Growth Arrest-Specific Protein 6 Is Elevated in Endometriosis but Shows Poor Diagnostic Performance
Abstract
1. Introduction
2. Results
2.1. Clinical Characteristics of Patients
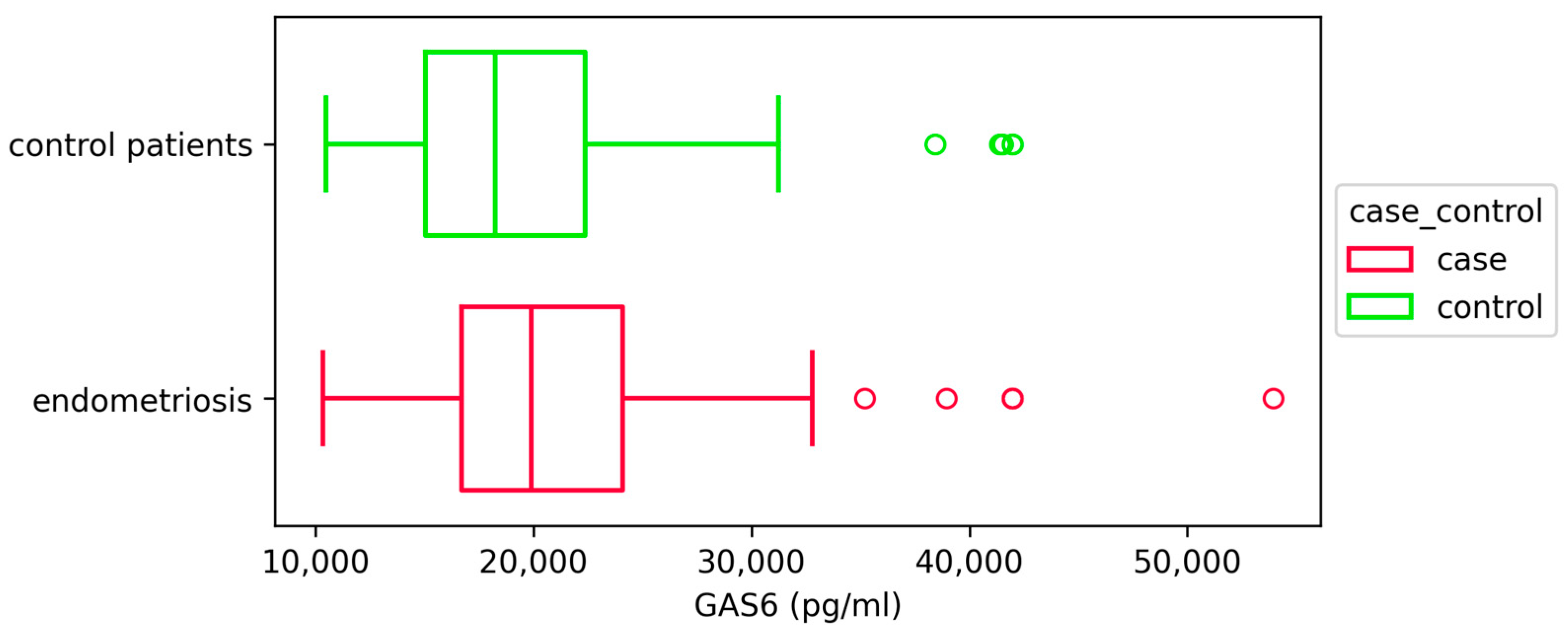
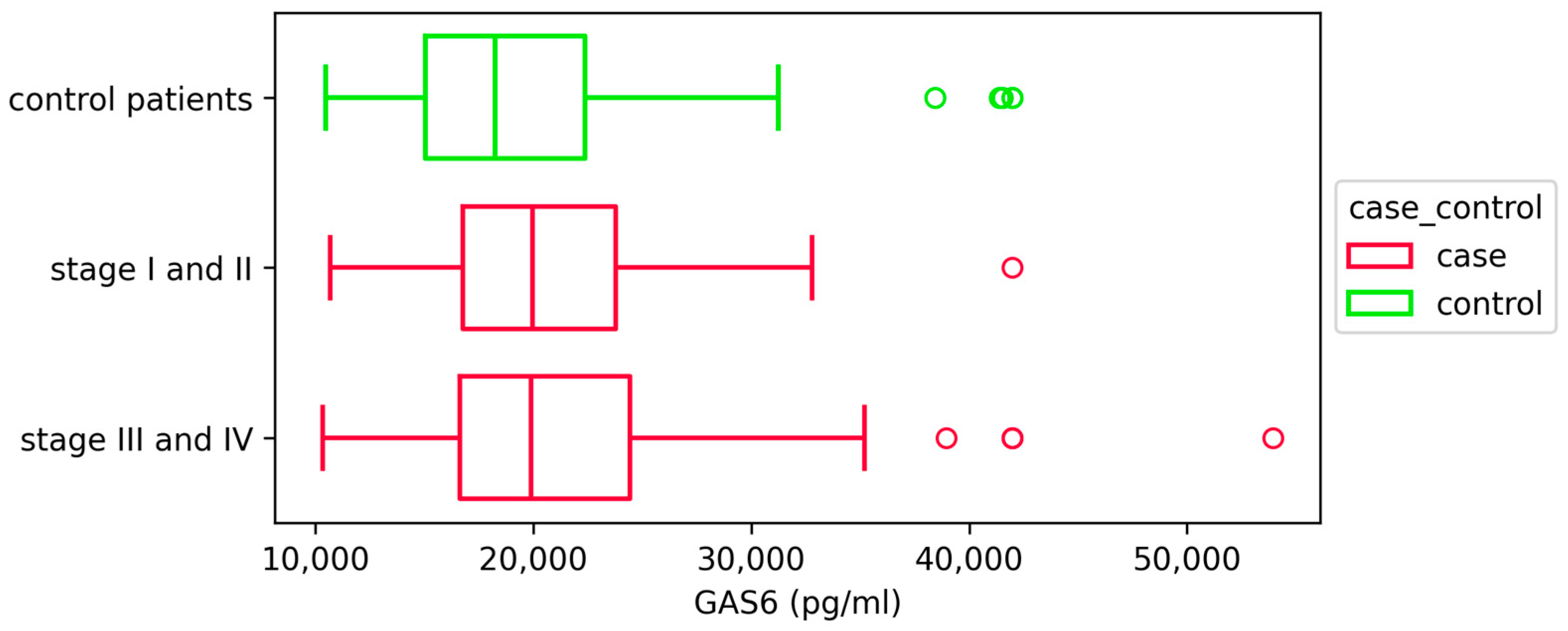
2.2. Plasma GAS6 Concentrations Are Increased in Patients with Endometriosis
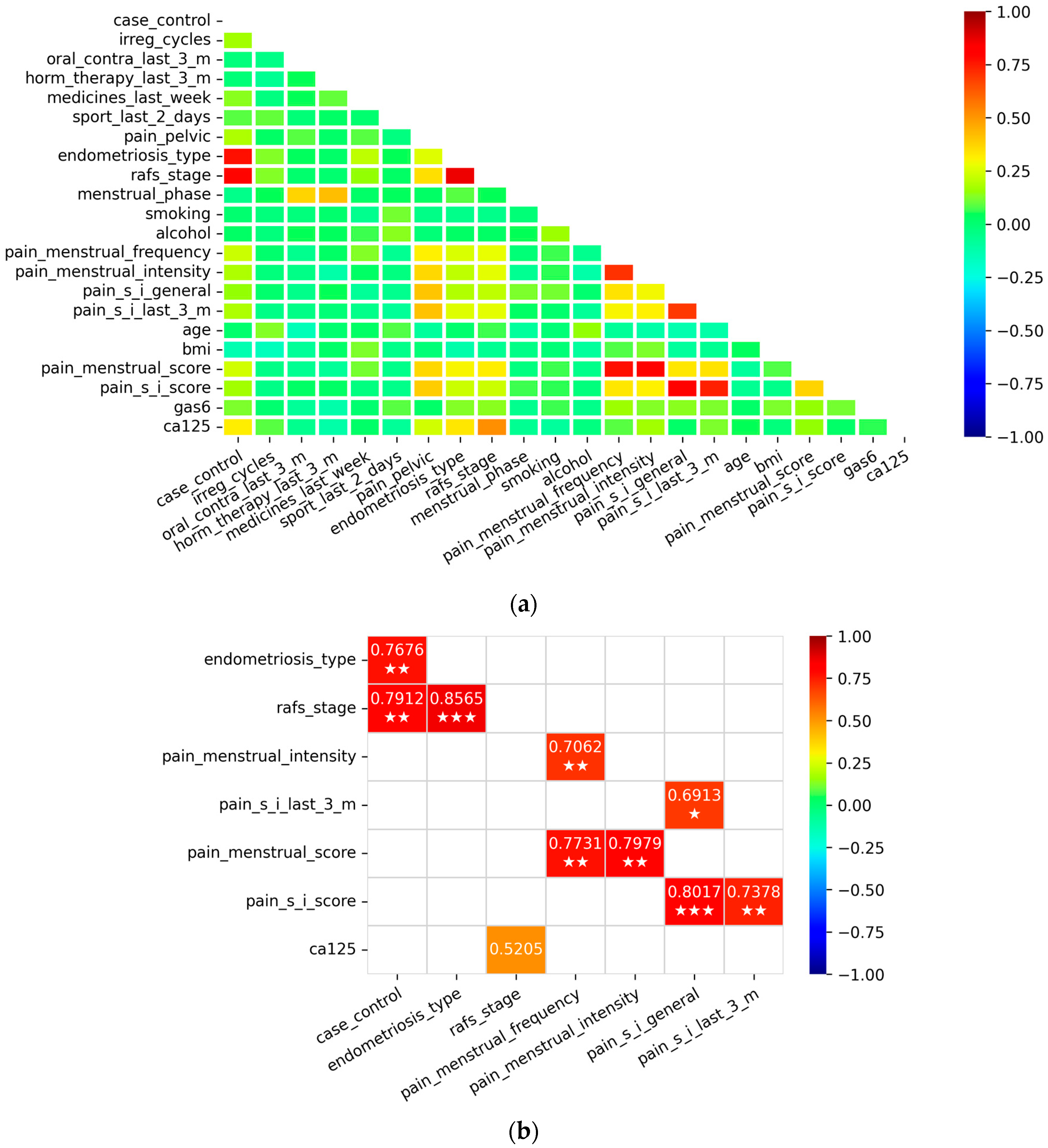
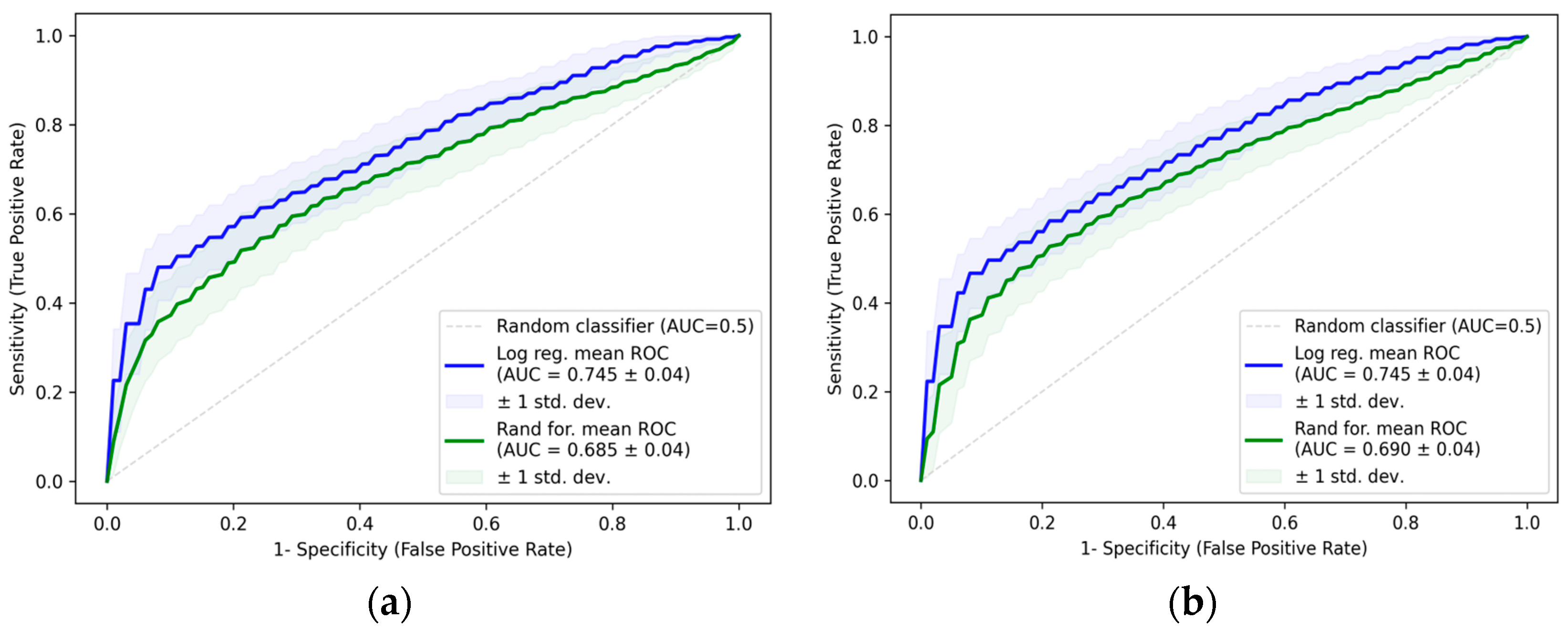
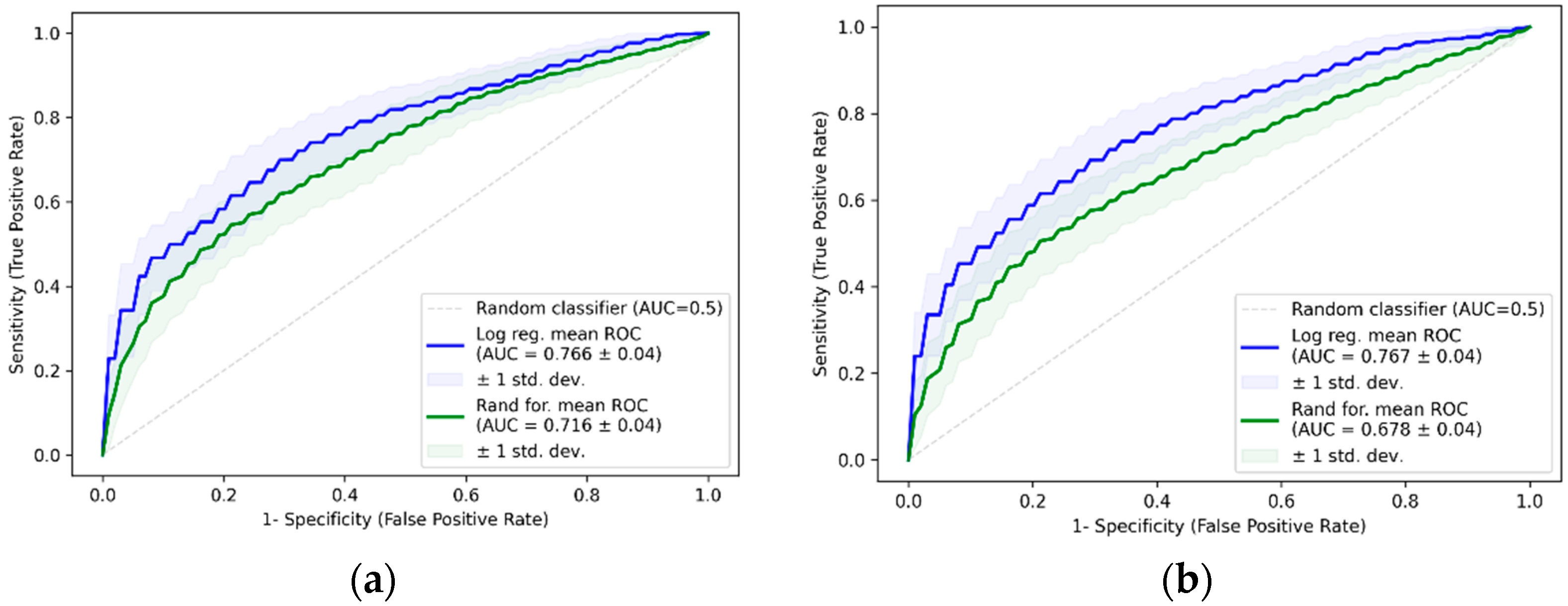
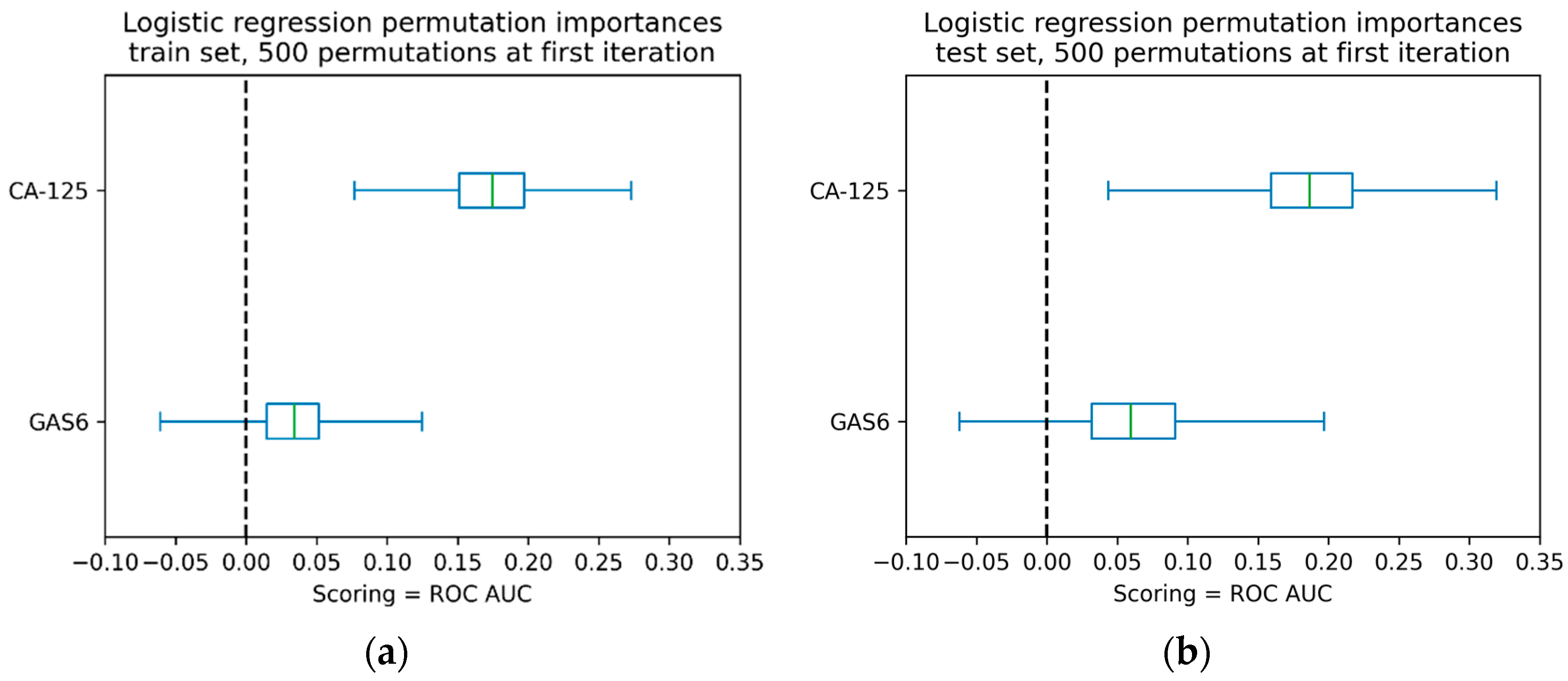
2.3. Logistic Regression Models Including GAS6 and CA-125
3. Discussion
4. Materials and Methods
4.1. Study Design and Patient Selection
4.2. Sample and Data Collection
4.3. ELISA and ECLIA
4.4. Statistical Analyses
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AIC | Akaike’s Information Criterion |
| AKT | Proteinase kinase B |
| ARG2 | Arginase 2 |
| AUC | Area under the curve |
| AXL | AXL receptor tyrosine kinase |
| BMI | Body mass index |
| C3 | Complement component 3 |
| CA-125 | Cancer antigen 125 |
| ECLIA | Electrochemiluminescence immunoassay |
| EDTA | Ethylenediamine tetraacetic acid |
| ELISA | Enzyme-linked immunosorbent assay |
| ERK1/2 | Extracellular signal-regulated kinase 1/2 |
| FAK | Focal adhesion kinase |
| Grb2 | Growth factor receptor-bound protein 2 |
| GAS6 | Growth arrest-specific protein 6 |
| IHC | Immunohistochemistry |
| JAK/STAT3 | Janus kinase/signal transducer and activator of transcription 3 pathway |
| MEK | Mitogen-activated protein kinase kinase |
| MRI | Magnetic resonance imaging |
| mTOR | Mechanistic target of rapamycin |
| NF kappa B | Nuclear factor kappa-light-chain-enhancer of activated B cells |
| PIK3 | Phosphatidylinositol 3-kinase |
| qRT-PCR | Quantitative real-time polymerase chain reaction |
| rAFS | revised American Fertility Society score |
| RAS | Rat sarcoma |
| ROC | Receiver operating characteristic curve |
| SOP | Standard operating procedure |
