Stem Cell Niche Concept: Search for Current Expert Consensus
Abstract
1. Introduction
2. General Concept (Definition) of the Stem Cell Niche
- The fundamental property of a stem cell is self-renewal, which depends on the microenvironment in which the stem cell is seen in association with other cells, determining its behavior [10].
- The stem cell niche is a specialized microenvironment in which stem cells reside primarily in a quiescent state by providing anti-proliferation signals [19].
- The stem cell niche is a specific microenvironment that directly controls the dual capacity for self-renewal and multilineage differentiation of stem cells, maintaining the HSC in a quiescent state [20].
- The niche is a restricted site in an organ that supports the stem cell self-renewal [21].
- Stem cells reside in specialized microenvironments, also called niches, which maintain them in an undifferentiated and self-renewing state [22].
- Niches are the anatomical regions within the microenvironment [60].
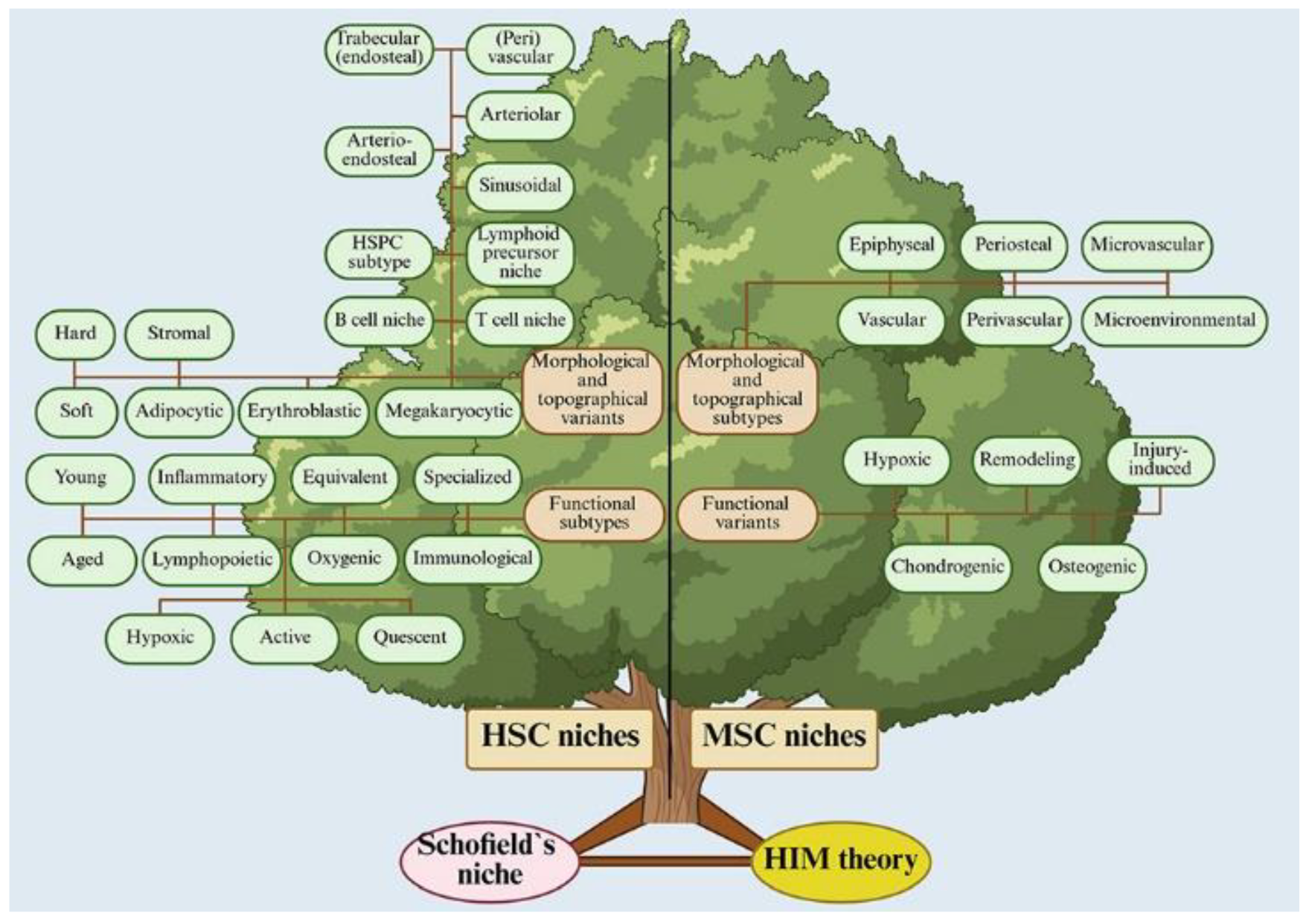
- The stem cell niche is a group of cells in a special tissue location that are intended for the maintenance of stem cells. It has a variable structure, and different cell types can provide the niche environment. There is a niche hierarchy of HSC due to HIM heterogeneity at diverse regions of the BM [23].
- There are the hierarchical HSC osteoblastic (and MSC) niches in terms of their localization, composition, function, and multi-layer regulation systems [24].
- The stem cell niches are the distinct, structurally functional, energetically favorable microterritories in a contiguous space of HIM where quantitative parameters of a microenvironment promote the qualitative control of stem cell fate [13].
- According to Pinho et al. (2018), the HSC pool is functionally and molecularly heterogeneous; consequently, there are distinct ‘specialized’ niches for distinct subpopulations of HSCs [25].
- The HSC niche is considered to be a complex multicellular network that provides molecular signals and physical interactions that are essential for HSC localization, maintenance, and differentiation [26].
- The niche provides a microenvironment that supports the self-renewal and multi-lineage differentiation of stem cells [27].
- The niche represents a sophisticated and dynamic system of cellular and molecular components coupled with heterogeneous signaling mechanisms. It serves as an interface between stem cells and the organism, orchestrating their adaptive responses to tissue damage [62].
- The BM niche is a complex environment composed of heterogeneous cell populations that regulate the hematopoietic stem and progenitor cells (HSPCs) function and activity through the secretion of a wide array of cytokines and growth factors [63].
- The stem cell niche is a dynamic and specialized microenvironment with a specific architecture that regulates self-renewal of stem cells, the balance between their quiescent and proliferative status, as well as their choice of fate and differentiation of their progenitor cells [28].
2.1. Core Components and Regulatory Mechanisms in Stem Cell Niches
2.1.1. Cellular and Non-Cellular Components of Stem Cell Niches in Mammalian Tissues
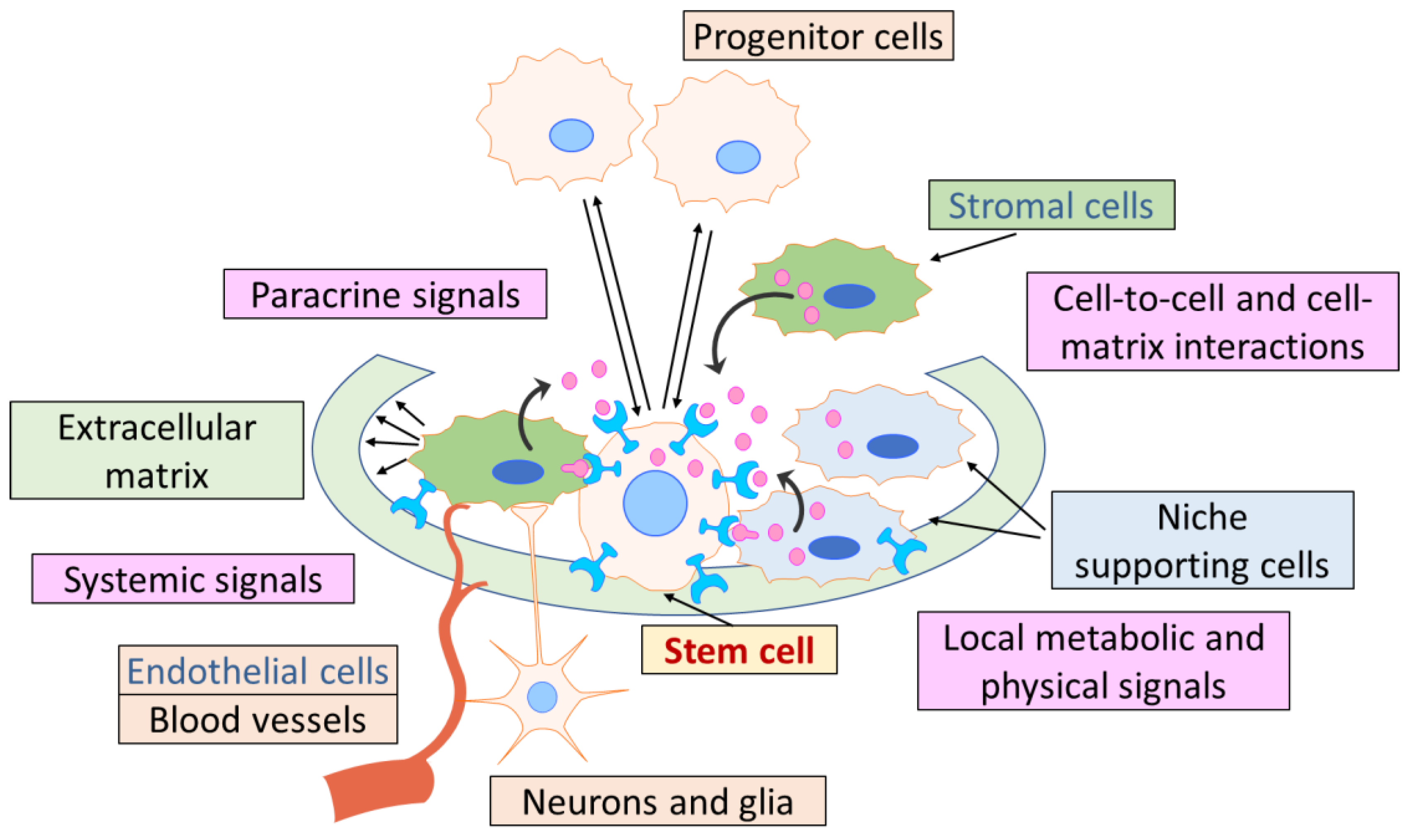
2.1.2. Comparison of HSPC Niche Structure and HIM Morphologic Components in the BM
- Tissue-resident and migrating cells.
- ECM with fibers and soluble molecules.
- Blood vessels.
- Nerve terminals.
- Various hematopoietic and stromal support cells, including cell–cell adhesion molecules and secreted soluble factors, are located in close proximity to stem cells.
- ECM, which serves as an “anchor” for stem cells and forms a mechanical scaffold for the transmission of stem cell signaling.
