1. Introduction
Natural products refer to compounds capable of exerting one or more biological activities, obtained from bacteria, fungi, terrestrial and marine plants, and/or marine animals [
1]. Bioactive compounds represent an important source of diverse and largely complex molecules, capable of addressing human needs and challenges [
2]. Biologically active compounds primarily belong to secondary metabolism of organisms, which provide them with the necessary tools for adaptation and survival in their native environments; this is why these molecules exhibit unique characteristics and properties [
3,
4]. Although few natural products are approved for medical use or direct human consumption, many serve as models for synthesis of more effective compounds—those with higher bioavailability [
5,
6].
Genus
Porophyllum belongs to the Asteraceae family, which includes plants known for their diverse and high content of bioactive compounds, as well as their use in food production [
7]. Species of genus
Porophyllum are native to the Western Hemisphere, ranging from southern United States to South America, and thrive in tropical and subtropical areas [
8,
9]. Although there are some reports regarding the bioactive capacity and presence of natural products in plants of genus
Porophyllum [
8,
10,
11,
12,
13,
14,
15,
16,
17], studies on bioactive properties of
P. gracile are scarce [
18], despite its long-time use in traditional medicine by Sonoran ethnic groups in Mexico [
19].
The use of traditional medicine based on plants still dominates many therapeutic practices worldwide. Bioactive compounds in plants remain essential for the development of modern medicine and represent a substantial portion of current pharmaceutical agents [
4].
Besides what was reported by Guillet et al. [
18], there are no studies that have evaluated phytochemical composition and bioactive compounds in
P. gracile, which creates a gap in the existing knowledge about this species. Therefore, a detailed characterization of phytochemical composition of
P. gracile will not only describe the compounds present, but also help identify previously unreported compounds, bioactive compounds, and those with potential future applications.
Due to the traditional medicinal use of P. gracile and considering the studies reported for other species of the same genus, it has been hypothesized that P. gracile contains a high diversity of molecules with different chemical natures and high bioactive potential, specifically in vitro antioxidant effects and antiproliferative effects on cancer cells. Therefore, the aim of this study was to determine the profile of volatile compounds, phenolic compounds, and fatty acids in the dried leaves and stems of P. gracile, as well as to explore the antioxidant (in vitro and in cells), antiproliferative (in cancer cells), and cytoprotective (in apparently healthy cells) effects of P. gracile extracts obtained using polar and non-polar solvents.
3. Discussion
Plants of genus
Porophyllum belong to the Asteraceae family, which have been used in traditional medicine throughout history, and there are various reports regarding their bioactive components [
9]. However, specifically within genus
Porophyllum, only eight species have been investigated in terms of their phytochemical composition or evaluation of their bioactive properties, out of a total of 25 species comprising the genus [
8,
9,
11,
12,
13,
17,
18,
22,
23,
24,
25,
26,
27,
28]. It has been reported that plants of this genus contain terpenes, aldehydes and alcohols, alkenes and alkanes, furan and thiophene derivatives, certain aromatic ethers, thiols and fatty acids, among other compounds.
However, specifically for the species
Porophyllum gracile, only identification of seven compounds has been reported: α-pinene, sabinene, myrcene, β-cubebene, 7-tetradecene, pentadecanal, and heptadecanal [
18]. Therefore, the results obtained in the present study represent a significant contribution to the knowledge of the phytochemical composition of
P. gracile, as they allowed the identification of more than 120 distinct compounds in leaves and stems. This phytochemical diversity, previously unreported for the species, includes mainly fatty acids, chlorogenic acid and its derivatives, quercetin-derived compounds, terpenes, aldehydes, ketones, and alcohols, among others. These findings substantially expand the known phytochemical profile of
P. gracile and provide a solid foundation for future research on its biological and pharmacological potential.
It is important to highlight that, although some of compounds reported in the present study coincide with those previously identified in other species of the genus
Porophyllum [
9], the detection of chlorogenic acid derivatives, quercetin-derived compounds, and certain fatty acids represent novel findings within the genus. This reinforces the uniqueness of the phytochemical profile of
