Abstract
A simple and novel route for the synthesis of new spirocyclic propionamide derivative is developed. The present work involves N-arylation of pyrazolone (1) using copper(I) iodide catalyst followed by reduction to give amine (2). The coupling of 2 with 3-(4-fluorophenyl)propionic acid and deprotection of Boc group yields the title compound (3).
Introduction
Spirocyclic structures are found in wide range of natural compounds isolated from various sources [1,2]. These compounds play a very important role in many fields like chiral medicine, chiral LCD materials, macromolecule bulking agents and biological pesticides [3,4,5]. In continuation of our work on synthesis of spirocyclic derivatives [6,7], a new spirocyclic amide derivative was synthesized and characterized.
Results and Discussion
The title compound, 3-(4-fluorophenyl)-N-[4-(4-furan-2-yl-1-oxo-2,3,7-triazaspiro[4.5]dec-3-en-2-yl)phenyl]propionamide hydrochloride (3) was prepared by the coupling of tert-butyl 3-(4-aminophenyl)-1-(furan-2-yl)-4-oxo-2,3,7-triazaspiro[4.5]dec-1-ene-7-carboxylate (2) with 3-(4-fluorophenyl)-propionic acid using HATU followed by deprotection of Boc group using HCl in 1,4-dioxane (Scheme 1). The intermediate 2 in turn prepared by N-arylation of tert-butyl 1-(furan-2-yl)-4-oxo-2,3,7-triazaspiro[4.5]dec-1-ene-7-carboxylate (1) using copper(I) iodide catalyst followed by reduction. The final product 3 was well characterized by using NMR, IR and mass spectral data.
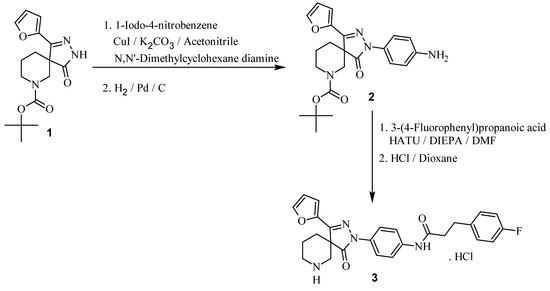
Scheme 1.
Synthesis of 3-(4-fluorophenyl)-N-[4-(4-furan-2-yl-1-oxo-2,3,7-triazaspiro[4.5]dec-3-en-2-yl)phenyl]propionamide hydrochloride, 3.
The IR spectrum of compound 3 showed a broad absorption band at 3,424 cm−1 due to the presence of NH in the molecule. Two sharp bands appeared at 1,697 and 1,603 cm−1 was due to pyrazolone carbonyl and amide carbonyl group respectively. In 1H-NMR spectrum, the signals of the respective protons of the title compound 3 were verified on the basis of their chemical shifts, multiplicities, and coupling constants. A singlet observed at δ 10.16 ppm was due to the amide NH. Another singlet observed at δ 9.85 ppm was due to the piperidine NH which was D2O exchangeable. All the aromatic protons resonated in the region δ 6.8–8.0 ppm. Protons of two methylene groups appeared at δ 2.88 and 2.60 ppm respectively as triplets. The eight protons of the piperidine ring resonated in the region δ 1.8–3.8 ppm as multiplets due to chemical non-equivalence of these protons. The mass spectrum showed a molecular ion peak at m/z 461 corresponding to (M+-HCl) +1 as the HCl gets instantly dissociated in the mass spectral conditions. Elemental analysis and 13C-NMR spectrum also gave satisfactory results for the title compound.
Experimental
Melting point was taken in open capillary tube and was uncorrected. The purity of the compound was confirmed by thin layer chromatography using Merck silica gel 60 F254 coated aluminium plates. IR spectrum was recorded on Shimadzu-FTIR Infrared spectrometer in KBr (νmax in cm−1). 1H-NMR (400 MHz) spectrum was recorded on a Varian 400 spectrometer, with 5 mm PABBO BB-1H TUBES and 13C-NMR (100 MHz) spectrum was recorded for approximately 0.03 M solutions in CD3OD at 100 MHz with TMS as internal standard. All exchangeable protons were confirmed by addition of D2O. LCMS was obtained using Agilent 1200 series LC and Micromass zQ spectrometer. Elemental analysis was carried out by using VARIO EL-III (Elementar Analysensysteme GmBH).
The synthesis of tert-butyl 1-(furan-2-yl)-4-oxo-2,3,7-triazaspiro[4.5]dec-1-ene-7-carboxylate (1) was described in our earlier work [6]. The reaction of 1 with 1-iodo-4-nitrobenzene under copper(I) iodide catalyzed condition followed by the reduction of the nitro compound using Pd/C afforded the intermediate 2 [8].
To a solution of 2 (0.250 g, 0.609 mmol) in DMF (2 mL), DIPEA (0.3 mL, 1.8 mmol) was added at 0–5 °C followed by 3-(4-fluorophenyl)propionic acid (0.102 g, 0.609 mmol). After stirring at 0–5 °C for 15 min, HATU (0.277 g, 0.73 mmol) was added and stirring continued at ambient temperature for 6 h. After the completion of reaction as indicated by TLC, the reaction mixture was quenched into crushed ice and filtered. The solid product was reacted with HCl in 1,4-dioxane [9] to afford the title compound. Yield was 160 mg, 53%.