- Blood vessels that supply the niche with nutrients and systemic signals from other organs and are involved in the recruitment of circulating stem cells from and into the niche.
- Nerve terminals and Schwann cells.
- The entire HIM space of the BM consists of niches for HSCs, MSCs, and their progenitors, which are in close contact with each other.
- Between the niches, there is a non-specialized HIM space, which has extrinsic effects on its functioning.
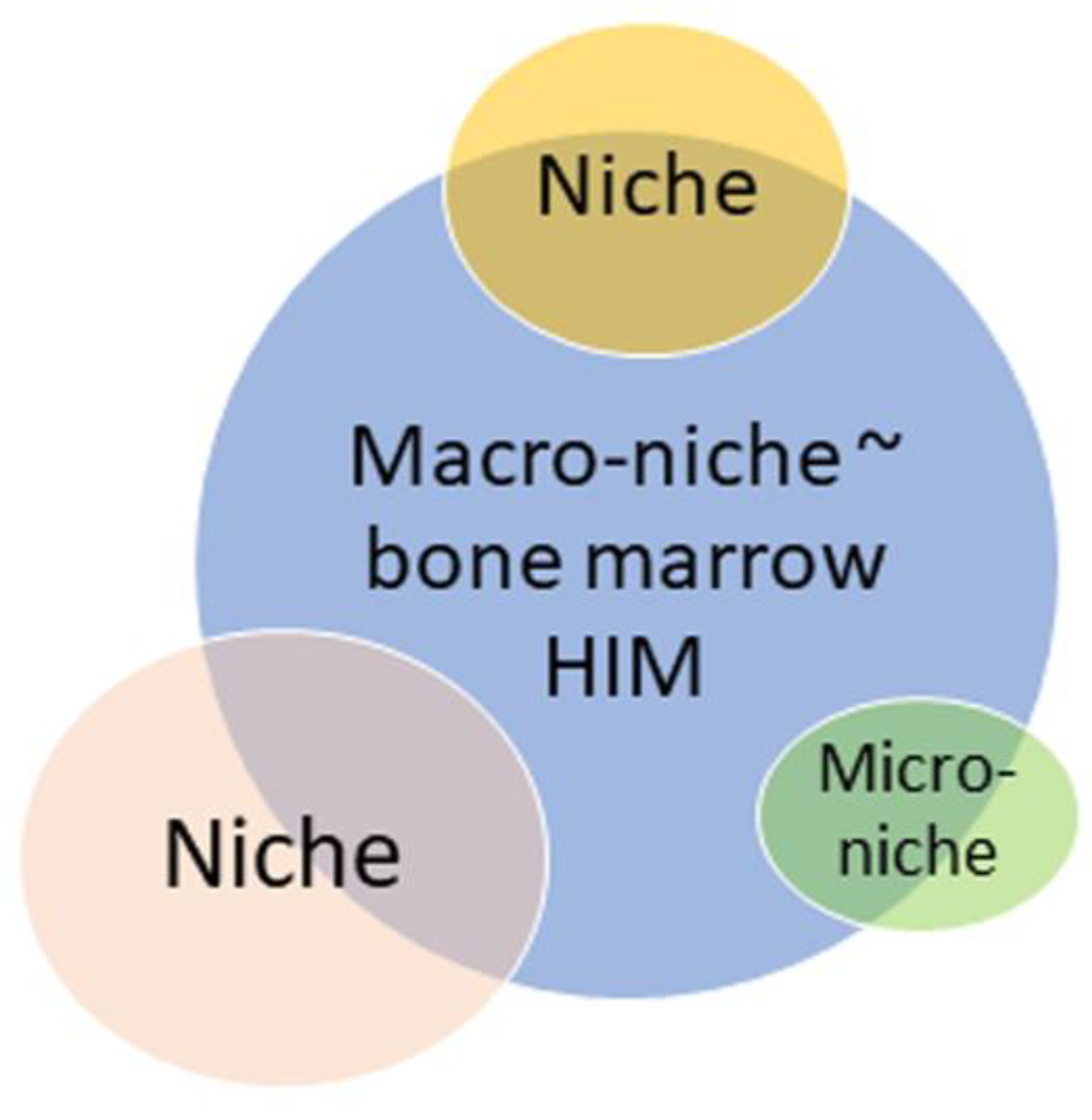
- It is unclear whether there are significant (qualitative or quantitative) differences between the local specialized microterritories (referred to as the niches) and the known components of the entire HIM space. What are the external borders of stem cell niches to distinguish their structural and functional features?
3. Hierarchy of Potential Niche-Candidates for Stem and Progenitor Cells
3.1. Erythroblastic Islands as Candidate Niche for HSPCs
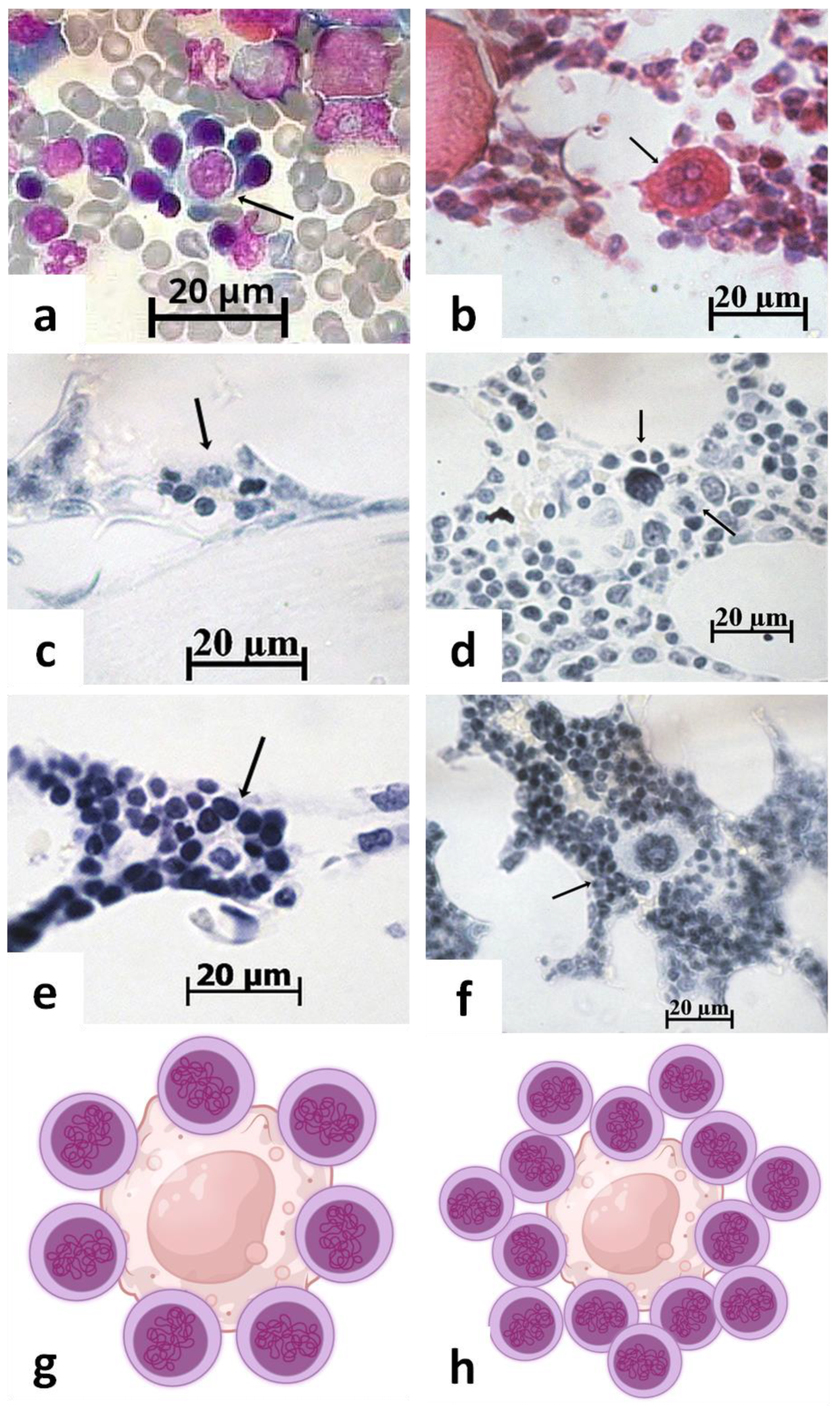
3.2. Megakaryocytic Candidate for HSPC Niches
3.3. MSC Niche
3.4. Live Imaging of Niches
3.5. Niche Aging as One of the Functional Manifestations of Specialized Microterritories
4. Challenges of the Dimension and Geometry of Stem and Progenitor Cell Niches
5. Prospects for Further Research
6. Primary Minimal Criteria for HSPC Niche
7. Conclusions
- Fundamental and applied scientific solutions:
- -
- Enhanced international research collaboration by defining clear priorities and directions;
- -
- Systematic development of a unified classification system for diverse structural-functional units within the native hierarchy of hematopoietic tissues, building upon previous work outlined in [101];
- -
- Innovative digital solutions for three-dimensional prototyping and printing of synthetic stem cell niches and other microscale tissue constructs, enabling precise bioengineering applications.
- Educational and terminological contributions:
- -
- Specialized training programs to equip early-career researchers with cutting-edge knowledge in the field;
- -
- Development and refinement of a standardized glossary to harmonize terminology related to stem cells and their niches.
