The Central Role of GSNOR: Decoding Nitric Oxide Signaling for Crop Stress Tolerance
Abstract
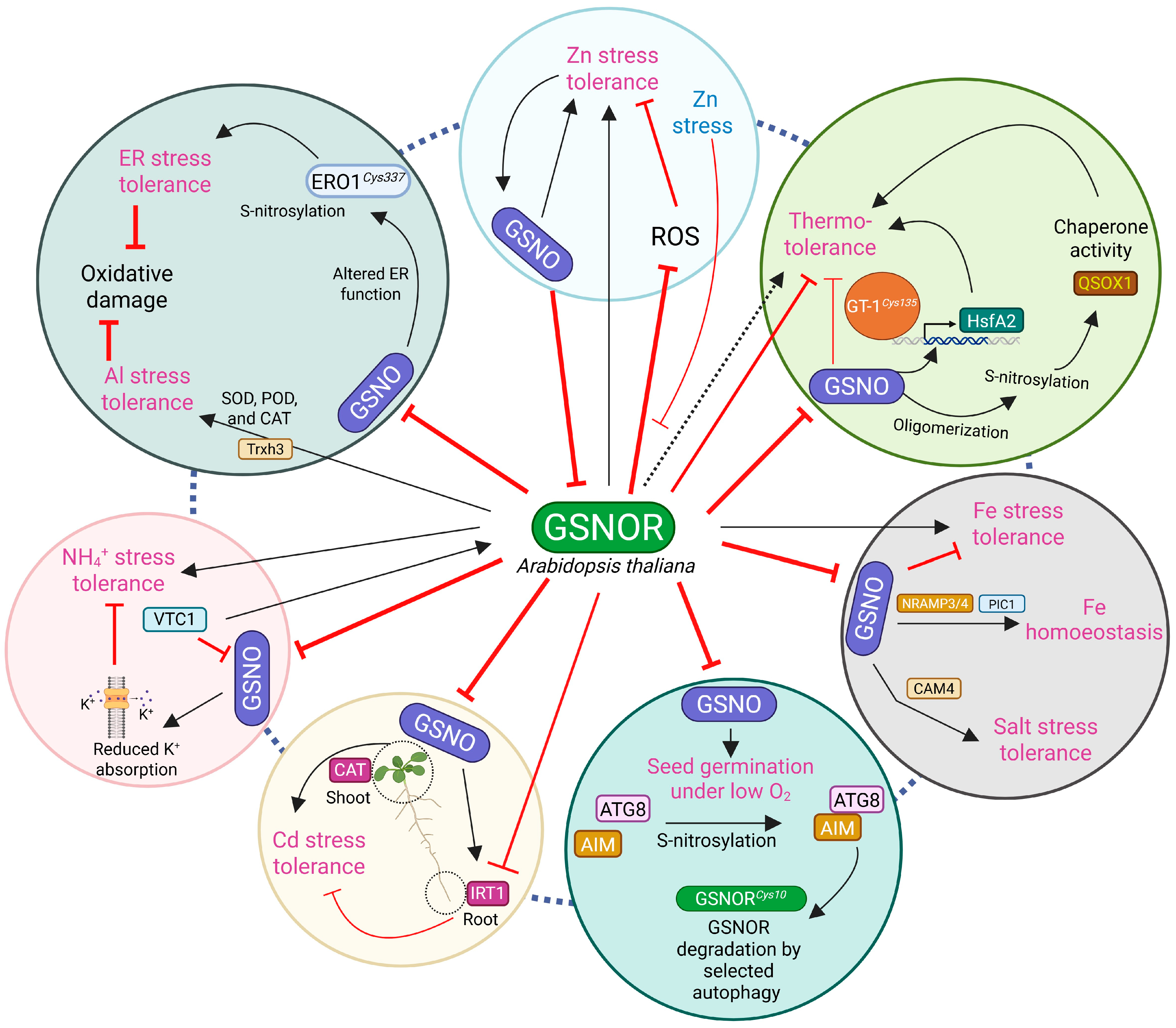
1. Introduction
1.1. Nitric Oxide
1.2. An Introduction to the GSNOR Enzyme and Its Role in GSNO Turnover
| Species | S-Nitrosylation Sites | Target Protein | Upstream Signal | Detection Method | Reference |
|---|---|---|---|---|---|
| Arabidopsis thaliana | Cys-80 | BIK1 | P/MAMPs | Biotin switch assay | [39] |
| Arabidopsis thaliana | Cys-425, Cys-607 | COP1 | Light | Biotin switch assay, nano-LC-MS/MS | [40] |
| Arabidopsis thaliana | - | QSOX1 | Heat stress | Biotin switch assay | [41] |
| Arabidopsis thaliana | Cys-337 | ERO1 | ER stress | TMT labeling, LC-MS/MS | [42] |
| Arabidopsis thaliana | Cys-137 | HDA19 | Oxidative stress | Biotin switch assay, TMT labeling, LC-MS/MS | [43] |
| Arabidopsis thaliana | Cys-374 | RGA | Salt stress | Biotin switch assay, LC-MS/MS | [44] |
| Arabidopsis thaliana | Cys-164 | HFR1 | High temperature | Biotin switch assay | [45] |
| Arabidopsis thaliana | - | AtNRAMP3, AtNRAMP4, AtPIC1 | Iron deficiency | GPS-SNO 1.0 software (in silico and protein stability assay) | [46] |
| Arabidopsis thaliana | Cys-10 | GSNOR1 | Hypoxia | Biotin switch assay, DAN Assay, LC-MS/MS | [47] |
| Arabidopsis thaliana | Cys-137 | SnRK 2.6 | Drought | Biotin switch assay, LC-MS/MS | [48] |
| Arabidopsis thaliana | Cys-32 | APX1 | Oxidative stress | Biotin switch assay, DAN assay, LC-MS/MS | [49] |
| Arabidopsis thaliana | Cys-890 | RBOHD | Pathogen | Biotin switch assay, LC-MS | [50] |
| Arabidopsis thaliana | Cys-28 | AtSABP3 | Pathogen infection | Biotin switch assay, LC-MS/MS | [51] |
| Arabidopsis thaliana | Cys-156 | NPR1 | Pathogen infection | Biotin switch assay | [52] |
| Tomato (Solanum lycopersicum) | Cys-316, Cys-258, Cys-316 | SlGABA-TP1, SlGABA-TP2, SlGABA-TP3 | Saline-alkaline stress | Biotin switch assay | [53] |
| Tomato (Solanum lycopersicum) | Cys-5 | SlP5CR | Drought and salt stress | Biotin switch assay | [54] |
| Tomato (Solanum lycopersicum) | Cys-54 | SlTrxh | Nitrate stress | Biotin switch assay, LC-MS/MS | [55] |
| Tomato (Solanum lycopersicum) | Cys-172 | ACOh4 | Salt stress | Biotin switch assay, LC-MS/MS | [56] |
| Mini Chinese Cabbage (Brassica rapa ssp. pekinensis) | - | BrGSNOR | Low temperature stress | Biotin switch assay | [57] |
| Peach (Prunus persica (L.) Batsch) | Cys-85 | - | Pathogen infection | Iodo-TMT labeling, LC-MS/MS | [58] |
2. Changes in GSNOR Activity Affect Abiotic Stress Tolerance
2.1. High- and Low-Temperature Stress Tolerance
2.2. Iron Stress Tolerance and Homeostasis
| Mode of Genetic Modification | Stress Conditions | GSNOR Activity/Expression | NO/SNO Levels | Crosstalk with Other Proteins | Stress Effects | ROS: Antioxidant | Crosstalk with Hormones | Reference |
|---|---|---|---|---|---|---|---|---|
| hot1 | Heat | – | – | QSOX1 | Tolerance | – | – | [41] |
| gsnor1-3 | Cd | – | – | – | Tolerance | ↓:↑ | – | [80] |
| GSNOR | Sensitive | ↑:↓ | ||||||
| hot5-4 | ER | – | – | ERO1 | Tolerance | – | – | [42] |
| gsnor1 | Oxidative | – | –/Increased | HDA19 | – | – | – | [43] |
| hot5-2 | Heat | – | Increased/– | GT-1 | Tolerance | – | – | [66] |
| hot5-2 | NH4+ | – | Increased/– | – | Sensitive | – | – | [81] |
| gsnor1-3 | Oxidative | – | – | – | Sensitive | – | – | [82] |
| gsnor1-3 | Light intensity | – | Increased/Increased | HDA6 | – | – | – | [83] |
| gsnor1-3 | Oxidative | Inhibited | –/Increased | ICS1 | – | – | SA | [84] |
| gsnor1-3 | Oxidative | – | – | ROG1 | Tolerance | – | – | [85] |
| gsnor1-3 | Zn | – | – | APX1 | Tolerance | ↑:↓ | – | [86] |
| 35S:FLAG-GSNOR1 | Inhibited | Decreased/Increased | Sensitive | ↑:↓ | ||||
| gsnor1-3 | Cd | – | – | IRT1 and APX | Sensitive | – | – | [87] |
| GSNOR | Induced | Tolerance | ||||||
| gsnor1-3 | Fe | – | Increased/– | – | Sensitive | – | – | [77] |
| gsnor1-3 | Hypoxia | – | – | ATG8 | Sensitive | – | – | [47] |
| gsnor | Salt | Inhibited | Increased/– | CaM | Tolerance | – | – | [88] |
| GSNOR | Induced | Decreased/– | Sensitive | |||||
| gsnor1-3 | Oxidative | – | – | APX1 | Tolerance | –:↑ | – | [49] |
| gsnor1 | Nitrate | Inhibited | – | – | Sensitive | – | – | [89] |
| 35S:FLAG-GSNOR1 | Induced | Tolerance | ||||||
| hot5 | Heat | – | – | – | Sensitive | – | – | [62] |
2.3. Salt, Drought, and Metal Stress Tolerance
2.4. Nutrient Stress Tolerance
2.5. Saline–Alkaline Stress Tolerance
2.6. Other Stress Responses
| Species | Mode of Genetic Modification | Stress Conditions | GSNOR Activity/Expression | NO/SNO Levels | Crosstalk with Other Proteins | Stress Effects | ROS: Antioxidant | Crosstalk with Hormones | Reference |
|---|---|---|---|---|---|---|---|---|---|
| Solanum lycopersicum L. | GSNOR | Saline–Alkali Stress | – | Decreased/Decreased | HA2 | Tolerance | ↓:– | Melatonin | [101] |
| GSNOR-RNAi | Increased/Increased | Sensitive | ↑:– | ||||||
| Nicotiana tabacum | GSNOR1 | Aluminum Stress | Induced | Decreased/Decreased | Trxh3 | Tolerance | ↓:↑ | – | [104] |
| Solanum lycopersicum L. | GSNOR-RNAi | Salt Stress | – | – | P5CR | Sensitive | – | – | [54] |
| Solanum lycopersicum cv. Ailsa | GSNOR-RNAi | Salt Stress | – | – | ACOh4 | Sensitive | – | Ethylene | [56] |
| Ganoderma lucidum | GSNOR-RNAi | Heat Stress | Inhibited | – | CAT | Tolerance | ↓:↑ | – | [105] |
| Solanum lycopersicum L. | GSNOR-RNAi | Salt Stress | – | Increased/– | MAPK3, ACO1 | Sensitive | ↑:– | – | [106] |
| Solanum lycopersicum L. cv. Ailsa | GSNOR-Silenced | High-Temperature Stress | Inhibited | –/Increased | RBOH1 | Sensitive | ↑:↓ | ABA and SA | [63] |
| Nicotiana tabacum | GSNOR | Nitrate Stress | Induced | Decreased/Decreased | – | Tolerance | ↓:↑ | – | [92] |
| Solanum lycopersicum L. | GSNOR | Fe Deficiency Stress | Induced | Decreased/Decreased | – | Tolerance | ↓:↑ | – | [78] |
| Solanum lycopersicum L. cv. Condine Red | GSNOR-Silenced | Cold Acclimation | Inhibited | Increased/ | NR, MPK1/2 | Tolerance | – | – | [72] |