| Src | Proto-oncogene tyrosine-protein kinase |
| TGFB1 | Transforming growth factor beta 1 |
| TVU | Transvaginal ultrasound |
| VIF | Variance inflation factor |
References
- Zhai, X.; Pu, D.; Wang, R.; Zhang, J.; Lin, Y.; Wang, Y.; Zhai, N.; Peng, X.; Zhou, Q.; Li, L. Gas6/AXL pathway: Immunological landscape and therapeutic potential. Front. Oncol. 2023, 13, 1121130. [Google Scholar] [CrossRef]
- Tanaka, M.; Siemann, D.W. Therapeutic Targeting of the Gas6/Axl Signaling Pathway in Cancer. Int. J. Mol. Sci. 2021, 22, 9953. [Google Scholar] [CrossRef]
- Pei, F.L.; Jia, J.J.; Lin, S.H.; Chen, X.X.; Wu, L.Z.; Lin, Z.X.; Sun, B.-W.; Zeng, C. Construction and evaluation of endometriosis diagnostic prediction model and immune infiltration based on efferocytosis-related genes. Front. Mol. Biosci. 2023, 10, 1298457. [Google Scholar] [CrossRef]
- Henlon, Y.; Panir, K.; McIntyre, I.; Hogg, C.; Dhami, P.; Cuff, A.O.; Senior, A.; Moolchandani-Adwani, N.; Courtois, E.T.; Horne, A.W.; et al. Single-cell analysis identifies distinct macrophage phenotypes associated with prodisease and proresolving functions in the endometriotic niche. Proc. Natl. Acad. Sci. USA 2024, 121, e2405474121. [Google Scholar] [CrossRef] [PubMed]
- Giudice, L.C.; Oskotsky, T.T.; Falako, S.; Opoku-Anane, J.; Sirota, M. Endometriosis in the era of precision medicine and impact on sexual and reproductive health across the lifespan and in diverse populations. Faseb J. 2023, 37, e23130. [Google Scholar] [CrossRef]
- Becker, C.M.; Bokor, A.; Heikinheimo, O.; Horne, A.; Jansen, F.; Kiesel, L.; King, K.; Kvaskoff, M.; Nap, A.; Petersen, K.; et al. ESHRE guideline: Endometriosis. Hum. Reprod. Open 2022, 2022, hoac009. [Google Scholar] [CrossRef]
- Imperiale, L.; Nisolle, M.; Noël, J.C.; Fastrez, M. Three Types of Endometriosis: Pathogenesis, Diagnosis and Treatment. State of the Art. J. Clin. Med. 2023, 12, 994. [Google Scholar] [CrossRef]