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Glossary
| HIM | The microenvironment of hematopoietic tissues (bone marrow, spleen, and liver in rodents); its morphological components (stromal cells, ECM, blood vessels, and nerve terminals) control the hematopoietic cell fate via direct (cell–cell and cell–matrix) and indirect (soluble molecules) signals. |
| Niche | No common definition exists. The simplest definition is that the niche is the specific microenvironment of a stem cell. Our description of the niche is as follows: the stem cell niches are the distinct, structural-functional, energetically favorable microterritories (structural-functional regions) in a contiguous spatial space of HIM where quantitative parameters of a microenvironment promote the qualitative control of stem cell fate. |
| Domain | HSC regulatory volume in mouse bone marrow, with the value of ~0.1 mm3, consisted of ~90,000 cells (stem, progenitor, and mature hematopoietic cells + HIM components) [55,109]. |
Abbreviations
| HSCs | Hematopoietic stem cells |
| HIM | Hemopoietic-inductive microenvironment |
| HER | Hemopoiesis engendered randomly |
| MSCs | Mesenchymal stromal/stem cells |
| BM | Bone marrow |
| HSPCs | Hematopoietic stem and progenitor cells |
| CFU | Colony-forming units |
| ECM | Extracellular matrix |
| LepR+ | leptin receptor-expressing (cells) |
| SSCs | Skeletal stem cells |
| CAR | CXC chemokine ligand 12 (CXCL12)-abundant reticular cells |
| SCF | Stem cell factor |
| BMP | Bone morphogenetic protein |
| TGF | Transforming growth factor |
| SDF-1 | Stromal cell-derived factor 1 |
| FGF-4 | Fibroblast growth factor 4 |
| VEGF | Vascular endothelial growth factor |
| MMP | Metalloproteinase |
| KitL | Kit ligand |
| LGR5 | Leucine-rich repeat-containing G-protein-coupled receptor 5 |
| EGF | Epidermal growth factor |
| DLL4 | Delta-like protein 4 |
| FGFs | Fibroblast growth factors |
| EIs | Erythroblastic islands |
| HIs | Hematopoietic islands |
| MKs | Megakaryocytes |
| SSPCs | Skeletal stem and progenitor cells |
| MLM | Multiphoton laser microscopy |
| ISCT | International Society for Cellular Therapy |
References
- Shiba, Y. Pluripotent Stem Cells for Cardiac Regeneration—Current Status, Challenges, and Future Perspectives. Circ. J. 2020, 84, 2129–2135. [Google Scholar] [CrossRef] [PubMed]
- Berika, M.; El-Hashash, A.H.K. Stem Cells in Bone Repair and Regeneration. Curr. Stem Cell Res. Ther. 2023, 18, 460–469. [Google Scholar] [CrossRef] [PubMed]
- Trompet, D.; Melis, S.; Chagin, A.S.; Maes, C. Skeletal Stem and Progenitor Cells in Bone Development and Repair. J. Bone Miner. Res. 2024, 39, 633–654. [Google Scholar] [CrossRef]
- Augustine, R.; Gezek, M.; Nikolopoulos, V.K.; Buck, P.L.; Bostanci, N.S.; Camci-Unal, G. Stem Cells in Bone Tissue Engineering: Progress, Promises and Challenges. Stem Cell Rev. Rep. 2024, 20, 1692–1731. [Google Scholar] [CrossRef]
- Kent, D.G. Hematopoietic Stem Cells Depend on HIM and HER. Exp. Hematol. 2022, 116, 15–17. [Google Scholar] [CrossRef] [PubMed]
- Curry, J.L.; Trentin, J.J.; Wolf, N. Hemopoietic Spleen Colony Studies. II. Erythropoiesis. J. Exp. Med. 1967, 125, 703–720. [Google Scholar] [CrossRef]
- McCulloch, E.A.; Siminovitch, L.; Till, J.E.; Russell, E.S.; Bernstein, S.E. The Cellular Basis of the Genetically Determined Hemopoietic Defect in Anemic Mice of Genotype Sl/Sld. Blood 1965, 26, 399–410. [Google Scholar] [CrossRef]
- Till, J.E.; McCulloch, E.A. Hemopoietic Stem Cell Differentiation. Biochim. Biophys. Acta 1980, 605, 431–459. [Google Scholar] [CrossRef]
- Schofield, R. The Relationship between the Spleen Colony-Forming Cell and the Haemopoietic Stem Cell. Blood Cells 1978, 4, 7–25. [Google Scholar]
- Schofield, R. The stem cell system. Biomed. Pharmacother. 1983, 37, 375–380. [Google Scholar]
- Huang, D.; Chen, C.; Xie, L.; Yu, Z.; Zheng, J. Hematopoietic Stem Cell Metabolism and Stemness. Blood Sci. 2019, 1, 12–18. [Google Scholar] [CrossRef]
- Sagaradze, G.D.; Basalova, N.A.; Efimenko, A.Y.; Tkachuk, V.A. Mesenchymal Stromal Cells as Critical Contributors to Tissue Regeneration. Front. Cell Dev. Biol. 2020, 8, 576176. [Google Scholar] [CrossRef]
- Khlusov, I.A.; Litvinova, L.S.; Khlusova, M.Y.; Yurova, K.A. Concept of Hematopoietic and Stromal Niches for Cell-Based Diagnostics and Regenerative Medicine (a Review). Curr. Pharm. Des. 2018, 24, 3034–3054. [Google Scholar] [CrossRef]
- Yurova, K.A.; Khaziakhmatova, O.G.; Melashchenko, E.S.; Malashchenko, V.V.; Shunkin, E.O.; Shupletsova, V.V.; Ivanov, P.A.; Khlusov, I.A.; Litvinova, L.S. Cellular and Molecular Basis of Osteoblastic and Vascular Niches in the Processes of Hematopoiesis and Bone Remodeling (A Short Review of Modern Views). Curr. Pharm. Des. 2019, 25, 663–669. [Google Scholar] [CrossRef]
- Calvo, W.; Fliedner, T.M.; Herbst, E.; Hügl, E.; Bruch, C. Regeneration of Blood-Forming Organs after Autologous Leukocyte Transfusion in Lethally Irradiated Dogs. II. Distribution and Cellularity of the Marrow in Irradiated and Transfused Animals. Blood 1976, 47, 593–601. [Google Scholar] [CrossRef]
- Purton, L.E.; Scadden, D.T. The Hematopoietic Stem Cell Niche. In StemBook; Harvard Stem Cell Institute: Cambridge, MA, USA, 2008. [Google Scholar]
- Scadden, D.T. The Stem-Cell Niche as an Entity of Action. Nature 2006, 441, 1075–1079. [Google Scholar] [CrossRef] [PubMed]
- Scadden, D.T. The Stem Cell Niche in Health and Leukemic Disease. Best Pract. Res. Clin. Haematol. 2007, 20, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Neaves, W.B. Normal Stem Cells and Cancer Stem Cells: The Niche Matters. Cancer Res. 2006, 66, 4553–4557. [Google Scholar] [CrossRef] [PubMed]
- Wagner, W.; Horn, P.; Bork, S.; Ho, A.D. Aging of Hematopoietic Stem Cells Is Regulated by the Stem Cell Niche. Exp. Gerontol. 2008, 43, 974–980. [Google Scholar] [CrossRef]
- Gentry, S.N.; Jackson, T.L. A Mathematical Model of Cancer Stem Cell Driven Tumor Initiation: Implications of Niche Size and Loss of Homeostatic Regulatory Mechanisms. PLoS ONE 2013, 8, e71128. [Google Scholar] [CrossRef]
- Birbrair, A. Stem Cell Microenvironments and Beyond. Adv. Exp. Med. Biol. 2017, 1041, 1–3. [Google Scholar] [CrossRef]
- Li, L.; Xie, T. Stem Cell Niche: Structure and Function. Annu. Rev. Cell Dev. Biol. 2005, 21, 605–631. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Zhang, P.; Liu, L.; Zou, L. Hierarchical Organization and Regulation of the Hematopoietic Stem Cell Osteoblastic Niche. Crit. Rev. Oncol. Hematol. 2013, 85, 1–8. [Google Scholar] [CrossRef]
- Pinho, S.; Marchand, T.; Yang, E.; Wei, Q.; Nerlov, C.; Frenette, P.S. Lineage-Biased Hematopoietic Stem Cells Are Regulated by Distinct Niches. Dev. Cell 2018, 44, 634–641.e4. [Google Scholar] [CrossRef]
- Pinho, S.; Frenette, P.S. Haematopoietic Stem Cell Activity and Interactions with the Niche. Nat. Rev. Mol. Cell Biol. 2019, 20, 303–320. [Google Scholar] [CrossRef]
- Chen, S.; Jing, J.; Yuan, Y.; Feng, J.; Han, X.; Wen, Q.; Ho, T.-V.; Lee, C.; Chai, Y. Runx2+ Niche Cells Maintain Incisor Mesenchymal Tissue Homeostasis through IGF Signaling. Cell Rep. 2020, 32, 108007. [Google Scholar] [CrossRef] [PubMed]
- Kurenkova, A.D.; Medvedeva, E.V.; Newton, P.T.; Chagin, A.S. Niches for Skeletal Stem Cells of Mesenchymal Origin. Front. Cell Dev. Biol. 2020, 8, 592. [Google Scholar] [CrossRef] [PubMed]
- Ferraro, F.; Celso, C.L.; Scadden, D. Adult Stem Cels and Their Niches. Adv. Exp. Med. Biol. 2010, 695, 155–168. [Google Scholar] [CrossRef]
- Morrison, S.J.; Scadden, D.T. The Bone Marrow Niche for Haematopoietic Stem Cells. Nature 2014, 505, 327–334. [Google Scholar] [CrossRef]
- Calvi, L.M.; Link, D.C. Cellular Complexity of the Bone Marrow Hematopoietic Stem Cell Niche. Calcif. Tissue Int. 2014, 94, 112–124. [Google Scholar] [CrossRef]
- Gattazzo, F.; Urciuolo, A.; Bonaldo, P. Extracellular Matrix: A Dynamic Microenvironment for Stem Cell Niche. Biochim. Biophys. Acta 2014, 1840, 2506–2519. [Google Scholar] [CrossRef]
- Domingues, M.J.; Cao, H.; Heazlewood, S.Y.; Cao, B.; Nilsson, S.K. Niche Extracellular Matrix Components and Their Influence on HSC. J. Cell. Biochem. 2017, 118, 1984–1993. [Google Scholar] [CrossRef]
- Muncie, J.M.; Weaver, V.M. The Physical and Biochemical Properties of the Extracellular Matrix Regulate Cell Fate. Curr. Top. Dev. Biol. 2018, 130, 1–37. [Google Scholar] [CrossRef]
