Pathological Mechanisms in Sjögren’s Disease Likely Involve the ADP-Ribosyl Cyclase Family Members: CD38 and CD157
Abstract
1. Introduction
2. Results
2.1. Patients
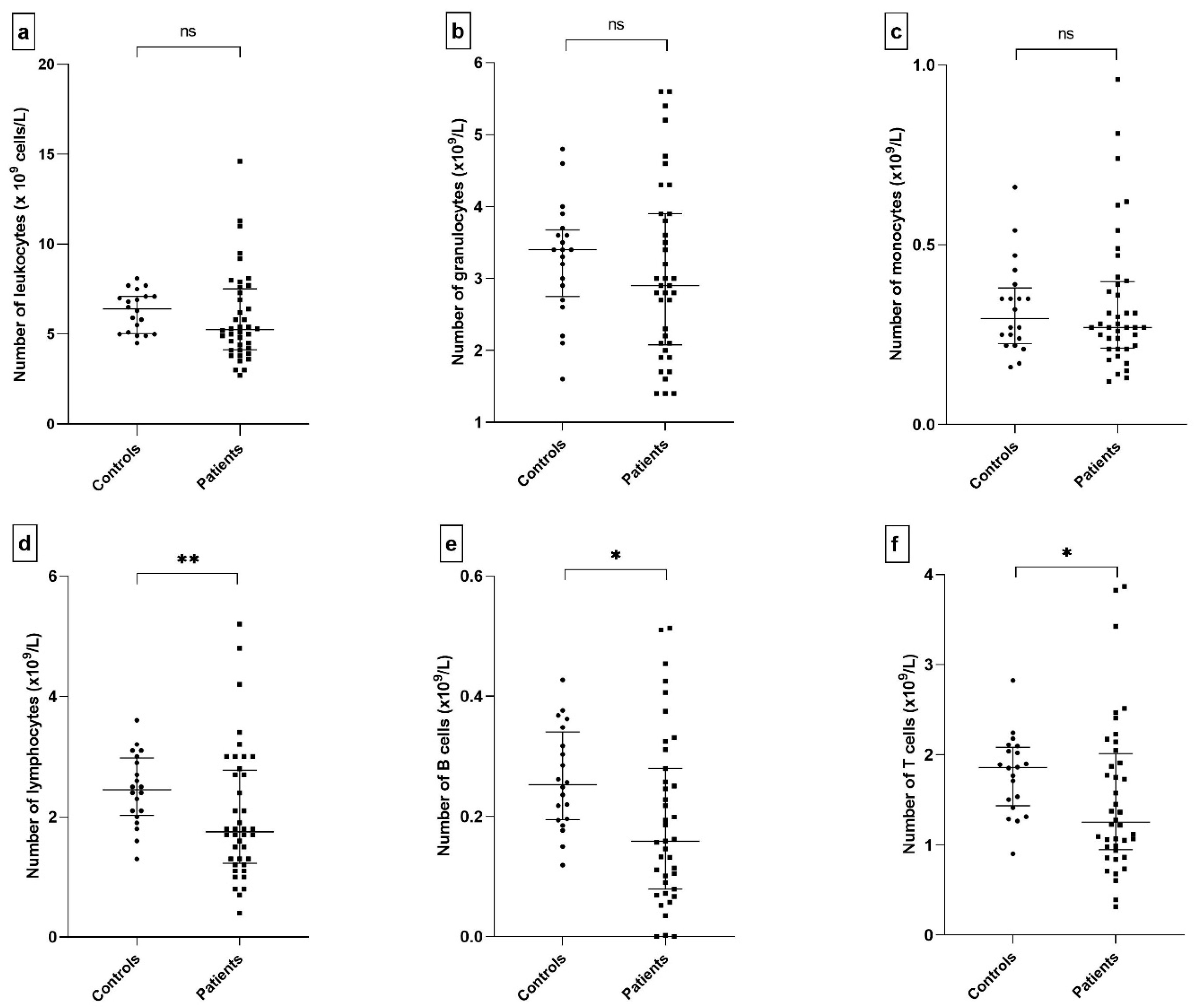
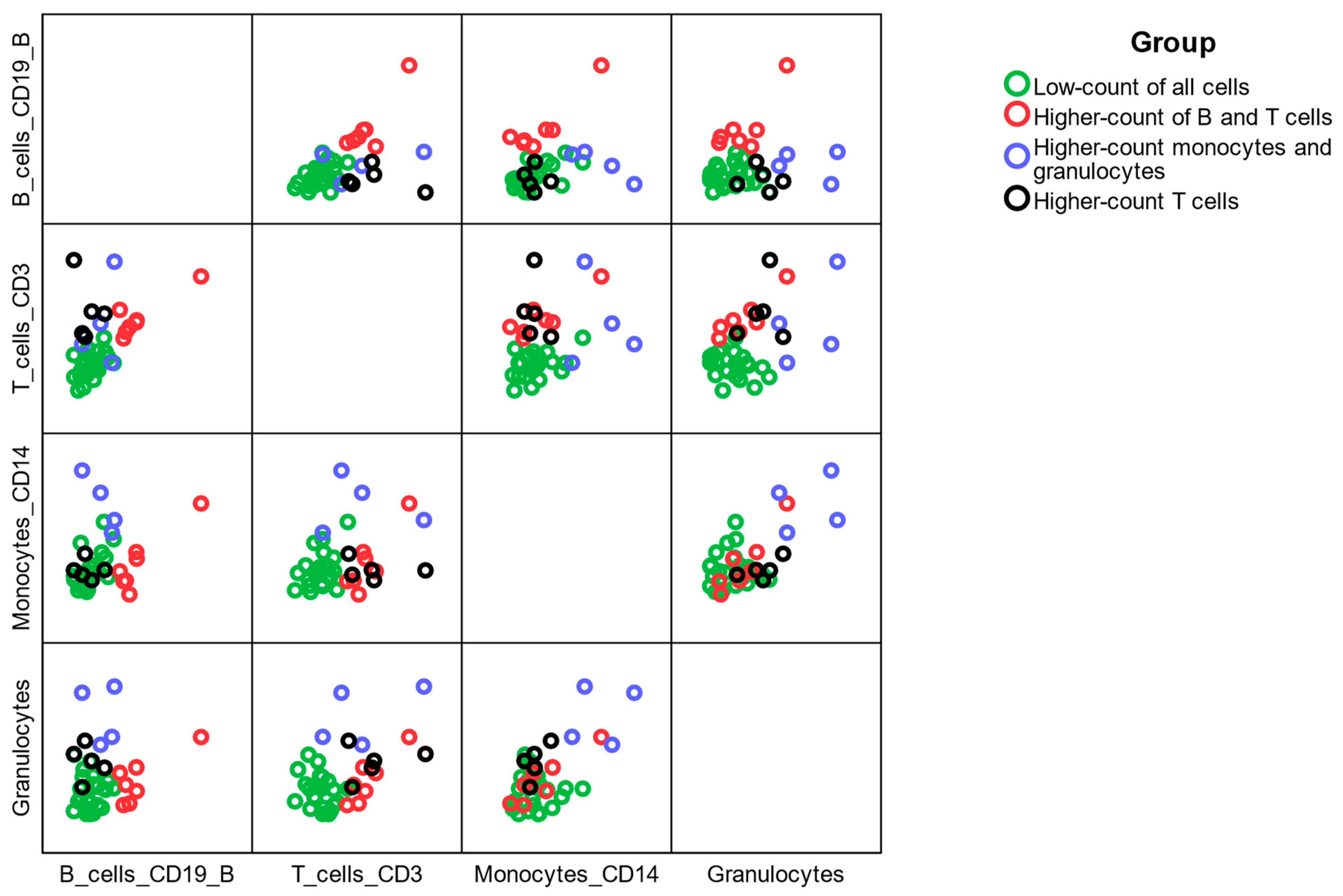
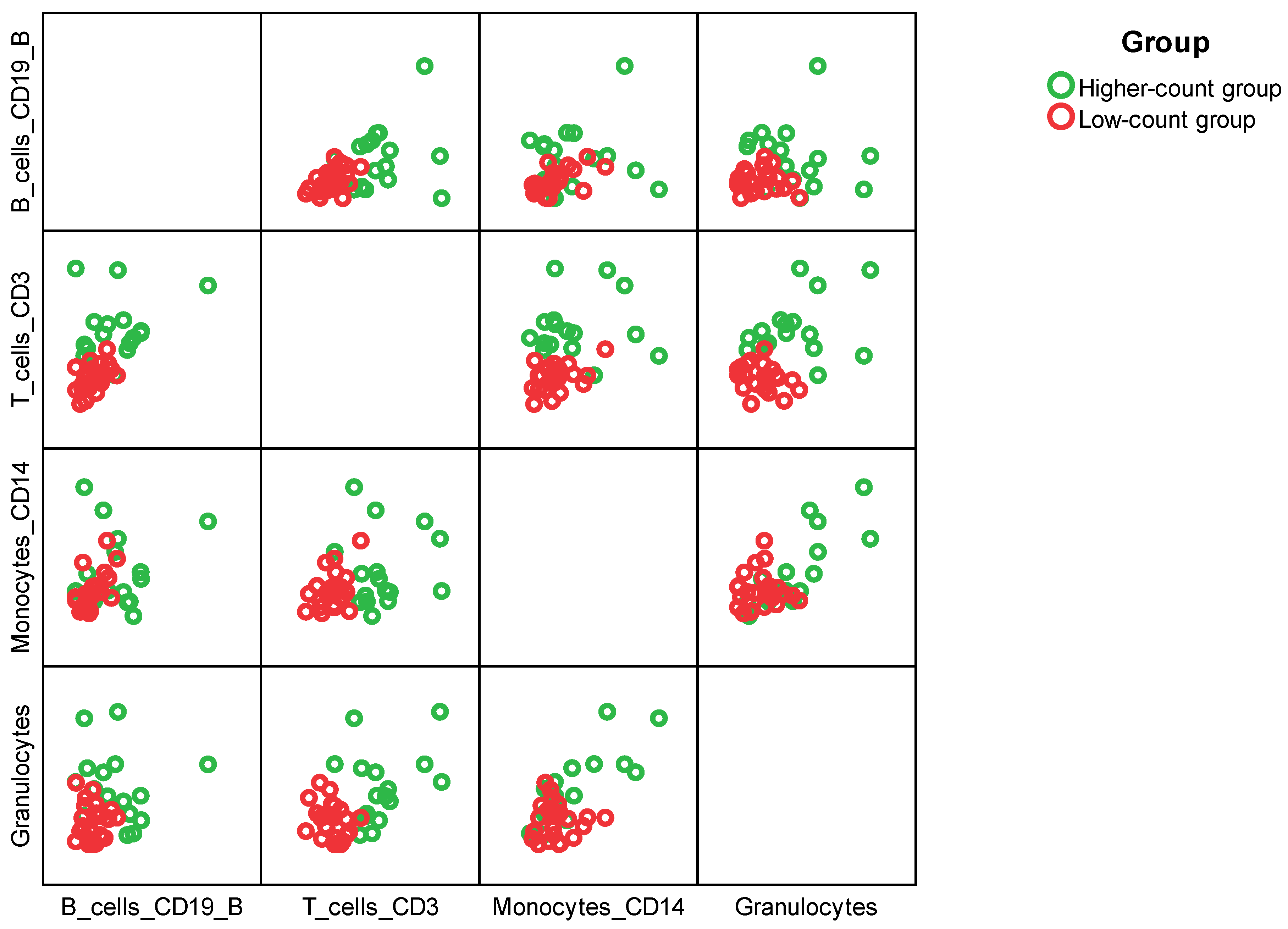
2.2. Leukocytes and Their (Sub)Populations
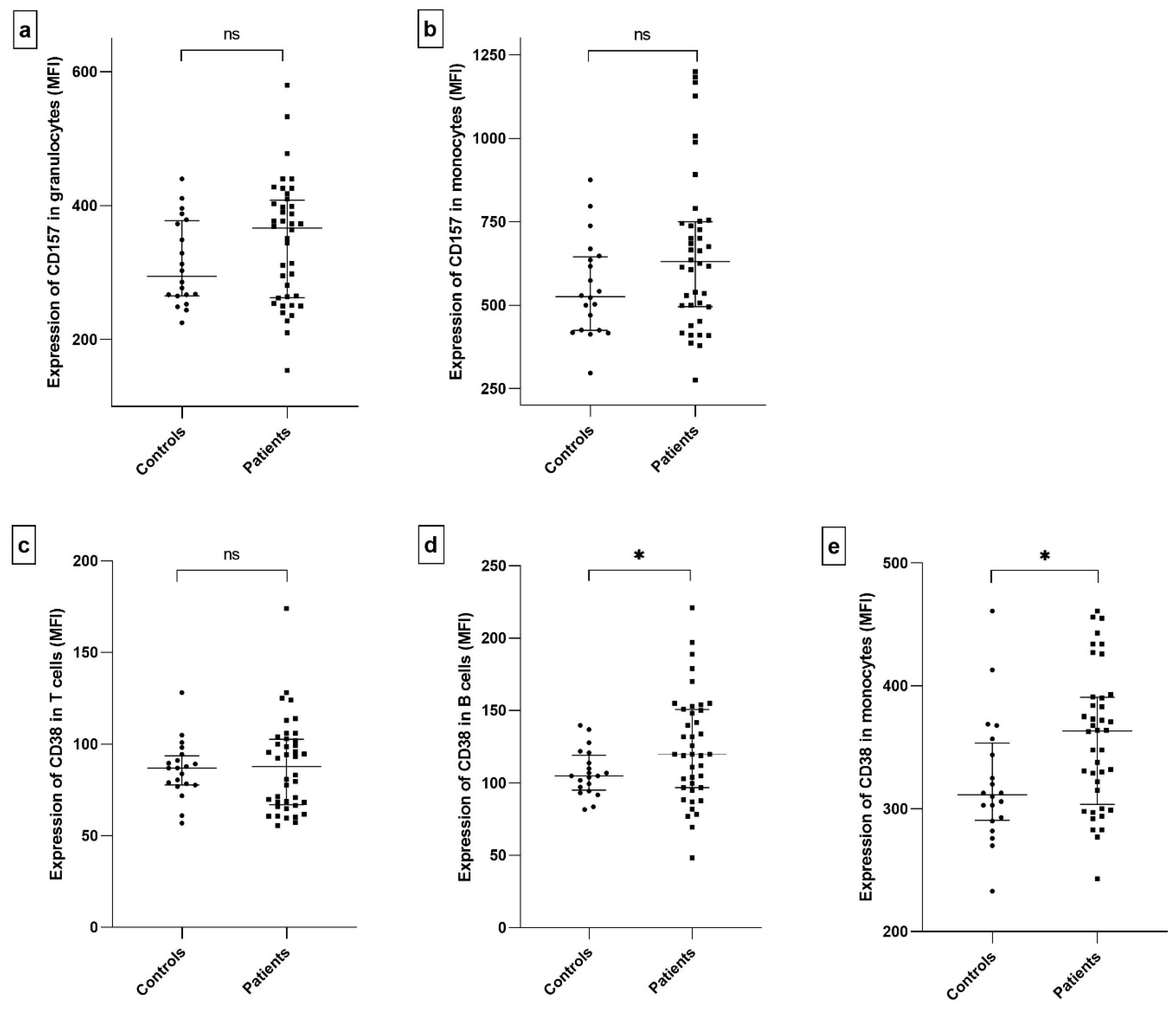
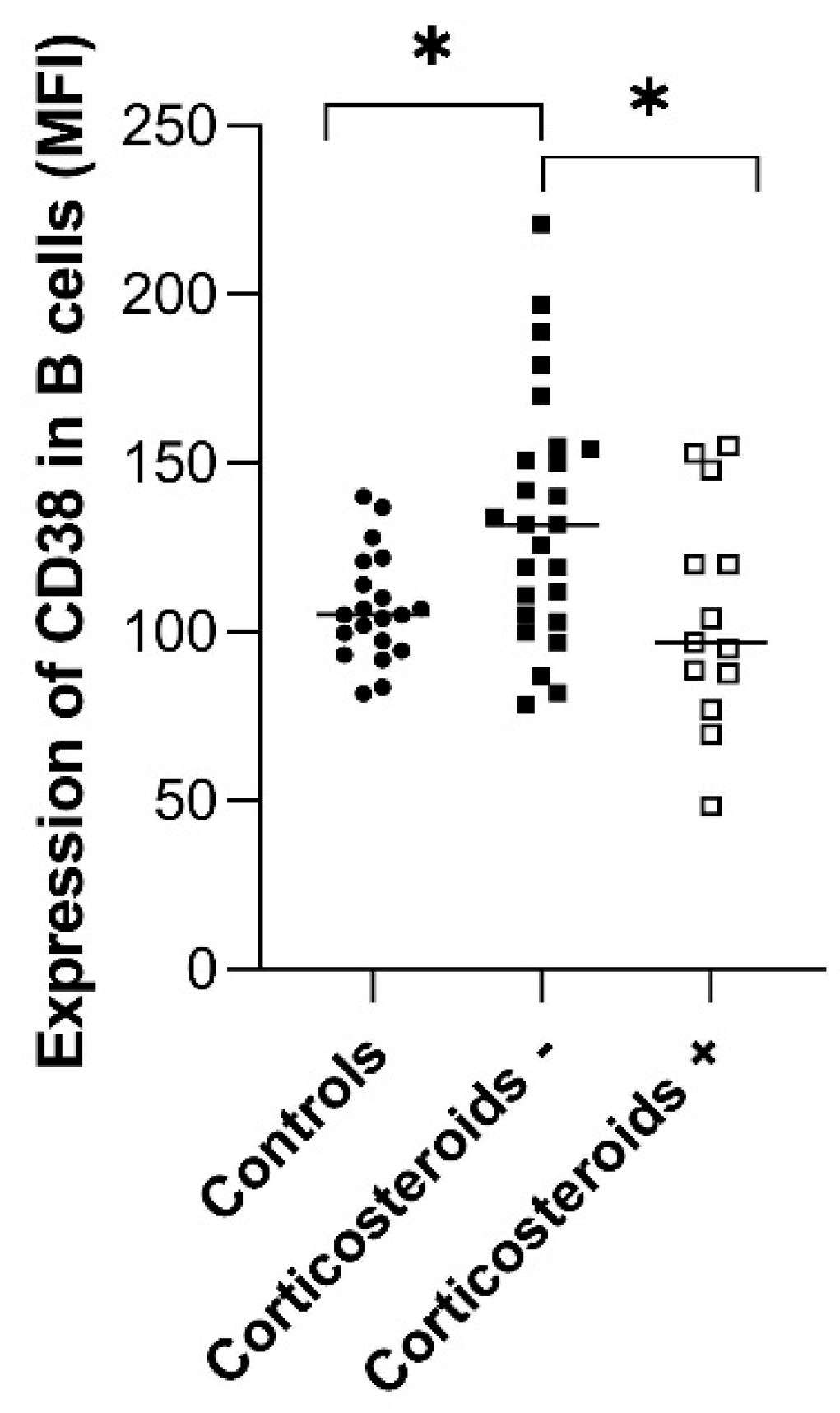
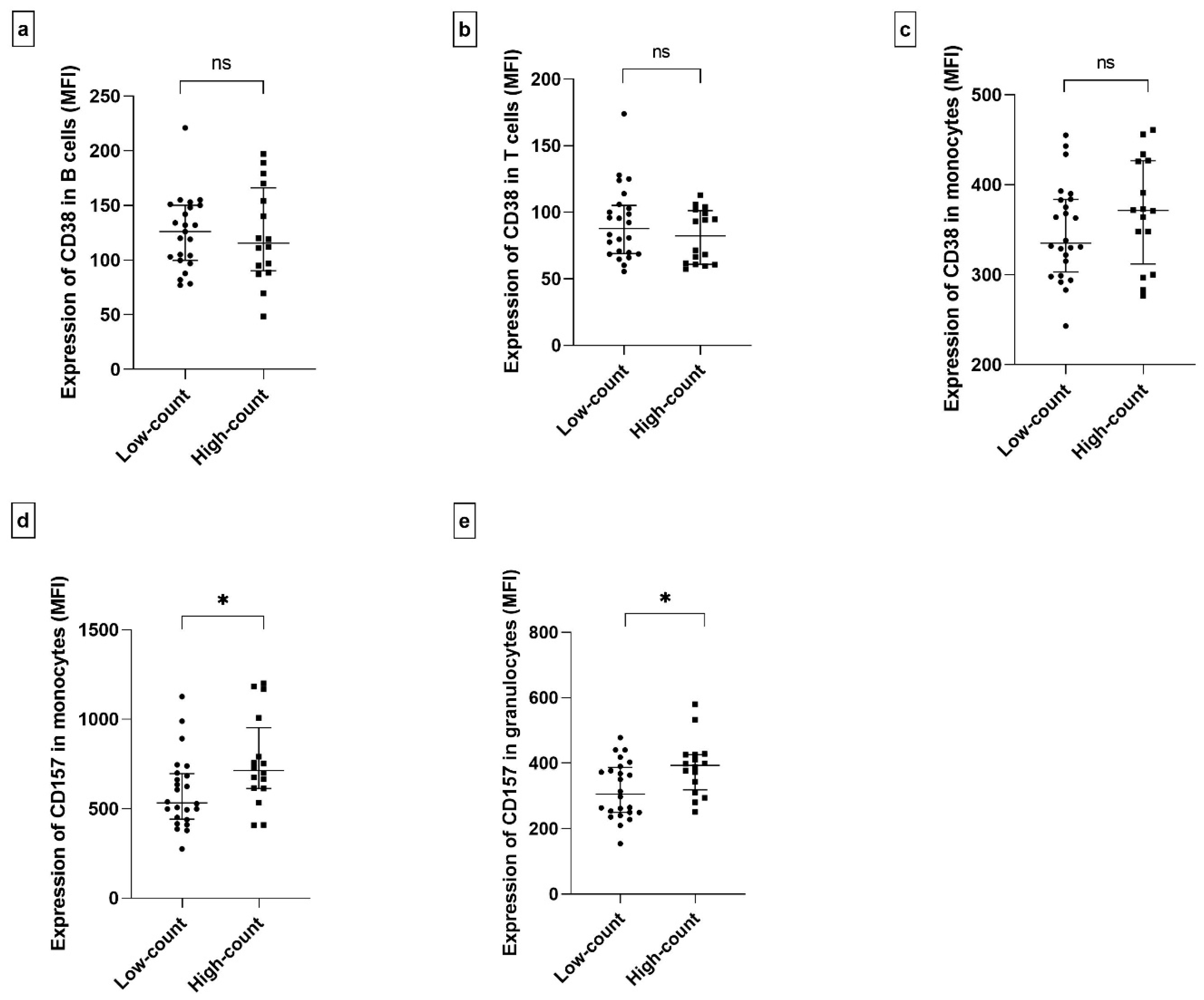
2.3. The Expression of CD157 and CD38 on the Cell Surface
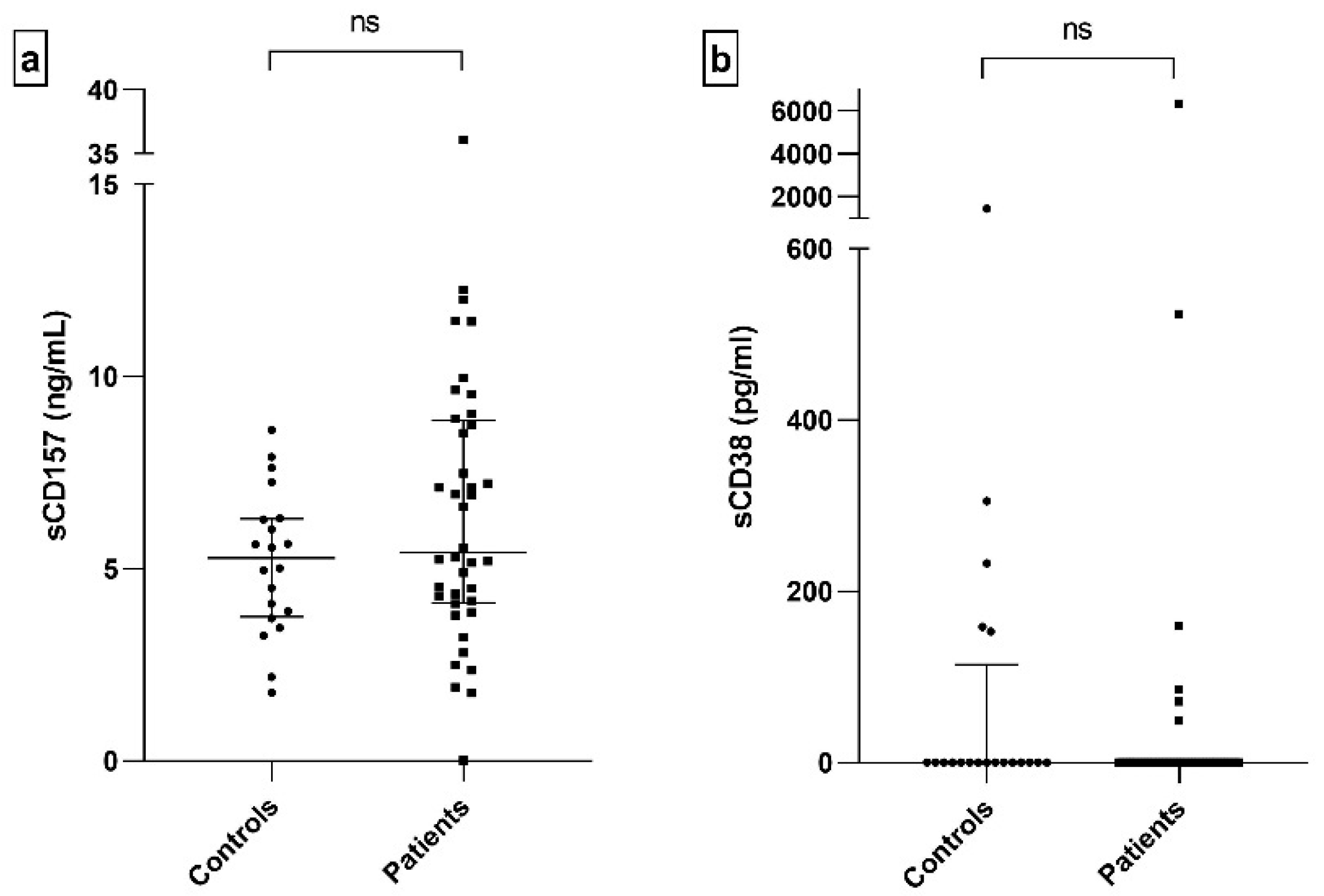
2.4. Serum Level of sCD157 and sCD38
2.5. Hierarchical Clustering
3. Discussion
4. Materials and Methods
4.1. Participants
4.2. Flow Cytometry
4.3. Enzyme-Linked Immunosorbent Assay (ELISA)
4.4. Statistical Evaluation
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Committee on Selected Immune Disorders and Disability; Board on Health Care Services; Health and Medicine Division; National Academies of Sciences, Engineering, and Medicine. Selected Immune Disorders and Disability; National Academies Press: Washington, DC, USA, 2022; p. 26595. ISBN 978-0-309-68949-6. [Google Scholar]
- Dianzani, U.; Funaro, A.; DiFranco, D.; Garbarino, G.; Bragardo, M.; Redoglia, V.; Buonfiglio, D.; De Monte, L.B.; Pileri, A.; Malavasi, F. Interaction between Endothelium and CD4+CD45RA+ Lymphocytes. Role of the Human CD38 Molecule. J. Immunol. 1994, 153, 952–959. [Google Scholar] [CrossRef]
- Li, J.; Li, H.; Cai, S.; Bai, S.; Cai, H.; Zhang, X. CD157 in Bone Marrow Mesenchymal Stem Cells Mediates Mitochondrial Production and Transfer to Improve Neuronal Apoptosis and Functional Recovery after Spinal Cord Injury. Stem Cell Res. Ther. 2021, 12, 289. [Google Scholar] [CrossRef]
- Carafoli, E.; Krebs, J. Why Calcium? How Calcium Became the Best Communicator. J. Biol. Chem. 2016, 291, 20849–20857. [Google Scholar] [CrossRef]
- Malavasi, F.; Deaglio, S.; Funaro, A.; Ferrero, E.; Horenstein, A.L.; Ortolan, E.; Vaisitti, T.; Aydin, S. Evolution and Function of the ADP Ribosyl Cyclase/CD38 Gene Family in Physiology and Pathology. Physiol. Rev. 2008, 88, 841–886. [Google Scholar] [CrossRef] [PubMed]
- Amici, S.A.; Young, N.A.; Narvaez-Miranda, J.; Jablonski, K.A.; Arcos, J.; Rosas, L.; Papenfuss, T.L.; Torrelles, J.B.; Jarjour, W.N.; Guerau-de-Arellano, M. CD38 Is Robustly Induced in Human Macrophages and Monocytes in Inflammatory Conditions. Front. Immunol. 2018, 9, 1593. [Google Scholar] [CrossRef]
- Chini, C.C.S.; Peclat, T.R.; Warner, G.M.; Kashyap, S.; Espindola-Netto, J.M.; De Oliveira, G.C.; Gomez, L.S.; Hogan, K.A.; Tarragó, M.G.; Puranik, A.S.; et al. CD38 Ecto-Enzyme in Immune Cells Is Induced during Aging and Regulates NAD+ and NMN Levels. Nat. Metab. 2020, 2, 1284–1304. [Google Scholar] [CrossRef] [PubMed]
- Camacho-Pereira, J.; Tarragó, M.G.; Chini, C.C.S.; Nin, V.; Escande, C.; Warner, G.M.; Puranik, A.S.; Schoon, R.A.; Reid, J.M.; Galina, A.; et al. CD38 Dictates Age-Related NAD Decline and Mitochondrial Dysfunction through a SIRT3-Dependent Mechanism. Cell Metab. 2016, 23, 1127–1139. [Google Scholar] [CrossRef]
- Ruskovska, T.; Bernlohr, D.A. The Role of NAD+ in Metabolic Regulation of Adipose Tissue: Implications for Obesity-Induced Insulin Resistance. Biomedicines 2023, 11, 2560. [Google Scholar] [CrossRef] [PubMed]
- Santiago-Cruz, W.; García-García, F. T Lymphocytes in Aging: CD38 as a Novel Contributor between Inflammaging and Immunosenescence. J. Cell. Immunol. 2024, 6, 148–162. [Google Scholar] [CrossRef]
