Targeted Sequencing Identifies SNPs Associated with Antimalarial Drug Resistance and the CSP Vaccine Antigen in Plasmodium falciparum from Southwest Cameroon
Abstract
1. Introduction
2. Results
2.1. General Characteristics of Study Population
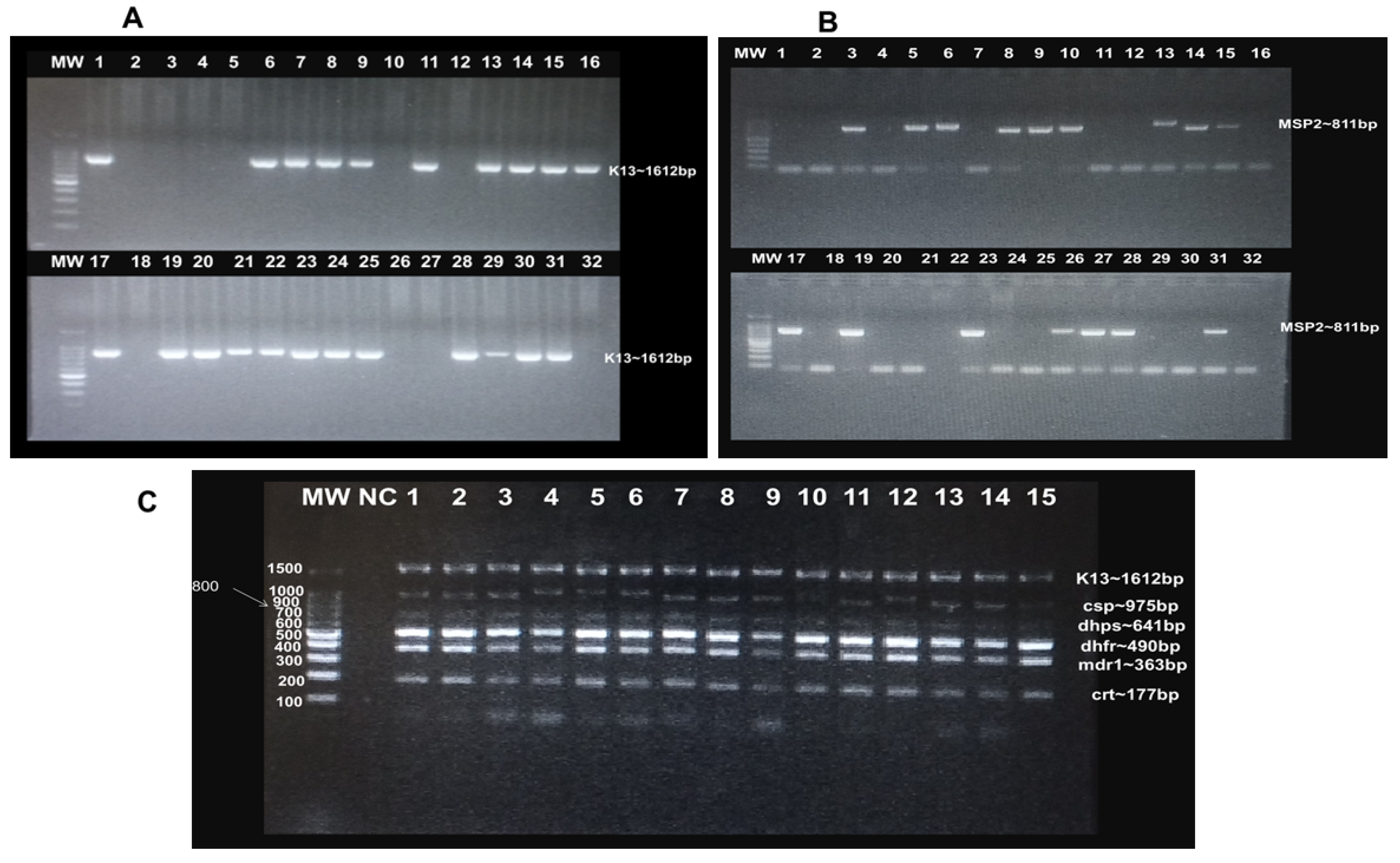
2.2. Screening of Samples and Polymerase Chain Reaction (PCR) Amplification of pfkelch13, pfdhfr, pfdhps, pfcrt, pfmdr1, and pfcsp Gene Targets
2.3. Targeted Sequencing Identified Single-Nucleotide Polymorphisms in Gene Targets
2.4. Frequency of Identified Single-Nucleotide Polymorphisms Associated with Drug Resistance
2.5. Frequency of SNPs in the P. falciparum Circumsporozoite Protein That May Impact Malaria Vaccine Efficacy
3. Discussion
4. Materials and Methods
4.1. Study Design and Ethical Considerations
4.2. Study Site and Sample Collection
4.3. Malaria Parasitemia Determination
4.4. DNA Extraction and Purification
4.5. Molecular Identification of Actively Infected Samples and Amplification of Drug-Resistance and Vaccine Gene Targets
4.6. Confirmation of PCR Amplicons by Agarose Gel Electrophoresis
4.7. Purification and Quantification of Amplicons
4.8. Targeted Sequencing to Identify Single-Nucleotide Polymorphisms
4.9. Bioinformatics Analysis
4.10. Data Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| SNP | Single-nucleotide polymorphism |
| ACT | Artemisinin-based combination therapy |
| pfcrt | Plasmodium falciparum chloroquine resistant transporter |
| pfdhfr | Plasmodium falciparum dihydrofolate reductase |
| pfdhps | Plasmodium falciparum dihydropteroate synthase |
| pfmdr1 | Plasmodium falciparum multidrug resistance 1 |
| pfcsp | Plasmodium falciparum circumsporozoite protein |
| PCR | Polymerase chain reaction |
| ONT | Oxford Nanopore technology |
| SP | Sulfadoxine-pyrimethamine |
| EPI | Expanded Programme on Immunization |
| AL | Artemether-lumefantrine |
| ASAQ | Artesunate-amodiaquine |
| BTB/POZ | Bric-a-brac, Tramtrack, and Broad-complex/Poxvirus and zinc finger |
| WHO | World Health Organization |
| CTR | C-terminal region |
| GTS | Global Technical Strategy |
| RDT | Rapid Diagnostic Test |
| RTS,S/AS01 | Repeat T epitopes and Surface antigen |