| Solanum lycopersicum L. | GSNOR | Alkaline Stress | Induced | Decreased/Decreased | – | Tolerance | ↓:↑ | – | [100] |
| GSNOR-Suppressed | Inhibited | Increased/Increased | Sensitive | ↑:↓ | |||||
| Oryza sativa | GSNOR | Oxidative Stress | Induced | –/Decreased | – | Tolerance | ↑:↓ | – | [107] |
| GSNOR-RNAi | Inhibited | –/Increased | Sensitive | – | |||||
| Solanum lycopersicum L. | GSNOR | Botrytis cinerea | – | Decreased/Decreased | COMT2 | Sensitive | – | JA, Melatonin | [108] |
| GSNOR-RNAi | Increased/Increased | Tolerance | |||||||
| GSNOR-Silenced | |||||||||
| Solanum lycopersicum L. | gsnor | P. capsici, flg22 | Inhibited | – | PcRD18, ATG8c | Sensitive | ↓:– | SA | [109] |
| GSNOR-silenced | |||||||||
| Solanum lycopersicum L. | GSNOR | Botrytis cinerea | Induced | Decreased/– | – | Tolerance | – | – | [110] |
| GSNOR-RNAi | Inhibited | Increased/– | Sensitive | ||||||
| Solanum lycopersicum L. | GSNOR | Pst DC3000 | Induced | – | – | Tolerance | – | – | [111] |
| Solanum lycopersicum L. | GSNOR | Pst DC3000 | Induced | – | PR1 | Tolerance | – | SA | [112] |
| GSNOR-RNAi | Inhibited | Sensitive | |||||||
| Medicago truncatula L. | 35S:GSNOR | Aphanomyces euteiches | Induced | –/Increased | NR | Tolerance | – | – | [113] |
3. Changes in GSNOR Activity Affect Biotic Stress Tolerance
| Mode of Genetic Modification | Stress Conditions | GSNOR Activity/Expression | NO/SNO Levels | Crosstalk with Other Proteins | Stress Effects | ROS: Antioxidant | Crosstalk with Hormones | Reference |
|---|---|---|---|---|---|---|---|---|
| gsnor1-3 | Pst DC3000 hrcC−, flg22 | – | –/Increased | BIK1, RBOHD, FLS2, BAK1 | Sensitive | ↑:– | – | [39] |
| par2-1 | ||||||||
| gsnor1-1 | Phytophthora parasitica | – | – | – | – | – | – | [119] |
| gsnor1-3 | Sensitive | ↓:– | SA | |||||
| par2-1 | – | – | ||||||
| gsnor1-3 | Pst DC3000 (avrRpt2), Pst DC3000 (avrRpm1), Pst DC3000 (avrRps4) | Inhibited | –/Increased | RBOHD | Sensitive | – | – | [65] |
| gsnor1-3 | Pst DC3000 (avrB) | – | –/Increased | – | Sensitive | – | SA | [118] |
| gsnor1-3 | Pst DC3000, Pst DC3000 (avrRpm1) | – | –/Increased | SRG1 | Sensitive | ↓:– | SA | [117] |
| gsnor1-3 | Pst DC3000, Pst DC 3000 (avrB) | – | –/Increased | – | Sensitive | – | SA | [116] |
| gsnor1-1 | Pst DC3000 (avrB), Pst DC3000 (avrRps4), Pst DC3000 (virulent), Psp | – | Decreased/Decreased | – | Tolerance | – | – | [122] |
| 35S:FLAG-GSNOR1 | – | |||||||
| gnsor1-3 | Inhibited | Increased/Increased | Sensitive | SA | ||||
| par2-1 | – | – | – | |||||
| gsnor1-1 | Pst DC3000 (avrB), Pst DC3000 (avrRps4), Pst DC3000, H. arabidopsidis Emwa1 | Induced | Decreased/Decreased | RBOHD | Sensitive | ↑:– | SA | [50] |
| gnsor1-3 | Inhibited | Increased/Increased | Tolerance | ↓:– | ||||
| gsnor | Pst DC3000 (avrRpt2), Pst DC3000 | Inhibited | – | – | – | –:↑ | – | [123] |
| gsnor1-1 | Pst DC3000 (avrB) | – | –/Decreased | SABP3 | Tolerance | – | SA | [51] |
| gsnor1-3 | –/Increased | Sensitive | ||||||
| gsnor1-1 | Pst DC3000 (avrB), Pst DC3000 | Induced | –/Decreased | – | Tolerance | – | SA | [30] |
| gsnor1-2 | ||||||||
| gsnor1-3 | Inhibited | –/Increased | Sensitive |
4. Nitric Oxide for Innovative Agricultural Application: Potential Nanotechnology and Its Limitations
5. Conclusions and Future Perspectives
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ADH3 | Class III alcohol dehydrogenase |
| APX1 | Ascorbate Peroxidase 1 |
| BIK1 | Botrytis-induced kinase 1 |
| COP1 | Constitutive Photomorphogenesis Protein 1 |
| Cys | Cystein |
| ER | Endoplasmic Reticulum |
| ERO1 | Endoplasmic Reticulum Oxidoreductin 1 |
| ET | Ethylene |
| ETI | Effector-Triggered Immunity |
| FRO1 | Ferric-Reductase Oxidase1 |
| GABA-TP1/2 | Gamma-aminobutyrate Transaminase 1/2 |
| GSH | Reduced Glutathione |
| GSNO | S-nitrosoglutathione |
| GSNOR | S-nitrosoglutathione Reductase |
| GSSG | Glutathione disulfide |
| HDA19 | Histone Deacetylase 19 |
| HFR1 | Far-red elongated hypocotyl 1 |
| JA | Jasmonic Acid |
| MDHAR | Monodehydroascorbate Reductase |
| NADH | Nicotinamide adenine dinucleotide |
| NIA1/2 | Nitrate Reductase 1/2 |
| NO | Nitric Oxide |
| NOA1 | Nitric Oxide-Associated 1 |
| NOS | Nitric Oxide Synthase |
| NOX1 | NADPH oxidase 1 |
| NPR1 | Nonexpressor of Pathogenesis-Related genes 1 |
| NRAMP3/4 | Natural Resistance-Associated Macrophage Protein 3/4 |
| OPT | Oligopeptide transporter |
| P5CR | Pyrroline-5-Carboxylate Reductase |
| PDC1 | Pyruvate Decarboxylase 1 |
| PIC1 | Permease in Chloroplasts 1 |
| QSOX1 | Quiescin Sulfhydryl Oxidase 1 |
| RBOHD | Respiratory Burst Oxidase Homologue D |
| RGA | Repressor of Gibberellin 1-3 |
| RNS | Reactive Nitrogen Species |
| ROS | Reactive Oxygen Species |
| SA | Salicylic Acid |
| SABP3 | Salicylic Acid-Binding Protein 3 |
| SAR | Systemic Acquired Resistance |
| SNO | S-nitrosothiol |
| SNP | Single-Nucleotide Polymorphism |
| SnRK | Sucrose Non-Fermenting 1-related Protein Kinase 2.6 |
| Trxh3 | Thioredoxin H3 |
References
- Tennyson, A.G.; Lippard, S.J. Generation, Translocation, and Action of Nitric Oxide in Living Systems. Chem. Biol. 2011, 18, 1211–1220. [Google Scholar] [CrossRef]
- Dixit, V.D.; Parvizi, N. Nitric oxide and the control of reproduction. Anim. Reprod. Sci. 2001, 65, 1–16. [Google Scholar] [CrossRef]
- Furchgott, R.F.; Zawadzki, J.V. The obligatory role of endothelial cells in the relaxation of arterial smooth muscle by acetylcholine. Nature 1980, 288, 373–376. [Google Scholar] [CrossRef] [PubMed]
- Ignarro, L.J.; Buga, G.M.; Wood, K.S.; Byrns, R.E.; Chaudhuri, G. Endothelium-derived relaxing factor produced and released from artery and vein is nitric oxide. Proc. Natl. Acad. Sci. USA 1987, 84, 9265–9269. [Google Scholar] [CrossRef] [PubMed]
- Palmer, R.M.J.; Ferrige, A.G.; Moncada, S. Nitric oxide release accounts for the biological activity of endothelium-derived relaxing factor. Nature 1987, 327, 524–526. [Google Scholar] [CrossRef]
- Priestley, J. Experiments and Observations on Different Kinds of Air; J. Johnson: Chicago, IL, USA, 1776; Volume 2. [Google Scholar]