- Hogg, C.; Panir, K.; Dhami, P.; Rosser, M.; Mack, M.; Soong, D.; Pollard, J.W.; Jenkins, S.J.; Horne, A.W.; Greaves, E. Macrophages inhibit and enhance endometriosis depending on their origin. Proc. Natl. Acad. Sci. USA 2021, 118, e2013776118. [Google Scholar] [CrossRef] [PubMed]
- Agostinis, C.; Balduit, A.; Mangogna, A.; Zito, G.; Romano, F.; Ricci, G.; Kishore, U.; Bulla, R. Immunological Basis of the Endometriosis: The Complement System as a Potential Therapeutic Target. Front. Immunol. 2021, 11, 2020. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.S.; Misao, R.; Iwagaki, S.; Fujimoto, J.; Tamaya, T. Coexpression of growth arrest-specific gene 6 and receptor tyrosine kinases, Axl and Sky, in human uterine endometrium and ovarian endometriosis. Mol. Hum. Reprod. 2002, 8, 552–558. [Google Scholar] [CrossRef][Green Version]
- Tian, J.; Liu, X. Understanding the Molecular Landscape of Endometriosis: A Bioinformatics Approach to Uncover Signaling Pathways and Hub Genes. Iran. J. Pharm. Res. 2024, 23, e144266. [Google Scholar] [CrossRef]
- Janša, V.; Pušić Novak, M.; Ban Frangež, H.; Rižner, T.L. TGFBI as a candidate biomarker for non-invasive diagnosis of early-stage endometriosis. Hum. Reprod. 2023, 38, 1284–1296. [Google Scholar] [CrossRef]
- Nisenblat, V.; Prentice, L.; Bossuyt, P.M.; Farquhar, C.; Hull, M.L.; Johnson, N. Combination of the non-invasive tests for the diagnosis of endometriosis. Cochrane Database Syst. Rev. 2016, 2016, CD012281. [Google Scholar] [CrossRef]
- Knific, T.; Vouk, K.; Vogler, A.; Osredkar, J.; Gstöttner, M.; Wenzl, R.; Rižner, T.L. Models including serum CA-125, BMI, cyst pathology, dysmenorrhea or dyspareunia for diagnosis of endometriosis. Biomarkers Med. 2018, 12, 737–747. [Google Scholar] [CrossRef] [PubMed]
- Vouk, K.; Hevir, N.; Ribić-Pucelj, M.; Haarpaintner, G.; Scherb, H.; Osredkar, J.; Möller, G.; Prehn, C.; Rižner, T.L.; Adamski, J. Discovery of phosphatidylcholines and sphingomyelins as biomarkers for ovarian endometriosis. Hum. Reprod. 2012, 27, 2955–2965. [Google Scholar] [CrossRef]