| IPTp | Intermittent preventive treatment of malaria in pregnancy |
| IPTi | Intermittent preventive treatment of infants |
| SMC | Seasonal malaria chemoprevention |
Appendix A
Appendix A.1
| Gene Target | Primer Sequence (Forward and Reverse) | Reaction Conditions | Reaction Components |
|---|---|---|---|
| MSP2 | 5′-ATG AAG GTA ATT AAA ACA TTG TCT ATT-3′ 5′-ATA TGG CAA AAG ATA AAA CAA GTG TTG-3′ | 94 °C 3 min, 94 °C 30 s, 55 °C 1 min, 72 °C 1 min, 72 °C 4 min, 4 °C ∞, 40× | Go taq (7.5 µL), F/R primer (2 µL), nfH2O (3.5 µL), gDNA (3 µL), Total volume = 15 µL |
| kelch13 ampl | 5′-GGG AAT CTG GTG GTA ACA GC-3′ 5′-CGG AGT GAC CAA ATC TGG GA-3′ | 94 °C 3 min; 94 °C 30 s; 61 °C 1 min 30 s; 72 °C 1 min 30 s; 72 °C 10 min, 4 °C ∞; 40× | |
| Kelch13 nested | 5′-AAG CCT TGT TGA AAG AAG CA-3′ 5′-GGG AAC TAA TAA AGA TGG GC-3′ | ||
| Pfcsp | 5′-TGG GAA ACA GGA AAA TTG GTA T-3′ 5′-TAC GAC ATT AAA CAC ACT GGA A-3′ | 94 °C 5 min; 94 °C 30 s; 58 °C 1 min; 72 °C 1 min 30 s; 72 °C 5 min, 4 °C ∞; 40× | Go taq (12.5 µL), F/R primer (csp 2 µL; dhfr 1.2 µL; dhps2 µL), nfH2O (3.3 µL), gDNA (4 µL), Total volume = 25 µL |
| Pfdhfr | 5′-GTT TTC GAT ATT TAT GCC ATA TGT G-3′ 5′-TGA TAA ACA ACG GAA CCT CC-3′ | ||
| Pfdhps | 5′-TTT GTT GAA CCT AAA CGT GC-3′ 5′AAC ATT TTG ATC ATT CAT GCA AT-3′ | ||
| Pfmdr1 | 5′-TGT GTT TGG TGT AAT ATT AAA GAA CA-3′ 5′-ACA TAA AGT CAA ACG TGC ATT T-3′ | Go taq (7.5 µL), F/R primer (2 µL) nfH2O (3.5 µL), gDNA (2 µL), Total volume = 25 µL | |
| Pfcrt | 5′-TGT CTT GGT AAA CAC GCT CA-3′ 5′-AGT TGA GAG TTT CGG ATG TT-3′ |
Appendix A.2
| Chromosome | Length | Variants | Variants Rate |
|---|---|---|---|
| Pf3D7_03_v3 | 1,067,971 | 82 | 13,024 |
| Pf3D7_04_v3 | 1,200,490 | 3 | 400,163 |
| Pf3D7_05_v3 | 1,343,557 | 2 | 671,778 |
| Pf3D7_07_v3 | 1,445,207 | 3 | 481,735 |
| Pf3D7_08_v3 | 1,472,805 | 6 | 245,467 |
| Pf3D7_13_v3 | 2,925,236 | 3 | 1,462,618 |
| Total | 9,455,266 | 98 | 96,482 |
Appendix B

References
- World Health Organization. WHO 2024 Malaria Report: Addressing Inequity in the Global Malaria Response. Available online: https://www.who.int/teams/global-malaria-programme/reports/world-malaria-report-2024 (accessed on 26 March 2025).