- Klepper, L.A. Nitric Oxide Emissions from Soybean Leaves during in Vivo Nitrate Reductase Assays. Plant Physiol. 1987, 85, 96–99. [Google Scholar] [CrossRef]
- Klepper, L. Comparison between NOx Evolution Mechanisms of Wild-Type and nr1 Mutant Soybean Leaves. Plant Physiol. 1990, 93, 26–32. [Google Scholar] [CrossRef]
- Dean, J.V.; Harper, J.E. Nitric Oxide and Nitrous Oxide Production by Soybean and Winged Bean during the in Vivo Nitrate Reductase Assay. Plant Physiol. 1986, 82, 718–723. [Google Scholar] [CrossRef] [PubMed]
- Förstermann, U.; Sessa, W.C. Nitric oxide synthases: Regulation and function. Eur. Heart J. 2011, 33, 829–837. [Google Scholar] [CrossRef]
- Wilson, I.D.; Neill, S.J.; Hancock, J.T. Nitric oxide synthesis and signalling in plants. Plant Cell Environ. 2008, 31, 622–631. [Google Scholar] [CrossRef]
- Yamasaki, H.; Sakihama, Y. Simultaneous production of nitric oxide and peroxynitrite by plant nitrate reductase: In Vitro evidence for the NR-dependent formation of active nitrogen species. FEBS Lett. 2000, 468, 89–92. [Google Scholar] [CrossRef] [PubMed]
- Rockel, P.; Strube, F.; Rockel, A.; Wildt, J.; Kaiser, W.M. Regulation of nitric oxide (NO) production by plant nitrate reductase in vivo and In Vitro. J. Exp. Bot. 2002, 53, 103–110. [Google Scholar] [CrossRef]
- Jahnová, J.; Luhová, L.; Petřivalský, M. S-Nitrosoglutathione Reductase—The Master Regulator of Protein S-Nitrosation in Plant NO Signaling. Plants 2019, 8, 48. [Google Scholar] [CrossRef] [PubMed]
- Sarma, H.; Ramchiary, C.; Sen, B.; Daimary, M.; Prasad, R. Integration of nitric oxide signaling in plant stress responses: Unveiling its role in enhancing crop tolerance to abiotic stress. Discov. Plants 2025, 2, 32. [Google Scholar] [CrossRef]
- Stuehr, D.J. Arginine metabolism: Enzymology, nutrition, and clinical significance. J. Nutr. 2004, 134, 2748S–2751S. [Google Scholar] [CrossRef] [PubMed]
- Jeandroz, S.; Wipf, D.; Stuehr, D.J.; Lamattina, L.; Melkonian, M.; Tian, Z.; Zhu, Y.; Carpenter, E.J.; Wong, G.K.-S.; Wendehenne, D. Occurrence, structure, and evolution of nitric oxide synthase–like proteins in the plant kingdom. Sci. Signal. 2016, 9, re2. [Google Scholar] [CrossRef]
- Gaston, B.; Reilly, J.; Drazen, J.M.; Fackler, J.; Ramdev, P.; Arnelle, D.; Mullins, M.E.; Sugarbaker, D.J.; Chee, C.; Singel, D.J.; et al. Endogenous nitrogen oxides and bronchodilator S-nitrosothiols in human airways. Proc. Natl. Acad. Sci. USA 1993, 90, 10957–10961. [Google Scholar] [CrossRef]
- Spadaro, D.; Yun, B.-W.; Spoel, S.H.; Chu, C.; Wang, Y.-Q.; Loake, G.J. The redox switch: Dynamic regulation of protein function by cysteine modifications. Physiol. Plant. 2010, 138, 360–371. [Google Scholar] [CrossRef]
- Durner, J.; Klessig, D.F. Nitric oxide as a signal in plants. Curr. Opin. Plant Biol. 1999, 2, 369–374. [Google Scholar] [CrossRef]
- Espunya, M.C.; Díaz, M.; Moreno-Romero, J.; Martínez, M.C. Modification of intracellular levels of glutathione-dependent formaldehyde dehydrogenase alters glutathione homeostasis and root development. Plant Cell Environ. 2006, 29, 1002–1011. [Google Scholar] [CrossRef]
- Hogg, N. The Biochemistry and Physiology of S-Nitrosothiols. Annu. Rev. Pharmacol. Toxicol. 2002, 42, 585–600. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Ruiz, A.; Lamas, S. Signalling by NO-induced protein S-nitrosylation and S-glutathionylation: Convergences and divergences. Cardiovasc. Res. 2007, 75, 220–228. [Google Scholar] [CrossRef]
- Stamler, J.S.; Lamas, S.; Fang, F.C. Nitrosylation: The Prototypic Redox-Based Signaling Mechanism. Cell 2001, 106, 675–683. [Google Scholar] [CrossRef]
- Wang, Y.; Yun, B.-W.; Kwon, E.; Hong, J.K.; Yoon, J.; Loake, G.J. S-Nitrosylation: An emerging redox-based post-translational modification in plants. J. Exp. Bot. 2006, 57, 1777–1784. [Google Scholar] [CrossRef]
- Corpas, F.J.; Alché, J.d.; Barroso, J.B. Current overview of S-nitrosoglutathione (GSNO) in higher plants. Front. Plant Sci. 2013, 4, 126. [Google Scholar] [CrossRef]
- Liu, L.; Hausladen, A.; Zeng, M.; Que, L.; Heitman, J.; Stamler, J.S. A metabolic enzyme for S-nitrosothiol conserved from bacteria to humans. Nature 2001, 410, 490–494. [Google Scholar] [CrossRef] [PubMed]
- Jensen, D.E.; Belka, G.K.; Bois, G.C.D. S-Nitrosoglutathione is a substrate for rat alcohol dehydrogenase class III isoenzyme. Biochem. J. 1998, 331, 659–668. [Google Scholar] [CrossRef]
- Sakamoto, A.; Ueda, M.; Morikawa, H. Arabidopsis glutathione-dependent formaldehyde dehydrogenase is an S-nitrosoglutathione reductase. FEBS Lett. 2002, 515, 20–24. [Google Scholar] [CrossRef] [PubMed]
- Feechan, A.; Kwon, E.; Yun, B.-W.; Wang, Y.; Pallas, J.A.; Loake, G.J. A central role for S-nitrosothiols in plant disease resistance. Proc. Natl. Acad. Sci. USA 2005, 102, 8054–8059. [Google Scholar] [CrossRef]
- Koivusalo, M.; Baumann, M.; Uotila, L. Evidence for the identity of glutathione-dependent formaldehyde dehydrogenase and class III alcohol dehydrogenase. FEBS Lett. 1989, 257, 105–109. [Google Scholar] [CrossRef]
- Jaffrey, S.R.; Erdjument-Bromage, H.; Ferris, C.D.; Tempst, P.; Snyder, S.H. Protein S-nitrosylation: A physiological signal for neuronal nitric oxide. Nat. Cell Biol. 2001, 3, 193–197. [Google Scholar] [CrossRef]
- Chen, Y.-J.; Ching, W.-C.; Lin, Y.-P.; Chen, Y.-J. Methods for detection and characterization of protein S-nitrosylation. Methods 2013, 62, 138–150. [Google Scholar] [CrossRef] [PubMed]
- Feng, J.; Chen, L.; Zuo, J. Protein S-Nitrosylation in plants: Current progresses and challenges. J. Integr. Plant Biol. 2019, 61, 1206–1223. [Google Scholar] [CrossRef]
- Machchhu, F.; Wany, A. Protein S-nitrosylation in plants under biotic stress. Theor. Exp. Plant Physiol. 2023, 35, 331–339. [Google Scholar] [CrossRef]
- Li, B.; Sun, C.; Lin, X.; Busch, W. The Emerging Role of GSNOR in Oxidative Stress Regulation. Trends Plant Sci. 2021, 26, 156–168. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Liu, Z.; Wu, X.; Fang, H.; Huang, D.; Pan, X.; Liao, W. Role of protein S-nitrosylation in plant growth and development. Plant Cell Rep. 2024, 43, 204. [Google Scholar] [CrossRef]