P. gracile and highlights its potential as a source of bioactive metabolites.
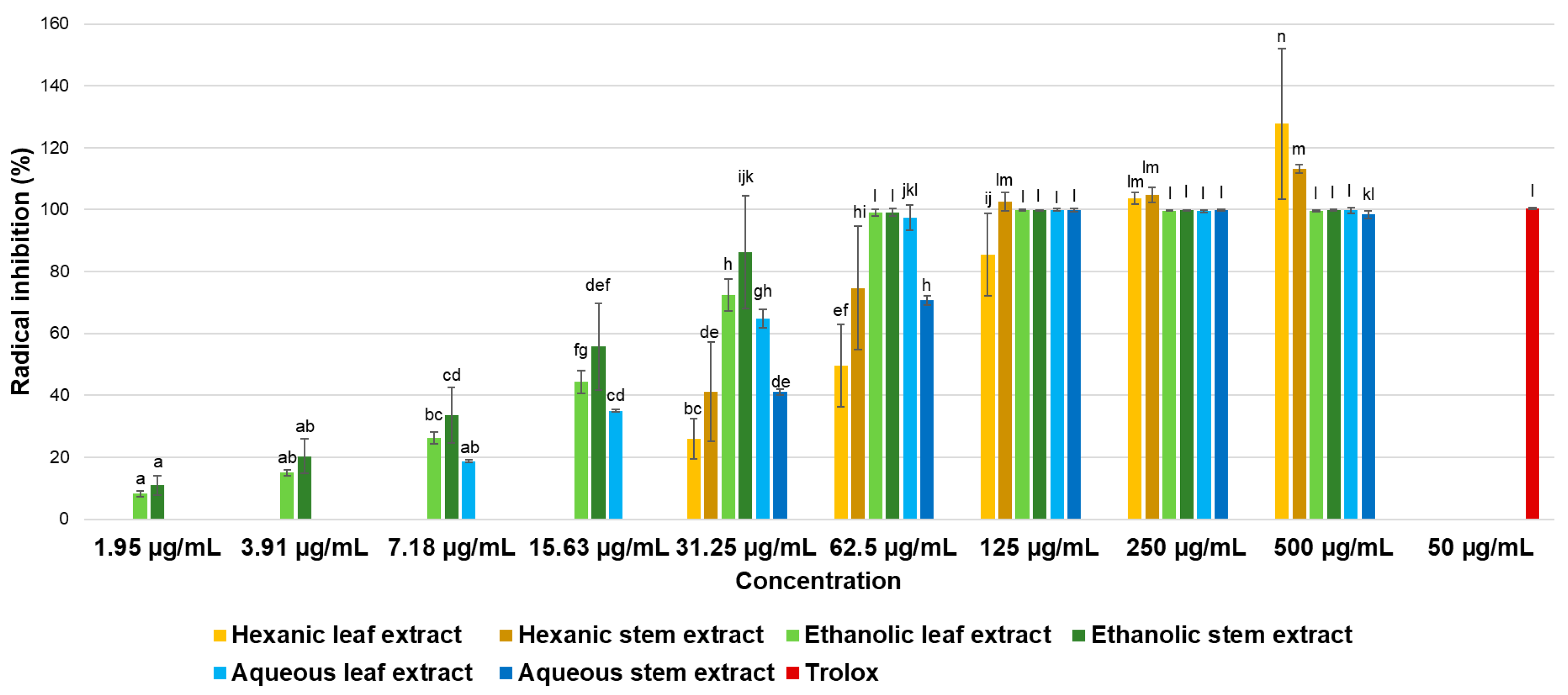
On the other hand, regarding evaluated antioxidant activity, ABTS and DPPH radical-based assays are considered mixed-mode methods capable of assessing antioxidant activity through both single electron transfer (SET) and hydrogen atom transfer (HAT) mechanisms. However, the specific information they provide depends on reaction conditions. Rapid decolorization reactions of ABTS reagent indicate radical stabilization via electron transfer [
29] as observed in the present study, suggesting the possible existence of an electron transfer-based antioxidant mechanism by compounds present in extracts. This hypothesis is supported by the limited activity observed against DPPH radical, an assay that primarily evaluates hydrogen atom transfer mechanisms in the conditions evaluated [
29]. Further support for this proposed mechanism comes from FRAP assay results, as the extracts exhibiting the highest activity in this method (ethanolic leaf extract, ethanolic stem extract, and aqueous leaf extract) also demonstrated the greatest antioxidant capacities against ABTS radical.
Conde-Hernández & Guerrero-Beltrán [
17] reported antioxidant activity against ABTS radical from extracts of the plant
Porophyllum ruderale, obtaining the highest activity from ethanol–water extracts compared to aqueous or pure ethanol extracts. They also observed better results when using fresh plant material rather than dried material as, according to authors, some antioxidant molecules are sensitive to heat, while others may evaporate during the drying process. It is important to note that in the present study, no extractions were performed with solvent mixtures, as this is the first analysis of this nature using
P. gracile, so the possibility of obtaining higher activity using ethanol–water for serial extraction, as well as using fresh plant material, cannot be ruled out. On the other hand, Jiménez et al. [
25] reported high antioxidant activity from
Porophyllum tagetoides extracts against the DPPH radical, which contrasts with the findings of the present study where extracts obtained from
P. gracile did not show activity against this radical according to the established limits. In both studies, authors attributed the antioxidant activity to presence of phenolic compounds in plants; however, in present research, although phenolic compounds were detected, their concentration is likely to have been lower compared to other compounds present in extracts, which may have contributed to reduced antioxidant activity observed against DPPH.