- Lauritzen, K.H.; Yang, K.; Frisk, M.; Louwe, M.C.; Olsen, M.B.; Ziegler, M.; Louch, W.E.; Halvorsen, B.; Aukrust, P.; Yndestad, A.; et al. Apigenin Inhibits NLRP3 Inflammasome Activation in Monocytes and Macrophages Independently of CD38. Front. Immunol. 2025, 15, 1497984. [Google Scholar] [CrossRef]
- Wen, S.; Arakawa, H.; Yokoyama, S.; Shirasaka, Y.; Higashida, H.; Tamai, I. Functional Identification of Soluble Uric Acid as an Endogenous Inhibitor of CD38. eLife 2024, 13, RP96962. [Google Scholar] [CrossRef]
- Tarragó, M.G.; Chini, C.C.S.; Kanamori, K.S.; Warner, G.M.; Caride, A.; de Oliveira, G.C.; Rud, M.; Samani, A.; Hein, K.Z.; Huang, R.; et al. A Potent and Specific CD38 Inhibitor Ameliorates Age-Related Metabolic Dysfunction by Reversing Tissue NAD+ Decline. Cell Metab. 2018, 27, 1081–1095.e10. [Google Scholar] [CrossRef]
- Bagavant, H.; Durslewicz, J.; Pyclik, M.; Makuch, M.; Papinska, J.A.; Deshmukh, U.S. Age-Associated B Cell Infiltration in Salivary Glands Represents a Hallmark of Sjögren’s-like Disease in Aging Mice. GeroScience 2024, 46, 6085–6099. [Google Scholar] [CrossRef]
- Jackson, D.G.; Bell, J.I. Isolation of a cDNA Encoding the Human CD38 (T10) Molecule, a Cell Surface Glycoprotein with an Unusual Discontinuous Pattern of Expression during Lymphocyte Differentiation. J. Immunol. 1990, 144, 2811–2815. [Google Scholar] [CrossRef]
- Itoh, M.; Ishihara, K.; Tomizawa, H.; Tanaka, H.; Kobune, Y.; Ishikawa, J.; Kaisho, T.; Hirano, T. Molecular Cloning of Murine BST-1 Having Homology with CD38 and Aplysia ADP-Ribosyl Cyclase. Biochem. Biophys. Res. Commun. 1994, 203, 1309–1317. [Google Scholar] [CrossRef]
- Kaisho, T.; Ishikawa, J.; Oritani, K.; Inazawa, J.; Tomizawa, H.; Muraoka, O.; Ochi, T.; Hirano, T. BST-1, a Surface Molecule of Bone Marrow Stromal Cell Lines That Facilitates Pre-B-Cell Growth. Proc. Natl. Acad. Sci. USA 1994, 91, 5325–5329. [Google Scholar] [CrossRef] [PubMed]
- Morone, S.; Augeri, S.; Cuccioloni, M.; Mozzicafreddo, M.; Angeletti, M.; Lo Buono, N.; Giacomino, A.; Ortolan, E.; Funaro, A. Binding of CD157 Protein to Fibronectin Regulates Cell Adhesion and Spreading. J. Biol. Chem. 2014, 289, 15588–15601. [Google Scholar] [CrossRef] [PubMed]
- Partida-Sánchez, S.; Goodrich, S.; Kusser, K.; Oppenheimer, N.; Randall, T.D.; Lund, F.E. Regulation of Dendritic Cell Trafficking by the ADP-Ribosyl Cyclase CD38: Impact on the Development of Humoral Immunity. Immunity 2004, 20, 279–291. [Google Scholar] [CrossRef] [PubMed]
- Funaro, A.; Ortolan, E.; Ferranti, B.; Gargiulo, L.; Notaro, R.; Luzzatto, L.; Malavasi, F. CD157 Is an Important Mediator of Neutrophil Adhesion and Migration. Blood 2004, 104, 4269–4278. [Google Scholar] [CrossRef]
- Lo Buono, N.; Parrotta, R.; Morone, S.; Bovino, P.; Nacci, G.; Ortolan, E.; Horenstein, A.L.; Inzhutova, A.; Ferrero, E.; Funaro, A. The CD157-Integrin Partnership Controls Transendothelial Migration and Adhesion of Human Monocytes. J. Biol. Chem. 2011, 286, 18681–18691. [Google Scholar] [CrossRef]
- Deaglio, S.; Morra, M.; Mallone, R.; Ausiello, C.M.; Prager, E.; Garbarino, G.; Dianzani, U.; Stockinger, H.; Malavasi, F. Human CD38 (ADP-Ribosyl Cyclase) Is a Counter-Receptor of CD31, an Ig Superfamily Member1. J. Immunol. 1998, 160, 395–402. [Google Scholar] [CrossRef]