- Huang, D.; Huo, J.; Zhang, J.; Wang, C.; Wang, B.; Fang, H.; Liao, W. Protein S-nitrosylation in programmed cell death in plants. Cell. Mol. Life Sci. 2019, 76, 1877–1887. [Google Scholar] [CrossRef]
- Cui, B.; Pan, Q.; Cui, W.; Wang, Y.; Loake, V.I.P.; Yuan, S.; Liu, F.; Loake, G.J. S-nitrosylation of a receptor-like cytoplasmic kinase regulates plant immunity. Sci. Adv. 2024, 10, eadk3126. [Google Scholar] [CrossRef]
- Zhang, Q.; Cai, X.; Wu, B.; Tong, B.; Xu, D.; Wang, J.; Cui, B.; Yin, R.; Lin, L. S-nitrosylation may inhibit the activity of COP1 in plant photomorphogenesis. Biochem. Biophys. Res. Commun. 2024, 719, 150096. [Google Scholar] [CrossRef]
- Chae, H.B.; Bae, S.B.; Paeng, S.K.; Wi, S.D.; Thi Phan, K.A.; Lee, S.Y. S-nitrosylation switches the Arabidopsis redox sensor protein, QSOX1, from an oxidoreductase to a molecular chaperone under heat stress. Plant Physiol. Biochem. 2024, 206, 108219. [Google Scholar] [CrossRef]
- Qin, G.; Qu, M.; Jia, B.; Wang, W.; Luo, Z.; Song, C.-P.; Tao, W.A.; Wang, P. FAT-switch-based quantitative S-nitrosoproteomics reveals a key role of GSNOR1 in regulating ER functions. Nat. Commun. 2023, 14, 3268. [Google Scholar] [CrossRef]
- Zheng, Y.; Li, Z.; Cui, X.; Yang, Z.; Bao, C.; Pan, L.; Liu, X.; Chatel-Innocenti, G.; Vanacker, H.; Noctor, G.; et al. S-Nitrosylation of the histone deacetylase HDA19 stimulates its activity to enhance plant stress tolerance in Arabidopsis. Plant J. 2023, 114, 836–854. [Google Scholar] [CrossRef]
- Chen, L.; Sun, S.; Song, C.-P.; Zhou, J.-M.; Li, J.; Zuo, J. Nitric oxide negatively regulates gibberellin signaling to coordinate growth and salt tolerance in Arabidopsis. J. Genet. Genom. 2022, 49, 756–765. [Google Scholar] [CrossRef]
- Ying, S.; Yang, W.; Li, P.; Hu, Y.; Lu, S.; Zhou, Y.; Huang, J.; Hancock, J.T.; Hu, X. Phytochrome B enhances seed germination tolerance to high temperature by reducing S-nitrosylation of HFR1. EMBO Rep. 2022, 23, e54371. [Google Scholar] [CrossRef]
- Shee, R.; Ghosh, S.; Khan, P.; Sahid, S.; Roy, C.; Shee, D.; Paul, S.; Datta, R. Glutathione regulates transcriptional activation of iron transporters via S-nitrosylation of bHLH factors to modulate subcellular iron homoeostasis. Plant Cell Environ. 2022, 45, 2176–2190. [Google Scholar] [CrossRef] [PubMed]
- Zhan, N.; Wang, C.; Chen, L.; Yang, H.; Feng, J.; Gong, X.; Ren, B.; Wu, R.; Mu, J.; Li, Y.; et al. S-Nitrosylation Targets GSNO Reductase for Selective Autophagy during Hypoxia Responses in Plants. Mol. Cell 2018, 71, 142–154.e146. [Google Scholar] [CrossRef]
- Wang, P.; Du, Y.; Hou, Y.-J.; Zhao, Y.; Hsu, C.-C.; Yuan, F.; Zhu, X.; Tao, W.A.; Song, C.-P.; Zhu, J.-K. Nitric oxide negatively regulates abscisic acid signaling in guard cells by S-nitrosylation of OST1. Proc. Natl. Acad. Sci. USA 2015, 112, 613–618. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Mu, J.; Chen, L.; Feng, J.; Hu, J.; Li, L.; Zhou, J.-M.; Zuo, J. S-Nitrosylation Positively Regulates Ascorbate Peroxidase Activity during Plant Stress Responses. Plant Physiol. 2015, 167, 1604–1615. [Google Scholar] [CrossRef] [PubMed]
- Yun, B.-W.; Feechan, A.; Yin, M.; Saidi, N.B.B.; Le Bihan, T.; Yu, M.; Moore, J.W.; Kang, J.-G.; Kwon, E.; Spoel, S.H.; et al. S-nitrosylation of NADPH oxidase regulates cell death in plant immunity. Nature 2011, 478, 264–268. [Google Scholar] [CrossRef]
- Wang, Y.-Q.; Feechan, A.; Yun, B.-W.; Shafiei, R.; Hofmann, A.; Taylor, P.; Xue, P.; Yang, F.-Q.; Xie, Z.-S.; Pallas, J.A.; et al. S-Nitrosylation of AtSABP3 Antagonizes the Expression of Plant Immunity. J. Biol. Chem. 2009, 284, 2131–2137. [Google Scholar] [CrossRef]
- Tada, Y.; Spoel, S.H.; Pajerowska-Mukhtar, K.; Mou, Z.; Song, J.; Wang, C.; Zuo, J.; Dong, X. Plant Immunity Requires Conformational Charges of NPR1 via S-Nitrosylation and Thioredoxins. Science 2008, 321, 952–956. [Google Scholar] [CrossRef]
- Liu, M.; Cao, B.; Wei, J.-W.; Gong, B. Redesigning a S-nitrosylated pyruvate-dependent GABA transaminase 1 to generate high-malate and saline–alkali-tolerant tomato. New Phytol. 2024, 242, 2148–2162. [Google Scholar] [CrossRef]
- Liu, W.; Wei, J.-W.; Shan, Q.; Liu, M.; Xu, J.; Gong, B. Genetic engineering of drought- and salt-tolerant tomato via Δ1-pyrroline-5-carboxylate reductase S-nitrosylation. Plant Physiol. 2024, 195, 1038–1052. [Google Scholar] [CrossRef]
- Zeng, S.; Sun, X.; Zhai, J.; Li, X.; Pedro, G.-C.; Nian, H.; Li, K.; Xu, H. SlTrxh functions downstream of SlMYB86 and positively regulates nitrate stress tolerance via S-nitrosation in tomato seedling. Hortic. Res. 2024, 11, uhae184. [Google Scholar] [CrossRef]
- Liu, M.; Wei, J.-W.; Liu, W.; Gong, B. S-nitrosylation of ACO homolog 4 improves ethylene synthesis and salt tolerance in tomato. New Phytol. 2023, 239, 159–173. [Google Scholar] [CrossRef]
- Gao, X.; Ma, J.; Tie, J.; Li, Y.; Hu, L.; Yu, J. BR-Mediated Protein S-Nitrosylation Alleviated Low-Temperature Stress in Mini Chinese Cabbage (Brassica rapa ssp. pekinensis). Int. J. Mol. Sci. 2022, 23, 10964. [Google Scholar] [CrossRef]
- Yu, Z.; Cao, J.; Zhu, S.; Zhang, L.; Peng, Y.; Shi, J. Exogenous Nitric Oxide Enhances Disease Resistance by Nitrosylation and Inhibition of S-Nitrosoglutathione Reductase in Peach Fruit. Front. Plant Sci. 2020, 11, 543. [Google Scholar] [CrossRef] [PubMed]
- Leterrier, M.; Chaki, M.; Airaki, M.; Valderrama, R.; Palma, J.M.; Barroso, J.B.; Corpas, F.J. Function of S-nitrosoglutathione reductase (GSNOR) in plant development and under biotic/abiotic stress. Plant Signal. Behav. 2011, 6, 789–793. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Hou, X.; Wei, L.; Deng, Y.; Zhao, Z.; Liang, C.; Liao, W. Protein S-nitrosylation under abiotic stress: Role and mechanism. Plant Physiol. Biochem. 2024, 207, 108329. [Google Scholar] [CrossRef] [PubMed]