Furthermore, regarding antioxidant activity observed and compounds identified in the present study, chlorogenic acid has been reported to exert various bioactive properties related to its ability to regulate inflammatory and oxidative processes, as well as to maintain metabolic homeostasis by effectively eliminating excess free radicals [
30]. Quercetin, for its part, is a flavonoid that has been reported as a potent antioxidant, as well as having anti-inflammatory, antiproliferative, anticancer, antidiabetic, antiviral properties, and protective effects against neurodegenerative diseases. In food sources, quercetin is typically found in the form of quercetin glycosides, such as those identified in
P. gracile in the present study [
31,
32,
33]. Regarding fatty acids, α-linolenic acid, a major fatty acid found in leaves and one of the main components in stems in this study, is an essential omega-3 fatty acid which has already been described for its ability to counteract oxidative stress by decreasing the concentration of ROS, reducing lipid peroxidation, and increasing levels of antioxidant enzymes such as superoxide dismutase, glutathione peroxidase, and catalase, besides being the precursor of EPA and DHA [
34,
35]. In addition, it has been associated with reduced incidence of stroke and coronary heart disease, as well as antitumor properties, among others [
36]. Moreover, the presence of several terpenes was detected; these compounds are main constituents of essential oils and have been widely reported to exhibit various bioactivities, including antioxidant, antiplasmodial, antiviral, anticancer, antidiabetic, anti-inflammatory, and antiseptic properties, among others [
37].
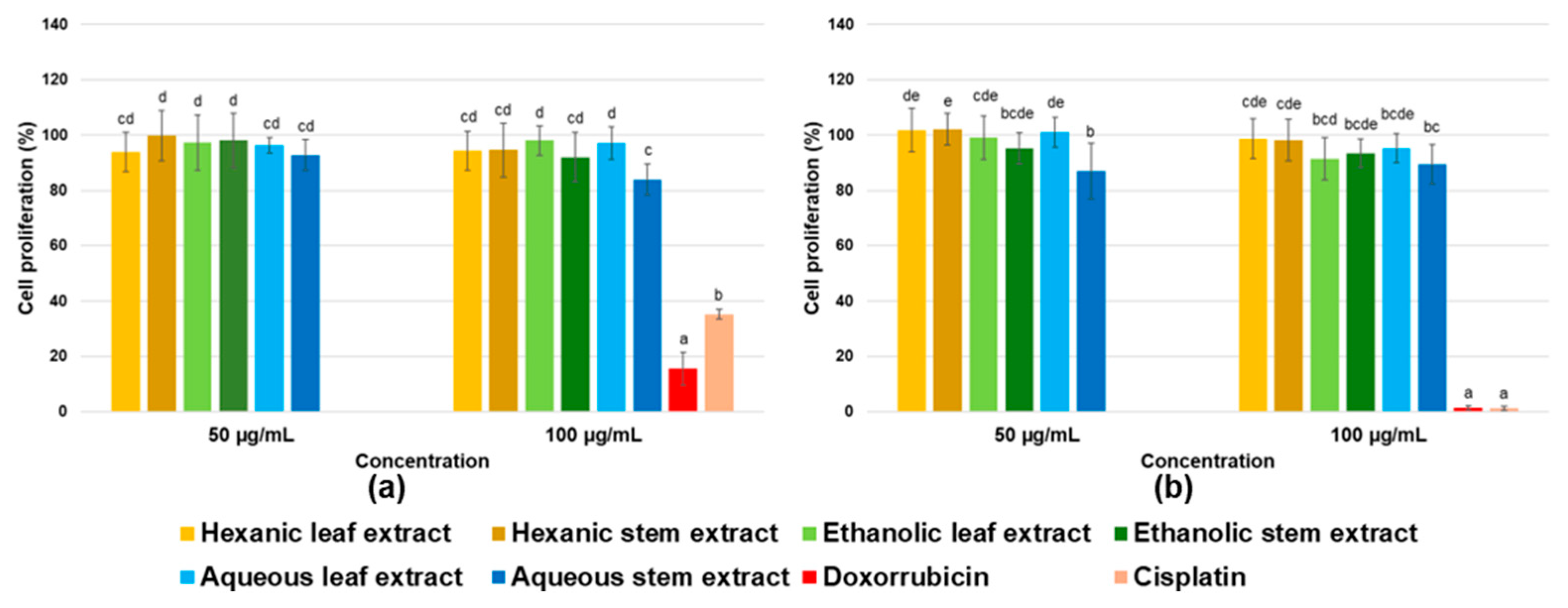
Regarding the evaluation of cell viability across different human cancer cell lines, it is important to highlight that one of the most crucial controls when analyzing effects on cancer cells is the use of non-cancerous cells. This is because, in search for potential anticancer drugs, the aim is for these compounds to be specific and to primarily target malignant tissues. However, such specificity remains one of the main challenges of current treatments, which often results in side effects and long-term consequences for patients [
38,
39].