- Zubiaur, M.; Fernández, O.; Ferrero, E.; Salmerón, J.; Malissen, B.; Malavasi, F.; Sancho, J. CD38 Is Associated with Lipid Rafts and upon Receptor Stimulation Leads to Akt/Protein Kinase B and Erk Activation in the Absence of the CD3-ζ Immune Receptor Tyrosine-Based Activation Motifs*. J. Biol. Chem. 2002, 277, 13–22. [Google Scholar] [CrossRef]
- Liao, S.; Liang, L.; Yue, C.; He, J.; He, Z.; Jin, X.; Luo, G.; Zhou, Y. CD38 Is Involved in Cell Energy Metabolism via Activating the PI3K/AKT/mTOR Signaling Pathway in Cervical Cancer Cells. Int. J. Oncol. 2020, 57, 338–354. [Google Scholar] [CrossRef]
- Lund, F.E.; Muller-Steffner, H.M.; Yu, N.; Stout, C.D.; Schuber, F.; Howard, M.C. CD38 Signaling in B Lymphocytes Is Controlled by Its Ectodomain but Occurs Independently of Enzymatically Generated ADP-Ribose or Cyclic ADP-Ribose1. J. Immunol. 1999, 162, 2693–2702. [Google Scholar] [CrossRef] [PubMed]
- Fumaro, A.; Horenstein, A.L.; Calosso, L.; Morra, M.; Tarocco, R.P.; Franco, L.; Flora, A.D.; Malavasi, F. Identification and Characterization of an Active Soluble Form of Human CD38 in Normal and Pathological Fluids. Int. Immunol. 1996, 8, 1643–1650. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.O.; Ishihara, K.; Denno, K.; Kobune, Y.; Itoh, M.; Muraoka, O.; Kaisho, T.; Sasaki, T.; Ochi, T.; Hirano, T. Elevated levels of the soluble form of bone marrow stromal cell antigen 1 in the sera of patients with severe rheumatoid arthritis. Arthritis Rheum. 1996, 39, 629–637. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Kriksunov, I.A.; Graeff, R.; Munshi, C.; Lee, H.C.; Hao, Q. Crystal Structure of Human CD38 Extracellular Domain. Structure 2005, 13, 1331–1339. [Google Scholar] [CrossRef]
- Lebedev, M.J.; Egorova, N.I.; Sholkina, M.N.; Vilkov, S.A.; Baryshnikov, A.J.; Novikov, V.V. Serum Levels of Different Forms of Soluble CD38 Antigen in Burned Patients. Burns 2004, 30, 552–556. [Google Scholar] [CrossRef]
- Augeri, S.; Capano, S.; Morone, S.; Fissolo, G.; Giacomino, A.; Peola, S.; Drace, Z.; Rapa, I.; Novello, S.; Volante, M.; et al. Soluble CD157 in Pleural Effusions: A Complementary Tool for the Diagnosis of Malignant Mesothelioma. Oncotarget 2018, 9, 22785–22801. [Google Scholar] [CrossRef][Green Version]
- Wallace, D.; Church, J.; Vivino, F.; Lee, C.; Makara, M.; Bowman, S.; Fisher, B.; Fox, R.; Carsons, S.; Wilson, J.; et al. The Sjögren’s Book; Oxford University Press: Oxford, UK, 2022. [Google Scholar] [CrossRef]
- Jonsson, R.; Brokstad, K.A.; Jonsson, M.V.; Delaleu, N.; Skarstein, K. Current Concepts on Sjögren’s Syndrome—Classification Criteria and Biomarkers. Eur. J. Oral Sci. 2018, 126, 37–48. [Google Scholar] [CrossRef]
- Adamson, T.C., 3rd; Fox, R.I.; Frisman, D.M.; Howell, F.V. Immunohistologic Analysis of Lymphoid Infiltrates in Primary Sjogren’s Syndrome Using Monoclonal Antibodies. J. Immunol. 1983, 130, 203–208. [Google Scholar] [CrossRef] [PubMed]
- Christodoulou, M.I.; Kapsogeorgou, E.K.; Moutsopoulos, H.M. Characteristics of the Minor Salivary Gland Infiltrates in Sjögren’s Syndrome. J. Autoimmun. 2010, 34, 400–407. [Google Scholar] [CrossRef]
- Szyszko, E.A.; Brokstad, K.A.; Øijordsbakken, G.; Jonsson, M.V.; Jonsson, R.; Skarstein, K. Salivary Glands of Primary Sjögren’s Syndrome Patients Express Factors Vital for Plasma Cell Survival. Arthritis Res. Ther. 2011, 13, R2. [Google Scholar] [CrossRef] [PubMed]