- Xuan, Y.; Zhou, S.; Wang, L.; Cheng, Y.; Zhao, L. Nitric Oxide Functions as a Signal and Acts Upstream of AtCaM3 in Thermotolerance in Arabidopsis Seedlings. Plant Physiol. 2010, 153, 1895–1906. [Google Scholar] [CrossRef]
- Lee, U.; Wie, C.; Fernandez, B.O.; Feelisch, M.; Vierling, E. Modulation of Nitrosative Stress by S-Nitrosoglutathione Reductase Is Critical for Thermotolerance and Plant Growth in Arabidopsis. Plant Cell 2008, 20, 786–802. [Google Scholar] [CrossRef]
- Song, X.; Wang, T.; Zhang, Y.; Yu, J.-Q.; Xia, X.-J. S-Nitrosoglutathione Reductase Contributes to Thermotolerance by Modulating High Temperature-Induced Apoplastic H2O2 in Solanum lycopersicum. Front. Plant Sci. 2022, 13, 862649. [Google Scholar] [CrossRef] [PubMed]
- Volkov, R.A.; Panchuk, I.I.; Mullineaux, P.M.; Schöffl, F. Heat stress-induced H2O2 is required for effective expression of heat shock genes in Arabidopsis. Plant Mol. Biol. 2006, 61, 733–746. [Google Scholar] [CrossRef] [PubMed]
- Chae, H.B.; Kim, M.G.; Kang, C.H.; Park, J.H.; Lee, E.S.; Lee, S.-U.; Chi, Y.H.; Paeng, S.K.; Bae, S.B.; Wi, S.D.; et al. Redox sensor QSOX1 regulates plant immunity by targeting GSNOR to modulate ROS generation. Mol. Plant 2021, 14, 1312–1327. [Google Scholar] [CrossRef] [PubMed]
- He, N.-Y.; Chen, L.-S.; Sun, A.-Z.; Zhao, Y.; Yin, S.-N.; Guo, F.-Q. A nitric oxide burst at the shoot apex triggers a heat-responsive pathway in Arabidopsis. Nat. Plants 2022, 8, 434–450. [Google Scholar] [CrossRef]
- Parankusam, S.; Adimulam, S.S.; Bhatnagar-Mathur, P.; Sharma, K.K. Nitric Oxide (NO) in Plant Heat Stress Tolerance: Current Knowledge and Perspectives. Front. Plant Sci. 2017, 8, 1582. [Google Scholar] [CrossRef]
- Cui, J.; Huang, M.; Qi, J.; Yu, W.; Li, C. Nitric Oxide in Plant Cold Stress: Functions, Mechanisms and Challenges. Agronomy 2025, 15, 1072. [Google Scholar] [CrossRef]
- Babuta, P.; Sougrakpam, Y.; Deswal, R. Nitric oxide cross-talks during low-temperature stress in plants. Plant Sci. 2025, 360, 112708. [Google Scholar] [CrossRef]
- Zhao, M.-G.; Chen, L.; Zhang, L.-L.; Zhang, W.-H. Nitric Reductase-Dependent Nitric Oxide Production Is Involved in Cold Acclimation and Freezing Tolerance in Arabidopsis. Plant Physiol. 2009, 151, 755–767. [Google Scholar] [CrossRef]
- Costa-Broseta, Á.; Perea-Resa, C.; Castillo, M.-C.; Ruíz, M.F.; Salinas, J.; León, J. Nitric Oxide Controls Constitutive Freezing Tolerance in Arabidopsis by Attenuating the Levels of Osmoprotectants, Stress-Related Hormones and Anthocyanins. Sci. Rep. 2018, 8, 9268. [Google Scholar] [CrossRef]
- Lv, X.; Ge, S.; Jalal Ahammed, G.; Xiang, X.; Guo, Z.; Yu, J.; Zhou, Y. Crosstalk between Nitric Oxide and MPK1/2 Mediates Cold Acclimation-induced Chilling Tolerance in Tomato. Plant Cell Physiol. 2017, 58, 1963–1975. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Ma, J.; Wang, G.; Huang, S.; Wu, X.; Hu, L.; Yu, J. The S-nitrosylation of monodehydroascorbate reductase positively regulated the low temperature tolerance of mini Chinese cabbage. Int. J. Biol. Macromol. 2024, 281, 136047. [Google Scholar] [CrossRef]
- Stamler, J.S.; Singel, D.J.; Loscalzo, J. Biochemistry of Nitric Oxide and Its Redox-Activated Forms. Science 1992, 258, 1898–1902. [Google Scholar] [CrossRef]
- Wink, D.A.; Mitchell, J.B. Chemical biology of nitric oxide: Insights into regulatory, cytotoxic, and cytoprotective mechanisms of nitric oxide. Free Radic. Biol. Med. 1998, 25, 434–456. [Google Scholar] [CrossRef]
- Dixon, S.J.; Stockwell, B.R. The role of iron and reactive oxygen species in cell death. Nat. Chem. Biol. 2014, 10, 9–17. [Google Scholar] [CrossRef]
- Li, B.; Sun, L.; Huang, J.; Göschl, C.; Shi, W.; Chory, J.; Busch, W. GSNOR provides plant tolerance to iron toxicity via preventing iron-dependent nitrosative and oxidative cytotoxicity. Nat. Commun. 2019, 10, 3896. [Google Scholar] [CrossRef]
- Wen, D.; Sun, S.; Yang, W.; Zhang, L.; Liu, S.; Gong, B.; Shi, Q. Overexpression of S-nitrosoglutathione reductase alleviated iron-deficiency stress by regulating iron distribution and redox homeostasis. J. Plant Physiol. 2019, 237, 1–11. [Google Scholar] [CrossRef]
- García, M.J.; Corpas, F.J.; Lucena, C.; Alcántara, E.; Pérez-Vicente, R.; Zamarreño, Á.M.; Bacaicoa, E.; García-Mina, J.M.; Bauer, P.; Romera, F.J. A Shoot Fe Signaling Pathway Requiring the OPT3 Transporter Controls GSNO Reductase and Ethylene in Arabidopsis thaliana Roots. Front. Plant Sci. 2018, 9, 1325. [Google Scholar] [CrossRef]
- Guan, M.; Zheng, X.; Zhu, Y. S-nitrosoglutathione reductase disfavors cadmium tolerance in shoots of Arabidopsis. Sci. Rep. 2024, 14, 26401. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Song, H.; Li, B.; Wang, M.; Di, D.; Lin, X.; Kronzucker, H.J.; Shi, W.; Li, G. Induction of S-nitrosoglutathione reductase protects root growth from ammonium toxicity by regulating potassium homeostasis in Arabidopsis and rice. J. Exp. Bot. 2021, 72, 4548–4564. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.; Al Azawi, T.N.I.; Pande, A.; Mun, B.-G.; Lee, D.-S.; Hussain, A.; Lee, B.-H.; Yun, B.-W. The Role of Nitric Oxide-Induced ATILL6 in Growth and Disease Resistance in Arabidopsis thaliana. Front. Plant Sci. 2021, 12, 685156. [Google Scholar] [CrossRef]
- Ageeva-Kieferle, A.; Georgii, E.; Winkler, B.; Ghirardo, A.; Albert, A.; Hüther, P.; Mengel, A.; Becker, C.; Schnitzler, J.-P.; Durner, J.; et al. Nitric oxide coordinates growth, development, and stress response via histone modification and gene expression. Plant Physiol. 2021, 187, 336–360. [Google Scholar] [CrossRef]
- Zhang, T.; Ma, M.; Chen, T.; Zhang, L.; Fan, L.; Zhang, W.; Wei, B.; Li, S.; Xuan, W.; Noctor, G.; et al. Glutathione-dependent denitrosation of GSNOR1 promotes oxidative signalling downstream of H2O2. Plant Cell Environ. 2020, 43, 1175–1191. [Google Scholar] [CrossRef]
- Chen, L.; Wu, R.; Feng, J.; Feng, T.; Wang, C.; Hu, J.; Zhan, N.; Li, Y.; Ma, X.; Ren, B.; et al. Transnitrosylation Mediated by the Non-canonical Catalase ROG1 Regulates Nitric Oxide Signaling in Plants. Dev. Cell 2020, 53, 444–457.e445. [Google Scholar] [CrossRef] [PubMed]