Melting point: 210–220 °C.
LCMS: m/z = 461, (M+-HCl) + 1.
IR (KBr): νmax (cm−1), 3424 (NH), 1697 (pyrazolone C=O), 1603 (amide C=O), 1535 (C=N), 1222 (C-F).
1H-NMR (400 MHz, DMSO-d6): δ ppm, 1.8 (d, 1H, Piperidine-H, J = 8.4 Hz), 2.1 (d, 1H, Piperidine-H, J = 8.4 Hz), 2.2 (m, 2H, Piperidine-H), 2.6 (t, 2H, CH2, J = 7.6 Hz), 2.88 (t, 2H, CH2, J = 7.6 Hz), 3.2 (m, 3H, Piperidine-H), 3.85 (d, 1H, Piperidine-H, J = 12.8 Hz), 6.8 (m, 1H, Furyl-H), 7.1 (t, 2H, 4-Fluorophenyl-H, J = 8.8 Hz), 7.3 (dd, 2H, 4-Fluorophenyl-H, J = 5.6 Hz, 8.4 Hz), 7.5 (d, 1H, Furyl-H, J = 1.6 Hz), 7.7 (2d, 4H, Aryl-H, J = 9.2 Hz), 7.8 (m, 1H, HCl, D2O exchangeable), 8.0 (d, 1H, Furyl-H, J = 1.6 Hz), 9.85 (s,1H, Piperidine-NH, D2O exchangeable), 10.16 (s, 1H, amide-NH).
13C-NMR (100 MHz, CD3OD): δ ppm, 17.68, 28.82, 44.95, 46.19, 50,10 (Piperidine C’s), 31.92, 39.80, 113.56, 114.11, 115.97, 116.19, 121.14, 121.53, 131.1, 131.17, 134.75, 137.89, 138.13, 146.94 (C=N), 151.31 (Aryl C-F), 173.47 (Pyrazolone C=O), 175.02 (Amide C=O).
Elemental analysis: Calculated for C26H25FN4O3, C, 67.81%; H, 5.47%; N, 12.17%; Found: C, 67.75%; H, 5.44%; N,12.13%.
Supplementary materials
Supplementary File 1Supplementary File 2Supplementary File 3Supplementary File 4Acknowledgments
RS thanks Aurigene Discovery Technologies Limited and Mangalore University for research facilities.
References
- Mead, K.T.; Brewer, B.N. Strategies in spiro ketal synthesis revisited: Recent applications and advances. Curr. Org. Chem. 2003, 7, 227–256. [Google Scholar] [CrossRef]
- Shi, Z.-J.; Zhang, S.-L.; Cao, W.-G.; Deng, H.-M. Facile synthesis of a series of perfluoroalkyl-containing tetra-spirocyclic compounds and their spectral analysis. Chin. J. Chem. 2008, 26, 2103–2106. [Google Scholar] [CrossRef]
- Zhang, Z.-H. Synthesis and application of chiral spiro ligands in asymmetric catalysis. Chin. J. Org. Chem. 2005, 25, 355–363. [Google Scholar]
- Arai, M.A.; Kuraishi, M.; Arai, T.; Sasai, H. A new asymmetric Wacker-type cyclization and tandem cyclization promoted by Pd(II)-spiro bis(isoxazoline) catalyst. J. Am. Chem. Soc. 2001, 123, 2907–2908. [Google Scholar] [CrossRef] [PubMed]
- Pan, C.-Y.; Wang, Y.; William, J.B. The investigation of polymerization mechanism of 7-methylene-2-methyl-1,4,6-triox-aspiro(4,4)nonane. Acta Polym. Sin. 1989, 1, 18–24. [Google Scholar]
- Srinivasan, R.; Narayana, B.; Samshuddin, S.; Sarojini, B.K. tert-Butyl 1-(furan-2-yl)-4-oxo-2,3,7-triazaspiro[4.5]dec-1-ene-7-carboxylate. Molbank 2012, 2012, M757. [Google Scholar] [CrossRef]
- Srinivasan, R.; Narayana, B.; Samshuddin, S.; Sarojini, B.K. N-(3,4-Dimethoxyphenyl)-4-oxo-2,3,7-triazaspiro[4.5]dec-1-ene-1-carboxamide hydrochloride. Molbank 2012, 2012, M769. [Google Scholar] [CrossRef]
- Klapars, A.; Huang, X.; Buchwald, S. A general and efficient copper catalyst for the amidation of aryl halides. J. Am. Chem. Soc. 2002, 124, 7421–7428. [Google Scholar]
- Han, G.; Tamaki, M.; Hruby, V.J. Fast, efficient and selective deprotection of the tert-butoxycarbonyl (Boc) group using HCl/dioxane (4 M). J. Pept. Res. 2001, 58, 338–341. [Google Scholar] [CrossRef] [PubMed]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).