- Gabriel, M.; Fey, V.; Heinosalo, T.; Adhikari, P.; Rytkönen, K.; Komulainen, T.; Huhtinen, K.; Laajala, T.D.; Siitari, H.; Virkki, A.; et al. A relational database to identify differentially expressed genes in the endometrium and endometriosis lesions. Sci. Data 2020, 7, 284. [Google Scholar] [CrossRef]
- Bacci, M.; Capobianco, A.; Monno, A.; Cottone, L.; Di Puppo, F.; Camisa, B.; Mariani, M.; Brignole, C.; Ponzoni, M.; Ferrari, S.; et al. Macrophages are alternatively activated in patients with endometriosis and required for growth and vascularization of lesions in a mouse model of disease. Am. J. Pathol. 2009, 175, 547–556. [Google Scholar] [CrossRef]
- Ekman, C.; Stenhoff, J.; Dahlbäck, B. Gas6 is complexed to the soluble tyrosine kinase receptor Axl in human blood. J. Thromb. Haemost. 2010, 8, 838–844. [Google Scholar] [CrossRef] [PubMed]
- van der Meer, J.H.; van der Poll, T.; van’t Veer, C. TAM receptors, Gas6, and protein S: Roles in inflammation and hemostasis. Blood 2014, 123, 2460–2469. [Google Scholar] [CrossRef]
- Tjwa, M.; Bellido-Martin, L.; Lin, Y.; Lutgens, E.; Plaisance, S.; Bono, F.; Delesque-Touchard, N.; Hervé, C.; Moura, R.; Billiau, A.D.; et al. Gas6 promotes inflammation by enhancing interactions between endothelial cells, platelets, and leukocytes. Blood 2008, 111, 4096–4105. [Google Scholar] [CrossRef] [PubMed]
- Tondo, G.; Perani, D.; Comi, C. TAM Receptor Pathways at the Crossroads of Neuroinflammation and Neurodegeneration. Dis. Markers 2019, 2019, 2387614. [Google Scholar] [CrossRef] [PubMed]
- Hurtado, B.; de Frutos, P.G. GAS6 in systemic inflammatory diseases: With and without infection. Crit. Care 2010, 14, 1003. [Google Scholar] [CrossRef] [PubMed]