In this case, evaluated extracts did not significantly affect the apparently healthy cells, which is a desirable outcome. However, as previously mentioned, they also showed no effect on cancer cells, indicating that at the concentrations used in this study, they do not possess antiproliferative activity. According to the United States National Cancer Institute, an extract is considered to have antiproliferative activity if its IC
50 value is below 30 µg/mL [
40]; therefore, it can be confirmed that analyzed extracts do not exhibit activity against cancer cell lines included in this study.
Nevertheless, it cannot be ruled out that extracts may contain individual compounds with the ability to reduce cell viability, but which are present at very low concentrations, potentially masking their effect within the complex mixture of the extract. Likewise, the presence of antagonistic compounds that interfere with or counteract activity of those with antiproliferative potential cannot be excluded, thereby preventing the desired effects on cancer cells from being observed.
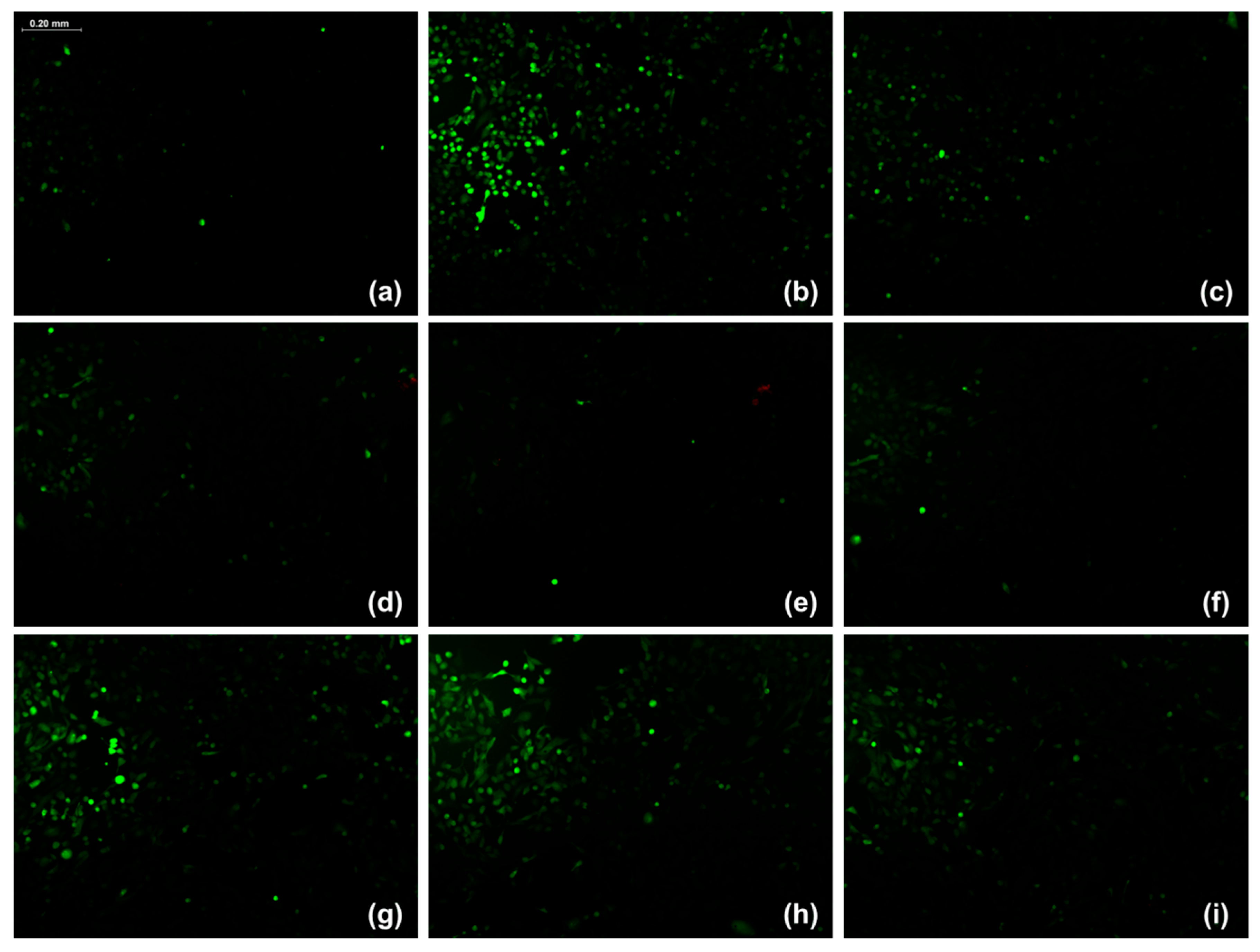
The DCFH-DA probe is a non-fluorescent, lipophilic compound that, upon entering the cytoplasm, is hydrolyzed by intracellular esterases to 2′,7′-dichlorodihydrofluorescein (DCFH) through the removal of acetate groups. DCFH is subsequently oxidized by intracellular ROS to form 2′,7′-dichlorofluorescein (DCF), a highly fluorescent molecule; therefore, the intracellular ROS concentration is directly proportional to the observed fluorescence intensity [
41,
42].