- de Fatima Ferreira-da-Cruz, M.; Almeida-de-Oliveira, N.K.; de Abreu-Fernandes, R.; de Lavigne Mello, A.R.; Zanini, G.; de Abreu, F.V.S.; de Pina-Costa, A.; Lourenco-de-Oliveira, R.; Brasil, P.; Daniel-Ribeiro, C.T.; et al. Simium zoonotic malaria infection in the Rio de Janeiro Atlantic forest, Brazil. Sci. Rep. 2025, 15, 23695. [Google Scholar] [CrossRef] [PubMed]
- Andrade, M.V.; Noronha, K.; Diniz, B.P.C.; Guedes, G.; Carvalho, L.R.; Silva, V.A.; Calazans, J.A.; Santos, A.S.; Silva, D.N.; Castro, M.C. The economic burden of malaria: A systematic review. Malar. J. 2022, 21, 283. [Google Scholar] [CrossRef]
- Sachs, J.; Malaney, P. The economic and social burden of malaria. Nature 2002, 415, 680–685. [Google Scholar] [CrossRef]
- Ricci, F. Social implications of malaria and their relationships with poverty. Mediterr. J. Hematol. Infect. Dis. 2012, 4, e2012048. [Google Scholar] [CrossRef]
- World Health Organization World Malaria Report. Available online: https://www.who.int/teams/global-malaria-programme/reports/world-malaria-report-2023 (accessed on 26 March 2025).
- Sarfo, J.O.; Amoadu, M.; Kordorwu, P.Y.; Adams, A.K.; Gyan, T.B.; Osman, A.G.; Asiedu, I.; Ansah, E.W. Malaria amongst children under five in sub-Saharan Africa: A scoping review of prevalence, risk factors and preventive interventions. Eur. J. Med. Res. 2023, 28, 80. [Google Scholar] [CrossRef] [PubMed]
- U.S. President’s Malaria Initiative Cameroon Malaria Profile Cameroon-Country-Profile. Available online: https://mesamalaria.org/wp-content/uploads/2025/04/CAMEROON_Malaria_Profile_PMI_FY_2024.pdf (accessed on 14 September 2025).
- National Malaria Control Programme. Cameroon Malaria Indicators Survey. Available online: https://dhsprogram.com/pubs/pdf/PR145/PR145.pdf (accessed on 14 September 2025).
- World Health Organization World Malaria Report: Tracking Progress and Gaps in the Global Response to Malaria. Available online: https://www.who.int/teams/global-malaria-programme/reports/world-malaria-report-2022 (accessed on 26 August 2025).
- El-Moamly, A.A.; El-Sweify, M.A. Malaria vaccines: The 60-year journey of hope and final success-lessons learned and future prospects. Trop. Med. Health 2023, 51, 29. [Google Scholar] [CrossRef] [PubMed]
- The RTS,S Clinical Trials Partnership. Efficacy and Safety of the RTS,S/AS01 Malaria Vaccine during 18 Months after Vaccination: A Phase 3 Randomized, Controlled Trial in Children and Young Infants at 11 African Sites. PLoS Med. 2014, 11, e1001685. [Google Scholar]
- Datoo, M.S.; Natama, H.M.; Somé, A.; Bellamy, D.; Traoré, O.; Rouamba, T.; Tahita, M.C.; Ido, N.F.A.; Yameogo, P.; Valia, D.; et al. Efficacy and immunogenicity of R21/Matrix-M vaccine against clinical malaria after 2 years’ follow-up in children in Burkina Faso: A phase 1/2b randomised controlled trial. Lancet Infect. Dis. 2022, 22, 1728–1736. [Google Scholar] [CrossRef]
- Ndoula, S.T.; Mboussou, F.; Njoh, A.A.; Nembot, R.; Baonga, S.F.; Njinkeu, A.; Biey, J.; Kaba, M.I.; Amani, A.; Farham, B.; et al. Malaria Vaccine Introduction in Cameroon: Early Results 30 Days into Rollout. Vaccines 2024, 12, 346. [Google Scholar] [CrossRef]
- World Health Organization Cameroon Kicks Malaria Vaccine Rollout. Available online: https://www.afro.who.int/countries/cameroon/news/cameroon-kicks-malaria-vaccine-rollout (accessed on 2 September 2024).