- Rizner, T.L.; Adamski, J. Paramount importance of sample quality in pre-clinical and clinical research-Need for standard operating procedures (SOPs). J. Steroid Biochem. Mol. Biol. 2019, 186, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Hudson, Q.J.; Perricos, A.; Wenzl, R.; Yotova, I. Challenges in uncovering non-invasive biomarkers of endometriosis. Exp. Biol. Med. 2020, 245, 437–447. [Google Scholar] [CrossRef]
- Azeze, G.G.; Wu, L.; Alemu, B.K.; Lee, W.F.; Fung, L.W.Y.; Cheung, E.C.W.; Zhang, T.; Wang, C.C. Proteomics approach to discovering non-invasive diagnostic biomarkers and understanding the pathogenesis of endometriosis: A systematic review and meta-analysis. J. Transl. Med. 2024, 22, 685. [Google Scholar] [CrossRef]
- Bendifallah, S.; Dabi, Y.; Suisse, S.; Delbos, L.; Spiers, A.; Poilblanc, M.; Golfier, F.; Jornea, L.; Bouteiller, D.; Fernandez, H.; et al. Validation of a Salivary miRNA Signature of Endometriosis—Interim Data. NEJM Evid. 2023, 2, EVIDoa2200282. [Google Scholar] [CrossRef]
- Bjorkman, S.; Taylor, H.S. MicroRNAs in endometriosis: Biological function and emerging biomarker candidates. Biol. Reprod. 2019, 100, 1135–1146, Erratum in Biol. Reprod. 2019, 101, 1179. [Google Scholar] [CrossRef]
- Adamyan, L.; Pivazyan, L.; Zarova, E.; Avetisyan, J.; Laevskaya, A.; Sarkisova, A.; Stepanian, A. Metabolomic biomarkers of endometriosis: A systematic review. J. Endometr. Uterine Disord. 2024, 7, 100077. [Google Scholar] [CrossRef]
- Andrieu, T.; Duo, A.; Duempelmann, L.; Patzak, M.; Saner, F.A.M.; Skrabalova, J.; Donato, C.; Nestorov, P.; Mueller, M.D. Single-Cell RNA Sequencing of PBMCs Identified Junction Plakoglobin (JUP) as Stratification Biomarker for Endometriosis. Int. J. Mol. Sci. 2024, 25, 13071. [Google Scholar] [CrossRef]
- Wang, H.; Gan, Z.; Wang, Y.; Hu, D.; Zhang, L.; Ye, F.; Duan, P. A Noninvasive Menstrual Blood-Based Diagnostic Platform for Endometriosis Using Digital Droplet Enzyme-Linked Immunosorbent Assay and Single-Cell RNA Sequencing. Research 2025, 8, 0652. [Google Scholar] [CrossRef]