- Lampert, I.A.; Van Noorden, S.; Wotherspoon, A.C. Centrocytoid Plasma Cells of the Germinal Center. Appl. Immunohistochem. Mol. Morphol. 2005, 13, 124–131. [Google Scholar] [CrossRef] [PubMed]
- Salomonsson, S.; Jonsson, M.V.; Skarstein, K.; Brokstad, K.A.; Hjelmström, P.; Wahren-Herlenius, M.; Jonsson, R. Cellular Basis of Ectopic Germinal Center Formation and Autoantibody Production in the Target Organ of Patients with Sjögren’s Syndrome. Arthritis Rheum. 2003, 48, 3187–3201. [Google Scholar] [CrossRef]
- Zuo, J.; Williams, A.E.G.; Park, Y.-J.; Choi, K.; Chan, A.L.; Reeves, W.H.; Bubb, M.R.; Lee, Y.J.; Park, K.; Stewart, C.M.; et al. Muscarinic Type 3 Receptor Autoantibodies Are Associated with Anti-SSA/Ro Autoantibodies in Sjögren’s Syndrome. J. Immunol. Methods 2016, 437, 28–36. [Google Scholar] [CrossRef]
- Takeshita, M.; Suzuki, K.; Kaneda, Y.; Yamane, H.; Ikeura, K.; Sato, H.; Kato, S.; Tsunoda, K.; Arase, H.; Takeuchi, T. Antigen-Driven Selection of Antibodies against SSA, SSB and the Centromere ‘Complex’, Including a Novel Antigen, MIS12 Complex, in Human Salivary Glands. Ann. Rheum. Dis. 2020, 79, 150–158. [Google Scholar] [CrossRef]
- Ahmed, S.S.; Tan, F.K.; Arnett, F.C.; Jin, L.; Geng, Y.-J. Induction of Apoptosis and Fibrillin 1 Expression in Human Dermal Endothelial Cells by Scleroderma Sera Containing Anti–Endothelial Cell Antibodies. Arthritis Rheum. 2006, 54, 2250–2262. [Google Scholar] [CrossRef]
- Amft, N.; Curnow, S.J.; Scheel-Toellner, D.; Devadas, A.; Oates, J.; Crocker, J.; Hamburger, J.; Ainsworth, J.; Mathews, J.; Salmon, M.; et al. Ectopic Expression of the B Cell-Attracting Chemokine BCA-1 (CXCL13) on Endothelial Cells and within Lymphoid Follicles Contributes to the Establishment of Germinal Center-like Structures in Sjögren’s Syndrome. Arthritis Rheum. 2001, 44, 2633–2641. [Google Scholar] [CrossRef]
- Sène, D.; Ismael, S.; Forien, M.; Charlotte, F.; Kaci, R.; Cacoub, P.; Diallo, A.; Dieudé, P.; Lioté, F. Ectopic Germinal Center-Like Structures in Minor Salivary Gland Biopsy Tissue Predict Lymphoma Occurrence in Patients with Primary Sjögren’s Syndrome. Arthritis Rheumatol. 2018, 70, 1481–1488. [Google Scholar] [CrossRef]
- Park, H.-S.; Martínez-Martínez, L.; Magallares López, B.; Castellví, I.; Moya, P.; Codes-Mendez, H.; Hernandez Sosa, N.; Diaz-Torne, C.; Laiz, A.; Sainz, L.; et al. Prognostic Significance of Lymphocytic Foci Composition in Minor Salivary Gland Biopsies for Severe Disease Flare and Severity in Sjögren’s Syndrome: A 3-Year Follow-up Cohort Study. Front. Immunol. 2024, 15, 1332924. [Google Scholar] [CrossRef]
- Gurlevik, U.; Karakoyun, A.; Yasar, E. Does Sjogren’s Syndrome Affect Only the Lacrimal Gland in the Eye? Time to Replace the Missing Stones. Indian J. Ophthalmol. 2021, 69, 53–57. [Google Scholar] [CrossRef]
- Budden, A.K.; te West, N.I.D.; Sturgess, A.D.; Moore, K.H. Pelvic Floor Dysfunction in Female Sjögren’s Syndrome: An 8-Year Audit. Int. Urogynecol. J. 2016, 27, 1367–1373. [Google Scholar] [CrossRef] [PubMed]
- Katayama, I.; Yokozeki, H.; Nishioka, K. Impaired Sweating as an Exocrine Manifestation in Sjögren’s Syndrome. Br. J. Dermatol. 1995, 133, 716–720. [Google Scholar] [CrossRef] [PubMed]
- Stojan, G.; Baer, A.N.; Danoff, S.K. Pulmonary Manifestations of Sjögren’s Syndrome. Curr. Allergy Asthma Rep. 2013, 13, 354–360. [Google Scholar] [CrossRef] [PubMed]