- Lyimo, B.M.; Bakari, C.; Popkin-Hall, Z.R.; Giesbrecht, D.J.; Seth, M.D.; Pereus, D.; Shabani, Z.I.; Moshi, R.; Boniface, R.; Mandara, C.I.; et al. Genetic polymorphism and evidence of signatures of selection in the Plasmodium falciparum circumsporozoite protein gene in Tanzanian regions with different malaria endemicity. Malar. J. 2024, 23, 139. [Google Scholar] [CrossRef]
- Amegashie, E.A.; Amenga-Etego, L.; Adobor, C.; Ogoti, P.; Mbogo, K.; Amambua-Ngwa, A.; Ghansah, A. Population genetic analysis of the Plasmodium falciparum circumsporozoite protein in two distinct ecological regions in Ghana. Malar. J. 2020, 19, 437. [Google Scholar] [CrossRef]
- Huang, H.Y.; Liang, X.Y.; Lin, L.Y.; Chen, J.T.; Ehapo, C.S.; Eyi, U.M.; Li, J.; Jiang, T.T.; Zheng, Y.Z.; Zha, G.C.; et al. Genetic polymorphism of Plasmodium falciparum circumsporozoite protein on Bioko Island, Equatorial Guinea and global comparative analysis. Malar. J. 2020, 19, 245. [Google Scholar] [CrossRef]
- He, Z.Q.; Zhang, Q.Q.; Wang, D.; Hu, Y.B.; Zhou, R.M.; Qian, D.; Yang, C.Y.; Lu, D.L.; Li, S.H.; Liu, Y.; et al. Genetic polymorphism of circumsporozoite protein of Plasmodium falciparum among Chinese migrant workers returning from Africa to Henan Province. Malar. J. 2022, 21, 248. [Google Scholar] [CrossRef]
- Maina, M.; Musundi, S.; Kuja, J.; Waweru, H.; Kiboi, D.; Kanoi, B.N.; Gitaka, J. Genetic variation of the Plasmodium falciparum circumsporozoite protein in parasite isolates from Homabay County in Kenya. Front. Parasitol. 2024, 3, 1346017. [Google Scholar] [CrossRef]
- Dieng, C.C.; Ford, C.T.; Lerch, A.; Doniou, D.; Vegesna, K.; Janies, D.; Cui, L.; Amoah, L.; Afrane, Y.; Lo, E. Genetic variations of Plasmodium falciparum circumsporozoite protein and the impact on interactions with human immunoproteins and malaria vaccine efficacy. Infect. Genet. Evol. 2023, 110, 105418. [Google Scholar] [CrossRef]
- Wicht, K.J.; Mok, S.; Fidock, D.A. Molecular Mechanisms of Drug Resistance in Plasmodium falciparum Malaria. Annu. Rev. Microbiol. 2020, 74, 431–454. [Google Scholar] [CrossRef] [PubMed]
- Lei, Z.N.; Wu, Z.X.; Dong, S.; Yang, D.H.; Zhang, L.; Ke, Z.; Zou, C.; Chen, Z.S. Chloroquine and hydroxychloroquine in the treatment of malaria and repurposing in treating COVID-19. Pharmacol. Ther. 2020, 216, 107672. [Google Scholar] [CrossRef]
- Amambua-Ngwa, A.; Button-Simons, K.A.; Li, X.; Kumar, S.; Brenneman, K.V.; Ferrari, M.; Checkley, L.A.; Haile, M.T.; Shoue, D.A.; McDew-White, M.; et al. Chloroquine resistance evolution in Plasmodium falciparum is mediated by the putative amino acid transporter AAT1. Nat. Microbiol. 2023, 8, 1213–1226. [Google Scholar] [CrossRef] [PubMed]
- Sidhu, A.B.; Verdier-Pinard, D.; Fidock, D.A. Chloroquine resistance in Plasmodium falciparum malaria parasites conferred by pfcrt mutations. Science 2002, 298, 210–213. [Google Scholar] [CrossRef] [PubMed]
- Miriam, K.L.; Christopher, V.P. Withdrawing antimalarial drugs: Impact on parasite resistance and implications for malaria treatment policies, Drug Resistance Updates. Drug Resist. Updates 2004, 7, 279–288. [Google Scholar]
- Sayang, C.; Gausseres, M.; Vernazza-Licht, N.; Malvy, D.; Bley, D.; Millet, P. Treatment of malaria from monotherapy to artemisinin-based combination therapy by health professionals in urban health facilities in Yaounde, central province, Cameroon. Malar. J. 2009, 8, 176. [Google Scholar] [CrossRef]
- Niba, P.T.N.; Nji, A.M.; Evehe, M.-S.; Ali, I.M.; Netongo, P.M.; Ngwafor, R.; Moyeh, M.N.; Ngum, L.N.; Ndum, O.E.; Acho, F.A.; et al. Drug resistance markers within an evolving efficacy of anti-malarial drugs in Cameroon: A systematic review and meta-analysis (1998–2020). Malar. J. 2021, 20, 32. [Google Scholar] [CrossRef]