- Dungate, B.; Tucker, D.R.; Goodwin, E.; Yong, P.J. Assessing the Utility of artificial intelligence in endometriosis: Promises and pitfalls. Women’s Health 2024, 20, 17455057241248121. [Google Scholar] [CrossRef] [PubMed]
- Wewers, M.E.; Lowe, N.K. A critical review of visual analogue scales in the measurement of clinical phenomena. Res. Nurs. Health 1990, 13, 227–236. [Google Scholar] [CrossRef] [PubMed]
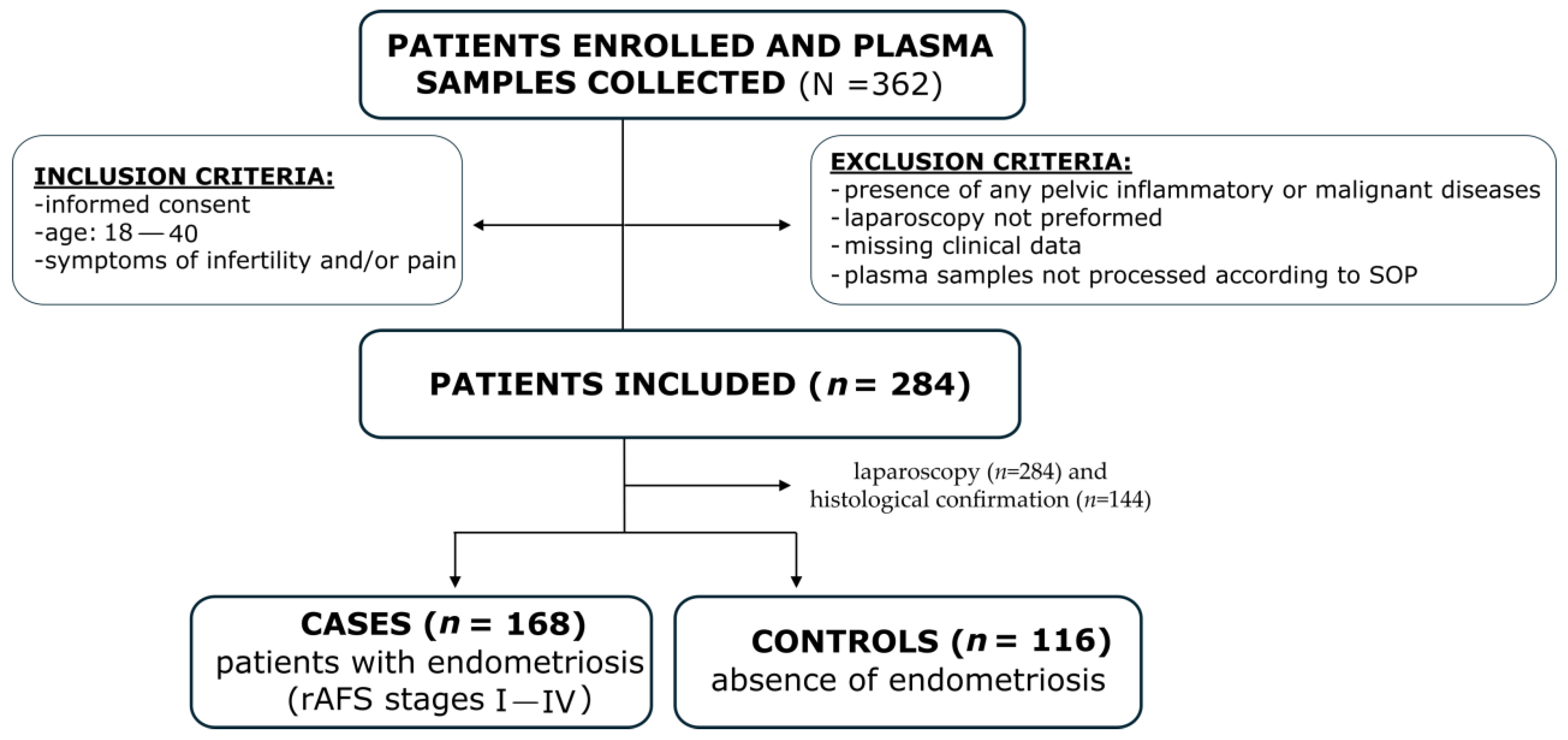

| Parameter | Subgroup | Cases, n = 168 Mean ± SD/n (%) | Controls, n = 116 Mean ± SD/n (%) | p-Value * |
|---|---|---|---|---|
| age (years) | - | 30.51 ± 4.11 | 30.38 ± 4.88 | ns |
| BMI (kg/m2) | - | 22.82 ± 3.83 | 24.01 ± 4.64 | p < 0.05 |
| menstrual phase | OHC | 17 (0.10) | 14 (0.12) | ns |
| proliferative | 82 (0.49) | 51 (0.44) | ||
| secretory | 69 (0.41) | 51 (0.44) | ||
| type of endometriosis | 0_no_endometriosis | 0 (0.00) | 116 (1.0) | 0 = control |
| 1_ovarian | 21 (0.12) | 0 (0.0) | ||
| 2_peritoneal | 71 (0.42) | 0 (0.0) | ||
| 3_ovarian_and_peritoneal | 38 (0.23) | 0 (0.0) | ||
| 4_deep | 6 (0.04) | 0 (0.0) | ||
| 5_ovarian_and_peritoneal_and_deep | 18 (0.11) | 0 (0.0) | ||
| 6_ovarian_and_deep | 9 (0.05) | 0 (0.0) | ||
| 7_peritoneal_and_deep | 5 (0.03) | 0 (0.0) | ||
| rAFS stage | 0_no_endometriosis | 0 (0.00) | 116 (1.0) | 0 = control |
| I | 61 (0.36) | 0 (0.0) | ||
| II | 25 (0.15) | 0 (0.0) | ||
| III | 52 (0.31) | 0 (0.0) | ||
| IV | 30 (0.18) | 0 (0.0) | ||
| regularity of menstrual cycle | no | 4 (0.02) | 12 (0.1) | p < 0.05 |
| yes | 164 (0.98) | 104 (0.9) | ||
| oral contraception last 3 months | no | 155 (0.92) | 106 (0.91) | ns |
| yes | 13 (0.08) | 10 (0.09) | ||
| hormonal therapy last 3 months | no | 143 (0.85) | 98 (0.84) | ns |
| yes | 25 (0.15) | 18 (0.16) | ||
| medicines—last week | no | 107 (0.64) | 89 (0.77) | p < 0.05 |
| yes | 61 (0.36) | 27 (0.23) | ||
| dysmenorrhea score | visual analogue scale (1–10) | 6.00 ± 2.50 | 4.73 ± 2.67 | p < 0.05 |
