Abstract
A novel compound, methyl 3-(quinolin-2-yl)indolizine-1-carboxylate (2) has been synthesized by cycloaddition reaction of 1-(quinolin-2-ylmethyl)pyridinium ylide (1) with methyl propiolate in presence of sodium hydride in THF. The structure of this compound was established by IR, 1H-NMR, 13C-NMR and MS data.
1. Introduction
Indolizines are aromatic organic compounds containing condensed five and six-membered rings with bridging nitrogen (isomer of indole) [1]. Heterocycles, possessing indolizine core have also found numerous biological and pharmacological activities, such as anti-inflammatory [2,3], antiviral [4], aromatase inhibitory [5], analgestic [6], antitumor [7,8] activities. Some methods for the synthesis of various types of indolizines and benzoindolizines have been well reviewed in the literature, such as the condensation reactions [9], 1,3-dipolar cycloadditions [10,11], and 1,5-dipolar cyclizations [12]. Among these, the 1,3-dipolar cycloaddition of N-pyridinium ylide and related heteroaromatic ylides, e.g., quinolinium or isoquinolinium ylides, with various dipolarophiles, is one of the simplest approaches for the construction of indolizine ring [13,14,15].
In continuation of our research interest in heteroaromatic N-ylide [16,17], we report here our result concerning the reactivity of 1-((quinol-2-yl)methyl) pyridinium ylide toward methyl propiolate as dipolarophile. Indolizine derivative (2) containing a quinoline unit was achieved as a result of this reaction. The position of the ester group on the new heterocyclic ring could not be determined efficiently by NMR spectroscopy. However, it has been established by analogy and by comparison with previous reported compounds [17].
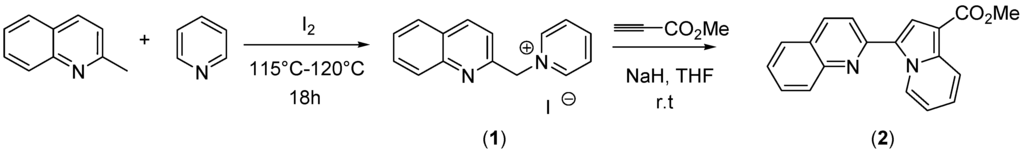
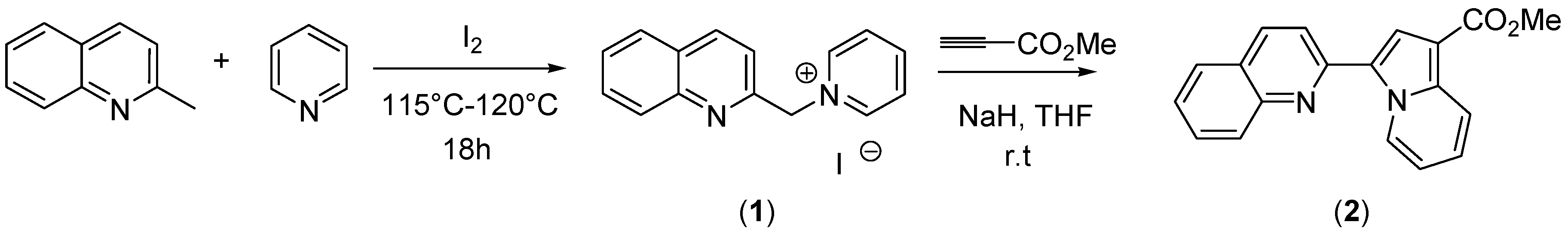
Scheme 1.
The synthesis of methyl 3-(quinolin-2-yl)indolizine-1-carboxylate (2).
Scheme 1.
The synthesis of methyl 3-(quinolin-2-yl)indolizine-1-carboxylate (2).
2. Experimental Section
2.1. General Information
The starting materials were generally used as received (Acros, Fontenay-sous-Bois, France) without any further purification. THF was freshly distilled from sodium/benzophenone. Melting point was determined on an Electrothermal Digital Melting Points Apparatus IA 9200 (Mentouri University, Constantine, Algeria) and is uncorrected. 1H-NMR and 13C-NMR spectra were recorded on Brüker Avance DPX250 spectrometers (Mentouri university, Constantine, Algeria). The purity of the final compound (greater than 95%) was determined by HPLC/MS on an Agilent 1290 system (Lyon 1 University, Lyon, France) using a Agilent 1290 Infinity ZORBAX Eclipse Plus C18 column (2.1 mm × 50 mm, 1.8 μm particle size) with a gradient mobile phase of H2O/CH3CN (90:10, v/v) with 0.1% of formic acid to H2O/CH3CN (10:90, v/v) with 0.1% of formic acid at a flow rate of 0.5 mL/min, with UV monitoring at the wavelength of 254 nm with a run time of 10 min. 1-((Quinol-2-yl)methyl) pyridinium iodide (1) was synthesized following a literature procedure starting from quinaldine and its structure has been confirmed by spectroscopic methods [18].