- Lakshmanan, V.; Bray, P.G.; Verdier-Pinard, D.; Johnson, D.J.; Horrocks, P.; Muhle, R.A.; Alakpa, G.E.; Hughes, R.H.; Ward, S.A.; Krogstad, D.J.; et al. A critical role for PfCRT K76T in Plasmodium falciparum verapamil-reversible chloroquine resistance. EMBO J. 2005, 24, 2294–2305. [Google Scholar] [CrossRef] [PubMed]
- Tchuenkam, P.V.K.; Ngum, L.N.; Ali, I.M.; Chedjou, J.P.K.; Nji, A.M.; Netongo, P.M.; Ngwafor, R.; Niba, P.T.N.; Tah, C.F.; Nana, W.D.; et al. Plasmodium falciparum dhps and dhfr markers of resistance to sulfadoxine-pyrimethamine five years (2016–2020) after the implementation of seasonal malaria chemoprevention in Cameroon. Wellcome Open Res. 2024, 9, 323. [Google Scholar] [CrossRef]
- Deloron, P.; Bertin, G.; Briand, V.; Massougbodji, A.; Cot, M. Sulfadoxine/pyrimethamine intermittent preventive treatment for malaria during pregnancy. Emerg. Infect. Dis. 2010, 16, 1666–1670. [Google Scholar] [CrossRef] [PubMed]
- Amimo, F.; Lambert, B.; Magit, A.; Sacarlal, J.; Hashizume, M.; Shibuya, K. Plasmodium falciparum resistance to sulfadoxine-pyrimethamine in Africa: A systematic analysis of national trends. BMJ Glob. Health 2020, 5, e003217. [Google Scholar] [CrossRef]
- Guemas, E.; Coppee, R.; Menard, S.; du Manoir, M.; Nsango, S.; Makaba Mvumbi, D.; Nakoune, E.; Eboumbou Moukoko, C.E.; Bouyou Akotet, M.K.; Mirabeau, T.Y.; et al. Evolution and spread of Plasmodium falciparum mutations associated with resistance to sulfadoxine-pyrimethamine in central Africa: A cross-sectional study. Lancet Microbe 2023, 4, e983–e993. [Google Scholar] [CrossRef] [PubMed]
- Gatton, M.L.; Martin, L.B.; Cheng, Q. Evolution of resistance to sulfadoxine-pyrimethamine in Plasmodium falciparum. Antimicrob. Agents Chemother. 2004, 48, 2116–2123. [Google Scholar] [CrossRef]
- Flegg, J.A.; Metcalf, C.J.E.; Gharbi, M.; Venkatesan, M.; Shewchuk, T.; Hopkins Sibley, C.; Guerin, P.J. Trends in antimalarial drug use in Africa. Am. J. Trop. Med. Hyg. 2013, 89, 857–865. [Google Scholar] [CrossRef]
- Hassett, M.R.; Roepe, P.D. Origin and Spread of Evolving Artemisinin-Resistant Plasmodium falciparum Malarial Parasites in Southeast Asia. Am. J. Trop. Med. Hyg. 2019, 101, 1204–1211. [Google Scholar] [CrossRef]
- Nji, A.M.; Ali, I.M.; Moyeh, M.N.; Ngongang, E.O.; Ekollo, A.M.; Chedjou, J.P.; Ndikum, V.N.; Evehe, M.S.; Froeschl, G.; Heumann, C.; et al. Randomized non-inferiority and safety trial of dihydroartemisin-piperaquine and artesunate-amodiaquine versus artemether-lumefantrine in the treatment of uncomplicated Plasmodium falciparum malaria in Cameroonian children. Malar. J. 2015, 14, 27. [Google Scholar] [CrossRef]
- Assefa, A.; Fola, A.A.; Tasew, G. Emergence of Plasmodium falciparum strains with artemisinin partial resistance in East Africa and the Horn of Africa: Is there a need to panic? Malar. J. 2024, 23, 34. [Google Scholar] [CrossRef]
- Breman, J.G. Resistance to artemisinin-based combination therapy. Lancet Infect. Dis. 2012, 12, 820–822. [Google Scholar] [CrossRef]
- World Health Organization Malaria: Artemisinin Partial Resistance. Available online: https://www.who.int/news-room/questions-and-answers/item/artemisinin-resistance (accessed on 26 August 2025).
- Fidock, D.A.; Rosenthal, P.J. Artemisinin resistance in Africa: How urgent is the threat? Med 2021, 2, 1287–1288. [Google Scholar] [CrossRef]
- Jeang, B.; Zhong, D.; Lee, M.C.; Atieli, H.; Yewhalaw, D.; Yan, G. Molecular surveillance of Kelch 13 polymorphisms in Plasmodium falciparum isolates from Kenya and Ethiopia. Malar. J. 2024, 23, 36. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization Report on Antimalarial Drug Efficacy and Resistance: ACT Markers. Available online: https://www.who.int/teams/global-malaria-programme/reports/world-malaria-report-2020 (accessed on 26 March 2025).