| dyspareunia score | visual analogue scale (1–10) | 2.70 ± 2.40 | 1.91 ± 2.02 | p < 0.05 |
| dysmenorrhea—frequency | 1_never | 0 (0.00) | 2 (0.02) | p < 0.05 |
| 2_almost_never | 11 (0.07) | 21 (0.18) | ||
| 3_sometimes | 34 (0.20) | 22 (0.19) | ||
| 4_quite_often | 25 (0.15) | 31 (0.27) | ||
| 5_very_often | 98 (0.58) | 40 (0.34) | ||
| dysmenorrhea—intensity | 0_no_pain | 16 (0.10) | 22 (0.19) | p < 0.05 |
| 1_slight_pain | 62 (0.37) | 55 (0.47) | ||
| 2_medium_pain | 60 (0.36) | 24 (0.21) | ||
| 3_strong_pain | 30 (0.18) | 15 (0.13) | ||
| dyspareunia (general) | 1_never | 43 (0.26) | 38 (0.33) | p < 0.05 |
| 2_almost_never | 39 (0.23) | 31 (0.27) | ||
| 3_sometimes | 52 (0.31) | 37 (0.32) | ||
| 4_quite_often | 17 (0.10) | 7 (0.06) | ||
| 5_very_often | 17 (0.10) | 3 (0.03) | ||
| dyspareunia (last 3 months) | 0_no_pain | 56 (0.33) | 58 (0.50) | p < 0.05 |
| 1_slight_pain | 72 (0.43) | 42 (0.36) | ||
| 2_medium_pain | 36 (0.21) | 15 (0.13) | ||
| 3_strong_pain | 4 (0.02) | 1 (0.01) | ||
| pelvic, abdominal or back pain | no | 106 (0.63) | 93 (0.8) | p < 0.05 |
| yes | 62 (0.37) | 23 (0.2) | ||
| smoking status | 1_non_smoker | 105 (0.62) | 73 (0.63) | ns |
| 2_smoker | 42 (0.25) | 28 (0.24) | ||
| 3_smoker_occas_week | 6 (0.04) | 3 (0.03) | ||
| 4_smoker_occas_month | 2 (0.01) | 4 (0.03) | ||
| 5_smoker_former | 13 (0.08) | 8 (0.07) | ||
| alcohol | 1_never | 47 (0.28) | 30 (0.26) | ns |
| 2_rarely | 102 (0.61) | 76 (0.66) | ||
| 3_once_a_week | 9 (0.05) | 7 (0.06) | ||
| 4_2_to_3_times_a_week | 8 (0.05) | 2 (0.02) | ||
| 5_more_than_3_times_a_week | 2 (0.01) | 1 (0.01) | ||
| sport/recreation—last 2 days (before surgery) | no | 109 (0.65) | 85 (0.73) | ns |
| yes | 59 (0.35) | 31 (0.27) |
| Parameter | Cases, n = 168 Mean ± SD/n (%) | Controls, n = 116 Mean ± SD/n (%) | p-Value * |
|---|---|---|---|
| GAS6 (pg/mL) | 21,056.09 ± 6664.89 | 19,499.93 ± 6465.49 | p < 0.05 |
| CA-125 (U/mL) | 50.33 ± 61.46 | 17.50 ± 13.33 | p < 0.05 |