- Nishimori, I.; Yamamoto, Y.; Okazaki, K.; Morita, M.; Onodera, M.; Kino, J.; Tamura, S.; Yamamoto, Y. Identification of Autoantibodies to a Pancreatic Antigen in Patients with Idiopathic Chronic Pancreatitis and Sjögren’s Syndrome. Pancreas 1994, 9, 374–381. [Google Scholar] [CrossRef]
- Mihai, A.; Caruntu, C.; Jurcut, C.; Blajut, F.C.; Casian, M.; Opris-Belinski, D.; Ionescu, R.; Caruntu, A. The Spectrum of Extraglandular Manifestations in Primary Sjögren’s Syndrome. J. Pers. Med. 2023, 13, 961. [Google Scholar] [CrossRef]
- Nocturne, G.; Marmontel, O.; di Filippo, M.; Chretien, P.; Krzysiek, R.; Lifermann, F.; Rahal, N.; Belkhir, R.; Moulin, P.; Mariette, X. Efficacy of Daratumumab in Refractory Primary Sjögren Disease. RMD Open 2023, 9, e003464. [Google Scholar] [CrossRef]
- Hirata, Y.; Kimura, N.; Sato, K.; Ohsugi, Y.; Takasawa, S.; Okamoto, H.; Ishikawa, J.; Kaisho, T.; Ishihara, K.; Hirano, T. ADP Ribosyl Cyclase Activity of a Novel Bone Marrow Stromal Cell Surface Molecule, BST-1. FEBS Lett. 1994, 356, 244–248. [Google Scholar] [CrossRef]
- Yamamoto-Katayama, S.; Ariyoshi, M.; Ishihara, K.; Hirano, T.; Jingami, H.; Morikawa, K. Crystallographic Studies on Human BST-1/CD157 with ADP-Ribosyl Cyclase and NAD Glycohydrolase Activities 1. J. Mol. Biol. 2002, 316, 711–723. [Google Scholar] [CrossRef]
- Matsui, Y.; Muramatsu, F.; Nakamura, H.; Noda, Y.; Matsumoto, K.; Kishima, H.; Takakura, N. Brain-Derived Endothelial Cells Are Neuroprotective in a Chronic Cerebral Hypoperfusion Mouse Model. Commun. Biol. 2024, 7, 338. [Google Scholar] [CrossRef]
- Higashida, H.; Liang, M.; Yoshihara, T.; Akther, S.; Fakhrul, A.; Stanislav, C.; Nam, T.-S.; Kim, U.-H.; Kasai, S.; Nishimura, T.; et al. An Immunohistochemical, Enzymatic, and Behavioral Study of CD157/BST-1 as a Neuroregulator. BMC Neurosci. 2017, 18, 35. [Google Scholar] [CrossRef]
- Yakymiv, Y.; Augeri, S.; Bracci, C.; Marchisio, S.; Aydin, S.; D’Ardia, S.; Massaia, M.; Ferrero, E.; Ortolan, E.; Funaro, A. CD157 Signaling Promotes Survival of Acute Myeloid Leukemia Cells and Modulates Sensitivity to Cytarabine through Regulation of Anti-Apoptotic Mcl-1. Sci. Rep. 2021, 11, 21230. [Google Scholar] [CrossRef]
- Tang, Y.; Zhou, Y.; Wang, X.; Che, N.; Tian, J.; Man, K.; Rui, K.; Peng, N.; Lu, L. The Role of Epithelial Cells in the Immunopathogenesis of Sjögren’s Syndrome. J. Leukoc. Biol. 2024, 115, 57–67. [Google Scholar] [CrossRef] [PubMed]
- Rivière, E.; Pascaud, J.; Tchitchek, N.; Boudaoud, S.; Paoletti, A.; Ly, B.; Dupré, A.; Chen, H.; Thai, A.; Allaire, N.; et al. Salivary Gland Epithelial Cells from Patients with Sjögren’s Syndrome Induce B-Lymphocyte Survival and Activation. Ann. Rheum. Dis. 2020, 79, 1468–1477. [Google Scholar] [CrossRef] [PubMed]
- Morva, A.; Kapsogeorgou, E.K.; Konsta, O.D.; Moutsopoulos, H.M.; Tzioufas, A.G. OP0079 Salivary Gland Epithelial Cells (SGECS) Promote the Differentiation of B Cells. Ann. Rheum. Dis. 2013, 72, A77. [Google Scholar] [CrossRef]
- Wildenberg, M.E.; Welzen-Coppens, J.M.C.; van Helden-Meeuwsen, C.G.; Bootsma, H.; Vissink, A.; van Rooijen, N.; van de Merwe, J.P.; Drexhage, H.A.; Versnel, M.A. Increased Frequency of CD16+ Monocytes and the Presence of Activated Dendritic Cells in Salivary Glands in Primary Sjögren Syndrome. Ann. Rheum. Dis. 2009, 68, 420–426. [Google Scholar] [CrossRef]
- Ichikawa, Y.; Shimizu, H.; Yoshida, M.; Arimori, S. Activation Antigens Expressed on T-Cells of the Peripheral Blood in Sjögren’s Syndrome and Rheumatoid Arthritis. Clin. Exp. Rheumatol. 1990, 8, 243–249. [Google Scholar]