- Avci, K.D.; Karakus, M.; Kart Yasar, K. Molecular survey of pfmdr-1, pfcrt, and pfk13 gene mutations among patients returning from Plasmodium falciparum endemic areas to Turkey. Malar. J. 2024, 23, 286. [Google Scholar] [CrossRef] [PubMed]
- Manoj, T.D.; Alan, F.C. Contribution of the pfmdr1 gene to antimalarial drug-resistance. Acta Trop. 2005, 94, 181–190. [Google Scholar] [CrossRef] [PubMed]
- Plowe, C.V. Malaria chemoprevention and drug resistance: A review of the literature and policy implications. Malar. J. 2022, 21, 104. [Google Scholar] [CrossRef]
- Rosenthal, P.J. Artemisinin Resistance Outside of Southeast Asia. Am. J. Trop. Med. Hyg. 2018, 99, 1357–1359. [Google Scholar] [CrossRef]
- Rosenthal, P.J.; Asua, V.; Bailey, J.A.; Conrad, M.D.; Ishengoma, D.S.; Kamya, M.R.; Rasmussen, C.; Tadesse, F.G.; Uwimana, A.; Fidock, D.A. The emergence of artemisinin partial resistance in Africa: How do we respond? Lancet Infect. Dis. 2024, 24, e591–e600. [Google Scholar] [CrossRef]
- Antonio-Nkondjio, C.; Ndo, C.; Njiokou, F.; Bigoga, J.D.; Awono-Ambene, P.; Etang, J.; Ekobo, A.S.; Wondji, C.S. Review of malaria situation in Cameroon: Technical viewpoint on challenges and prospects for disease elimination. Parasit. Vectors 2019, 12, 501. [Google Scholar] [CrossRef] [PubMed]
- Quan, H.; Igbasi, U.; Oyibo, W.; Omilabu, S.; Chen, S.B.; Shen, H.M.; Okolie, C.; Chen, J.H.; Zhou, X.N. High multiple mutations of Plasmodium falciparum-resistant genotypes to sulphadoxine-pyrimethamine in Lagos, Nigeria. Infect. Dis. Poverty 2020, 9, 91. [Google Scholar] [CrossRef]
- Moyeh, M.N.; Njimoh, D.L.; Evehe, M.S.; Ali, I.M.; Nji, A.M.; Nkafu, D.N.; Masumbe, P.N.; Barbara, A.T.; Ndikum, V.N.; Mbacham, W.F. Effects of Drug Policy Changes on Evolution of Molecular Markers of Plasmodium falciparum Resistance to Chloroquine, Amodiaquine, and Sulphadoxine-Pyrimethamine in the South West Region of Cameroon. Malar. Res. Treat. 2018, 2018, 7071383. [Google Scholar] [CrossRef] [PubMed]
- Takala-Harrison, S.; Jacob, C.G.; Arze, C.; Cummings, M.P.; Silva, J.C.; Dondorp, A.M.; Fukuda, M.M.; Hien, T.T.; Mayxay, M.; Noedl, H.; et al. Independent emergence of artemisinin resistance mutations among Plasmodium falciparum in Southeast Asia. J. Infect. Dis. 2015, 211, 670–679. [Google Scholar] [CrossRef]
- Liu, Y.; Liang, X.; Li, J.; Chen, J.; Huang, H.; Zheng, Y.; He, J.; Ehapo, C.S.; Eyi, U.M.; Yang, P.; et al. Molecular Surveillance of Artemisinin-Based Combination Therapies Resistance in Plasmodium falciparum Parasites from Bioko Island, Equatorial Guinea. Microbiol. Spectr. 2022, 10, e00413–e00422. [Google Scholar] [CrossRef]
- Amusan, A.; Akinola, O.; Akano, K.; Hernandez-Castaneda, M.; Dick, J.K.; Sowunmi, A.; Hart, G.; Gbotosho, G. Polymorphisms in Pfkelch13 domains before and after the introduction of artemisinin-based combination therapy in Southwest Nigeria. PLoS ONE 2025, 20, e0316479. [Google Scholar] [CrossRef] [PubMed]
- Sansonetti, P.J.; Lebras, C.; Verdier, F.; Charmot, G.; Dupont, B.; Lapresle, C. Chloroquine-resistant Plasmodium falciparum in Cameroon. Lancet 1985, 1, 1154–1155. [Google Scholar] [CrossRef]
- Leonado, K.B.; Vincent, F.N.; Mathieu, N.; Albert, S.; Jean-Christian, Y.; Raphael, T.O.; Georges, S. Molecular Epidemiology of Malaria in Cameroon. XXI. Baseline Therapeutic efficacy of chloroquine, amodiaquine, and sulfadoxine-pyrimethamine monotherapies in children before national drug policy change. Am. J. Trop. Med. Hyg. 2006, 75, 388–395. [Google Scholar] [CrossRef]