- Kolbert, Z.; Molnár, Á.; Oláh, D.; Feigl, G.; Horváth, E.; Erdei, L.; Ördög, A.; Rudolf, E.; Barth, T.; Lindermayr, C. S-Nitrosothiol Signaling Is involved in Regulating Hydrogen Peroxide Metabolism of Zinc-Stressed Arabidopsis. Plant Cell Physiol. 2019, 60, 2449–2463. [Google Scholar] [CrossRef]
- Guan, M.Y.; Zhu, Y.X.; Liu, X.X.; Jin, C.W. Induction of S-nitrosoglutathione reductase reduces root cadmium uptake by inhibiting Iron-regulated transporter 1. Plant Soil 2019, 438, 251–262. [Google Scholar] [CrossRef]
- Zhou, S.; Jia, L.; Chu, H.; Wu, D.; Peng, X.; Liu, X.; Zhang, J.; Zhao, J.; Chen, K.; Zhao, L. Arabidopsis CaM1 and CaM4 Promote Nitric Oxide Production and Salt Resistance by Inhibiting S-Nitrosoglutathione Reductase via Direct Binding. PLoS Genet. 2016, 12, e1006255. [Google Scholar] [CrossRef] [PubMed]
- Frungillo, L.; Skelly, M.J.; Loake, G.J.; Spoel, S.H.; Salgado, I. S-nitrosothiols regulate nitric oxide production and storage in plants through the nitrogen assimilation pathway. Nat. Commun. 2014, 5, 5401. [Google Scholar] [CrossRef]
- Nabi, R.B.S.; Tayade, R.; Hussain, A.; Kulkarni, K.P.; Imran, Q.M.; Mun, B.-G.; Yun, B.-W. Nitric oxide regulates plant responses to drought, salinity, and heavy metal stress. Environ. Exp. Bot. 2019, 161, 120–133. [Google Scholar] [CrossRef]
- Zulfiqar, F.; Ashraf, M. Proline Alleviates Abiotic Stress Induced Oxidative Stress in Plants. J. Plant Growth Regul. 2023, 42, 4629–4651. [Google Scholar] [CrossRef]
- Wang, M.; Dong, Y.; Yan, J.; Han, Q.; Li, K.; Xu, H. Overexpression of the spinach S-nitrosoglutathione reductase (SoGSNOR) in tobacco resulted in enhanced nitrate stress tolerance. Plant Cell Tissue Organ Cult. 2020, 143, 173–187. [Google Scholar] [CrossRef]
- Lay-Pruitt, K.S.; Takahashi, H. Integrating N signals and root growth: The role of nitrate transceptor NRT1.1 in auxin-mediated lateral root development. J. Exp. Bot. 2020, 71, 4365–4368. [Google Scholar] [CrossRef] [PubMed]
- Barth, C.; Gouzd, Z.A.; Steele, H.P.; Imperio, R.M. A mutation in GDP-mannose pyrophosphorylase causes conditional hypersensitivity to ammonium, resulting in Arabidopsis root growth inhibition, altered ammonium metabolism, and hormone homeostasis. J. Exp. Bot. 2009, 61, 379–394. [Google Scholar] [CrossRef]
- Sagardoy, R.; Morales, F.; López-Millán, A.-F.; Abadía, A.; Abadía, J. Effects of zinc toxicity on sugar beet (Beta vulgaris L.) plants grown in hydroponics. Plant Biol. 2009, 11, 339–350. [Google Scholar] [CrossRef] [PubMed]
- Feigl, G.; Lehotai, N.; Molnár, Á.; Ördög, A.; Rodríguez-Ruiz, M.; Palma, J.M.; Corpas, F.J.; Erdei, L.; Kolbert, Z. Zinc induces distinct changes in the metabolism of reactive oxygen and nitrogen species (ROS and RNS) in the roots of two Brassica species with different sensitivity to zinc stress. Ann. Bot. 2014, 116, 613–625. [Google Scholar] [CrossRef]
- Oláh, D.; Kondak, S.; Molnár, Á.; Adedokun, O.P.; Czékus, Z.; Gémes, K.; Galbács, G.; Kolbert, Z. Suboptimal zinc supply affects the S-nitrosoglutathione reductase enzyme and nitric oxide signaling in Arabidopsis. Plant Stress 2023, 10, 100250. [Google Scholar] [CrossRef]
- Gong, B.; Wen, D.; Bloszies, S.; Li, X.; Wei, M.; Yang, F.; Shi, Q.; Wang, X. Comparative effects of NaCl and NaHCO3 stresses on respiratory metabolism, antioxidant system, nutritional status, and organic acid metabolism in tomato roots. Acta Physiol. Plant. 2014, 36, 2167–2181. [Google Scholar] [CrossRef]
- Sharma, T.; Dreyer, I.; Kochian, L.; Piñeros, M.A. The ALMT Family of Organic Acid Transporters in Plants and Their Involvement in Detoxification and Nutrient Security. Front. Plant Sci. 2016, 7, 1488. [Google Scholar] [CrossRef]
- Gong, B.; Wen, D.; Wang, X.; Wei, M.; Yang, F.; Li, Y.; Shi, Q. S-Nitrosoglutathione Reductase-Modulated Redox Signaling Controls Sodic Alkaline Stress Responses in Solanum lycopersicum L. Plant Cell Physiol. 2014, 56, 790–802. [Google Scholar] [CrossRef]
- Wei, J.-W.; Liu, M.; Zhao, D.; Du, P.; Yan, L.; Liu, D.; Shi, Q.; Yang, C.; Qin, G.; Gong, B. Melatonin confers saline-alkali tolerance in tomato by alleviating nitrosative damage and S-nitrosylation of H+-ATPase 2. Plant Cell 2025, 37, koaf035. [Google Scholar] [CrossRef]
- Back, K.; Tan, D.-X.; Reiter, R.J. Melatonin biosynthesis in plants: Multiple pathways catalyze tryptophan to melatonin in the cytoplasm or chloroplasts. J. Pineal Res. 2016, 61, 426–437. [Google Scholar] [CrossRef] [PubMed]
- Voesenek, L.; Sasidharan, R.; Visser, E.J.; Bailey, J. Flooding stress signaling through perturbations in oxygen, ethylene, nitric oxide and light. New Phytol. 2016, 209, 39–43. [Google Scholar] [CrossRef]
- Pan, C.; Li, X.; Jian, C.; Zhou, Y.; Wang, A.; Xiao, D.; Zhan, J.; He, L. AhGSNOR1 negatively regulates Al-induced programmed cell death by regulating intracellular NO and redox levels. Plant Sci. 2024, 349, 112275. [Google Scholar] [CrossRef]
- Liu, R.; Zhu, T.; Chen, X.; Wang, Z.; Yang, Z.; Ren, A.; Shi, L.; Yu, H.; Zhao, M. GSNOR regulates ganoderic acid content in Ganoderma lucidum under heat stress through S-nitrosylation of catalase. Commun. Biol. 2022, 5, 32. [Google Scholar] [CrossRef]
- Zhang, X.; Du, H.; Shi, Q.; Gong, B. Loss of GSNOR increases abiotic stress sensitivity via regulating MAPK-ethylene cascade signaling in Solanum lycopersicum L. Environ. Exp. Bot. 2022, 199, 104872. [Google Scholar] [CrossRef]
- Lin, A.; Wang, Y.; Tang, J.; Xue, P.; Li, C.; Liu, L.; Hu, B.; Yang, F.; Loake, G.J.; Chu, C. Nitric Oxide and Protein S-Nitrosylation Are Integral to Hydrogen Peroxide-Induced Leaf Cell Death in Rice. Plant Physiol. 2011, 158, 451–464. [Google Scholar] [CrossRef]
- Shan, Q.; Zhao, D.; Cao, B.; Zhu, X.; Wang, C.; Deng, L.; Li, C.; Zhang, Y.; Shi, Q.; Gong, B. Jasmonic acid and nitric oxide orchestrate a hierarchical melatonin cascade for Botrytis cinerea resistance in tomato. Plant Physiol. 2025, 197, kiaf078. [Google Scholar] [CrossRef]