2.2. Synthesis of Methyl 3-(Quinolin-2-yl)indolizine-1-carboxylate (2)
To a suspension of 50 % sodium hydride dispersion in mineral oil (30 mg, 1.25 mmol) placed in 10 mL of tetrahydrofurane, was added, at 0 °C, 348 mg (1 mmol) of 1-((quinol-2-yl)methyl) pyridinium iodide (1) and 126 mg (1.5 mmol) of methyl propiolate. The ice bath was then removed, and the contents were allowed to cool to room temperature. The reaction mixture was kept, under stirring at room temperature, for 24 h (the progress of the reaction was monitored by TLC). Water was added and the residue was extracted threefold with CH2Cl2 (2 × 20 mL). The organic layers were separated and dried over anhydrous MgSO4. The solvent was removed and the residue was purified by column chromatography on silica gel, using cyclohexane/AcOEt (2/1) as eluent.
Yellow solid; Yield: 40%; m.p. = 164 °C. MS (ES-API): m/z [M + H] = 303.1. Anal. calcd. for C19H14N2O2. 0.09 CHCl3: C 73.24, H 4.54, N 8.95, found: C 73.20, H 4.11, N 8.85. IR(KBr) ν cm−1 : 3838, 2360, 1685, 1211, 740. 1H-NMR δ (ppm) (250 MHz, CDCl3): 10.63 (d, 1H, J = 7.2 Hz), 8.35 (dt, 1H, J = 9.0 Hz, J = 1.2 Hz), 8.10 (d, 2H, J = 8.7 Hz), 7.95 (s, 1H), 7.90–7.68 (m, 3H), 7.49 (td, 1H, J = 8.0 Hz, J = 1.1 Hz), 7.40–7.19 (m, 1H), 6.98 (td, 1H, J = 7.1 Hz, J = 1.4 Hz), 3.97 (s, 3H, COOMe). 13C-NMR δ (ppm) (62.9 MHz, CDCl3): 165.2 (C=O), 151.3, 147.3, 138.6, 136.2, 129.8, 128.9, 128.8, 127.6, 126.2, 125.9, 124.5, 123.6, 119.6, 119.5, 119.4, 113.6, 104.6 and 51.2 (CH3). (see supplementary material for more details, Figures S1–S4).
Supplementary Materials
Supplementary File 1Supplementary File 2Supplementary File 3Supplementary File 4The molfiles and the other supplementary materials can be found at http://www.mdpi.com/1422-8599/2016/1/M883.
Acknowledgements
We are grateful to the Ministère de l’Enseignement Supérieur et de la Recherche Scientifique, Algérie (MESRS) for financial support.
Author Contributions
Roumaissa Belguedj and Esma Lemarahave contributed to the experimental part of this work. Abdelmalek Bouraiou, Zouhair Bouaziz and Aissa Chibani contributed to the Experimental characterization and the preparation of the manuscript. All authors read and approve the final manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Georgescu, E.; Dumitrascu, F.; Georgescu, F.; Draghici, C.; Barbu, L. A Novel Approach for the Synthesis of 5-Pyridylindolizine Derivatives via 2-(2-Pyridyl)pyridinium Ylides. J. Heterocycl. Chem. 2013, 50, 78–82. [Google Scholar] [CrossRef]
- Malonne, H.; Hanuise, J.; Fontaine, J. Topical Anti-inflammatory Activity of New 2-(1-Indolizinyl)propionic Acid Derivatives in Mice. Pharm. Pharmacol. Commun. 1998, 4, 241–242. [Google Scholar]
- Kitadokoro, K.; Hagishita, S.; Sato, T.; Ohtani, M.; Miki, K. Crystal Structure of Human Secretory Phospholipase A2-IIA Complex with the Potent Indolizine Inhibitor 120–1032. J. Biochem. 1998, 123, 619–623. [Google Scholar] [CrossRef] [PubMed]
- Bolle, L.D.; Andrei, G.; Snoeck, R.; Zhang, Y.; Lommel, A.V.; Otto, M.; Bousseau, A.; Roy, C.; Clercq, E.D.; Naesens, L. Potent, selective and cell-mediated inhibition of human herpesvirus 6 at an early stage of viral replication by the non-nucleoside compound CMV423. Biochem. Pharmacol. 2004, 67, 325–336. [Google Scholar] [CrossRef] [PubMed]