- Lu, F.; Zhang, M.; Culleton, R.L.; Xu, S.; Tang, J.; Zhou, H.; Zhu, G.; Gu, Y.; Zhang, C.; Liu, Y.; et al. Return of chloroquine sensitivity to Africa? Surveillance of African Plasmodium falciparum chloroquine resistance through malaria imported to China. Parasit. Vectors 2017, 10, 355. [Google Scholar] [CrossRef]
- Ndam, N.T.; Basco, L.K.; Ngane, V.F.; Ayouba, A.; Ngolle, E.M.; Deloron, P.; Peeters, M.; Tahar, R. Reemergence of chloroquine-sensitive pfcrt K76 Plasmodium falciparum genotype in southeastern Cameroon. Malar. J. 2017, 16, 130. [Google Scholar] [CrossRef]
- Moyeh, M.N.; Fankem, S.N.; Ali, I.M.; Sofeu, D.; Sandie, S.M.; Njimoh, D.L.; Ghogomu, S.M.; Kimbi, H.K.; Mbacham, W.F. Current status of 4-aminoquinoline resistance markers 18 years after cessation of chloroquine use for the treatment of uncomplicated falciparum malaria in the littoral coastline region of Cameroon. Pathog. Glob. Health 2022, 116, 509–514. [Google Scholar] [CrossRef] [PubMed]
- Pamela, C.; Sandie, M.; Xavier, I.; Sandrine, E.N.; Majoline, T.T.; Luc, A.; Parfait, H.A.; Isabelle, M.; Antoine, B. Prevalence of Plasmodium falciparum parasites resistant to sulfadoxine/pyrimethamine in pregnant women in Yaoundé, Cameroon: Emergence of highly resistant pfdhfr/pfdhps alleles. J. Antimicrob. Chemother. 2015, 70, 2566–2571. [Google Scholar]
- Ajit, L.; Phillipe, M.; Gerald, V.; Ansar, A.P.; Kent, E.K.; Roger, B.; Edwin, L.; Marguerite, K.; Magdalena, P.; Martine, D.; et al. Potent Induction of Focused Th1-Type Cellular and Humoral Immune Responses by RTS,S/SBAS2, a Recombinant Plasmodium falciparum Malaria Vaccine. J. Infect. Dis. 1999, 180, 1656–1664. [Google Scholar]
- Ballou, W.R.; Voss, G.; Kester, K.E.; Heppner, D.G.; Krzych, U. Protective immunity induced with malaria vaccine, RTS,S, is linked to Plasmodium falciparum circumsporozoite protein-specific CD4+ and CD8+ T cells producing IFN-gamma. J. Immunol. 2003, 171, 6961–6967. [Google Scholar]
- Esum, M.E.; Roland, N.N.; Sumbele, I. Trends of Malaria in the South West Region of Cameroon: Overview, Challenges and Perspectives (SDG3). In Proceedings of the 1st International Conference on Sustainable Development Goals "Why It Matters" As Part of the Decade of Action, Utah Valley University, Orem, UT, USA, 5–7 October 2022; Volume 1, pp. 237–254. [Google Scholar]
- Apinjoh, T.O.; Tata, R.B.; Anchang-Kimbi, J.K.; Chi, H.F.; Fon, E.M.; Mugri, R.N.; Tangoh, D.A.; Nyingchu, R.V.; Ghogomu, S.M.; Nkuo-Akenji, T.; et al. Plasmodium falciparum merozoite surface protein 1 block 2 gene polymorphism in field isolates along the slope of mount Cameroon: A cross—Sectional study. BMC Infect. Dis. 2015, 15, 309. [Google Scholar] [CrossRef] [PubMed]
- Adrien, L.; Tommaso, L. pycoQC, interactive quality control for Oxford Nanopore Sequencing. J. Open Source Softw. 2019, 4, 1236. [Google Scholar] [CrossRef]
- Heng, L. Minimap2: Pairwise alignment for nucleotide sequences. Bioinformatics 2018, 34, 3094–3100. [Google Scholar] [CrossRef]
- Petr, D.; James, K.B.; Jennifer, L.; John, M.; Valeriu, O.; Thomas, K.; Martin, O.P.; Andrew, W.; Shane, A.M.; Robert, M.D.; et al. Twelve years of SAMtools and BCFtools. GigaScience 2021, 10, giab008. [Google Scholar] [CrossRef]
- Junhao, S.; Zhenxian, Z.; Syed, S.A.; Tak-Wah, L.; Ruibang, L. Clair3-trio: High-performance Nanopore long-read variant calling in family trios with trio-to-trio deep neural networks. Brief. Bioinform. 2022, 22, bbac301. [Google Scholar]


| Characteristics | Percentages (n/N) | |
|---|---|---|
| Gender | Male | 40.9% (70/171) |
| Female | 59.1% (101/171 | |
| Age (Years) | <5 | 19.3% (33/171) |
| 5–14 | 20.5% (35/171) | |