- Li, T.; Kang, J.; Zhang, H.; Wang, L.; Lu, M.; Cai, L.; Li, J.; Joosten, M.H.A.J.; Du, Y. Phytophthora Disrupts Plant Immunity by Manipulating Nitric Oxide Homeostasis Through GSNOR Inhibition. Adv. Sci. 2025, 12, e03633. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Cao, J.; Qiao, J.; Pan, J.; Zhang, S.; Li, Q.; Wang, Q.; Gong, B.; Shi, J. GABA keeps nitric oxide in balance by regulating GSNOR to enhance disease resistance of harvested tomato against Botrytis cinerea. Food Chem. 2022, 392, 133299. [Google Scholar] [CrossRef]
- Rasool, G.; Buchholz, G.; Yasmin, T.; Shabbir, G.; Abbasi, N.A.; Malik, S.I. Overexpression of SlGSNOR impairs In Vitro shoot proliferation and developmental architecture in tomato but confers enhanced disease resistance. J. Plant Physiol. 2021, 261, 153433. [Google Scholar] [CrossRef]
- Hussain, A.; Yun, B.W.; Kim, J.H.; Gupta, K.J.; Hyung, N.I.; Loake, G.J. Novel and conserved functions of S-nitrosoglutathione reductase in tomato. J. Exp. Bot. 2019, 70, 4877–4886. [Google Scholar] [CrossRef]
- Thalineau, E.; Truong, H.-N.; Berger, A.; Fournier, C.; Boscari, A.; Wendehenne, D.; Jeandroz, S. Cross-Regulation between N Metabolism and Nitric Oxide (NO) Signaling during Plant Immunity. Front. Plant Sci. 2016, 7, 472. [Google Scholar] [CrossRef]
- Lindermayr, C.; Saalbach, G.; Durner, J.r. Proteomic Identification of S-Nitrosylated Proteins in Arabidopsis. Plant Physiol. 2005, 137, 921–930. [Google Scholar] [CrossRef]
- Romero-Puertas, M.C.; Campostrini, N.; Mattè, A.; Righetti, P.G.; Perazzolli, M.; Zolla, L.; Roepstorff, P.; Delledonne, M. Proteomic analysis of S-nitrosylated proteins in Arabidopsis thaliana undergoing hypersensitive response. Proteomics 2008, 8, 1459–1469. [Google Scholar] [CrossRef]
- Imran, Q.M.; Falak, N.; Hussain, A.; Mun, B.-G.; Sharma, A.; Lee, S.-U.; Kim, K.-M.; Yun, B.-W. Nitric Oxide Responsive Heavy Metal-Associated Gene AtHMAD1 Contributes to Development and Disease Resistance in Arabidopsis thaliana. Front. Plant Sci. 2016, 7, 1712. [Google Scholar] [CrossRef] [PubMed]
- Cui, B.; Pan, Q.; Clarke, D.; Villarreal, M.O.; Umbreen, S.; Yuan, B.; Shan, W.; Jiang, J.; Loake, G.J. S-nitrosylation of the zinc finger protein SRG1 regulates plant immunity. Nat. Commun. 2018, 9, 4226. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.; Imran, Q.M.; Shahid, M.; Mun, B.-G.; Lee, S.-U.; Khan, M.A.; Hussain, A.; Lee, I.-J.; Yun, B.-W. Nitric oxide- induced AtAO3 differentially regulates plant defense and drought tolerance in Arabidopsis thaliana. BMC Plant Biol. 2019, 19, 602. [Google Scholar] [CrossRef]
- Cui, B.; Ma, X.; Li, Y.; Zhou, Y.; Ju, X.; Hussain, A.; Umbreen, S.; Yuan, B.; Tabassum, A.; Lubega, J.; et al. Perturbations in nitric oxide homeostasis promote Arabidopsis disease susceptibility towards Phytophthora parasitica. Mol. Plant Pathol. 2021, 22, 1134–1148. [Google Scholar] [CrossRef] [PubMed]
- Hurali, D.T.; Bhurta, R.; Tyagi, S.; Sathee, L.; Sandeep, A.B.; Singh, D.; Mallick, N.; Vinod; Jha, S.K. Analysis of NIA and GSNOR family genes and nitric oxide homeostasis in response to wheat-leaf rust interaction. Sci. Rep. 2022, 12, 803. [Google Scholar] [CrossRef]
- Wünsche, H.; Baldwin, I.T.; Wu, J. S-Nitrosoglutathione reductase (GSNOR) mediates the biosynthesis of jasmonic acid and ethylene induced by feeding of the insect herbivore Manduca sexta and is important for jasmonate-elicited responses in Nicotiana attenuata. J. Exp. Bot. 2011, 62, 4605–4616. [Google Scholar] [CrossRef]
- Yun, B.-W.; Skelly, M.J.; Yin, M.; Yu, M.; Mun, B.-G.; Lee, S.-U.; Hussain, A.; Spoel, S.H.; Loake, G.J. Nitric oxide and S-nitrosoglutathione function additively during plant immunity. New Phytol. 2016, 211, 516–526. [Google Scholar] [CrossRef] [PubMed]
- Holzmeister, C.; Fröhlich, A.; Sarioglu, H.; Bauer, N.; Durner, J.; Lindermayr, C. Proteomic analysis of defense response of wildtype Arabidopsis thaliana and plants with impaired NO-homeostasis. Proteomics 2011, 11, 1664–1683. [Google Scholar] [CrossRef] [PubMed]
- Aslam, M.M.; Waseem, M.; Felix, A.; Shaheen, I. Nitrogen Uptake and Utilization Toward Sustainable Agriculture Development. In Nitric Oxide in Plants: A Molecule with Dual Roles; John Wiley & Sons: Hoboken, NJ, USA, 2022; pp. 78–94. [Google Scholar]
- Pramanik, P.; Krishnan, P.; Maity, A.; Mridha, N.; Mukherjee, A.; Rai, V. Application of Nanotechnology in Agriculture. In Environmental Nanotechnology Volume 4; Dasgupta, N., Ranjan, S., Lichtfouse, E., Eds.; Springer International Publishing: Cham, Switzerland, 2020; pp. 317–348. [Google Scholar]
- de Souza, G.F.P.; Denadai, J.P.; Picheth, G.F.; de Oliveira, M.G. Long-term decomposition of aqueous S-nitrosoglutathione and S-nitroso-N-acetylcysteine: Influence of concentration, temperature, pH and light. Nitric Oxide 2019, 84, 30–37. [Google Scholar] [CrossRef]
- Manoj, V.M.; Aravindakumar, C.T. Hydroxyl radical induced decomposition of S-nitrosoglutathione. Chem. Commun. 2000, 2361–2362. [Google Scholar] [CrossRef]
- Trujillo, M.; Alvarez, M.a.N.; Peluffo, G.; Freeman, B.A.; Radi, R. Xanthine Oxidase-mediated Decomposition of S-Nitrosothiols. J. Biol. Chem. 1998, 273, 7828–7834. [Google Scholar] [CrossRef]
- Seabra, A.B.; Silveira, N.M.; Ribeiro, R.V.; Pieretti, J.C.; Barroso, J.B.; Corpas, F.J.; Palma, J.M.; Hancock, J.T.; Petřivalský, M.; Gupta, K.J.; et al. Nitric oxide-releasing nanomaterials: From basic research to potential biotechnological applications in agriculture. New Phytol. 2022, 234, 1119–1125. [Google Scholar] [CrossRef]
- Pereira, A.; Narciso, A.; Seabra, A.; Fraceto, L. Evaluation of the effects of nitric oxide-releasing nanoparticles on plants. J. Phys. Conf. Ser. 2015, 617, 012025. [Google Scholar] [CrossRef]
- Oliveira, H.C.; Gomes, B.C.R.; Pelegrino, M.T.; Seabra, A.B. Nitric oxide-releasing chitosan nanoparticles alleviate the effects of salt stress in maize plants. Nitric Oxide 2016, 61, 10–19. [Google Scholar] [CrossRef]