- Ghosh, J.; Koussa, R.E.; Mohamad, S.F.; Liu, J.; Kacena, M.A.; Srour, E.F. Cellular Components of the Hematopoietic Niche and Their Regulation of Hematopoietic Stem Cell Function. Curr. Opin. Hematol. 2021, 28, 243–250. [Google Scholar] [CrossRef]
- Lee-Thedieck, C.; Schertl, P.; Klein, G. The Extracellular Matrix of Hematopoietic Stem Cell Niches. Adv. Drug Deliv. Rev. 2022, 181, 114069. [Google Scholar] [CrossRef]
- Wu, Q.; Zhang, J.; Lucas, D. Anatomy of Hematopoiesis and Local Microenvironments in the Bone Marrow. Where to? Front. Immunol. 2021, 12, 768439. [Google Scholar] [CrossRef]
- Quesenberry, P.; Abedi, M.; Dooner, M.; Colvin, G.; Sanchez-Guijo, F.M.; Aliotta, J.; Pimentel, J.; Dooner, G.; Greer, D.; Demers, D.; et al. The Marrow Cell Continuum: Stochastic Determinism. Folia Histochem. Cytobiol. 2005, 43, 187–190. [Google Scholar] [PubMed]
- Shimoto, M.; Sugiyama, T.; Nagasawa, T. Numerous Niches for Hematopoietic Stem Cells Remain Empty during Homeostasis. Blood 2017, 129, 2124–2131. [Google Scholar] [CrossRef] [PubMed]
- Kandarakov, O.; Belyavsky, A.; Semenova, E. Bone Marrow Niches of Hematopoietic Stem and Progenitor Cells. Int. J. Mol. Sci. 2022, 23, 4462. [Google Scholar] [CrossRef] [PubMed]
- Drummond-Barbosa, D. Stem Cells, Their Niches and the Systemic Environment: An Aging Network. Genetics 2008, 180, 1787–1797. [Google Scholar] [CrossRef]
- Chasis, J.A. Erythroblastic Islands: Specialized Microenvironmental Niches for Erythropoiesis. Curr. Opin. Hematol. 2006, 13, 137–141. [Google Scholar] [CrossRef]
- Khlusov, I.A.; Shevtsova, N.M.; Khlusova, M.Y. Detection in Vitro and Quantitative Estimation of Artificial Microterritories Which Promote Osteogenic Differentiation and Maturation of Stromal Stem Cells. Methods Mol. Biol. 2013, 1035, 103–119. [Google Scholar] [CrossRef]
- Bruns, I.; Lucas, D.; Pinho, S.; Ahmed, J.; Lambert, M.P.; Kunisaki, Y.; Scheiermann, C.; Schiff, L.; Poncz, M.; Bergman, A.; et al. Megakaryocytes Regulate Hematopoietic Stem Cell Quiescence through CXCL4 Secretion. Nat. Med. 2014, 20, 1315–1320. [Google Scholar] [CrossRef]
- Young, K.A.; Telpoukhovskaia, M.A.; Hofmann, J.; Mistry, J.J.; Kokkaliaris, K.D.; Trowbridge, J.J. Variation in Mesenchymal KITL/SCF and IGF1 Expression in Middle Age Underlies Steady-State Hematopoietic Stem Cell Aging. Blood 2024, 144, 378–391. [Google Scholar] [CrossRef]
- Kopp, H.-G.; Avecilla, S.T.; Hooper, A.T.; Rafii, S. The Bone Marrow Vascular Niche: Home of HSC Differentiation and Mobilization. Physiology 2005, 20, 349–356. [Google Scholar] [CrossRef]
- Yin, T.; Li, L. The Stem Cell Niches in Bone. J. Clin. Investig. 2006, 116, 1195–1201. [Google Scholar] [CrossRef]
- Zhang, J.; Niu, C.; Ye, L.; Huang, H.; He, X.; Tong, W.-G.; Ross, J.; Haug, J.; Johnson, T.; Feng, J.Q.; et al. Identification of the Haematopoietic Stem Cell Niche and Control of the Niche Size. Nature 2003, 425, 836–841. [Google Scholar] [CrossRef] [PubMed]
- Calvi, L.M.; Adams, G.B.; Weibrecht, K.W.; Weber, J.M.; Olson, D.P.; Knight, M.C.; Martin, R.P.; Schipani, E.; Divieti, P.; Bringhurst, F.R.; et al. Osteoblastic Cells Regulate the Haematopoietic Stem Cell Niche. Nature 2003, 425, 841–846. [Google Scholar] [CrossRef]
- Newton, P.T.; Li, L.; Zhou, B.; Schweingruber, C.; Hovorakova, M.; Xie, M.; Sun, X.; Sandhow, L.; Artemov, A.V.; Ivashkin, E.; et al. A Radical Switch in Clonality Reveals a Stem Cell Niche in the Epiphyseal Growth Plate. Nature 2019, 567, 234–238. [Google Scholar] [CrossRef] [PubMed]
- Chan, C.K.; Chen, C.C.; Luppen, C.A.; Kim, J.B.; DeBoer, A.T.; Wei, K.; Helms, J.A.; Kuo, C.J.; Kraft, D.L.; Weissman, I.L. Endochondral ossification is required for haematopoietic stem-cell niche formation. Nature 2009, 457, 490–494. [Google Scholar] [CrossRef] [PubMed]
- Yu, V.W.C.; Scadden, D.T. Heterogeneity of the Bone Marrow Niche. Curr. Opin. Hematol. 2016, 23, 331–338. [Google Scholar] [CrossRef] [PubMed]
- Baccin, C.; Al-Sabah, J.; Velten, L.; Helbling, P.M.; Grünschläger, F.; Hernández-Malmierca, P.; Nombela-Arrieta, C.; Steinmetz, L.M.; Trumpp, A.; Haas, S. Combined Single-Cell and Spatial Transcriptomics Reveal the Molecular, Cellular and Spatial Bone Marrow Niche Organization. Nat. Cell Biol. 2020, 22, 38–48. [Google Scholar] [CrossRef]
- Khlusov, I.; Litvinova, L.; Yurova, K.; Khlusova, M. Precise Tissue Bioengineering and Niches of Mesenchymal Stem Cells: Their Size and Hierarchy Matter. BIOCELL 2022, 46, 1365–1373. [Google Scholar] [CrossRef]
- Maloney, M.A.; Dorie, M.J.; Lamela, R.A.; Rogers, Z.R.; Patt, H.M. Hematopoietic Stem Cell Regulatory Volumes as Revealed in Studies of the Bgj/Bgj:W/WV Chimera. J. Exp. Med. 1978, 147, 1189–1197. [Google Scholar] [CrossRef]
- Crocker, P.R.; Gordon, S. Isolation and Characterization of Resident Stromal Macrophages and Hematopoietic Cell Clusters from Mouse Bone Marrow. J. Exp. Med. 1985, 162, 993–1014. [Google Scholar] [CrossRef]
- Blazsek, I.; Misset, J.L.; Comisso, M.; Mathé, G. Hematon: A Multicellular Functional Unit in Primary Hematopoiesis. Biomed. Pharmacother. 1988, 42, 661–668. [Google Scholar]
- Wang, L.; Benedito, R.; Bixel, M.G.; Zeuschner, D.; Stehling, M.; Sävendahl, L.; Haigh, J.J.; Snippert, H.; Clevers, H.; Breier, G.; et al. Identification of a Clonally Expanding Haematopoietic Compartment in Bone Marrow. EMBO J. 2013, 32, 219–230. [Google Scholar] [CrossRef]
- Vodyanoy, V.; Pustovyy, O.; Globa, L.; Kulesza, R.J.; Sorokulova, I. Hemmule: A Novel Structure with the Properties of the Stem Cell Niche. Int. J. Mol. Sci. 2020, 21, 539. [Google Scholar] [CrossRef]
- Ju, F.; Atyah, M.M.; Horstmann, N.; Gul, S.; Vago, R.; Bruns, C.J.; Zhao, Y.; Dong, Q.-Z.; Ren, N. Characteristics of the Cancer Stem Cell Niche and Therapeutic Strategies. Stem Cell Res. Ther. 2022, 13, 233. [Google Scholar] [CrossRef] [PubMed]
- Lucas, D. Structural Organization of the Bone Marrow and Its Role in Hematopoiesis. Curr. Opin. Hematol. 2021, 28, 36–42. [Google Scholar] [CrossRef]
- Nimiritsky, P.; Sagaradze, G.D.; Efimenko, A.Y.; Makarevich, P.; Tkachuk, V.A. The Stem Cell Niche. Tsitologiya 2018, 60, 575–586. [Google Scholar] [CrossRef]
- Emmons, R.; Niemiro, G.M.; De Lisio, M. Hematopoiesis with Obesity and Exercise: Role of the Bone Marrow Niche. Exerc. Immunol. Rev. 2017, 23, 82–95. [Google Scholar] [PubMed]
- Morrison, S.J.; Spradling, A.C. Stem Cells and Niches: Mechanisms That Promote Stem Cell Maintenance throughout Life. Cell 2008, 132, 598–611. [Google Scholar] [CrossRef] [PubMed]
- Wagers, A.J. The Stem Cell Niche in Regenerative Medicine. Cell Stem Cell 2012, 10, 362–369. [Google Scholar] [CrossRef]
- Scadden, D.T. Nice Neighborhood: Emerging Concepts of the Stem Cell Niche. Cell 2014, 157, 41–50. [Google Scholar] [CrossRef]
- Hicks, M.R.; Pyle, A.D. The Emergence of the Stem Cell Niche. Trends Cell Biol. 2023, 33, 112–123. [Google Scholar] [CrossRef]
- Nakatani, T.; Nagasawa, T. Bone Marrow Niches for Hematopoietic Stem Cells in Homeostasis and Aging. Exp. Hematol. 2025, 144, 104749. [Google Scholar] [CrossRef]
- Yip, R.K.H.; Hawkins, E.D.; Bowden, R.; Rogers, K.L. Towards Deciphering the Bone Marrow Microenvironment with Spatial Multi-Omics. Semin. Cell Dev. Biol. 2025, 167, 10–21. [Google Scholar] [CrossRef]
- Boulais, P.E.; Frenette, P.S. Making sense of hematopoietic stem cell niches. Blood 2015, 125, 2621–2629. [Google Scholar] [CrossRef]
- Ding, L.; Saunders, T.L.; Enikolopov, G.; Morrison, S.J. Endothelial and Perivascular Cells Maintain Haematopoietic Stem Cells. Nature 2012, 481, 457–462. [Google Scholar] [CrossRef]
- Crippa, S.; Bernardo, M.E. Mesenchymal Stromal Cells: Role in the BM Niche and in the Support of Hematopoietic Stem Cell Transplantation. Hemasphere 2018, 2, e151. [Google Scholar] [CrossRef] [PubMed]
- Kfoury, Y.; Scadden, D.T. Mesenchymal Cell Contributions to the Stem Cell Niche. Cell Stem Cell 2015, 16, 239–253. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.; Iqbal, Z.; Zhao, Z.; Liu, J.; Alabsi, A.M.; Shabbir, M.; Mahmood, A.; Liang, Y.; Li, W.; Deng, Z. Cellular Crosstalk in the Bone Marrow Niche. J. Transl. Med. 2024, 22, 1096. [Google Scholar] [CrossRef] [PubMed]
- Méndez-Ferrer, S.; Michurina, T.V.; Ferraro, F.; Mazloom, A.R.; Macarthur, B.D.; Lira, S.A.; Scadden, D.T.; Ma’ayan, A.; Enikolopov, G.N.; Frenette, P.S. Mesenchymal and Haematopoietic Stem Cells Form a Unique Bone Marrow Niche. Nature 2010, 466, 829–834. [Google Scholar] [CrossRef]
- Chan, C.K.F.; Gulati, G.S.; Sinha, R.; Tompkins, J.V.; Lopez, M.; Carter, A.C.; Ransom, R.C.; Reinisch, A.; Wearda, T.; Murphy, M.; et al. Identification of the Human Skeletal Stem Cell. Cell 2018, 175, 43–56.e21. [Google Scholar] [CrossRef]