In this study, ARPE-19 cells exposed to the hexanic extract from leaves, hexanic extract from stems, and ethanolic extract from stems exhibited lower relative fluorescence intensity compared to the control without extracts. Notably, these extracts produced a fluorescence intensity even lower than that produced by the positive control (vitamin E). Although all six
P. gracile extracts demonstrated in vitro antioxidant activity, primarily through electron transfer as evidenced by the ABTS and FRAP assays, only the three above-mentioned extracts exerted this effect within the cells. This outcome may be attributed to the chemical nature and polarity of the compounds present in the hexanic and ethanolic extracts, which typically include mainly lipophilic or moderate polar molecules, consistent with the nature of these solvents, and are more capable of permeating into the cell membrane and exerting antioxidant effects intracellularly [
43]. In contrast, the polar compounds present in the aqueous extracts likely had limited membrane permeability, preventing them from displaying antioxidant activity in the cellular assay [
44].
Although P. gracile has been identified as containing compounds with highly promising antioxidant activity, based on these results, it is recommended to evaluate its capacity to scavenge reactive nitrogen species (RNS) as well as to assess its activity in vivo and potential anti-inflammatory effects. Furthermore, it is essential to conduct bioactivity-guided purification and isolation studies to identify the compounds responsible for the observed bioactivity and to elucidate underlying mechanisms of action.
In addition, P. gracile is commonly used in traditional medicine in the state of Sonora, Mexico, to treat circulation-related disorders, such as varicose vein formation. Therefore, it is advisable to investigate its bioactive potential in this context, including its possible antihypertensive activity, for instance, through inhibition of key enzymes such as angiotensin-converting enzyme (ACE).
4. Materials and Methods
4.1. Plant Material
Porophyllum gracile was collected from a single location, specifically the Port of Yavaros, Huatabampo, Sonora, Mexico (26°42′27.3″ N, 109°33′00.4″ W), during October 2023 and October 2024. The plant material was taxonomically identified at the Herbarium of the Universidad de Sonora (Catalog#:31731, Occurrence ID: 6bfc9716-c0a3-47d7-9617-a0e6582bfc6b). For each collection season, approximately 10 kg of plant material was harvested. Samples were dried at room temperature in the dark for three days, and leaves and stems were manually separated and milled using a coffee grinder mill. Each milled sample was homogenized to ensure uniformity, and representative portions were taken, as appropriate, for each of the subsequent analyses.
4.2. Analysis of Volatile Compounds
Extraction of volatile compounds was carried out using headspace solid-phase microextraction (HS-SPME) [
45]. A 1 g sample (crushed leaves and stems) was placed in 20 mL vials with polypropylene caps and PTFE/silicone septa. Extraction was performed using an automatic sampler AOC-6000 Plus (Shimadzu, Kyoto, Japan), employing a DVB/CAR/PDMS fiber (50/30 μm, 1 cm) at 40 °C for 40 min. Analysis was performed on a gas chromatograph GC2030 (Shimadzu, Canby, OR, USA) coupled with a triple quadrupole mass spectrometer TQ8040 NX, with a Sapiens X5MS column (30 m × 0.25 mm × 0.25 μm, Teknokroma, Cugat del Vallès, Spain). The Q3 Scan acquisition mode (5000 uma/s, 35–400 m/z) was used, with the following oven temperature program: 80 °C, increasing by 3 °C/min to 210 °C, then 20 °C/min to 280 °C (2 min). Helium was used as carrier gas (1.0 mL/min), with a split injection (1:25) and pressure of 65.2 kPa. The injector, ion source, and interface were adjusted to 230, 230, and 280 °C, respectively. Compound identification was based on three criteria: retention index, comparison with retention times of pure standards, and mass spectrum analysis using the National Institute of Standards and Technology (NIST) database. Only fully identified compounds are reported. The analyses were performed in duplicate.