- Aziz, K.E.; Wakefield, D. In Vivo and in Vitro Expression of Adhesion Molecules by Peripheral Blood Lymphocytes from Patients with Primary Sjogren’s Syndrome: Culture-Associated Enhancement of LECAM-1 and CD44. Rheumatol. Int. 1995, 15, 69–74. [Google Scholar] [CrossRef] [PubMed]
- Kudryavtsev, I.; Benevolenskaya, S.; Serebriakova, M.; Grigor’yeva, I.; Kuvardin, E.; Rubinstein, A.; Golovkin, A.; Kalinina, O.; Zaikova, E.; Lapin, S.; et al. Circulating CD8+ T Cell Subsets in Primary Sjögren’s Syndrome. Biomedicines 2023, 11, 2778. [Google Scholar] [CrossRef]
- Su, R.; Wang, Y.Y.; Wang, C. Ab0017 Immune Characteristics of Peripheral Blood in Secondary Sjogren’s Syndrome Patients with Rheumatoid Arthritis. Ann. Rheum. Dis. 2021, 80, 1043. [Google Scholar] [CrossRef]
- Funauchi, M.; Sugishima, H.; Minoda, M.; Horiuchi, A. Serum Level of Interferon-Gamma in Autoimmune Diseases. Tohoku J. Exp. Med. 1991, 164, 259–267. [Google Scholar] [CrossRef]
- Halse, A.; Tengnér, P.; Wahren-Herlenius, M.; Haga, H.; Jonsson, R. Increased Frequency of Cells Secreting Interleukin-6 and Interleukin-10 in Peripheral Blood of Patients with Primary Sjögren’s Syndrome. Scand. J. Immunol. 1999, 49, 533–538. [Google Scholar] [CrossRef] [PubMed]
- Diz-Lopes, M.; Marques-Gomes, C.; Santos, I.; Costa, L.; Coelho, D.B.; Bernardes, M.; Rocha, T.M. Tocilizumab as a Treatment Option in Sjögren Disease with Associated Lymphocytic Interstitial Pneumonia. Clin. Rheumatol. 2025, 44, 4435–4436. [Google Scholar] [CrossRef] [PubMed]
- Nakahara, H.; Kawamoto, K.; Mori, H.; Nozato, S.; Hirai, M.; Matuoka, H.; Higa, S.; Fusama, M.; Takeuchi, E.; Igarashi, T.; et al. AB0485 Tocilizumab Is Effective for the Patient with Sjogren’s Syndrome Complicated with Rheumatoid Arthritis. Ann. Rheum. Dis. 2014, 73, 967–968. [Google Scholar] [CrossRef]
- Sudzius, G.; Mieliauskaite, D.; Siaurys, A.; Viliene, R.; Butrimiene, I.; Characiejus, D.; Dumalakiene, I. Distribution of Peripheral Lymphocyte Populations in Primary Sjögren’s Syndrome Patients. J. Immunol. Res. 2015, 2015, 854706. [Google Scholar] [CrossRef]
- Davies, R.; Hammenfors, D.; Bergum, B.; Jakobsen, K.; Solheim, M.; Vogelsang, P.; Brun, J.G.; Bryceson, Y.; Jonsson, R.; Appel, S. Patients with Primary Sjögren’s Syndrome Have Alterations in Absolute Quantities of Specific Peripheral Leucocyte Populations. Scand. J. Immunol. 2017, 86, 491–502. [Google Scholar] [CrossRef] [PubMed]
- Ichikawa, Y.; Shimizu, H.; Takahashi, K.; Yoshida, M.; Moriuchi, J.; Takaya, M.; Arimori, S. Lymphocyte Subsets of the Peripheral Blood in Sjögren’s Syndrome and Rheumatoid Arthritis. Clin. Exp. Rheumatol. 1989, 7, 55–61. [Google Scholar]
- Sarkar, I.; Davies, R.; Aarebrot, A.K.; Solberg, S.M.; Petrovic, A.; Joshi, A.M.; Bergum, B.; Brun, J.G.; Hammenfors, D.; Jonsson, R.; et al. Aberrant Signaling of Immune Cells in Sjögren’s Syndrome Patient Subgroups upon Interferon Stimulation. Front. Immunol. 2022, 13, 854183. [Google Scholar] [CrossRef]
- He, Y.; Chen, R.; Zhang, M.; Wang, B.; Liao, Z.; Shi, G.; Li, Y. Abnormal Changes of Monocyte Subsets in Patients With Sjögren’s Syndrome. Front. Immunol. 2022, 13, 864920. [Google Scholar] [CrossRef]
- Yoshimoto, K. Elevated Expression of BAFF Receptor, BR3, on Monocytes Correlates with B Cell Activation and Clinical Features of Patients with Primary Sjögren’s Syndrome. Arthritis Res. Ther. 2020, 22, 157. [Google Scholar] [CrossRef]