- Soteriou, D.; Fuchs, Y. A Matter of Life and Death: Stem Cell Survival in Tissue Regeneration and Tumour Formation. Nat. Rev. Cancer 2018, 18, 187–201. [Google Scholar] [CrossRef]
- Novoseletskaya, E.; Grigorieva, O.; Nimiritsky, P.; Basalova, N.; Eremichev, R.; Milovskaya, I.; Kulebyakin, K.; Kulebyakina, M.; Rodionov, S.; Omelyanenko, N.; et al. Mesenchymal Stromal Cell-Produced Components of Extracellular Matrix Potentiate Multipotent Stem Cell Response to Differentiation Stimuli. Front. Cell Dev. Biol. 2020, 8, 555378. [Google Scholar] [CrossRef]
- Avigad Laron, E.; Aamar, E.; Enshell-Seijffers, D. The Mesenchymal Niche of the Hair Follicle Induces Regeneration by Releasing Primed Progenitors from Inhibitory Effects of Quiescent Stem Cells. Cell Rep. 2018, 24, 909–921.e3. [Google Scholar] [CrossRef]
- Sagaradze, G.; Basalova, N.; Kirpatovsky, V.; Ohobotov, D.; Nimiritsky, P.; Grigorieva, O.; Popov, V.; Kamalov, A.; Tkachuk, V.; Efimenko, A. A Magic Kick for Regeneration: Role of Mesenchymal Stromal Cell Secretome in Spermatogonial Stem Cell Niche Recovery. Stem Cell Res. Ther. 2019, 10, 342. [Google Scholar] [CrossRef]
- Shimizu, S.; Kubota, Y. Intra-Bone Marrow Diversity of Endothelial Cells and Its Impact on Hematopoietic Stem Cell Development and Maintenance. Exp. Hematol. 2025, 149, 104817. [Google Scholar] [CrossRef]
- Xu, H.; Li, Y.; Gao, Y. The Role of Immune Cells Settled in the Bone Marrow on Adult Hematopoietic Stem Cells. Cell. Mol. Life Sci. 2024, 81, 420. [Google Scholar] [CrossRef] [PubMed]
- Quarato, E.R.; Salama, N.A.; Calvi, L.M. Interplay Between Skeletal and Hematopoietic Cells in the Bone Marrow Microenvironment in Homeostasis and Aging. Curr. Osteoporos. Rep. 2024, 22, 416–432. [Google Scholar] [CrossRef]
- Akinyemi, D.E.; Chevre, R.; Soehnlein, O. Neuro-immune crosstalk in hematopoiesis, inflammation, and repair. Trends Immunol. 2024, 45, 597–608. [Google Scholar] [CrossRef]
- Novoseletskaya, E.S.; Grigorieva, O.A.; Efimenko, A.Y.; Kalinina, N.I. Extracellular Matrix in the Regulation of Stem Cell Differentiation. Biochemistry 2019, 84, 232–240. [Google Scholar] [CrossRef]
- Khurana, S.; Schouteden, S.; Manesia, J.K.; Santamaria-Martínez, A.; Huelsken, J.; Lacy-Hulbert, A.; Verfaillie, C.M. Outside-in integrin signalling regulates haematopoietic stem cell function via Periostin-Itgav axis. Nat. Commun. 2016, 7, 13500. [Google Scholar] [CrossRef] [PubMed]
- Nakatani, T.; Sugiyama, T.; Omatsu, Y.; Watanabe, H.; Kondoh, G.; Nagasawa, T. Ebf3+ Niche-Derived CXCL12 Is Required for the Localization and Maintenance of Hematopoietic Stem Cells. Nat. Commun. 2023, 14, 6402. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, P.; Isringhausen, S.; Li, H.; Paterson, A.J.; He, J.; Gomariz, Á.; Nagasawa, T.; Nombela-Arrieta, C.; Bhatia, R. Mesenchymal Niche-Specific Expression of Cxcl12 Controls Quiescence of Treatment-Resistant Leukemia Stem Cells. Cell Stem Cell 2019, 24, 769–784.e6. [Google Scholar] [CrossRef]
- Niu, C.; Yu, J.; Zou, T.; Lu, Y.; Deng, L.; Yun, H.; Si, C.-Y.; Wu, X.; Jiang, H.; Guo, T.; et al. Identification of Hematopoietic Stem Cells Residing in the Meninges of Adult Mice at Steady State. Cell Rep. 2022, 41, 111592. [Google Scholar] [CrossRef]
- Gao, L.; Lee, H.; Goodman, J.H.; Ding, L. Hematopoietic Stem Cell Niche Generation and Maintenance Are Distinguishable by an Epitranscriptomic Program. Cell 2024, 187, 2801–2816.E17. [Google Scholar] [CrossRef]
- Oda, A.; Tezuka, T.; Ueno, Y.; Hosoda, S.; Amemiya, Y.; Notsu, C.; Kasahara, T.; Nishiyama, C.; Goitsuka, R. Niche-induced extramedullary hematopoiesis in the spleen is regulated by the transcription factor Tlx1. Sci. Rep. 2018, 8, 8308. [Google Scholar] [CrossRef]
- Conrad, C.; Magnen, M.; Tsui, J.; Wismer, H.; Naser, M.; Venkataramani, U.; Samad, B.; Cleary, S.J.; Qiu, L.; Tian, J.J.; et al. Decoding Functional Hematopoietic Progenitor Cells in the Adult Human Lung. Blood 2025, 145, 1975–1986. [Google Scholar] [CrossRef]
- Lewis, K.; Yoshimoto, M.; Takebe, T. Fetal liver hematopoiesis: From development to delivery. Stem Cell Res. Ther. 2021, 12, 139. [Google Scholar] [CrossRef]
- Yoshida, S. Heterogeneous, Dynamic, and Stochastic Nature of Mammalian Spermatogenic Stem Cells. Curr. Top. Dev. Biol. 2019, 135, 245–285. [Google Scholar] [CrossRef]
- Kitadate, Y.; Jörg, D.J.; Tokue, M.; Maruyama, A.; Ichikawa, R.; Tsuchiya, S.; Segi-Nishida, E.; Nakagawa, T.; Uchida, A.; Kimura-Yoshida, C.; et al. Competition for Mitogens Regulates Spermatogenic Stem Cell Homeostasis in an Open Niche. Cell Stem Cell 2019, 24, 79–92.e6. [Google Scholar] [CrossRef] [PubMed]
- Trentin, J.J. Determination of Bone Marrow Stem Cell Differentiation by Stromal Hemopoietic Inductive Microenvironments (HIM). Am. J. Pathol. 1971, 65, 621–628. [Google Scholar]
- Cossío, I.; Lucas, D.; Hidalgo, A. Neutrophils as Regulators of the Hematopoietic Niche. Blood 2019, 133, 2140–2148. [Google Scholar] [CrossRef] [PubMed]
- Tavassoli, M. Studies on Hemopoietic Microenvironments. Report of a Workshop Held in La Jolla, California, August 8–9, 1974. Exp. Hematol. 1975, 3, 213–226. [Google Scholar] [PubMed]
- Crane, G.M.; Jeffery, E.; Morrison, S.J. Adult Haematopoietic Stem Cell Niches. Nat. Rev. Immunol. 2017, 17, 573–590. [Google Scholar] [CrossRef]
- Walker, M.R.; Patel, K.K.; Stappenbeck, T.S. The Stem Cell Niche. J. Pathol. 2009, 217, 169–180. [Google Scholar] [CrossRef]
- Norozi, F.; Shahrabi, S.; Hajizamani, S.; Saki, N. Regulatory Role of Megakaryocytes on Hematopoietic Stem Cells Quiescence by CXCL4/PF4 in Bone Marrow Niche. Leuk. Res. 2016, 48, 107–112. [Google Scholar] [CrossRef]
- Wuchter, P.; Diehlmann, A.; Klüter, H. Closer to Nature: The Role of MSCs in Recreating the Microenvironment of the Hematopoietic Stem Cell Niche in Vitro. Transfus. Med. Hemother. 2022, 49, 258–267. [Google Scholar] [CrossRef] [PubMed]
- Morikawa, T.; Takubo, K. Hypoxia Regulates the Hematopoietic Stem Cell Niche. Pflugers Arch. 2016, 468, 13–22. [Google Scholar] [CrossRef]
- Niazi, V.; Parseh, B.; Ahani, M.; Karami, F.; Gilanchi, S.; Atarodi, K.; Soufi, M.; Soleimani, M.; Ghafouri-Fard, S.; Taheri, M.; et al. Communication between Stromal and Hematopoietic Stem Cell by Exosomes in Normal and Malignant Bone Marrow Niche. Biomed. Pharmacother. 2020, 132, 110854. [Google Scholar] [CrossRef]
- Krenn, P.W.; Montanez, E.; Costell, M.; Fässler, R. Integrins, Anchors and Signal Transducers of Hematopoietic Stem Cells during Development and in Adulthood. Curr. Top. Dev. Biol. 2022, 149, 203–261. [Google Scholar] [CrossRef]
- Zhang, J.; Wu, X.; Ma, J.; Long, K.; Sun, J.; Li, M.; Ge, L. Hypoxia and Hypoxia-Inducible Factor Signals Regulate the Development, Metabolism, and Function of B Cells. Front. Immunol. 2022, 13, 967576. [Google Scholar] [CrossRef]
- Binder, V.; Li, W.; Faisal, M.; Oyman, K.; Calkins, D.L.; Shaffer, J.; Teets, E.M.; Sher, S.; Magnotte, A.; Belardo, A.; et al. Microenvironmental Control of Hematopoietic Stem Cell Fate via CXCL8 and Protein Kinase C. Cell Rep. 2023, 42, 112528. [Google Scholar] [CrossRef] [PubMed]
- Kokkaliaris, K.D.; Kunz, L.; Cabezas-Wallscheid, N.; Christodoulou, C.; Renders, S.; Camargo, F.; Trumpp, A.; Scadden, D.T.; Schroeder, T. Adult blood stem cell localization reflects the abundance of reported bone marrow niche cell types and their combinations. Blood 2020, 136, 2296–2307. [Google Scholar] [CrossRef]
- Khlusov, I.A.; Khlusova, M.Y.; Litvinova, L.S. Native cell domains as stem cell regulatory microterritories for precise tissue engineering. Next Mater. 2023, 1, 100021. [Google Scholar] [CrossRef]
- Garrett, R.W.; Emerson, S.G. Bone and Blood Vessels: The Hard and the Soft of Hematopoietic Stem Cell Niches. Cell Stem Cell 2009, 4, 503–506. [Google Scholar] [CrossRef]
- Kiel, M.J.; Yilmaz, O.H.; Iwashita, T.; Yilmaz, O.H.; Terhorst, C.; Morrison, S.J. SLAM family receptors distinguish hematopoietic stem and progenitor cells and reveal endothelial niches for stem cells. Cell 2005, 121, 1109–1121. [Google Scholar] [CrossRef] [PubMed]
- Bandyopadhyay, S.; Duffy, M.P.; Ahn, K.J.; Sussman, J.H.; Pang, M.; Smith, D.; Duncan, G.; Zhang, I.; Huang, J.; Lin, Y.; et al. Mapping the cellular biogeography of human bone marrow niches using single-cell transcriptomics and proteomic imaging. Cell 2024, 187, 3120–3140.e29. [Google Scholar] [CrossRef]