4.3. Identification of Phenolic Compounds
Analysis was performed on an HPLC Agilent 1200 Series (Agilent Technologies, Santa Clara, CA, USA); equipped with a photodiode array detector (G1315D), a tandem mass spectrometer, a binary pump (G1376A), a degasser (G1379B), and an automatic sampler (G1313A), using Nucleosil
® 100-5 C18 column (25.0 cm × 0.46 cm, 5 µm; Macherey-Nagel, Düren, Germany). A 20 µL sample was injected in triplicate. Mobile phase consisted of a mixture of water and formic acid (99:1,
v/
v) as eluent A, and acetonitrile as eluent B, with the following gradient: 10% B (start), 24% (24 min), 32% (28 min), 40% (36 min), 60% (40 min), and 95% (42 min), followed by a 5 min wash and a return to initial conditions. Mass detector conditions were capillary temperature of 350 °C, capillary voltage of 4 kV, nebulizer pressure of 65 psi, nitrogen flow rate of 11 L/min, and mobile phase flow rate of 0.8 mL/min. Helium was used as collision gas in an ion trap with fragmentation voltages ranging from 0.3 to 2 V [
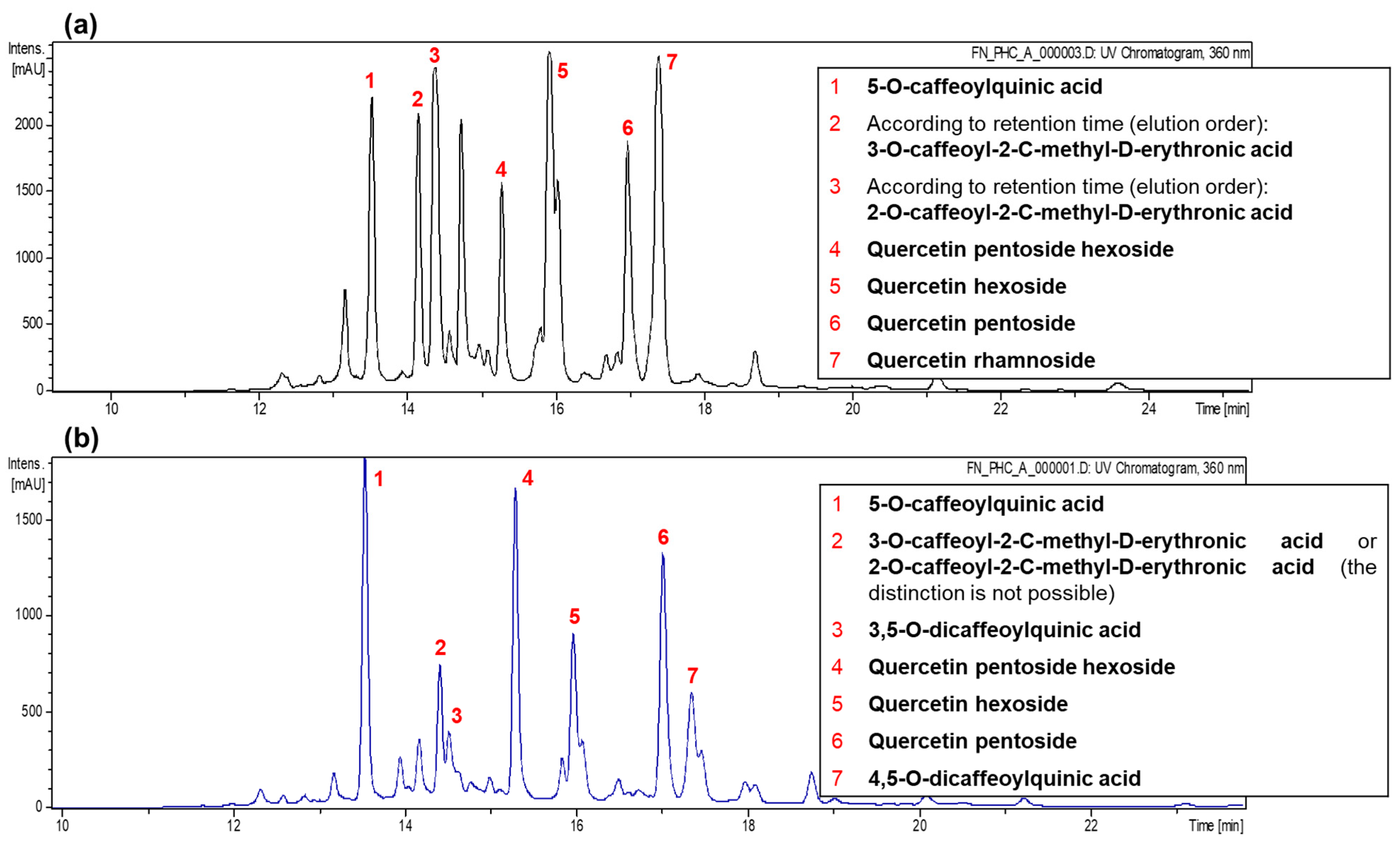
46]. Data acquisition was performed in negative ionization mode and automated MSn for the most abundant fragment ion. The ChemStation for LC 3D Systems Rev. B.01.03SR2 software (Agilent, Madrid, Spain) controlled equipment was used. Identification of phenolic compounds was based on order of elution, retention time, and UV-Vis and mass spectra, compared with authentic standards and previous data.
