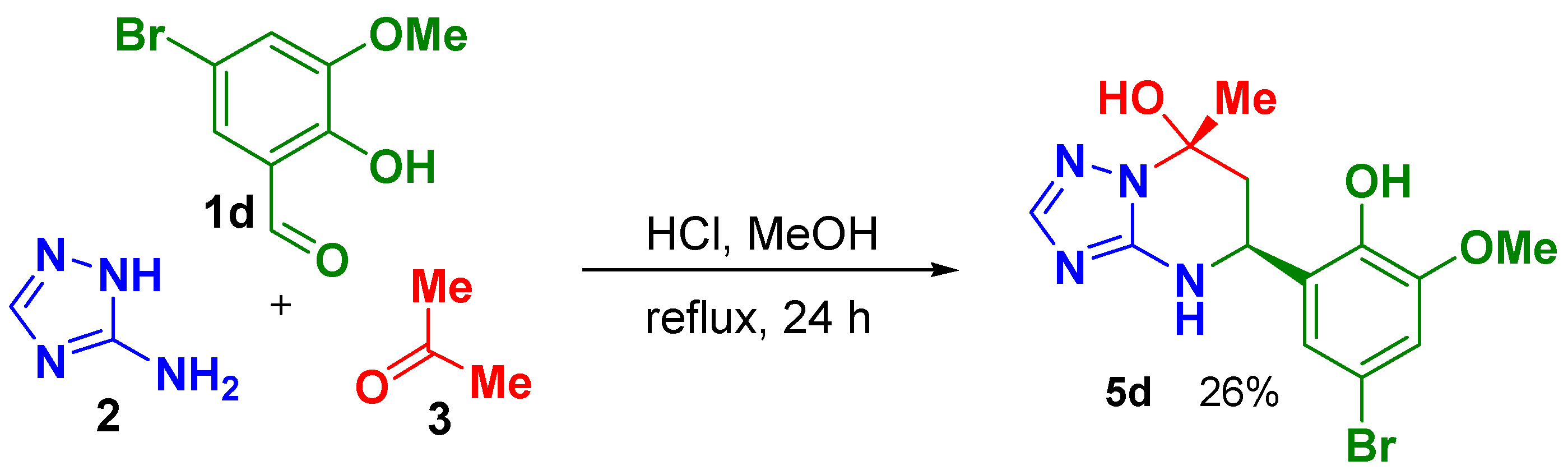
The 2-hydroxybenzaldehydes 1 with following substituents: 3-methoxy-(1b), 4-methoxy-(1c), 5-bromo-3-methoxy-(1d), 3-ethoxy-(1e), 5-bromo-(1f), 3,5-dibromo-(1g) and 4-hydroxy-(1h), were used in this study. In our first attempt, the conditions described in work [5] (40 °C, 16 h in the presence of HCl) were applied for aldehydes 1b–h that resulted in formation of compounds 4 as the main product for all aldehydes as was determined by TLC. Further prolongation of the reaction up to 4 days at 40 °C led to formation of the desired product 5 only in the case of 5-bromo-2-hydroxy -3-methoxybenzaldehyde (1d) but with a very small yield (18%). Reactions with other aldehydes resulted in formation of oxygen-bridged product 6 or its mixture with the desired product 5 and the azamethyne derivatives 4 as determined by 1H-NMR spectra of precipitated products. Variation of reaction conditions, such as application of different solvents and acids (DMF, MeOH, water, acetic acid, trifluoroacetic acid, methylsulfuric acid), application of ultrasonic agitation and microwave irradiation under different temperatures did not improve the results, except for the case of aldehyde 1d, where the desired product 5d was obtained with moderate yield using MeOH and HCl under reflux conditions (Scheme 2).
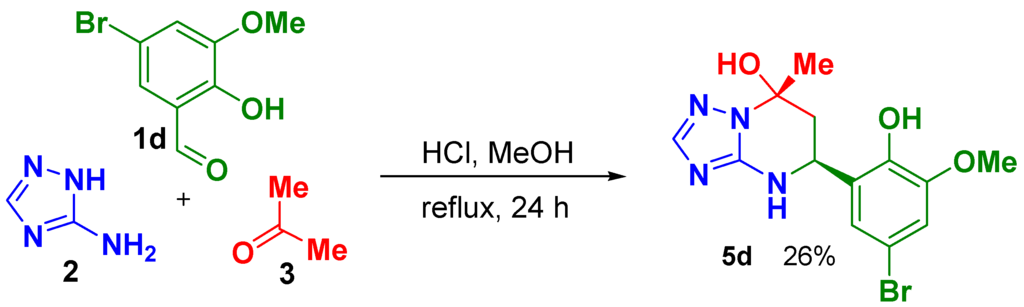
Scheme 2.
Synthesis of 5-(5-Bromo-2-hydroxy-3-methoxyphenyl)-7-methyl-4,5,6,7-tetrahydro[1,2,4]triazolo[1,5-a]pyrimidin-7-ol, (5R,7S)- and (5S,7R)- racemic mixture (5d), relative configuration of stereogenic centers is shown.