| Method | Predictor | Avg AUC ± sd | Avg AIC ± sd | Avg Sensitivity ± sd (%) | Avg Specificity ± sd (%) |
|---|---|---|---|---|---|
| Log reg. | GAS6 | 0.583 ± 0.05 | 258.17 ± 2.09 | 96 ± 6 | 5 ± 7 |
| Rand for. | GAS6 | 0.536 ± 0.05 | / | 63 ± 7 | 46 ± 8 |
| Log reg. | CA-125 | 0.745 ± 0.04 | 217.74 ± 6.81 | 66 ± 7 | 67 ± 7 |
| Rand for. | CA-125 | 0.685 ± 0.04 | / | 69 ± 6 | 55 ± 8 |
| Log reg. | GAS6 + CA-125 | 0.745 ± 0.04 | 218.52 ± 6.81 | 66 ± 7 | 68 ± 7 |
| Rand for. | GAS6 + CA-125 | 0.690 ± 0.04 | / | 70 ± 6 | 56 ± 8 |
| Log reg. | CA-125 + dysmenorrhea frequency | 0.766 ± 0.04 | 214.37 ± 7.18 | 74 ± 7 | 63 ± 9 |
| Log reg. | CA-125 + dysmenorrhea score + sport last two days before surgery | 0.767 ± 0.04 | 214.08 ± 7.05 | 73 ± 7 | 66 ± 7 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pušić, M.N.; Marijan, R.; Klančič, T.; Knific, T.; Ban Frangež, H.; Rižner, T.L. Growth Arrest-Specific Protein 6 Is Elevated in Endometriosis but Shows Poor Diagnostic Performance. Int. J. Mol. Sci. 2025, 26, 8348. https://doi.org/10.3390/ijms26178348
Pušić MN, Marijan R, Klančič T, Knific T, Ban Frangež H, Rižner TL. Growth Arrest-Specific Protein 6 Is Elevated in Endometriosis but Shows Poor Diagnostic Performance. International Journal of Molecular Sciences. 2025; 26(17):8348. https://doi.org/10.3390/ijms26178348
Chicago/Turabian StylePušić, Maja Novak, Robert Marijan, Teja Klančič, Tamara Knific, Helena Ban Frangež, and Tea Lanišnik Rižner. 2025. "Growth Arrest-Specific Protein 6 Is Elevated in Endometriosis but Shows Poor Diagnostic Performance" International Journal of Molecular Sciences 26, no. 17: 8348. https://doi.org/10.3390/ijms26178348
APA StylePušić, M. N., Marijan, R., Klančič, T., Knific, T., Ban Frangež, H., & Rižner, T. L. (2025). Growth Arrest-Specific Protein 6 Is Elevated in Endometriosis but Shows Poor Diagnostic Performance. International Journal of Molecular Sciences, 26(17), 8348. https://doi.org/10.3390/ijms26178348