4.4. Identification of Fatty Acids
For derivatization of fatty acids [
45], 25 mg of a sample were mixed with 2 mL of sodium methoxide in test tubes with caps and incubated in an orbital bath at 50 °C for 10 min. The mixture was then rapidly cooled to 4 °C and 3 mL of 10% acetylene chloride in methanol were added, followed by a 50 °C bath with stirring for 10 min. After reaching room temperature, 7.5 mL of 6% K
2CO
3 and 1 mL of internal standard C13:0 (1 mg/mL in hexane) were added, and the mixture was vortexed for 30 s. Samples were centrifuged at 200×
g at 4 °C for 10 min. The organic phase was transferred to Eppendorf tubes, and a second centrifugation was performed at 7500×
g at 4 °C for 10 min. Finally, the organic phase was deposited into amber vials for chromatography. Analysis was carried out on a gas chromatograph (Shimadzu Nexis GC-2030; Shimadzu Corporation, Kyoto, Japan) with a flame ionization detector and an automatic injector (AOC-20i) [
47]. Helium was used as carrier gas (flow rate 1.05 L/min) and a CP-Sil 88 capillary column (100 m, 0.25 mm internal diameter, 0.20 μm thickness; Agilent Technologies, Santa Clara, CA, USA) was employed. Injector and detector temperatures were set to 250 and 260 °C, respectively, with a split ratio of 1:20. Oven temperature was programmed as follows: 45 °C (4 min), increasing by 13 °C/min to 175 °C (27 min), followed by a 4 °C/min increase to 215 °C (35 min). Fatty acid methyl esters were identified by comparing retention times with the Supelco 37 Component FAME MIX (Merck KGaA, Darmstadt, Germany). Results were expressed as a percentage of each fatty acid in the total profile and as mg/kg of a sample. The analyses were performed in duplicate.
4.5. Extraction of Crude Extracts
Milled samples of leaves and stems were separately extracted in darkness using a 1:9 ratio (
w/v) of different solvents, sequentially from low to high polarity (hexane, ethanol and water), for 24 h in each under constant agitation at 25 °C. Extracts were filtered and dried under reduced pressure using a rotary evaporator (Yamato, RE300, Tokyo, Japan) at no more than 45 °C, then suspended in their respective extraction solvents at a known concentration. Finally, extracts were stored at −20 °C in amber glass vials until further use [
48,
49,
50].
4.6. Antioxidant Activity
4.6.1. 2,2′-Azino-Bis(3-Ethylbenzothiazoline-6-Sulfonic Acid) (ABTS•+) Radical Assay
Antioxidant activity was assessed using the assay described by Re et al. [
51]. The ABTS reagent was prepared by mixing ABTS solution (3.84 mg/mL) and potassium persulfate solution (0.66 mg/mL) in a 1:1 ratio (
v/
v), followed by incubation in the dark at room temperature for 24 h. ABTS solution was adjusted to an absorbance of 0.7 ± 0.01 at 713 nm. Then, 900 µL of ABTS solution was mixed with 100 µL of extracts to achieve concentrations of 31.25, 62.5, 125, 250, and 500 µg/mL. The mixture was incubated for 30 min at room temperature, and absorbance was measured at 713 nm using a microplate reader (Agilent 800 TS; Agilent Technologies, Santa Clara, CA, USA). Trolox was used as a positive control. Three independent assays with five replicates were performed. Results were expressed as a percentage of radical inhibition using the following formula:
where
ABSsample: ABTS + Sample Absorbance.
ABSneg.control: Sample Absorbance.
ABSABTS: ABTS Absorbance.
ABSsolvent: Solvent Absorbance.
4.6.2. 2,2-Diphenyl-1-Picrylhydrazyl (DPPH•) Radical Assay
Antioxidant activity was assessed following the methodology described by Osuna-Ruiz et al. [
50]. A DPPH solution was prepared by dissolving reagent in the assay solvent until an absorbance value of 0.7 ± 0.01 was obtained at a wavelength of 520 nm. Subsequently, 900 µL of the DPPH reagent was mixed with 100 µL of extracts at final concentrations of 31.25, 62.5, 125, 250, and 500 µg/mL. The mixture was then incubated for 30 min in the dark at room temperature. Absorbance was measured at 520 nm using a microplate reader (Agilent 800 TS; Agilent Technologies, Santa Clara, CA, USA). Trolox was used as a positive control. Three independent assays with five replicates were performed. Results were expressed as a percentage of radical inhibition using the following formula:
where
ABSsample: ABTS + Sample Absorbance.
ABSneg.control: Sample Absorbance.
ABSDPPH: DPPH Absorbance.