The structure of the obtained product 5d was established by 1H-NMR, 13C-NMR, IR spectroscopy and LC-MS. Relative configuration of the stereogenic centers in the molecule is given by analogy with earlier obtained unsubstituted open-bridged compound 5a (R = H) and confirmed by NOE experiments; structure of 5a have been previously proven via single crystal X-ray diffraction study [5] (comparison of the NMR spectra is given in the supplementary materials).
To obtain 5-(5-Bromo-2-hydroxy-3-methoxyphenyl)-7-methyl-4,5,6,7-tetrahydro[1,2,4]triazolo[1,5-a]pyrimidin-7-ol (5d) 10.0 mmol of 3-amino-1,2,4-triazole, 10.0 mmol of 5-bromo-2-hydroxy-3-methoxybenzaldehyde and 60.0 mmol (4.4 mL) of acetone in 25 mL of MeOH containing 625 mg HCl (15% solution in EtOH) were mixed in a round-bottom flask with condenser. The mixture was heated to reflux in oil bath for 24 h. After cooling and standing overnight the precipitate formed was filtered out and washed with 3 × 5 mL of MeOH and dried on air to give a cream-white powder with yield 26%.
Melting Point: 199–201 °C. LC-MS (APCI): Purity 99.3%; m/z: 355 (M + H+, Pos.), 353 (M − H+, Neg.). IR (KBr, cm−1): 3320 (OH), 3150 (CH, aromatic), 2943 (CH, aliphatic), 1634 (C=N), 1536, 1493 (C-C, C=C, NH bend), 1263, 1151, 1085 (C-C, C-O, C-N). 1H-NMR (DMSO-d6, 400 MHz, ppm): δ 9.11 (s, 1H), 7.41 (s, 1H), 7.30 (s, 1H), 7.11–7.17 (m, 1H), 7.03–7.09 (m, 1H), 6.55 (s, 1H), 4.98–5.10 (m, 1H), 3.83 (s, 3H), 2.13–2.22 (m, 1H), 1.76–1.88 (m, 1H), 1.66 (s, 3H). 13C-NMR (DMSO-d6, 100 MHz, ppm): δ 154.0, 148.74, 148.70, 143.2, 130.8, 121.7, 113.9, 110.4, 80.5, 56.7, 44.8, 43.4, 27.1. Elemental Analysis: calcd. for C13H15BrN4O3: C, 43.96; H, 4.26; N, 15.77. Found: C, 43.73; H, 4.34; N, 15.54.
Supplementary Materials
Supplementary File 1Supplementary File 2Supplementary File 3Supplementary File 4The supplementary materials and the molfile can be found at http://www.mdpi.com/1422-8599/2016/2/M898.
Acknowledgments
Authors are thankful to Enamine Ltd (Kyiv, Ukraine), and personally to Maxim A. Nechayev for gratuitous LC-MS analysis of the sample.
Author Contributions
M.K., Y.V.S. and M.K.G. performed all the preliminary studies and the final experiment and analyzed the data, N.Yu.G. and Y.V.S. wrote the paper and composed the supplementary material, S.M.D. acted as the general supervisor.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Dömling, A.; Wang, W.; Wang, K. Chemistry and biology of multicomponent reactions. Chem. Rev. 2012, 112, 3083–3135. [Google Scholar] [CrossRef] [PubMed]
- Ruijter, E.; Scheffelaar, R.; Orru, R.V.A. Multicomponent reaction design in the quest for molecular complexity and diversity. Angew. Chem. Int. Ed. 2011, 50, 6234–6246. [Google Scholar] [CrossRef] [PubMed]
- Van der Heijden, G.; Ruijter, E.; Orru, R.V.A. Efficiency, diversity, and complexity with multicomponent reactions. Synlett 2013, 24, 666–685. [Google Scholar]
- Chebanov, V.A.; Desenko, S.M. Multicomponent heterocyclization reactions with controlled selectivity (review). Chem. Heterocycl. Compd. 2012, 48, 566–583. [Google Scholar] [CrossRef]
- Gorobets, N.Y.; Sedash, Y.V.; Ostras, K.S.; Zaremba, O.V.; Shishkina, S.V.; Baumer, V.N.; Shishkin, O.V.; Kovalenko, S.M.; Desenko, S.M.; van der Eycken, E.V. Unexpected alternative direction of a Biginelli-like multicomponent reaction with 3-amino-1,2,4-triazole as the urea component. Tetrahedron Lett. 2010, 51, 2095–2098. [Google Scholar] [CrossRef]
- Svetlik, J.; Kettmann, V. The chameleon-like behaviour of 3-amino-1,2,4-triazole in the Biginelli reaction: Unexpected formation of a novel spiroheterocyclic system. Tetrahedron Lett. 2011, 52, 1062–1066. [Google Scholar] [CrossRef]
- Sedash, Y.V.; Gorobets, N.Y.; Chebanov, V.A.; Konovalova, I.S.; Shishkin, O.V.; Desenko, S.M. Dotting the i’S in three-component Biginelli-like condensations using 3-amino-1,2,4-triazole as a 1,3-binucleophile. RSC Adv. 2012, 2, 6719–6728. [Google Scholar] [CrossRef]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).