- Wei, Q.; Frenette, P.S. Niches for hematopoietic stem cells and their progeny. Immunity 2018, 48, 632–648. [Google Scholar] [CrossRef]
- Bello, A.B.; Park, H.; Lee, S.-H. Current Approaches in Biomaterial-Based Hematopoietic Stem Cell Niches. Acta Biomater. 2018, 72, 1–15. [Google Scholar] [CrossRef]
- Vercellino, J.; Małachowska, B.; Kulkarni, S.; Bell, B.I.; Shajahan, S.; Shinoda, K.; Eichenbaum, G.; Verma, A.K.; Ghosh, S.P.; Yang, W.-L.; et al. Thrombopoietin Mimetic Stimulates Bone Marrow Vascular and Stromal Niches to Mitigate Acute Radiation Syndrome. Stem Cell Res. Ther. 2024, 15, 123. [Google Scholar] [CrossRef]
- Bessis, M. Erythroblastic island, functional unity of bone marrow. Rev. Hematol. 1958, 13, 8–11. [Google Scholar] [PubMed]
- May, A.; Forrester, L.M. The Erythroblastic Island Niche: Modeling in Health, Stress, and Disease. Exp. Hematol. 2020, 91, 10–21. [Google Scholar] [CrossRef]
- Yahata, T.; Ibrahim, A.A.; Muguruma, Y.; Eren, M.; Shaffer, A.M.; Watanabe, N.; Kaneko, S.; Nakabayashi, T.; Dan, T.; Hirayama, N.; et al. TGF-β-induced intracellular PAI-1 is responsible for retaining hematopoietic stem cells in the niche. Blood 2017, 130, 2283–2294. [Google Scholar] [CrossRef] [PubMed]
- Ugarte, F.; Forsberg, E.C. Haematopoietic Stem Cell Niches: New Insights Inspire New Questions. EMBO J. 2013, 32, 2535–2547. [Google Scholar] [CrossRef]
- Sottoriva, K.; Pajcini, K.V. Notch Signaling in the Bone Marrow Lymphopoietic Niche. Front. Immunol. 2021, 12, 723055. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Pronk, E.; van Dijk, C.; Bian, Y.; Feyen, J.; van Tienhoven, T.; Yildirim, M.; Pisterzi, P.; de Jong, M.M.E.; Bastidas, A.; et al. A Single-Cell Taxonomy Predicts Inflammatory Niche Remodeling to Drive Tissue Failure and Outcome in Human AML. Blood Cancer Discov. 2023, 4, 394–417. [Google Scholar] [CrossRef]
- Taylor, C.T.; Colgan, S.P. Regulation of Immunity and Inflammation by Hypoxia in Immunological Niches. Nat. Rev. Immunol. 2017, 17, 774–785. [Google Scholar] [CrossRef] [PubMed]
- Piccin, D.; Tufford, A.; Morshead, C.M. Neural Stem and Progenitor Cells in the Aged Subependyma Are Activated by the Young Niche. Neurobiol. Aging 2014, 35, 1669–1679. [Google Scholar] [CrossRef]
- Matteini, F.; Mulaw, M.A.; Florian, M.C. Aging of the Hematopoietic Stem Cell Niche: New Tools to Answer an Old Question. Front. Immunol. 2021, 12, 738204. [Google Scholar] [CrossRef]
- Bianco, P. Minireview: The stem cell next door: Skeletal and hematopoietic stem cell “niches” in bone. Endocrinology 2011, 152, 2957–2962. [Google Scholar] [CrossRef]
- Kolf, C.M.; Cho, E.; Tuan, R.S. Mesenchymal Stromal Cells. Biology of Adult Mesenchymal Stem Cells: Regulation of Niche, Self-Renewal and Differentiation. Arthritis Res. Ther. 2007, 9, 204. [Google Scholar] [CrossRef][Green Version]
- Kuhn, N.Z.; Tuan, R.S. Regulation of Stemness and Stem Cell Niche of Mesenchymal Stem Cells: Implications in Tumorigenesis and Metastasis. J. Cell. Physiol. 2010, 222, 268–277. [Google Scholar] [CrossRef]
- Lin, H.; Sohn, J.; Shen, H.; Langhans, M.T.; Tuan, R.S. Bone Marrow Mesenchymal Stem Cells: Aging and Tissue Engineering Applications to Enhance Bone Healing. Biomaterials 2019, 203, 96–110. [Google Scholar] [CrossRef] [PubMed]
- Guo, T.; Pei, F.; Zhang, M.; Yamada, T.; Feng, J.; Jing, J.; Ho, T.-V.; Chai, Y. Vascular Architecture Regulates Mesenchymal Stromal Cell Heterogeneity via P53-PDGF Signaling in the Mouse Incisor. Cell Stem Cell 2024, 31, 904–920.e6. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.-F.; Zhang, C.; Han, M.-M.; Wang, Y.; Zhou, T.-J.; Xing, L.; Wei, N.; Wang, J.; Jeong, J.-H.; Zhou, F.; et al. Engineered MSCs Break Endothelial-Myofibroblast Crosstalk in Pulmonary Fibrosis: Reconstructing the Vascular Niche. Adv. Mater. 2025, 37, e2414601. [Google Scholar] [CrossRef]
- Schraufstatter, I.U.; Discipio, R.G.; Khaldoyanidi, S. Mesenchymal Stem Cells and Their Microenvironment. Front. Biosci. (Landmark Ed) 2011, 16, 2271–2288. [Google Scholar] [CrossRef]
- Pereira, A.R.; Trivanović, D.; Herrmann, M. Approaches to Mimic the Complexity of the Skeletal Mesenchymal Stem/Stromal Cell Niche in Vitro. Eur. Cell Mater. 2019, 37, 88–112. [Google Scholar] [CrossRef]
- Skiles, M.L.; Sahai, S.; Rucker, L.; Blanchette, J.O. Use of Culture Geometry to Control Hypoxia-Induced Vascular Endothelial Growth Factor Secretion from Adipose-Derived Stem Cells: Optimizing a Cell-Based Approach to Drive Vascular Growth. Tissue Eng. Part A 2013, 19, 2330–2338. [Google Scholar] [CrossRef]
- Chen, L.-C.; Wang, H.-W.; Huang, C.-C. Modulation of Inherent Niches in 3D Multicellular MSC Spheroids Reconfigures Metabolism and Enhances Therapeutic Potential. Cells 2021, 10, 2747. [Google Scholar] [CrossRef]
- Kan, C.; Ding, N.; Yang, J.; Tan, Z.; McGuire, T.L.; Lu, H.; Zhang, K.; Berger, D.M.P.; Kessler, J.A.; Kan, L. BMP-Dependent, Injury-Induced Stem Cell Niche as a Mechanism of Heterotopic Ossification. Stem Cell Res. Ther. 2019, 10, 14. [Google Scholar] [CrossRef]
- Khlusov, I.A.; Khlusova, M.Y.; Zaitsev, K.V.; Kolokol’tsova, T.D.; Sharkeev, Y.P.; Pichugin, V.F.; Legostaeva, E.V.; Trofimova, I.E.; Klimov, A.S.; Zhdanova, A.I. Pilot in Vitro Study of the Parameters of Artificial Niche for Osteogenic Differentiation of Human Stromal Stem Cell Pool. Bull. Exp. Biol. Med. 2011, 150, 535–542. [Google Scholar] [CrossRef]
- Yeo, J.H.; Lam, Y.W.; Fraser, S.T. Cellular Dynamics of Mammalian Red Blood Cell Production in the Erythroblastic Island Niche. Biophys. Rev. 2019, 11, 873–894. [Google Scholar] [CrossRef] [PubMed]
- Han, H.; Rim, Y.A.; Ju, J.H. Recent Updates of Stem Cell-Based Erythropoiesis. Hum. Cell 2023, 36, 894–907. [Google Scholar] [CrossRef] [PubMed]
- Sathyanarayana, P.; Menon, M.P.; Bogacheva, O.; Bogachev, O.; Niss, K.; Kapelle, W.S.; Houde, E.; Fang, J.; Wojchowski, D.M. Erythropoietin Modulation of Podocalyxin and a Proposed Erythroblast Niche. Blood 2007, 110, 509–518. [Google Scholar] [CrossRef][Green Version]
- Chasis, J.A.; Mohandas, N. Erythroblastic islands: Niches for erythropoiesis. Blood 2008, 112, 470–478. [Google Scholar] [CrossRef]
- Pearse, A.G.E. Histochemistry: Theoretical and Applied. Am. J. Med. Sci. 1961, 241, 136. [Google Scholar] [CrossRef]
- Chan, C.K.F.; Seo, E.Y.; Chen, J.Y.; Lo, D.; McArdle, A.; Sinha, R.; Tevlin, R.; Seita, J.; Vincent-Tompkins, J.; Wearda, T.; et al. Identification and Specification of the Mouse Skeletal Stem Cell. Cell 2015, 160, 285–298. [Google Scholar] [CrossRef] [PubMed]
- Ambrosi, T.H.; Chan, C.K.F. Skeletal Stem Cells as the Developmental Origin of Cellular Niches for Hematopoietic Stem and Progenitor Cells. Curr. Top. Microbiol. Immunol. 2021, 434, 1–31. [Google Scholar] [CrossRef] [PubMed]
- Herrmann, M.; Jakob, F. Bone Marrow Niches for Skeletal Progenitor Cells and Their Inhabitants in Health and Disease. Curr. Stem Cell Res. Ther. 2019, 14, 305–319. [Google Scholar] [CrossRef]
- Melis, S.; Trompet, D.; Chagin, A.S.; Maes, C. Skeletal stem and progenitor cells in bone physiology, ageing and disease. Nat. Rev. Endocrinol. 2025, 21, 135–153. [Google Scholar] [CrossRef]
- Chagin, A.S.; Newton, P.T. Postnatal Skeletal Growth Is Driven by the Epiphyseal Stem Cell Niche: Potential Implications to Pediatrics. Pediatr. Res. 2020, 87, 986–990. [Google Scholar] [CrossRef]
- Kaucka, M.; Joven Araus, A.; Tesarova, M.; Currie, J.D.; Boström, J.; Kavkova, M.; Petersen, J.; Yao, Z.; Bouchnita, A.; Hellander, A.; et al. Altered Developmental Programs and Oriented Cell Divisions Lead to Bulky Bones during Salamander Limb Regeneration. Nat. Commun. 2022, 13, 6949. [Google Scholar] [CrossRef]
- Doherty, M.J.; Canfield, A.E. Gene Expression during Vascular Pericyte Differentiation. Crit. Rev. Eukaryot. Gene Expr. 1999, 9, 1–17. [Google Scholar] [CrossRef]
- Barrett, O.; Sottocornola, R.; Lo Celso, C. In Vivo Imaging of Hematopoietic Stem Cells in the Bone Marrow Niche. Methods Mol. Biol. 2012, 916, 231–242. [Google Scholar] [CrossRef]
- Lo Celso, C.; Fleming, H.E.; Wu, J.W.; Zhao, C.X.; Miake-Lye, S.; Fujisaki, J.; Côté, D.; Rowe, D.W.; Lin, C.P.; Scadden, D.T. Live-Animal Tracking of Individual Haematopoietic Stem/Progenitor Cells in Their Niche. Nature 2009, 457, 92–96. [Google Scholar] [CrossRef] [PubMed]
- Photonics Spectra; Coherent Multiphoton Microscopy Sets the Standard for Live-Cell Imaging. Available online: https://www.photonics.com/Articles/Multiphoton_Microscopy_Sets_the_Standard_for/a63225 (accessed on 14 June 2025).