- Aarhus, R.; Graeff, R.M.; Dickey, D.M.; Walseth, T.F.; Hon, C.L. ADP-Ribosyl Cyclase and CD38 Catalyze the Synthesis of a Calcium-Mobilizing Metabolite from NADP+(∗). J. Biol. Chem. 1995, 270, 30327–30333. [Google Scholar] [CrossRef]
- Lee, H.C.; Aarhus, R. A Derivative of NADP Mobilizes Calcium Stores Insensitive to Inositol Trisphosphate and Cyclic ADP-Ribose (∗). J. Biol. Chem. 1995, 270, 2152–2157. [Google Scholar] [CrossRef]
- Abdul-Awal, S.M.; Hotta, C.T.; Davey, M.P.; Dodd, A.N.; Smith, A.G.; Webb, A.A.R. NO-Mediated [Ca2+]Cyt Increases Depend on ADP-Ribosyl Cyclase Activity in Arabidopsis1[OPEN]. Plant Physiol. 2016, 171, 623–631. [Google Scholar] [CrossRef]
- De Flora, A.; Guida, L.; Franco, L.; Zocchi, E. The CD38/Cyclic ADP-Ribose System: A Topological Paradox. Int. J. Biochem. Cell Biol. 1997, 29, 1149–1166. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.J.; Lam, C.M.C.; Lee, H.C. The Membrane-Bound Enzyme CD38 Exists in Two Opposing Orientations. Sci. Signal. 2012, 5, ra67. [Google Scholar] [CrossRef]
- Kim, H.; Jacobson, E.L.; Jacobson, M.K. Synthesis and Degradation of Cyclic ADP-Ribose by NAD Glycohydrolases. Science 1993, 261, 1330–1333. [Google Scholar] [CrossRef]
- Vu, C.Q.; Lu, P.-J.; Chen, C.-S.; Jacobson, M.K. 2′-Phospho-Cyclic ADP-Ribose, a Calcium-Mobilizing Agent Derived from NADP. J. Biol. Chem. 1996, 271, 4747–4754. [Google Scholar] [CrossRef]
- Kontani, K.; Kukimoto, I.; Kanda, Y.; Inoue, S.; Kishimoto, H.; Hoshino, S.; Nishina, H.; Takahashi, K.; Hazeki, O.; Katada, T. Signal Transduction Via the CD38/NAD+ Glycohydrolase. In ADP-Ribosylation in Animal Tissues; Springer: Boston, MA, USA, 1997; pp. 421–430. ISBN 978-1-4419-8632-0. [Google Scholar]
- Partida-Sanchez, S.; Cockayne, D.A.; Monard, S.; Jacobson, E.L.; Oppenheimer, N.; Garvy, B.; Kusser, K.; Goodrich, S.; Howard, M.; Harmsen, A.; et al. Cyclic ADP-Ribose Production by CD38 Regulates Intracellular Calcium Release, Extracellular Calcium Influx and Chemotaxis in Neutrophils and Is Required for Bacterial Clearance in Vivo. Nat. Med. 2001, 7, 1209–1216. [Google Scholar] [CrossRef] [PubMed]
- Szlasa, W.; Czarny, J.; Sauer, N.; Rakoczy, K.; Szymańska, N.; Stecko, J.; Kołodziej, M.; Kaźmierczak, M.; Barg, E. Targeting CD38 in Neoplasms and Non-Cancer Diseases. Cancers 2022, 14, 4169. [Google Scholar] [CrossRef] [PubMed]
- Deterre, P.; Berthelier, V.; Bauvois, B.; Dalloul, A.; Schuber, F.; Lund, F. CD38 in T- and B-Cell Functions. In Chemical Immunology and Allergy; Mehta, K., Malavasi, F., Eds.; KARGER: Basel, Switzerland, 2000; Volume 75, pp. 146–168. ISBN 978-3-8055-6923-1. [Google Scholar]
- Partida-Sánchez, S.; Randall, T.D.; Lund, F.E. Innate Immunity Is Regulated by CD38, an Ecto-Enzyme with ADP-Ribosyl Cyclase Activity. Microbes Infect. 2003, 5, 49–58. [Google Scholar] [CrossRef]
- Ma, Y.; Zhang, Y.; Liu, X.; Yang, X.; Guo, H.; Ding, X.; Ye, C.; Guo, C. Deletion of CD38 Mitigates the Severity of NEC in Experimental Settings by Modulating Macrophage-Mediated Inflammation. Redox Biol. 2024, 77, 103336. [Google Scholar] [CrossRef]
- Quintana, D.S.; Rokicki, J.; van der Meer, D.; Alnæs, D.; Kaufmann, T.; Córdova-Palomera, A.; Dieset, I.; Andreassen, O.A.; Westlye, L.T. Oxytocin Pathway Gene Networks in the Human Brain. Nat. Commun. 2019, 10, 668. [Google Scholar] [CrossRef]