ABSsolvent: Solvent Absorbance
4.6.3. Ferric Reducing Antioxidant Power (FRAP) Assay
The FRAP reagent was prepared by mixing 10 mM TPTZ (2,4,6-Tris(2-pyridyl)-s-triazine) in 40 mM HCl, 20 mM FeCl
3 in ultrapure water, and CH
3COONa buffer (pH 3.6, 1.55 g/L) in a 1:1:10 ratio. For the assay, 990 µL of FRAP reagent was mixed with 10 µL of the extracts at concentrations of 31.25 or 62.5 µg/mL, followed by incubation at room temperature for 10 min. Absorbance was measured at 593 nm using a UV-Vis spectrophotometer (Uvikon XS, Bio-Tek Instruments, Saint Quentin Yvelines, France) [
52]. One assay with three replicates was performed and results were expressed as Trolox equivalents (mmol TE/g of the initial sample).
4.7. Cell Culture
For cell viability assays, five cancer cell lines were used: MDA-MB-231 from invasive breast adenocarcinoma, HeLa from cervical carcinoma, A549 from lung carcinoma, HCT 116 from colon carcinoma, and 22Rv1 from prostate carcinoma. Two apparently healthy cell lines were also used: ARPE-19 from retinal epithelium and hFOB 1.19 from osteoblasts (ATCC, Rockville, MD, USA). Cells were cultured in growth medium using Roswell Park Memorial Institute (RPMI) medium (Sigma-Aldrich, St. Louis, MO, USA) for 22Rv1 cell line and Dulbecco’s Modified Eagle Medium (DMEM) (Sigma-Aldrich, St. Louis, MO, USA) for the other cell lines; both mediums were supplemented with 10% fetal bovine serum (Gibco®, Thermo Fisher Scientific, Waltham, MA, USA) and maintained in a Binder incubator (BINDER GmbH, Tuttlingen, Germany) at 37 °C, with a 5% CO2 atmosphere and 80% humidity.
4.8. Cell Viability Assay
The effect of
P. gracile extracts on cell viability was evaluated using the MTT assay (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) [
53]. Briefly, 10,000 cells were seeded per well in a 96-well microplate; after 24 h of incubation at 37 °C, culture medium was replaced with treatments at concentrations of 50 and 100 μg/mL in culture medium. Cells were then incubated for 48 h; after this period, 10 μL of MTT solution (5 mg/mL in water) was added, and cells were incubated for 4 h. Finally, the medium was removed and the MTT formazan crystals were dissolved in DMSO for absorbance readings at 570 nm, using 630 nm as reference (Agilent 800 TS microplate reader). Cisplatin and doxorubicin were used as positive controls. Three independent assays with three replicates were performed.
4.9. Cellular Antioxidant Activity Assay
The cellular antioxidant potential of the extracts was evaluated using a fluorescence intensity assay with 2′,7′-dichlorodihydrofluorescein diacetate (DCFH-DA), following the methodology described by Kim and Xue [
41] with slight modifications. Briefly, ARPE-19 cells were seeded in 96-well microplates (10,000 cells/well) and incubated for 24 h at 37 °C. The culture medium was then replaced with 50 µL of the test extracts diluted in medium to a final concentration of 100 µg/mL. After 24 h of incubation with the treatments, 50 µL of doxorubicin solution (4 µM in culture medium) was added to induce ROS generation. Following an additional 24 h incubation, the medium was removed, and the cells were washed with 100 µL of PBS. Cells were then stained with 100 µL of working solution (10 µM DCFH-DA in culture medium) and incubated for 30 min at 37 °C. The staining solution was discarded, and the wells were washed twice with 50 µL of PBS before adding 100 µL of PBS for subsequent fluorescence intensity measurement in a microplate fluorescence reader FLUOstar Omega (BMG LABTECH GmbH, Ortenberg, Germany) at an excitation wavelength of 485 nm and an emission wavelength of 520 nm, with gain set to 1200. Vitamin E (0.4 mM) was used as a positive control [
54]. All assays were performed in triplicate. Data were expressed as relative fluorescence intensity (I/I
o) according to the following equation:
where
FlCells+Tx+Doxo: Fluorescence intensity of cells treated with extract plus doxorubicin.
FlTx+Doxo: Fluorescence intensity of extract plus doxorubicin, without cells.
FlCells+Doxo: Fluorescence intensity of cells treated with doxorubicin alone.
4.10. Experimental Design
For the phytochemical characterization, descriptive statistics were used. For the evaluation of biological activities, a completely randomized experimental design was employed.
4.11. Statistical Analysis
For analysis of the obtained data, a significance level of 95% (α < 0.05) was established and the JMP 9 statistical package was used. A Probit regression model was employed to determine median inhibitory concentrations (IC50). Additionally, a one-way analysis of variance (ANOVA) was performed followed by Tukey’s multiple comparison test.