- Seidenari, S.; Arginelli, F.; Dunsby, C.; French, P.M.W.; König, K.; Magnoni, C.; Talbot, C.; Ponti, G. Multiphoton Laser Tomography and Fluorescence Lifetime Imaging of Melanoma: Morphologic Features and Quantitative Data for Sensitive and Specific Non-Invasive Diagnostics. PLoS ONE 2013, 8, e70682. [Google Scholar] [CrossRef]
- Park, S.; Greco, V.; Cockburn, K. Live Imaging of Stem Cells: Answering Old Questions and Raising New Ones. Curr. Opin. Cell Biol. 2016, 43, 30–37. [Google Scholar] [CrossRef]
- Yang, D.; de Haan, G. Inflammation and Aging of Hematopoietic Stem Cells in Their Niche. Cells 2021, 10, 1849. [Google Scholar] [CrossRef]
- Lovegrove, H.E.; Hulmes, G.E.; Ghadaouia, S.; Revell, C.; Giralt-Pujol, M.; Alhashem, Z.; Pena, A.; Nogare, D.D.; Appleton, E.; Costa, G.; et al. Interphase Cell Morphology Defines the Mode, Symmetry, and Outcome of Mitosis. Science 2025, 388, eadu9628. [Google Scholar] [CrossRef]
- Hitrik, A.; Popliker, M.; Gancz, D.; Mukamel, Z.; Lifshitz, A.; Schwartzman, O.; Tanay, A.; Gilboa, L. Combgap Promotes Ovarian Niche Development and Chromatin Association of EcR-Binding Regions in BR-C. PLoS Genet. 2016, 12, e1006330. [Google Scholar] [CrossRef] [PubMed]
- Tjin, G.; Flores-Figueroa, E.; Duarte, D.; Straszkowski, L.; Scott, M.; Khorshed, R.A.; Purton, L.E.; Lo Celso, C. Imaging Methods Used to Study Mouse and Human HSC Niches: Current and Emerging Technologies. Bone 2019, 119, 19–35. [Google Scholar] [CrossRef]
- Salazar-Terreros, M.J.; Vernot, J.-P. In Vitro and In Vivo Modeling of Normal and Leukemic Bone Marrow Niches: Cellular Senescence Contribution to Leukemia Induction and Progression. Int. J. Mol. Sci. 2022, 23, 7350. [Google Scholar] [CrossRef]
- Hong, J.K.; Bang, J.Y.; Xu, G.; Lee, J.-H.; Kim, Y.-J.; Lee, H.-J.; Kim, H.S.; Kwon, S.-M. Thickness-Controllable Electrospun Fibers Promote Tubular Structure Formation by Endothelial Progenitor Cells. Int. J. Nanomed. 2015, 10, 1189–1200. [Google Scholar] [CrossRef] [PubMed]
- Ahn, J.; Jang, H.; Jeong, Y.; Choi, S.; Ko, J.; Hwang, S.H.; Jeong, J.-H.; Jung, Y.S.; Park, I. Illuminating Recent Progress in Nanotransfer Printing: Core Principles, Emerging Applications, and Future Perspectives. Adv. Sci. 2024, 11, e2303704. [Google Scholar] [CrossRef]
- Santos Rosalem, G.; Gonzáles Torres, L.A.; de Las Casas, E.B.; Mathias, F.A.S.; Ruiz, J.C.; Carvalho, M.G.R. Microfluidics and Organ-on-a-Chip Technologies: A Systematic Review of the Methods Used to Mimic Bone Marrow. PLoS ONE 2020, 15, e0243840. [Google Scholar] [CrossRef]
- Weißenbruch, K.; Lemma, E.D.; Hippler, M.; Bastmeyer, M. Micro-Scaffolds as Synthetic Cell Niches: Recent Advances and Challenges. Curr. Opin. Biotechnol. 2022, 73, 290–299. [Google Scholar] [CrossRef] [PubMed]
- Chimenti, I.; Massai, D.; Morbiducci, U.; Beltrami, A.P.; Pesce, M.; Messina, E. Stem Cell Spheroids and Ex Vivo Niche Modeling: Rationalization and Scaling-Up. J. Cardiovasc. Transl. Res. 2017, 10, 150–166. [Google Scholar] [CrossRef]
- Xiao, Y.; McGuinness, C.S.; Doherty-Boyd, W.S.; Salmeron-Sanchez, M.; Donnelly, H.; Dalby, M.J. Current insights into the bone marrow niche: From biology in vivo to bioengineering ex vivo. Biomaterials 2022, 286, 121568. [Google Scholar] [CrossRef]
- Gilchrist, A.E.; Harley, B.A.C. Engineered Tissue Models to Replicate Dynamic Interactions within the Hematopoietic Stem Cell Niche. Adv. Healthc. Mater. 2022, 11, e2102130. [Google Scholar] [CrossRef]
- Hadland, B.; Varnum-Finney, B.; Dozono, S.; Dignum, T.; Nourigat-McKay, C.; Heck, A.M.; Ishida, T.; Jackson, D.L.; Itkin, T.; Butler, J.M.; et al. Engineering a niche supporting hematopoietic stem cell development using integrated single-cell transcriptomics. Nat. Commun. 2022, 13, 1584. [Google Scholar] [CrossRef]
- Matsushita, Y.; Noguchi, A.; Ono, W.; Ono, N. Multi-omics analysis in developmental bone biology. Jpn. Dent. Sci. Rev. 2023, 59, 412–420. [Google Scholar] [CrossRef]
- Szade, K.; Gulati, G.S.; Chan, C.K.F.; Kao, K.S.; Miyanishi, M.; Marjon, K.D.; Sinha, R.; George, B.M.; Chen, J.Y.; Weissman, I.L. Where Hematopoietic Stem Cells Live: The Bone Marrow Niche. Antioxid. Redox Signal. 2018, 29, 191–204. [Google Scholar] [CrossRef]
- Park, S.; Hainaut, P. Editorial: Dissecting the pathological bone marrow niche through single-cell omics and artificial intelligence. Curr. Opin. Oncol. 2020, 32, 137–138. [Google Scholar] [CrossRef] [PubMed]
- Vanegas, N.P.; Vernot, J.P. Loss of quiescence and self-renewal capacity of hematopoietic stem cell in an in vitro leukemic niche. Exp. Hematol. Oncol. 2017, 6, 2. [Google Scholar] [CrossRef] [PubMed]
- Oh, I.H.; Jeong, S.Y.; Kim, J.A. Normal and leukemic stem cell niche interactions. Curr. Opin. Hematol. 2019, 26, 249–257. [Google Scholar] [CrossRef] [PubMed]
- Yuan, N.; Wei, W.; Ji, L.; Qian, J.; Jin, Z.; Liu, H.; Xu, L.; Li, L.; Zhao, C.; Gao, X.; et al. Young donor hematopoietic stem cells revitalize aged or damaged bone marrow niche by transdifferentiating into functional niche cells. Aging Cell 2023, 22, e13889. [Google Scholar] [CrossRef]
- Sagaradze, G.D.; Monakova, A.O.; Basalova, N.A.; Popov, V.S.; Balabanyan, V.Y.; Efimenko, A.Y. Regenerative medicine for male infertility: A focus on stem cell niche injury models. Biomed. J. 2022, 45, 607–614. [Google Scholar] [CrossRef]
- He, W.; Deng, J.; Ma, B.; Tao, K.; Zhang, Z.; Ramakrishna, S.; Yuan, W.; Ye, T. Recent Advancements of Bioinks for 3D Bioprinting of Human Tissues and Organs. ACS Appl. Bio Mater. 2024, 7, 17–43. [Google Scholar] [CrossRef]
- Matai, I.; Kaur, G.; Seyedsalehi, A.; McClinton, A.; Laurencin, C.T. Progress in 3D bioprinting technology for tissue/organ regenerative engineering. Biomaterials 2020, 226, 119536. [Google Scholar] [CrossRef]
- Tashman, J.W.; Shiwarski, D.J.; Feinberg, A.W. Development of a high-performance open-source 3D bioprinter. Sci. Rep. 2022, 12, 22652. [Google Scholar] [CrossRef] [PubMed]
- Dominici, M.; Le Blanc, K.; Mueller, I.; Slaper-Cortenbach, I.; Marini, F.; Krause, D.; Deans, R.; Keating, A.; Prockop, D.; Horwitz, E. Minimal Criteria for Defining Multipotent Mesenchymal Stromal Cells. The International Society for Cellular Therapy Position Statement. Cytotherapy 2006, 8, 315–317. [Google Scholar] [CrossRef] [PubMed]
- Levine, R.F.; Hazzard, K.C.; Lamberg, J.D. The Significance of Megakaryocyte Size. Blood 1982, 60, 1122–1131. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Vainchenker, W.; Raslova, H. Megakaryocyte Polyploidization: Role in Platelet Production. Platelets 2020, 31, 707–716. [Google Scholar] [CrossRef]
| Postulates of Stem Cell Niche Hypothesis Proposed by Schofield [9,10] | Current Development of Schofield’s Hypothesis | Selective References and Sources | Crucial Issues and Relevant Questions of Niche Hypothesis Development |
|---|---|---|---|
| General concept (definition) of the stem cell niche | |||
| The fundamental property of a stem cell is self-renewal, which depends on the microenvironment in which the stem cell is seen in association with other cells, determining its behavior. The cellular environment, which retains the stem cell, is a stem cell ‘niche’. | The niches are the osteal sites in the trabecular bones where CFU give rise to different lineages of hematopoiesis | [15] | There is no agreement on a common ‘niche’ definition from concept proposal to the present day. |