| 15–24 | 24.6% (42/171) | |
| 25–64 | 29.2% (50/171) | |
| ≥65 | 6.4% (11/171) | |
| Temperatures (°C) | 36.4–37.8 | 39% (60/154) |
| ≥37.8 | 61% (94/154) |
| N | Minimum | Maximum | Mean | Std. Deviation | |
|---|---|---|---|---|---|
| DNA Conc (ng/ uL) | 72 | 99.950 | 247.430 | 166.765 | 36.327766 |
| A260/280 | 72 | 1.69 | 1.94 | 1.8538 | 0.05345 |
| Valid N | 72 |
| Sequencing Batch Code | ONT Kit | Number of Clinical Samples per Run | Run Time | Reads Generated | Estimated Bases | Basecalled Bases | Basecalled % |
|---|---|---|---|---|---|---|---|
| A | Flongle R10.1 | 40 | 7 h | 345.11 K | 166.28 Mb | 189.57 Mb | 100% |
| B | Flongle R10.1 | 32 (+8 duplicates) | 19 h 52 min | 613.98 K | 294.42 Mb | 274 Mb | 96.05% |
| Total | 72 |
| Gene Name | Associated Antimalarial Drug | Identified SNP | Allele Frequency %(n/N) | Status and Reference |
|---|---|---|---|---|
| Chloroquine-resistant transporter, pfcrt | Chloroquine | K76T | 4.2% (3/72) | Resistant mutation [25,29] |
| Dihydrofolate reductase, pfdhfr | Pyrimethamine | N51I | 98.6% (71/72) | Associated with SP resistance [28,33,49,50,51] |
| C59R | 98.6% (71/72) | |||
| S108N | 97.7% (70/72) | |||
| Dihydropteroate synthase, pfdhps | Sulfadoxine | S436A | 54.2% (39/72) | Associated with SP resistance [28,33,49,50,51] |
| A437G | 2.8% (2/72) | |||
| A581G | 28.9% (28/72) | |||
| A613S | 63.4% (32/72) | |||
| Multidrug resistance protein 1, pfmdr1 | Several antimalarials | Y184F | 75% (54/72) | Associated with mefloquine, lumefantrine, chloroquine and artemisinin resistance [44,45,51] |
| Pfkelch13 | Artemisinin | K189T | 63.4% (46/72) | May reduce susceptibility to artemisinin [52,53,54] |
| R255K | 4.2% (3/72) | |||
| Pfdhfr triple mutant | N51I/C59R/S108N | 98.6% (71/72) | SP resistance [28,33,50] | |
| Pfdhfr + Pfdhps quadruple mutant | N51I/C59R/S108N+ A437G | 2.8% (2/72) | Associated with SP resistance [33,50] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Efeti, M.T.; Fankem, S.N.; Diallo, M.T.; Lahngong, M.S.; Acha, N.L.; Shey, R.A.; Demeyer, K.; Souopgui, J.; Ghogomu, S.M.; Njemini, R. Targeted Sequencing Identifies SNPs Associated with Antimalarial Drug Resistance and the CSP Vaccine Antigen in Plasmodium falciparum from Southwest Cameroon. Int. J. Mol. Sci. 2025, 26, 10764. https://doi.org/10.3390/ijms262110764
Efeti MT, Fankem SN, Diallo MT, Lahngong MS, Acha NL, Shey RA, Demeyer K, Souopgui J, Ghogomu SM, Njemini R. Targeted Sequencing Identifies SNPs Associated with Antimalarial Drug Resistance and the CSP Vaccine Antigen in Plasmodium falciparum from Southwest Cameroon. International Journal of Molecular Sciences. 2025; 26(21):10764. https://doi.org/10.3390/ijms262110764
Chicago/Turabian StyleEfeti, Mary T., Sandra N. Fankem, Mariama T. Diallo, Methodius S. Lahngong, Nelson L. Acha, Robert A. Shey, Kristiaan Demeyer, Jacob Souopgui, Stephen M. Ghogomu, and Rose Njemini. 2025. "Targeted Sequencing Identifies SNPs Associated with Antimalarial Drug Resistance and the CSP Vaccine Antigen in Plasmodium falciparum from Southwest Cameroon" International Journal of Molecular Sciences 26, no. 21: 10764. https://doi.org/10.3390/ijms262110764
APA StyleEfeti, M. T., Fankem, S. N., Diallo, M. T., Lahngong, M. S., Acha, N. L., Shey, R. A., Demeyer, K., Souopgui, J., Ghogomu, S. M., & Njemini, R. (2025). Targeted Sequencing Identifies SNPs Associated with Antimalarial Drug Resistance and the CSP Vaccine Antigen in Plasmodium falciparum from Southwest Cameroon. International Journal of Molecular Sciences, 26(21), 10764. https://doi.org/10.3390/ijms262110764