- Todd, R.F.; Roach, J.A.; Arnaout, M.A. The Modulated Expression of Mo5, a Human Myelomonocytic Plasma Membrane Antigen. Blood 1985, 65, 964–973. [Google Scholar] [CrossRef] [PubMed]
- Ishioka-Takei, E.; Yoshimoto, K.; Suzuki, K.; Nishikawa, A.; Yasuoka, H.; Yamaoka, K.; Takeuchi, T. Increased Proportion of a CD38highIgD+ B Cell Subset in Peripheral Blood Is Associated with Clinical and Immunological Features in Patients with Primary Sjögren’s Syndrome. Clin. Immunol. 2018, 187, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Vitali, C.; Bombardieri, S.; Jonsson, R.; Moutsopoulos, H.M.; Alexander, E.L.; Carsons, S.E.; Daniels, T.E.; Fox, P.C.; Fox, R.I.; Kassan, S.S.; et al. Classification Criteria for Sjögren’s Syndrome: A Revised Version of the European Criteria Proposed by the American-European Consensus Group. Ann. Rheum. Dis. 2002, 61, 554–558. [Google Scholar] [CrossRef] [PubMed]
| Patients (40) | No. (Frequency) |
|---|---|
| Additional symptoms | |
| TMJ disorder | 20 (50%) |
| Thyroid dysfunction | 11 (27.5%) |
| Arthritis | 25 (62.5%) |
| Weight loss | 9 (22.5%) |
| Fatigue | 27 (67.5%) |
| Systemic pharmacological treatment | |
| NSAIDs | 11 (27.5%) |
| Cyclosporin A | 5 (12.5%) |
| Corticosteroids | 13 (32.5%) |
| Antimalarials | 11 (27.5%) |
| Combined treatment | 8 (20%) |
| Yes | No | |
|---|---|---|
| Low-count group (24) | 24 (100%) | 0 (0%) |
| Higher-count group (16) | 11 (68.8%) | 5 (31.3%) |
| Yes | No | |
|---|---|---|
| Low-count group (24) | 20 (83.3%) | 4 (16.7%) |
| Higher-count group (16) | 7 (43.8%) | 9 (56.3%) |
| Controls (20) | Patients (40) | |
|---|---|---|
| Men/Women (no.) | 1/19 | 3/37 |
| Age (years) | 54 | 55.5 |
| No. of leukocytes (×109/L) | 6.4 | 5.25 |
| Oral symptoms and signs, and their laboratory evaluation | ||
| Xerostomia (no.) | 0 | 33 |
| Dysphagia (no.) | 0 | 23 |
| Decreased salivary flow (no.) | 0 | 24 |
| MSG biopsy (no.) | N/A | 24 (15) |
| Ocular symptoms and signs | ||
| Xerophtalmia (no.) | 0 | 34 |
| Schirmer’s test (no.) | N/A | 34 |
| Presence of anti-Ro autoantibodies | 0 | 35 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rosecka, M.; Kolackova, M.; Afshari, M.; Jozifkova, E.; Slezak, R.; Krejsek, J.; Radochova, V. Pathological Mechanisms in Sjögren’s Disease Likely Involve the ADP-Ribosyl Cyclase Family Members: CD38 and CD157. Int. J. Mol. Sci. 2025, 26, 11544. https://doi.org/10.3390/ijms262311544
Rosecka M, Kolackova M, Afshari M, Jozifkova E, Slezak R, Krejsek J, Radochova V. Pathological Mechanisms in Sjögren’s Disease Likely Involve the ADP-Ribosyl Cyclase Family Members: CD38 and CD157. International Journal of Molecular Sciences. 2025; 26(23):11544. https://doi.org/10.3390/ijms262311544
Chicago/Turabian StyleRosecka, Michaela, Martina Kolackova, Moeina Afshari, Eva Jozifkova, Radovan Slezak, Jan Krejsek, and Vladimira Radochova. 2025. "Pathological Mechanisms in Sjögren’s Disease Likely Involve the ADP-Ribosyl Cyclase Family Members: CD38 and CD157" International Journal of Molecular Sciences 26, no. 23: 11544. https://doi.org/10.3390/ijms262311544
APA StyleRosecka, M., Kolackova, M., Afshari, M., Jozifkova, E., Slezak, R., Krejsek, J., & Radochova, V. (2025). Pathological Mechanisms in Sjögren’s Disease Likely Involve the ADP-Ribosyl Cyclase Family Members: CD38 and CD157. International Journal of Molecular Sciences, 26(23), 11544. https://doi.org/10.3390/ijms262311544