- Sonnet, P.; Dallemagne, P.; Guillon, J.; Engueard, C.; Stiebing, S.; Tangue, J.; Bureau, B.; Rault, S.; Auvray, P.; Moslemi, S.; et al. New aromatase inhibitors. Synthesis and biological activity of aryl-substituted pyrrolizine and indolizine derivatives. Bioorg. Med. Chem. 2000, 8, 945–955. [Google Scholar] [CrossRef]
- Campagna, F.; Carotti, A.; Casini, G.; Macripo, M. Synthesis of New Heterocyclic Ring Systems: Indeno[2,1-b]benzo[g]indolizine and indeno[1′,2′:5,4]pyrrolo[2,1-a]phthalazine. Heterocycles 1990, 31, 97–107. [Google Scholar] [CrossRef]
- Bols, M.; Lillelund, V.H.; Jensen, H.H.; Liang, X. Recent Developments of Transition-State Analogue Glycosidase Inhibitors of Non-Natural Product Origin. Chem. Rev. 2002, 102, 515–553. [Google Scholar]
- Asano, N.; Nash, R.J.; Molyneux, R.J.; Fleet, G.W.J. Sugar-mimic glycosidase inhibitors: natural occurrence, biological activity and prospects for therapeutic application. Tetrahedron: Asymmetry 2000, 11, 1645–1680. [Google Scholar] [CrossRef]
- Shipman, M. Indolizines in science of synthesis. In In Fused Five-Membered Hetarenes with One Heteroatom; Thomas, E.J., Ed.; Georg Thieme Verlag: New York, NY, USA, 2000; Volume 10, pp. 745–787. [Google Scholar]
- Agejas, J.; Cuadro, A.M.; Pastor, M.; Vaquero, J.J.; García-Navío, J.L.; Alvarez-Builla, J. N-(Pyridylmethyl)azinium Salts: Precursors of Pyridyl-stabilised Azinium N-Ylides. Tetrahedron 1995, 51, 12425–12438. [Google Scholar] [CrossRef]
- Jaung, J.Y.; Jung, Y.S. 1,3-Dipolar Cycloaddition Reactions of Pyridinium Azomethine Ylides Containing 5,6-Dicyanopyrazines. Bull. Korean Chem. Soc. 2003, 24, 1565–1566. [Google Scholar] [CrossRef]
- Mao, Z.; Li, X.; Lin, X.; Lu, P.; Wang, Y. One-pot multicomponent synthesis of polysubstituted indolizines. Tetrahedron 2012, 68, 85–91. [Google Scholar] [CrossRef]
- Boeklheide, V.; Farenholtz, K. The Formation of Pyrrocolines by the Reaction of Dimethyl Acetylenedicarboxylate with Heterocyclic Zwitterions. J. Am. Chem. Soc. 1961, 83, 458–462. [Google Scholar] [CrossRef]
- Hendrick, C.A.; Ritchie, E.; Taylor, W.C. Pyridinium ylids in synthesis. III. Synthesis of indolizines. Aust. J. Chem. 1967, 20, 2467–2477. [Google Scholar] [CrossRef]
- Padwa, A.; Austin, D.J.; Precedo, L.; Zhi, L. Cycloaddition reactions of pyridinium and related azomethine ylides. J. Org. Chem. 1993, 58, 1144–1150. [Google Scholar] [CrossRef]
- Belguedj, R.; Bouraiou, A.; Bouacida, S.; Merazig, H.; Chibani, A. Pyridinium ylides in the one-pot synthesis of a new quinoline/indolizine hybrid. Z. Naturforsch. 2015, 70, 885–887. [Google Scholar] [CrossRef]
- Belguedj, R.; Bouacida, S.; Merazig, H.; Belfaitah, A.; Bouraiou, A. 1-(2′-Benzimidazolylmethyl)-pyridinium ylide in the one-pot synthesis of indolizine and benzimidazo[1,2-a]pyridine derivatives. Z. Naturforsch. 2015, 70, 555–561. [Google Scholar] [CrossRef]
- Albright, J.D.; Shepherd, R.G. Reactions of 1,2-Dimethyl-5-nitroimidazole, novel methods of conversion of the 2-Methyl group to a nitrile. J. Heterocycl. Chem. 1973, 10, 899–907. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