| Orthodox definition A niche is a confined site (specialized microenvironment) in an organ that supports the stem cell self-renewal and maintains the HSC in a quiescent (undifferentiated) state. | [17,18,19,20,21,22] | ||
| Alternative (dynamic) definition A stem cell niche is a distinct, dynamic, hierarchical, and specialized microenvironment that provides for localization and self-renewal, regulates the balance between quiescent and proliferative states, and allows for the choice of fate and differentiation of stem cells and their progenitors. | [13,23,24,25,26,27,28] | ||
| Core components and regulatory mechanisms in stem cell niches | |||
| The microenvironment is a major component of the stem cell system. Three different supporting cell lines are required for stem cells to produce different hematopoietic lineages. | The most tangible advancement of views since Schofield’s hypothesis. Virtually all BM cells and molecules may be components of the niches for stem and progenitor cells. | Figure 1 [13,29,30,31,32,33,34,35,36] | There is no clear understanding of how a local niche is fundamentally different from the formal microenvironment of a stem cell within a specific tissue compartment [37]. This issue is particularly relevant for the stem cells of mesenchymal origin. If a cell niche is a specific anatomical microterritory, spatial restrictions should exist. Otherwise, the molecular and cellular regulatory mechanisms in the niche are not distinguished from those observed in the total HIM (Figure 2). At least, minimal quantitative and qualitative criteria are required for a stem cell niche composition, specifically the HSC, HSPC and MSC niches in adult mammals. |
| Hierarchy of potential niche-candidates for stem and progenitor cells. Topographical distribution | |||
| Stem and progenitor cells can occupy a vacant niche in which they become a stem cell, i.e., they stop their commitment, but their capacity for self-renewal is reduced. | The classical model of hematopoiesis is hierarchical. Therefore, there can also be a niche hierarchy. According to Schofield’s hypothesis, outside the niche, the HSC begins to commit (i.e., becomes a progenitor cell); if the progenitor cell finds another niche, it can return to the ‘quiescent’ state. In this way, a hierarchy of niches emerges, as there are microterritories for both true HSCs and their progenitor cells. Indeed, many niche options have been proposed for HSCs, MSCs, and their progenies. There are numerous empty HSC niches in the BM unoccupied by transplanted HSCs. HSC niches are abundant and are not a limiting factor in vivo. | Figure 3 (Table S2) [38,39,40] | To define that stem cells have found a new niche, it is critically important to know the boundaries of niche space. |
| Their progeny, unless they can occupy a similar stem cell ‘niche’, are first-generation colony-forming cells that proliferate and mature to acquire a high probability of differentiation, i.e., they have an age-structure | There are young and old niches that regulate the ‘aging’ of stem cells. It is assumed that niches for hematopoietic progenitor cells exist within the HIM, in which they proliferate, differentiate, and mature. True anatomical (structural and functional) candidate niches for HSPCs are erythroblastic islands and megakaryocytes. MSCs have an ex vivo reproducible candidate niche whose space they preferentially differentiate into osteoblasts. | Table S2 [26,40,41,42,43,44,45] | To understand that the stem cell has moved out of its niche, we need to clearly define the niche borders. |
| The fundamental property of a stem cell is self-renewal. | In addition to the sites supporting self-renewal stem cells (quiescent niches?), active (activated) niches for hematopoietic precursor cells have been proposed, for example hematopoietic islands (HIs) (Figure 4). Multiple cellular and molecular niche signals that maintain the stem cell pool in a specific functional state have already been identified. | Figure 1 Table S2 [13,46,47] | To what extent do the intercellular interactions and cellular-molecular signals of specialized niches differ from those in the entire HIM? Are there qualitative differences in the signals or in their high concentration in a limited niche volume? To highlight, knowledge of the finite boundaries and dimensions of stem microterritories is required. |
| The hematopoietic niches are in close contact with the bone. | There are many variations in topographical, anatomical, and functional candidate niches for HSPCs and MSCs that are visualized in the BM. | Table S2 [48,49,50] | The location of a stem cell in a tissue site is not sufficient to define its niche, as the niche must have both anatomical and functional characteristics [26]. For example, HSPCs and MSCs are often in close contact in the BM. The question arises: whether this is an MSC-derived niche for HSPC or the MSC niche itself? This could be particularly related to the niche found by Newton in the epiphyseal growth plate [50], as the relationship between hematopoiesis, chondropoiesis, and enchondral ossification is well known [51]. |
| Challenges of dimension and geometry of stem and progenitor cell niches | |||
| Every stem cell niche is occupied. The cells may not have enough space in the niche. | It follows from Schofield’s postulate that a niche has a certain size. However, HSC territories are now considered as micro-niches, niches themselves, as well as macro-niches with a size corresponding to the entire BM. At the same time, erythroblastic islands and megakaryocytic niches for HSPCs have definitive dimensions that can be determined on cytologic and histologic preparations. Furthermore, ex vivo modeling of individual MSC niches demonstrates a preferable size range for enhancing osteogenic differentiation. | Figure 2 [40,42,44,52,53,54] | There is still no generally accepted, clear meaning of the niche boundaries (the shape and size of the stem microterritories). |
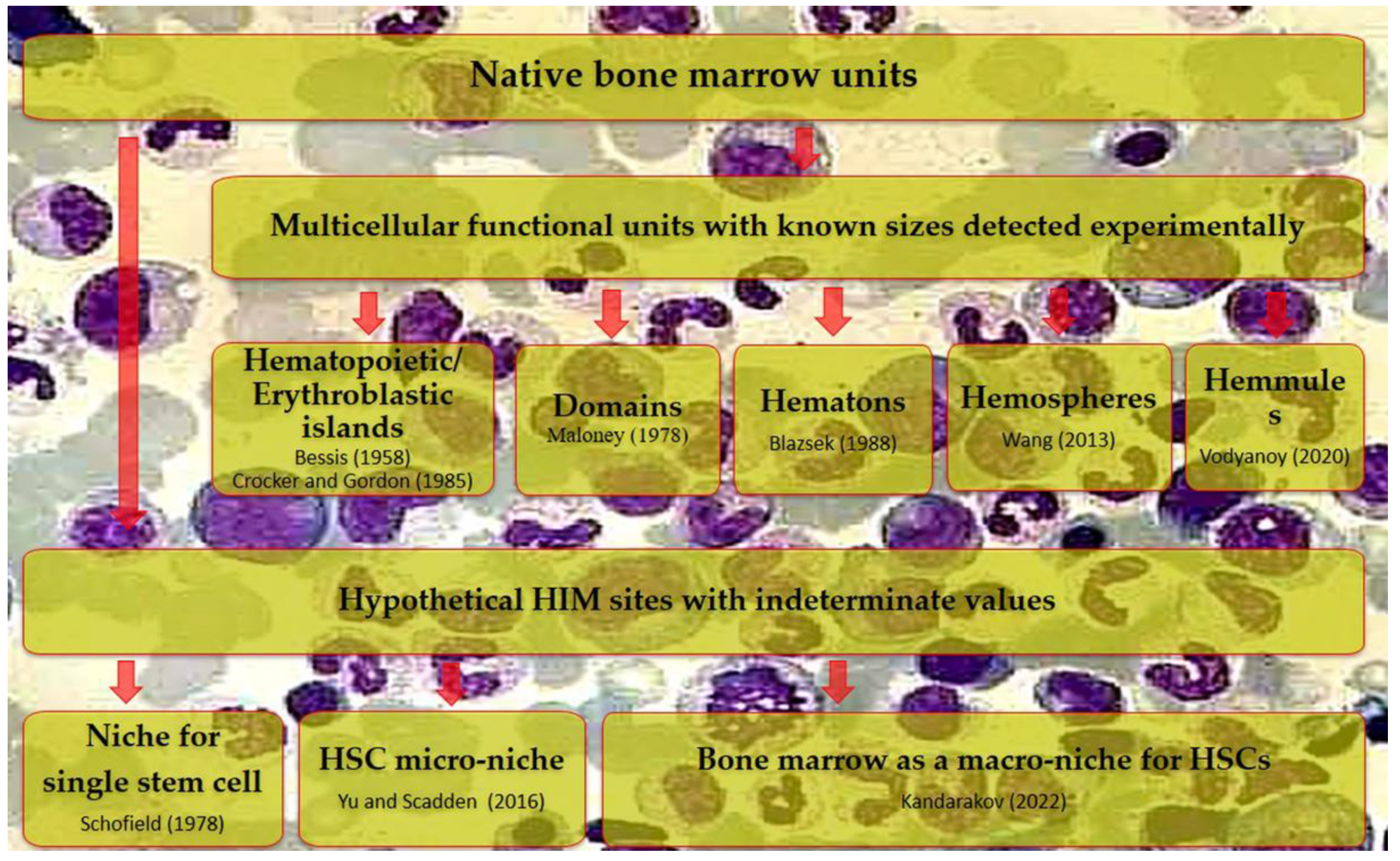
| Stem cells are dependent on their microenvironment. | In addition to the hypothetical niches, large multicellular associations (hematopoietic islands, domains, hematons, hemospheres, hemmules) have been experimentally defined in the BM, reflecting its structural and functional hierarchy as a hematopoietic organ. | Figure 5 [52,55,56,57,58,59] | What is the place of niches in the structural and functional organization of the BM? Are they components of larger cellular (tissue) associations, or can the entire HIM be considered a ‘niche’ with wide variations in its size? |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khlusov, I.; Litvinova, L.; Efimenko, A. Stem Cell Niche Concept: Search for Current Expert Consensus. Int. J. Mol. Sci. 2025, 26, 8422. https://doi.org/10.3390/ijms26178422
Khlusov I, Litvinova L, Efimenko A. Stem Cell Niche Concept: Search for Current Expert Consensus. International Journal of Molecular Sciences. 2025; 26(17):8422. https://doi.org/10.3390/ijms26178422
Chicago/Turabian StyleKhlusov, Igor, Larisa Litvinova, and Anastasia Efimenko. 2025. "Stem Cell Niche Concept: Search for Current Expert Consensus" International Journal of Molecular Sciences 26, no. 17: 8422. https://doi.org/10.3390/ijms26178422
APA StyleKhlusov, I., Litvinova, L., & Efimenko, A. (2025). Stem Cell Niche Concept: Search for Current Expert Consensus. International Journal of Molecular Sciences, 26(17), 8422. https://doi.org/10.3390/ijms26178422