- Methela, N.J.; Pande, A.; Islam, M.S.; Rahim, W.; Hussain, A.; Lee, D.-S.; Mun, B.-G.; Maria Joseph Raj, N.P.; Kim, S.-J.; Kim, Y.; et al. Chitosan-GSNO nanoparticles: A positive modulator of drought stress tolerance in soybean. BMC Plant Biol. 2023, 23, 639. [Google Scholar] [CrossRef] [PubMed]
- Kondak, D.; Deák, Á.; Rónavári, A.; Bodor, T.; Kondak, S.; Adedokun, O.P.; Benkő, P.; Szőllősi, R.; Szalai, G.; Janda, T.; et al. Chitosan encapsulated S-nitrosoglutathione is an efficient nanodonor in Brassica napus seedlings. Plant Physiol. Biochem. 2025, 227, 110184. [Google Scholar] [CrossRef]
- Cardozo, V.F.; Lancheros, C.A.C.; Narciso, A.M.; Valereto, E.C.S.; Kobayashi, R.K.T.; Seabra, A.B.; Nakazato, G. Evaluation of antibacterial activity of nitric oxide-releasing polymeric particles against Staphylococcus aureus and Escherichia coli from bovine mastitis. Int. J. Pharm. 2014, 473, 20–29. [Google Scholar] [CrossRef] [PubMed]
- Silveira, N.M.; Seabra, A.B.; Marcos, F.C.C.; Pelegrino, M.T.; Machado, E.C.; Ribeiro, R.V. Encapsulation of S-nitrosoglutathione into chitosan nanoparticles improves drought tolerance of sugarcane plants. Nitric Oxide 2019, 84, 38–44. [Google Scholar] [CrossRef]
- Ma, Y.; Fu, L.; Hussain, Z.; Huang, D.; Zhu, S. Enhancement of storability and antioxidant systems of sweet cherry fruit by nitric oxide-releasing chitosan nanoparticles (GSNO-CS NPs). Food Chem. 2019, 285, 10–21. [Google Scholar] [CrossRef]
- Karn, B.; Kuiken, T.; Otto, M. Nanotechnology and In Situ Remediation: A Review of the Benefits and Potential Risks. Environ. Health Perspect. 2009, 117, 1813–1831. [Google Scholar] [CrossRef]
- Santos, P.A.; Biraku, X.; Nielsen, E.; Ozketen, A.C.; Ozketen, A.A.; Hakki, E.E. Agricultural nanotechnology for a safe and sustainable future: Current status, challenges, and beyond. J. Sci. Food Agric. 2025, 105, 3159–3169. [Google Scholar] [CrossRef]
- Yadav, A.; Yadav, K. Exploring the Effect of Engineered Nanomaterials on Soil Microbial Diversity and Functions: A Review. J. Environ. Nanotechnol. 2024, 13, 48–62. [Google Scholar] [CrossRef]
- Jafar, G.; Hamzeh, G. Ecotoxicity of nanomaterials in soil. Ann. Biol. Res. 2013, 4, 86–92. [Google Scholar]
- Colman, B.P.; Arnaout, C.L.; Anciaux, S.; Gunsch, C.K.; Hochella, M.F., Jr.; Kim, B.; Lowry, G.V.; McGill, B.M.; Reinsch, B.C.; Richardson, C.J.; et al. Low Concentrations of Silver Nanoparticles in Biosolids Cause Adverse Ecosystem Responses under Realistic Field Scenario. PLoS ONE 2013, 8, e57189. [Google Scholar] [CrossRef] [PubMed]
- Yang, P.; Zhang, L.; Huang, Y.; Ouyang, Z. Research status on soil and water pollution remediation and environmental impact of nanomaterials. IOP Conf. Ser. Earth Environ. Sci. 2019, 371, 032015. [Google Scholar] [CrossRef]
- Kumar, R.; Kishan Singh, R.; Mishra, A.K. Nanoparticles in the soil environment and their behaviour: An overview. J. Appl. Nat. Sci. 2012, 4, 310–324. [Google Scholar] [CrossRef]
- Lewis, R.W.; Bertsch, P.M.; McNear, D.H. Nanotoxicity of engineered nanomaterials (ENMs) to environmentally relevant beneficial soil bacteria—A critical review. Nanotoxicology 2019, 13, 392–428. [Google Scholar] [CrossRef]
- Gupta, R.; Xie, H. Nanoparticles in daily life: Applications, toxicity and regulations. J. Environ. Pathol. Toxicol. Oncol. 2018, 37, 209–230. [Google Scholar] [CrossRef]
- Kaya, C.; Uğurlar, F.; Seth, C.S. Sodium nitroprusside modulates oxidative and nitrosative processes in Lycopersicum esculentum L. under drought stress. Plant Cell Rep. 2024, 43, 152, Correction in Plant Cell Rep. 2025, 44, 145. https://doi.org/10.1007/s00299-025-03529-3. [Google Scholar] [CrossRef] [PubMed]
- Ghita, R.V.; Predoi, D.; Iconaru, S.L.; Cimpeanu, C.L.; Raita, S.M. Application of Nanotechnology Solutions in Plants Fertilization. In Urban Horticulture-Necessity of the Future; Solankey, S.S., Akhtar, S., Luna Maldonado, A.I., Rodriguez-Fuentes, H., Vidales Contreras, J.A., Reyes, J.M.M., Eds.; IntechOpen: London, UK, 2020. [Google Scholar][Green Version]
- Yadav, A.; Yadav, K.; Ahmad, R.; Abd-Elsalam, K.A. Emerging Frontiers in Nanotechnology for Precision Agriculture: Advancements, Hurdles and Prospects. Agrochemicals 2023, 2, 220–256. [Google Scholar] [CrossRef]
- Klaine, S.J.; Koelmans, A.A.; Horne, N.; Carley, S.; Handy, R.D.; Kapustka, L.; Nowack, B.; von der Kammer, F. Paradigms to assess the environmental impact of manufactured nanomaterials. Environ. Toxicol. Chem. 2012, 31, 3–14. [Google Scholar] [CrossRef] [PubMed]
- Pandey, G.; Tripathi, S.; Bajpai, S.; Kamboj, M. Approaches, Challenges, and Prospects of Nanotechnology for Sustainable Agriculture. In Agricultural and Environmental Nanotechnology: Novel Technologies and Their Ecological Impact; Fernandez-Luqueno, F., Patra, J.K., Eds.; Springer Nature Singapore: Singapore, 2023; pp. 83–103. [Google Scholar]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Das, A.K.; Lee, D.-S.; Lee, G.-J.; Kim, Y.-S.; Hussain, S.; Lee, M.-S.; Yun, B.-W.; Mun, B.-G. The Central Role of GSNOR: Decoding Nitric Oxide Signaling for Crop Stress Tolerance. Int. J. Mol. Sci. 2025, 26, 11486. https://doi.org/10.3390/ijms262311486
Das AK, Lee D-S, Lee G-J, Kim Y-S, Hussain S, Lee M-S, Yun B-W, Mun B-G. The Central Role of GSNOR: Decoding Nitric Oxide Signaling for Crop Stress Tolerance. International Journal of Molecular Sciences. 2025; 26(23):11486. https://doi.org/10.3390/ijms262311486
Chicago/Turabian StyleDas, Ashim Kumar, Da-Sol Lee, Geum-Jin Lee, Ye-Song Kim, Sajeel Hussain, Moon-Sub Lee, Byung-Wook Yun, and Bong-Gyu Mun. 2025. "The Central Role of GSNOR: Decoding Nitric Oxide Signaling for Crop Stress Tolerance" International Journal of Molecular Sciences 26, no. 23: 11486. https://doi.org/10.3390/ijms262311486
APA StyleDas, A. K., Lee, D.-S., Lee, G.-J., Kim, Y.-S., Hussain, S., Lee, M.-S., Yun, B.-W., & Mun, B.-G. (2025). The Central Role of GSNOR: Decoding Nitric Oxide Signaling for Crop Stress Tolerance. International Journal of Molecular Sciences, 26(23), 11486. https://doi.org/10.3390/ijms262311486






